Sea Buckthorn Oil—A Valuable Source for Cosmeceuticals
Abstract
:1. Introduction
1.1. Botanical Features
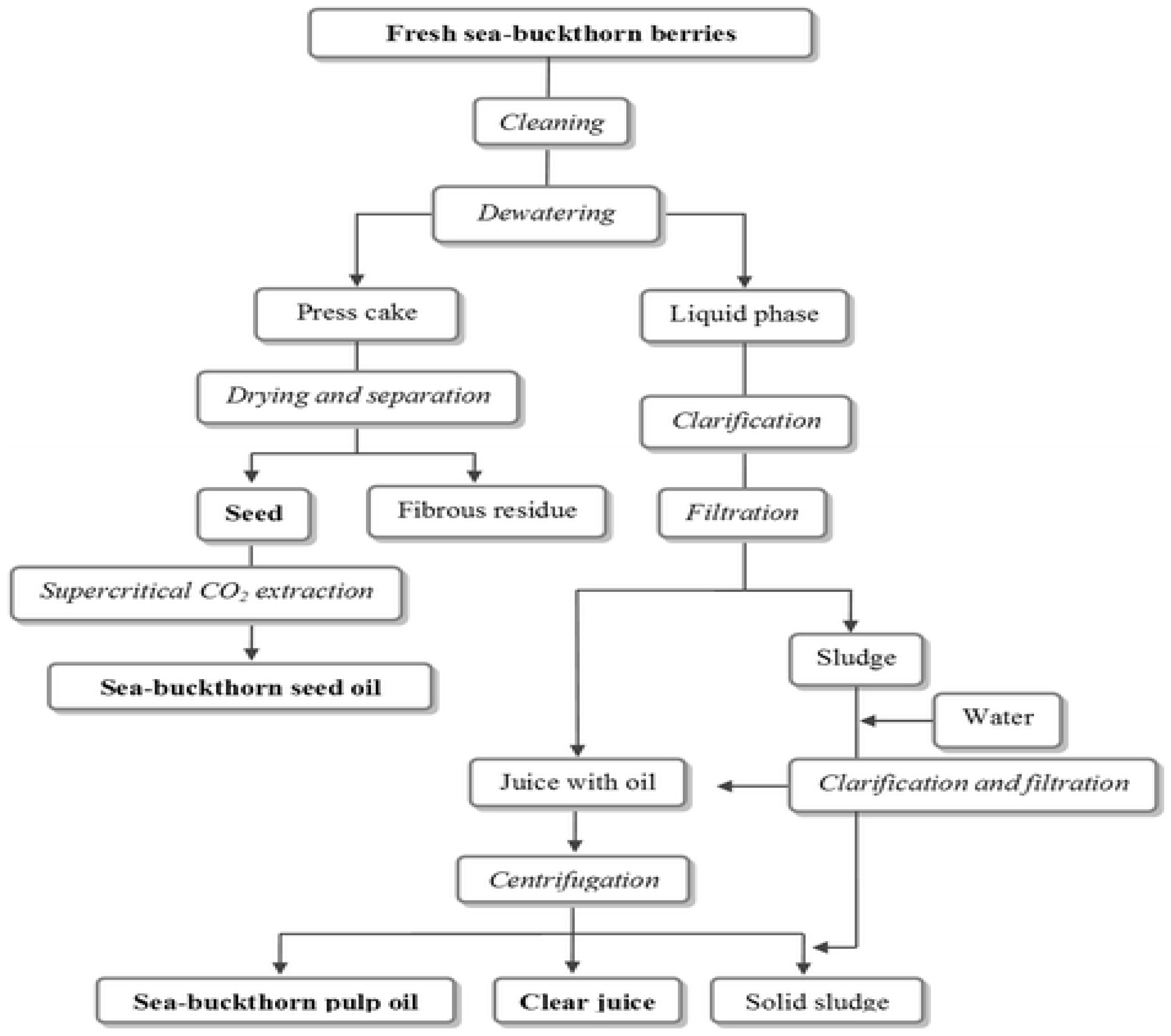
1.2. Methods for Obtaining the SB Oil
2. Sea Buckthorn Berries-Chemical Composition
3. Lipophilic Profile of SB Oil
3.1. Unsaturated Fatty Acids
3.2. Saturated Fatty Acids
3.3. Complex Lipids
3.3.1. Phospholipids and Glycolipids
3.3.2. Sterols
4. Internal Application of Sea Buckthorn Benefits for Human Health
5. External Application of Sea Buckthorn Benefits for Human Health
SB Oil in Cosmetics
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, R.; Zong, S.X.; Yu, L.F.; Lu, P.F.; Luo, Y.Q. Rhythms of volatile release from female and male sea buckthorn plants and electrophysiological response of sea buckthorn carpenter moths. J. Plant Interact. 2014, 9, 763–774. [Google Scholar] [CrossRef]
- Fu, L.; Su, H.; Li, R.; Cui, Y. Harvesting technologies for sea buckthorn fruit. Eng. Agric. Environ. Food 2014, 7, 64–69. [Google Scholar] [CrossRef]
- Li, T.S.C. Product development of sea buckthorn. In Trends in New Crops and New Uses; Janick, J., Whipkey, A., Eds.; ASHS Press: Alexandria, VA, USA, 2002; pp. 393–398. [Google Scholar]
- Bartish, I.V.; Jeppsson, N.; Nybom, H.; Swenson, U. Phylogeny of hippophae (elaeagnaceae) inferred from parsimony analysis of chloroplast DNA and morphology. Syst. Bot. 2002, 27, 41–54. [Google Scholar]
- Zielińska, A.; Nowak, I. Fatty acids in vegetable oils and their importance in cosmetic industry. Chem. Aust. 2014, 68, 103–110. [Google Scholar]
- Isayev, J.I.; Karimov, Y.B.; Kazimov, H.A. New technology of sea-buckthorn oil extraction. Azerbaijan Med. J. 2005, 2, 7–9. [Google Scholar]
- United States Department of Agriculture. PLANTS Profile for Hippophae rhamnoides L. (seaberry); United States Department of Agriculture: Washington, DC, USA, 2007.
- Zielińska, A.; Nowak, I. Abundance of active ingredients in sea-buckthorn oil. Lipids Health Dis. 2017, 16, 95. [Google Scholar] [CrossRef] [PubMed]
- Korekar, G.; Dolkar, P.; Singh, H.; Srivastava, R.B.; Stobdan, T. Genotypic and morphometric effect on fruit oil content in seventeen natural population of Seabuckthorn (Hippophae rhamnoides L.) from trans-Himalaya. Natl. Acad. Sci. Lett. 2013, 36, 603–607. [Google Scholar] [CrossRef]
- Arumughan, C.; Venugopalan, V.V.; Ranjith, A.; Sarinkumar, K.; Mangalagowri, P.; Sawhney, R.C.; Attrey, D.P.; Banerjee, P.K.; Chaurasia, O.P. A novel Green Approach to the Integrated Processing of Sea Buckthorn Berries for Therapeutic and Nutraceutical Values. Indian Patent 648/DEL, 2 June 2006. [Google Scholar]
- Arif, S.; Ahmed, S.D.; Shah, A.H.; Hassan, L.; Awan, S.I.; Hamid, A.; Batool, F.L. Determination of optimum harvesting time for Vitamin C, oil and mineral elements in berries sea buckthorn (Hippophae rhamnoides). Pak. J. Bot. 2010, 42, 3561–3568. [Google Scholar]
