A Recent Update on the Potential Use of Catechins in Cosmeceuticals
Abstract
1. Introduction
2. Physicochemical Properties of Catechins
3. Activities of Catechin
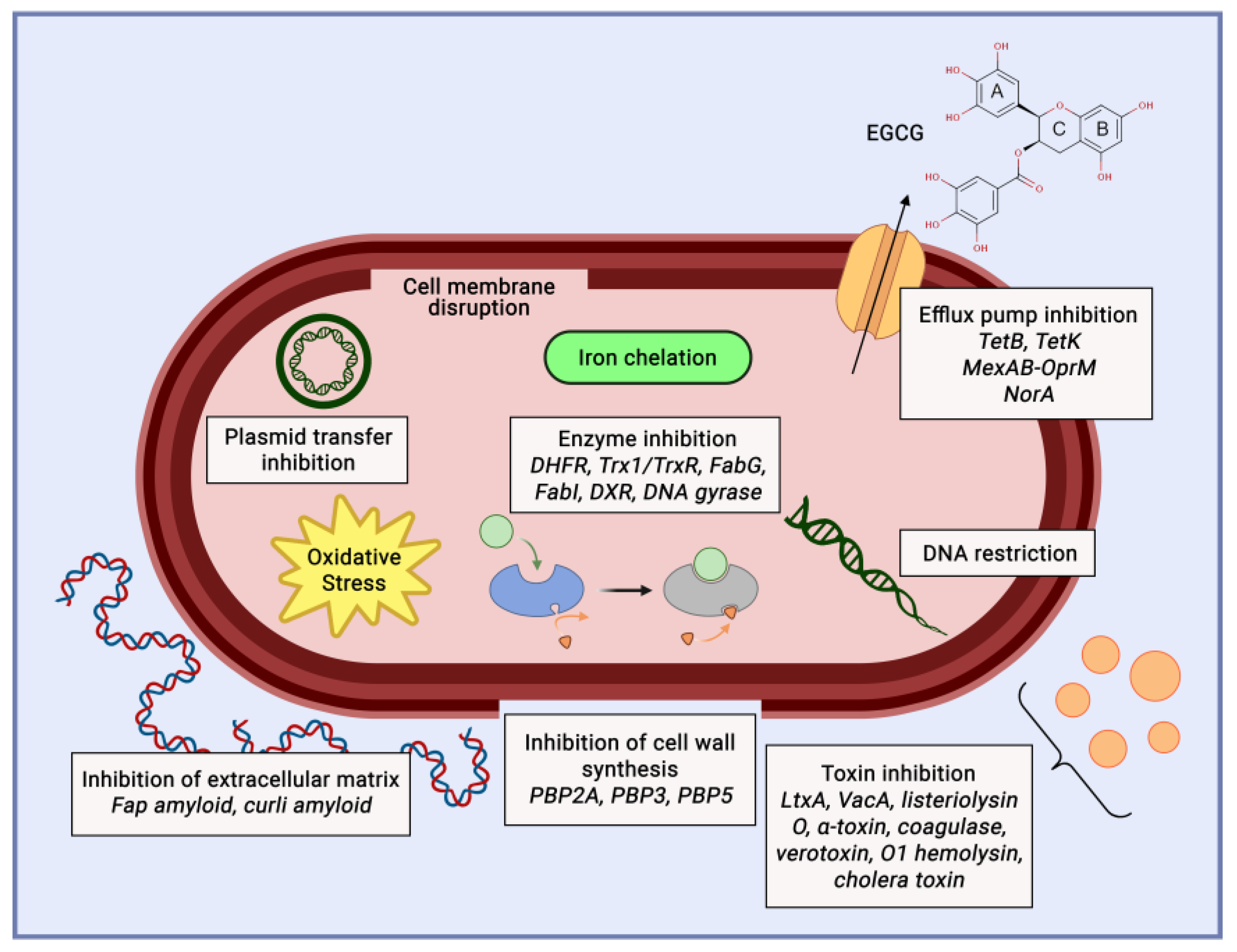
3.1. Antioxidant
3.2. Anti-Ageing
3.3. Skin Brightener
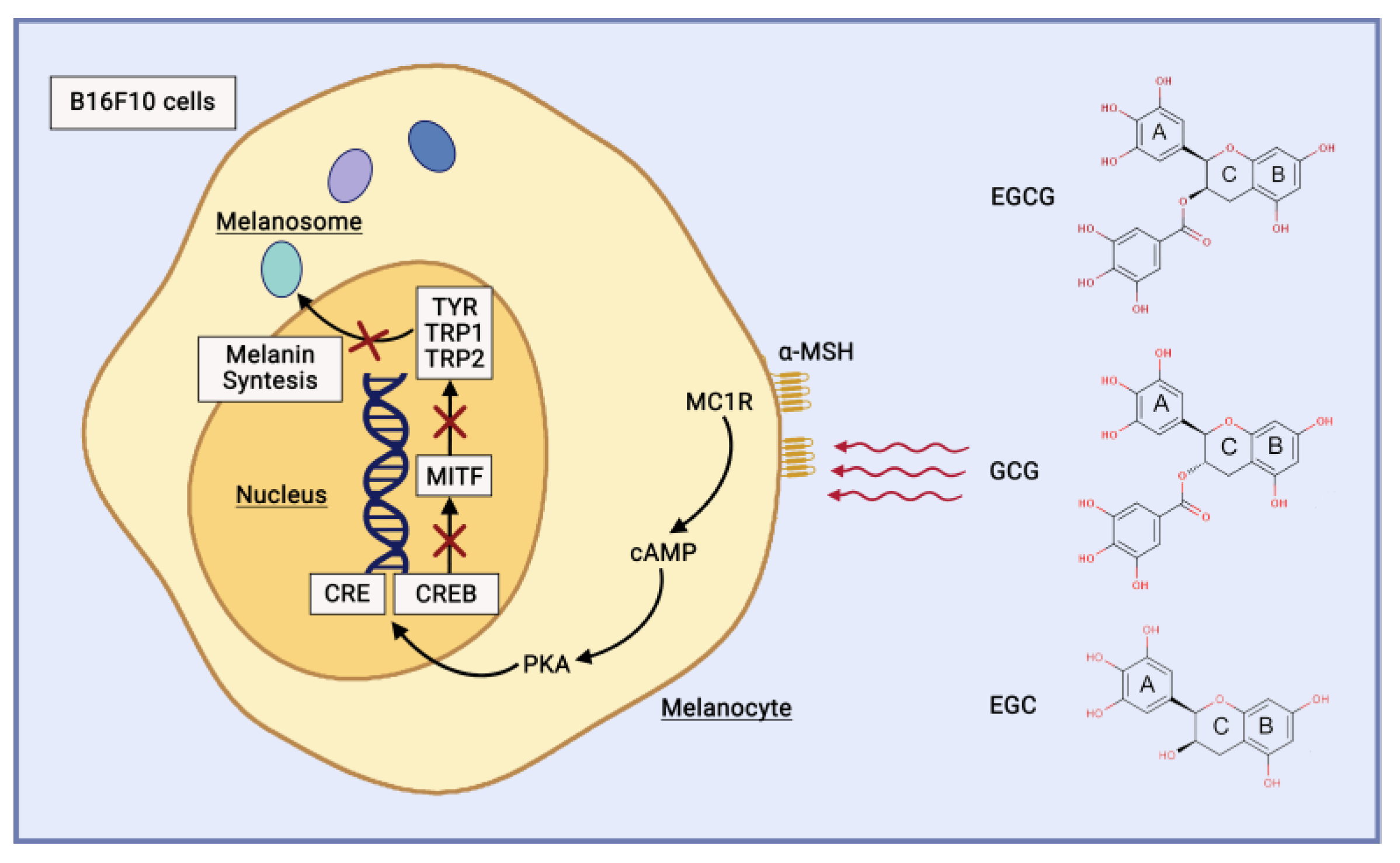
3.4. Anti-Hyperpigmentation
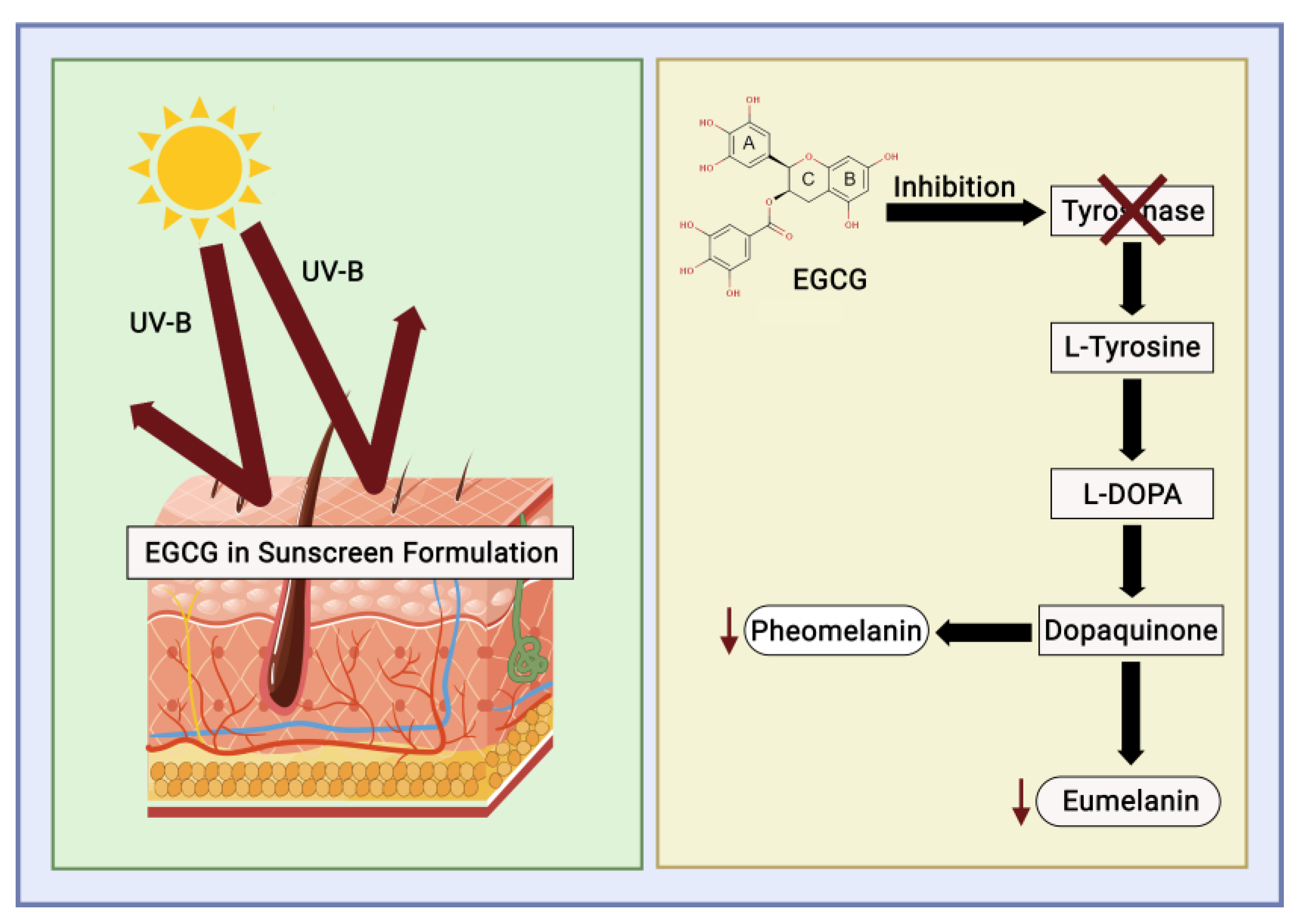
3.5. UV-Reduction and Sunscreen
3.6. Anti-Acne
4. Application and Development of Catechin Formulations in Cosmetic Preparations
5. Pre-Clinical Dan Clinical Trials of Catechins in Cosmetics and Skincare
| Study Title | Catechin | Result/Conclusion | Ref. |
|---|---|---|---|
| Double-blinded, placebo-controlled trial of green tea extracts in the clinical and histologic appearance of photoageing skin | Green Tea, EGCG | Skin elasticity No significant differences, although histologic grading showed improvement in elastic tissue Experiment: supplement of green tea and addition of 10% green tea cream in an 8-week trial | [75] |
| The green tea polyphenol (-) epigallocatechin gallate and green tea can protect human cellular DNA from UV and visible radiation-induced damage | Green Tea, EGCG | Photoprotective UV radiation inhibition, prevention, and minimal DNA cell damage Experiment: study of 540 mL of green tea in 10 subjects | [76] |
| A randomised controlled trial of green tea beverages on the in vivo radical scavenging activity in human skin | Green Tea, EGCG | Antioxidant Increasing radical scavenging of skin by 28–29% compared to the control group Experiment: 3 cups of tea (Benifuuki tea and Yabukita tea) for 3 weeks | [77] |
| UV radiation-induced degradation of the dermal extracellular matrix and protection by green tea catechins | Green Tea, EGCG | Photoprotective Specific UVR protection and significant changes in acute UVR Experiment: 50 subjects were randomised to green tea catechin and vitamin C for 12 weeks with twice-daily consumption | [78] |
| Treatment of atopic dermatitis associated with Malassezia sympodialis by green tea extracts bath therapy: a pilot study | Green Tea, EGCG | Atopic dermatitis Significant improvement in 1 of 3 patients with a total reduction of 50.3% Experiment: 3 subjects bathed in a combination of green tea extract and tap water for 1 month | [79] |
| A randomised controlled trial of green tea catechins in protection against UV radiation-induced cutaneous inflammation | Green Tea, EGCG | Photoprotective No significant difference between the test and placebo groups Experiment: 50 subjects were randomly placed in 2 groups; group 1 was given encapsulated green tea extract with the addition of vitamins and group 2 was given a placebo twice daily for 3 months | [73] |
| Formulation of Gambir (Uncaria gambir Roxb.) ethanol extract as acne powder | Gambir Extract, Catechin | Anti-acne Inhibition diameters of 3%, 6%, and 9% produced a diameter of 3.6 mm, 4.2 mm, and 6.8 mm respectively Experiment: catechin from an ethanol extract of gambir (3%, 6%, and 9%) tested on Staphylococcus epidermidis | [60] |
| The use of green tea extract in cosmetic formulation: not only antioxidant active ingredient | Green Tea Leaf Extract | Moisturiser Significant increase in skin moisture and improved skin texture Experiment: cosmetic formulation with 6% Camellia sinensis extract with 24 volunteers | [80] |
