Novel Lipidized Derivatives of the Bioflavonoid Hesperidin: Dermatological, Cosmetic and Chemopreventive Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. General
2.2. Chemistry
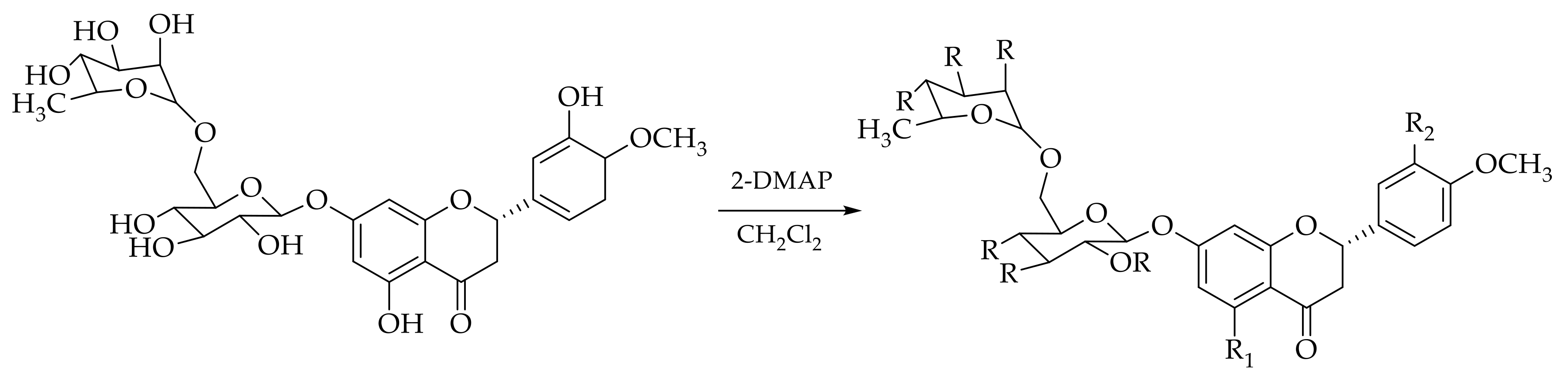
2.2.1. Synthesis of Epta-Tert-Butyl Hesperidin (Esp1)
2.2.2. Synthesis of Epta-Cyclohexylacetyl Hesperidin (Esp2)
2.2.3. Synthesis of Epta-Benzoyl Hesperidin (Esp3)
2.2.4. Synthesis of Epta-Naphtoyl Hesperidin (Esp4)
2.2.5. Synthesis of Octa-Cinnamoyl Hesperidin (Esp5)
2.3. Determination of LogP
2.4. Antioxidant Assays
2.4.1. PCL Test (Photochemiluminescence)
2.4.2. DPPH Test (Diphenylpicrylhydrazyl)
2.4.3. FRAP Assay (Ferric Reducing Antioxidant Power)
2.5. Antifungal Activity
2.5.1. Microorganisms
2.5.2. Antifungal Inhibition Growth Test
2.6. Preparation and Stability Test of Cosmetic Formulations
2.7. Cell Cultures and Assays
2.7.1. K562 Cell Culture
2.7.2. Anti-Proliferative Activities
2.7.3. Pro-Apoptotic Effects
2.8. Statistical Evaluations
3. Results and Discussion
3.1. Chemistry
3.2. LogP Values
3.3. Antifungal Activity
3.4. Antioxidant Activity
3.5. Stability Studies
3.6. Antiproliferative Activity on Human Leukemic K562 Cells
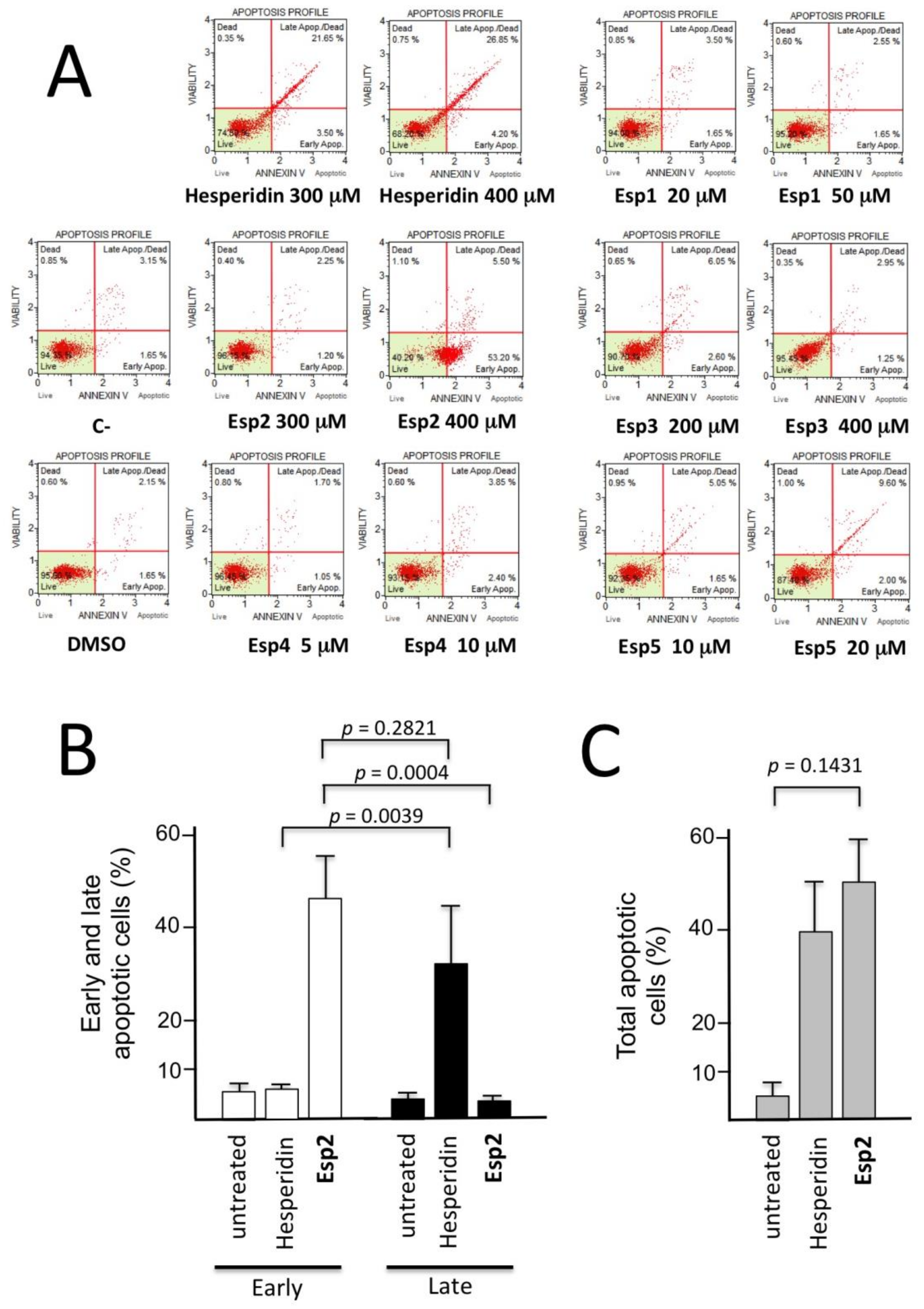
3.7. Pro-Apoptotic Effect of Hesperidin and Its Analogues Esp1-5 on the K562 Cell Line
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- King, A.; Young, G. Characteristics and Occurrence of Phenolic Phytochemicals. J. Am. Diet. Assoc. 1999, 99, 213–218. [Google Scholar] [CrossRef]
- Lin, D.; Xiao, M.; Zhao, J.; Li, Z.; Xing, B.; Li, X.; Kong, M.; Li, L.; Zhang, Q.; Liu, Y.; et al. An Overview of Plant Phenolic Compounds and Their Importance in Human Nutrition and Management of Type 2 Diabetes. Molecules 2016, 21, 1374. [Google Scholar] [CrossRef] [PubMed]
- Banjarnahor, S.D.S.; Artanti, N. Antioxidant properties of flavonoids. Med. J. Indones. 2014, 23, 239–244. [Google Scholar] [CrossRef]
- Kostyuk, V.; Potapovich, A.; Albuhaydar, A.R.; Mayer, W.; De Luca, C.; Korkina, L. Natural Substances for Prevention of Skin Photoaging: Screening Systems in the Development of Sunscreen and Rejuvenation Cosmetics. Rejuvenation Res. 2018, 21, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Choi, S.I.; Jung, T.D.; Cho, B.Y.; Lee, J.H.; Kim, S.H.; Yoon, S.A.; Ham, Y.M.; Yoon, W.J.; Cho, J.H.; et al. Anti-Photoaging Effect of Jeju Putgyul (Unripe Citrus) Extracts on Human Dermal Fibroblasts and Ultraviolet B-induced Hairless Mouse Skin. Int. J. Mol. Sci. 2017, 25, 2052. