Episodic Ultradian Events—Ultradian Rhythms
Abstract
1. Introduction
2. Ultradian Rhythms: What Are They?
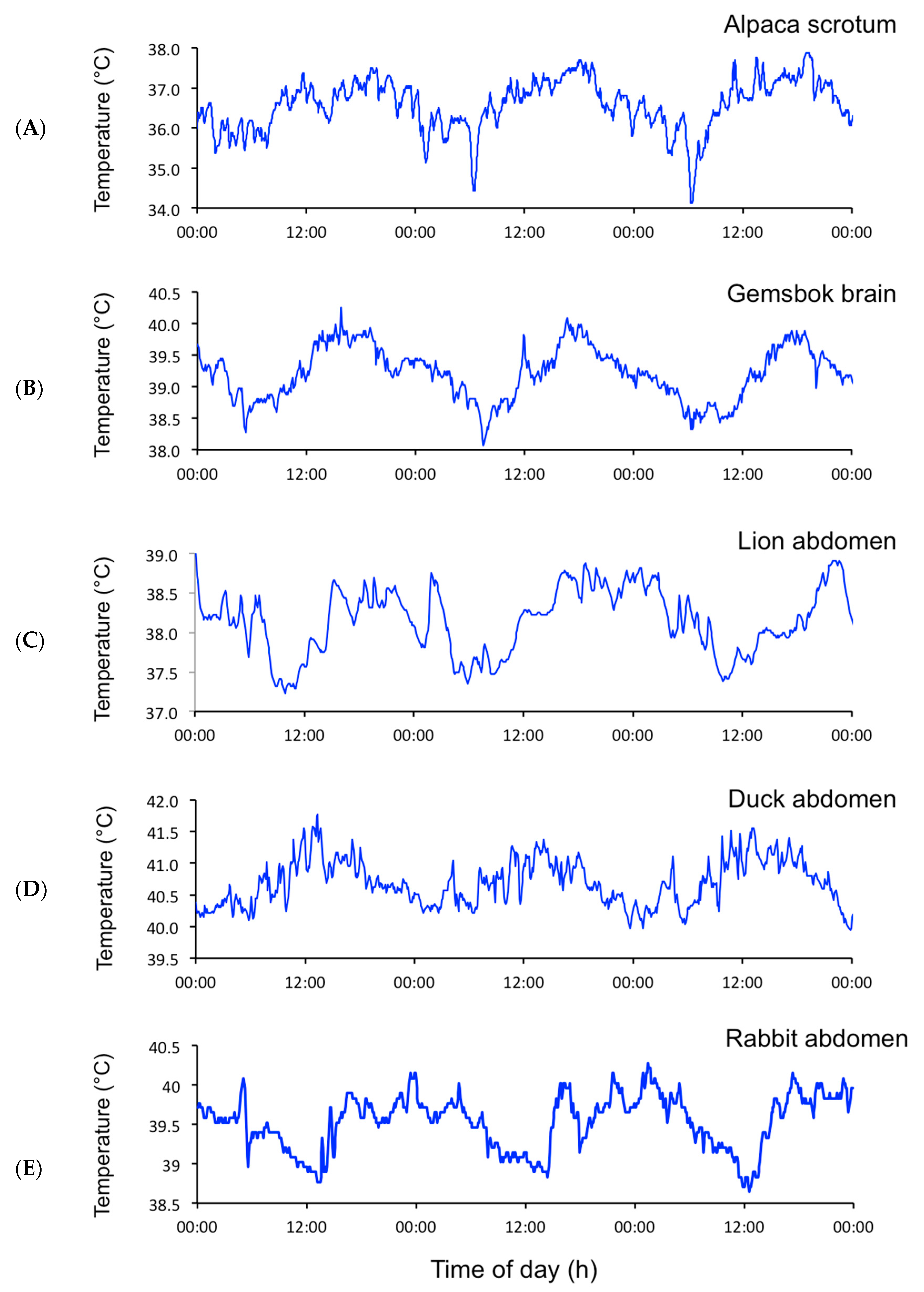
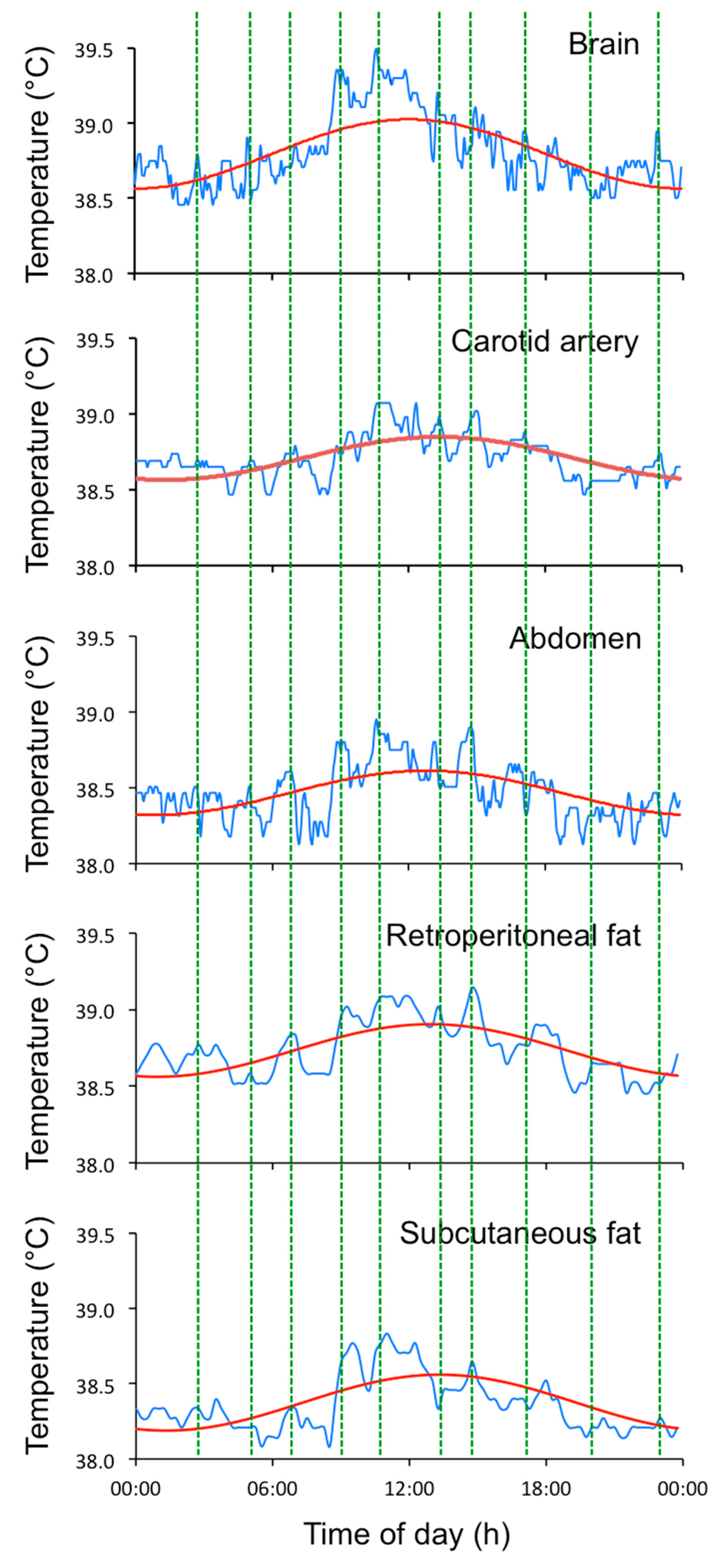
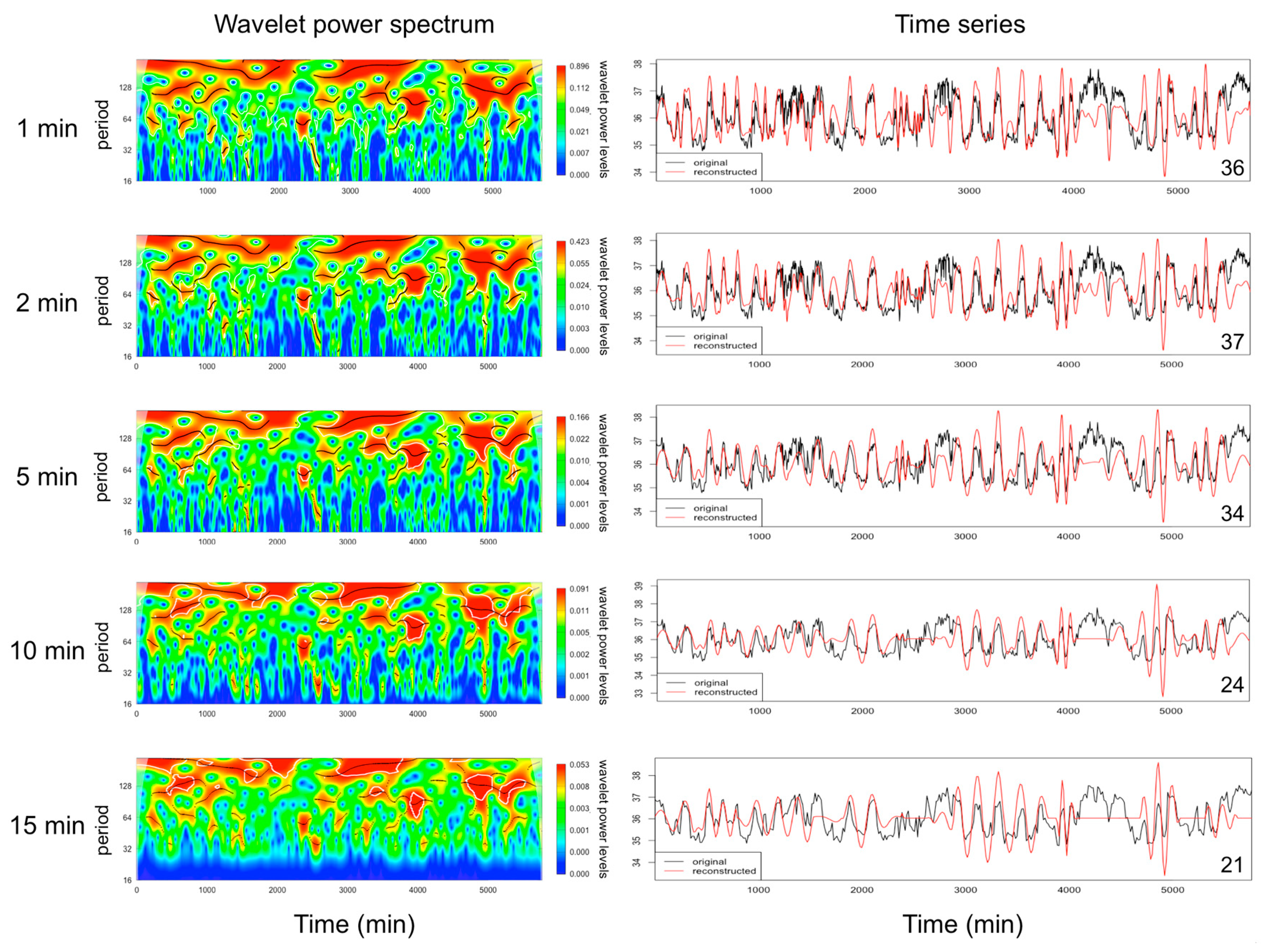
2.1. Data Collection and Analysis of EUEs
3. Origins and Mechanisms Driving EUEs
3.1. Mechanisms of Cellular Ultradian Oscillators
3.2. A Case for Coupling amongst Ultradian Oscillators
3.3. Evidence for the Dopaminergic Ultradian Oscillator
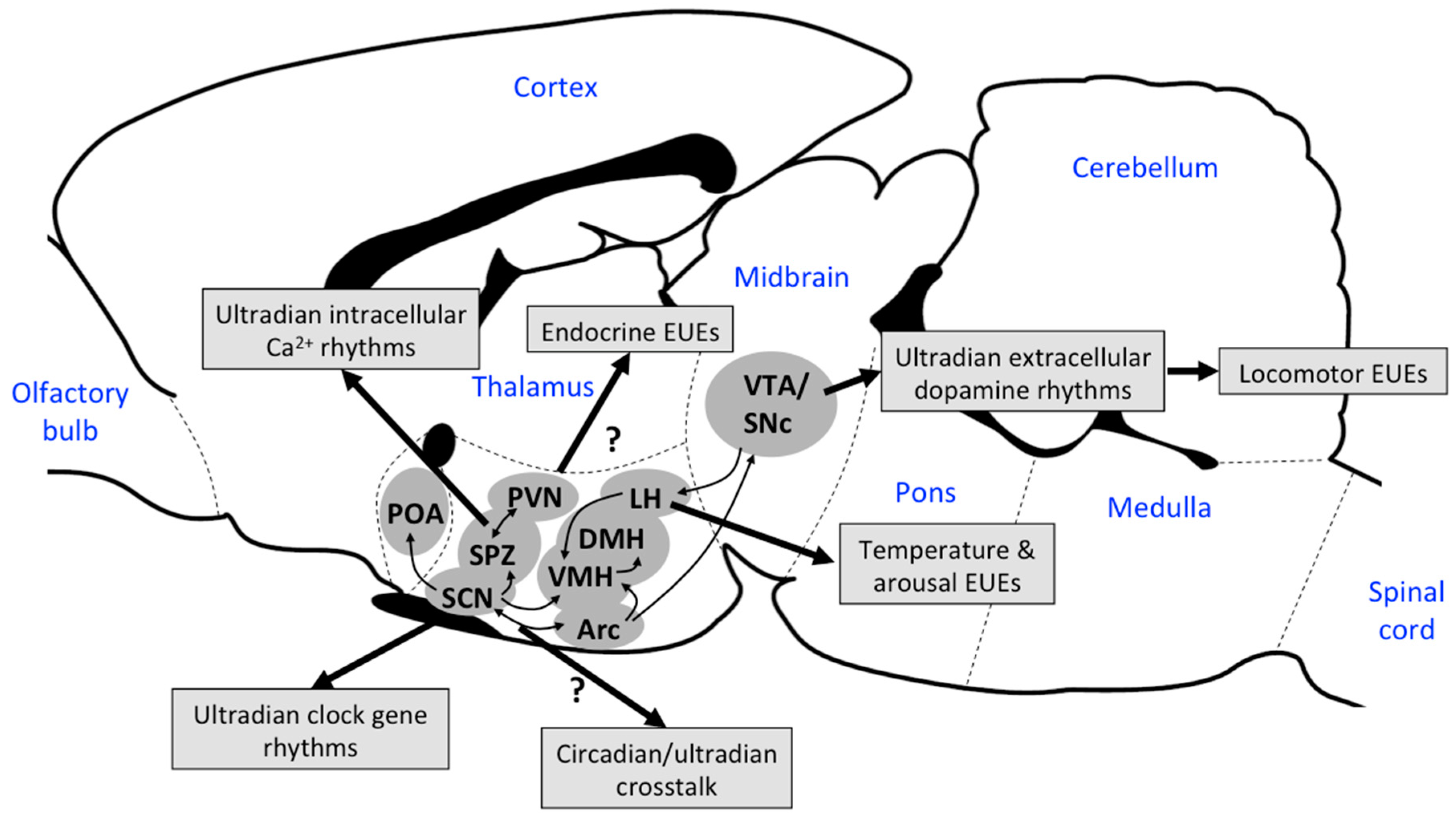
3.4. Other Neuronal Mechanisms Driving EUEs
4. The Role of EUEs
- (1)
- synchronizing compatible processes, and preventing the simultaneous activation of incompatible processes [10];
- (2)
- (3)
- interacting with circadian rhythms.
4.1. EUEs and the Synchronization of Compatible Processes
4.2. Role of EUEs in Preparedness
4.3. EUEs and Circadian Rhythms
5. Conclusions: What Next in the Field of EUEs?
- (1)
- the origin of EUEs and further exploration of the molecular basis of EUEs;
- (2)
- the regulation of EUEs by neuromediators such as serotonin and dopamine, and neurohormones such as orexin and kisspeptin;
- (3)
- the relationship between EUEs and other biological rhythms, such as circadian rhythms;
- (4)