- Cenkowski, S.; Yakimishen, R.; Przybylski, R.; Muir, W.E. Quality of extracted sea buckthorn seed and pulp oil. Can. Biosyst. Eng. 2006, 48, 309–316. [Google Scholar]
- Kallio, H.; Yang, B.; Peippo, P.; Tahvonen, R.; Pan, R. Triacylglycerols, glycerophospholipids, tocopherols, tocotrienols in berries and seeds of two sspecies (ssp.sinensis and mongolica) of sea buckthorn (Hippophaë rhamnoides). J. Agric. Food Chem. 2002, 50, 3004–3009. [Google Scholar] [CrossRef] [PubMed]
- Yakimishen, R.; Cenkowski, S.; Muir, W.E. Oil recoveries from sea buckthorn seeds and pulp. Appl. Eng. Agric. 2005, 21, 1047–1055. [Google Scholar] [CrossRef]
- Chen, L.; Xin, X.; Yuan, Q.; Su, D.; Liu, W. Phytochemical properties and antioxidant capacities of various colored berries. J. Agric. Food Chem. 2014, 94, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Karlsson, R.M.; Oksman, P.H.; Kallio, H.P. Phytosterols in sea buckthorn (Hippophaë rhamnoides L.) berries: Identification and effects of different origins and harvesting times. J. Agric. Food Chem. 2001, 49, 5620–5629. [Google Scholar] [CrossRef] [PubMed]
- Kallio, H.P.; Yang, B. Health effects of sea buckthorn berries; research and strategies at the university of Turku, Finland. Acta Hortic. 2014, 1017, 343–349. [Google Scholar] [CrossRef]
- Górnaś, P.; Šne, E.; Siger, A.; Segliņa, D. Sea buckthorn (Hippophae rhamnoides L.) leaves as valuable source of lipophilic antioxidants: The effect of harvest time, sex, drying and extraction methods. Ind. Crops Prod. 2014, 60, 1–7. [Google Scholar] [CrossRef]
- Kallio, H.; Yang, B.; Peippo, P. Effects of different origins and harvesting time on vitamin C, tocopherols, and tocotrienols in sea buckthorn (Hippophaë rhamnoides) berries. J. Agric. Food Chem. 2002, 50, 6136–6142. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Kallio, H. Effects of harvesting time on triacylglycerols and glycerophospholipids of sea buckthorn (Hippophaë rhamnoides L.) berries of different origins. J. Food. Compos. Anal. 2002, 15, 143–157. [Google Scholar] [CrossRef]
- Seglina, D.; Karklina, D.; Ruisa, S.; Krasnova, I. The effect of processing on the composition of sea buckthorn juice. J. Fruit. Ornam. Plant Res. 2006, 14, 257–263. [Google Scholar]
- Rösch, D.; Bergmann, M.; Knorr, D.; Kroh, L.W. Structure−antioxidant efficiency relationships of phenolic compounds and their contribution to the antioxidant activity of sea buckthorn juice. J. Agric. Food Chem. 2003, 51, 4233–4239. [Google Scholar] [CrossRef] [PubMed]
- Zeb, A. Chemical and nutritional constituents of sea buckthorn juice. Pak. J. Nutr. 2004, 3, 99–106. [Google Scholar]
- Selvamuthukumaran, M.; Khanum, F. Development of spiced seabuckthorn [Elaeagnus rhamnoides (L.) a. Nelson syn. Hippophae rhamnoides L.] mixed fruit squash. Indian J. Tradit. Knowl. 2014, 13, 132–141. [Google Scholar]