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Chen, J.; Xiao, A.; Liu, L. Antibacterial Activity of Polyphenols: Structure-Activity Relationship and Influence of Hyperglycemic Condition. Molecules 2017, 6, 1913. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, R.W. History, world distribution, botany and varieties. In The Citrus Industry; Reuther, W., Webber, H., Batchelor, L., Eds.; University of California Press: Berkeley, CA, USA, 1967; pp. 431–591. [Google Scholar]
- Fluckinger, F.A.; Hanbury, D. Pharmacographia—A History of the Principle Drugs of Vegetable Origin; International Book Distributors: Delhi, India, 1986; pp. 104–105. [Google Scholar] [CrossRef]
- Chen, X.M.; Tait, A.R.; Kitts, D.D. Flavonoid composition of orange peel and its association with antioxidant and anti-inflammatory activities. Food Chem. 2017, 218, 15–21. [Google Scholar] [CrossRef]
- Ratty, A.K.; Das, N.P. Effects of flavonoids on nonenzymatic lipid peroxidation: Structure-activity relationship. Biochem. Med. Metab. Biol. 1988, 39, 69–79. [Google Scholar] [CrossRef]
- Ahmadi, A.; Shadboorestan, A. Oxidative stress and cancer; the role of hesperidin, a citrus natural bioflavonoid, as a cancer chemoprotective agent. Nutr. Cancer 2016, 68, 29–39. [Google Scholar] [CrossRef]
- Jeong, H.; Phan, A.N.H.; Choi, J.W. Anti-cancer Effects of Polyphenolic Compounds in Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitor-resistant Non-small Cell Lung Cancer. Pharmacogn. Mag. 2017, 13, 595–599. [Google Scholar] [CrossRef]
- Montanari, A.; Chen, J.; Widmer, W. Citrus flavonoids: A review of past biological activity against disease. Discovery of new flavonoids from Dancy tangerine cold pressed peel oil solids and leaves. Adv. Exp. Med. Biol. 1998, 439, 103–116. [Google Scholar] [PubMed]
- Wolfram, J.; Scott, B.; Boom, K.; Shen, J.; Borsoi, C.; Suri, K.; Grande, R.; Fresta, M.; Celia, C.; Zhao, Y.; et al. Hesperetin Liposomes for Cancer Therapy. Curr. Drug. Deliv. 2016, 13, 711–719. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Alam, K.; Hossain, M.M.; Fatima, M.; Firdaus, F.; Zafeer, M.F.; Arif, Z.; Ahmed, M.; Nafees, K.A. Anti-arthritogenic and cardioprotective action of hesperidin and daidzein in collagen-induced rheumatoid arthritis. Mol. Cell. Biochem. 2016, 423, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Zaghloul, R.A.; Elsherbiny, N.M.; Kenawy, H.I.; El-Karef, A.; Eissa, L.A.; El-Shishtawy, M.M. Hepatoprotective effect of hesperidin in hepatocellular carcinoma: Involvement of Wnt signaling pathways. Life Sci. 2017, 185, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Wu, J.; Xie, H.; Liu, M.; Takau, I.; Zhang, H.; Xiong, Y.; Xia, C. Inhibitory effect of Hesperetin and Naringenin on human UDP-Glucuronosyltransferase enzymes: Implications for herbal-drug interactions. Biol. Pharm. Bull. 2016, 39, 2052–2059. [Google Scholar] [CrossRef] [PubMed]
