Quercetin: A Bioactive Compound Imparting Cardiovascular and Neuroprotective Benefits: Scope for Exploring Fresh Produce, Their Wastes, and By-Products
Abstract
Simple Summary
Abstract
1. Introduction
2. Discussion
2.1. Quercetin as a Cardioprotective Agent
2.2. Quercetin as a Neuroprotective Agent
3. Quercetin from Food Industry Wastes and By-Products
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kelly, G.S. Quercetin. Altern. Med. Rev. 2011, 16, 172–195. [Google Scholar]
- Wang, G.; Li, Y.; Lei, C.; Lei, X.; Zhu, X.; Yang, L.; Zhang, R. Quercetin exerts antidepressant and cardioprotective effects in estrogen receptor α-deficient female mice via BDNF-AKT/ERK1/2 signaling. J. Steroid Biochem. Mol. Biol. 2021, 206, 105795. [Google Scholar] [CrossRef] [PubMed]
- Sengul, E.; Gelen, V.; Gedikli, S. Cardioprotective activities of quercetin and rutin in sprague dawley rats treated with 5-fluorouracil. J. Anim. Plant Sci. 2021, 31, 423–431. [Google Scholar]
- Lesjak, M.; Beara, I.; Simin, N.; Pintać, D.; Majkić, T.; Bekvalac, K.; Orčić, D.; Mimica-Dukić, N. Antioxidant and anti-inflammatory activities of quercetin and its derivatives. J. Funct. Foods 2018, 40, 68–75. [Google Scholar] [CrossRef]
- Davis, J.M.; Murphy, A.; Carmichael, M.D.; Davis, B. Quercetin increases brain and muscle mitochondrial biogenesis and exercise tolerance. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R1071–R1077. [Google Scholar] [CrossRef]
- Saeedi-Boroujeni, A.; Mahmoudian-Sani, M.-R. Anti-inflammatory potential of Quercetin in COVID-19 treatment. J. Inflamm. 2021, 18, 1. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.; Jõudu, I.; Bhat, R. Dietary Fiber from Underutilized Plant Resources—A Positive Approach for Valorization of Fruit and Vegetable Wastes. Sustainability 2020, 12, 5401. [Google Scholar] [CrossRef]
- Jõgi, K.; Bhat, R. Valorization of food processing wastes and by-products for bioplastic production. Sustain. Chem. Pharm. 2020, 18, 100326. [Google Scholar] [CrossRef]
- Malenica, D.; Bhat, R. Current research trends in fruit and vegetables wastes and by-products management-Scope and opportunities in the Estonian context. Agron. Res. 2020, 18, 1760–1795. [Google Scholar]
- Sharma, M.; Bhat, R. Extraction of Carotenoids from Pumpkin Peel and Pulp: Comparison between Innovative Green Extraction Technologies (Ultrasonic and Microwave-Assisted Extractions Using Corn Oil). Foods 2021, 10, 787. [Google Scholar] [CrossRef]
- Ay, M.; Charli, A.; Jin, H.; Anantharam, V.; Kanthasamy, A.; Kanthasamy, A.G. Quercetin. In Book Nutraceuticals, Ramesh C. Gupta, 1st ed.; Academic Press; Elsevier: New York, NY, USA, 2016; Volume 32, pp. 447–452. [Google Scholar]
- Yu, S.; Kim, S.R.; Jiang, K.; Ogrodnik, M.; Zhu, X.Y.; Ferguson, C.M.; Tchkonia, T.; Lerman, A.; Kirkland, J.L.; Lerman, L.O. Quercetin Reverses Cardiac Systolic Dysfunction in Mice Fed with a High-Fat Diet: Role of Angiogenesis. Oxid Med. Cell. Longev. 2021, 2021, 8875729. [Google Scholar] [CrossRef]
- Rolnik, A.; Żuchowski, J.; Stochmal, A.; Olas, B. Quercetin and kaempferol derivatives isolated from aerial parts of Lens culinaris Medik as modulators of blood platelet functions. Ind. Crop. Prod. 2020, 152, 112536. [Google Scholar] [CrossRef]
- Elbarbry, F.; Abdelkawy, K.; Moshirian, N.; Abdel-Megied, A.M. The antihypertensive effect of quercetin in young spontaneously hypertensive rats; role of arachidonic acid metabolis. Int. J. Mol. Sci. 2020, 21, 6554. [Google Scholar] [CrossRef]
- Liang, Y.; Zhang, Y.; Liu, M.; Han, X.; Zhang, J.; Zhang, X.; Chu, L. Protective effect of quercetin against myocardial ischemia as a Ca2+ channel inhibitor: Involvement of inhibiting contractility and Ca2+ influx via L-type Ca2+ channels. Arch. Pharm. Res. 2020, 43, 808–820. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Zhao, Y.; Wang, S.; Wang, Y.; Chen, Q.; Zhao, H.; Wang, H.; Tian, S.; Yu, H.; Wu, Z. Zi Shen Huo Luo Formula Enhances the Therapeutic Effects of Angiotensin-Converting Enzyme Inhibitors on Hypertensive Left. Ventricular Hypertrophy by Interfering with Aldosterone Breakthrough and Affecting Caveolin-1/Mineralocorticoid Receptor Colocalization and Downstream Extracellular Signal-Regulated Kinase Signaling. Front. Pharmacol. 2020, 11, 383. [Google Scholar]
- Lu, J.; Meng, Y.; Wang, R.; Zhang, R. Anti-arrhythmogenic effects of quercetin postconditioning in myocardial ischemia/reperfusion injury in a rat model. J. King Saud Univ. Sci. 2020, 32, 1910–1916. [Google Scholar] [CrossRef]
- Ferenczyova, K.; Kalocayova, B.; Bartekova, M. Potential implications of quercetin and its derivatives in cardioprotection. Int. J. Mol. Sci. 2020, 21, 1585. [Google Scholar] [CrossRef]
- Ferenczyova, K.; Kalocayova, B.; Kindernay, L.; Jelemensky, M.; Balis, P.; Berenyiova, A.; Zemancikova, A.; Farkasova, V.; Sykora, M.; Tothova, L.; et al. quercetin exerts age-dependent beneficial effects on blood pressure and vascular function, but is inefficient in preventing myocardial ischemia-reperfusion injury in zucker diabetic fatty rats. Molecules 2020, 25, 187. [Google Scholar] [CrossRef]
- Wang, M.-N.; Cao, Y.-G.; Wei, Y.-X.; Ren, Y.-J.; Liu, Y.-L.; Chen, X.; He, C.; Zheng, X.-K.; Feng, W.-S. Saffloflavone, a new flavonoid from the flowers of Carthamus tinctorius L. and its cardioprotective activity. Nat. Prod. Res. 2020, 12, 1–6. [Google Scholar]
- Stechyshyn, I.; Pavliuk, B. The quercetine containing drugs in pharmacological correction of experimental diabetes with myocardial injury. Rom. J. Diabetes Nutr. Metab. Dis. 2020, 26, 393–399. [Google Scholar] [CrossRef]
- Binder, P.; Wang, S.; Radu, M.; Zin, M.; Collins, L.; Khan, S.; Li, Y.; Sekeres, K.; Humphreys, N.; Swanton, E.; et al. Pak2 as a Novel Therapeutic Target for Cardioprotective Endoplasmic Reticulum Stress Response. Circ. Res. 2019, 124, 696–711. [Google Scholar] [CrossRef] [PubMed]
- Ogar, I.; Egbung, G.E.; Nna, V.U.; Atangwho, I.J.; Itam, E.H. Hyptis verticillata attenuates dyslipidaemia, oxidative stress and hepato-renal damage in streptozotocin-induced diabetic rats. Life Sci. 2019, 219, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Burak, C.; Wolffram, S.; Zur, B.; Langguth, P.; Fimmers, R.; Alteheld, B.; Stehle, P.; Egert, S. Effect of alpha-linolenic acid in combination with the flavonol Quercetin on markers of cardiovascular disease risk in healthy, non-obese adults: A randomized, double-blinded placebo-controlled crossover trial. Nutrition 2019, 58, 47–56. [Google Scholar] [CrossRef]
- Shu, Z.; Yang, Y.; Yang, L.; Jiang, H.; Yu, X.; Wang, Y. Cardioprotective effects of dihydro quercetin against ischemia reperfusion injury by inhibiting oxidative stress and endoplasmic reticulum stress-induced apoptosis: Via the PI3K/Akt pathway. Food Funct. 2019, 10, 213–215. [Google Scholar] [CrossRef]
- Wang, Y.; Xie, X.; Liu, L.; Zhang, H.; Ni, F.; Wen, J.; Wu, Y.; Wang, Z.; Xiao, W. Four new flavonol glycosides from the leaves of Ginkgo biloba. Nat. Prod. Res. 2019. [Google Scholar] [CrossRef]
- Chakraborty, M.; Ahmed, M.G.; Bhattacharjee, A. Effect of quercetin on myocardial potency of curcumin against ischemia reperfusion induced myocardial toxicity. Synergy 2018, 7, 25–29. [Google Scholar] [CrossRef]
- Chiş, I.C.; Baltaru, D.; Dumitrovici, A.; Coseriu, A.; Radu, B.C.; Moldovan, R.; Mureşan, A. Protective effects of quercetin from oxidative/nitrosative stress under intermittent hypobaric hypoxia exposure in the rat’s heart. Physiol. Int. 2018, 105, 233–246. [Google Scholar] [CrossRef]
- Zakaria, N.; Khalil, S.R.; Awad, A.; Khairy, G.M. Quercetin Reverses Altered Energy Metabolism in the Heart of Rats Receiving Adriamycin Chemotherapy. Cardiovasc. Toxicol. 2018, 18, 109–119. [Google Scholar] [CrossRef]
- Zhang, X.; Li, P.; Guo, S.; Wang, S.; Liu, D. Quantitation of β-carboline and quercetin in alligator weed (Alternanthera philoxeroides (Mart.) Griseb.) by LC-MS/MS and evaluation of cardioprotective effects of the methanol extracts. Drug Discov. Ther. 2018, 12, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Castillo, R.L.; Herrera, E.A.; Gonzalez-Candia, A.; Reyes-Farias, M.; De La Jara, N.; Peña, J.P.; Carrasco-Pozo, C. Quercetin Prevents Diastolic Dysfunction Induced by a High-Cholesterol Diet: Role of Oxidative Stress and Bioenergetics in Hyperglycemic Rats. Oxidative Med. Cell. Longev. 2018, 2018, 7239123. [Google Scholar] [CrossRef]
- Hayamizu, K.; Morimoto, S.; Nonaka, M.; Hoka, S.; Sasaguri, T. Cardiotonic actions of quercetin and its metabolite tamarixetin through a digitalis-like enhancement of Ca2+ transients. Arch. Biochem. Biophys. 2018, 637, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Garjani, A.; Tila, D.; Hamedeyazdan, S.; Vaez, H.; Rameshrad, M.; Pashaii, M.; Fathiazad, F. An investigation on cardioprotective potential of Marrubium vulgare aqueous fraction against ischaemia-reperfusion injury in isolated rat heart. Folia Morphol. 2017, 76, 361–371. [Google Scholar] [CrossRef]
