Broussonetia papyrifera Promotes Hair Growth Through the Regulation of β-Catenin and STAT6 Target Proteins: A Phototrichogram Analysis of Clinical Samples
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture, Chemicals and Antibodies
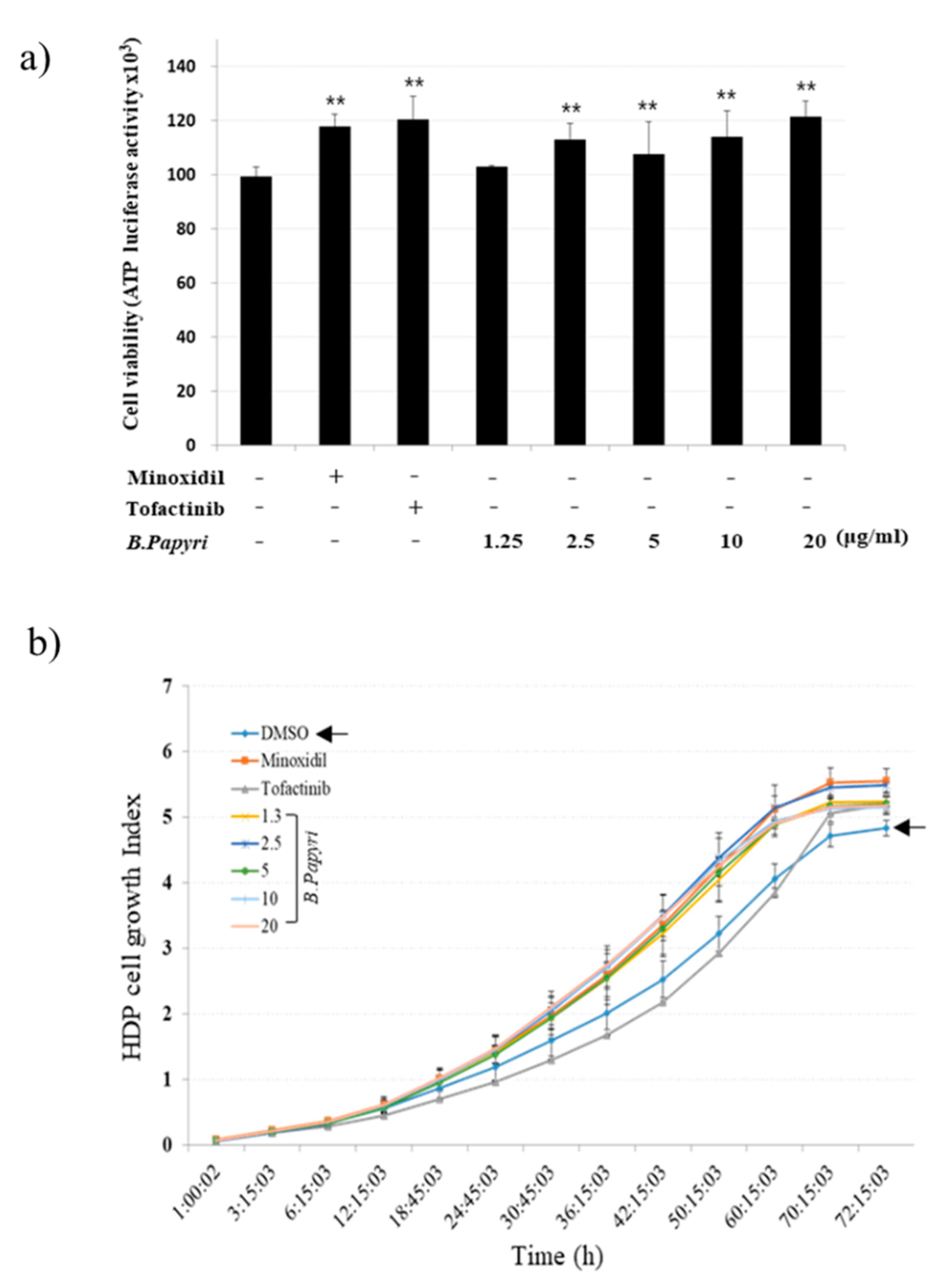
2.2. Real-Time Cell Analyzer (RTCA) System
2.3. CellTiter-Glo® Luminescent Cell Proliferation Assay
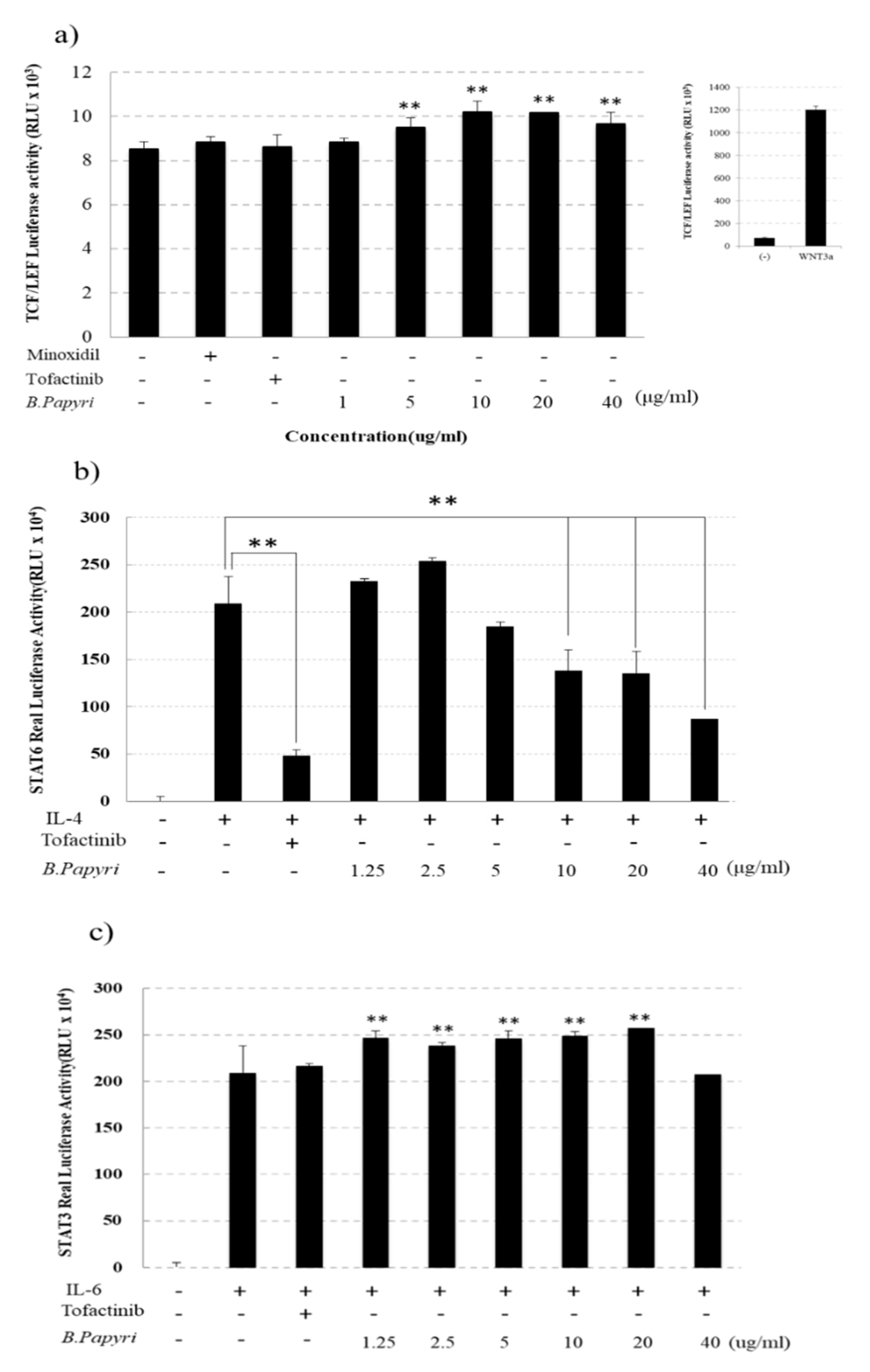
2.4. Measurement of Luciferase-Reporter Activity
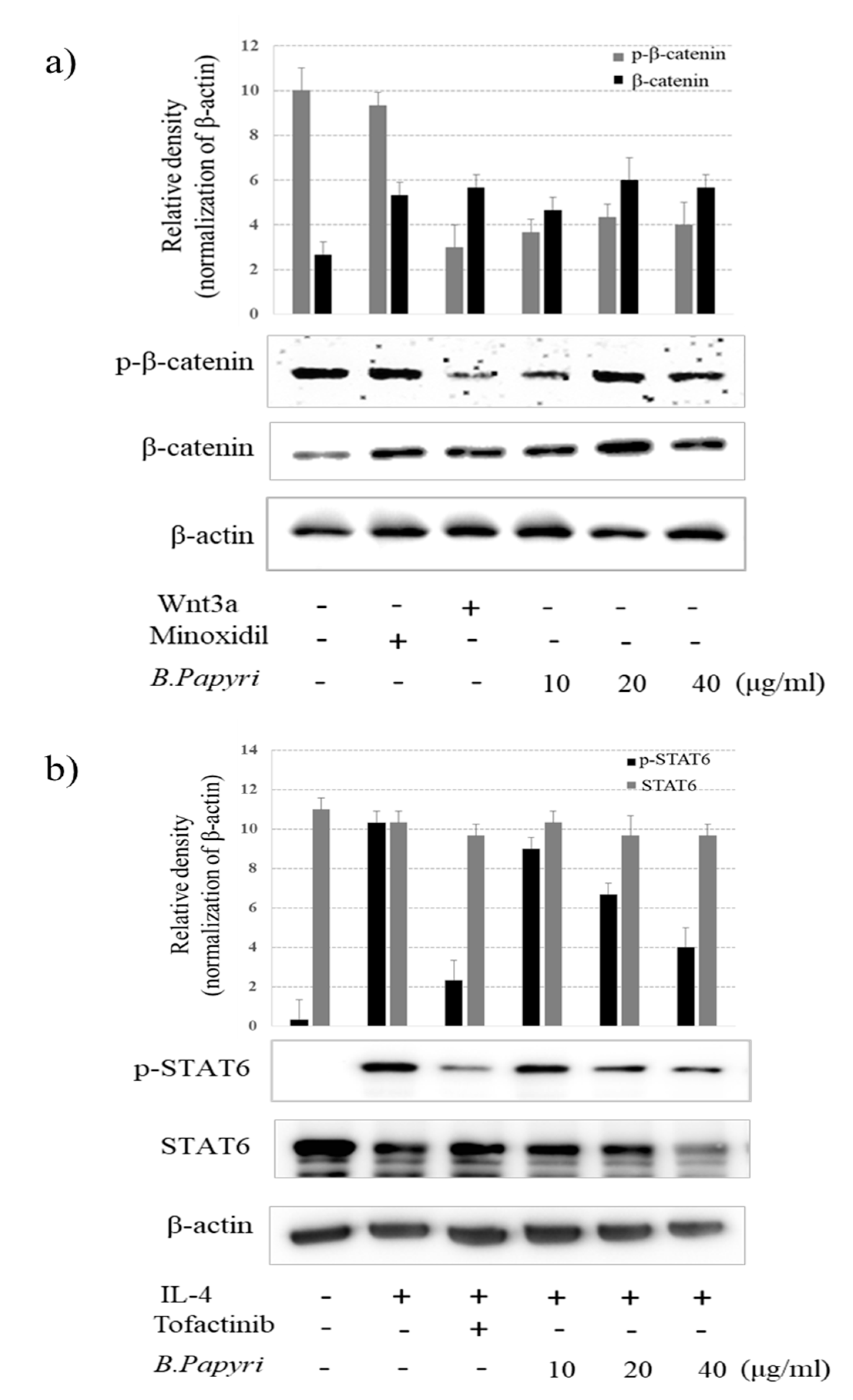
2.5. Western Blotting
2.6. Clinical Subjects and Study Design
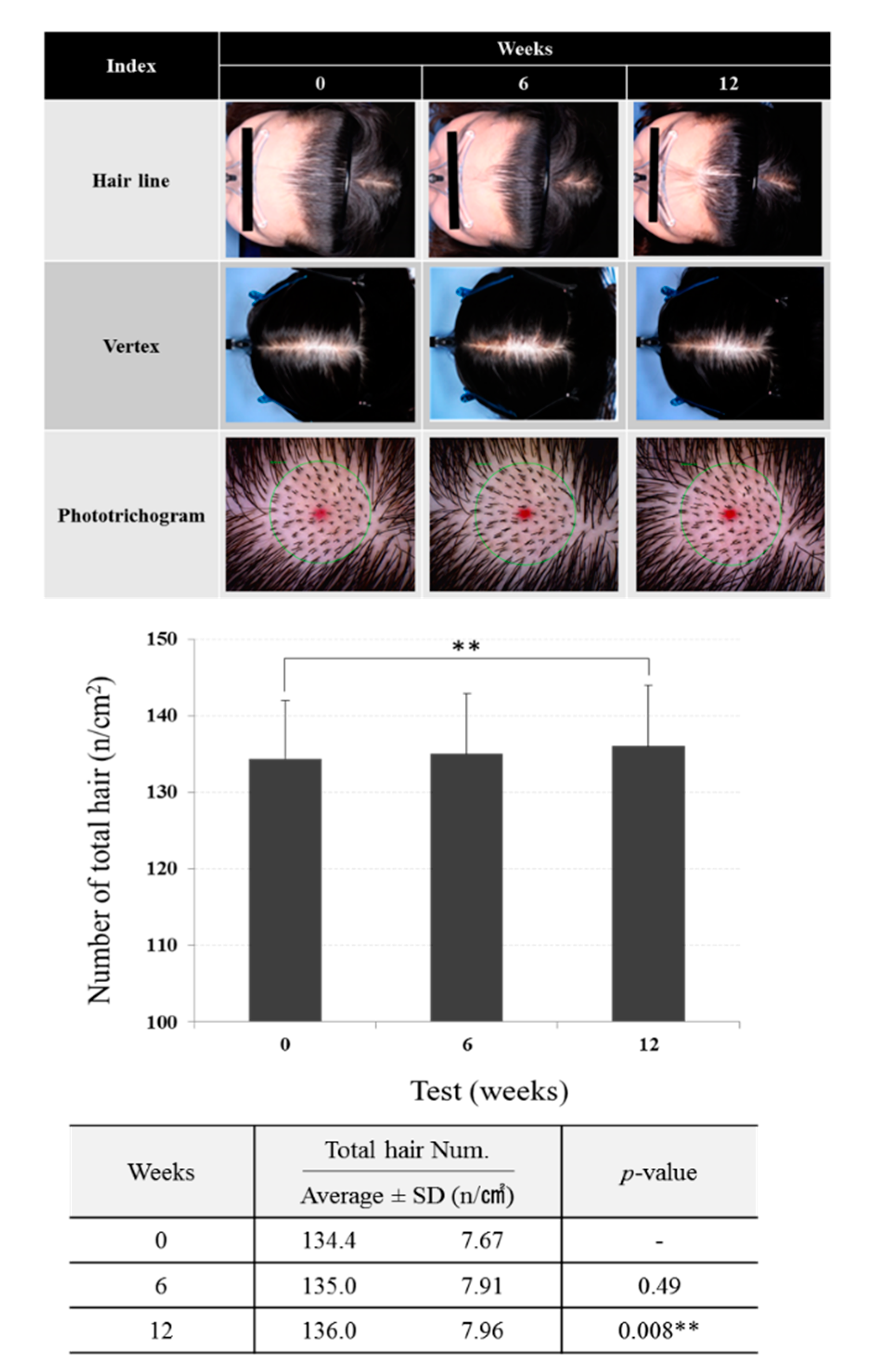
2.7. Hair Density and Total Hair Counts
2.8. Statistical Analysis
3. Results
3.1. The Effects of B. papyrifera on the Growth of Hair Follicle Dermal Papilla Cells
3.2. Effects of B. Papyrifera on the Reporter Gene Activity of Tcf/Lef, stat3 and stat6 in hHFDP Cell
3.3. Effects of B. Papyrifera on Phosphorylation of β-Catenin and stat6 in hHFDP Cells.
3.4 Effects of Scalp Tonic Enriched With B. papyrifera in Human Subjects
Background information of the Subjects:
3.5. Effects of Scalp Tonic on Hair Density and Total Hair Counts
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cotsarelis, G.; Millar, S.E. Towards a molecular understanding of hair loss and its treatment. Trends Mol. Med. 2001, 7, 293–301. [Google Scholar] [CrossRef]
- Miyashita, H.; Hakamata, Y.; Kobayashi, E.; Kobayashi, K. Characterization of hair follicles induced in implanted, cultured rat keratinocyte sheets. Exp. Dermatol. 2004, 13, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H. A novel concept for determining the direction of implanted hair in hairline correction surgery in East Asian women. Arch. Plast. Surg. 2018, 45, 292–294. [Google Scholar] [CrossRef] [PubMed]