- Thilakarathna, S.H.; Rupasinghe, H.P.V. Flavonoid Bioavailability and Attempts for Bioavailability Enhancement. Nutrients 2013, 5, 3367–3387. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.Y.; Lin, Y.K.; Lin, C.F.; Wang, P.W.; Chen, E.L.; Fang, J.Y. Elucidating the Skin Delivery of Aglycone and Glycoside Flavonoids: How the Structures Affect Cutaneous Absorption. Nutrients 2017, 9, 1304. [Google Scholar] [CrossRef] [PubMed]
- Hultberg, B.; Sjoblad, S.; Ockerman, P.A. Glycosidases in human skin fibroblast cultures α-fucosidase, α-galactosidase, α-glucosidase, β-mannosidase, and N-acetyl-α-glueosaminidase. Acta Paediatr. 1975, 64, 123–131. [Google Scholar] [CrossRef]
- Engen, A.; Maeda, J.; Wozniak, D.E.; Brents, C.A.; Bell, J.J.; Uesaka, M.; Aizawa, Y.; Kato, T.A. Induction of cytotoxic and genotoxic responses by natural and novel Quercetin glycosides. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2015, 784–785, 15–22. [Google Scholar] [CrossRef]
- Souza, C.; de Freitas, L.A.; Maia Campos, P.M. Topical Formulation Containing Beeswax-Based Nanoparticles Improved in vivo Skin Barrier Function. AAPS Pharm. Sci. Tech. 2017, 18, 2505–2516. [Google Scholar] [CrossRef]
- Depieri, L.V.; Praça, F.S.; Campos, P.M.; Bentley, M.V. Advances in the bioanalytical study of drug delivery across the skin. Ther. Deliv. 2015, 6, 571–594. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Figueroa, J.M.; Pessoa-Mahana, D.C.; Palavecino-González, E.M.; Mella-Raipán, J.; Espinosa-Bustos, C.; Lagos-Muñoz, M.E. Evaluation of the Membrane Permeability (PAMPA and Skin) of Benzimidazoles with Potential Cannabinoid Activity and their Relation with the Biopharmaceutics Classification System (BCS). AAPS Pharm. Sci. Tech. 2011, 12, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Herman, A.; Herman, A.P. Essential oils and their constituents as skin penetration enhancer for transdermal drug delivery: A review. J. Pharm. Pharmacol. 2015, 67, 473–485. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, S.; Srirangam, R. Solubility, Stability, Physicochemical Characteristics and In Vitro Ocular Tissue Permeability of Hesperidin: A Natural Bioflavonoid. Pharm. Res. 2009, 26, 1217–1225. [Google Scholar] [CrossRef] [PubMed]
