Exploring the Potential Anti-Inflammatory and Wound-Healing Proprieties of Cepaea hortensis Snail Mucin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Snail Crude Material
2.2. Animals
2.2.1. Evaluation of Dermal and Ocular Toxicity and Analysis of Wound-Healing Activity
2.2.2. Evaluation of Anti-Inflammatory Activity
2.3. Dermal Toxicity Evaluation
2.4. Ocular Toxicity Evaluation
2.5. Topical Anti-Inflammatory Activity Assessment
2.6. Histological Study of the Ear Pieces
2.7. Evaluation of the In Vivo Healing Effect
2.8. Statistical Analysis
3. Results and Discussion
3.1. Toxicological Tests
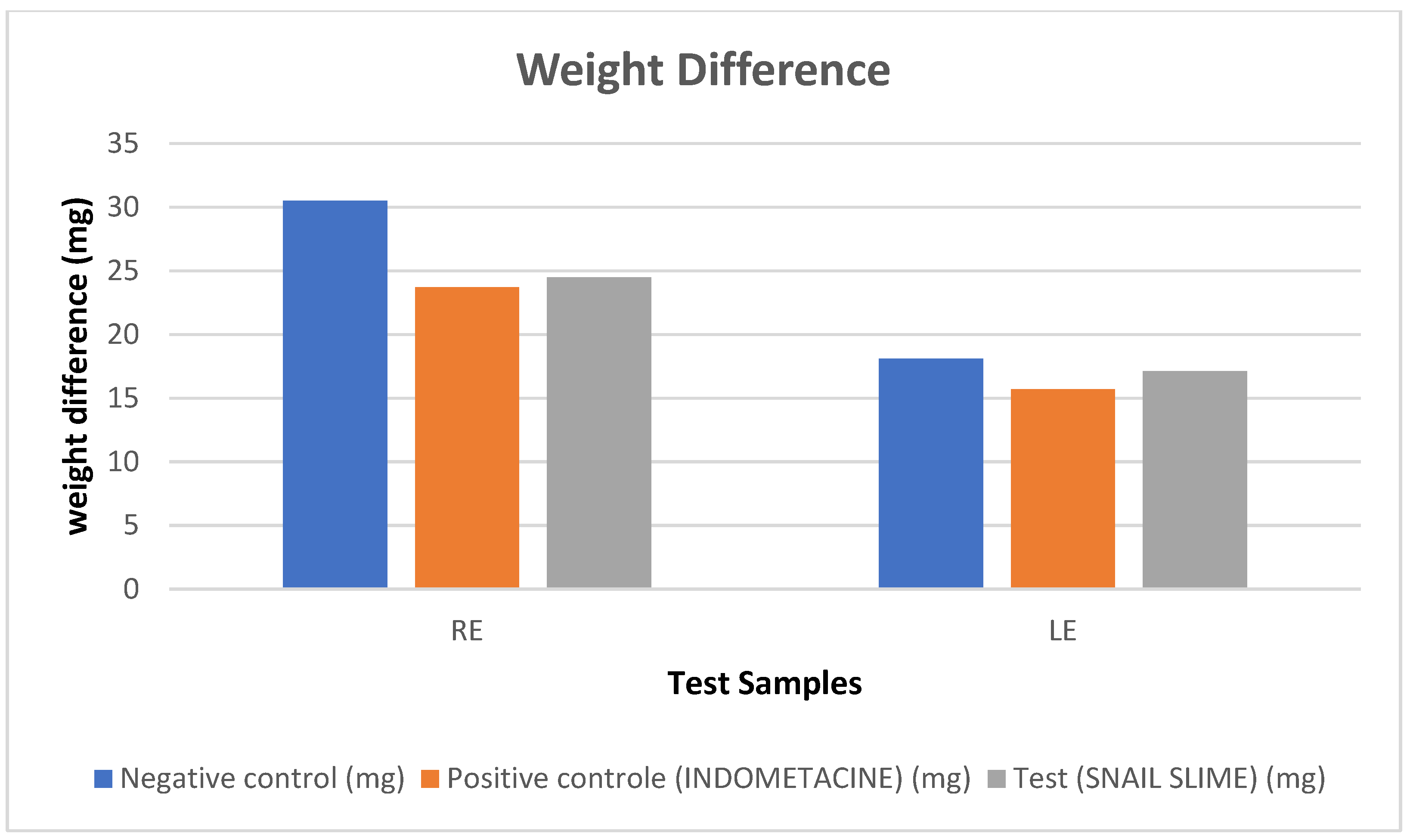
3.2. Evaluation of the Anti-Inflammatory Activity of Snail Slime
3.3. Evaluation of Ear Tissue through Microscopic Examination
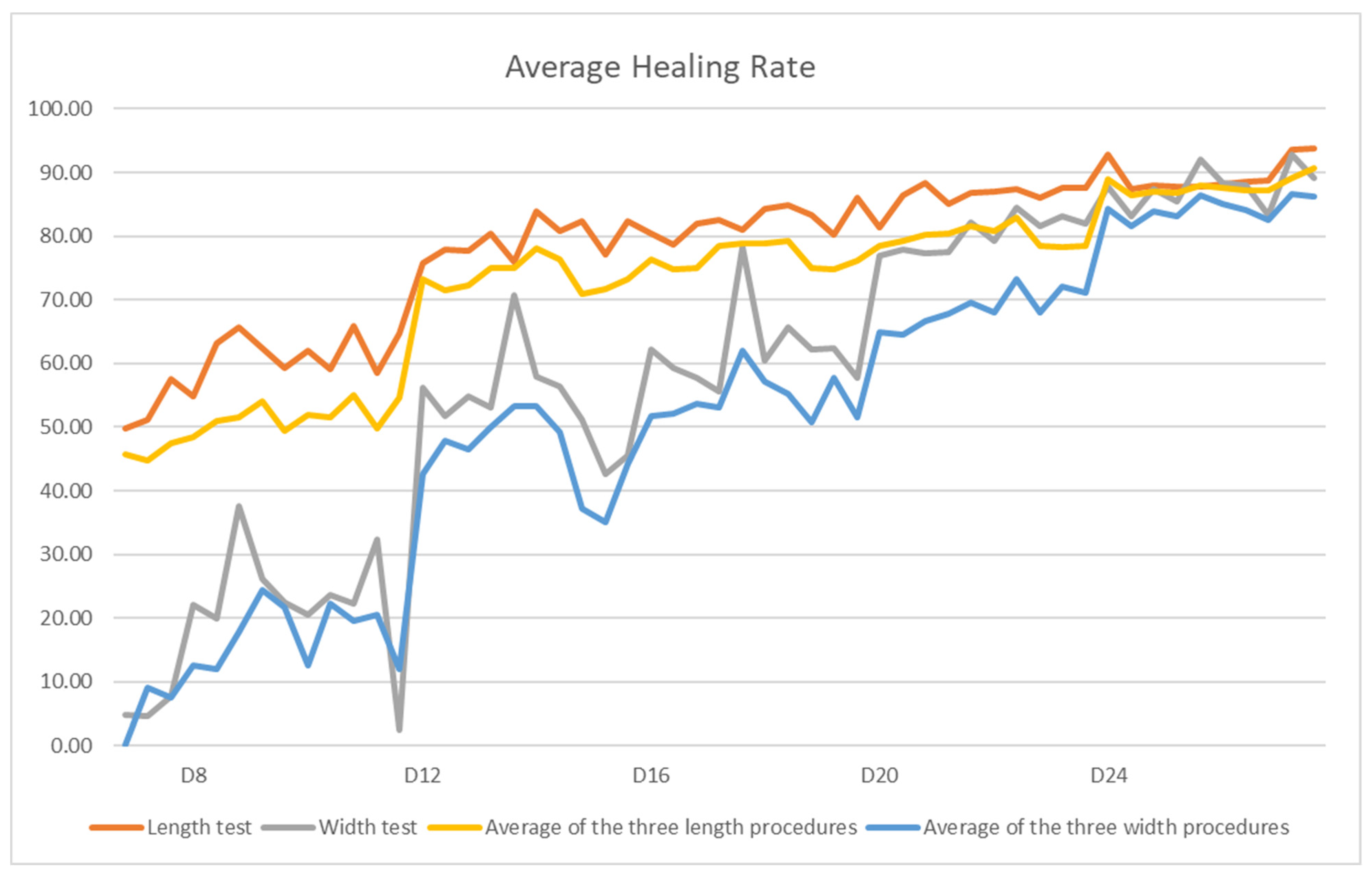
3.4. Evaluation of the In Vivo Healing Effect
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Libby, P. Inflammatory Mechanisms: The Molecular Basis of Inflammation and Disease. Nutr. Rev. 2007, 65, 140–146. [Google Scholar] [CrossRef]
- Abdulkhaleq, L.; Assi, M.; Abdullah, R.; Zamri-Saad, M.; Taufiq-Yap, Y.; Hezmee, M. The crucial roles of inflammatory mediators in inflammation: A review. Vet. World 2018, 11, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Mastrangelo, F.; Ronconi, G.; Kritas, S.; Conti, P. Stimulated mast cells release inflammatory cytokines: Potential suppression and therapeutical aspects. J. Biol. Regul. Homeost. Agents 2018, 32, 1355–1360. [Google Scholar]
- Kaminska, B. MAPK signalling pathways as molecular targets for anti-inflammatory therapy—From molecular mechanisms to therapeutic benefits. Biochim. Biophys. Acta 2005, 1754, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Sheela, M.K.; Sasikala, M.S.; Padmini, M.R.; Sivaranjani, M.D.; Shobana, M.M. Biomolecules; Associationofindian Biologists Publications: New Delhi, India, 2021; ISBN 978-81-950231-2-7. [Google Scholar]
