Anti-Aging Properties of Plant Stem Cell Extracts
Abstract
1. Introduction
2. The Characteristics of Plant Stem Cells
- extending the life of fibroblasts and stimulating their activity (e.g., Oryza sativa, Gardenia jasminoides);
- increasing the flexibility of the epidermis (e.g., Symphytum officinale, Capsicum annuum, Opuntia spp.);
- regulating cell division (e.g., Oryza sativa, Lotus japonicus);
- rebuilding damaged epidermis (e.g., Panax ginsgen, Opuntia spp.);
- activating DNA repair of the cells, protecting them from oxidative stress (e.g., Rubus ideaus, Lycopersicon esculentum, e.g., Citrus limon);
- protecting against UV radiation (e.g., Dolichos biflorus, Opuntia ficus indica).
3. Plant Stem Cells with Anti-Aging Effects
4. Antioxidant Activity of Steam Cell Extracts
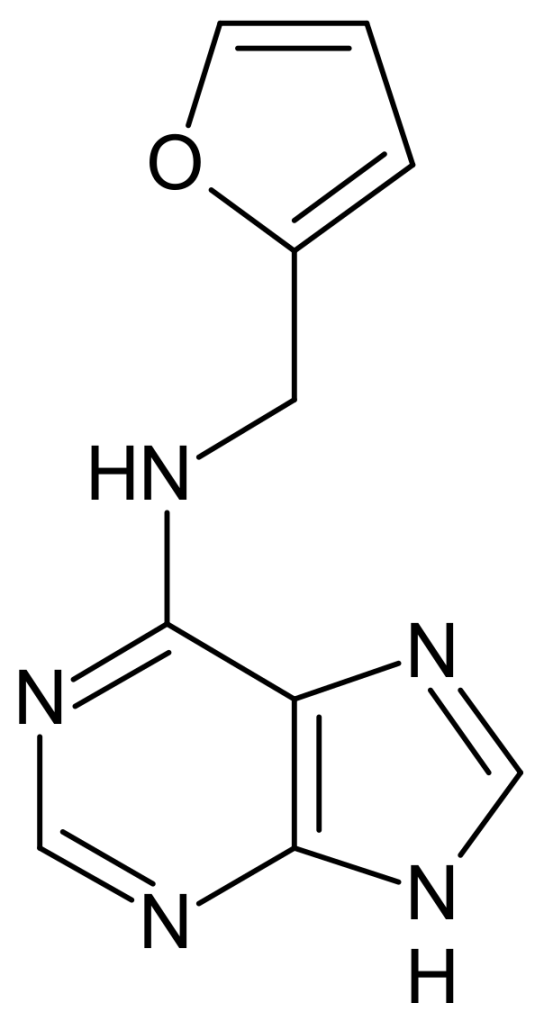
Kinetin—Powerful Antioxidant
5. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Ksiazek, K. Oxidative stress as an universal cause of aging—From human somatic cells to the unicellular yeast and bacteria. Postęp. Biochem. 2010, 56, 260–268. [Google Scholar]
- Trehan, S.; Michniak-Kohn, B.; Beri, K. Plant stem cells in cosmetics: Current trends and future directions. Future Sci. OA 2017, 3, FSO226. [Google Scholar] [CrossRef] [PubMed]
- Sosińska, P.; Mikuła-Pietrasik, J.; Książek, K. Molecular bases of cellular senescence: Hayflick phenomenon 50 years later. Postep. Hig. Med. Dosw. 2016, 70, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Rhein, L.D.; Fluhr, J.W. Starzenie Skóry. Aktualne Strategie Terapeutyczne; MedPharm Polska: Wrocław, Poland, 2013; pp. 3–23. [Google Scholar]
- Industry Experts, Antiaging: Physiology to Formulation; Collective work; Allured Publishing Corporation: Carol Stream, IL, USA, 2006.