- Cao, R.; Xue, Y.; Strappe, P.; Blanchard, C.; Zhou, Z. Natural products derived from tea on the solubility of hesperidin by LC-TOF/MS and NMR. Int. J. Food Prop. 2017, 20 (Suppl. 1), S270–S278. [Google Scholar] [CrossRef]
- Tozuka, Y.; Imono, M.; Uchiyama, H.; Takeuchi, H. A novel application of α-glucosyl hesperidin for nanoparticle formation of active pharmaceutical ingredients by dry grinding. Eur. J. Pharm. Biopharm. 2011, 79, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Kilor, V.A. Design and development of novel microemulsion based topical formulation of Hesperidin. Int. J. Pharm. Pharm. Sci. 2015, 7, 238–245. [Google Scholar]
- Vertuani, S.; Beghelli, E.; Scalambra, E.; Malisardi, G.; Copetti, S.; Dal Toso, R.; Baldisserotto, A.; Manfredini, S. Activity and stability studies of verbascoside, a novel antioxidant, in dermo-cosmetic and pharmaceutical topical formulations. Molecules 2011, 16, 7068–7080. [Google Scholar] [CrossRef]
- Baldisserotto, A.; Malisardi, G.; Scalambra, E.; Andreotti, E.; Romagnoli, C.; Vicentini, C.B.; Manfredini, S.; Vertuani, S. Synthesis, antioxidant and antimicrobial activity of a new phloridzin derivative for dermo-cosmetic applications. Molecules 2012, 17, 13275–13289. [Google Scholar] [CrossRef]
- Baldisserotto, A.; Vertuani, S.; Bino, A.; De Lucia, D.; Lampronti, I.; Milani, R.; Gambari, R.; Manfredini, S. Design, synthesis and biological activity of a novel Rutin analogue with improved lipid soluble properties. Bioorg. Med. Chem. 2015, 23, 264–271. [Google Scholar] [CrossRef]
- Lewin, G.; Popov, I. Oxidants and Antioxidants Part B—Antioxidative Homeostasis: Characterization by Means of Chemiluminescent Technique. Methods Enzymol. 1999, 300, 437–456. [Google Scholar] [CrossRef]
- Schlesier, K.; Harwat, M.; Böhm, V.; Bitsch, R. Assessment of antioxidant activity by using different in vitro methods. Free Radic. Res. 2002, 36, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Li, J.; Rangarajan, M.; Shao, Y.; La Voie, E.J.; Huang, T.-C.; Ho, C.-T. Antioxidative phenolic compound from sage (salvia officinalis). J. Agric. Food Chem. 1998, 46, 4868–4873. [Google Scholar] [CrossRef]
- Xu, G.; Ye, X.; Chen, J.; Liu, D. Effect of heat treatment on the phenolic compounds and antioxidant capacity of citrus peel extract. J. Agric. Food Chem. 2007, 55, 330–335. [Google Scholar] [CrossRef] [PubMed]
- Romagnoli, C.; Baldisserotto, A.; Vicentini, C.B.; Mares, D.; Andreotti, E.; Vertuani, S.; Manfredini, S. Antidermatophytic Action of Resorcinol Derivatives: Ultrastructural Evidence of the Activity of Phenylethyl Resorcinol against Microsporum gypseum. Molecules 2016, 21, 1306. [Google Scholar] [CrossRef] [PubMed]