- Bonnemain, B. Helix and Drugs: Snails for Western Health Care From Antiquity to the Present. Evid.-Based Complement. Altern. Med. 2005, 2, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.K.; Hirasawa, N.; Lee, Y.S.; Kim, Y.S.; Shin, K.H.; Ryu, N.; Ohuchi, K. Inhibition by acharan sulphate of angiogenesis in experimental inflammation models. Br. J. Pharmacol. 2002, 137, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Lee, Y.H.; Lee, Y.R.; Im, S.-A.; Lee, J.K.; Kim, Y.S.; Sim, J.S.; Choi, H.S.; Lee, C.K. Activation of professional antigen presenting cells by acharan sulfate isolated from giant african snail, achatina fulica. Arch. Pharmacal. Res. 2007, 30, 866–870. [Google Scholar] [CrossRef]
- Dhiman, V.; Pant, D. Human health and snails. J. Immunoass. Immunochem. 2021, 42, 211–235. [Google Scholar] [CrossRef]
- Gabriel, U.I.; Mirela, S.; Ionel, J. Quantification of mucoproteins (glycoproteins) from snails mucus, Helix aspersa and Helix Pomatia. J. Agroaliment. Process. Technol. 2011, 17, 410–413. [Google Scholar]
- Gubitosa, J.; Rizzi, V.; Fini, P.; Fanelli, F.; Sibillano, T.; Corriero, N.; Cosma, P. Chitosan/snail slime films as multifunctional platforms for potential biomedical and cosmetic applications: Physical and chemical characterization. J. Mater. Chem. B 2023, 11, 2638–2649. [Google Scholar] [CrossRef]
- Truchuelo, M.T.; Vitale, M. A cosmetic treatment based on the secretion of Cryptomphalus aspersa 40% improves the clinical results after the use of nonablative fractional laser in skin aging. J. Cosmet. Dermatol. 2020, 19, 622–628. [Google Scholar] [CrossRef]
- Atta, S.A.; Ibrahim, A.M.; Megahed, F.A.K. In-Vitro Anticancer and Antioxidant Activities of Eremina desertorum (Forsskal, 1775) Snail Mucin. Asian Pac. J. Cancer Prev. APJCP 2021, 22, 3467–3474. [Google Scholar] [CrossRef]
- Ho, C.-Y.; Hu, D.-W.; Chen, B.-R.; Yang, C.-C.; Yao, C.-H.; Ni, T.; Ho, W.-C.; Chen, Y.-S.; Tu, C.-Y.; Chang, W.-C.; et al. Snail Mucus Enhances Chemosensitivity of Triple-negative Breast Cancer Via Activation of the Fas Pathway. Anticancer Res. 2022, 42, 845–855. [Google Scholar] [CrossRef]
- Ricci, A.; Gallorini, M.; Feghali, N.; Sampò, S.; Cataldi, A.; Zara, S. Snail Slime Extracted by a Cruelty Free Method Preserves Viability and Controls Inflammation Occurrence: A Focus on Fibroblasts. Molecules 2023, 28, 1222. [Google Scholar] [CrossRef]