- Suchonwanit, P.; Srisuwanwattana, P.; Chalermroj, N.; Khunkhet, S. A randomized, double-blind controlled study of the efficacy and safety of topical solution of 0.25% finasteride admixed with 3% minoxidil vs. 3% minoxidil solution in the treatment of male androgenetic alopecia. J. Eur. Acad. Dermatol. Venereol. 2018, 32, 2257–2263. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.; De-Eknamkul, W. Potential targets in the discovery of new hair growth promoters for androgenic alopecia. Expert Opin. Ther. Targets 2014, 18, 787–806. [Google Scholar] [CrossRef] [PubMed]
- Lopedota, A.; Denora, N.; Laquintana, V.; Cutrignelli, A.; Lopalco, A.; Tricarico, D.; Maqoud, F.; Curci, A.; Mastrodonato, M.; la Forgia, F.; et al. Alginate-Based Hydrogel Containing Minoxidil/Hydroxypropyl-beta-Cyclodextrin Inclusion Complex for Topical Alopecia Treatment. J. Pharm. Sci. 2018, 107, 1046–1054. [Google Scholar] [CrossRef]
- O’Leary, M. By the way, doctor. Finasteride has been prescribed for my BPH. I think that the most frequent side effect is erectile dysfunction or loss of sexual desire. Would Levita or a similar drug (like Viagra or Cialis) overcome the side effects of finasteride? Harv. Health Lett. 2009, 34, 8. [Google Scholar]
- Ahshawat, M.S.; Saraf, S. Preparation and characterization of herbal creams for improvement of skin viscoelastic properties. Int. J. Cosmet. Sci. 2008, 30, 183–193. [Google Scholar] [CrossRef]