- Lozzio, C.B.; Lozzio, B.B. Human chronic myelogenous leukemia cell-line with positive Philadelphia-chromosome. Blood 1975, 45, 321–334. [Google Scholar] [PubMed]
- Lampronti, I.; Khan, M.T.; Borgatti, M.; Bianchi, N.; Gambari, R. Inhibitory Effects of Bangladeshi Medicinal Plant Extracts on Interactions between Transcription Factors and Target DNA Sequences. Evid.-Based Complement. Altern. Med. 2008, 5, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Lampronti, I.; Borgatti, M.; Vertuani, S.; Manfredini, S.; Gambari, R. Modulation of the expression of the pro-inflammatory IL-8 gene in cystic fibrosis cells by extracts deriving from olive mill waste water. Evid.-Based Complement. Altern. Med. 2013, 2013, 960603. [Google Scholar] [CrossRef]
- Wu, T.; He, M.; Zang, X.; Zhou, Y.; Qiu, T.; Pan, S.; Xu, X. A structure–activity relationship study of flavonoids as inhibitors of E. coli by membrane interaction effect. Biochim. Biophys. Acta 2013, 1828, 2751–2756. [Google Scholar] [CrossRef]
- Mishra, K. Structure-Activity Relationship of Antioxidative Property of Hesperidin. Int. J. Pharma. Erud. 2013, 2, 40–53. [Google Scholar]
- Khan, M.T.; Lampronti, I.; Martello, D.; Bianchi, N.; Jabbar, S.; Choudhuri, M.S.; Datta, B.K.; Gambari, R. Identification of pyrogallol as an antiproliferative compound present in extracts from the medicinal plant Emblica officinalis: Effects on in vitro cell growth of human tumor cell lines. Int. J. Oncol. 2002, 21, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.A.; Lee, S.H.; Lee, Y.J.; Baeg, S.M.; Shim, J.H. Hesperidin Induces Apoptosis by Inhibiting Sp1 and Its Regulatory Protein in MSTO-211H Cells. Biomol. Ther. (Seoul) 2012, 20, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Fischer, N.; Seo, E.J.; Efferth, T. Prevention from radiation damage by natural products. Phytomedicine 2018, 47, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Waldman, A.; Schmults, C. Cutaneous Squamous Cell Carcinoma. Hematol. Oncol. Clin. N. Am. 2019, 33, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Rabbie, R.; Ferguson, P.; Molina-Aguilar, C.; Adams, D.J.; Robles-Espinoza, C.D. Melanoma subtypes: Genomic profiles, prognostic molecular markers and therapeutic possibilities. J. Pathol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Polini, B.; Digiacomo, M.; Carpi, S.; Bertini, S.; Gado, F.; Saccomanni, G.; Macchia, M.; Nieri, P.; Manera, C.; Fogli, S. Oleocanthal and oleacein contribute to the in vitro therapeutic potential of extra virgin oil-derived extracts in non-melanoma skin cancer. Toxicol. In Vitro 2018, 52, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Rajan, N.; Langtry, J.A. Generalized exacerbation of psoriasis associated with imiquimod cream treatment of superficial basal cell carcinomas. Clin. Exp. Dermatol. 2006, 31, 140–141. [Google Scholar] [CrossRef]