- Schmid, D.; Schürch, C.; Blum, P.; Belser, E.; Zülli, F. Plant Stem Cell Extract for Longevity of Skin and Hair. SOFW J. 2008, 134, 30–35. [Google Scholar]
- Moruś, M.; Baran, M.; Rost-Roszkowska, M.; Skotnicka-Graca, U. Plant stem cells as innovation in cosmetics. Acta Pol. Pharm. 2014, 71, 701–707. [Google Scholar] [PubMed]
- Directive No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on Cosmetic Products. Available online: https://ec.europa.eu/health/sites/health/files/endocrine_disruptors/docs/cosmetic_1223_2009_regulation_en.pdf (accessed on 21 September 2018).
- Available online: http://biotechnologia.pl/kosmetologia/artykuly/potencjal-komorek-macierzystych-zamkniety-w-kosmetykach,14952 (accessed on 25 March 2018).
- Schürch, C.; Blum, P.; Zulli, F. Potential of plant cells in culture for cosmetic application. Phytochem. Rev. 2008, 7, 599–605. [Google Scholar] [CrossRef]
- Barbulova, A.; Apone, F.; Colluci, G. Plant cell cultures as source of cosmetic active ingredients. Cosmetics 2014, 1, 94–104. [Google Scholar] [CrossRef]
- Georgiev, V.; Slavov, A.; Vasileva, I.; Pavlov, A. Plant cell culture as emerging technology for production of active cosmetic ingredients. Eng. Life Sci. 2018, 0, 1–20. [Google Scholar] [CrossRef]
- Draelos, Z. Plant stem cells and skin care. Cosmet. Dermatol. 2012, 25, 395–396. [Google Scholar]
- Available online: http://www.naolys.com/media/refine_ginger_en.pdf (accessed on 15 July 2018).
- Price, C. An in-depth look at lilac stem cell extract in skin care. Pract. Dermatol. 2014, 5, 46–48. [Google Scholar]
- Wu, J.; Zhong, J.-J. Production of ginseng and its bioactive components in plant cell culture: Current technological and applied aspects. J. Biotechnol. 1999, 68, 89–99. [Google Scholar] [CrossRef]
- Tito, A.; Carola, A.; Bimonte, M.; Barbulova, A.; Arciello, S.; de Laurentiis, F.; Monoli, I.; Hill, J.; Gibertoni, S.; Colucci, G.; et al. A tomato stem cell extract, containing antioxidant compounds and metal chelating factors, protects skin cells from heavy metal induced damages. Int. J. Cosmet. Sci. 2011, 33, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Caucanas, M.; Montastier, C.; Piérard, G.E.; Quatresooz, P. Dynamics of skin barrier repair following preconditioning by a biotechnology-driven extract from samphire (Crithmum maritimum) stem cells. J. Cosmet. Dermatol. 2011, 10, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Lequeux, C.; Lhoste, A.; Rovere, M.R.; Montastier, C.; Damour, O. Model of in vitro healing to test the influence of dedifferentiated Crithmum maritimum cells on dermal repair and epidermal regeneration. Skin Pharmacol. Physiol. 2011, 24, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, J.A.; Ortuño, A.; Marin, E.R.; Puig, D.G.; Sabater, F. Bioproduction of neohesperidin and naringin in callus cultures of Citrus aurantium. Plant Cell Rep. 1992, 11, 592–596. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.centerchem.com/Products/DownloadFile.aspx?FileID=7918 (accessed on 28 July 2018).