- the biological importance or biological function of EUEs, from energy optimization, to alertness, or possibly a combination of both—a theory that has often been proposed but rarely tested experimentally.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ARC | arcuate nucleus |
| PVN | paraventricular nucleus |
| SCN | suprachiasmatic nucleus |
| SNc | substantia nigra pars compacta |
| SPZ | subparaventricular zone |
| VTA | ventral tegmental area |
References
- Aschoff, J. Biological Rhythms; Plenum Press: New York, NY, USA, 1981; p. 563. [Google Scholar]
- Szymanski, J.S. Aktivität und ruhe bei tieren und menschen. Z. Fjur. Allg. Physiol. 1920, 18, 105–162. [Google Scholar]
- Daan, S.; Aschoff, J. Short-term rhythms in activity. In Biological Rhythms; Aschoff, J., Ed.; Plenum Press: New York, NY, USA, 1981; pp. 491–498. [Google Scholar]
- Acharya, U.R.; Hagiwara, Y.; Deshpande, S.N.; Suren, S.; Koh, J.E.W.; Oh, S.L.; Arunkumar, N.; Ciaccio, E.J.; Lim, C.M. Characterization of focal EEG signals: A review. Future Gener. Comput. Syst. 2019, 91, 290–299. [Google Scholar] [CrossRef]