- Alrushaid, S.; Zhao, Y.; Sayre, C.L.; Maayah, Z.H.; Laird Forrest, M.; Senadheera, S.N.; Chaboyer, K.; Anderson, H.D.; El-Kadi, A.O.S.; Davies, N.M. Mechanistically elucidating the in vitro safety and efficacy of a novel doxorubicin derivative. Drug Deliv. Transl. Res. 2017, 7, 582–597. [Google Scholar] [CrossRef]
- Li, W.; Li, Y.; Sun, R.; Zhou, S.; Li, M.; Feng, M.; Xie, Y. Dual character of flavonoids in attenuating and aggravating ischemia-reperfusion-induced myocardial injury. Exp. Ther. Med. 2017, 14, 1307–1314. [Google Scholar] [CrossRef] [PubMed]
- Ballmann, C.; Denney, T.; Beyers, R.J.; Quindry, T.; Romero, M.; Selsby, J.T.; Quindry, J.C. Long-term dietary quercetin enrichment as a cardioprotective countermeasure in mdx mice. Exp. Physiol. 2017, 102, 635–649. [Google Scholar] [CrossRef] [PubMed]
- Syama, H.P.; Arya, A.D.; Dhanya, R.; Nisha, P.; Sundaresan, A.; Jacob, E.; Jayamurthy, P. Quantification of phenolics in Syzygium cumini seed and their modulatory role on tertiary butyl-hydrogen peroxide-induced oxidative stress in H9c2 cell lines and key enzymes in cardioprotection. J. Food Sci. Technol. 2017, 54, 2115–2125. [Google Scholar] [CrossRef]
- Roslan, J.; Giribabu, N.; Karim, K.; Salleh, N. Quercetin ameliorates oxidative stress, inflammation and apoptosis in the heart of streptozotocin-nicotinamide-induced adult male diabetic rats. Biomed. Pharmacother. 2017, 86, 570–582. [Google Scholar] [CrossRef]
- Kumar, M.; Kasala, E.R.; Bodduluru, L.N.; Kumar, V.; Lahkar, M. Molecular and biochemical evidence on the protective effects of quercetin in isoproterenol-induced acute myocardial injury in rats. J. Biochem. Mol. Toxicol. 2017, 31, 8. [Google Scholar] [CrossRef]
- Bin-Jaliah, I. Quercetin inhibits chronic stress–Induced myocardial infarction in rats. [La Quercetina inhibe el infarto al miocardio inducido por estrés crónico en ratas]. Int. J. Morphol. 2017, 35, 1363–1369. [Google Scholar] [CrossRef]
- Chekalina, N.I.; Shut, S.V.; Trybrat, T.A.; Burmak, Y.H.; Petrov, Y.Y.; Manusha, Y.I.; Kazakov, Y.M. Effect of quercetin on parameters of central hemodynamics and myocardial ischemia in patients with stable coronary heart disease. Wiad. Lek. 2017, 70, 707–711. [Google Scholar]
- Ramachandran, R.; Mani, M.; Soman, S. Mediation of FOXO3 gene on momism of quercetin against isoproterenol induced cardiotoxicity in H9C2 cardiomyoblasts. Int. Cardiovasc. Res. J. 2017, 11, 49–54. [Google Scholar]
- Soman, S.; Rajamanickam, C.; Rauf, A.A.; Madambath, I. Molecular mechanisms of the antiglycative and cardioprotective activities of Psidium guajava leaves in the rat diabetic myocardium. Pharm. Biol. 2016, 54, 3078–3085. [Google Scholar] [CrossRef]
- Cheserek, M.J.; Wu, G.; Li, L.; Li, L.; Karangwa, E.; Shi, Y.; Le, G. Cardioprotective effects of lipoic acid, quercetin and resveratrol on oxidative stress related to thyroid hormone alterations in long-term obesity. J. Nutr. Biochem. 2016, 33, 36–44. [Google Scholar] [CrossRef]
- Li, C.; Wang, T.; Zhang, C.; Xuan, J.; Su, C.; Wang, Y. Quercetin attenuates cardiomyocyte apoptosis via inhibition of JNK and p38 mitogen-activated protein kinase signaling pathways. Gene 2016, 577, 275–280. [Google Scholar] [CrossRef]
- Grande, F.; Parisi, O.I.; Mordocco, R.A.; Rocca, C.; Puoci, F.; Scrivano, L.; Quintieri, A.M.; Cantafio, P.; Ferla, S.; Brancale, A.; et al. Quercetin derivatives as novel antihypertensive agents: Synthesis and physiological characterization. Eur. J. Pharm. Sci. 2016, 82, 161–170. [Google Scholar] [CrossRef]
- Bartekova, M.; Radosinska, J.; Pancza, D.; Barancik, M.; Ravingerova, T. Cardioprotective effects of quercetin against ischemia-reperfusion injury are age-dependent. Psychol. Res. 2016, 65, S101–S107. [Google Scholar] [CrossRef] [PubMed]
- Kavya, S.; Kumar, M.S. Cardioprotective effect of bioflavonoids against isoproterenol induced cardiotoxicity in rats. Int. J. Pharma Bio Sci. 2016, 7, P158–P162. [Google Scholar]
- Dobhal, Y.; Parcha, V.; Dhasmana, D.C. Characterization of new cardioprotective principle isolated from methanolic extract of Allium humile leaves from Himalayan region. Bangladesh J. Pharmacol. 2016, 11, 383–388. [Google Scholar] [CrossRef]
- Liu, X.; Yu, Z.; Huang, X.; Gao, Y.; Wang, X.; Gu, J.; Xue, S. Peroxisome proliferator-activated receptor γ (PPARγ) mediates the protective effect of quercetin against myocardial ischemia-reperfusion injury via suppressing the NF-κB pathway. Am. J. Transl. Res. 2016, 8, 5169–5186. [Google Scholar]
- Galinski, C.N.; Zwicker, J.I.; Kennedy, D.R. Revisiting the mechanistic basis of the French Paradox: Red wine inhibits the activity of protein disulfide isomerase in vitro. Thromb. Res. 2016, 137, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Ozbek, N.; Bali, E.B.; Karasu, C. Quercetin and hydroxytyrosol attenuates xanthine/xanthine oxidase-induced toxicity in H9c2 cardiomyocytes by regulation of oxidative stress and stress-sensitive signaling pathways. Gen. Physiol. Biophys. 2015, 34, 407–414. [Google Scholar] [PubMed]
- Brüll, V.; Burak, C.; Stoffel-Wagner, B.; Wolffram, S.; Nickenig, G.; Müller, C.; Langguth, P.; Alteheld, B.; Fimmers, R.; Naaf, S.; et al. Effects of a quercetin-rich onion skin extract on 24 h ambulatory blood pressure and endothelial function in overweight-to-obese patients with (pre-)hypertension: A randomised double-blinded placebo-controlled cross-over trial. Br. J. Nutr. 2015, 114, 1263–1277. [Google Scholar] [CrossRef]
- Daubney, J.; Bonner, P.L.; Hargreaves, A.J.; Dickenson, J.M. Cardioprotective and cardiotoxic effects of quercetin and two of its in vivo metabolites on differentiated H9c2 cardiomyocytes. Basic Clin. Pharmacol. Toxicol. 2015, 116, 96–109. [Google Scholar] [CrossRef] [PubMed]
- Dower, J.I.; Geleijnse, J.M.; Gijsbers, L.; Schalkwijk, C.; Kromhout, D.; Hollman, P.C. Supplementation of the pure flavonoids epicatechin and quercetin affects some biomarkers of endothelial dysfunction and inflammation in (pre)hypertensive adults: A randomized double-blind, placebo-controlled, crossover trial. J. Nutr. 2015, 145, 1459–1463. [Google Scholar] [CrossRef]
- Dower, J.I.; Geleijnse, J.M.; Gijsbers, L.; Zock, P.L.; Kromhout, D.; Hollman, P.C.H. Effects of the pure flavonoids epicatechin and quercetin on vascular function and cardiometabolic health: A randomized, double-blind, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 2015, 101, 914–921. [Google Scholar] [CrossRef]
- El-Mahalaway, A.M.; Selim, A.A.; Mahboub, F.A.R. The effect of quercetin on cyclosporine-A-induced cardiotoxicity in adult male albino rats: A histological and immunohistochemical study. Egypt. J. Histol. 2015, 38, 464–473. [Google Scholar] [CrossRef]
- Daskalova, E.; Delchev, S.; Peeva, Y.; Vladimirova-Kitova, L.; Kratchanova, M.; Kratchanov, C.; Denev, P. Antiatherogenic and Cardioprotective Effects of Black Chokeberry (Aronia melanocarpa) Juice in Aging Rats. Evid. Based Complement. Altern. Med. 2015, 2015, 717439. [Google Scholar] [CrossRef]
- Adaramoye, O.A.; Lawal, S.O. Kolaviron, a biflavonoid fraction from Garcinia kola, protects against isoproterenol-induced injury by mitigating cardiac dysfunction and oxidative stress in rats. J. Basic Clin. Physiol. Pharmacol. 2015, 26, 65–72. [Google Scholar] [CrossRef]
- Matouk, A.I.; Taye, A.; Heeba, G.H.; El-Moselhy, M.A. Quercetin augments the protective effect of losartan against chronic doxorubicin cardiotoxicity in rats. Environ. Toxicol. Pharmacol. 2013, 36, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Milton Prabu, S.; Muthumani, M.; Shagirtha, K. Quercetin potentially attenuates cadmium induced oxidative stress mediated cardiotoxicity and dyslipidemia in rats. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 582–595. [Google Scholar]
- Li, M.; Jiang, Y.; Jing, W.; Sun, B.; Miao, C.; Ren, L. Quercetin provides greater cardioprotective effect than its glycoside derivative rutin on isoproterenol-induced cardiac fibrosis in the rat. Can. J. Physiol. Pharmacol. 2013, 91, 951–959. [Google Scholar] [CrossRef]
- Chen, Y.-W.; Chou, H.-C.; Lin, S.-T.; Chen, Y.-H.; Chang, Y.-J.; Chen, L.; Chan, H.-L. Cardioprotective effects of quercetin in cardiomyocyte under ischemia/reperfusion injury. Evid. Based Complement. Altern. Med. 2013. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-Q.; Zhu, X.-F.; Wang, X.-N.; Shen, T.; Xiang, F.; Lou, H.-X. Flavonoids from Malus hupehensis and their cardioprotective effects against doxorubicin-induced toxicity in H9c2 cells. Phytochem 2013, 87, 119–125. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Z.Z.; Wu, Y.; Ke, J.J.; He, X.H.; Wang, Y.L. Quercetin postconditioning attenuates myocardial ischemia/reperfusion injury in rats through the PI3K/Akt pathway. Braz. J. Med. Biol. Res. 2013, 46, 861–867. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Croft, K.D.; Hodgson, J.M.; Kyle, R.; Lee, I.-L.E.; Wang, Y.; Stocker, R.; Ward, N.C. Quercetin and its metabolites improve vessel function by inducing eNOS activity via phosphorylation of AMPK. Biochem. Pharmacol. 2012, 84, 1036–1044. [Google Scholar] [CrossRef]
- Yadav, H.N.; Singh, M.; Sharma, P.L. Pharmacological inhibition of GSK-3β produces late phase of cardioprotection in hyperlipidemic rat: Possible involvement of HSP 72. Mol. Cell. Biochem. 2012, 369, 227–233. [Google Scholar] [CrossRef]