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Bellad, K.A.; Nanjwade, B.K.; Kamble, M.S.; Srichana, T.; Idris, N.F. Development of cosmeceuticals. World J. Pharm. Pharm. Sci. 2017, 6, 643–691. [Google Scholar]
- Yamada, M.; Mohammed, Y.; Prow, T.W. Advances and controversies in studying sunscreen delivery and toxicity. Adv. Drug Deliv. Rev. 2020, 153, 72–86. [Google Scholar] [CrossRef] [PubMed]
- Cavinato, M.; Jansen-Durr, P. Molecular mechanisms of UVB-induced senescence of dermal fibroblasts and its relevance for photoaging of the human skin. Exp. Gerontol. 2017, 94, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Rinnerthaler, M.; Bischof, J.; Streubel, M.K.; Trost, A.; Richter, K. Oxidative stress in aging human skin. Biomolecules 2015, 5, 545–589. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.H.; Chien, C.Y.; Chen, K.H.; Huang, S.C.; Chien, C.T. Catechins Blunt the Effects of oxLDL and its Primary Metabolite Phosphatidylcholine Hydroperoxide on Endothelial Dysfunction Through Inhibition of Oxidative Stress and Restoration of eNOS in Rats. Kidney Blood Press. Res. 2017, 42, 919–932. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Yu, J.S.; Phung, H.M.; Lee, J.G.; Kim, K.H.; Kang, K.S. Potential Anti-Skin Aging Effect of (-)-Catechin Isolated from the Root Bark of Ulmus davidiana var. japonica in Tumor Necrosis Factor-alpha-Stimulated Normal Human Dermal Fibroblasts. Antioxidants 2020, 9, 981. [Google Scholar] [CrossRef]
- Braicu, C.; Ladomery, M.R.; Chedea, V.S.; Irimie, A.; Berindan-Neagoe, I. The relationship between the structure and biological actions of green tea catechins. Food Chem. 2013, 141, 3282–3289. [Google Scholar] [CrossRef] [PubMed]
- Bernatoniene, J.; Kopustinskiene, D.M. The Role of Catechins in Cellular Responses to Oxidative Stress. Molecules 2018, 23, 965. [Google Scholar] [CrossRef]
- Matsubara, T.; Wataoka, I.; Urakawa, H.; Yasunaga, H. Effect of reaction pH and CuSO4 addition on the formation of catechinone due to oxidation of (+)-catechin. Int. J. Cosmet. Sci. 2013, 35, 362–367. [Google Scholar] [CrossRef]
- Ferreira-Nunes, R.; da Silva, S.M.M.; de Souza, P.E.N.; de Oliveira Magalhães, P.; Cunha-Filho, M.; Gratieri, T.; Gelfuso, G.M. Incorporation of Eugenia dysenterica extract in microemulsions preserves stability, antioxidant effect and provides enhanced cutaneous permeation. J. Mol. Liq. 2018, 265, 408–415. [Google Scholar] [CrossRef]
- Mita, S.R.; Abdassah, M.; Supratman, U.; Shiono, Y.; Rahayu, D.; Sopyan, I.; Wilar, G. Nanoparticulate System for the Transdermal Delivery of Catechin as an Antihypercholesterol: In Vitro and In Vivo Evaluations. Pharmaceuticals 2022, 15, 1142. [Google Scholar] [CrossRef] [PubMed]
- Munggari, I.P.; Kurnia, D.; Deawati, Y.; Julaeha, E. Current Research of Phytochemical, Medicinal and Non-Medicinal Uses of Uncaria gambir Roxb.: A Review. Molecules 2022, 27, 6551. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Heo, H.J. The roles of catechins in regulation of systemic inflammation. Food Sci. Biotechnol. 2022, 31, 957–970. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Hwang, K.; Lee, J.; Han, S.Y.; Kim, E.-M.; Park, J.; Cho, J.Y. Skin protective effect of epigallocatechin gallate. Int. J. Mol. Sci. 2018, 19, 173. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.; Kim, N.; Shin, Y.; Kim, S.-Y.; Kim, Y.-J. Activity of catechins and their applications. Biomed. Dermatol. 2020, 4, 1–10. [Google Scholar] [CrossRef]