- Gambarotta, N.; Aletti, F.; Baselli, G.; Ferrario, M. A review of methods for the signal quality assessment to improve reliability of heart rate and blood pressures derived parameters. Med. Biol. Eng. Comput. 2016, 54, 1025–1035. [Google Scholar] [CrossRef]
- Kippert, F.; Hunt, P. Ultradian clocks in eukaryotic microbes: From behavioural observation to functional genomics. BioEssays 2000, 22, 16–22. [Google Scholar] [CrossRef]
- Branicky, R.; Hekimi, S. What keeps C. elegans regular: The genetics of defecation. Trends Genet. 2006, 22, 571–579. [Google Scholar] [CrossRef]
- Kadono, H.; Usami, E. Ultradian rhythm of chicken body temperature under continuous light. Nihon Juigaku Zasshi 1983, 45, 401–405. [Google Scholar] [CrossRef] [PubMed]
- Brodsky, V. Circahoralian (Ultradian) metabolic rhythms. Biochemistry 2014, 79, 483–495. [Google Scholar] [CrossRef]
- Lloyd, D.; Stupfel, M. The occurrence and functions of ultradian rhythms. Biol. Rev. 1991, 66, 275–299. [Google Scholar] [CrossRef]
- Blessing, W.; Ootsuka, Y. Timing of activities of daily life is jaggy: How episodic ultradian changes in body and brain temperature are integrated into this process. Temperature 2016, 3, 371–383. [Google Scholar] [CrossRef]
- Maloney, S.K.; Blache, D.; The University of Western Australia, Crawley, Western Australia, Australia. Unpublished data. 2019.