- Panda, S.; Kar, A.; Banerjee, T.; Sharma, N. Combined effects of quercetin and atenolol in reducing isoproterenol- induced cardiotoxicity in rats: Possible mediation through scavenging free radicals. Cardiovasc. Toxicol. 2012, 12, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, S.F.; Nabavi, S.M.; Mirzaei, M.; Moghaddam, A.H. Protective effect of quercetin against sodium fluoride induced oxidative stress in rat’s heart. Food Funct. 2012, 3, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-Y.; Chan, H.-L.; Chou, H.-C. Proteomic analysis of quercetin-induced cardioprotective effects. Genom. Med. Biomark. Health Sci. 2012, 4, 51–53. [Google Scholar] [CrossRef][Green Version]
- Wang, Z.; Wan, H.; Tong, X.; He, Y.; Yang, J.; Zhang, L.; Shao, C.; Ding, Z.; Wan, H.; Li, C. An integrative strategy for discovery of functional compound combination from Traditional Chinese Medicine: Danhong Injection as a model. Biomed Pharm. 2021, 138, 111451. [Google Scholar] [CrossRef]
- Santo, G.D.; de Veras, B.O.; Rico, E.; Magro, J.D.; Agostini, J.F.; Vieira, L.D.; Calisto, J.F.F.; Mocelin, R.; de Sá Fonseca, V.; Wanderley, A.G. Hexane extract from SpoSndias mombin L. (Anacardiaceae) prevents behavioral and oxidative status changes on model of Parkinson’s disease in zebrafish. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2021, 241, 108953. [Google Scholar] [CrossRef]
- Bhatia, G.; Singh, J.; Nehru, B. Neuroprotective effects of hydro-alcoholic extract of Eclipta alba against 1-methyl-4-phenylpyridinium-induced in vitro and in vivo models of Parkinson’s disease. Environ. Sci. Pollut. Res. 2021, 28, 9390–9406. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Zheng, Y.; Chen, R.; Huang, X.; Shi, G. Neuroprotective effects of Quercetin 3-O-sophoroside from Hibiscus rosa-sinensis Linn. on scopolamine-induced amnesia in mice. J. Funct. Foods 2021, 76, 104291. [Google Scholar] [CrossRef]
- Sehaber-Sierakowski, C.C.; Vieira-Frez, F.C.; Hermes-Uliana, C.; Martins, H.A.; Bossolani, G.D.P.; Lima, M.M.; Blegniski, F.P.; Guarnier, F.A.; Baracat, M.M.; Perles, J.V.C.M.; et al. Protective effects of Quercetin-loaded microcapsules on the enteric nervous system of diabetic rats. Auton. Neurosci. Basic Clin. 2021, 230, 102759. [Google Scholar] [CrossRef] [PubMed]
- Jalili-Baleh, L.; Nadri, H.; Forootanfar, H.; Küçükkılınç, T.T.; Ayazgök, B.; Sharifzadeh, M.; Rahimifard, M.; Baeeri, M.; Abdollahi, M.; Foroumadi, A.; et al. Chromone–lipoic acid conjugate: Neuroprotective agent having acceptable butyrylcholinesterase inhibition, antioxidant and copper-chelation activities. DARU J. Pharm. Sci. 2021. [Google Scholar] [CrossRef] [PubMed]
- Ortega, J.T.; Parmar, T.; Golczak, M.; Jastrzebska, B. Protective effects of flavonoids in acute models of light-induced retinal degeneration. Mol. Pharmacol. 2021, 99, 60–77. [Google Scholar] [CrossRef]
- Zhang, J.; Ning, L.; Wang, J. Dietary Quercetin attenuates depressive-like behaviors by inhibiting astrocyte reactivation in response to stress. Biochem. Biophys. Res. Commun. 2020, 533, 1338–1346. [Google Scholar] [CrossRef] [PubMed]
- Le, K.; Song, Z.; Deng, J.; Peng, X.; Zhang, J.; Wang, L.; Zhou, L.; Bi, H.; Liao, Z.; Feng, Z. Quercetin alleviates neonatal hypoxic-ischemic brain injury by inhibiting microglia-derived oxidative stress and TLR4-mediated inflammation. J. Inflamm. Res. 2020, 69, 1201–1213. [Google Scholar] [CrossRef]
- Baraka, S.M.; Saleh, D.O.; Ghaly, N.S.; Melek, F.R.; Gamal el Din, A.A.; Khalil, W.K.B.; Said, M.M.; Medhat, A.M. Flavonoids from Barnebydendron riedelii leaf extract mitigate thioacetamide-induced hepatic encephalopathy in rats: The interplay of NF-κB/IL-6 and Nrf2/HO-1 signaling pathways. Bioorg. Chem. 2020, 105, 104444. [Google Scholar] [CrossRef]
- Dallas, P.; Rekkas, D.M.; Demetzos, C.; Colombo, G.; Banella, S.; Javornik, U.; Plavec, J.; Mavromoustakos, T.; Tzakos, A.G.; Valsami, G. Preparation and Biophysical Characterization of Quercetin Inclusion Complexes with β-Cyclodextrin Derivatives to be Formulated as Possible Nose-to-Brain Quercetin Delivery Systems. Mol. Pharm. 2020, 17, 4241–4255. [Google Scholar]
- Singh, V.; Shri, R.; Krishan, P.; Singh, I.P.; Shah, P. Isolation and characterization of components responsible for neuroprotective effects of Allium cepa outer scale extract against ischemia reperfusion induced cerebral injury in mice. J. Food Sci. 2020, 85, 4009–4017. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, P.K.; Pradhan, L.K.; Aparna, S.; Agarwal, K.; Banerjee, A.; Das, S.K. Quercetin abrogates bisphenol A induced altered neurobehavioral response and oxidative stress in zebrafish by modulating brain antioxidant defence system. Environ. Toxicol. Pharmacol. 2020, 80, 103483. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhu, D.; Luo, J.; Chen, X.; Gao, L.; Liu, W.; Chen, T. NIR-II-Activated Yolk-Shell Nanostructures as an Intelligent Platform for Parkinsonian Therapy. ACS Appl. Bio Mater. 2020, 3, 6876–6887. [Google Scholar] [CrossRef]
- Rifaai, R.A.; Mokhemer, S.A.; Saber, E.A.; El-Aleem, S.A.A.; El-Tahawy, N.F.G. Neuroprotective effect of Quercetin nanoparticles: A possible prophylactic and therapeutic role in alzheimer’s disease. J. Chem. Neuroanat. 2020, 107, 101795. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Chang, C.-Y.; Lin, S.-Y.; Wang, J.-D.; Wu, C.-C.; Chen, W.-Y.; Kuan, Y.-H.; Liao, S.-L.; Wang, W.-Y.; Chen, C.-J. Quercetin protects against cerebral ischemia/reperfusion and oxygen glucose deprivation/reoxygenation neurotoxicity. J. Nutr. Biochem. 2020, 83, 108436. [Google Scholar] [CrossRef]
- Martins-Perles, J.V.C.; Bossolani, G.D.P.; Zignani, I.; de Souza, S.R.G.; Frez, F.C.V.; de Souza Melo, C.G.; Barili, E.; de Souza Neto, F.P.; Guarnier, F.A.; Armani, A.L.C.; et al. Quercetin increases bioavailability of nitric oxide in the jejunum of euglycemic and diabetic rats and induces neuronal plasticity in the myenteric plexus. Auton. Neurosci. 2020, 227, 102675. [Google Scholar] [CrossRef]
- An, L.; Zhang, W.; Ma, G.; Wang, K.; Ji, Y.; Ren, H.; Wang, Y. Neuroprotective effects of Camellia nitidissima Chi leaf extract in hydrogen peroxide-treated human neuroblastoma cells and its molecule mechanisms. Food Sci. Nutr. 2020, 8, 4782–4793. [Google Scholar] [CrossRef]
- Sharma, L.; Sharma, A.; Goyal, R.; Alam, J. Pinus Roxburghii Sarg. Ameliorates alzheimer’s disease-type neurodegeneration and cognitive deficits caused by intracerebroventricular-streptozotocin in rats: An in vitro and in vivo study. Indian J. Pharm. Sci. 2020, 82, 861–870. [Google Scholar] [CrossRef]
- Karaman, M.; Tesanovic, K.; Novakovic, A.; Jakovljevic, D.; Janjusevic, L.; Sibul, F.; Pejin, B. Coprinus comatus filtrate extract, a novel neuroprotective agent of natural origin. Nat. Prod. Res. 2020, 34, 2346–2350. [Google Scholar] [CrossRef]
- Pakrashi, S.; Chakraborty, J.; Bandyopadhyay, J. Neuroprotective Role of Quercetin on Rotenone-Induced Toxicity in SH-SY5Y Cell Line Through Modulation of Apoptotic and Autophagic Pathways. Neurochem. Res. 2020, 45, 1962–1973. [Google Scholar] [CrossRef]
- Xie, J.; Song, W.; Liang, X.; Zhang, Q.; Shi, Y.; Liu, W.; Shi, X. Protective effect of Quercetin on streptozotocin-induced diabetic peripheral neuropathy rats through modulating gut microbiota and reactive oxygen species level. Biomed. Pharmacother. 2020, 127, 110147. [Google Scholar] [CrossRef]
- Chaturvedi, S.; Malik, M.Y.; Rashid, M.; Singh, S.; Tiwari, V.; Gupta, P.; Shukla, S.; Singh, S.; Wahajuddin, M. Mechanistic exploration of Quercetin against metronidazole induced neurotoxicity in rats: Possible role of nitric oxide isoforms and inflammatory cytokines. NeuroToxicology 2020, 79, 1–10. [Google Scholar] [CrossRef]
- Pinheiro, R.G.R.; Granja, A.; Loureiro, J.A.; Pereira, M.C.; Pinheiro, M.; Neves, A.R.; Reis, S. RVG29-Functionalized Lipid Nanoparticles for Quercetin Brain Delivery and Alzheimer’s Disease. Pharm. Res. 2020, 37, 139. [Google Scholar] [CrossRef] [PubMed]
- Nzekoue, F.K.; Angeloni, S.; Navarini, L.; Angeloni, C.; Freschi, M.; Hrelia, S.; Vitali, L.A.; Sagratini, G.; Vittori, S.; Caprioli, G. Coffee silverskin extracts: Quantification of 30 bioactive compounds by a new HPLC-MS/MS method and evaluation of their antioxidant and antibacterial activities. Food Res. Int. 2020, 133, 109128. [Google Scholar] [CrossRef]
- de Mattos, B.D.S.; Soares, M.S.P.; Spohr, L.; Pedra, N.S.; Teixeira, F.C.; de Souza, A.A.; Stefanello, F.M.; Baldissarelli, J.; Gamaro, G.D.; Spanevello, R.M. Quercetin prevents alterations of behavioral parameters, delta-aminolevulinic dehydratase activity, and oxidative damage in brain of rats in a prenatal model of autism. Int. J. Dev. Neurosci. 2020, 80, 287–302. [Google Scholar] [CrossRef] [PubMed]
- Lossi, L.; Merighi, A.; Novello, V.; Ferrandino, A. Protective effects of some grapevine polyphenols against naturally occurring neuronal death. Molecules 2020, 25, 2925. [Google Scholar] [CrossRef]
