Ecotoxicological Evaluation of Sunscreens on Marine Plankton
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Exposure Media
2.3. Test Organisms and Bioassays
2.4. Statistical Analysis
3. Results
3.1. Growth Inhibition in Tisochrysis lutea
3.2. Sea Urchin Embryo Test (SET)
3.3. Survival Test in Copepods
3.4. PBSA Sea Urchin Embryo Test (SET)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Item | Characteristics |
|---|---|
| CR1 | Colored fluid solution |
| CR2 | Body oil |
| CR3 | Fluid solution without color |
| CR4 | Gel cream |
| CR5 | High-protection fluid solution |
| CR6 | High-protection fluid solution |
| CR7 | Body spray |
| CR8 | Water fluid |
| Item | Dilutions | NOEC | LOEC | EC10 | EC50 | EC10 (mg/L) | EC50 (mg/L) | TU |
|---|---|---|---|---|---|---|---|---|
| CR1 | ×1/100, ×1/30, ×1/10, ×1/3, ×1/2 | ×1/30 | ×1/10 | 1/12.31 | 1/4.96 | 812.35 | 2016.13 | 4.96 (3.92–6.44) |
| CR2 | ×1/100, ×1/30, ×1/10, ×1/3, ×1/2 | ×1/100 | ×1/30 | 1/62.71 | 1/4.09 | 159.46 | 2445 | 4.09 (2.66–5.74) |
| CR3 | ×1/100, ×1/30, ×1/10, ×1/3, ×1/2 | ×1/30 | ×1/10 | 1/31.36 | 1/9.59 | 318.88 | 1042.75 | 9.60 (7.25–13) |
| CR4 | ×1/100, ×1/30, ×1/10, ×1/3, ×1/2 | ×1/30 | ×1/10 | 1/27.71 | 1/5.52 | 360.88 | 1811.60 | 5.52 (4.34–6.98) |
| CR5 | ×1/100, ×1/30, ×1/10, ×1/3, ×1/2 | ×1/30 | ×1/10 | 1/40.53 | 1/5.10 | 246.73 | 1960.78 | 5.10 (3.16–7.65) |
| CR6 | ×1/100, ×1/30, ×1/10, ×1/3, ×1/2 | ×1/100 | ×1/30 | 1/103.11 | 1/4.16 | 96.98 | 2403.85 | 4.16 (1.84–7.13) |
| CR7 | ×1/100, ×1/30, ×1/10, ×1/3, ×1/2 | ×1/100 | ×1/30 | 1/415.33 | 1/0.49 | 24.08 | 20,430.04 | <2 (0.01–1.67) |
| CR8 | ×1/100, ×1/30, ×1/10, ×1/3, ×1/2 | ×1/10 | ×1/3 | 1/4.64 | 1/1.85 | 215.33 | 5411.26 | <2 (1.10–2.29) |
| Item | Dilutions | NOEC | LOEC | EC10 | EC50 | EC10 (mg/L) | EC50 (mg/L) | TU |
|---|---|---|---|---|---|---|---|---|
| CR1 | ×1/30, ×1/10, ×1/3, ×1 | ×1/30 | ×1/10 | 1/258.92 | 1/20.14 | 38.62 | 496.52 | 20.14 (11.87–51.23) |
| CR2 | ×1/30, ×1/10, ×1/3, ×1 | ×1/30 | ×1/10 | 1/100 | 1/10.14 | 100 | 986.19 | 10.14 (6.20–20) |
| CR3 | ×1/1000, ×1/300, ×1/100, ×1/30, ×1/10, ×1/3, ×1 | ×1/100 | ×1/30 | 1/120.65 | 1/16.23 | 82.90 | 616.14 | 16.23 (11.46–23) |
| CR4 | ×1/1000, ×1/300, ×1/100, ×1/30, ×1/10, ×1/3, ×1 | ×1/300 | ×1/100 | 1/977.20 | 1/81.67 | 10.23 | 122.44 | 81.67 (52.21–134.56) |
| CR5 | ×1/30, ×1/10, ×1/3, ×1 | ×1/30 | ×1/10 | 1/67.17 | 1/9.7 | 148.90 | 1030.93 | 9.70 (6.3–16.80) |
| CR6 | ×1/1000, ×1/300, ×1/100, ×1/30, ×1/10, ×1/3, ×1 | ×1/100 | ×1/30 | 1/203.86 | 1/21.86 | 49.10 | 457.46 | 21.86 (13.85–35.10) |
| CR7 | ×1/1000, ×1/300, ×1/100, ×1/30, ×1/10, ×1/3, ×1 | ×1/100 | ×1/30 | 1/15.53 | 1/3.10 | 643.92 | 3225.81 | 3.10 (2.67–3.56) |
| CR8 | ×1/1000, ×1/300, ×1/100, ×1/30, ×1/10, ×1/3, ×1 | ×1/100 | ×1/30 | 1/1097.46 | 1/52.20 | 9.11 | 191.57 | 52.20 (34.20–86.70) |
| Item | Dilutions | NOEC | LOEC | EC10 | EC50 | EC10 (mg/L) | EC50 (mg/L) | TU |
|---|---|---|---|---|---|---|---|---|
| CR1 | ×1/30, ×1/10, ×1/3, ×1 | ×1/30 | ×1/10 | 1/25.7 | 1/11.17 | 389.11 | 895.26 | 11.17 (1.5–42.78) |
| CR2 | ×1/30, ×1/10, ×1/3, ×1 | ×1/30 | ×1/10 | 1/30 | 1/9.43 | 333.33 | 1060.45 | 9.43 (2.11–48.02) |
| CR3 | ×1/30, ×1/10, ×1/3, ×1 | ×1/10 | ×1/3 | 1/16.40 | 1/7.85 | 609.76 | 1273.88 | 7.85 (1.10–21.23) |
| CR4 | ×1/30, ×1/10, ×1/3, ×1 | n.c | n.c | 1/52.10 | 1/28.22 | 191.94 | 354.36 | 28.22 (n.c) |
| CR5 | ×1/30, ×1/10, ×1/3, ×1 | ×1/10 | ×1/3 | 1/14.65 | 1/4.65 | 682.60 | 2150.57 | 4.65 (1.21–16.42) |
| CR6 | ×1/30, ×1/10, ×1/3, ×1 | ×1/3 | ×1 | 1/11.37 | 1/4.41 | 879.51 | 2267.57 | 4.41 (1.31–13.14) |
| CR7 | ×1/30, ×1/10, ×1/3, ×1 | ×1/10 | ×1/3 | 1/14.27 | 1/4.60 | 700.79 | 2181.11 | 4.60 (1.36–13. 85) |
| CR8 | ×1/30, ×1/10, ×1/3, ×1 | ×1/10 | ×1/3 | 1/27.31 | 1/8.47 | 366.17 | 1180.64 | 8.46 (2.57–34.56) |
| Incubation Conditions | Dilutions | NOEC | LOEC | EC10 | EC50 | EC10 (mg/L) | EC50 (mg/L) | TU |
|---|---|---|---|---|---|---|---|---|
| Darkness | ×1/30, ×1/10, ×1/3, ×1 | ×1/3 | ×1/1 | 1/5.37 | 1/2.32 | 1861.50 | 4312.2 | 2.32 (1.99–2.70) |
| Light/darkness (16:8) | ×1/30, ×1/10, ×1/3, ×1 | ×1/30 | ×1/10 | 1/14.29 | 1/3.63 | 699.59 | 2751.79 | 3.63 (2.91–4.50) |
Appendix B
| UV Filter | CR1 | CR2 | CR3 | CR4 | CR5 | CR6 | CR7 | CR8 |
|---|---|---|---|---|---|---|---|---|
| EHMC | 499 | 999 | ||||||
| OC | 1000 | 1000 | 800 | |||||
| TiO2 | 479 | 533 | 72 | |||||
| PBSA | 200 |
References