- Rizzi, V.; Gubitosa, J.; Fini, P.; Nuzzo, S.; Agostiano, A.; Cosma, P. Snail slime-based gold nanoparticles: An interesting potential ingredient in cosmetics as an antioxidant, sunscreen, and tyrosinase inhibitor. J. Photochem. Photobiol. B Biol. 2021, 224, 112309. [Google Scholar] [CrossRef]
- Di Filippo, M.F.; Di Matteo, V.; Dolci, L.S.; Albertini, B.; Ballarin, B.; Cassani, M.C.; Passerini, N.; Gentilomi, G.A.; Bonvicini, F.; Panzavolta, S. Effectiveness of Snail Slime in the Green Synthesis of Silver Nanoparticles. Nanomaterials 2022, 12, 3447. [Google Scholar] [CrossRef]
- Noothuan, N.; Apitanyasai, K.; Panha, S.; Tassanakajon, A. Snail mucus from the mantle and foot of two land snails, Lissachatina fulica and Hemiplecta distincta, exhibits different protein profile and biological activity. BMC Res. Notes 2021, 14, 138. [Google Scholar] [CrossRef]
- El-Zawawy, N.A.; Mona, M.M. Antimicrobial efficacy of Egyptian Eremina desertorum and Helix aspersa snail mucus with a novel approach to their anti-inflammatory and wound healing potencies. Sci. Rep. 2021, 11, 24317. [Google Scholar] [CrossRef]
- OCDE. Essai n° 404: Effet Irritant/Corrosif Aigu sur la Peau; OCDE: Paris, France, 2015. [Google Scholar]
- OCDE. Essai n° 405: Effet Irritant/Corrosif Aigu sur les Yeux; OCDE: Paris, France, 2023. [Google Scholar]
- Tubaro, A.; Dri, P.; Delbello, G.; Zilli, C.; Della Loggia, R. The Croton oil ear test revisited. Agents Actions 1986, 17, 347–349. [Google Scholar] [CrossRef]
- Arvouet-Grand, A.; Lejeune, B.; Bastide, P.; Pourrat, A.; Privat, A.M.; Legret, P. Étude de la cicatrisation des plaies chez le lapin et le rat. J. PharmBelg. 1993, 48, 171–178. [Google Scholar]
- De Sá, J.P.M. Applied Statistics Using SPSS, Statistica, MatLab and R; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007; SPIN: 11908944. [Google Scholar]
- Hourfane, S.; Mechqoq, H.; Errajouani, F.; Rocha, J.M.; El Aouad, N. In Vitro and In Silico Evaluations of Boswellia carterii Resin Dermocosmetic Activities. Cosmetics 2022, 9, 131. [Google Scholar] [CrossRef]
