Amyloidosis and Longevity: A Lesson from Plants
Abstract
1. Introduction
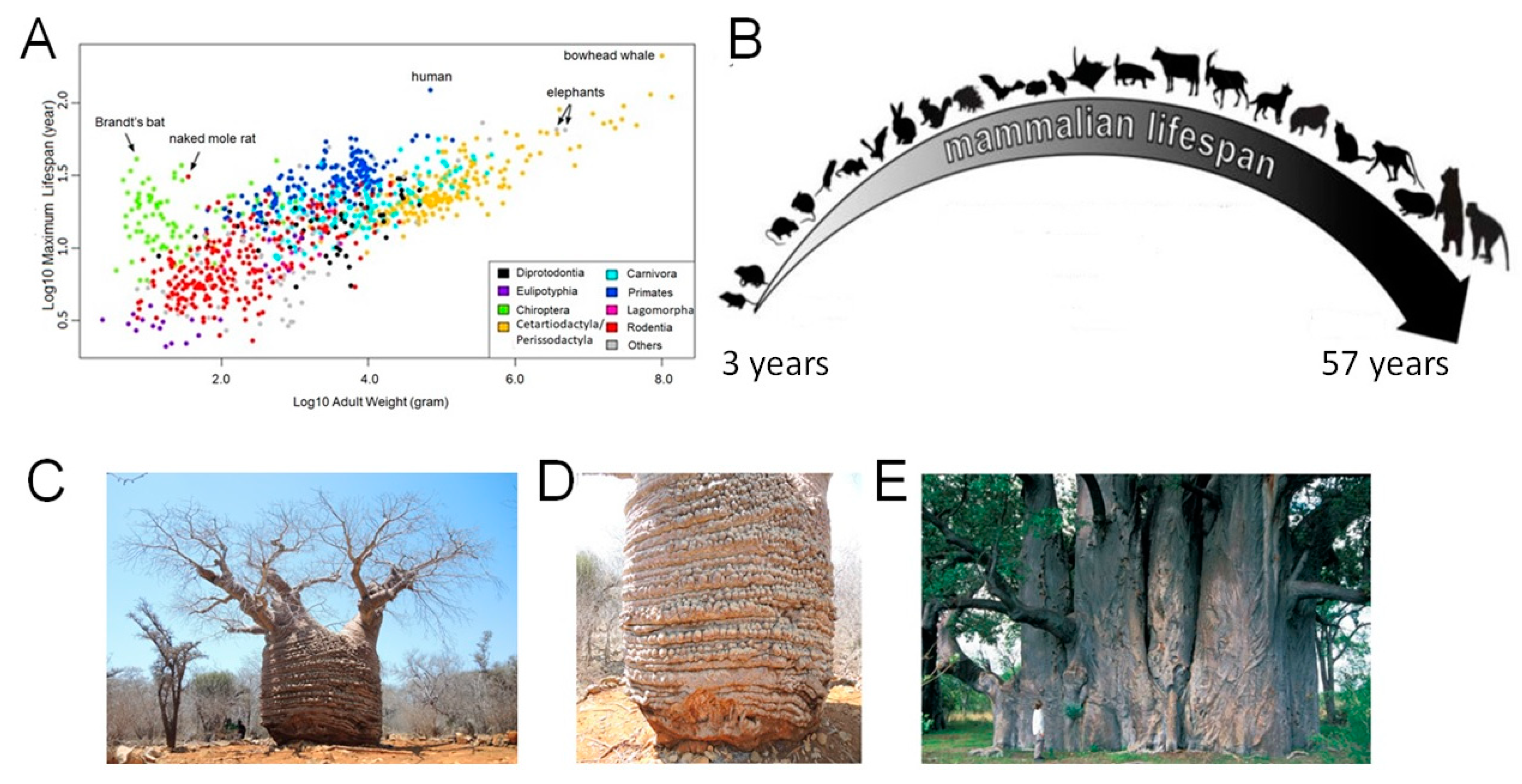
Species Diversity in Maximal Longevity
2. An Overview
2.1. Trees Longevity
2.2. Why Do Vertebrate Animals Not Live as Long as Woody Plants?
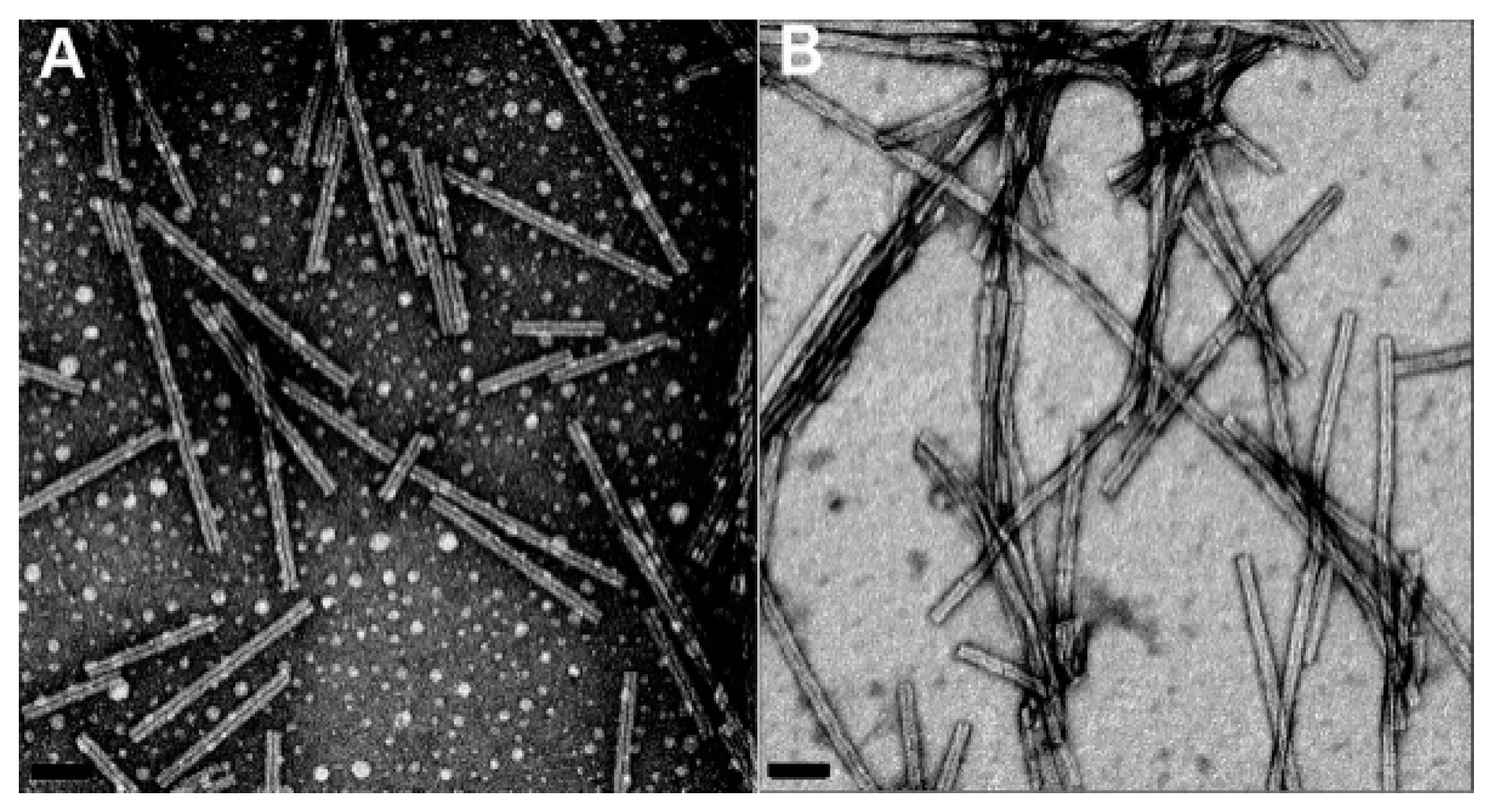
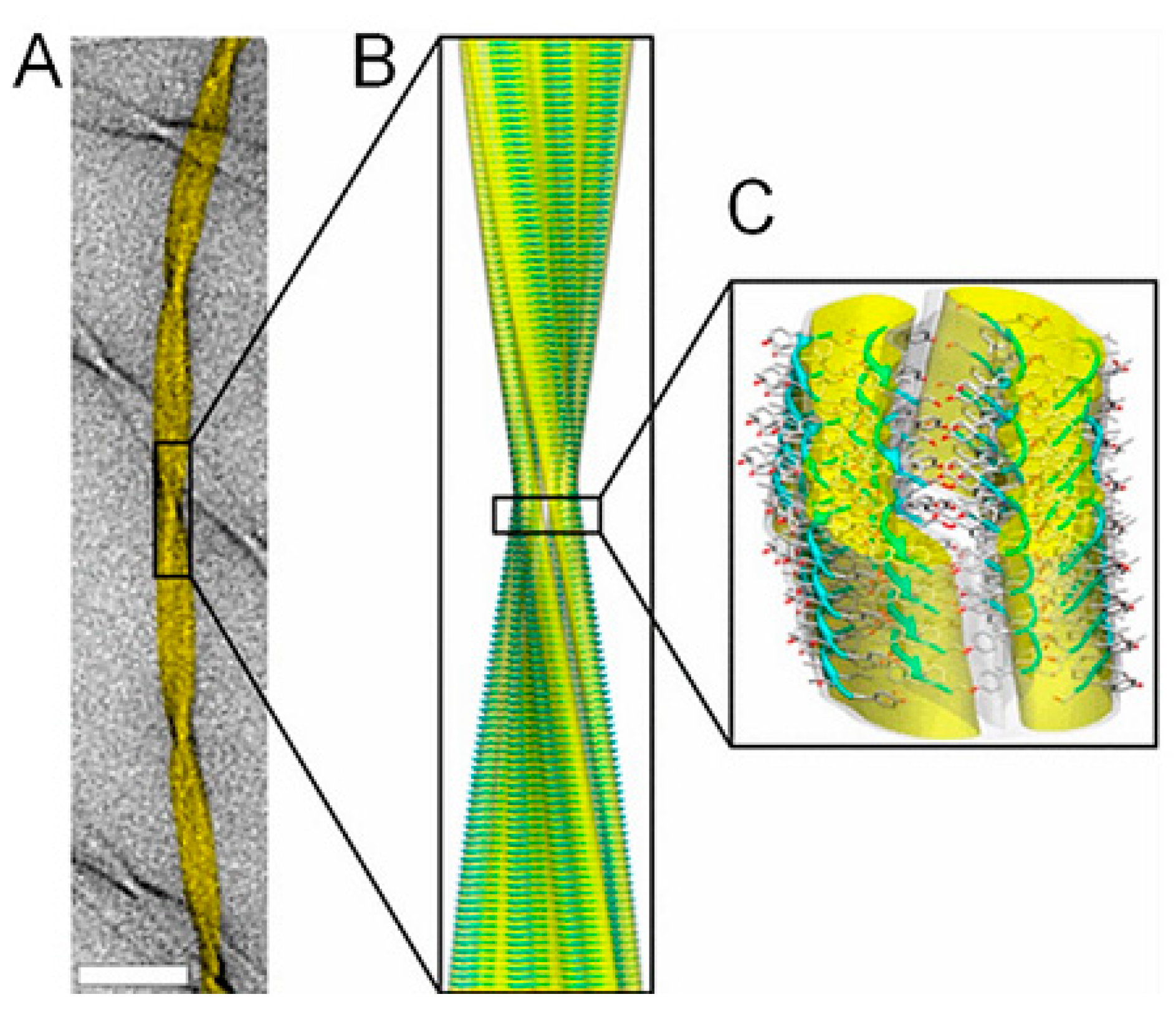
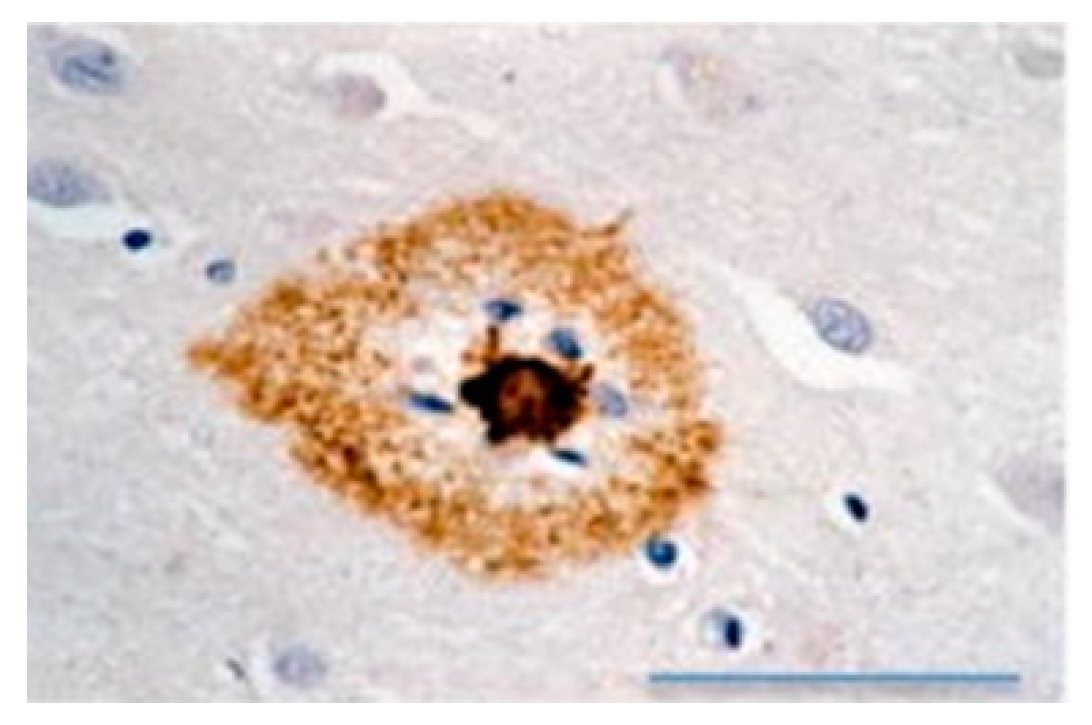
2.3. Amyloid Fibrils and Amyloidosis
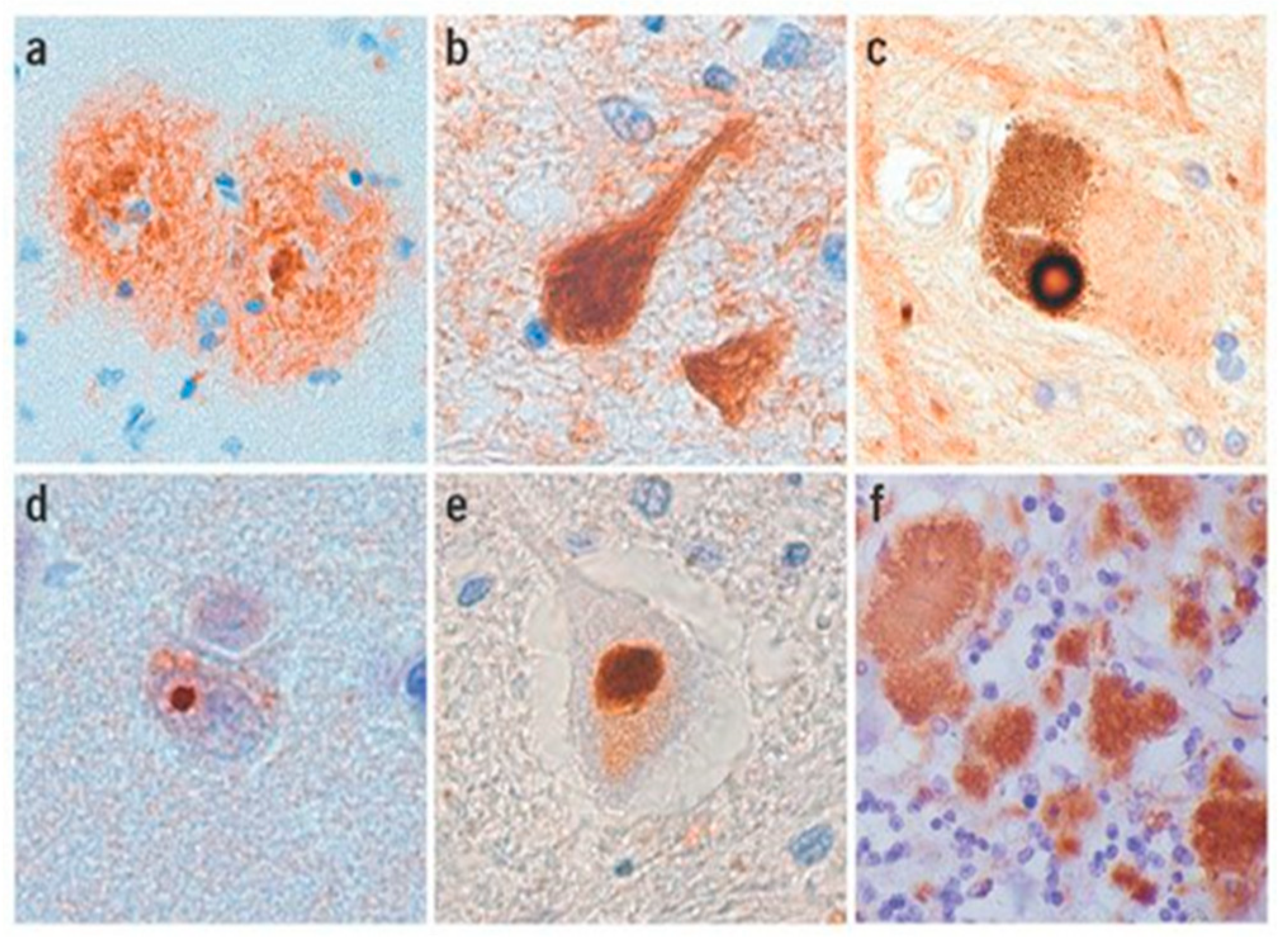
2.4. Amyloidosis in Humans
2.5. Amyloidosis in Animals
2.6. Functional Amyloidosis
2.7. Rare Amyloidosis in Plants
2.8. Certain Amyloidogenic Proteins Highly Expressed in Vertebrates are Absent in Plants—An Example is the Synuclein Family
2.9. Plant Longevity and Secondary Metabolites Inhibiting Amyloidosis and Preventing Aging
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Barja, G. The gene cluster hypothesis of aging and longevity. Biogerontology 2008, 9, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Gladyshev, V.N. Signatures of longevity: Insights from cross-species comparative studies. Semin Cell Dev. Biol. 2017, 70, 190–203. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Seim, I.; Huang, Z.; Gerashchenko, M.V.; Xiong, Z.; Turanov, A.A.; Zhu, Y.; Lobanov, A.V.; Fan, D.; Yim, S.H.; et al. Adaptations to a subterranean environment and longevity revealed by the analysis of mole rat genomes. Cell Rep. 2014, 8, 1354–1364. [Google Scholar] [CrossRef] [PubMed]
- Skulachev, M.V.; Severin, F.F.; Skulachev, V.P. Aging as an evolvability-increasing program which can be switched off by organism to mobilize additional resources for survival. Curr. Aging Sci. 2015, 8, 95–109. [Google Scholar] [CrossRef] [PubMed]
- Buffenstein, R. Negligible senescence in the longest living rodent, the naked mole-rat: Insights from a successfully aging species. J. Comp. Physiol. B. 2008, 178, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Tacutu, R.; Craig, T.; Budovsky, A.; Wuttke, D.; Lehmann, G.; Taranukha, D.; Costa, J.; Fraifeld, V.E.; de Magalhães, J.P. Human Ageing Genomic Resources: Integrated databases and tools for the biology and genetics of ageing. Nucleic Acids Res. 2013, 41, 1027–1033. [Google Scholar] [CrossRef] [PubMed]