- Bimonte, M.; Tito, A.; Carola, A.; Barbulova, A.; Monoli, I.; Cucchiara, M.; Hill, J.; Colucci, G.; Apone, F. Coffea bengalensis for anti-wrinkle and skin toning applications. Cosmet. Toilet. 2011, 126, 644–650. [Google Scholar]
- Apone, F.; Tito, A.; Carola, A.; Arciello, S.; Tortora, A.; Filippini, L.; Monoli, I.; Cucchiara, M.; Gibertoni, S.; Chrispeels, M.; et al. A mixture of peptides and sugars derived from plant cell walls increases plant defense responses to stress and attenuates ageing-associated molecular changes in cultured skin cells. J. Biotechnol. 2010, 145, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Knott, A.; Koop, U.; Mielke, H.; Reuschlein, K.; Peters, N.; Muhr, G.M.; Lenz, H.; Wensorra, U.; Jaspers, S.; Kolbe, L.; et al. A novel treatment option for photoaged skin. J. Cosmet. Dermatol. 2008, 7, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Velioglu, Y.S.; Mazza, G.; Gao, L.; Oomah, B.D. Antioxidant activity and total phenolics in selected fruits, vegetables, and grain products. J. Agric. Food Chem. 1998, 46, 4113–4117. [Google Scholar] [CrossRef]
- Burda, S.; Oleszek, W. Antioxidant and antiradical activities of flavonoids. J. Agric. Food Chem. 2001, 49, 2774–2779. [Google Scholar] [CrossRef] [PubMed]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; Sofic, E.; Prior, R.L. Antioxidant and prooxidant behavior of flavonoids: structure-activity relationships. Free Radic. Biol. Med. 1997, 22, 749–760. [Google Scholar] [CrossRef]
- Korkina, L.G.; Afanasev, I.B. Antioxidant and chelating properties of flavonoids. Adv. Pharmacol. 1997, 38, 151–163. [Google Scholar] [PubMed]
- Kumar, S.; Pandey, A.K. Chemistry and biological activities of flavonoids: An overview. Sci. World J. 2013. [Google Scholar] [CrossRef] [PubMed]
- Goutzourelas, N.; Stagos, D.; Spanidis, Y.; Liosi, M.; Apostolou, A.; Priftis, A.; Haroutounian, S.; Spandidos, D.A.; Tsatsakis, A.M.; Kouretas, D. Polyphenolic composition of grape stem extracts affects antioxidant activity in endothelial and muscle cells. Mol. Med. Rep. 2015, 12, 5846–5856. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, K. Buddleja davidii stem cell for oxidative and photodamage protection. Available online: https://www.cosmeticsandtoiletries.com/formulating/function/active/142630376.html (accessed on 21 September 2018).
- US20110123646A1. Available online: https://patentimages.storage.googleapis.com/a3/d9/09/0ab7822bff126e/US20110123646A1.pdf (accessed on 21 September 2018).
- Barbulova, A.; Tito, A.; Carola, A.; Bimonte, M.; De Laurentis, F.; D’Ambrosio, P.; Apone, F.; Colucci, G.; Monoli, I.; Cucchiara, M. Raspberry stem cell extract to protect skin from inflammation and oxidative stress. Cosmet. Toiletries 2010, 125, 38–47. [Google Scholar]
- Bazylak, G.; Gryn, A. Antioxidant activity and total flavonoid content in variable phyto-stem cells extracts obtained by high-pressure homogenization method and assigned for use in biocosmetics. Planta Med. 2015, 81. [Google Scholar] [CrossRef]
- Miller, C.O.; Skoog, F.; Von Saltza, M.H.; Strong, F.M. Kinetin, a cell division factor from deoxyribonucleic acid. J. Am. Chem. Soc. 1955, 77, 1392. [Google Scholar] [CrossRef]
- Miller, C.O.; Skoog, F.; Okumura, F.S.; Von Saltza, M.H.; Strong, F.M. Isolation, structure and synthesis of kinetin, a substance promoting cell division. J. Am. Chem. Soc. 1956, 78, 1375–1380. [Google Scholar] [CrossRef]
- Barciszewski, J.; Rattan, S.I.S.; Siboska, G.; Clark, B.F.C. Kinetin—45 years on. Plant Sci. 1999, 148, 37–45. [Google Scholar] [CrossRef]