- Nathan, C. Points of control in inflammation. Nature 2002, 420, 846–852. [Google Scholar] [CrossRef]
- Barton, G.M. A calculated response: Control of inflammation by the innate immune system. J. Clin. Investig. 2008, 118, 413–420. [Google Scholar] [CrossRef]
- Altinier, G.; Sosa, S.; Aquino, R.P.; Mencherini, T.; Della Loggia, R.; Tubaro, A. Characterization of Topical Antiinflammatory Compounds in Rosmarinus officinalis L. J. Agric. Food Chem. 2007, 55, 1718–1723. [Google Scholar] [CrossRef]
- Delaporte, R.; Sarragiotto, M.; Takemura, O.; Sanchez, G.; Nakamura, C. Evaluation of the antioedematogenic, free radical scavenging and antimicrobial activities of aerial parts of Tillandsia streptocarpa Baker–Bromeliaceae. J. Ethnopharmacol. 2004, 95, 229–233. [Google Scholar] [CrossRef]
- Gubitosa, J.; Rizzi, V.; Fini, P.; Laurenzana, A.; Fibbi, G.; Veiga-Villauriz, C.; Fanelli, F.; Fracassi, F.; Onzo, A.; Bianco, G.; et al. Biomolecules from snail mucus (Helix aspersa) conjugated gold nanoparticles, exhibiting potential wound healing and anti-inflammatory activity. Soft Matter 2020, 16, 10876–10888. [Google Scholar] [CrossRef]
- Putranti, I.O.; Sutrisna, E.; Ekowati, N.; Mawardi, P.; Novrial, D. The Role of Achatina Fulica Snail Slime Extract Enzymatic Antioxidants as Photoprotector in Sunburn Model Mice. Int. J. Med. Sci. Clin. Res. Stud. 2022, 02, 624–630. [Google Scholar] [CrossRef]
- Anju, M.; Vijayan, K. Comparison of antimicrobial properties of the slime of invasive alien pest Lissachatina fulica and an indigenous pest Cryptozona bistrialis snails. Crop Res. 2021, 56, 159–164. [Google Scholar]
- Cilia, G.; Fratini, F. Antimicrobial properties of terrestrial snail and slug mucus. J. Complement. Integr. Med. 2018, 15, 20170168. [Google Scholar] [CrossRef]
- Perpelek, M.; Tamburaci, S.; Aydemir, S.; Tihminlioglu, F.; Baykara, B.; Karakasli, A.; Havitcioglu, H. Bioactive snail mucus-slime extract loaded chitosan scaffolds for hard tissue regeneration: The effect of mucoadhesive and antibacterial extracts on physical characteristics and bioactivity of chitosan matrix. Biomed. Mater. 2021, 16, 065008. [Google Scholar] [CrossRef]