- Pop, R.M.; Weesepoel, Y.; Socaciu, C.; Vincken, J.P.; Gruppen, H.; et al. Carotenoid composition of berries and leaves from six Romanian sea buckthorn (Hippophae rhamnoides L.) varieties. Food Chem. 2014, 147, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Stobdan, T.; Korekar, G.; Srivastava, R.B. Nutritional attributes and health application of seabuckthorn (Hippophae rhamnoides L.)—A review. Curr. Nutr. Food Sci. 2013, 9, 151–165. [Google Scholar] [CrossRef]
- Zadernowski, R.; Naczk, M.; Czaplicki, S.; Rubinskiene, M.; Szałkiewicz, M. Composition of phenolic acids in sea buckthorn (Hippophae rhamnoides L.) berries. J. Am. Oil Chem. Soc. 2005, 82, 175–179. [Google Scholar] [CrossRef]
- Cupara, S.M.; Sobajic, S.S.; Tadic, V.M.; Arsic, I.A.; Djordjevic, S.M.; Runjajic-Antic, D.; et al. Dry sea buckthorn berries (Hippophae rhamnoides L.)-fatty acid and carotene content in pericarp and seed oil. HealthMed 2010, 4, 789–793. [Google Scholar]
- Yang, B.; Kallio, H. Composition and physiological effects of sea buckthorn lipids. Trends Food Sci. Technol. 2002, 13, 160–167. [Google Scholar] [CrossRef]
- Yang, B.; Kallio, H.P. Fatty acid composition of lipids in sea buckthorn (Hippophaë rhamnoides L.) berries of different origins. J. Agric. Food Chem. 2011, 49, 1939–1947. [Google Scholar] [CrossRef]
- Kim, K.B.; Nam, Y.A.; Kim, H.S.; Hayes, A.W.; Lee, B.M. α-Linolenic acid: Nutraceutical, pharmacological and toxicological evaluation. Food Chem. Toxicol. 2014, 70, 163–178. [Google Scholar] [CrossRef] [PubMed]
- Geetha, R.M.; Sai, V.; Singh, G.; Ilavazhagan, M.; Sawhney, R.C. Anti-oxidant and immunomodulatory properties of seabuckthorn (Hippophae rhamnoides)—An in vitro study. J. Ethnopharmacol. 2002, 79, 373–378. [Google Scholar] [CrossRef]
- Yang, B.; Kalimo, K.; Mattila, L.; Kallio, S.; Katajisto, J.; Peltola, O.J.; Kallio, H.P. Effects of dietary supplementation with sea buckthorn seed and pulp oils on atopic dermatitis. J. Nutr. Biochem. 1999, 10, 622–630. [Google Scholar] [CrossRef]
- Rustan, A.; Drevon, C. Fatty acids: Structures and properties. Encycl. Life Sci. 2005. [Google Scholar] [CrossRef]
- Proksch, E.; Brandner, J.M.; Jensen, J.M. The skin: An indispensable barrier. Exp. Dermatol. 2008, 17, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Krejcarová, J.; Straková, E.; Suchý, P.; Herzig, I.; Karásková, K. Sea buckthorn (Hippophae rhamnoides L.) as a potential source of nutraceutics and its therapeutic possibilities—A review. Acta Vet. Brno 2015, 84, 257–268. [Google Scholar] [CrossRef]
- Edraki, M.; Akbarzadeh, A.; Hosseinzadeh, M.; Salehi, A.; Koohi-Hosseinabadi, O. Healing effect of sea buckthorn, olive oil, and their mixture on full-thickness burn wounds. Adv. Skin Wound Care 2014, 27, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Kallio, H.; Koponen, J.; Tahvonen, R. Free and esterified sterols in seed oil and pulp/peel oil of sea buckthorn (Hippophae rhamnoides L.). In Biologicaly-Active Phytochemicals; Pfannhauser, W., Fenwick, R., Khokhar, S., Eds.; The Royal Chemistry Society: Cambridge, UK, 2001; pp. 24–27. [Google Scholar]