- Fuller, A.; Brain Function Research Group, School of Physiology, Medical School, University of the Witwatersrand, South Africa. Unpublished data. 2019.
- Lavie, P. Nonstationarity in human perceptual ultradian rhythms. Chronobiologia 1977, 4, 38–48. [Google Scholar] [PubMed]
- Dowse, H.B. Mid-range ultrdian rhythms in Drosophila and the circadian clock problem. In Ultradian Rhythms from Molecules to Mind; Lloyd, D., Rossi, E.L., Eds.; Springer: New York, NY, USA, 2008; pp. 175–199. [Google Scholar]
- Kleitman, N. Basic Rest-Activity Cycle—22 Years Later. Sleep 1982, 5, 311–317. [Google Scholar] [CrossRef]
- Nelson, W.; Liang Tong, Y.; Lee, J.; Halberg, F. Methods for cosinor-rhythmometry. Chronobiologia 1979, 6, 305–323. [Google Scholar] [PubMed]
- Zhang, S.; Blache, D.; Blackberry, M.A.; Martin, G.B. Dynamics of the responses in secretion of LH, leptin and insulin following an acute increase in nutrition in mature male sheep. Reprod. Fertil. Dev. 2004, 16, 823–829. [Google Scholar] [CrossRef]
- Zhang, S.; Blache, D.; Blackberry, M.A.; Martin, G.B. Body reserves affect the reproductive endocrine responses to an acute change in nutrition in mature male sheep. Anim. Reprod. Sci. 2005, 88, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Martin, G.B.; Thomas, G.B.; Terqui, M.; Warner, P. Pulsatile LH secretion during the preovulatory surge in the ewe: Experimental observations and theoretical considerations. Reprod. Nutr. Dév. 1987, 27, 1023–1040. [Google Scholar] [CrossRef] [PubMed]
- Blache, D.; Martin, G.B. Neural and endocrine mechanisms underlying the synchronicity of sexual behaviour and ovulation in the sheep. Oxf. Rev. Reprod. Biol. 1995, 17, 205–254. [Google Scholar]
- Clarke, I.J. Exactitude in the relationship between GnRH and LH secretion. In Modes of Action of GnRH and GnRH Analogs; Crowley, W.F., Conn, P.M., Eds.; Springer: Berlin, Germany, 1992; pp. 179–192. [Google Scholar]
- Rietema, S.E.; Blackberry, M.A.; Maloney, S.K.; Martin, G.B.; Hawken, P.A.R.; Blache, D. Twenty four-hour profiles of metabolic and stress hormones in sheep selected for a calm or nervous temperament. Dom. Anim. Endocr. 2015, 53, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Leise, T.L. Wavelet analysis of circadian and ultradian behavioral rhythms. J. Circadian Rhythm. 2013, 11, 5. [Google Scholar] [CrossRef] [PubMed]
- Merriam, G.R.; Wachter, K.W. Algorithms for the study of episodic hormone secretion. Am. J. Physiol. 1982, 243, E310–E318. [Google Scholar] [CrossRef] [PubMed]
- Veldhuis, J.D.; Johnson, M.L. Cluster analysis: A simple, versatile, and robust algorithm for endocrine pulse detection. Am. J. Physiol. 1986, 250, E486–E493. [Google Scholar] [CrossRef] [PubMed]
- Genazzani, A.D.; Guardabasso, V.; Petraglia, F.; Genazzani, A.R. Specific concordance index defines the physiological lag between LH and progesterone in women during the midluteal phase of the menstrual cycle. Gynecol. Endocr. 1991, 5, 175–184. [Google Scholar] [CrossRef]
- Van Cauter, E. Estimating false-positive and false-negative errors in analyses of hormonal pulsatility. Am. J. Physiol. 1988, 254, E786–E794. [Google Scholar] [CrossRef] [PubMed]
- Keenan, D.M.; Veldhuis, J.D. Stochastic model of admixed basal and pulsatile hormone secretion as modulated by a deterministic oscillator. Am. J. Physiol. 1997, 42, R1182–R1192. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, J.; Signoret, J.P. Controle de la decharge de LH dans le sang par la progesterone et le benzoate d’oestradiol chez la brebis castree. C. R. Séances Acad. Sci. III 1969, 269, 2595–2598. [Google Scholar]
- Refinetti, R. Non-stationary time series and the robustness of circadian rhythms. J. Therm. Biol. 2004, 227, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Le Fur, I.; Reinberg, A.; Lopez, S.; Morizot, F.; Mechkouri, M.; Tschachler, E. Analysis of circadian and ultradian rhythms of skin surface properties of face and forearm of healthy women. J. Investig. Dermatol. 2001, 117, 718–724. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.N.; Alwan, A.; Kossan, G.; Cody, M.L.; Taylor, C.E. Dynamic time warping and sparse representation classification for birdsong phrase classification using limited training data. JASA 2015, 137, 1069–1080. [Google Scholar] [CrossRef] [PubMed]
- Ono, D.; Honma, K.-I.; Honma, S. Circadian and ultradian rhythms of clock gene expression in the suprachiasmatic nucleus of freely moving mice. Sci. Rep. 2015, 5, 12310. [Google Scholar] [CrossRef]
- Gerkema, M.P.; Groos, G.A.; Daan, S. Differential elimination of circadian and ultradian rhythmicity by hypothalamic lesions in the Common Vole, Microtus arvalis. J. Biol. Rhythm. 1990, 5, 81–95. [Google Scholar] [CrossRef]