- Patrut, A.; von Reden, K.F.; Danthu, P.; Leong Pock-Tsy, J.-M.; Patrut, R.T.; Lowy, D.A. Searching for the Oldest Baobab of Madagascar: Radiocarbon Investigation of Large Adansonia rubrostipa Trees. PLoS ONE 2015, 10, e0121170. [Google Scholar] [CrossRef] [PubMed]
- Livingstone, D. A Popular Account of Missionary Travels and Researches in South Africa; Murray: London, UK, 1857; p. 732. [Google Scholar]
- Patrut, A.; von Reden, K.F.; Lowy, D.A.; Alberts, A.H.; Pohlman, J.W.; Wittmann, R.; Gerlach, D.; Xu, L.; Mitchell, C.S. Radiocarbon dating of a very large African baobab. Tree Physiol. 2007, 27, 1569–1570. [Google Scholar] [CrossRef]
- Carmona, J.J.; Michan, S. Biology of Healthy Aging and Longevity. Rev. Invest Clin. 2016, 68, 7–16. [Google Scholar]
- Carey, J.R. Longevity minimalists: Life table studies of two species of northern Michigan adult mayflies. Exp. Gerontol. 2002, 37, 567–570. [Google Scholar] [CrossRef]
- Schulman, E. Bristlecone pine, oldest known living thing. Natl. Geogr. Mag. 1958, 113, 354–372. [Google Scholar]
- Flanary, B.E.; Kletetschka, G. Analysis of telomere length and telomerase activity in tree species of various life-spans, and with age in the bristlecone pine Pinus longaeva. Biogerontology 2005, 6, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Brutovská, E.; Sámelová, A.; Dušička, J.; Mičieta, K. Ageing of trees: Application of general ageing theories. Ageing Res. Rev. 2013, 12, 855–866. [Google Scholar] [CrossRef] [PubMed]
- Chambers, J.Q.; Higuchi, N.; Schimel, J.P. Ancient trees in Amazonia. Nature 1998, 39, 135–136. [Google Scholar] [CrossRef]
- Skulachev, M.V.; Skulachev, V.P. Programmed Aging of Mammals: Proof of Concept and Prospects of Biochemical Approaches for Antiaging Therapy. Biochemistry (Moscow) 2017, 82, 1403–1422. [Google Scholar] [CrossRef]
- Lanner, R.M. Why do trees live so long? Ageing Res. Rev. 2002, 1, 653–671. [Google Scholar] [CrossRef]
- Daniell, H.; Lin, C.S.; Yu, M.; Chang, W.J. Chloroplast genomes: Diversity, evolution, and applications in genetic engineering. Genome Biol. 2016, 17, 134. [Google Scholar] [CrossRef]
- Fowler, D.M.; Koulov, A.V.; Balch, W.E.; Kelly, J.W. Functional amyloid–from bacteria to humans. Trends Biochem Sci. 2007, 32, 217–224. [Google Scholar] [CrossRef]