- Isemura, M. Catechin in Human Health and Disease. Molecules 2019, 24, 528. [Google Scholar] [CrossRef] [PubMed]
- Syukri, D.; Azima, F.; Aprialdho, R. Study on the Utilization of Catechins from Gambir (Uncaria Gambir Roxb) Leaves as Antioxidants Cooking Oil. Andalasian Int. J. Agric. Nat. Sci. 2022, 3, 12–25. [Google Scholar] [CrossRef]
- Zillich, O.; Schweiggert-Weisz, U.; Eisner, P.; Kerscher, M. Polyphenols as active ingredients for cosmetic products. Int. J. Cosmet. Sci. 2015, 37, 455–464. [Google Scholar] [CrossRef]
- Moldoveanu, S.C.; David, V. Essentials in Modern HPLC Separations; Elsevier: Amsterdam, The Netherlands, 2022. [Google Scholar]
- Hariyati, N. Ministry of Health of the Republic of Indonesia Farmakope Herbal Indonesia, 2nd ed.; Ministry of Health Indonesia: Kota Jakarta, Indonesia, 2017. [Google Scholar]
- Cuevas-Valenzuela, J.; González-Rojas, Á.; Wisniak, J.; Apelblat, A.; Pérez-Correa, J.R. Solubility of (+)-catechin in water and water-ethanol mixtures within the temperature range 277.6–331.2 K: Fundamental data to design polyphenol extraction processes. Fluid Phase Equilibria 2014, 382, 279–285. [Google Scholar] [CrossRef]
- Riwu, A.G.; Nugraha, J.; Purwanto, D.A.; Triyono, E.A. Determination of (+)-Catechin and Antioxidant Activity in Faloak (Sterculia quadrifida R. Br) Stem Bark Infusion. Sci. Technol. Indones. 2023, 8, 59–65. [Google Scholar] [CrossRef]
- Delarosa, A.; Hendrawan, R.P.; Halimah, E. Screening of Costus speciosus and Determination of Antioxidant Potential Using DPPH Method: A Review. Eur. J. Med. Plants 2023, 34, 17–28. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, Q.; Zhao, Z.; Bai, B.; Sun, Z.; Cai, L.; Fu, Y.; Ma, Y.; Wang, Q.; Xi, G. Effect of hydroxyl on antioxidant properties of 2, 3-dihydro-3, 5-dihydroxy-6-methyl-4 H-pyran-4-one to scavenge free radicals. RSC Adv. 2021, 11, 34456–34461. [Google Scholar] [CrossRef] [PubMed]
- Hoang, H.T.; Moon, J.-Y.; Lee, Y.-C.J.C. Natural antioxidants from plant extracts in skincare cosmetics: Recent applications, challenges and perspectives. Cosmetics 2021, 8, 106. [Google Scholar] [CrossRef]
- Grzesik, M.; Naparło, K.; Bartosz, G.; Sadowska-Bartosz, I. Antioxidant properties of catechins: Comparison with other antioxidants. Food Chem. 2018, 241, 480–492. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Y.; Sun, Y.; Tang, Y.; Yu, Y.; Wang, J.; Zheng, F.; Li, Y.; Sun, Y. Catechins: Protective mechanism of antioxidant stress in atherosclerosis. Front. Pharmacol. 2023, 14, 1144878. [Google Scholar] [CrossRef] [PubMed]
- Šeruga, M.; Tomac, I. Influence of chemical structure of some flavonols on their electrochemical behaviour. Int. J. Electrochem. Sci. 2017, 12, 7616–7637. [Google Scholar] [CrossRef]
- Hassanpour, S.H.; Doroudi, A. Review of the antioxidant potential of flavonoids as a subgroup of polyphenols and partial substitute for synthetic antioxidants. Avicenna J. Phytomed. 2023, 13, 354. [Google Scholar] [PubMed]