- Swastini, I.G.A.A.P.; Widnyana, A.N.K.P. Antimicrobial properties of snail slime (Achatina fulica) on the growth of actinobacilus actinomycetemcomitans bacteria causing periodontitis. J. Glob. Pharma Technol. 2009, 11, 170–174. [Google Scholar]
- Monaco, J.L.; Lawrence, W.T. Acute wound healing. Clin. Plast. Surg. 2003, 30, 1–12. [Google Scholar] [CrossRef]
- Guo, S.; DiPietro, L.A. Factors Affecting Wound Healing. J. Dent. Res. 2010, 89, 219–229. [Google Scholar] [CrossRef]
- Song, Y.; Cui, Y.; Hao, L.; Zhu, J.; Yi, J.; Kang, Q.; Huang, J.; Lu, J. Wound-healing activity of glycoproteins from white jade snail (Achatina fulica) on experimentally burned mice. Int. J. Biol. Macromol. 2021, 175, 313–321. [Google Scholar] [CrossRef]




| Rabbit n° | Time | Erythema and Eschar (0: Absence) | Edema (0: Absence) | ||
|---|---|---|---|---|---|
| Scarred Area | Unscarred Area | Scarred Area | Unscarred Area | ||
| 1 | 30 min | 0 | 0 | 0 | 0 |
| 24 h | 0 | 0 | 0 | 0 | |
| 72 h | 0 | 0 | 0 | 0 | |
| 2 | 30 min | 0 | 0 | 0 | 0 |
| 24 h | 0 | 0 | 0 | 0 | |
| 72 h | 0 | 0 | 0 | 0 | |
| 3 | 30 min | 0 | 0 | 0 | 0 |
| 24 h | 0 | 0 | 0 | 0 | |
| 72 h | 0 | 0 | 0 | 0 | |
| 4 | 30 min | 0 | 0 | 0 | 0 |
| 24 h | 0 | 0 | 0 | 0 | |
| 72 h | 0 | 0 | 0 | 0 | |
| 5 | 30 min | 0 | 0 | 0 | 0 |
| 24 h | 0 | 0 | 0 | 0 | |
| 72 h | 0 | 0 | 0 | 0 | |
| 6 | 30 min | 0 | 0 | 0 | 0 |
| 24 h | 0 | 0 | 0 | 0 | |
| 72 h | 0 | 0 | 0 | 0 | |
| Evaluation of Damage | 1 h | 24 h | 48 h | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Rabbit Number | 1 | 2 | 3 | 1 | 2 | 3 | 1 | 2 | 3 | |
| Conjunctiva | A: chemosis | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| B: lachrymation | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| C: enanthem | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| (a + b + c) × 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Iris | Direct photomotor reflex of the pupil | N | N | N | N | N | N | N | N | N |