- Li, J.; Hoop, C.L.; Kodali, R.; Sivanandam, V.N.; van der Wel, P.C. Amyloid-like fibrils from a domain-swapping protein feature a parallel, in-register conformation without native-like interactions. J. Biol. Chem. 2011, 286, 28988–28995. [Google Scholar] [CrossRef]
- Fitzpatrick, A.W.; Debelouchina, G.T.; Bayro, M.J.; Clare, D.K.; Caporini, M.A.; Bajaj, V.S.; Jaroniec, C.P.; Wang, L.; Ladizhansky, V.; Müller, S.A.; et al. Atomic structure and hierarchical assembly of a cross-β amyloid fibril. Proc. Natl. Acad. Sci. USA 2013, 110, 5468–5473. [Google Scholar] [CrossRef]
- López delaPaz, M.; Serrano, L. Sequence determinants of amyloid fibril formation. Proc. Natl. Acad. Sci. USA 2004, 101, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Forman, M.S.; Trojanowski, J.Q.; Lee, V.M.-Y. Neurodegenerative diseases: A decade of discoveries paves the way for therapeutic breakthroughs. Nature Med. 2004, 10, 1055–1563. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, D.; Jucker, M. The amyloid state of proteins in human diseases. Cell 2012, 148, 1188–1203. [Google Scholar] [CrossRef] [PubMed]
- Chiti, F.; Dobson, C.M. Protein Misfolding, Amyloid Formation, and Human Disease: A Summary of Progress over the Last Decade. Annu. Rev. Biochem. 2017, 86, 27–68. [Google Scholar] [CrossRef] [PubMed]
- Soto, C.; Pritzkow, S. Protein misfolding, aggregation, and conformational strains in neurodegenerative diseases. Nat Neurosci. 2018, 21, 1332–1340. [Google Scholar] [CrossRef] [PubMed]
- Surguchev, A.; Surguchov, A. Conformational Diseases: Looking into the eyes. Brain Res. Bull. 2010, 81, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Luheshi, L.M.; Crowther, D.C.; Dobson, C.M. Protein misfolding and disease: From the test tube to the organism. Curr. Opinion Chem. Biol. 2008, 12, 25–31. [Google Scholar] [CrossRef]
- Pham, C.L.L.; Kwan, A.H.; Sunde, M. Functional amyloid: Widespread in nature, diverse in purpose. Essays Biochem. 2014, 56, 207–219. [Google Scholar] [CrossRef]
- Van Gerven, N.; Van der Verren, S.E.; Reiter, D.M.; Remaut, H. The Role of Functional Amyloids in Bacterial Virulence. J. Mol. Biol. 2018, 430, 3657–3684. [Google Scholar] [CrossRef]