- Li, T.S.; Beveridge, T.; Drover, J. Phytosterol content of sea buckthorn (Hippophae rhamnoides L.) seed oil: Extraction and identification. Food Chem. 2007, 101, 1633–1639. [Google Scholar] [CrossRef]
- Zhou, Y.; Jiang, J.; Song, Y.; Sun, S. Research on the anti-gastric ulcer effect of sea buckthorn seed oil. Hippophae 1994, 7, 33–36. [Google Scholar]
- Wu, A.R.; Su, Y.C.; Li, J.F.; Liu, Q.L.; Lu, J.X.; Che, X.P.; Qian, C.M. Observation on the clinical effect of sea buckthorn oil suppository on chronic cervicitis. Hippophae 1992, 5, 22–25. [Google Scholar]
- Wang, X.Q.; Hu, Q.H.; Liu, Y.Z.; Zhao, C.; Wu, R.F.; Cui, X.H.; Liu, J.M.; Feng, X.J. Studies on effects of sea buckthorn on humoral immune function of experimental animals. Ningxia Med. J. 1989, 11, 281–282. [Google Scholar]
- Larmo, P.S.; Yang, B.; Hurme, S.A.; Alin, J.A.; Kallio, H.P.; Salminen, E.K.; Tahvonen, R.L. Effect of a low dose of sea buckthorn berries on circulating concentrations of cholesterol, triacylglycerols, and flavonols in healthy adults. Eur. J. Nutr. 2009, 48, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Basu, M.; Prasad, R.; Jayamurthy, P.; Pal, K.; Arumughan, C.; Sawhney, R.C. Anti-atherogenic effects of seabuckthorn (Hippophaea rhamnoides) seed oil. Phytomedicine 2007, 14, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Kumar, G.P.; Chaurasia, O.P.; Singh, S.B. Phytochemical and pharmacological profile of seabuckthorn oil: A review. Res. J. Med. Pl. 2011, 5, 491–499. [Google Scholar] [CrossRef]
- Shikov, A.; Pozharitskaya, O.; Makarov, V.; Wagner, H.; Verpoorte, R.; Heinrich, M. Medicinal plants of the Russian pharmacopoeia; their history and applications. J. Ethnopharmacol. 2014, 154, 481–536. [Google Scholar] [CrossRef] [PubMed]
- Uauy, R.; Dangour, A.D. Nutrition in brain development and aging: Role of essential fatty acids. Nutr. Rev. 2006, 64, 24–33. [Google Scholar] [CrossRef]
- Gao, X.; Ohlander, M.; Jeppsson, N.; Björk, L.; Trajkovski, V. Changes in antioxidant effects and their relationship to phytonutrients in fruits of sea buckthorn (Hippophae rhamnoides L.) during maturation. J. Agric. Food Chem. 2000, 48, 1485–1490. [Google Scholar] [CrossRef] [PubMed]
- Ito, H.; Asmussen, S.; Traber, D.L.; Cox, R.A.; Hawkins, H.K.; Connelly, R.; Traberal, L.D.; Walker, T.W.; Malgerud, E.; Sakurai, H.; et al. Healing efficacy of sea buckthorn (Hippophae rhamnoides L.) seed oil in an ovine burn wound model. Burns 2014, 40, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Cupara, S.; Arsic, I.; Homsek, I.; Tadic, V.; Jankovic, S.; Djordjevic, S. Moisturizing effect of o/w oleosom structure creams containing seabuckthorn fatty oil and olive oil. In Proceedings of the 6th World Meeting on Pharmaceutics, Biopharmaceutics and Phramaceutical Technology, Barcelona, Spain, 7–10 April 2008; p. 35. [Google Scholar]