- Refinetti, R.; Kaufman, C.M.; Menaker, M. Complete suprachiasmatic lesions eliminate circadian rhythmicity of body temperature and locomotor activity in golden hamsters. J. Comp. Physiol. A 1994, 175, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Vitaterna, M.H.; King, D.P.; Chang, A.-M.; Kornhauser, J.M.; Lowrey, P.L.; McDonald, J.D.; Dove, W.F.; Pinto, L.H.; Turek, F.W.; Takahashi, J.S. Mutagenesis and mapping of a mouse gene, Clock, essential for circadian behavior. Science 1994, 264, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Bunger, M.K.; Wilsbacher, L.D.; Moran, S.M.; Clendenin, C.; Radcliffe, L.A.; Hogenesch, J.B.; Simon, M.C.; Takahashi, J.S.; Bradfield, C.A. Mop3 is an essential component of the master circadian pacemaker in mammals. Cell 2000, 103, 1009–1017. [Google Scholar] [CrossRef]
- Baker, F.; Angara, C.; Szymusiak, R.; McGinty, D. Persistence of sleep-temperature coupling after suprachiasmatic nuclei lesions in rats. Am. J. Physiol. 2005, 289, R827–R838. [Google Scholar] [CrossRef] [PubMed]
- Feillet, C.A.; Ripperger, J.A.; Magnone, M.C.; Dulloo, A.; Albrecht, U.; Challet, E. Lack of food anticipation in Per2 mutant mice. Curr. Biol. 2006, 16, 2016–2022. [Google Scholar] [CrossRef]
- McDearmon, E.L.; Patel, K.N.; Ko, C.H.; Walisser, J.A.; Schook, A.C.; Chong, J.L.; Wilsbacher, L.D.; Song, E.J.; Hong, H.-K.; Bradfield, C.A. Dissecting the functions of the mammalian clock protein BMAL1 by tissue-specific rescue in mice. Science 2006, 314, 1304–1308. [Google Scholar] [CrossRef]
- Waite, E.J.; McKenna, M.; Kershaw, Y.; Walker, J.J.; Cho, K.; Piggins, H.D.; Lightman, S.L. Ultradian corticosterone secretion is maintained in the absence of circadian cues. Eur. J. Neurosci. 2012, 36, 3142–3150. [Google Scholar] [CrossRef]
- Seki, Y.; Tanimura, T. Ultradian rhythm unmasked in the Pdf clock mutant of Drosophila. J. Biosci. 2014, 39, 585–594. [Google Scholar] [CrossRef]
- Honma, K.I.; Hiroshige, T. Simultaneous determination of circadian rhythms of locomotor activity and body temperature in the rat. Jpn. J. Physiol. 1978, 28, 159–169. [Google Scholar] [CrossRef]
- Gerkema, M.P.; van der Leest, F. Ongoing ultradian activity rhythms in the common vole, Microtus arvalis, during deprivations of food, water and rest. J. Comp. Physiol. A 1991, 168, 591–597. [Google Scholar] [CrossRef]
- Stephenson, R.; Lim, J.; Famina, S.; Caron, A.M.; Dowse, H.B. Sleep-wake behavior in the rat: Ultradian rhythms in a light-dark cycle and continuous bright light. J. Biol. Rhythm. 2012, 27, 490–501. [Google Scholar] [CrossRef]
- Isomura, A.; Kageyama, R. Ultradian oscillations and pulses: Coordinating cellular responses and cell fate decisions. Development 2014, 141, 3627–3636. [Google Scholar] [CrossRef]
- Hirata, H.; Yoshiura, S.; Ohtsuka, T.; Bessho, Y.; Harada, T.; Yoshikawa, K.; Kageyama, R. Oscillatory expression of the bHLH factor Hes1 regulated by a negative feedback loop. Science 2002, 298, 840–843. [Google Scholar] [CrossRef]
- Takebayashi, K.; Sasai, Y.; Sakai, Y.; Watanabe, T.; Nakanishi, S.; Kageyama, R. Structure, chromosomal locus, and promoter analysis of the gene encoding the mouse helix-loop-helix factor HES-1. Negative autoregulation through the multiple N box elements. J. Biol. Chem. 1994, 269, 5150–5156. [Google Scholar]
- Hogenesch, J.B.; Gu, Y.-Z.; Jain, S.; Bradfield, C.A. The basic-helix–loop–helix-PAS orphan MOP3 forms transcriptionally active complexes with circadian and hypoxia factors. Proc. Natl. Acad. Sci. USA 1998, 95, 5474–5479. [Google Scholar] [CrossRef]
- Shearman, L.P.; Sriram, S.; Weaver, D.R.; Maywood, E.S.; Chaves, I.; Zheng, B.; Kume, K.; Lee, C.C.; Hastings, M.H.; Reppert, S.M. Interacting molecular loops in the mammalian circadian clock. Science 2000, 288, 1013–1019. [Google Scholar] [CrossRef]
- Hirata, H.; Bessho, Y.; Kokubu, H.; Masamizu, Y.; Yamada, S.; Lewis, J.; Kageyama, R. Instability of Hes7 protein is crucial for the somite segmentation clock. Nat. Genet. 2004, 36, 750–754. [Google Scholar] [CrossRef]
- Shimojo, H.; Ohtsuka, T.; Kageyama, R. Oscillations in notch signaling regulate maintenance of neural progenitors. Neuron 2008, 58, 52–64. [Google Scholar] [CrossRef]
- Kobayashi, T.; Mizuno, H.; Imayoshi, I.; Furusawa, C.; Shirahige, K.; Kageyama, R. The cyclic gene Hes1 contributes to diverse differentiation responses of embryonic stem cells. Genes Dev. 2009, 23, 1870–1875. [Google Scholar] [CrossRef]
- Yoshiura, S.; Ohtsuka, T.; Takenaka, Y.; Nagahara, H.; Yoshikawa, K.; Kageyama, R. Ultradian oscillations of Stat, Smad, and Hes1 expression in response to serum. Proc. Natl. Acad. Sci. USA 2007, 104, 11292–11297. [Google Scholar] [CrossRef]
- Santorelli, M.; Perna, D.; Isomura, A.; Garzilli, I.; Annunziata, F.; Postiglione, L.; Tumaini, B.; Kageyama, R.; di Bernardo, D. Reconstitution of an ultradian oscillator in mammalian cells by a synthetic biology approach. ACS Synth. Biol. 2018, 7, 1447–1455. [Google Scholar] [CrossRef]
- Mohawk, J.A.; Green, C.B.; Takahashi, J.S. Central and peripheral circadian clocks in mammals. Ann. Rev. Neurosci. 2012, 35, 445–462. [Google Scholar] [CrossRef] [PubMed]