- Zoschke, C.; Schilrreff, P.; Romero, E.L.; Brandner, J.M.; Schafer-Korting, M. Dendritic nanoparticles for cutaneous drug delivery—Testing in human skin and reconstructed human skin. Curr. Pharm. Des. 2015, 21, 2784–2800. [Google Scholar] [CrossRef]
- Brauchle, E.; Johannsen, H.; Nolan, S.; Thude, S.; Schenke-Layland, K. Design and analysis of a squamous cell carcinoma in vitro model system. Biomaterials 2013, 34, 7401–7407. [Google Scholar] [CrossRef]
- Saleh, J. Murine models of melanoma. Pathol. Res. Pract. 2018, 214, 1235–1238. [Google Scholar] [CrossRef]
- Amberg, N.; Holcmann, M.; Glitzner, E.; Novoszel, P.; Stulnig, G.; Sibilia, M. Mouse models of nonmelanoma skin cancer. Methods Mol. Biol. 2015, 1267, 217–250. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Yang, Y.R.; Chen, W.; Chen, M.H.; Wang, H.; Wang, X.D.; Sun, L.L.; Wang, F.Z.; Wang, D.C. Fluoxetine synergizes with temozolomide to induce the CHOP-dependent endoplasmic reticulum stress-related apoptosis pathway in glioma cells. Oncol. Rep. 2016, 36, 676–684. [Google Scholar] [CrossRef] [PubMed]
- Giacomelli, C.; Daniele, S.; Natali, L.; Iofrida, C.; Flamini, G.; Braca, A.; Trincavelli, M.L.; Martini, C. Carnosol controls the human glioblastoma stemness features through the epithelial-mesenchymal transition modulation and the induction of cancer stem cell apoptosis. Sci. Rep. 2017, 7, 15174. [Google Scholar] [CrossRef] [PubMed]

| Analytical Conditions | |||||
|---|---|---|---|---|---|
| Compound | % Solvent A | % Solvent B | Flow Rate (mL/min) | λ (nm) | Column Temperature (°C) |
| Hesperidin | 77 | 23 | 1.5 | 270 | 27 |
| Esp1 | 2 | 98 | 1.5 | 270 | 27 |
| Esp2 | 77 | 23 | 1.2 | 280 | 27 |
| Esp3, Esp5 | 5 | 95 | 1.2 | 280 | 27 |
| Esp4 | 55 | 45 | 1.2 | 280 | 27 |
| Compounds | R | R1 | R2 |
|---|---|---|---|
| Esp1 | tert-butyl-acetyl | -H | tert-butyl-acetyl |
| Esp2 | cyclo-hexyl-acetyl | -H | cyclo-hexyl-acetyl |
| Esp3 | benzoyl | -H | benzoyl |
| Esp4 | naphthoyl | -H | naphthoyl |
| Esp5 | cinnamoyl | cinnamoyl | cinnamoyl |
| Compounds | LogP [*] |
|---|---|
| Hesperidin | −0.87 |
| Esp1 | 1.38 |
| Esp2 | 0.40 |
| Esp3 | 2.04 |
| Esp4 | 1.08 |
| Esp5 | 1.04 |
| Compounds | PCL µmolTE/g | FRAP µmolTE/g | DPPH µmolTE/g |
|---|---|---|---|