- Baldwin, A.J.; Knowles, T.P.; Tartaglia, G.G.; Fitzpatrick, A.W.; Devlin, G.L.; Shammas, S.L.; Waudby, C.A.; Mossuto, M.F.; Meehan, S.; Gras, S.L.; et al. Metastability of native proteins and the phenomenon of amyloid formation. J. Am. Chem. Soc. 2011, 133, 14160–14163. [Google Scholar] [CrossRef]
- Antonets, K.S.; Nizhnikov, A.A. Amyloids and prions in plants: Facts and perspectives. Prion 2017, 1, 300–312. [Google Scholar] [CrossRef] [PubMed]
- Mohammad-Beigi, H.; Kjaer, L.; Eskandari, H.; Aliakbari, F.; Christiansen, G.; Ruvo, G.; Ward, J.L.; Otzen, D.E. A Possible Connection between Plant Longevity and the Absence of Protein Fibrillation: Basis for Identifying Aggregation Inhibitors in Plants. Front. Plant Sci. 2019, 10, 148. [Google Scholar] [CrossRef] [PubMed]
- Blackett, T.A.; Simpson, V.R.; Haugland, S.; Everest, D.J.; Muir, C.F.; Smith, K.C.; Mill, A.C. Mortalities, amyloidosis and other diseases in free-living red squirrels (Sciurus vulgaris) on Jersey, Channel Islands. Vet. Rec. 2018, 183, 503. [Google Scholar] [CrossRef] [PubMed]
- Terio, K.; O’Brien, T.; Lamberski, N.; Famula, T.R.; Munson, L. Amyloidosis in black-footed cats (Felis nigvipes). Vet. Pathol. 2008, 45, 393–400. [Google Scholar] [CrossRef]
- Woldemeskel, M. A concise review of amyloidosis in animals. Vet. Med. Int. 2012, 427296, 1–11. [Google Scholar] [CrossRef]
- Hara, H.; Ono, F.; Nakamura, S.; Matsumoto, S.E.; Jin, H.; Hattori, N.; Tabira, T. An Oral Aβ Vaccine Using a Recombinant Adeno-Associated Virus Vector in Aged Monkeys: Reduction in Plaque Amyloid and Increase in Aβ Oligomers. J. Alzheimers Dis. 2016, 54, 1047–1059. [Google Scholar] [CrossRef]
- Ji, S.; Luo, Y.; Cai, Q.; Cao, Z.; Zhao, Y.; Mei, J.; Li, C.; Xia, P.; Xie, Z.; Xia, Z.; et al. LC Domain-Mediated Coalescence Is Essential for Otu Enzymatic Activity to Extend Drosophila Lifespan. Mol. Cell. 2019, 74, 363–377. [Google Scholar] [CrossRef]
- Antonets, K.S.; Nizhnikov, A.A. Predicting Amyloidogenic Proteins in the Proteomes of Plants. Int. J. Mol. Sci. 2017, 18, 2155. [Google Scholar] [CrossRef]