- Barciszewski, J.; Massino, F.; Clark, B.F.C. Kinetin—A multiactive molecule. Int. J. Biol. Macromol. 2007, 40, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Goswami, K.; Sharma, R.; Singh, P.K.; Singh, G. Micropropagation of seedless lemon (Citrus limon L. cv. Kaghzi Kalan) and assessment of genetic fidelity of micropropagated plants using RAPD markers. Physiol. Mol. Biol. Plants 2013, 19, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Nohynek, L.; Bailey, M.; Tähtiharju, J.; Seppänen-Laakso, T.; Rischer, H.; Oksman-Caldentey, K.-M.; Puupponen-Pimiä, R. Cloudberry (Rubus chamaemorus) cell culture with bioactive substances: Establishment and mass propagation for industrial use. Eng. Life Sci. 2014, 14, 667–675. [Google Scholar] [CrossRef]
- Verbeke, P.; Siboska, G.E.; Clark, B.F.C.; Rattan, S.I.S. Kinetin inhibits protein oxidation and glycoxidation in vitro. Biochem. Biophys. Res. Commun. 2000, 276, 1265–1270. [Google Scholar] [CrossRef] [PubMed]
- Olsen, A.; Siboska, E.; Clark, B.F.C.; Rattan, S.I.S. N6-Furfuryladenine, kinetin, protects against fenton reaction-mediated oxidative damage to DNA. Biochem. Biophys. Res. Commun. 1999, 265, 499–502. [Google Scholar] [CrossRef] [PubMed]
- Jabłońska-Trypyć, A.; Czerpak, R. Cytokinins, their biochemical activity in division process, ageing and apoptosis in human and animal cells. Postęp. Biol. Komorki 2009, 36, 135–154. [Google Scholar]
- Lee, J.H.; Chung, K.Y.; Bang, D.; Lee, K.H. Searching for aging-related proteins in human dermal microvascular endothelial cells treated with anti-aging agents. Proteomics 2006, 6, 1351–1361. [Google Scholar] [CrossRef] [PubMed]
- Tournas, J.A.; Lin, F.H.; Burch, J.A.; Selim, M.A.; Monteiro-Riviere, N.A.; Zielinski, J.E.; Pinnell, S.R. Ubiquinone, idebenone, and kinetin provide ineffective photoprotection to skin when compared to a topical antioxidant combination of Vitamins C and E with ferulic acid. J. Investig. Dermatol. 2006, 126, 1185–1187. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Doi, K. Depigmentation and rejuvenation effects of kinetin on the aged skin of hairless descendants of Mexican hairless dogs. Rejuvenation Res. 2004, 7, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.J.; Weinstein, G.D.; Kricorian, G.J.; Kormeili, T.; McCullough, J.L. Topical kinetin 0.1% lotion for improving the signs and symptoms of rosacea. Clin. Exp. Dermatol. 2007, 32, 693–695. [Google Scholar] [PubMed]
- Jabłońska-Trypuc, A.; Matejczyk, M.; Czerpak, R. N6-benzyladenine and kinetin influence antioxidative stress parameters in human skin fibroblasts. Mol. Cell. Biochem. 2016, 413, 97–107. [Google Scholar] [CrossRef] [PubMed]
- An, S.; Cha, H.J.; Ko, J.M.; Han, H.; Kim, S.Y.; Kim, K.S.; Lee, S.J.; An, I.S.; Kim, S.; Youn, H.J.; et al. Kinetin improves barrier function of the skin by modulating keratinocyte differentiation markers. Ann. Dermatol. 2017, 29, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Gan, S.; Amasino, R.M. Inhibition of leaf senescence by autoregulated production of cytokinin. Science 1995, 270, 1986–1988. [Google Scholar] [CrossRef] [PubMed]
- Rattan, S.I.; Clark, B.F. Kinetin delays the onset of ageing characteristics in human fibroblasts. Biochem. Biophys. Res. Commun. 1994, 201, 665–672. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miastkowska, M.; Sikora, E. Anti-Aging Properties of Plant Stem Cell Extracts. Cosmetics 2018, 5, 55. https://doi.org/10.3390/cosmetics5040055
Miastkowska M, Sikora E. Anti-Aging Properties of Plant Stem Cell Extracts. Cosmetics. 2018; 5(4):55. https://doi.org/10.3390/cosmetics5040055
Chicago/Turabian StyleMiastkowska, Małgorzata, and Elżbieta Sikora. 2018. "Anti-Aging Properties of Plant Stem Cell Extracts" Cosmetics 5, no. 4: 55. https://doi.org/10.3390/cosmetics5040055
APA StyleMiastkowska, M., & Sikora, E. (2018). Anti-Aging Properties of Plant Stem Cell Extracts. Cosmetics, 5(4), 55. https://doi.org/10.3390/cosmetics5040055