- Patel, S.; Sharma, V.; Chauhan, N.S.; Thakur, M.; Dixit, V.K. Hair Growth: Focus on Herbal Therapeutic Agent. Curr. Drug Discov. Technol. 2015, 12, 21–42. [Google Scholar] [CrossRef]
- Pervin, M.; Hasnat, M.A.; Lee, Y.M.; Kim, D.H.; Jo, J.E.; Lim, B.O. Antioxidant activity and acetylcholinesterase inhibition of grape skin anthocyanin (GSA). Molecules 2014, 19, 9403–9418. [Google Scholar] [CrossRef]
- Zaid, A.N.; Jaradat, N.A.; Eid, A.M.; Al Zabadi, H.; Alkaiyat, A.; Darwish, S.A. Ethnopharmacological survey of home remedies used for treatment of hair and scalp and their methods of preparation in the West Bank-Palestine. BMC Complement. Altern. Med. 2017, 17, 355. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Garcovich, S. Advances in Regenerative Stem Cell Therapy in Androgenic Alopecia and Hair Loss: Wnt pathway, Growth-Factor, and Mesenchymal Stem Cell Signaling Impact Analysis on Cell Growth and Hair Follicle Development. Cells 2019, 8, 466. [Google Scholar] [CrossRef] [PubMed]
- Premanand, A.; Reena Rajkumari, B. Androgen modulation of Wnt/beta-catenin signaling in androgenetic alopecia. Arch. Dermatol. Res. 2018, 310, 391–399. [Google Scholar] [CrossRef]
- Ramot, Y.; Zlotogorski, A. [Jak Inhibitors for the Treatment of Alopecia Areata]. Harefuah 2020, 159, 38–42. [Google Scholar] [PubMed]
- Wang, E.H.C.; Sallee, B.N.; Tejeda, C.I.; Christiano, A.M. JAK Inhibitors for Treatment of Alopecia Areata. J. Investig. Dermatol. 2018, 138, 1911–1916. [Google Scholar] [CrossRef]