- Garvey, M.; Meehan, S.; Gras, S.L.; Schirra, H.J.; Craik, D.J.; Van der Weerden, N.L.; Anderson, M.A.; Gerrard, J.A.; Carver, J.A. A radish seed antifungal peptide with a high amyloid fibril-forming propensity. Biochim. Biophys. Acta. 2013, 1834, 1615–1623. [Google Scholar] [CrossRef]
- Gour, S.; Kaushik, V.; Kumar, V.; Bhat, P.; Yadav, S.C.; Yadav, J.K. Antimicrobial peptide (Cn-AMP2) from liquid endosperm of Cocos nucifera forms amyloid-like fibrillar structure. J. Pept. Sci. 2016, 22, 201–207. [Google Scholar] [CrossRef]
- Mostaert, A.S.; Higgins, M.J.; Fukuma, T.; Rindi, F.; Jarvis, S.P. Nanoscale mechanical characterisation of amyloid fibrils discovered in a natural adhesive. J. Biol. Phys. 2006, 32, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Mostaert, A.S.; Giordani, C.; Crockett, R.; Karsten, U.; Schumann, R.; Jarvis, S.P. Characterisation of Amyloid Nanostructures in the Natural Adhesive of Unicellular Subaerial Algae. J. Adhes. 2009, 85, 465–483. [Google Scholar] [CrossRef]
- Berthelot, K.; Lecomte, S.; Estevez, Y.; Coulary-Salin, B.; Bentaleb, A.; Cullin, C.; Deffieux, A.; Peruch, F. Rubber elongation factor (REF), a major allergen component in Hevea brasiliensis latex has amyloid properties. PLoS ONE 2012, 7, e48065. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Kim, J.G.; Jeon, E.; Yoo, C.H.; Moon, J.S.; Rhee, S.; Hwang, I. Amyloidogenesis of type III-dependent harpins from plant pathogenic bacteria. J. Biol. Chem. 2007, 282, 13601–13609. [Google Scholar] [CrossRef] [PubMed]
- Villar-Piqué, A.; Sabaté, R.; Lopera, O.; Gibert, J.; Torne, J.M.; Santos, M.; Ventura, S. Amyloid-like protein inclusions in tobacco transgenic plants. PLoS ONE 2010, 5, e13625. [Google Scholar]
- Cook, N.P.; Martí, A.A. Facile methodology for monitoring amyloid-β fibrillization. ACS Chem. Neurosci. 2012, 3, 896–899. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Veloso, A.J.; Hung, V.W.; Sindhu, G.; Constantinof, A.; Kerman, K. Electrochemical oxidation of benzothiazole dyes for monitoring amyloid formation related to the Alzheimer’s disease. Anal. Chem. 2009, 81, 9410–9415. [Google Scholar] [CrossRef]
- Cook, N.P.; Torres, V.; Jain, D.; Martí, A.A. Sensing amyloid-β aggregation using luminescent dipyridophenazine ruthenium(II) complexes. J. Am. Chem. Soc. 2011, 133, 11121–11123. [Google Scholar] [CrossRef]