- Cupara, S.M.; Ninkovic, M.B.; Knezevic, M.G.; Vuckovic, I.M.; Jankovic, S.M. Wound healing potential of liquid crystal structure emulsion with sea buckthorn oil. HealthMed 2011, 5, 1218–1223. [Google Scholar]
- Yang, B.; Kalimo, K.; Tahvonen, R.; Mattila, L.; Katajisto, J.; Kallio, H. Effects of dietary supplementation with sea buckthorn (Hippophaë rhamnoides) seed and pulp oils on the fatty acid composition of skin glycerophospholipids of patients with atopic dermatitis. J. Nutr. Biochem. 2000, 11, 338–340. [Google Scholar] [CrossRef]
- Lee, S.; Gura, K.M.; Kim, S.; Arsenault, D.A.; Bistrian, B.R.; Puder, M. Current clinical applications of Ω-6 and Ω-3 fatty acids. Nutr. Clin. Pract. 2006, 21, 323–341. [Google Scholar] [CrossRef] [PubMed]
- Yen, C.H.; Dai, Y.S.; Yang, Y.H.; Lee, J.H.; Chiang, B.L. Linoleic acid metabolite levels and transepidermal water loss in children with atopic dermatitis. Ann. Allergy Asthma Immunol. 2008, 100, 66–73. [Google Scholar] [CrossRef]
- Cupara, S.; Arsic, I.; Homsek, I.; Tadic, V.; Jevtovic, I.; Petrovic, M. In vivo case study: Investigation of o/w cream containing sea buckthorn oil on skin moisture. In Proceedings of the 7th central European Symposium on Pharmaceutical Technology and Biotechnology, Ljubljana, Slovenia, 18–20 September 2008. [Google Scholar]
- Hwang, I.S.; Kim, J.E.; Choi, S.I.; Lee, H.R.; Lee, Y.J.; Jang, M.J. UV radiation-induced skin aging in hairless mice is effectively prevented by oral intake of sea buckthorn (Hippophae rhamnoides L.) fruit blend for 6 weeks through MMP suppression and increase of SOD activity. Int. J. Mol. Med. 2012, 30, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Bath-Hextall, F.J.; Jenkinson, C.; Humphreys, R.; Williams, H.C. Dietary supplements for established atopic eczema. Cochrane Database Syst. Rev. 2012, 2, CD005205. [Google Scholar]
- Korać, R.R.; Khambholja, K.M. Potential of herbs in skin protection from ultraviolet radiation. Pharmacogn. Rev. 2011, 5, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Beveridge, T.; Li, T.S.; Oomah, B.D.; Smith, A. Sea buckthorn products: Manufacture and composition. J. Agric. Food Chem. 1999, 47, 3480–3488. [Google Scholar] [CrossRef] [PubMed]
| Common Name | Systematic Name | Content in wt % | General Formula | Numerical Symbol | Omega Family |
|---|---|---|---|---|---|
| saturated fatty acids | |||||
| palmitic acid | hexadecanoic acid | 30–33 | CH3(CH2)14COOH | C16:0 | - |
| stearic acid | octadecanoic acid | <1 | CH3(CH2)16COOH | C18:0 | - |
| unsaturated fatty acids | |||||
| palmitoleic acid | (Z)-9-hexadecenoic acid | 30–35 | C16H30O2 | C16:1 | 7 |