- Masamizu, Y.; Ohtsuka, T.; Takashima, Y.; Nagahara, H.; Takenaka, Y.; Yoshikawa, K.; Okamura, H.; Kageyama, R. Real-time imaging of the somite segmentation clock: Revelation of unstable oscillators in the individual presomitic mesoderm cells. Proc. Natl. Acad. Sci. USA 2006, 103, 1313–1318. [Google Scholar] [CrossRef] [PubMed]
- Lim, C.; Allada, R. Emerging roles for post-transcriptional regulation in circadian clocks. Nat. Neurosci. 2013, 16, 1544–1550. [Google Scholar] [CrossRef]
- Goodfellow, M.; Phillips, N.E.; Manning, C.; Galla, T.; Papalopulu, N. microRNA input into a neural ultradian oscillator controls emergence and timing of alternative cell states. Nat. Commun. 2014, 5, 3399. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Fiskum, G.; Schubert, D. Generation of reactive oxygen species by the mitochondrial electron transport chain. J. Neurochem. 2002, 80, 780–787. [Google Scholar] [CrossRef]
- Sumimoto, H. Structure, regulation and evolution of Nox-family NADPH oxidases that produce reactive oxygen species. FEBS 2008, 275, 3249–3277. [Google Scholar] [CrossRef]
- Ventre, S.; Indrieri, A.; Fracassi, C.; Franco, B.; Conte, I.; Cardone, L.; di Bernardo, D. Metabolic regulation of the ultradian oscillator Hes1 by reactive oxygen species. J. Mol. Biol. 2015, 427, 1887–1902. [Google Scholar] [CrossRef]
- Ashall, L.; Horton, C.A.; Nelson, D.E.; Paszek, P.; Harper, C.V.; Sillitoe, K.; Ryan, S.; Spiller, D.G.; Unitt, J.F.; Broomhead, D.S. Pulsatile stimulation determines timing and specificity of NF-κB-dependent transcription. Science 2009, 324, 242–246. [Google Scholar] [CrossRef]
- Lahav, G.; Rosenfeld, N.; Sigal, A.; Geva-Zatorsky, N.; Levine, A.J.; Elowitz, M.B.; Alon, U. Dynamics of the p53-Mdm2 feedback loop in individual cells. Nat. Genet. 2004, 36, 147. [Google Scholar] [CrossRef]
- Shankaran, H.; Ippolito, D.L.; Chrisler, W.B.; Resat, H.; Bollinger, N.; Opresko, L.K.; Wiley, H.S. Rapid and sustained nuclear–cytoplasmic ERK oscillations induced by epidermal growth factor. Mol. Syst. Biol. 2009, 5, 332. [Google Scholar] [CrossRef]
- Waters, K.M.; Cummings, B.S.; Shankaran, H.; Scholpa, N.E.; Weber, T.J. ERK oscillation-dependent gene expression patterns and deregulation by stress response. Chem. Res. Toxicol. 2014, 27, 1496–1503. [Google Scholar] [CrossRef]
- Nakayama, K.; Satoh, T.; Igari, A.; Kageyama, R.; Nishida, E. FGF induces oscillations of Hes1 expression and Ras/ERK activation. Curr. Biol. 2008, 18, R332–R334. [Google Scholar] [CrossRef] [PubMed]
- Varusai, T.M.; Kolch, W.; Kholodenko, B.N.; Nguyen, L.K. Protein–protein interactions generate hidden feedback and feed-forward loops to trigger bistable switches, oscillations and biphasic dose–responses. Mol. BioSyst. 2015, 11, 2750–2762. [Google Scholar] [CrossRef] [PubMed]
- Arkun, Y.; Yasemi, M. Dynamics and control of the ERK signaling pathway: Sensitivity, bistability, and oscillations. PLoS ONE 2018, 13, e0195513. [Google Scholar] [CrossRef]
- Ootsuka, Y.; de Menezes, R.C.; Zaretsky, D.V.; Alimoradian, A.; Hunt, J.; Stefanidis, A.; Oldfield, B.J.; Blessing, W.W. Brown adipose tissue thermogenesis heats brain and body as part of the brain-coordinated ultradian basic rest-activity cycle. Neuroscience 2009, 164, 849–861. [Google Scholar] [CrossRef]
- Grant, A.D.; Wilsterman, K.; Smarr, B.L.; Kriegsfeld, L.J. Evidence for a coupled oscillator model of endocrine ultradian rhythms. J Biol. Rhythm. 2018, 33, 475–496. [Google Scholar] [CrossRef]
- Hampl, R.; Starka, L.; Jansky, L. Steroids and thermogenesis. Physiol. Res. 2006, 55, 123. [Google Scholar]
- Ramage, L.E.; Akyol, M.; Fletcher, A.M.; Forsythe, J.; Nixon, M.; Carter, R.N.; van Beek, E.J.; Morton, N.M.; Walker, B.R.; Stimson, R.H. Glucocorticoids acutely increase brown adipose tissue activity in humans, revealing species-specific differences in UCP-1 regulation. Cell Metabol. 2016, 24, 130–141. [Google Scholar] [CrossRef]
- Smarr, B.L.; Burnett, D.C.; Mesri, S.M.; Pister, K.S.; Kriegsfeld, L.J. A wearable sensor system with circadian rhythm stability estimation for prototyping biomedical studies. IEEE Trans. Affect. Comput. 2016, 7, 220–230. [Google Scholar] [CrossRef]
- Sanchez-Alavez, M.; Alboni, S.; Conti, B. Sex-and age-specific differences in core body temperature of C57Bl/6 mice. Age 2011, 33, 89–99. [Google Scholar] [CrossRef]
- Rance, N.E.; Dacks, P.A.; Mittelman-Smith, M.A.; Romanovsky, A.A.; Krajewski-Hall, S.J. Modulation of body temperature and LH secretion by hypothalamic KNDy (kisspeptin, neurokinin B and dynorphin) neurons: A novel hypothesis on the mechanism of hot flushes. Front. Neuroendocr. 2013, 34, 211–227. [Google Scholar] [CrossRef] [PubMed]
- Mullur, R.; Liu, Y.-Y.; Brent, G.A. Thyroid hormone regulation of metabolism. Physiol. Rev. 2014, 94, 355–382. [Google Scholar] [CrossRef] [PubMed]
- Maloney, S.K.; Withers, P.C.; The University of Western Australia, Crawley, Western Australia, Australia. Unpublished data. 2019.