- Ryu, H.W.; Park, M.H.; Kwon, O.K.; Kim, D.Y.; Hwang, J.Y.; Jo, Y.H.; Ahn, K.S.; Hwang, B.Y.; Oh, S.R. Anti-inflammatory flavonoids from root bark of Broussonetia papyrifera in LPS-stimulated RAW264.7 cells. Bioorg. Chem. 2019, 92, 103233. [Google Scholar] [CrossRef]
- Wu, W.T. Evaluation of anti-inflammatory effects of Broussonetia papyrifera stem bark. Indian J. Pharmacol. 2012, 44, 26–30. [Google Scholar] [CrossRef]
- Si, B.; Tao, H.; Zhang, X.; Guo, J.; Cui, K.; Tu, Y.; Diao, Q. Effect of Broussonetia papyrifera L. (paper mulberry) silage on dry matter intake, milk composition, antioxidant capacity and milk fatty acid profile in dairy cows. Asian-Australas J. Anim. Sci. 2018, 31, 1259–1266. [Google Scholar] [CrossRef]
- Ko, H.H.; Chang, W.L.; Lu, T.M. Antityrosinase and antioxidant effects of ent-kaurane diterpenes from leaves of Broussonetia papyrifera. J. Nat. Prod. 2008, 71, 1930–1933. [Google Scholar] [CrossRef]
- Park, S.; Fudhaili, A.; Oh, S.S.; Lee, K.W.; Madhi, H.; Kim, D.H.; Yoo, J.; Ryu, H.W.; Park, K.H.; Kim, K.D. Cytotoxic effects of kazinol A derived from Broussonetia papyrifera on human bladder cancer cells, T24 and T24R2. Phytomedicine 2016, 23, 1462–1468. [Google Scholar] [CrossRef]
- Lin, L.W.; Chen, H.Y.; Wu, C.R.; Liao, P.M.; Lin, Y.T.; Hsieh, M.T.; Ching, H. Comparison with various parts of Broussonetia papyrifera as to the antinociceptive and anti-inflammatory activities in rodents. Biosci. Biotechnol. Biochem. 2008, 72, 2377–2384. [Google Scholar] [CrossRef] [PubMed]