- NCI Dictionary of Cancer Terms—National Cancer Institute. Available online: https://www.cancer.gov/publications/dictionaries/cancer-terms/def/sunscreen (accessed on 1 November 2020).
- Mildau, G. General Review of Official Methods of Analysis of Cosmetics. In Analysis of Cosmetic Products, 2nd ed.; Salvador, A., Chisvert, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 67–83. [Google Scholar]
- Daly, S.; Ouyang, H.; Maitra, P. Chemistry of Sunscreens. In Principles and Practice of Photoprotection; Springer: Berlin/Heidelberg, Germany, 2016; pp. 159–178. [Google Scholar] [CrossRef]
- Bowman, D.M.; May, N.D.; Maynard, A.D. Nanomaterials in Cosmetics: Regulatory Aspects. In Analysis of Cosmetic Products, 2nd ed.; Salvador, A., Chisvert, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 289–302. [Google Scholar]
- Giokas, D.L.; Salvador, A.; Chisvert, A. UV filters: From sunscreens to human body and the environment. TrAC Trends Anal. Chem. 2007, 26, 360–374. [Google Scholar] [CrossRef]
- Beiras, R. Environmental Risk Assessment of Pharmaceutical and Personal Care Products in Estuarine and Coastal Waters. In Pharmaceuticals in Marine and Coastal Environments; Elsevier: Amsterdam, The Netherlands, 2021; pp. 195–252. [Google Scholar]
- Corinaldesi, C.; Damiani, E.; Marcellini, F.; Falugi, C.; Tiano, L.; Brugè, F.; Danovaro, R. Sunscreen products impair the early developmental stages of the sea urchin Paracentrotus lividus. Sci. Rep. 2017, 7, 7815. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sánchez-Quiles, D.; Blasco, J.; Tovar-Sánchez, A. Sunscreen Components Are a New Environmental Concern in Coastal Waters: An Overview. In Handbook of Environmental Chemistry; Springer: Cham, Switzerland, 2020; Volume 94, pp. 1–14. [Google Scholar]
- Paredes, E.; Perez, S.; Rodil, R.; Quintana, J.B.; Beiras, R. Ecotoxicological evaluation of four UV filters using marine organisms from different trophic levels Isochrysis galbana, Mytilus galloprovincialis, Paracentrotus lividus, and Siriella armata. Chemosphere 2014, 104, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Bratkovics, S.; Wirth, E.; Sapozhnikova, Y.; Pennington, P.; Sanger, D. Baseline monitoring of organic sunscreen compounds along South Carolina’s coastal marine environment. Mar. Pollut. Bull. 2015, 101, 370–377. [Google Scholar] [CrossRef]
- Apel, C.; Joerss, H.; Ebinghaus, R. Environmental occurrence and hazard of organic UV stabilizers and UV filters in the sediment of European North and Baltic Seas. Chemosphere 2018, 212, 254–261. [Google Scholar] [CrossRef]
- Park, C.-B.; Jang, J.; Kim, S.; Kim, Y.J. Single- and mixture toxicity of three organic UV-filters, ethylhexyl methoxycinnamate, octocrylene, and avobenzone on Daphnia magna. Ecotoxicol. Environ. Saf. 2017, 137, 57–63. [Google Scholar] [CrossRef]
- Beiras, R.; Schönemann, A.M. Water quality criteria for selected pharmaceuticals and personal care products for the protection of marine ecosystems. Sci. Total Environ. 2021, 758, 143589. [Google Scholar] [CrossRef]
- Danovaro, R.; Bongiorni, L.; Corinaldesi, C.; Giovannelli, D.; Damiani, E.; Astolfi, P.; Greci, L.; Pusceddu, A. Sunscreens Cause Coral Bleaching by Promoting Viral Infections. Environ. Health Perspect. 2008, 116, 441–447. [Google Scholar] [CrossRef] [Green Version]
- Jovanović, B.; Guzmán, H.M. Effects of titanium dioxide (TiO2) nanoparticles on caribbean reef-building coral (Montastraea faveolata). Environ. Toxicol. Chem. 2014, 33, 1346–1353. [Google Scholar] [CrossRef]
- Tsui, M.M.; Leung, H.; Wai, T.-C.; Yamashita, N.; Taniyasu, S.; Liu, W.; Lam, K.S.P.; Murphy, M.B. Occurrence, distribution and ecological risk assessment of multiple classes of UV filters in surface waters from different countries. Water Res. 2014, 67, 55–65. [Google Scholar] [CrossRef]
- Lozano, C.; Givens, J.; Stien, D.; Matallana-Surget, S.; Lebaron, P. Bioaccumulation and Toxicological Effects of UV-Filters on Marine Species. In The Handbook of Environmental Chemistry; Springer Science and Business Media LLC.: Berlin/Heidelberg, Germany, 2020; Volume 94, pp. 85–130. [Google Scholar]