- Platzer, M.; Kiese, S.; Tybussek, T.; Herfellner, T.; Schneider, F.; Schweiggert-Weisz, U.; Eisner, P. Radical scavenging mechanisms of phenolic compounds: A quantitative structure-property relationship (QSPR) study. Front. Nutr. 2022, 9, 882458. [Google Scholar] [CrossRef]
- Anitha, S.; Krishnan, S.; Senthilkumar, K.; Sasirekha, V. Theoretical investigation on the structure and antioxidant activity of (+) catechin and (−) epicatechin–a comparative study. Mol. Phys. 2020, 118, e1745917. [Google Scholar] [CrossRef]
- Zhou, H.-C.; Tam, N.F.-y.; Lin, Y.-M.; Ding, Z.-H.; Chai, W.-M.; Wei, S.-D. Relationships between degree of polymerization and antioxidant activities: A study on proanthocyanidins from the leaves of a medicinal mangrove plant Ceriops tagal. PLoS ONE 2014, 9, e107606. [Google Scholar] [CrossRef]
- Spiegel, M.; Andruniów, T.; Sroka, Z. Flavones’ and Flavonols’ Antiradical Structure–Activity Relationship—A Quantum Chemical Study. Antioxidants 2020, 9, 461. [Google Scholar] [CrossRef] [PubMed]
- Munteanu, I.G.; Apetrei, C. Assessment of the Antioxidant Activity of Catechin in Nutraceuticals: Comparison between a Newly Developed Electrochemical Method and Spectrophotometric Methods. Int. J. Mol. Sci. 2022, 23, 8110. [Google Scholar] [CrossRef] [PubMed]
- Doshi, P.; Adsule, P.; Banerjee, K.; Oulkar, D. Technology. Phenolic compounds, antioxidant activity and insulinotropic effect of extracts prepared from grape (Vitis vinifera L) byproducts. J. Food Sci. Technol. 2015, 52, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Dias, M.C.; Pinto, D.C.; Silva, A.M.S. Plant flavonoids: Chemical characteristics and biological activity. Molecules 2021, 26, 5377. [Google Scholar] [CrossRef] [PubMed]
- Nuryana, I.; Ratnakomala, S.; Fahrurrozi, A.B.J.; Andriani, A.; Putra, F.J.N.; Rezamela, E.; Wulansari, R.; Prawira-Atmaja, M.I.; Lisdiyanti, P. Catechin Contents, Antioxidant and Antibacterial Activities of Different Types of Indonesian Tea (Camellia Sinensis). Ann. Bogor. 2020, 24, 107. [Google Scholar] [CrossRef]
- Anggraini, T.; Wilma, S.; Syukri, D.; Azima, F.J.I.J.o.F.S. Total phenolic, anthocyanin, Catechins, DPPH radical scavenging activity, and toxicity of Lepisanthes alata (Blume) Leenh. Int. J. Food Sci. 2019, 2019, 9703176. [Google Scholar] [CrossRef]
- Musdja, M.Y.; Rahman, H.A.; Hasan, D.J.L.I.J.H.L.-S. Antioxidant activity of catechins isolate of Uncaria gambier Roxb in male rats. LIFE Int. J. Health Life-Sci. 2018, 4, 34–46. [Google Scholar] [CrossRef]
- Rahmi, M.; Rita, R.S.; Yetti, H. Gambir Catechins (Uncaria gambir Roxb) Prevent Oxidative Stress in Wistar Male Rats Fed a High-Fat Diet. Maj. Kedokt. Andalas 2021, 44, 436–441. [Google Scholar]
- Yetuk, G.; Pandir, D.; Bas, H. Protective role of catechin and quercetin in sodium benzoate-induced lipid peroxidation and the antioxidant system in human erythrocytes in vitro. Sci. World J. 2014, 2014, 874824. [Google Scholar] [CrossRef]
- Amber, K.T.; Shiman, M.I.; Badiavas, E.V. The use of antioxidants in radiotherapy-induced skin toxicity. Integr. Cancer Ther. 2014, 13, 38–45. [Google Scholar] [CrossRef]
- Zheng, M.; Liu, Y.; Zhang, G.; Yang, Z.; Xu, W.; Chen, Q. The Applications and Mechanisms of Superoxide Dismutase in Medicine, Food, and Cosmetics. Antioxidants 2023, 12, 1675. [Google Scholar] [CrossRef] [PubMed]