- Sohn, H.Y.; Kwon, C.S.; Son, K.H. Fungicidal effect of prenylated flavonol, papyriflavonol A, isolated from Broussonetia papyrifera (L.) vent. against Candida albicans. J. Microbiol. Biotechnol. 2010, 20, 1397–1402. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.E.; Choi, H.C.; Lee, I.C.; Yuk, D.Y.; Lee, H.; Choi, B.Y. 3-Deoxysappanchalcone Promotes Proliferation of Human Hair Follicle Dermal Papilla Cells and Hair Growth in C57BL/6 Mice by Modulating WNT/beta-Catenin and STAT Signaling. Biomol. Ther. 2016, 24, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, A.; Kaneko, T. A New Classification of Early Female Pattern Hair Loss. Int. J. Trichology 2018, 10, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.S.; Ro, B.I.; Hong, S.P.; Bak, H.; Sim, W.Y.; Kim, D.W.; Park, J.K.; Ihm, C.W.; Eun, H.C.; Kwon, O.S.; et al. A new classification of pattern hair loss that is universal for men and women: Basic and specific (BASP) classification. J. Am. Acad. Dermatol. 2007, 57, 37–46. [Google Scholar] [CrossRef]
- Baek, J.H.; Lee, S.Y.; Yoo, M.; Park, W.S.; Lee, S.J.; Boo, Y.C.; Koh, J.S. Effects of a new mild shampoo for preventing hair loss in Asian by a simple hand-held phototrichogram technique. Int J. Cosmet. Sci. 2011, 33, 491–496. [Google Scholar] [CrossRef]
- Alonso, L.; Fuchs, E. The hair cycle. J. Cell Sci. 2006, 119, 391–393. [Google Scholar] [CrossRef]
- Zgonc Skulj, A.; Poljsak, N.; Kocevar Glavac, N.; Kreft, S. Herbal preparations for the treatment of hair loss. Arch. Dermatol. Res. 2019. [Google Scholar] [CrossRef]
- Botchkarev, V.A.; Kishimoto, J. Molecular control of epithelial-mesenchymal interactions during hair follicle cycling. J. Investig. Dermatol. Symp. Proc. 2003, 8, 46–55. [Google Scholar] [CrossRef]