- Blessing, W.; Mohammed, M.; Ootsuka, Y. Brown adipose tissue thermogenesis, the basic rest–activity cycle, meal initiation, and bodily homeostasis in rats. Physiol. Behav. 2013, 121, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates; Academic Press: Sydney, Australia, 1982. [Google Scholar]
- Blum, I.D.; Zhu, L.; Moquin, L.; Kokoeva, M.V.; Gratton, A.; Giros, B.; Storch, K.-F. A highly tunable dopaminergic oscillator generates ultradian rhythms of behavioral arousal. eLife 2014, 3, e05105. [Google Scholar] [CrossRef]
- Miyata, K.; Kuwaki, T.; Ootsuka, Y. The integrated ultradian organization of behavior and physiology in mice and the contribution of orexin to the ultradian patterning. Neuroscience 2016, 334, 119–133. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-E.; Enoki, R.; Oda, Y.; Huang, Z.-L.; Honma, K.-I.; Honma, S. Ultradian calcium rhythms in the paraventricular nucleus and subparaventricular zone in the hypothalamus. Proc. Natl. Acad. Sci. USA 2018, 115, E9469–E9478. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.E.; Basheer, R.; McKenna, J.T.; Strecker, R.E.; McCarley, R.W. Control of sleep and wakefulness. Physiol. Rev. 2012, 92, 1087–1187. [Google Scholar] [CrossRef]
- Zhou, Q.-Y.; Palmiter, R.D. Dopamine-deficient mice are severely hypoactive, adipsic, and aphagic. Cell 1995, 83, 1197–1209. [Google Scholar] [CrossRef]
- Giros, B.; Jaber, M.; Jones, S.R.; Wightman, R.M.; Caron, M.G. Hyperlocomotion and indifference to cocaine and amphetamine in mice lacking the dopamine transporter. Nature 1996, 379, 606. [Google Scholar] [CrossRef]
- Gainetdinov, R.R.; Jones, S.R.; Fumagalli, F.; Wightman, R.M.; Caron, M.G. Re-evaluation of the role of the dopamine transporter in dopamine system homeostasis. Brain Res. Rev. 1998, 26, 148–153. [Google Scholar] [CrossRef]
- Morice, E.; Denis, C.; Giros, B.; Nosten-Bertrand, M. Phenotypic expression of the targeted null-mutation in the dopamine transporter gene varies as a function of the genetic background. Eur. J. Neurosci. 2004, 20, 120–126. [Google Scholar] [CrossRef]
- Mohawk, J.A.; Baer, M.L.; Menaker, M. The methamphetamine-sensitive circadian oscillator does not employ canonical clock genes. Proc. Natl. Acad. Sci. USA 2009, 106, 3519–3524. [Google Scholar] [CrossRef]
- Greenwell, T.N.; Zangen, A.; Martin-Schild, S.; Wise, R.A.; Zadina, J.E. Endomorphin-1 and-2 immunoreactive cells in the hypothalamus are labeled by fluoro-gold injections to the ventral tegmental area. J. Comp. Neurol. 2002, 454, 320–328. [Google Scholar] [CrossRef]
- Gerkema, M.P.; Daan, S.; Wilbrink, M.; Hop, M.W.; van der Leest, F. Phase control of ultradian feeding rhythms in the common vole (Microtus arvalis): The roles of light and the circadian system. J Biol. Rhythm. 1993, 8, 151–171. [Google Scholar] [CrossRef]
- Yoshida, K.; McCormack, S.; España, R.A.; Crocker, A.; Scammell, T.E. Afferents to the orexin neurons of the rat brain. J. Comp. Neurol. 2006, 494, 845–861. [Google Scholar] [CrossRef]
- Yamanaka, A.; Muraki, Y.; Tsujino, N.; Goto, K.; Sakurai, T. Regulation of orexin neurons by the monoaminergic and cholinergic systems. Biochem. Biophys. Res. Commun. 2003, 303, 120–129. [Google Scholar] [CrossRef]
- Mohammed, M.; Ootsuka, Y.; Yanagisawa, M.; Blessing, W. Reduced brown adipose tissue thermogenesis during environmental interactions in transgenic rats with ataxin-3-mediated ablation of hypothalamic orexin neurons. Am. J. Physiol. 2014, 307, R978–R989. [Google Scholar] [CrossRef]
- Rivkees, S.A.; Mayes, L.; Jacobs, H.; Gross, I. Rest-activity patterns of premature infants are regulated by cycled lighting. Pediatrics 2004, 113, 833–839. [Google Scholar] [CrossRef]
- Watabe-Uchida, M.; Zhu, L.; Ogawa, S.K.; Vamanrao, A.; Uchida, N. Whole-brain mapping of direct inputs to midbrain dopamine neurons. Neuron 2012, 74, 858–873. [Google Scholar] [CrossRef]
- Rusak, B. The role of the suprachiasmatic nuclei in the generation of circadian rhythms in the golden hamster, Mesocricetus auratus. J. Comp. Physiol. 1977, 118, 145–164. [Google Scholar] [CrossRef]
- Ruby, N.F.; Ibuka, N.; Barnes, B.M.; Zucker, I. Suprachiasmatic nuclei influence torpor and circadian temperature rhythms in hamsters. Am. J. Physiol. 1989, 257, R210–R215. [Google Scholar] [CrossRef] [PubMed]
- Buijs, F.N.; Guzmán-Ruiz, M.; León-Mercado, L.; Basualdo, M.C.; Escobar, C.; Kalsbeek, A.; Buijs, R.M. Suprachiasmatic nucleus interaction with the arcuate nucleus; essential for organizing physiological rhythms. eNeuro 2017, 4. [Google Scholar] [CrossRef] [PubMed]
- Prendergast, B.J.; Zucker, I. Ultradian rhythms in mammalian physiology and behavior. Curr. Opin. Neurobiol. 2016, 40, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Buhr, E.D.; Yoo, S.-H.; Takahashi, J.S. Temperature as a universal resetting cue for mammalian circadian oscillators. Science 2010, 330, 379–385. [Google Scholar] [CrossRef]