- Pinheiro, R.G.R.; Granja, A.; Loureiro, J.A.; Pereira, M.C.; Pinheiro, M.; Neves, A.R.; Reis, S. Quercetin lipid nanoparticles functionalized with transferrin for Alzheimer’s disease. Eur. J. Pharm. Sci. 2020, 148, 105314. [Google Scholar]
- Viswanatha, G.L.; Drishya, M.; Kishore, D.V.; Shylaja, H.; Moolemath, Y. Characterization and Anti-stroke Activity of Stem Extract of Colebrookea oppositifolia Smith. J. Biol. Act. Prod. Nat. 2020, 10, 250–263. [Google Scholar] [CrossRef]
- Gugliandolo, E.; Peritore, A.F.; D’amico, R.; Licata, P.; Crupi, R. Evaluation of neuroprotective effects of Quercetin against aflatoxin b1-intoxicated mice. Animals 2020, 10, 898. [Google Scholar] [CrossRef]
- Woo, Y.; Lim, J.S.; Oh, J.; Lee, J.S.; Kim, J.-S. Neuroprotective effects of euonymus alatus extract on scopolamine-induced memory deficits in mice. Antioxidants 2020, 9, 449. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Zhou, H.; Wang, G.; Zhong, X.-H.; Shen, Q.-L.; Zhang, X.-J.; Li, R.-Y.; Chen, L.-H.; Zhang, Y.-H.; Wan, Z. Quercetin is protective against short-term dietary advanced glycation end products intake induced cognitive dysfunction in aged ICR mice. J. Food Biochem. 2020, 44, 4. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Wang, G.; Ma, Z.F.; Qin, L.-Q.; Zhai, Y.-J.; Yu, Z.-L.; Xue, M.; Zhang, Y.-H.; Wan, Z. DietaryAdvanced Glycation End Products–Induced Cognitive Impairment in Aged ICR Mice: Protective Role of quercetin. Mol. Nutr. Food Res. 2020, 64, 1901019. [Google Scholar] [CrossRef]
- Ibrahim, K.A.; Eleyan, M.; Abd El-Rahman, H.A.; Khwanes, S.A.; Mohamed, R.A. Quercetin Attenuates the Oxidative Injury–Mediated Upregulation of Apoptotic Gene Expression and Catecholaminergic Neurotransmitters of the Fetal Rats’ Brain Following Prenatal Exposure to Fenitrothion Insecticide. Neurotox. Res. 2020, 37, 871–882. [Google Scholar] [CrossRef]
- Shalavadi, M.H.; Chandrashekhar, V.M.; Muchchandi, I.S. Neuroprotective effect of Convolvulus pluricaulis Choisy in oxidative stress model of cerebral ischemia reperfusion injury and assessment of MAP2 in rats. J. Ethnopharmacol. 2020, 249, 112393. [Google Scholar] [CrossRef] [PubMed]
- Vieira-Frez, F.C.; Sehaber-Sierakowski, C.C.; Perles, J.V.C.M.; Bossolani, G.D.P.; Verri, W.A., Jr.; Nascimento, R.C.D.; Guarnier, F.A.; Bordini, H.P.; Blegniski, F.P.; Martins, H.A.; et al. Anti- and pro-oxidant effects of Quercetin stabilized by microencapsulation on interstitial cells of Cajal, nitrergic neurons and M2-like macrophages in the jejunum of diabetic rats. NeuroToxicology 2020, 77, 193–204. [Google Scholar] [CrossRef]
- Hu, T.; Lu, X.-Y.; Shi, J.-J.; Liu, X.-Q.; Chen, Q.-B.; Wang, Q.; Chen, Y.-B.; Zhang, S.-J. Quercetin protects against diabetic encephalopathy via SIRT1/NLRP3 pathway in db/db mice. J. Cell. Mol. Med. 2020, 24, 3449–3459. [Google Scholar] [CrossRef]
- Park, D.-J.; Kang, J.-B.; Shah, F.-A.; Jin, Y.-B.; Koh, P.-O. Quercetin Attenuates Decrease of Thioredoxin Expression Following Focal Cerebral Ischemia and Glutamate-induced Neuronal Cell Damage. Neuroscience 2020, 428, 38–49. [Google Scholar] [CrossRef]
- Sharma, S.; Raj, K.; Singh, S. Neuroprotective Effect of Quercetin in Combination with Piperine Against Rotenone- and Iron Supplement–Induced Parkinson’s Disease in Experimental Rats. Neurotox. Res. 2020, 37, 198–209. [Google Scholar] [CrossRef]
- Gül, Ş.; Aydoğmuş, E.; Bahadir, B.; Büyükuysal, Ç.; Güven, B. Neuroprotective effects of Quercetin on cerebral vasospasm following experimental subarachnoid haemorrhage in rats. Turk. J. Med. Sci. 2020, 50, 1106–1110. [Google Scholar] [CrossRef] [PubMed]
- Zubčić, K.; Radovanović, V.; Vlainić, J.; Hof, P.R.; Oršolić, N.; Šimić, G.; Jazvinšćak Jembrek, M. PI3K/Akt and ERK1/2 Signalling Are Involved in Quercetin-Mediated Neuroprotection against Copper-Induced Injury. Oxid. Med. Cell. Longev. 2020, 9834742. [Google Scholar] [CrossRef]
- Huang, C.; Fu, C.; Qi, Z.-P.; Guo, W.-L.; You, D.; Li, R.; Zhu, Z. Localised delivery of Quercetin by thermo-sensitive PLGA-PEG-PLGA hydrogels for the treatment of brachial plexus avulsion. Artif. Cells Nanomed. Biotechnol. 2020, 48, 1010–1021. [Google Scholar] [CrossRef]
- Yu, X.; Li, Y.; Mu, X. Effect of Quercetin on PC12 Alzheimer’s Disease Cell Model Induced by A β 25-35 and Its Mechanism Based on Sirtuin1/Nrf2/HO-1 Pathway. Biomed. Res. Int. 2020, 2020, 8210578. [Google Scholar]
- Moujahed, S.; Ruiz, A.; Hallegue, D.; Sakly, M. Quercetin alleviates styrene oxide-induced cytotoxicity in cortical neurons in vitro via modulation of oxidative stress and apoptosis. Drug Chem. Toxicol. 2020, 1–9. [Google Scholar] [CrossRef]
- Sallam, A.; Mira, A.; Sabry, M.A.; Abdel-Halim, O.B.; Gedara, S.R.; Galala, A.A. New prenylated flavonoid and neuroprotective compounds from Tephrosia purpurea subsp. Dunensis. Nat. Prod. Res. 2020, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.; Guo, L.; Jiang, Y.; Shi, Y.; Sui, H.; Zhao, L. Brain delivery of Quercetin-loaded exosomes improved cognitive function in AD mice by inhibiting phosphorylated tau-mediated neurofibrillary tangles. Drug Deliv. 2020, 27, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Malik, J.; Kaur, S.; Karan, M.; Choudhary, S. Neuroprotective effect of standardized extracts of three Lactuca sativa Linn. varieties against 3-NP induced Huntington’s disease like symptoms in rats. Nutr. Neurosci. 2020. [Google Scholar] [CrossRef]
- Gao, H.; Yuan, X.; Wang, Z.; Gao, Q.; Yang, J. Profiles and neuroprotective effects of Lycium ruthenicum polyphenols against oxidative stress-induced cytotoxicity in PC12 cells. J. Food Biochem. 2020, 44, 1. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Yeom, M.; Shim, I.; Lee, H.; Hahm, D.-H. Protective Effects of Quercetin on Anxiety-Like Symptoms and Neuroinflammation Induced by Lipopolysaccharide in Rats. Evid. Based Compl. Altern. Med. 2020. [Google Scholar] [CrossRef]
- Debnath, K.; Jana, N.R.; Jana, N.R. Quercetin Encapsulated Polymer Nanoparticle for Inhibiting Intracellular Polyglutamine Aggregation. ACS Appl. Bio Mater. 2019, 2, 5298–5305. [Google Scholar] [CrossRef]
- Piovezana Bossolani, G.D.; Silva, B.T.; Colombo Martins Perles, J.V.; Lima, M.M.; Vieira Frez, F.C.; Garcia de Souza, S.R.; Sehaber-Sierakowski, C.C.; Bersani-Amado, C.A.; Zanoni, J.N. Rheumatoid arthritis induces enteric neurodegeneration and jejunal inflammation, and Quercetin promotes neuroprotective and anti-inflammatory actions. Life Sci. 2019, 238, 116956. [Google Scholar] [CrossRef]
- Jamali-Raeufy, N.; Baluchnejadmojarad, T.; Roghani, M.; Keimasi, S.; Goudarzi, M. Isorhamnetin exerts neuroprotective effects in STZ-induced diabetic rats via attenuation of oxidative stress, inflammation and apoptosis. J. Chem. Neuroanat. 2019, 102, 101709. [Google Scholar] [CrossRef]
- Donoso, F.; Ramírez, V.T.; Golubeva, A.V.; Moloney, G.M.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Naturally Derived Polyphenols Protect Against Corticosterone-Induced Changes in Primary Cortical Neurons. Int. J. Neuropsychopharmacol. 2019, 22, 765–777. [Google Scholar] [CrossRef]
- Kwon, S.-H.; Lee, S.R.; Park, Y.J.; Ra, M.; Lee, Y.; Pang, C.; Kim, K.H. Suppression of 6-hydroxydopamine-induced oxidative stress by hyperoside via activation of nrf2/ho-1 signaling in dopaminergic neurons. Int. J. Mol. Sci. 2019, 20, 5832. [Google Scholar] [CrossRef]
- Nielsen, B.E.; Bermudez, I.; Bouzat, C. Flavonoids as positive allosteric modulators of α7 nicotinic receptors. Neuropharmacology 2019, 160, 107794. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Guo, T.-L.; Shen, L.-X. Research on estrogen-like protective effect of Quercetin on PC12 cells induced by Aβ25.35 and molecular mechanism. Chin. Pharm. J. 2019, 35, 1558–1564. [Google Scholar]
- Gapski, A.; Gomes, T.M.; Bredun, M.A.; Ferreira-Lima, N.E.; Ludka, F.K.; Bordignon-Luiz, M.T.; Burin, V.M. Digestion behavior and antidepressant-like effect promoted by acute administration of blueberry extract on mice. Food Res. Int. 2019, 125, 108618. [Google Scholar] [CrossRef] [PubMed]
- Moliner, C.; Barros, L.; Dias, M.I.; Reigada, I.; Ferreira, I.C.F.R.; López, V.; Langa, E.; Rincón, C.G. Viola cornuta and Viola × wittrockiana: Phenolic compounds, antioxidant and neuroprotective activities on Caenorhabditis elegans. J. Food Drug Anal. 2019, 27, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Azib, L.; Debbache-Benaida, N.; Costa, G.D.; Atmani-Kilani, D.; Saidene, N.; Ayouni, K.; Richard, T.; Atmani, D. Pistacia lentiscus L. leaves extract and its major phenolic compounds reverse aluminium-induced neurotoxicity in mice. Ind. Crops. Prod. 2019, 137, 576–584. [Google Scholar] [CrossRef]
- Li, Y.; Tian, Q.; Li, Z.; Dang, M.; Lin, Y.; Hou, X. Activation of Nrf2 signaling by sitagliptin and Quercetin combination against β-amyloid induced Alzheimer’s disease in rats. Drug Dev. Res. 2019, 80, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Liu, F.; Guo, Q. Quercetin attenuates hypoxia-ischemia induced brain injury in neonatal rats by inhibiting TLR4/NF-κB signaling pathway. Int. Immunopharmacol. 2019, 74, 105704. [Google Scholar] [CrossRef]