- Boo, Y.C. Arbutin as a Skin Depigmenting Agent with Antimelanogenic and Antioxidant Properties. Antioxidants 2021, 10, 1129. [Google Scholar] [CrossRef] [PubMed]
- Jesus, A.; Mota, S.; Torres, A.; Cruz, M.T.; Sousa, E.; Almeida, I.F.; Cidade, H. Antioxidants in Sunscreens: Which and What For? Antioxidants 2023, 12, 138. [Google Scholar] [CrossRef] [PubMed]
- Nahhas, A.F.; Abdel-Malek, Z.A.; Kohli, I.; Braunberger, T.L.; Lim, H.W.; Hamzavi, I.H. The potential role of antioxidants in mitigating skin hyperpigmentation resulting from ultraviolet and visible light-induced oxidative stress. Photodermatol. Photoimmunol. Photomed. 2019, 35, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Andrade, J.M.; Domínguez-Martín, E.M.; Nicolai, M.; Faustino, C.; Rodrigues, L.M.; Rijo, P. Screening the dermatological potential of plectranthus species components: Antioxidant and inhibitory capacities over elastase, collagenase and tyrosinase. J. Enzyme Inhib. Med. Chem. 2021, 36, 258–270. [Google Scholar] [CrossRef] [PubMed]
- Sonawane, G.B.; Jadhav, S.P.; Patil, C.D.; Kamble, P.R.; Somavanshi, D.B. A Review on the Antioxidant and Antiaging Properties of White Tea. J. Pharm. Res. Int. 2021, 33, 129–136. [Google Scholar]
- Wang, W.; Di, T.; Wang, W.; Jiang, H. EGCG, GCG, TFDG, or TSA inhibiting melanin synthesis by downregulating MC1R expression. Int. J. Mol. Sci. 2023, 24, 11017. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, J.; Li, Y.; Liu, Z.; Lin, Y.; Huang, J.-A. Anti-melanogenic effects of epigallocatechin-3-gallate (EGCG), epicatechin-3-gallate (ECG) and gallocatechin-3-gallate (GCG) via down-regulation of cAMP/CREB/MITF signaling pathway in B16F10 melanoma cells. Fitoterapia 2020, 145, 104634. [Google Scholar] [CrossRef]
- Jiang, T.; Qi, Y.; Wu, Y.; Zhang, J. Application of antioxidant and ultraviolet absorber into HDPE: Enhanced resistance to UV irradiation. e-Polymers 2019, 19, 499–510. [Google Scholar] [CrossRef]
- Laksmiani, N.P.L.; Sanjaya, I.K.N.; Leliqia, N.P.E. The activity of avocado (Persea americana Mill.) seed extract containing catechin as a skin lightening agent. J. Pharm. Pharmacogn. Res. 2020, 8, 449–456. [Google Scholar]
- Vale, E.P.; dos Santos Morais, E.; de Souza Tavares, W.; de Sousa, F.F.O. Epigallocatechin-3-gallate loaded-zein nanoparticles: Characterization, stability and associated antioxidant, anti-tyrosinase and sun protection properties. J. Mol. Liq. 2022, 358, 119107. [Google Scholar] [CrossRef]
- Donglikar, M.M.; Deore, S.L. Sunscreens: A review. Pharmacogn. J. 2016, 8, 171–179. [Google Scholar] [CrossRef]
- Sander, M.; Sander, M.; Burbidge, T.; Beecker, J. The efficacy and safety of sunscreen use for the prevention of skin cancer. CMAJ 2020, 192, E1802–E1808. [Google Scholar] [CrossRef] [PubMed]
- Dewi, D.A.R. Sunscreen Protection Against Visible Light: Is It Needed? Malahayati Nurs. J. 2022, 4, 2527–2536. [Google Scholar] [CrossRef]
- Latha, M.; Martis, J.; Shobha, V.; Shinde, R.S.; Bangera, S.; Krishnankutty, B.; Bellary, S.; Varughese, S.; Rao, P.; Kumar, B.N.; et al. Sunscreening agents: A review. J. Clin. Aesthet. Dermatol. 2013, 6, 16. [Google Scholar] [PubMed]