- Kang, B.M.; Shin, S.H.; Kwack, M.H.; Shin, H.; Oh, J.W.; Kim, J.; Moon, C.; Moon, C.; Kim, J.C.; Kim, M.K.; et al. Erythropoietin promotes hair shaft growth in cultured human hair follicles and modulates hair growth in mice. J. Dermatol. Sci. 2010, 59, 86–90. [Google Scholar] [CrossRef]
- Kishimoto, J.; Burgeson, R.E.; Morgan, B.A. Wnt signaling maintains the hair-inducing activity of the dermal papilla. Genes Dev. 2000, 14, 1181–1185. [Google Scholar] [PubMed]
- Miller, J.; Djabali, K.; Chen, T.; Liu, Y.; Ioffreda, M.; Lyle, S.; Christiano, A.M.; Holick, M.; Cotsarelis, G. Atrichia caused by mutations in the vitamin D receptor gene is a phenocopy of generalized atrichia caused by mutations in the hairless gene. J. Investig. Dermatol. 2001, 117, 612–617. [Google Scholar] [CrossRef]
- Cheon, H.I.; Bae, S.; Ahn, K.J. Flavonoid Silibinin Increases Hair-Inductive Property Via Akt and Wnt/beta-Catenin Signaling Activation in 3-Dimensional-Spheroid Cultured Human Dermal Papilla Cells. J. Microbiol. Biotechnol. 2019, 29, 321–329. [Google Scholar] [CrossRef]
- Lee, Y.R.; Bae, S.; Kim, J.Y.; Lee, J.; Cho, D.H.; Kim, H.S.; An, I.S.; An, S. Monoterpenoid Loliolide Regulates Hair Follicle Inductivity of Human Dermal Papilla Cells by Activating the Akt/beta-Catenin Signaling Pathway. J. Microbiol. Biotechnol. 2019, 29, 1830–1840. [Google Scholar] [CrossRef]
- Zhang, H.; Shi, Q.; Nan, W.; Wang, Y.; Wang, S.; Yang, F.; Li, G. Ginkgolide B and bilobalide promote the growth and increase beta-catenin expression in hair follicle dermal papilla cells of American minks. Biofactors 2019, 45, 950–958. [Google Scholar] [CrossRef]