- Surguchov, A. Intracellular dynamics of synucleins: Here, there and everywhere. Internat. Rev. Cell Mol. Biol. 2015, 320, 103–169. [Google Scholar]
- Iwai, A.; Masliah, E.; Yoshimoto, M.; Ge, N.; Flanagan, L.; de Silva, H.A.; Kittel, A.; Saitoh, T. The precursor protein of non-A beta component of Alzheimer’s disease amyloid is a presynaptic protein of the central nervous system. Neuron 1995, 14, 467–475. [Google Scholar] [CrossRef]
- Andersson, D.I.; Jerlström-Hultqvist, J.; Näsvall, J. Evolution of new functions de novo and from preexisting genes. Cold Spring Harb. Perspect. Biol. 2015, 7, a017996. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Zhang, Y. Evolution and function of de novo originated genes. Mol. Phylogenet. Evol. 2013, 67, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Tautz, D.; Domazet-Loső, T. The evolutionary origin of orphan genes. Nat. Rev. Genet. 2011, 12, 692–702. [Google Scholar] [CrossRef] [PubMed]
- Roman, A.Y.; Kovrazhkina, E.A.; Razinskaya, O.D.; Kukharsky, M.S.; Maltsev, A.V.; Ovchinnikov, R.K.; Lytkina, O.A.; Smirnov, A.P.; Moskovtsev, A.A.; Borodina, Y.V.; et al. Detection of autoantibodies to potentially amyloidogenic protein, gamma-synuclein, in the serum of patients with amyotrophic lateral sclerosis and cerebral circulatory disorders. Dokl. Biochem. Biophys. 2017, 472, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Paleologou, K.E.; Oueslati, A.; Shakked, G.; Rospigliosi, C.C.; Kim, H.Y.; Lamberto, G.R.; Fernandez, C.O.; Schmid, A.; Chegini, F.; Gai, W.P.; et al. Phosphorylation at S87 is enhanced in synucleinopathies, inhibits alpha-synuclein oligomerization, and influences synuclein-membrane interactions. J. Neurosci. 2010, 30, 3184–3198. [Google Scholar] [CrossRef] [PubMed]
- Bertoncini, C.W.; Jung, Y.S.; Fernandez, C.O.; Hoyer, W.; Griesinger, C.; Jovin, T.M.; Zweckstetter, M. Release of long-range tertiary interactions potentiates aggregation of natively unstructured alpha-synuclein. Proc. Natl. Acad. Sci. USA 2005, 102, 1430–1435. [Google Scholar] [CrossRef] [PubMed]
- Iyer, A.; Claessens, M.M.A.E. Disruptive membrane interactions of alpha-synuclein aggregates. Biochim. Biophys. Acta. Proteins Proteom. 2019, 1867, 468–482. [Google Scholar] [CrossRef] [PubMed]