- Stien, D.; Clergeaud, F.; Rodrigues, A.M.S.; Lebaron, K.; Pillot, R.; Romans, P.; Fagervold, S.; Lebaron, P. Metabolomics Reveal That Octocrylene Accumulates in Pocillopora damicornis Tissues as Fatty Acid Conjugates and Triggers Coral Cell Mitochondrial Dysfunction. Anal. Chem. 2018, 91, 990–995. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, S.; Oliviero, M.; Philippe, A.; Manzo, S. Nanoparticles Based Sunscreens Provoke Adverseeffects on Marine Microalgae Dunaliella Tertiolecta. Environ. Sci. Nano 2018, 5, 3011. [Google Scholar] [CrossRef]
- Miller, R.J.; Bennett, S.; Keller, A.A.; Pease, S.; Lenihan, H.S. TiO2 Nanoparticles Are Phototoxic to Marine Phytoplankton. PLoS ONE 2012, 7, e30321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barmo, C.; Ciacci, C.; Canonico, B.; Fabbri, R.; Cortese, K.; Balbi, T.; Marcomini, A.; Pojana, G.; Gallo, G.; Canesi, L. In Vivo Effects of N-TiO2 on Digestive Gland and Immune Function of the Marine Bivalve Mytilus Galloprovincialis. Aquat. Toxicol. 2013, 132–133, 9–18. [Google Scholar] [CrossRef]
- Chen, L.; Li, X.; Hong, H.; Shi, D. Multigenerational effects of 4-methylbenzylidene camphor (4-MBC) on the survival, development and reproduction of the marine copepod Tigriopus japonicus. Aquat. Toxicol. 2018, 194, 94–102. [Google Scholar] [CrossRef]
- Zhou, R.; Lu, G.; Yan, Z.; Bao, X.; Zhang, P.; Jiang, R. Bioaccumulation and biochemical effects of ethylhexyl methoxy cinnamate and its main transformation products in zebrafish. Aquat. Toxicol. 2019, 214, 105241. [Google Scholar] [CrossRef]
- Zucchi, S.; Oggier, D.M.; Fent, K. Global gene expression profile induced by the UV-filter 2-ethyl-hexyl-4-trimethoxycinnamate (EHMC) in zebrafish (Danio rerio). Environ. Pollut. 2011, 159, 3086–3096. [Google Scholar] [CrossRef]
- Guan, X.; Shi, W.; Zha, S.; Rong, J.; Su, W.; Liu, G. Neurotoxic impact of acute TiO2 nanoparticle exposure on a benthic marine bivalve mollusk, Tegillarca granosa. Aquat. Toxicol. 2018, 200, 241–246. [Google Scholar] [CrossRef]
- Guo, Y.; Chen, L.; Wu, J.; Hua, J.; Yang, L.; Wang, Q.; Zhang, W.; Lee, J.S.; Zhou, B. Parental Co-Exposure to Bisphenol A and Nano-TiO2 Causes Thyroid Endocrine Disruption and Developmental Neurotoxicity in Zebrafish Offspring. Sci. Total Environ. 2019, 650, 557–565. [Google Scholar] [CrossRef]
- Lee, I.; Lee, J.; Jung, D.; Kim, S.; Choi, K. Two-Generation Exposure to 2-Ethylhexyl 4-Methoxycinnamate (EHMC) in Japanese Medaka (Oryzias latipes) and Its Reproduction and Endocrine Related Effects. Chemosphere 2019, 228, 478–484. [Google Scholar] [CrossRef]
- Schlumpf, M.; Schmid, P.; Durrer, S.; Conscience, M.; Maerkel, K.; Henseler, M.; Gruetter, M.; Herzog, I.; Reolon, S.; Ceccatelli, R.; et al. Endocrine activity and developmental toxicity of cosmetic UV filters—An update. Toxicology 2004, 205, 113–122. [Google Scholar] [CrossRef]
- Christen, V.; Zucchi, S.; Fent, K. Effects of the UV-filter 2-ethyl-hexyl-4-trimethoxycinnamate (EHMC) on expression of genes involved in hormonal pathways in fathead minnows (Pimephales promelas) and link to vitellogenin induction and histology. Aquat. Toxicol. 2011, 102, 167–176. [Google Scholar] [CrossRef]
- Giraldo, A.; Montes, R.; Rodil, R.; Quintana, J.B.; Vidal-Liñán, L.; Beiras, R. Ecotoxicological Evaluation of the UV Filters Ethylhexyl Dimethyl p-Aminobenzoic Acid and Octocrylene Using Marine Organisms Isochrysis galbana, Mytilus galloprovincialis and Paracentrotus lividus. Arch. Environ. Contam. Toxicol. 2017, 72, 606–611. [Google Scholar] [CrossRef]
- Oliviero, M.; Schiavo, S.; Rametta, G.; Miglietta, M.L.; Manzo, S. Different sizes of ZnO diversely affected the cytogenesis of the sea urchin Paracentrotus lividus. Sci. Total Environ. 2017, 607-608, 176–183. [Google Scholar] [CrossRef]
- Thorel, E.; Clergeaud, F.; Jaugeon, L.; Rodrigues, A.M.S.; Lucas, J.; Stien, D.; Lebaron, P. Effect of 10 UV Filters on the Brine Shrimp Artemia salina and the Marine Microalga Tetraselmis sp. Toxics 2020, 8, 29. [Google Scholar] [CrossRef] [Green Version]
- Sendra, M.; Sánchez-Quiles, D.; Blasco, J.; Moreno-Garrido, I.; Lubián, L.M.; Pérez-García, S.; Tovar-Sánchez, A. Effects of TiO2 Nanoparticles and Sunscreens on Coastal Marine Microalgae: Ultraviolet Radiation Is Key Variable for Toxicity Assessment. Environ. Int. 2017, 98, 62–68. [Google Scholar] [CrossRef]
- Bachelot, M.; Li, Z.; Munaron, D.; Le Gall, P.; Casellas, C.; Fenet, H.; Gomez, E. Organic UV filter concentrations in marine mussels from French coastal regions. Sci. Total Environ. 2012, 420, 273–279. [Google Scholar] [CrossRef]