- Portilho, L.; Aiello, L.M.; Vasques, L.I.; Bagatin, E.; Leonardi, G.R. Effectiveness of sunscreens and factors influencing sun protection: A review. Braz. J. Pharm. Sci. 2023, 58, e20693. [Google Scholar] [CrossRef]
- Ebrahimzadeh, M.A.; Enayatifard, R.; Khalili, M.; Ghaffarloo, M.; Saeedi, M.; Charati, J.Y. Correlation between sun protection factor and antioxidant activity, phenol and flavonoid contents of some medicinal plants. Iran J. Pharm. Res. 2014, 13, 1041. [Google Scholar]
- Warnida, H.; Masliyana, A.; Sapri, S.J. Formulasi Ekstrak Etanol Gambir (Uncaria gambir Roxb.) dalam Bedak Anti Jerawat. J. Ilm. Manuntung 2016, 2, 99–106. [Google Scholar] [CrossRef]
- Messire, G.; Serreau, R.; Berteina-Raboin, S. Antioxidant Effects of Catechins (EGCG), Andrographolide, and Curcuminoids Compounds for Skin Protection, Cosmetics, and Dermatological Uses: An Update. Antioxidants 2023, 12, 1317. [Google Scholar] [CrossRef]
- Liu, B.; Li, A.; Xu, J.; Cui, Y. Single-Cell Transcriptional Analysis Deciphers the Inflammatory Response of Skin-Resident Stromal Cells. Front. Surg. 2022, 9, 935107. [Google Scholar] [CrossRef]
- Shanbhag, S.; Nayak, A.; Narayan, R.; Nayak, U.Y. Anti-aging and sunscreens: Paradigm shift in cosmetics. Adv. Pharm. Bull. 2019, 9, 348. [Google Scholar] [CrossRef]
- Estikomah, S.; Tussifah, H.; Kusumaningtyas, N.; Sholihatin, B.; Dinta, L. Formulation of Solid Soap Combination of Green Tea Leaf (Camellia sinnensis L.) and Corn Kernel (Zea mays) Extracts. In Proceedings of the U-Go Healthy International Conference, U-Go Healthy 2020, Pacitan, Indonesia, 29 March 2020. [Google Scholar]
- Kamal, S.; Rusdi, M.S. Utilization of catechins in sunscreen lotion formulation. Borneo J. Pharm. 2018, 1, 68–71. [Google Scholar] [CrossRef]
- Winarti, C. Physical Characteristics and UV Protection (In Vitro) of Gambier Leaf Extract Lotion. IOP Conf. Ser. Earth Environ. Sci. 2022, 1024, 012061. [Google Scholar] [CrossRef]
- Brito, I.; Ferreira, S.M.; Santos, L. On the Path to Sustainable Cosmetics: Development of a Value-Added Formulation of Solid Shampoo Incorporating Mango Peel Extract. Cosmetics 2023, 10, 140. [Google Scholar] [CrossRef]
- Supiati, S. The Quality of Cream Formulated From Gambier Leaf Extract. IOP Conf. Ser. Earth Environ. Sci. 2022, 1024, 012010. [Google Scholar]
- Rosaini, H.; Makmur, I.; Lestari, E.A.; Sidoretno, W.M.; Yetti, R.D. Formulation of Gel Peel Off Catechins Mask from Gambir (Uncaria gambir (Hunter) Roxb) with the PVP K-30 Concentration Variation. Int. J. Res. Rev. 2021, 8, 205–211. [Google Scholar]
- Zeng, L.; Ma, M.; Li, C.; Luo, L. Stability of tea polyphenols solution with different pH at different temperatures. Int. J. Food Prop. 2017, 20, 1–18. [Google Scholar] [CrossRef]
- Yamamoto, M.; Kirita, M.; Honma, D.; Yokota, T. Novel Methylated Catechin and Composition Containing the Same. U.S. Patent 20100324312A1, 23 December 2010. [Google Scholar]
- Figueroa-Robles, A.; Antunes-Ricardo, M.; Guajardo-Flores, D. Encapsulation of phenolic compounds with liposomal improvement in the cosmetic industry. Int. J. Pharm. 2021, 593, 120125. [Google Scholar] [CrossRef] [PubMed]
- Farrar, M.D.; Nicolaou, A.; Clarke, K.A.; Mason, S.; Massey, K.A.; Dew, T.P.; Watson, R.E.; Williamson, G.; Rhodes, L.E. A randomized controlled trial of green tea catechins in protection against ultraviolet radiation–induced cutaneous inflammation. Am. J. Clin. Nutr. 2015, 102, 608–615. [Google Scholar] [CrossRef]