- Ulrich-Lai, Y.M.; Herman, J.P. Neural regulation of endocrine and autonomic stress responses. Nat. Rev. Neurosci. 2009, 10, 397–409. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Zhang, Y.-H.; Chou, T.C.; Gaus, S.E.; Elmquist, J.K.; Shiromani, P.; Saper, C.B. Contrasting effects of ibotenate lesions of the paraventricular nucleus and subparaventricular zone on sleep–wake cycle and temperature regulation. J. Neurosci. 2001, 21, 4864–4874. [Google Scholar] [CrossRef] [PubMed]
- Vujovic, N.; Gooley, J.J.; Jhou, T.C.; Saper, C.B. Projections from the subparaventricular zone define four channels of output from the circadian timing system. J. Comp. Neurol. 2015, 523, 2714–2737. [Google Scholar] [CrossRef] [PubMed]
- Bernard, C. La fixité du milieu intérieur est la condition de la vie libre. In Leçons sur les Phénomènes de la vie Communs aux Animaux et aux Végétaux; Bernard, C., Ed.; Baillière: Paris, France, 1878; p. 246. [Google Scholar]
- Cannon, W.B. Organization for physiological homeostasis. Physiol. Rev. 1929, 9, 399–431. [Google Scholar] [CrossRef]
- Darwin, C. On the Origin of Species by Means of Natural Selection, or, the Preservation of Favoured Races in the Struggle of Life, 4th ed.; John Murray: London, UK, 1866; p. 593. [Google Scholar]
- Schibler, U. The mammalian circadian timekeeping system. In Ultradian Rhythms from Molecules to Mind: A New Vision of Life; Lloyd, D., Rossi, E.L., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 261–279. [Google Scholar]
- Daan, S. Adaptive daily strategies in behavior. In Biological Rhythms; Aschoff, J., Ed.; Plenum Press: New York, NY, USA, 1981; pp. 275–298. [Google Scholar]
- Ballesta, A.; Innominato, P.F.; Dallmann, R.; Rand, D.A.; Lévi, F.A. Systems chronotherapeutics. Pharmacol. Rev. 2017, 69, 161–199. [Google Scholar] [CrossRef]
- Brodsky, V.Y.; Lloyd, D. Self-organized intracellular ultradian rhythms provide direct cell-cell communication. In Ultradian Rhythms from Molecules to Mind: A New Vision of Life; Lloyd, D., Rossi, E.L., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 85–104. [Google Scholar]
- Meyer, C.W.; Blessing, W.; Heldmaier, G. Ultradian episodes of thermogenesis in mammals: Implications for the timing of torpor entry and arousal. In Living in a Seasonal World: Thermoregulatory and Metabolic Adaptations; Ruf, T., Bieber, C., Arnold, W., Millesi, E., Eds.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 219–229. [Google Scholar]
- Blache, D.; Zhang, S.; Martin, G.B. Dynamic and integrative aspects of the regulation of reproduction by metabolic status in male sheep. Reprod. Nut. Dev. 2006, 46, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Gerkema, M.P. Ultradian rhytms. In Biological Rhythms; Kumar, V., Ed.; Narosa Publishing House: New Delhi, India, 2002; pp. 207–215. [Google Scholar]
- Tattersall, G.J.; Roussel, D.; Voituron, Y.; Teulier, L. Novel energy-saving strategies to multiple stressors in birds: The ultradian regulation of body temperature. Proc. R. Soc. B Biol. Sci. 2016, 283, 20161551. [Google Scholar] [CrossRef]
- Kontos, A.; de Menezes, R.C.; Ootsuka, Y.; Blessing, W. Brown adipose tissue thermogenesis precedes food intake in genetically obese Zucker (fa/fa) rats. Physiol. Behav.r 2013, 118, 129–137. [Google Scholar] [CrossRef]
- Blessing, W.; Mohammed, M.; Ootsuka, Y. Heating and eating: Brown adipose tissue thermogenesis precedes food ingestion as part of the ultradian basic rest–activity cycle in rats. Physiol. Behav. 2012, 105, 966–974. [Google Scholar] [CrossRef]
- Gerkema, M.P. Biological rhytms: Mecahnism and adaptive values. In Rhythms ib Fishes; Ali, M.A., Ed.; Plenum Press: New York, NY, USA, 1992; pp. 27–37. [Google Scholar]
- Pathé, M. Surviving Stalking; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Aschoff, J.; Gerkema, M.P. On Diversity and Uniformity of Ultradian Rhythms. Exp. Brain Res. 1985, 12 (Suppl.), 321–334. [Google Scholar]
- Lumineau, S.; Guyomarc’h, C.; Richard, J.-P. Ultradian rhythm of activity in Japanese quail groups under semi-natural conditions during ontogeny: Functional aspects and relation to circadian rhythm. Biol. Rhythm. Res. 2001, 32, 373–400. [Google Scholar] [CrossRef]
- Maloney, S.K.; Goh, G.; Fuller, A.; Vesterdorf, K.; Blache, D. Amplitude of the circadian rhythm of temperature in homeotherms. Pers. Agric. Vet. Sci. Nutr. Nat. Res. 2019. accepted. [Google Scholar]
- Johnson, H.E.; Toettcher, J.E. Illuminating developmental biology with cellular optogenetics. Curr. Opin. Biotech. 2018, 52, 42–48. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goh, G.H.; Maloney, S.K.; Mark, P.J.; Blache, D. Episodic Ultradian Events—Ultradian Rhythms. Biology 2019, 8, 15. https://doi.org/10.3390/biology8010015
Goh GH, Maloney SK, Mark PJ, Blache D. Episodic Ultradian Events—Ultradian Rhythms. Biology. 2019; 8(1):15. https://doi.org/10.3390/biology8010015
Chicago/Turabian StyleGoh, Grace H., Shane K. Maloney, Peter J. Mark, and Dominique Blache. 2019. "Episodic Ultradian Events—Ultradian Rhythms" Biology 8, no. 1: 15. https://doi.org/10.3390/biology8010015
APA StyleGoh, G. H., Maloney, S. K., Mark, P. J., & Blache, D. (2019). Episodic Ultradian Events—Ultradian Rhythms. Biology, 8(1), 15. https://doi.org/10.3390/biology8010015