- Edrey, Y.H.; Medina, D.X.; Gaczynska, M.; Osmulski, P.A.; Oddo, S.; Caccamo, A.; Buffenstein, R. Amyloid beta and the longest-lived rodent: The naked mole-rat as a model for natural protection from Alzheimer’s disease. Neurobiol. Aging. 2013, 34, 2352–3260. [Google Scholar] [CrossRef] [PubMed]
- Dhouafli, Z.; Cuanalo-Contreras, K.; Hayouni, E.A.; Mays, C.E.; Soto, C.; Moreno-Gonzalez, I. Inhibition of protein misfolding and aggregation by natural phenolic compounds. Cell Mol. Life Sci. 2018, 75, 3521–3538. [Google Scholar] [CrossRef]
- Gambini, J.; Inglés, M.; Olaso, G.; Lopez-Grueso, R.; Bonet-Costa, V.; Gimeno-Mallench, L.; Mas-Bargues, C.; Abdelaziz, K.M.; Gomez-Cabrera, M.C.; Vina, J.; et al. Properties of Resveratrol: In Vitro and In Vivo Studies about Metabolism, Bioavailability, and Biological Effects in Animal Models and Humans. Oxid. Med. Cell Longev. 2015, 837042. [Google Scholar] [CrossRef]
- Pandey, T.; Sammi, S.R.; Nooreen, Z.; Mishra, A.; Ahmad, A.; Bhatta, R.S.; Pandey, R. Anti-ageing and anti-Parkinsonian effects of natural flavonol, tambulin from Zanthoxyllum aramatum promotes longevity in Caenorhabditis elegans. Exp. Gerontol. 2019, 120, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Howitz, K.T.; Bitterman, K.J.; Cohen, H.Y.; Lamming, D.W.; Lavu, S.; Wood, J.G.; Zipkin, R.E.; Chung, P.; Kisielewski, A.; Zhang, L.L.; et al. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature 2003, 425, 191–196. [Google Scholar] [CrossRef]
- Wood, J.G.; Regina, B.; Lavu, S.; Lamming, D.W.; Lavu, S.; Wood, J.G.; Zipkin, R.E.; Chung, P.; Kisielewski, A.; Zhang, L.L.; et al. Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature 2004, 430, 686–689. [Google Scholar] [CrossRef] [PubMed]
- Stefani, M.; Rigacci, S. Protein Folding and Aggregation into Amyloid: The Interference by Natural Phenolic Compounds. Int. J. Mol. Sci. 2013, 14, 12411–12457. [Google Scholar] [CrossRef] [PubMed]
- Martel, J.; Ojcius, D.M.; Ko, Y.F.; Chang, C.J.; Young, J.D. Antiaging effects of bioactive molecules isolated from plants and fungi. Med. Res. Rev. 2019. [Google Scholar] [CrossRef] [PubMed]