- Gomez, E.; Bachelot, M.; Boillot, C.; Munaron, D.; Chiron, S.; Casellas, C.; Fenet, H. Bioconcentration of two pharmaceuticals (benzodiazepines) and two personal care products (UV filters) in marine mussels (Mytilus galloprovincialis) under controlled laboratory conditions. Environ. Sci. Pollut. Res. 2012, 19, 2561–2569. [Google Scholar] [CrossRef]
- Vidal-Liñán, L.; Villaverde-De-Sáa, E.; Rodil, R.; Quintana, J.B.; Beiras, R. Bioaccumulation of UV filters in Mytilus galloprovincialis mussel. Chemosphere 2018, 190, 267–271. [Google Scholar] [CrossRef]
- Asztemborska, M.; Jakubiak, M.; Stęborowski, R.; Chajduk, E.; Bystrzejewska-Piotrowska, G. Titanium Dioxide Nanoparticle Circulation in an Aquatic Ecosystem. Water Air Soil Pollut. 2018, 229, 208. [Google Scholar] [CrossRef] [Green Version]
- Fastelli, P.; Renzi, M. Exposure of key marine species to sunscreens: Changing ecotoxicity as a possible indirect effect of global warming. Mar. Pollut. Bull. 2019, 149, 110517. [Google Scholar] [CrossRef]
- Prato, E.; Scardicchio, C.; Biandolino, F. Effects of temperature on the acute toxicity of cadmium to Corophium Insidiosum. Environ. Monit. Assess. 2007, 136, 161–166. [Google Scholar] [CrossRef]
- Rodil, R.; Benito Quintana, J.; López-Mahía, P.; Muniategui-Lorenzo, S.; Prada-Rodríguez, D. Multiclass Determination of Sunscreen Chemicals in Water Samples by Liquid Chromatography-Tandem Mass Spectrometry. Anal. Chem. 2008, 80, 1307–1315. [Google Scholar] [CrossRef]
- Orlikowska, A.; Fisch, K.; Schulz-Bull, D.E. Organic polar pollutants in surface waters of inland seas. Mar. Pollut. Bull. 2015, 101, 860–866. [Google Scholar] [CrossRef]
- Fisch, K.; Waniek, J.J.; Schulz-Bull, D.E. Occurrence of pharmaceuticals and UV-filters in riverine run-offs and waters of the German Baltic Sea. Mar. Pollut. Bull. 2017, 124, 388–399. [Google Scholar] [CrossRef]
- Bastien, N.; Millau, J.-F.; Rouabhia, M.; Davies, R.J.H.; Drouin, R. The Sunscreen Agent 2-Phenylbenzimidazole-5-Sulfonic Acid Photosensitizes the Formation of Oxidized Guanines in Cellulo after UV-A or UV-B Exposure. J. Investig. Dermatol. 2010, 130, 2463–2471. [Google Scholar] [CrossRef] [Green Version]
- Grabicova, K.; Fedorova, G.; Burkina, V.; Steinbach, C.; Schmidt-Posthaus, H.; Žlábek, V.; Kroupova, H.; Grabic, R.; Randak, T. Presence of UV filters in surface water and the effects of phenylbenzimidazole sulfonic acid on rainbow trout (Oncorhynchus mykiss) following a chronic toxicity test. Ecotoxicol. Environ. Saf. 2013, 96, 41–47. [Google Scholar] [CrossRef]
- Huang, X.; Li, Y.; Wang, T.; Liu, H.; Shi, J.; Zhang, X. Evaluation of the Oxidative Stress Status in Zebrafish (Danio rerio) Liver Induced by Three Typical Organic UV Filters (BP-4, PABA and PBSA). Int. J. Environ. Res. Public Health 2020, 17, 651. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Westphal, J.; Kümmerer, K.; Olsson, O. Experimental and in silico assessment of fate and effects of the UV filter 2-phenylbenzimidazole 5-sulfonic acid and its phototransformation products in aquatic solutions. Water Res. 2020, 171, 115393. [Google Scholar] [CrossRef]
- Beiras, R. Towards standard methods for the classification of aquatic toxicity for biologically active household chemicals (BAHC) present in plastics, pharmaceuticals, and cosmetic products. Environ. Monit. Assess. 2021, 193, 685. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, J.; Nieto, O.; Beiras, R. Effect of humic acids on speciation and toxicity of copper to Paracentrotus lividus larvae in seawater. Aquat. Toxicol. 2002, 58, 27–41. [Google Scholar] [CrossRef]
- ISO 10253; Marine Algal Growth Inhibition Test with Skeletonema sp. and Phaeodactylum Tricornutum-Water Quality. ISO: Geneva, Switzerland, 2016.
- Tato, T.; Beiras, R. The Use of the Marine Microalga Tisochrysis lutea (T-iso) in Standard Toxicity Tests; Comparative Sensitivity With Other Test Species. Front. Mar. Sci. 2019, 6, 488. [Google Scholar] [CrossRef] [Green Version]
- Saco-Álvarez, L.; Durán, I.; Lorenzo, J.I.; Beiras, R. Methodological basis for the optimization of a marine sea-urchin embryo test (SET) for the ecological assessment of coastal water quality. Ecotoxicol. Environ. Saf. 2010, 73, 491–499. [Google Scholar] [CrossRef] [PubMed]
- ISO 14669; Determination of Acute Lethal Toxicity to Marine Copepods (Copepoda, Crustacea)-Water Quality. ISO: Geneva, Switzerland, 1999.