- Hsieh, W.-C.; Fang, C.-W.; Suhail, M.; Vu, Q.L.; Chuang, C.-H.; Wu, P.-C. Improved skin permeability and whitening effect of catechin-loaded transfersomes through topical delivery. Int. J. Pharm. 2021, 607, 121030. [Google Scholar] [CrossRef]
- Chiu, A.E.; Chan, J.L.; Kern, D.G.; Kohler, S.; Rehmus, W.E.; Kimball, A.B. Double-blinded, placebo-controlled trial of green tea extracts in the clinical and histologic appearance of photoaging skin. Dermatol. Surg. 2005, 31, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Morley, N.; Clifford, T.; Salter, L.; Campbell, S.; Gould, D.; Curnow, A. The green tea polyphenol (−)-epigallocatechin gallate and green tea can protect human cellular DNA from ultraviolet and visible radiation-induced damage. Photodermatol. Photoimmunol. Photomed. 2005, 21, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Megow, I.; Darvin, M.E.; Meinke, M.C.; Lademann, J. A randomized controlled trial of green tea beverages on the in vivo radical scavenging activity in human skin. Skin. Pharmacol. Physiol. 2017, 30, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Charoenchon, N.; Rhodes, L.E.; Nicolaou, A.; Williamson, G.; Watson, R.E.; Farrar, M.D. Ultraviolet radiation-induced degradation of dermal extracellular matrix and protection by green tea catechins: A randomized controlled trial. Clin. Exp. Dermatol. 2022, 47, 1314–1323. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Chang, H.K.; Baek, S.Y.; Chung, J.O.; Rha, C.S.; Kim, S.Y.; Kim, B.J.; Kim, M.N. Treatment of atopic dermatitis associated with Malassezia sympodialis by green tea extracts bath therapy: A pilot study. Mycobiology 2012, 40, 124–128. [Google Scholar] [CrossRef]
- Gianeti, M.D.; Mercurio, D.G.; Maia Campos, P.M. The use of green tea extract in cosmetic formulations: Not only an antioxidant active ingredient. Dermatol. Ther. 2013, 26, 267–271. [Google Scholar] [CrossRef]
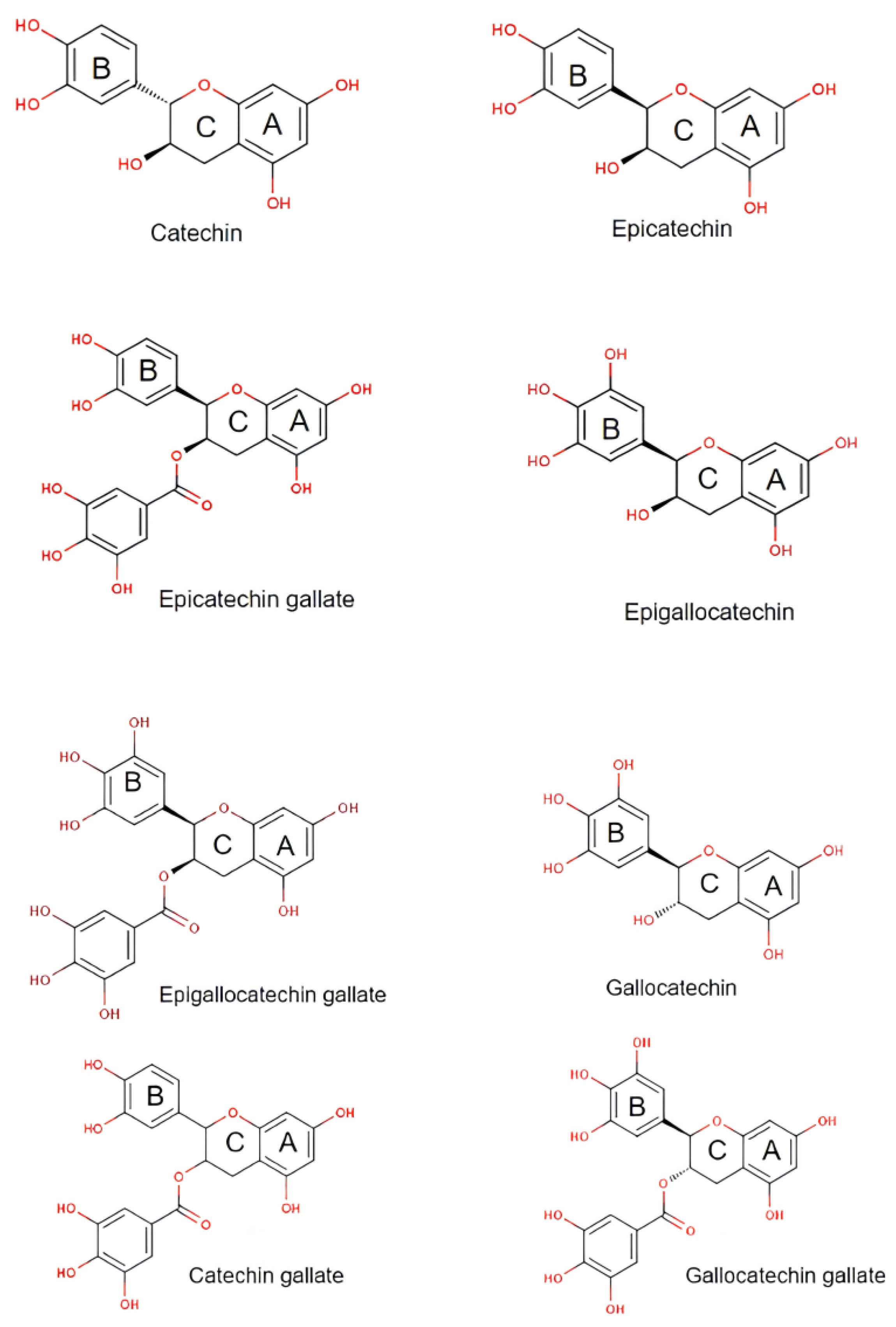
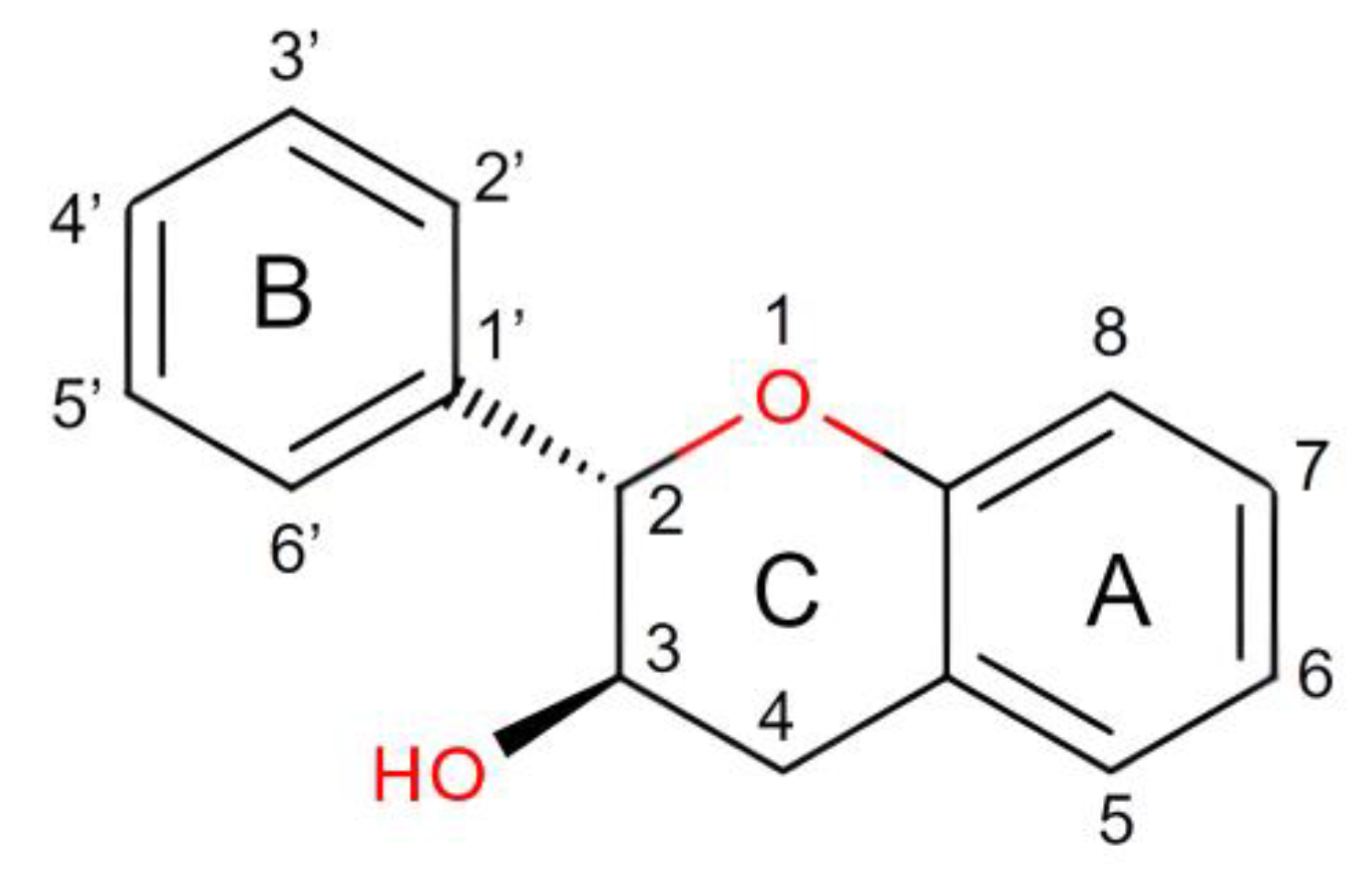
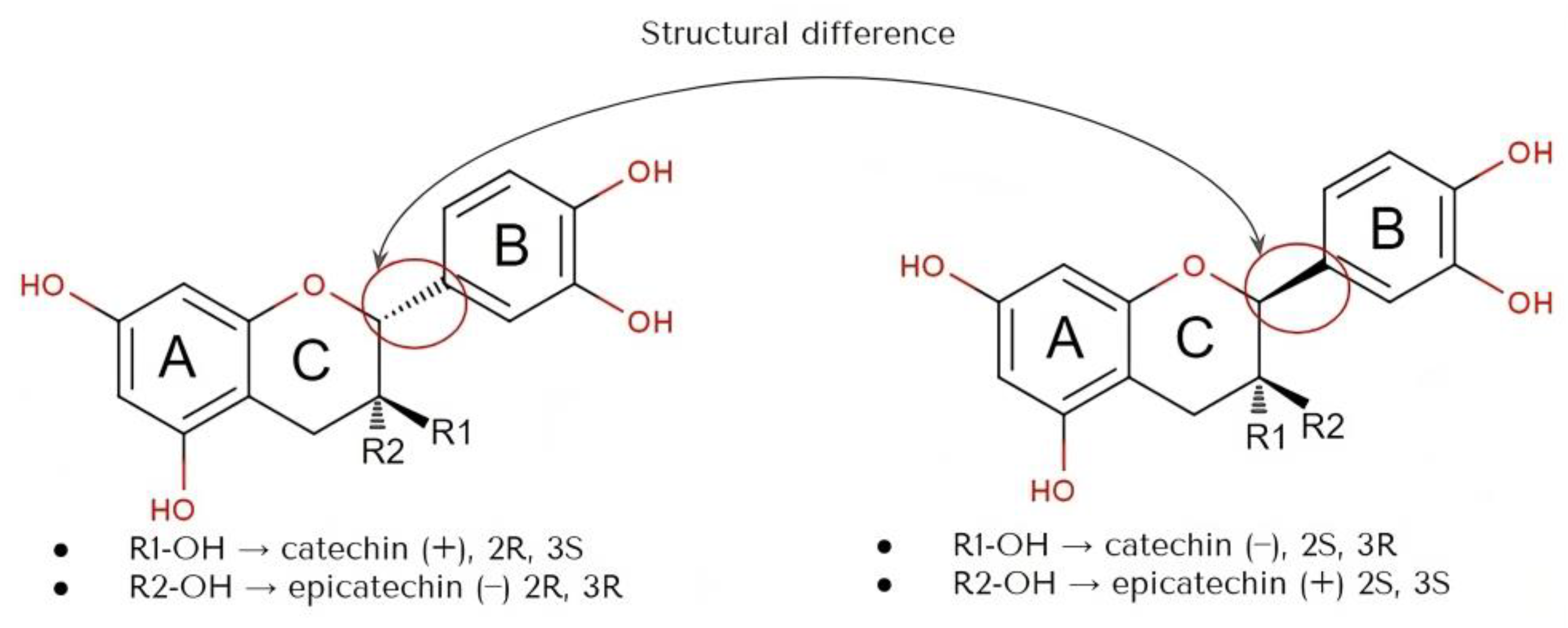
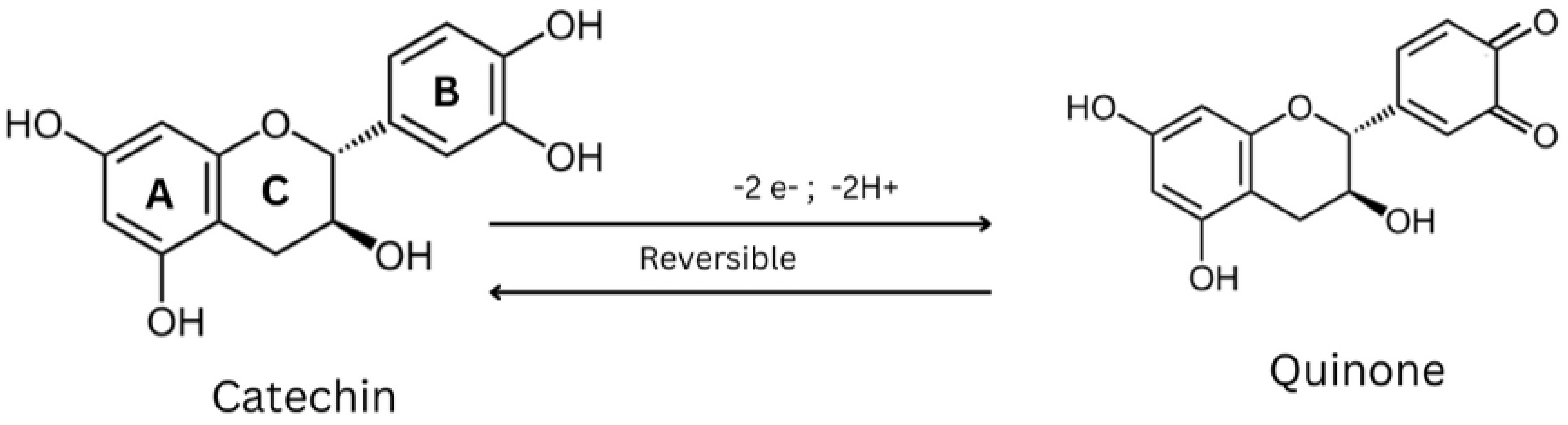


| Physicochemical Parameters | Accepted Value | Reference |
|---|---|---|
| Organoleptic | Solid form, it appears as a light brown to dark reddish-brown substance with a distinctive odour. It possesses a chelate taste that is slightly bitter at first but ends with a sweet aftertaste. | [20] |
| Water Content | Quantity should not exceed 14% | [20] |
| Ash Content | Quantity should not exceed 0.5% | [20] |
| Ash, not soluble in acid | Quantity should not exceed 0.1% | [20] |
| Purity | Contains no less than 90% tannins counted as catechins. | [20] |
| Identification | The assay was carried out using spectrophotometry with a wavelength of 294 nm. | [20] |
| Molecular weight | 290.27 g/mol | [20] |
| Solubility | Soluble in water and polar organic solvents; soluble in pressurised hot water between 298.75 to 415.85 K; soluble in mixtures of supercritical carbon dioxide (SC-CO2) and ethanol at 313 K and pressures ranging from 80 to 120 bar; soluble in SC-CO2 between 313.15 and 343.15 K and pressures ranging from 12 to 26 MPa using ethanol as the co-solvent. | [21] |