| Hesperidin | 1068.63 ± 1.12 | 1255.32 ± 4.04 | 216.61 ± 0.87 |
| Esp1 | 2.16 ± 0.09 | 44.37 ± 0.96 | No activity |
| Esp2 | 1.14 ± 0.50 | 20.15 ± 0.58 | No activity |
| Esp3 | 22.58 ± 2.92 | 14.18 ± 0.64 | 6.03 ± 0.31 |
| Esp4 | 6.06 ± 1.70 | 19.61 ± 0.84 | No activity |
| Esp5 | 8.12 ± 1.83 | 18.19 ± 0.77 | No activity |
| Compound | IC50 (µM) on K562 Cell Line |
|---|---|
| Hesperidin | 389.96 ± 35.49 |
| Esp1 | ˃50 (ppt) |
| Esp2 | 376.13 ± 32.29 |
| Esp3 | ˃400 (ppt) |
| Esp4 | 11.12 ± 1.97 |
| Esp5 | 21.68 ± 2.82 |
| Compound | Live Cells (%) | Early Apoptotic Cells (%) | Late Apoptotic Cells (%) | Total Apoptotic Cells (%) |
|---|---|---|---|---|
| C- (untreated cells) | 94.35 | 1.65 | 3.15 | 4.80 |
| Hesperidin 300 µM | 74.05 | 3.50 | 21.65 | 25.15 |
| Hesperidin 400 µM | 68.20 | 4.20 | 26.85 | 31.05 |
| Esp1 20 µM | 94.00 | 1.65 | 3.50 | 5.15 |
| Esp1 50 µM | 95.20 | 1.65 | 2.55 | 4.20 |
| Esp2 300 µM | 96.15 | 1.20 | 2.25 | 3.45 |
| Esp2 400 µM | 40.20 | 53.20 | 5.50 | 58.70 |
| Esp3 200 µM | 90.70 | 2.60 | 6.05 | 4.20 |
| Esp3 400 µM | 95.45 | 1.25 | 2.95 | 8.65 |
| Esp4 5 µM | 96.45 | 1.05 | 1.70 | 2.75 |
| Esp4 10 µM | 93.15 | 2.40 | 3.85 | 6.25 |
| Esp5 10 µM | 92.35 | 1.65 | 5.05 | 6.70 |
| Esp5 20 µM | 87.40 | 2.00 | 9.60 | 11.60 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bino, A.; Vicentini, C.B.; Vertuani, S.; Lampronti, I.; Gambari, R.; Durini, E.; Manfredini, S.; Baldisserotto, A. Novel Lipidized Derivatives of the Bioflavonoid Hesperidin: Dermatological, Cosmetic and Chemopreventive Applications. Cosmetics 2018, 5, 72. https://doi.org/10.3390/cosmetics5040072
Bino A, Vicentini CB, Vertuani S, Lampronti I, Gambari R, Durini E, Manfredini S, Baldisserotto A. Novel Lipidized Derivatives of the Bioflavonoid Hesperidin: Dermatological, Cosmetic and Chemopreventive Applications. Cosmetics. 2018; 5(4):72. https://doi.org/10.3390/cosmetics5040072
Chicago/Turabian StyleBino, Alessia, Chiara Beatrice Vicentini, Silvia Vertuani, Ilaria Lampronti, Roberto Gambari, Elisa Durini, Stefano Manfredini, and Anna Baldisserotto. 2018. "Novel Lipidized Derivatives of the Bioflavonoid Hesperidin: Dermatological, Cosmetic and Chemopreventive Applications" Cosmetics 5, no. 4: 72. https://doi.org/10.3390/cosmetics5040072
APA StyleBino, A., Vicentini, C. B., Vertuani, S., Lampronti, I., Gambari, R., Durini, E., Manfredini, S., & Baldisserotto, A. (2018). Novel Lipidized Derivatives of the Bioflavonoid Hesperidin: Dermatological, Cosmetic and Chemopreventive Applications. Cosmetics, 5(4), 72. https://doi.org/10.3390/cosmetics5040072