- Fereidouni, S.; Kumar, R.R.; Chadha, V.D.; Dhawan, D.K. Quercetin plays protective role in oxidative induced apoptotic events during chronic chlorpyrifos exposure to rats. J. Biochem. Mol. Toxicol. 2019, 33, 8. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Busi, M.; Arredondo, F.; González, D.; Echeverry, C.; Vega-Teijido, M.A.; Carvalho, D.; Rodríguez-Haralambides, A.; Rivera, F.; Dajas, F.; Abin-Carriquiry, J.A. Purification, structural elucidation, antioxidant capacity and neuroprotective potential of the main polyphenolic compounds contained in Achyrocline satureioides (Lam) D.C. (Compositae). Bioorg. Med. Chem. 2019, 27, 2579–2591. [Google Scholar] [CrossRef]
- Yan, R.; Tian, H.; Du, Z. Quercetin protects PC-12 cells against hypoxia injury by downregulation of miR-122. Iran. J. Basic Med. Sci. 2019, 22, 391–398. [Google Scholar] [PubMed]
- Cittadini, M.C.; Repossi, G.; Albrecht, C.; Di Paola Naranjo, R.; Miranda, A.R.; de Pascual-Teresa, S.; Soria, E.A. Effects of bioavailable phenolic compounds from Ilex paraguariensis on the brain of mice with lung adenocarcinoma. Phytother. Res. 2019, 33, 1142–1149. [Google Scholar] [CrossRef]
- Kaur, S.; Singla, N.; Dhawan, D.K. Neuro-protective potential of quercetin during chlorpyrifos induced neurotoxicity in rats. Drug Chem. Toxicol. 2019, 42, 220–230. [Google Scholar] [CrossRef]
- Lu, Y.; Du, Y.; Qin, X.; Wu, H.; Huang, Y.; Cheng, Y.; Wei, Y. Comprehensive evaluation of effective polyphenols in apple leaves and their combinatory antioxidant and neuroprotective activities. Ind. Crop. Prod. 2019, 129, 242–252. [Google Scholar] [CrossRef]
- Chatterjee, J.; Langhnoja, J.; Pillai, P.P.; Mustak, M.S. Neuroprotective effect of Quercetin against radiation-induced endoplasmic reticulum stress in neurons. J. Biochem. Mol. Toxicol. 2019, 33, e22242. [Google Scholar] [CrossRef]
- Haq, S.H.; AlAmro, A.A. Neuroprotective effect of Quercetin in murine cortical brain tissue cultures. Clin. Nutr. Exp. 2019, 23, 89–96. [Google Scholar] [CrossRef]
- Faggi, L.; Porrini, V.; Lanzillotta, A.; Benarese, M.; Mota, M.; Tsoukalas, D.; Parrella, E.; Pizzi, M. A polyphenol-enriched supplement exerts potent epigenetic-protective activity in a cell-based model of brain ischemia. Nutrients 2019, 11, 345. [Google Scholar] [CrossRef]
- Viswanatha, G.L.; Venkataranganna, M.V.; Prasad, N.B.L.; Shylaja, H. Achyranthes aspera Linn. alleviates cerebral ischemia-reperfusion-induced neurocognitive, biochemical, morphological and histological alterations in Wistar rats. J. Ethnopharmacol. 2019, 228, 58–69. [Google Scholar] [CrossRef]
- Park, D.-J.; Kang, J.-B.; Shah, M.-A.; Koh, P.-O. Quercetin alleviates the injury-induced decrease of protein phosphatase 2A subunit B in cerebral ischemic animal model and glutamate-exposed HT22 cells. J. Vet. Med. Sci. 2019, 81, 1047–1054. [Google Scholar] [CrossRef]
- Jing, B.-Q.; Zhao, L.; Jing, W.; Zhang, Y.-X. The neuroprotective effects of Quercetin on hypoxic-ischemic brain injury in neonatal mice. Lat. Am. J. Pharm. 2019, 38, 651–658. [Google Scholar]
- Ocal, O.; Borcek, A.O.; Pasaoglu, O.; Gundogdu, A.C.; Kaplanoglu, G.T.; Baykaner, M.K. Can Quercetin be an option for treatment of spinal cord injury? An experimental study. Turk. Neurosurg. 2019, 29, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Mehta, V.; Raj, U.; Varadwaj, P.K.; Udayabanu, M.; Yennamalli, R.M.; Singh, T.R. Computational and in-vitro validation of natural molecules as potential acetylcholinesterase inhibitors and neuroprotective agents. Curr. Alzheimer Res. 2019, 16, 116–127. [Google Scholar] [CrossRef]
- Sinha, S.; Kumar, B.; Luqman, S.; Singh, D.K. Neuroprotective potential of Cucurbita maxima Duchesne ex Poir, Caeselpenia bunduc (L.) Roxb and Bombax ceiba Linn extracts. S. Afr. J. Bot. 2019, 120, 319–325. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, Y.-N.; Wang, W.; Yao, G.-D.; Jiang, X.-F.; Song, S.-J. Flavones from a natural tea (the leaves of Armeniaca sibirica L.) prevent oxidative stress-induced neuronal death. J. Food Biochem. 2018, 42, 1–7. [Google Scholar] [CrossRef]
- Zhu, X.; Cheng, Y.-Q.; Lu, Q.; Du, L.; Yin, X.-X.; Liu, Y.-W. Enhancement of glyoxalase 1, a polyfunctional defense enzyme, by quercetin in the brain in streptozotocin-induced diabetic rats. Naunyn Schmiedeberg Arch. Pharmacol. 2018, 391, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Woo, M.; Kim, M.J.; Song, Y.O. Bioactive compounds in Kimchi improve the cognitive and memory functions impaired by amyloid beta. Nutrients 2018, 10, 1554. [Google Scholar] [CrossRef]
- Jembrek, M.J.; Vlainić, J.; Čadez, V.; Segota, S. Atomic force microscopy reveals new biophysical markers for monitoring subcellular changes in oxidative injury: Neuroprotective effects of quercetin at the nanoscale. PLoS ONE 2018, 13, e0200119. [Google Scholar]
- Enteshari Najafabadi, R.; Kazemipour, N.; Esmaeili, A.; Beheshti, S.; Nazifi, S. Using superparamagnetic iron oxide nanoparticles to enhance bioavailability of quercetin in the intact rat brain. BMC Pharmacol. Toxicol. 2018, 19, 59. [Google Scholar] [CrossRef]
- Viswanatha, G.L.; Venkataranganna, M.V.; Prasad, N.B.L.; Hanumanthappa, S. Chemical characterization and cerebroprotective effect of methanolic root extract of Colebrookea oppositifolia in rats. J. Ethnopharmacol. 2018, 223, 63–75. [Google Scholar] [CrossRef]
- Ghaffari, F.; Moghaddam, A.H.; Zare, M. Neuroprotective effect of quercetin nanocrystal in a 6-hydroxydopamine model of Parkinson disease: Biochemical and behavioral evidence. Basic Clin. Neurosci. 2018, 9, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Kumar, P. Piperine in combination with quercetin halt 6-OHDA induced neurodegeneration in experimental rats: Biochemical and neurochemical evidences. Neurosci. Res. 2018, 133, 38–47. [Google Scholar] [CrossRef]
- Kale, A.; Pişkin, O.; Baş, Y.; Aydln, B.G.; Can, M.; Elmas, Ö.; Büyükuysal, Ç. Neuroprotective effects of quercetin on radiation-induced brain injury in rats. J. Radiat. Res. 2018, 59, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Omar, S.H.; Scott, C.J.; Hamlin, A.S.; Obied, H.K. Biophenols: Enzymes (β-secretase, Cholinesterases, histone deacetylase and tyrosinase) inhibitors from olive (Olea europaea L.). Fitoterapia 2018, 128, 118–129. [Google Scholar] [CrossRef]
- Du, G.; Zhao, Z.; Chen, Y.; Li, Z.; Tian, Y.; Liu, Z.; Liu, B.; Song, J. Quercetin protects rat cortical neurons against traumatic brain injury. Mol. Med. Rep. 2018, 17, 7859–7865. [Google Scholar] [CrossRef]
- Ghahremani, S.; Soodi, M.; Atashi, A. quercetin ameliorates chlorpyrifos-induced oxidative stress in the rat brain: Possible involvment of PON2 pathway. J. Food Biochem. 2018, 42, 3. [Google Scholar] [CrossRef]
- Oliveira, A.I.; Pinho, C.; Fonte, P.; Sarmento, B.; Dias, A.C.P. Development, characterization, antioxidant and hepatoprotective properties of poly(Ɛ-caprolactone) nanoparticles loaded with a neuroprotective fraction of Hypericum perforatum. Int. J. Biol. Macromol. 2018, 110, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Park, D.-J.; Shah, F.-A.; Koh, P.-O. Quercetin attenuates neuronal cells damage in a middle cerebral artery occlusion animal model. J. Vet. Med. Sci. 2018, 80, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Türedi, S.; Yuluğ, E.; Alver, A.; Bodur, A.; İnce, İ. A morphological and biochemical evaluation of the effects of quercetin on experimental sciatic nerve damage in rats. Exp. Ther. Med. 2018, 15, 3215–3224. [Google Scholar] [PubMed]
- Wang, D.; Zhao, J.; Li, S.; Shen, G.; Hu, S. quercetin attenuates domoic acid-induced cognitive deficits in mice. Nutr. Neurosci. 2018, 21, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, H.; Wen, G.; Li, L.; Gao, Y.; Zhuang, Z.; Zhou, M.; Mao, L.; Fan, Y. Neuroprotection by quercetin via mitochondrial function adaptation in traumatic brain injury: PGC-1α pathway as a potential mechanism. J. Cell. Mol. Med. 2018, 22, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Rahvar, M.; Owji, A.A.; Mashayekhi, F.J. Effect of quercetin on the brain-derived neurotrophic factor gene expression in the rat brain. Bratisl. Med. J. 2018, 119, 28–31. [Google Scholar] [CrossRef]
- Ghosh, S.; Sarkar, S.; Choudhury, S.T.; Ghosh, T.; Das, N. Triphenyl phosphonium coated nano-quercetin for oral delivery: Neuroprotective effects in attenuating age related global moderate cerebral ischemia reperfusion injury in rats. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 2439–2450. [Google Scholar] [CrossRef]
- Godoy, J.A.; Lindsay, C.B.; Quintanilla, R.A.; Carvajal, F.J.; Cerpa, W.; Inestrosa, N.C. Quercetin Exerts Differential Neuroprotective Effects Against H2O2 and Aβ Aggregates in Hippocampal Neurons: The Role of Mitochondria. Mol. Neurobiol. 2017, 54, 7116–7128. [Google Scholar] [CrossRef] [PubMed]
- Soares, J.J.; Rodrigues, D.T.; Gonçalves, M.B.; Lemos, M.C.; Gallarreta, M.S.; Bianchini, M.C.; Gayer, M.C.; Puntel, R.L.; Roehrs, R.; Denardin, E.L.G. Paraquat exposure-induced Parkinson’s disease-like symptoms and oxidative stress in Drosophila melanogaster: Neuroprotective effect of Bougainvillea glabra Choisy. Biomed. Pharmacother. 2017, 95, 245–251. [Google Scholar] [CrossRef]