| Methods | Sample | Result | Ref. |
|---|---|---|---|
| DPPH Assay | Camellia sinensis |
| [37] |
| Lepisanthes alata (Blume) Leenh |
| [38] | |
| |||
| |||
| Sterculia quadrifida R. | Bark: 51.5 µg/mL (50%) | [22] | |
| Malondialdehyde | Uncaria Gambir Roxb |
| [39] |
| Uncaria Gambir Roxb. | Varies from 2.732% to 3.792% | [40] | |
| Combination of Isolated Catechin and Quercetin |
| [41] |
| Antioxidant Function | Mechanism of Action | Reference |
|---|---|---|
| Anti-ageing | Antioxidants inhibit the action of superoxide dismutase (SOD) enzymes, which play a role in degrading collagen. | [43] |
| Skin Brightening | Antioxidants have whitening effects by inhibiting tyrosinase and act as anti-inflammatory agents for hyperpigmentation caused by UV exposure (commonly known as melasma). | [44] |
| UV Filters | Topically administered antioxidants can enhance the photoprotective capabilities of UV filters by reducing erythema, inhibiting the development of sunburned skin cells, and causing immunosuppression. | [45] |
| Skin Hydration and Anti-Hyperpigmentation | Antioxidants suppress the production and secretion of melanin in melanoma cells to enhance skin hydration and improve hyperpigmentation. | [46] |
| Dosage Form | Formulation | Evaluation Available | Reference |
|---|---|---|---|
| Solid Soap | Catechin (obtained from extract), Aquadest, EDTA, Olive oil, Palm oil, Stearic acid |
| [64] |
| Sunscreen | Catechin, Cera alba, Tween 80, Ceryl Alcohol, Stearyl Alcohol |
| [65] |
| Gambir Leaf Extract, Glycerin, Triethanolamine, Propylene Glycol, Aquadest |
| [66] | |
| Solid Shampoo | Sodium Cocoyl Isethionate, Coco Glucoside, Beeswax, Shea Butter, Panthenol, Essential Oil, Lactic Acid, Tocopherol, BHT, Mango Peel Extract (containing catechin) |
| [67] |
| Lip Balm | Catechin Extract, Ethyl Alcohol, Lanolin, Cera alba, Propylene Glycol, Oleum rosae, Nipagin, Dye, Liquid Paraffin |
| [65] |
| Cream | Gambir Leaf Extract, Stearic Acid, Cetyl Alcohol, Paraffin, Isopropyl Myristic, Methylparaben, Triethanolamine, Glycerine, Perfume, Aquadest |
| [68] |
| Pell-Off Gel Mask | Catechins, PVA, PVP K-30, Propylene Glycol, Methylparaben, Propylparaben, Ethanol 70%, Citrus Essential Oil, Distilled Water |
| [69] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mita, S.R.; Husni, P.; Putriana, N.A.; Maharani, R.; Hendrawan, R.P.; Dewi, D.A. A Recent Update on the Potential Use of Catechins in Cosmeceuticals. Cosmetics 2024, 11, 23. https://doi.org/10.3390/cosmetics11010023
Mita SR, Husni P, Putriana NA, Maharani R, Hendrawan RP, Dewi DA. A Recent Update on the Potential Use of Catechins in Cosmeceuticals. Cosmetics. 2024; 11(1):23. https://doi.org/10.3390/cosmetics11010023
Chicago/Turabian StyleMita, Soraya Ratnawulan, Patihul Husni, Norisca Aliza Putriana, Rani Maharani, Ryan Proxy Hendrawan, and Dian Anggraeni Dewi. 2024. "A Recent Update on the Potential Use of Catechins in Cosmeceuticals" Cosmetics 11, no. 1: 23. https://doi.org/10.3390/cosmetics11010023
APA StyleMita, S. R., Husni, P., Putriana, N. A., Maharani, R., Hendrawan, R. P., & Dewi, D. A. (2024). A Recent Update on the Potential Use of Catechins in Cosmeceuticals. Cosmetics, 11(1), 23. https://doi.org/10.3390/cosmetics11010023