- Beiras, R.; Tato, T.; López-Ibáñez, S. A 2-Tier standard method to test the toxicity of microplastics in marine water using Paracentrotus lividu s and Acartia clausi larvae. Environ. Toxicol. Chem. 2019, 38, 630–637. [Google Scholar] [CrossRef]
- Alonso-López, O.; López-Ibáñez, S.; Beiras, R. Assessment of Toxicity and Biodegradability of Poly(vinyl alcohol)-Based Materials in Marine Water. Polymer 2021, 13, 3742. [Google Scholar] [CrossRef]
- European Commission. Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on Classification, Labelling and Packaging of Substances and Mixtures. Off. J. Eur. Union 2008, 353. Available online: https://eur-lex.europa.eu/eli/reg/2008/1272/oj (accessed on 17 January 2022).
- Beiras, R. Marine Pollution: Sources, Fate and Effects of Pollutants in Coastal Ecosystems; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
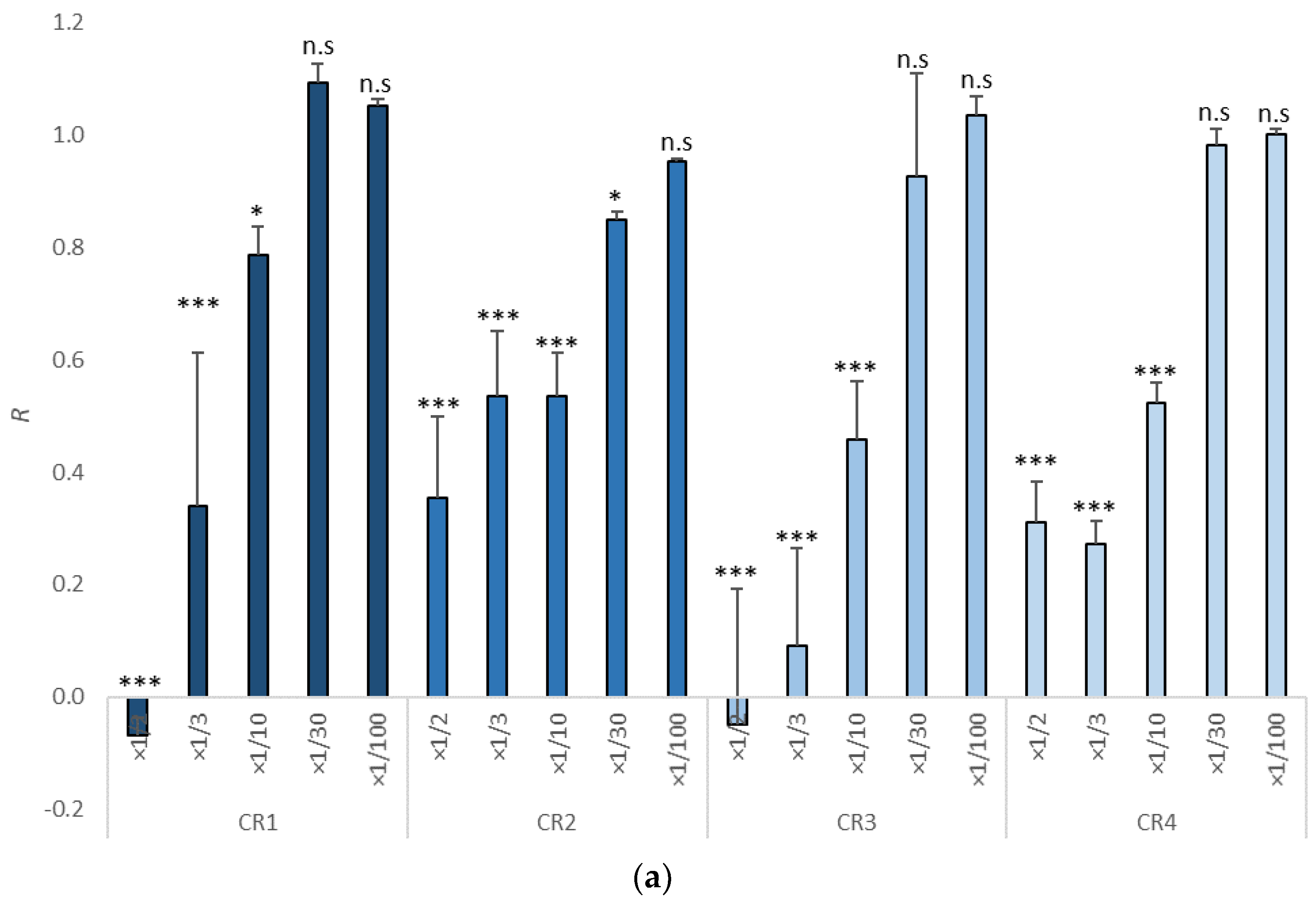
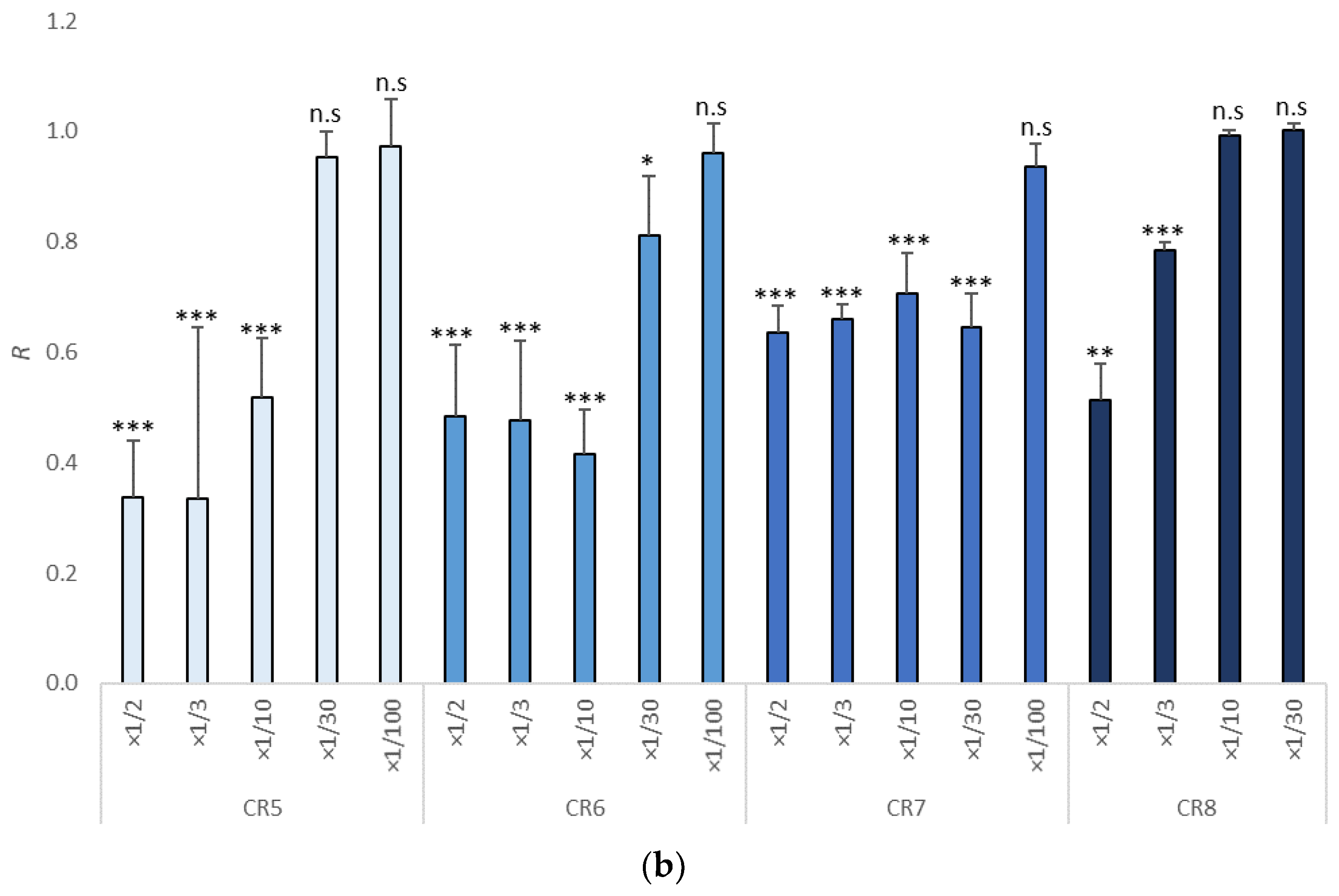
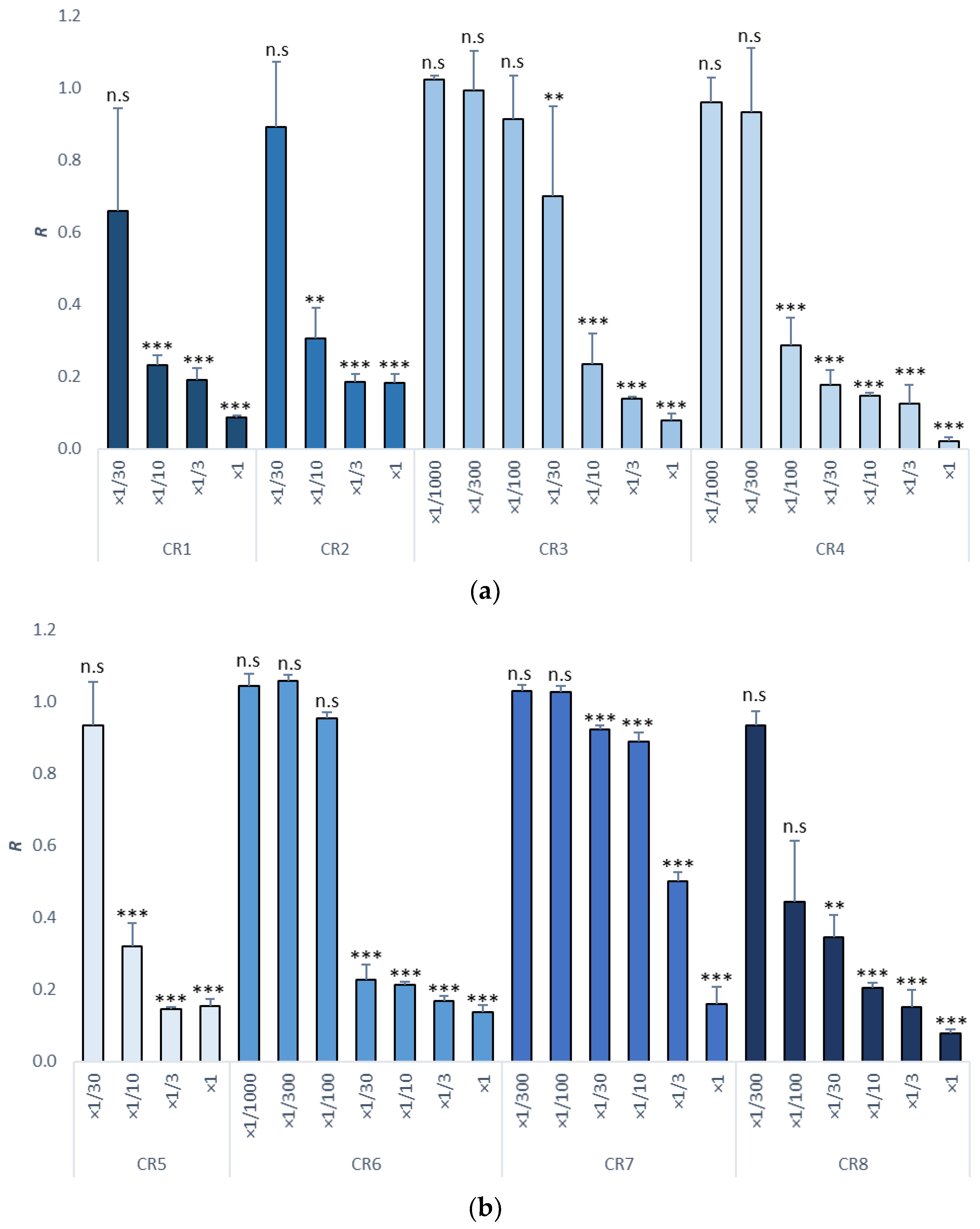
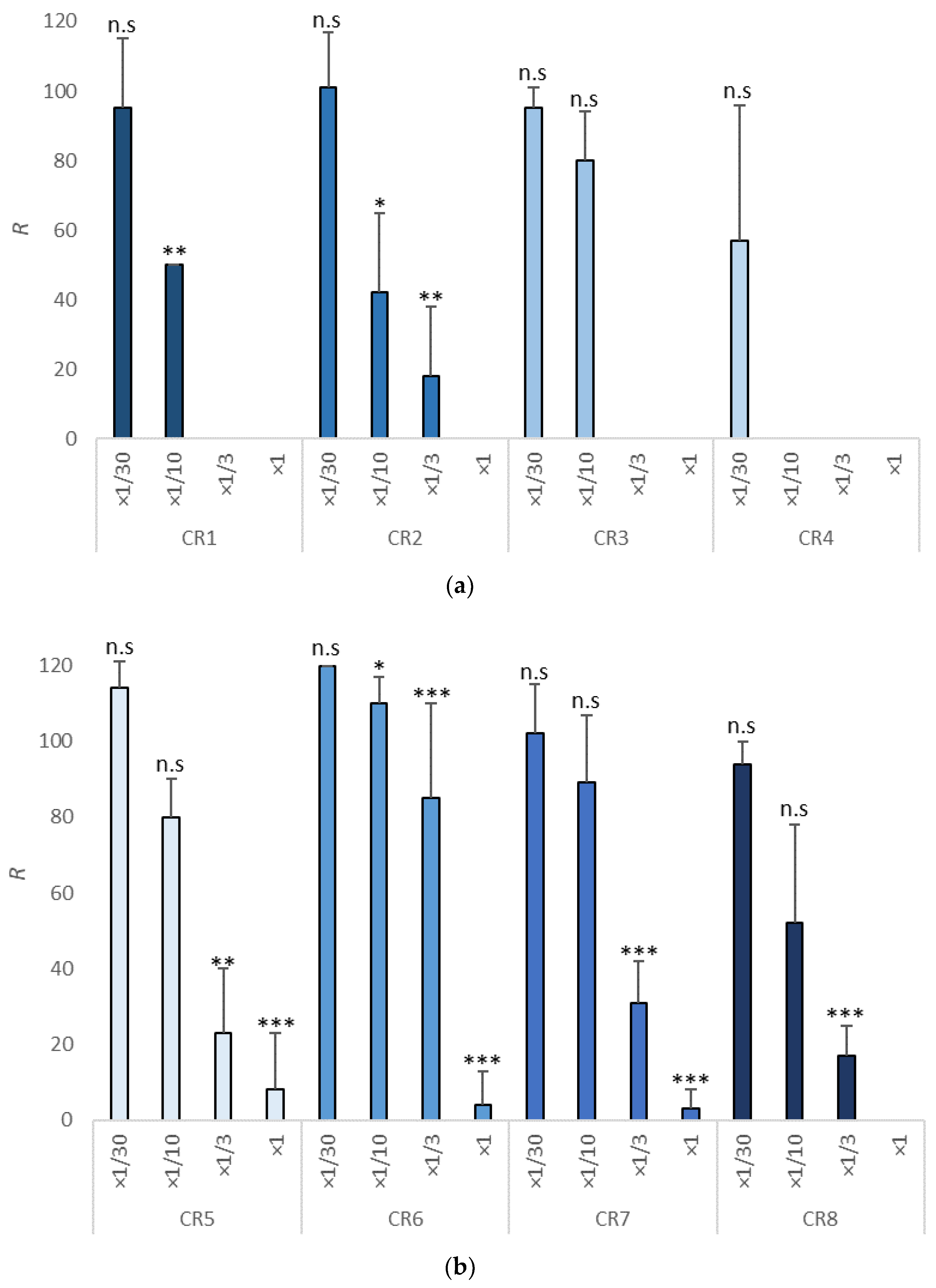
| Item | NOEC | LOEC | EC10 (mg/L) | EC50 (mg/L) | TU |
|---|---|---|---|---|---|
| CR1 | ×1/30 | ×1/10 | 812.35 | 2016 | 4.96 |
| CR2 | ×1/100 | ×1/30 | 159.46 | 2445 | 4.09 |
| CR3 | ×1/30 | ×1/10 | 318.88 | 1042.75 | 9.60 |
| CR4 | ×1/30 | ×1/10 | 360.88 | 1811.60 | 5.52 |
| CR5 | ×1/30 | ×1/10 | 246.73 | 1960.78 | 5.10 |
| CR6 | ×1/100 | ×1/30 | 96.98 | 2403.85 | 4.16 |
| CR7 | ×1/100 | ×1/30 | 24.08 | 20,430.04 | <2 |
| CR8 | ×1/10 * | ×1/3 * | 215.33 | 5411.26 | <2 |
| Item | NOEC | LOEC | EC10 (mg/L) | EC50 (mg/L) | TU |
|---|---|---|---|---|---|
| CR1 | ×1/30 * | ×1/10 * | 38.62 | 496.52 | 20.14 |
| CR2 | ×1/30 * | ×1/10 * | 100 | 986.19 | 10.14 |
| CR3 | ×1/100 | ×1/30 | 82.90 | 616.14 | 16.23 |