- Bhatia, G.; Dhuna, V.; Dhuna, K.; Kaur, M.; Singh, J. Bacopa monnieri extracts prevent hydrogen peroxide-induced oxidative damage in a cellular model of neuroblastoma IMR32 cells. Chin. J. Nat. Med. 2017, 15, 834–846. [Google Scholar] [CrossRef]
- Ademosun, A.O.; Oboh, G.; Oyeleye, S.I.; Ejakpovi, I.I.; Adewuni, T.M. Modulation of cholinergic, monoaminergic, and purinergic enzymes of the brain functions by bitter (Vernonia amygdalina) and water bitter (Struchium sparganophora) leaves extracts: Comparison of phenolic constituents versus nootropic potentials. Comp. Clin. Path. 2017, 26, 1267–1272. [Google Scholar] [CrossRef]
- Dong, F.; Wang, S.; Wang, Y.; Yang, X.; Jiang, J.; Wu, D.; Qu, X.; Fan, H.; Yao, R. Quercetin ameliorates learning and memory via the Nrf2-ARE signaling pathway in D-galactose-induced neurotoxicity in mice. Biochem. Biophys. Res. Commun. 2017, 491, 636–641. [Google Scholar] [CrossRef]
- Aluani, D.; Tzankova, V.; Yordanov, Y.; Kondeva-Burdina, M.; Yoncheva, K. In vitro protective effects of encapsulated Quercetin in neuronal models of oxidative stress injury. Biotechnol. Biotechnol. Equip. 2017, 31, 1055–1063. [Google Scholar] [CrossRef]
- Moreno, L.C.G.E.I.; Puerta, E.; Suárez-Santiago, J.E.; Santos-Magalhães, N.S.; Ramirez, M.J.; Irache, J.M. Effect of the oral administration of nanoencapsulated quercetin on a mouse model of Alzheimer’s disease. Int. J. Pharm. 2017, 517, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Shukla, R.K.; Chandravanshi, L.P.; Srivastava, P.; Dhuriya, Y.K.; Shanker, J.; Singh, M.P.; Pant, A.B.; Khanna, V.K. Protective Role of quercetin in Cadmium-Induced Cholinergic Dysfunctions in Rat Brain by Modulating Mitochondrial Integrity and MAP Kinase Signaling. Mol. Neurobiol. 2017, 54, 4560–4583. [Google Scholar] [CrossRef]
- Li, Y.-L.; Guo, H.; Zhao, Y.-Q.; Li, A.-F.; Ren, Y.-Q.; Zhang, J.-W. Quercetin protects neuronal cells from oxidative stress and cognitive degradation induced by amyloid β-peptide treatment. Mol. Med. Rep. 2017, 16, 1573–1577. [Google Scholar] [CrossRef]
- Živković, J.Č.; Barreira, J.C.M.; Šavikin, K.P.; Alimpić, A.Z.; Stojković, D.S.; Dias, M.I.; Santos-Buelga, C.; Duletić-Laušević, S.N.; Ferreira, I.C.F.R. Chemical Profiling and Assessment of Antineurodegenerative and Antioxidant Properties of Veronica teucrium L. and Veronica jacquinii Baumg. Chem. Biodivers. 2017, 14, e1700167. [Google Scholar] [CrossRef]
- Cho, C.H.; Jang, H.; Lee, M.; Kang, H.; Heo, H.J.; Kim, D.-O. Sea buckthorn (Hippophae rhamnoides L.) leaf extracts protect neuronal PC-12 cells from oxidative stress. J. Microbiol. Biotechnol. 2017, 27, 1257–1265. [Google Scholar] [CrossRef]
- Bao, D.; Wang, J.; Pang, X.; Liu, H. Protective Effect of quercetin against Oxidative Stress-Induced Cytotoxicity in Rat Pheochromocytoma (PC-12) Cells. Molecules 2017, 22, 1122. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Jamwal, S.; Kumar, P. Neuroprotective potential of quercetin in combination with piperine against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced neurotoxicity. Neural Regen. Res. 2017, 12, 1137–1144. [Google Scholar] [PubMed]
- Dureshahwar, K.; Mubashir, M.; Une, H. Quantification of quercetin obtained from Allium cepa Lam. leaves and its effects on streptozotocin-induced diabetic neuropathy. Pharmacogn. Res. 2017, 9, 287–293. [Google Scholar]
- Khamphukdee, C.; Chulikhit, Y.; Daodee, S.; Monthakantirat, O. Potential of Alternanthera philoxeroides on improvement of anxiety-like behavior induced by ovariectomized mice model. Indian J. Pharm. Educ. Res. 2017, 51, S494–S497. [Google Scholar] [CrossRef]
- Rocha, M.I.; Rodrigues, M.J.; Pereira, C.; Pereira, H.; da Silva, M.M.; da Neng, N.R.; Nogueira, J.M.F.; Varela, J.; Barreira, L.; Custódio, L. Biochemical profile and in vitro neuroprotective properties of Carpobrotus edulis L., a medicinal and edible halophyte native to the coast of South Africa. S. Afr. J. Bot. 2017, 111, 222–231. [Google Scholar] [CrossRef]
- Eom, M.R.; Weon, J.B.; Jung, Y.S.; Ryu, G.H.; Yang, W.S.; Ma, C.J. Neuroprotective compounds from Reynoutria sachalinensis. Arch. Pharm. Res. 2017, 40, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Ay, M.; Luo, J.; Langley, M.; Jin, H.; Anantharam, V.; Kanthasamy, A.; Kanthasamy, A.G. Molecular mechanisms underlying protective effects of quercetin against mitochondrial dysfunction and progressive dopaminergic neurodegeneration in cell culture and MitoPark transgenic mouse models of Parkinson’s Disease. J. Neurochem. 2017, 141, 766–782. [Google Scholar] [CrossRef]
- Yuan, Z.; Luan, G.; Wang, Z.; Hao, X.; Li, J.; Suo, Y.; Li, G.; Wang, H. Flavonoids from Potentilla parvifolia Fisch. and Their Neuroprotective Effects in Human Neuroblastoma SH-SY5Y Cells in vitro. Chem. Biodivers. 2017, 14, e1600487. [Google Scholar] [CrossRef]
- Oboh, G.; Ademiluyi, A.O.; Ogunsuyi, O.B.; Oyeleye, S.I.; Dada, A.F.; Boligon, A.A. Cabbage and cucumber extracts exhibited anticholinesterase, antimonoamine oxidase and antioxidant properties. J. Food Biochem. 2017, 41, e12358. [Google Scholar] [CrossRef]
- Seo, J.Y.; Kim, S.K.; Nguyen, P.H.; Lee, J.Y.; Tung, P.H.T.; Sung, S.H.; Oh, W.K. Chemical constituents from a Gynostemma laxum and their antioxidant and neuroprotective activities. Chin. Med. 2017, 12, 15. [Google Scholar] [CrossRef] [PubMed]
- Kalemci, O.; Aydin, H.E.; Kizmazoglu, C.; Kaya, I.; Yılmaz, H.; Arda, N.M. Effects of Quercetin and mannitol on erythropoietin levels in rats following acute severe traumatic brain injury. J. Korean Neurosurg. Soc. 2017, 60, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, M.; Singh, A.; Sachchidanand, S.; Bera, A.K. Quercetin inhibits acid-sensing ion channels through a putative binding site in the central vestibular region. Neuroscience 2017, 348, 264–272. [Google Scholar] [CrossRef]
- Kuo, Y.-C.; Tsao, C.-W. Neuroprotection against apoptosis of SK-N-MC cells using RMP-7- and lactoferrin-grafted liposomes carrying quercetin. Int. J. Nanomed. 2017, 12, 2857–2869. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.B.S.; Ruchel, J.B.; Adefegha, S.A.; Coelho, A.P.V.; Trelles, K.B.; Signor, C.; Rubin, M.A.; Oliveira, J.S.; Dornelles, G.L.; de Andrade, C.M.; et al. Neuroprotective effects of pretreatment with quercetin as assessed by acetylcholinesterase assay and behavioral testing in poloxamer-407 induced hyperlipidemic rats. Biomed. Pharmacother. 2017, 88, 1054–1063. [Google Scholar] [CrossRef] [PubMed]
- Adedara, I.A.; Ego, V.C.; Subair, T.I.; Oyediran, O.; Farombi, E.O. Quercetin Improves Neurobehavioral Performance Through Restoration of Brain Antioxidant Status and Acetylcholinesterase Activity in Manganese-Treated Rats. Neurochemical 2017, 42, 1219–1229. [Google Scholar] [CrossRef] [PubMed]
- Baral, S.; Pariyar, R.; Kim, J.; Lee, H.-S.; Seo, J. Quercetin-3-O-glucuronide promotes the proliferation and migration of neural stem cells. Neurobiol. Aging 2017, 52, 39–52. [Google Scholar] [CrossRef]
- Mesram, N.; Nagapuri, K.; Banala, R.R.; Nalagoni, C.R.; Karnati, P.R. Quercetin treatment against NaF induced oxidative stress related neuronal and learning changes in developing rats quercetin treatment against neuronal and learning changes in rats. J. King Saud Univ. Sci. 2017, 29, 221–229. [Google Scholar] [CrossRef]
- Mehta, V.; Parashar, A.; Sharma, A.; Singh, T.R.; Udayabanu, M. Quercetin ameliorates chronic unpredicted stress-mediated memory dysfunction in male Swiss albino mice by attenuating insulin resistance and elevating hippocampal GLUT4 levels independent of insulin receptor expression. Horm. Behav. 2017, 89, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Moghbelinejad, S.; Alizadeh, S.; Mohammadi, G.; Khodabandehloo, F.; Rashvand, Z.; Najafipour, R.; Nassiri-Asl, M. The effects of quercetin on the gene expression of the GABAA receptor α5 subunit gene in a mouse model of kainic acid-induced seizure. J. Physiol. Sci. 2017, 67, 339–343. [Google Scholar] [CrossRef]
- Kim, M.S.; Seo, J.Y.; Oh, J.; Jang, Y.K.; Lee, C.H.; Kim, J.-S. Neuroprotective Effect of Halophyte Salicornia herbacea L. Is Mediated by Activation of Heme Oxygenase-1 in Mouse Hippocampal HT22 Cells. J. Med. Food 2017, 20, 140–151. [Google Scholar] [CrossRef]
- Chen, B.H.; Park, J.H.; Ahn, J.H.; Cho, J.H.; Kim, I.H.; Lee, J.C.; Won, M.-H.; Lee, C.-H.; Hwang, I.K.; Kim, J.-D.; et al. Pretreated quercetin protects gerbil hippocampal CA1 pyramidal neurons from transient cerebral ischemic injury by increasing the expression of antioxidant enzymes. Neural Regen. Res. 2017, 12, 220–227. [Google Scholar]
- Pattanashetti, L.A.; Taranalli, A.D.; Parvatrao, V.; Malabade, R.H.; Kumar, D. Evaluation of neuroprotective effect of quercetin with donepezil in scopolamine-induced amnesia in rats. Indian J. Pharmacol. 2017, 49, 60–64. [Google Scholar] [PubMed]
- Ola, M.S.; Ahmed, M.M.; Shams, S.; Al-Rejaie, S.S. Neuroprotective effects of quercetin in diabetic rat retina. Saudi J. Biol. Sci. 2017, 24, 1186–1194. [Google Scholar] [CrossRef]