| A: congestion degree | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| A × 5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Cornea | A: degree of opacity | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| B: area reached | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| A × b × 5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Ulceration | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Area reached | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Granulation | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Individual Eye irritation Index (I.E.I) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Score per zone | Conjunctiva Iris Cornea | 0 0 0 | Conjunctiva Iris Cornea | 0 0 0 | Conjunctiva Iris Cornea | 0 0 0 | ||||
| Average Eye Index (A.E.I) | 0 | 0 | 0 | |||||||
| Administered Dose (µL in Ear) | Number of Animals | Average Weight Difference (mg) | Edema Inhibition % | |
|---|---|---|---|---|
| Negative control batch (µL) | 5 | 10 | 150 | 0% |
| Positive control (indomethacin) (µg) | 50 | 10 | 79.75 | 47% |
| Test batch (snail slime) (mg) | 15 | 10 | 61.33 | 59% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Errajouani, F.; Bakrim, H.; Hourfane, S.; Louajri, A.; Rocha, J.M.; El Aouad, N.; Laglaoui, A. Exploring the Potential Anti-Inflammatory and Wound-Healing Proprieties of Cepaea hortensis Snail Mucin. Cosmetics 2023, 10, 170. https://doi.org/10.3390/cosmetics10060170
Errajouani F, Bakrim H, Hourfane S, Louajri A, Rocha JM, El Aouad N, Laglaoui A. Exploring the Potential Anti-Inflammatory and Wound-Healing Proprieties of Cepaea hortensis Snail Mucin. Cosmetics. 2023; 10(6):170. https://doi.org/10.3390/cosmetics10060170
Chicago/Turabian StyleErrajouani, Fatima, Hanane Bakrim, Sohaib Hourfane, Adnane Louajri, João Miguel Rocha, Noureddine El Aouad, and Amin Laglaoui. 2023. "Exploring the Potential Anti-Inflammatory and Wound-Healing Proprieties of Cepaea hortensis Snail Mucin" Cosmetics 10, no. 6: 170. https://doi.org/10.3390/cosmetics10060170
APA StyleErrajouani, F., Bakrim, H., Hourfane, S., Louajri, A., Rocha, J. M., El Aouad, N., & Laglaoui, A. (2023). Exploring the Potential Anti-Inflammatory and Wound-Healing Proprieties of Cepaea hortensis Snail Mucin. Cosmetics, 10(6), 170. https://doi.org/10.3390/cosmetics10060170