- Ishigaki, Y.; Tanaka, H.; Akama, H.; Ogara, T.; Uwai, K.; Tokuraku, K. A microliter-scale high-throughput screening system with quantum-dot nanoprobes for amyloid-β aggregation inhibitors. PLoS ONE 2013, 8, e72992. [Google Scholar] [CrossRef] [PubMed]
- Santín-Márquez, R.; Alarcón-Aguilar, A.; López-Diazguerrero, N.E.; Chondrogianni, N.; Königsberg, M. Sulforaphane—Role in aging and neurodegeneration. Geroscience 2019. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Zhang, J.; Chang, N. Epigenetic modification of Nrf2 by sulforaphane increases the antioxidative and anti-inflammatory capacity in a cellular model of Alzheimer’s disease. Eur. J. Pharmacol. 2018, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Debernardi, J.M.; Mecchia, M.A.; Vercruyssen, L.; Smaczniak, C.; Kaufmann, K.; Inze, D.; Rodriguez, R.E.; Palatnik, J.F. Post-transcriptional control of GRF transcription factors by microRNA miR396 and GIF co-activator affects leaf size and longevity. Plant J. 2014, 79, 413–426. [Google Scholar] [CrossRef]
- Wang, N.; Liu, J.; Xie, F.; Gao, X.; Ye, J.H.; Sun, L.Y.; Wei, R.; Ai, J. miR-124/ATF-6, a novel lifespan extension pathway of Astragalus polysaccharide in Caenorhabditis elegans. J. Cell Biochem. 2015, 116, 242–251. [Google Scholar] [CrossRef]
- Carra, S.; Alberti, S.; Benesch, J.L.P.; Boelens, W.; Buchner, J.; Carver, J.A.; Cecconi, C.; Ecroyd, H.; Gusev, N.; Hightower, L.E.; et al. Small heat shock proteins: Multifaceted proteins with important implications for life. Cell Stress Chaperones 2019, 24, 295–308. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Surguchov, A.; Emamzadeh, F.N.; Surguchev, A.A. Amyloidosis and Longevity: A Lesson from Plants. Biology 2019, 8, 43. https://doi.org/10.3390/biology8020043
Surguchov A, Emamzadeh FN, Surguchev AA. Amyloidosis and Longevity: A Lesson from Plants. Biology. 2019; 8(2):43. https://doi.org/10.3390/biology8020043
Chicago/Turabian StyleSurguchov, Andrei, Fatemeh Nouri Emamzadeh, and Alexei A. Surguchev. 2019. "Amyloidosis and Longevity: A Lesson from Plants" Biology 8, no. 2: 43. https://doi.org/10.3390/biology8020043
APA StyleSurguchov, A., Emamzadeh, F. N., & Surguchev, A. A. (2019). Amyloidosis and Longevity: A Lesson from Plants. Biology, 8(2), 43. https://doi.org/10.3390/biology8020043