- Sano, S.; Kira, M.; Takagi, S.; Yoshikawa, K.; Takeda, J.; Itami, S. Two distinct signaling pathways in hair cycle induction: Stat3-dependent and -independent pathways. Proc. Natl. Acad. Sci. USA 2000, 97, 13824–13829. [Google Scholar] [CrossRef]
- Doles, J.; Storer, M.; Cozzuto, L.; Roma, G.; Keyes, W.M. Age-associated inflammation inhibits epidermal stem cell function. Genes Dev. 2012, 26, 2144–2153. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.; Fletcher, S.; Roth, E.; Wu, C.; Chun, A.; Horsley, V. Calcineurin/Nfatc1 signaling links skin stem cell quiescence to hormonal signaling during pregnancy and lactation. Genes Dev. 2014, 28, 983–994. [Google Scholar] [CrossRef] [PubMed]
- Harel, S.; Higgins, C.A.; Cerise, J.E.; Dai, Z.; Chen, J.C.; Clynes, R.; Christiano, A.M. Pharmacologic inhibition of JAK-STAT signaling promotes hair growth. Sci. Adv. 2015, 1, e1500973. [Google Scholar] [CrossRef]
- Garza, L.A.; Yang, C.C.; Zhao, T.; Blatt, H.B.; Lee, M.; He, H.; Stanton, D.C.; Carrasco, L.; Spiegel, J.H.; Tobias, J.W.; et al. Bald scalp in men with androgenetic alopecia retains hair follicle stem cells but lacks CD200-rich and CD34-positive hair follicle progenitor cells. J. Clin. Investig. 2011, 121, 613–622. [Google Scholar] [CrossRef]
- Gentile, P.; Scioli, M.G.; Cervelli, V.; Orlandi, A.; Garcovich, S. Autologous Micrografts from Scalp Tissue: Trichoscopic and Long-Term Clinical Evaluation in Male and Female Androgenetic Alopecia. Biomed. Res. Int. 2020, 2020, 7397162. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P. Autologous Cellular Method Using Micrografts of Human Adipose Tissue Derived Follicle Stem Cells in Androgenic Alopecia. Int. J. Mol. Sci. 2019, 20, 3446. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Test Group (n = 11) n (%) | |