- de Souza, S.R.G.; Neto, M.H.M.; Perles, J.V.C.M.; Frez, F.C.V.; Zignani, I.; Ramalho, F.V.; Hermes-Uliana, C.; Bossolani, G.D.P.; Zanoni, J.N. Antioxidant effects of the quercetin in the jejunal myenteric innervation of diabetic rats. Front. Med. 2017, 4, 8. [Google Scholar] [CrossRef][Green Version]
- Hajizadeh Moghaddam, A.; Zare, M.; Rostamian, S.; Khanjani, S. The protective effect of quercetin and it nanocrystal on anxiety-like behaviors induced by animal model of Alzheimer’s disease. J. Med. Plant Res. 2017, 16, 158–168. [Google Scholar]
- Li, J.; Yuan, B.; Wei, C.; Gao, J. Neuroprotective and neurorescue effects of eggplant extract in 6-OHDA-induced Parkinson’s rat model. Biomed. Res. J. 2017, 28, 7220–7229. [Google Scholar]
- Singh, V.; Kahol, A.; Singh, I.P.; Saraf, I.; Shri, R. Evaluation of anti-amnesic effect of extracts of selected Ocimum species using in-vitro and in-vivo models. J. Ethnopharmacol. 2016, 193, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Maciel, R.M.; Carvalho, F.B.; Olabiyi, A.A.; Schmatz, R.; Gutierres, J.M.; Stefanello, N.; Zanini, D.; Rosa, M.M.; Andrade, C.M.; Rubin, M.A.; et al. Neuroprotective effects of quercetin on memory and anxiogenic-like behavior in diabetic rats: Role of ectonucleotidases and acetylcholinesterase activities. Biomed. Pharmacother. 2016, 84, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Martín-Aragón, S.; Jiménez-Aliaga, K.L.; Benedí, J.; Bermejo-Bescós, P. Neurohormetic responses of quercetin and rutin in a cell line over-expressing the amyloid precursor protein (APPswe cells). Phytomedicine 2016, 23, 1285–1294. [Google Scholar] [CrossRef]
- Pogačnik, L.; Pirc, K.; Palmela, I.; Skrt, M.; Kwang, K.S.; Brites, D.; Brito, M.A.; Ulrih, N.P.; Silva, R.F.M. Potential for brain accessibility and analysis of stability of selected flavonoids in relation to neuroprotection in vitro. Brain Res. 2016, 1651, 17–26. [Google Scholar] [CrossRef]
- Kim, M.-S.; Bang, J.H.; Lee, J.; Han, J.-S.; Baik, T.G.; Jeon, W.K. Ginkgo biloba L. extract protects against chronic cerebral hypoperfusion by modulating neuroinflammation and the cholinergic system. Phytomedicine 2016, 23, 1356–1364. [Google Scholar] [CrossRef]
- Du, G.; Zhao, Z.; Chen, Y.; Li, Z.; Tian, Y.; Liu, Z.; Liu, B.; Song, J. Quercetin attenuates neuronal autophagy and apoptosis in rat traumatic brain injury model via activation of PI3K/Akt signaling pathway. Neurol. Res. 2016, 38, 1012–1019. [Google Scholar] [CrossRef]
- Park, E.; Chun, H.S. Protective effects of Quercetin on dieldrin-induced endoplasmic reticulum stress and apoptosis in dopaminergic neuronal cells. NeuroReport 2016, 27, 1140–1146. [Google Scholar] [CrossRef]
- Lee, M.; McGeer, E.G.; McGeer, P.L. Quercetin, not caffeine, is a major neuroprotective component in coffee. Neurobiol. Aging 2016, 46, 113–123. [Google Scholar] [CrossRef]
- Pei, B.; Yang, M.; Qi, X.; Shen, X.; Chen, X.; Zhang, F. Quercetin ameliorates ischemia/reperfusion-induced cognitive deficits by inhibiting ASK1/JNK3/caspase-3 by enhancing the Akt signaling pathway. Biochem. Biophys. Res. Commun. 2016, 478, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Zargar, S.; Siddiqi, N.J.; Ansar, S.; Alsulaimani, M.S.; El Ansary, A.K. Therapeutic role of quercetin on oxidative damage induced by acrylamide in rat brain. Pharm. Biol. 2016, 54, 1763–1767. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Shukla, S.C.; Bhattacharya, P.; Patnaik, R. A possible therapeutic potential of quercetin through inhibition of μ-calpain in hypoxia induced neuronal injury: A molecular dynamics simulation study. Neural Regen. Res. 2016, 11, 1247–1253. [Google Scholar] [CrossRef]
- El-Horany, H.E.; El-Latif, R.N.A.; ElBatsh, M.M.; Emam, M.N. Ameliorative Effect of Quercetin on Neurochemical and Behavioral Deficits in Rotenone Rat Model of Parkinson’s Disease: Modulating Autophagy (Quercetin on Experimental Parkinson’s Disease). J. Biochem. Mol. Toxicol. 2016, 30, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-J.; Bernstock, J.D.; Nagaraja, N.; Ko, B.; Hallenbeck, J.M. Global SUMOylation facilitates the multimodal neuroprotection afforded by quercetin against the deleterious effects of oxygen/glucose deprivation and the restoration of oxygen/glucose. J. Neurochem. 2016, 138, 101–116. [Google Scholar] [CrossRef]
- Sharma, D.R.; Wani, W.Y.; Sunkaria, A.; Kandimalla, R.J.; Sharma, R.K.; Verma, D.; Bal, A.; Gill, K.D. Quercetin attenuates neuronal death against aluminum-induced neurodegeneration in the rat hippocampus. Neuroscience 2016, 324, 163–176. [Google Scholar] [CrossRef]
- Moghbelinejad, S.; Rashvand, Z.; Khodabandehloo, F.; Mo, G.; Nassiri-Asl, M. Modulation of the expression of the GABAa receptor β1 and β3 subunits by pretreatment with Quercetin in the ka model of epilepsy in mice—The effect of Quercetin on GABAA receptor beta subunits. J. Pharmacopunct. 2016, 19, 163–166. [Google Scholar]
- Oboh, G.; Nwanna, E.E.; Oyeleye, S.I.; Olasehinde, T.A.; Ogunsuyi, O.B.; Boligon, A.A. In vitro neuroprotective potentials of aqueous and methanol extracts from Heinsia crinita leaves. Food Sci. Hum. Wellness 2016, 5, 95–102. [Google Scholar] [CrossRef]
- Wu, Y.; Sun, J.; George, J.; Ye, H.; Cui, Z.; Li, Z.; Liu, Q.; Zhang, Y.; Ge, D.; Liu, Y. Study of neuroprotective function of Ginkgo biloba extract (EGb761) derived-flavonoid monomers using a three-dimensional stem cell-derived neural model. Biotechnol. Prog. 2016, 32, 735–744. [Google Scholar] [CrossRef] [PubMed]
- Surapaneni, S.; Prakash, T.; Ansari, M.; Manjunath, P.M.; Kotresha, D.; Goli, D. Study on cerebroprotective actions of Clerodendron glandulosumleaves extract against long term bilateral common carotid artery occlusion in rats. Biomed. Pharmacother. 2016, 80, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Luo, T.; Li, S.; Zhou, Y.; Shen, X.-Y.; He, F.; Xu, J.; Wang, H.-Q. Quercetin protects against Okadaic acid-induced injury via MAPK and PI3K/Akt/GSK3β signaling pathways in HT22 hippocampal neurons. PLoS ONE 2016, 11, e0152371. [Google Scholar]
- Waseem, M.; Parvez, S. Neuroprotective activities of curcumin and quercetin with potential relevance to mitochondrial dysfunction induced by oxaliplatin. Protoplasma 2016, 253, 417–430. [Google Scholar] [CrossRef] [PubMed]
- Kanter, M.; Unsal, C.; Aktas, C.; Erboga, M. Neuroprotective effect of quercetin against oxidative damage and neuronal apoptosis caused by cadmium in hippocampus. Toxicol. Ind. Health 2016, 32, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Mu, X.; Yuan, X.; Du, L.-D.; He, G.-R.; Du, G.-H. Antagonism of quercetin against tremor induced by unilateral striatal lesion of 6-OHDA in rats. J. Asian Nat. Prod. Res. 2016, 18, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Sun, L.; Liu, Z.; Wang, H.; Xu, C. Protection afforded by quercetin against H2O2-induced apoptosis on PC12 cells via activating PI3K/Akt signal pathway. J. Recept. Signal Transduct. 2016, 36, 98–102. [Google Scholar] [CrossRef]
- Zabenko, Y.Y.; Pivneva, T.A. Flavonoid quercetin reduces gliosis after repetitive mild traumatic brain injury in mice. Fiziolohichnyĭ Zhurnal 2016, 62, 50–56. [Google Scholar] [CrossRef]
- Kong, Y.; Li, K.; Fu, T.; Wan, C.; Zhang, D.; Song, H.; Zhang, Y.; Liu, N.; Gan, Z.; Yuan, L. Quercetin ameliorates Aβ toxicity in Drosophila AD model by modulating cell cycle-related protein expression. Oncotarget 2016, 7, 67716–67731. [Google Scholar] [CrossRef]
- Lee, B.K.; Jung, Y.-S. Allium cepa Extract and quercetin Protect Neuronal Cells from Oxidative Stress via PKC- ϵ Inactivation/ERK1/2 Activation. Oxid. Med. Cell. Longev. 2016, 2016, 2495624. [Google Scholar] [CrossRef]
- Kumar, A.; Bora, K.S.; Jaggi, A.S.; Shri, R. Comparative evaluation of neuroprotective effect of three varieties of Allium cepa in chronic constriction injury induced neuropathic pain. Thai J. Pharm. Sci. 2016, 40, 9–20. [Google Scholar]
- Ma, J.-Q.; Luo, R.-Z.; Jiang, H.-X.; Liu, C.-M. Quercitrin offers protection against brain injury in mice by inhibiting oxidative stress and inflammation. Food Funct. 2016, 7, 549–556. [Google Scholar] [CrossRef]
- Nichols, M.; Zhang, J.; Polster, B.M.; Elustondo, P.A.; Thirumaran, A.; Pavlov, E.V.; Robertson, G.S. Synergistic neuroprotection by epicatechin and quercetin: Activation of convergent mitochondrial signaling pathways. Neuroscience 2015, 308, 75–94. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Asaduzzaman, M.; Hossain, M.S.; Sarker, J.; Rahman, S.M.A.; Rashid, M.; Rahman, M.M. In vitro antioxidant and cholinesterase inhibitory activities of methanolic fruit extract of Phyllanthus acidus. BMC Compl. Altern. Med. 2015, 15, 1. [Google Scholar] [CrossRef] [PubMed]
- Mrvová, N.; Škandík, M.; Kuniaková, M.; Račková, L. Modulation of BV-2 microglia functions by novel quercetin pivaloyl ester. Neurochem. Int. 2015, 90, 246–254. [Google Scholar] [CrossRef]
- Chakraborty, J.; Rajamma, U.; Jana, N.; Mohanakumar, K.P. Quercetin improves the activity of the ubiquitin-proteasomal system in 150Q mutated huntingtin-expressing cells but exerts detrimental effects on neuronal survivability. J. Neurosci. Res. 2015, 93, 1581–1591. [Google Scholar] [CrossRef] [PubMed]