| oleic acid | (Z)-9-octadecenoic acid | 14–18 | C18H34O2 | C18:1 | 9 |
| linoleic acid (LA) | (Z,Z)-9,12-octadecadienoic acid | 5–7 | C18H32O2 | C18:2 | 6 |
| α-linolenic acid (ALA) | (Z,Z,Z)-9,12,15- octadecatrienoic acid | 30 | C18H30O2 | C18:3 | 3 |
| γ-linolenic acid (GLA) | (Z,Z,Z)-6,9,12- octadecatrienoic acid | 35 | C18H30O2 | C18:3 | 6 |
| gondoic acid | (Z)-11-eicosenoic acid | 2 | C20H38O2 | C20:1 | 9 |
| Oil Components | Extraction Method | ||
|---|---|---|---|
| Screw Press | Aqueous | ||
| Seed oil | fatty acids | similar concentrations for most fatty acids | |
| tocopherols | low | n/a * | |
| carotenoids | low | n/a | |
| sterols | high | n/a | |
| Pulp oil | fatty acids | similar concentrations for most fatty acids | |
| tocopherols | n/a | high | |
| carotenoids | n/a | high | |
| sterols | n/a | low | |
| Micronutrients | Macronutrients | ||||||
|---|---|---|---|---|---|---|---|
| potassium | magnesium | calcium | iron | zinc | manganese | copper | nickel |
| 168–219 | 8.3–9.5 | 5–7.2 | 1.24 | 0.25 | 0.25 | 0.006 | 0.015 |
| affects muscle spasms | with calcium is responsible for the proper functioning of the nervous system | for the proper functioning of the muscular system | component of hemoglobin, myoglobin and coenzymes many enzymes involved, among others, in the formation of ATP | participates in various stages of protein biosynthesis, ingredient of insulin (also plays an important role in the storage of the pancreas), regulates the concentration of vitamin A is used in the formation of bone, stimulates growth and tissue repair (wound healing) | necessary for proper development of tissue (especially bone) and for the functioning of the central nervous system | cofactor of many enzymes | component of urease - an enzyme decomposing urea into ammonia and carbon dioxide |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koskovac, M.; Cupara, S.; Kipic, M.; Barjaktarevic, A.; Milovanovic, O.; Kojicic, K.; Markovic, M. Sea Buckthorn Oil—A Valuable Source for Cosmeceuticals. Cosmetics 2017, 4, 40. https://doi.org/10.3390/cosmetics4040040
Koskovac M, Cupara S, Kipic M, Barjaktarevic A, Milovanovic O, Kojicic K, Markovic M. Sea Buckthorn Oil—A Valuable Source for Cosmeceuticals. Cosmetics. 2017; 4(4):40. https://doi.org/10.3390/cosmetics4040040
Chicago/Turabian StyleKoskovac, Marijana, Snezana Cupara, Mihailo Kipic, Ana Barjaktarevic, Olivera Milovanovic, Ksenija Kojicic, and Marija Markovic. 2017. "Sea Buckthorn Oil—A Valuable Source for Cosmeceuticals" Cosmetics 4, no. 4: 40. https://doi.org/10.3390/cosmetics4040040
APA StyleKoskovac, M., Cupara, S., Kipic, M., Barjaktarevic, A., Milovanovic, O., Kojicic, K., & Markovic, M. (2017). Sea Buckthorn Oil—A Valuable Source for Cosmeceuticals. Cosmetics, 4(4), 40. https://doi.org/10.3390/cosmetics4040040