| CR4 | ×1/300 | ×1/100 | 10.23 | 122.44 | 81.67 |
| CR5 | ×1/30 | ×1/10 | 148.90 | 1030.93 | 9.70 |
| CR6 | ×1/100 | ×1/30 | 49.10 | 457.46 | 21.86 |
| CR7 | ×1/100 | ×1/30 | 643.92 | 3225.81 | 3.10 |
| CR8 | ×1/100 * | ×1/30* | 9.11 | 191.57 | 52.20 |
| Item | NOEC | LOEC | EC10 (mg/L) | EC50 (mg/L) | TU |
|---|---|---|---|---|---|
| CR1 | ×1/30 | ×1/10 | 389.11 | 895.26 | 11.17 |
| CR2 | ×1/30 | ×1/10 | 333.33 | 1060.45 | 9.43 |
| CR3 | ×1/10* | ×1/3 * | 609.76 | 1273.88 | 7.85 |
| CR4 | n.c. | n.c. | 191.94 | 354.36 | 28.22 |
| CR5 | ×1/10 | ×1/3 | 682.60 | 2150.57 | 4.65 |
| CR6 | ×1/3 | ×1 | 879.51 | 2267.57 | 4.41 |
| CR7 | ×1/10 | ×1/3 | 700.79 | 2181.11 | 4.60 |
| CR8 | ×1/10 * | ×1/3 * | 366.17 | 1180.64 | 8.46 |
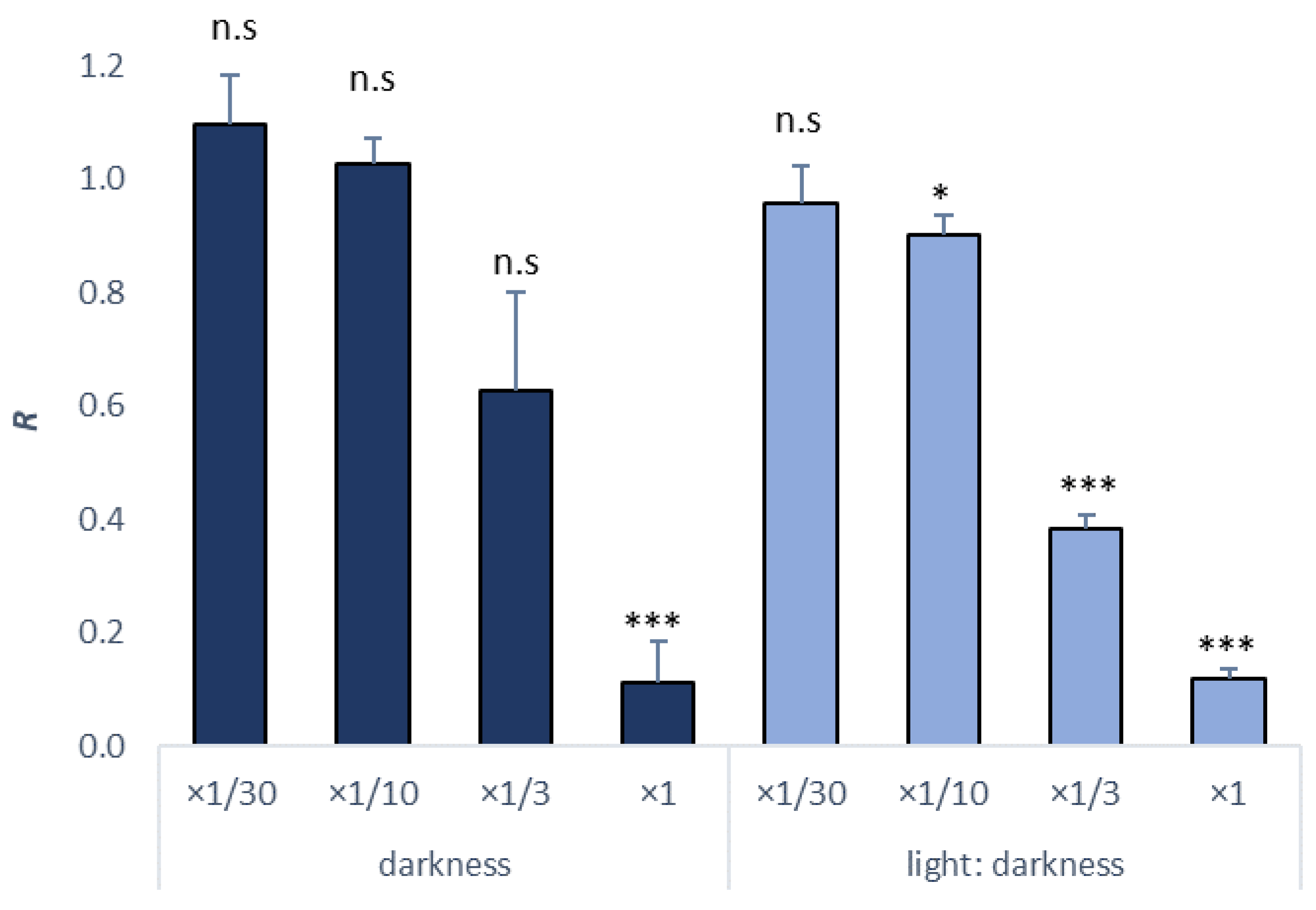
| Incubation Conditions | NOEC | LOEC | EC10 (mg/L) | EC50 (mg/L) | TU |
|---|---|---|---|---|---|
| darkness | ×1/3 | ×1 | 1861.50 | 4312.2 | 2.32 |
| Light/darkness (16:8) | ×1/30 | ×1/10 | 699.59 | 2751.79 | 3.63 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González, M.P.; Vilas, A.; Beiras, R. Ecotoxicological Evaluation of Sunscreens on Marine Plankton. Cosmetics 2022, 9, 20. https://doi.org/10.3390/cosmetics9010020
González MP, Vilas A, Beiras R. Ecotoxicological Evaluation of Sunscreens on Marine Plankton. Cosmetics. 2022; 9(1):20. https://doi.org/10.3390/cosmetics9010020
Chicago/Turabian StyleGonzález, María Pilar, Alejandro Vilas, and Ricardo Beiras. 2022. "Ecotoxicological Evaluation of Sunscreens on Marine Plankton" Cosmetics 9, no. 1: 20. https://doi.org/10.3390/cosmetics9010020
APA StyleGonzález, M. P., Vilas, A., & Beiras, R. (2022). Ecotoxicological Evaluation of Sunscreens on Marine Plankton. Cosmetics, 9(1), 20. https://doi.org/10.3390/cosmetics9010020