|---|---|---|
| Sex | Male | 2 (18) |
| Female | 9 (81) | |
| Age | Mean ± SD | 43.4 ± 9.25 |
| Median | 44 | |
| Max, Min | 53, 21 | |
| 20–29 | 1 (9) | |
| 30–39 | 1 (9) | |
| 40–49 | 6 (55) | |
| ≥50 | 3 (27) | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, Y.H.; Nam, G.; Kim, M.-K.; Cho, S.-C.; Choi, B.Y. Broussonetia papyrifera Promotes Hair Growth Through the Regulation of β-Catenin and STAT6 Target Proteins: A Phototrichogram Analysis of Clinical Samples. Cosmetics 2020, 7, 40. https://doi.org/10.3390/cosmetics7020040
Lee YH, Nam G, Kim M-K, Cho S-C, Choi BY. Broussonetia papyrifera Promotes Hair Growth Through the Regulation of β-Catenin and STAT6 Target Proteins: A Phototrichogram Analysis of Clinical Samples. Cosmetics. 2020; 7(2):40. https://doi.org/10.3390/cosmetics7020040
Chicago/Turabian StyleLee, Young Han, Gaewon Nam, Myong-Ki Kim, Seok-Cheol Cho, and Bu Young Choi. 2020. "Broussonetia papyrifera Promotes Hair Growth Through the Regulation of β-Catenin and STAT6 Target Proteins: A Phototrichogram Analysis of Clinical Samples" Cosmetics 7, no. 2: 40. https://doi.org/10.3390/cosmetics7020040
APA StyleLee, Y. H., Nam, G., Kim, M.-K., Cho, S.-C., & Choi, B. Y. (2020). Broussonetia papyrifera Promotes Hair Growth Through the Regulation of β-Catenin and STAT6 Target Proteins: A Phototrichogram Analysis of Clinical Samples. Cosmetics, 7(2), 40. https://doi.org/10.3390/cosmetics7020040