- Blasina, F.; Vaamonde, L.; Silvera, F.; Tedesco, A.C.; Dajas, F. Intravenous nanosomes of quercetin improve brain function and hemodynamic instability after severe hypoxia in newborn piglets. Neurochem. Int. 2015, 89, 149–156. [Google Scholar] [CrossRef] [PubMed]
- García-Morales, G.; Huerta-Reyes, M.; González-Cortazar, M.; Zamilpa, A.; Jiménez-Ferrer, E.; Silva-García, R.; Román-Ramos, R.; Aguilar-Rojas, A. Anti-inflammatory, antioxidant and anti-acetylcholinesterase activities of Bouvardia ternifolia: Potential implications in Alzheimer’s disease. Arch. Pharm. Res. 2015, 38, 1369–1379. [Google Scholar] [CrossRef] [PubMed]
- Denny Joseph, K.M. Muralidhara Combined Oral Supplementation of Fish Oil and quercetin Enhances Neuroprotection in a Chronic Rotenone Rat Model: Relevance to Parkinson’s Disease. Neurochem. Res. 2015, 40, 894–905. [Google Scholar] [CrossRef]
- Magalingam, K.B.; Radhakrishnan, A.; Ramdas, P.; Haleagrahara, N. Quercetin Glycosides Induced Neuroprotection by Changes in the Gene Expression in a Cellular Model of Parkinson’s Disease. J. Mol. Neurosci. 2015, 55, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Lei, X.; Chao, H.; Zhang, Z.; Lv, J.; Li, S.; Wei, H.; Xue, R.; Li, F.; Li, Z. Neuroprotective effects of quercetin in a mouse model of brain ischemic/reperfusion injury via anti-apoptotic mechanisms based on the Akt pathway. Mol. Med. Rep. 2015, 12, 3688–3696. [Google Scholar] [CrossRef]
- Ahn, T.-B.; Jeon, B.S. The role of quercetin on the survival of neuron-like PC12 cells and the expression of α-synuclein. Neural Regen. Res. 2015, 10, 1113–1119. [Google Scholar]
- Xia, S.-F.; Xie, Z.-X.; Qiao, Y.; Li, L.-R.; Cheng, X.-R.; Tang, X.; Shi, Y.-H.; Le, G.-W. Differential effects of quercetin on hippocampus-dependent learning and memory in mice fed with different diets related with oxidative stress. Physiol. Behav. 2015, 138, 325–331. [Google Scholar] [CrossRef]
- Unsal, C.; Kanter, M.; Aktas, C.; Erboga, M. Role of quercetin in cadmium-induced oxidative stress, neuronal damage, and apoptosis in rats. Toxicol. Ind. Health 2015, 31, 1106–1115. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, S.; Li, J.; Sun, Y.; Hasimu, H.; Liu, R.; Zhang, T. Quercetin protects human brain microvascular endothelial cells from fibrillar β-amyloid1-40-induced toxicity. Acta Pharm. Sin. B. 2015, 5, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Ashrafpour, M.; Parsaei, S.; Sepehri, H. Quercetin improved spatial memory dysfunctions in rat model of intracerebroventricular streptozotocin-induced sporadic Alzheimer’sdisease. Natl. J. Physiol. Pharm. Pharmacol. 2015, 5, 411–415. [Google Scholar] [CrossRef]
- Sabogal-Guáqueta, A.M.; Muñoz-Manco, J.I.; Ramírez-Pineda, J.R.; Lamprea-Rodriguez, M.; Osorio, E.; Cardona-Gómez, G.P. The flavonoid quercetin ameliorates Alzheimer’s disease pathology and protects cognitive and emotional function in aged triple transgenic Alzheimer’s disease model mice. Neuropharmacology 2015, 93, 134–145. [Google Scholar] [CrossRef]
- Roshanzamir, F.; Yazdanparast, R. Quercetin attenuates cell apoptosis of oxidant-stressed SK-N-MC cells while suppressing up-regulation of the defensive element, HIF-1α. Neuroscience 2014, 277, 780–793. [Google Scholar] [CrossRef]
- Abdalla, F.H.; Schmatz, R.; Cardoso, A.M.; Carvalho, F.B.; Baldissarelli, J.; de Oliveira, J.S.; Rosa, M.M.; Gonçalves Nunes, M.A.; Rubin, M.A.; da Cruz, I.B.M.; et al. Quercetin protects the impairment of memory and anxiogenic-like behavior in rats exposed to cadmium: Possible involvement of the acetylcholinesterase and Na+, K+-ATPase activities. Physiol. Behav. 2014, 135, 152–167. [Google Scholar] [CrossRef] [PubMed]
- Beckmann, D.V.; Carvalho, F.B.; Mazzanti, C.M.; Dos Santos, R.P.; Andrades, A.O.; Aiello, G.; Rippilinger, A.; Graça, D.L.; Abdalla, F.H.; Oliveira, L.S.; et al. Neuroprotective role of quercetin in locomotor activities and cholinergic neurotransmission in rats experimentally demyelinated with ethidium bromide. Life Sci. 2014, 103, 79–87. [Google Scholar] [CrossRef]
- Kumar, P.; Choonara, Y.E.; Modi, G.; Naidoo, D.; Pillay, V. Cur(Que)min: A neuroactive permutation of Curcumin and Quercetin for treating spinal cord injury. Med. Hypotheses 2014, 82, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Qi, D.; Dong, F.; Wang, B.; Guo, R.; Luo, M.; Yao, R. Quercetin improves hypoxia-ischemia induced cognitive deficits via promoting remyelination in neonatal rat. Brain Res. 2014, 1553, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.-S.; Wang, J.-L.; Feng, D.-Y.; Qin, H.-Z.; Wen, H.; Yin, Z.-M.; Gao, G.-D.; Li, C. Protective effect of quercetin against oxidative stress and brain edema in an experimental rat model of subarachnoid haemorrhage. Int. J. Med. Sci. 2014, 11, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.-C.; Yang, Y.-R.; Wang, P.S.; Wang, R.-Y. Quercetin enhances exercise-mediated neuroprotective effects in brain ischemic rats. Med. Sci. Sports Exerc. 2014, 46, 1908–1916. [Google Scholar] [CrossRef]
- Kumar, B.; Gupta, S.K.; Nag, T.C.; Srivastava, S.; Saxena, R.; Jha, K.A.; Srinivasan, B.P. Retinal neuroprotective effects of quercetin in streptozotocin-induced diabetic rats. Exp. Eye Res. 2014, 125, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Kong, B.; Gu, J.-W.; Kuang, Y.-Q.; Cheng, L.; Yang, W.-T.; Xia, X.; Shu, H.-F. Anti-apoptotic and anti-oxidative roles of quercetin after traumatic brain injury. Cell. Mol. Neurobiol. 2014, 34, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.; Liang, X.C.; Gu, B.; Liu, W. Quercetin alleviates high glucose-induced Schwann cell damage by autophagy. Neural Regen. Res. 2014, 9, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.-T.; Lu, C.-L.; Jiang, J.-G. Bioactivity evaluation of ingredients identified from the fruits of Amomum tsaoko Crevost et Lemaire, a Chinese spice. Food Funct. 2014, 5, 1747–1754. [Google Scholar] [CrossRef]
- Mathew, M.; Subramanian, S. In vitro evaluation of anti-Alzheimer effects of dry ginger (Zingiber officinale Roscoe) extract. Indian J. Exp. Biol. 2014, 52, 606–612. [Google Scholar] [PubMed]
- Rinwa, P.; Kumar, A. Quercetin suppress microglial neuroinflammatory response and induce antidepressent-like effect in olfactory bulbectomized rats. Neuroscience 2013, 255, 86–98. [Google Scholar] [CrossRef] [PubMed]
- Prasad, J.; Baitharu, I.; Sharma, A.K.; Dutta, R.; Prasad, D.; Singh, S.B. Quercetin reverses hypobaric hypoxia-induced hippocampal neurodegeneration and improves memory function in the rat. High Alt. Med. Biol. 2013, 14, 383–394. [Google Scholar] [CrossRef]
- Ferreira, P.E.B.; Lopes, C.R.P.; Alves, A.M.P.; Alves, É.P.B.; Linden, D.R.; Zanoni, J.N.; Buttow, N.C. Diabetic neuropathy: An evaluation of the use of quercetin in the cecum of rats. World J. Gastroenterol. 2013, 19, 6416–6426. [Google Scholar] [CrossRef]
- Pandey, A.K.; Bhattacharya, P.; Shukla, S.C.; Paul, S.; Patnaik, R. Neuroprotective effects of Quercetin in chemical hypoxia: In silico evaluation of the hypothesis exploring PKC inhibition-mediated pharmacotherapy. Med. Chem. Res. 2013, 22, 4836–4841. [Google Scholar] [CrossRef]
- Lu, C.-W.; Lin, T.-Y.; Wang, S.-J. Quercetin inhibits depolarization-evoked glutamate release in nerve terminals from rat cerebral cortex. NeuroToxicology 2013, 39, 1–9. [Google Scholar] [CrossRef]
- Selvakumar, K.; Bavithra, S.; Ganesh, L.; Krishnamoorthy, G.; Venkataraman, P.; Arunakaran, J. Polychlorinated biphenyls induced oxidative stress mediated neurodegeneration in hippocampus and behavioral changes of adult rats: Anxiolytic-like effects of quercetin. Toxicol. Lett. 2013, 222, 45–54. [Google Scholar] [CrossRef]
- Abdalla, F.H.; Cardoso, A.M.; Pereira, L.B.; Schmatz, R.; Gonçalves, J.F.; Stefanello, N.; Fiorenza, A.M.; Gutierres, J.M.; Da Silva Serres, J.D.; Zanini, D.; et al. Neuroprotective effect of quercetin in ectoenzymes and acetylcholinesterase activities in cerebral cortex synaptosomes of cadmium-exposed rats. Mol. Cell Biochem. 2013, 381, 1–8. [Google Scholar] [CrossRef]
- Costa, L.G.; Tait, L.; De Laat, R.; Dao, K.; Giordano, G.; Pellacani, C.; Cole, T.B.; Furlong, C.E. Modulation of paraoxonase 2 (PON2) in mouse brain by the polyphenol quercetin: A mechanism of neuroprotection? Neurochem. Res. 2013, 38, 1809–1818. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Liang, X.-C.; Zhang, H.; Wu, Q.-L.; Qu, L.; Sun, Q. Quercetin protects rat dorsal root ganglion neurons against high glucose-induced injury in vitro through Nrf-2/HO-1 activation and NF-κB inhibition. Acta Pharmacol. Sin. 2013, 34, 1140–1148. [Google Scholar] [CrossRef] [PubMed]
- Gómez del Rio, M.A.; Sánchez-Reus, M.I.; Iglesias, I.; Pozo, M.A.; García-Arencibia, M.; Fernández-Ruiz, J.; García-García, L.; Delgado, M.; Benedí, J. Neuroprotective properties of standardized extracts of hypericum perforatum on rotenone model of parkinson’s disease. CNS Neurol. Disord. Drug Targets 2013, 12, 665–679. [Google Scholar] [CrossRef]
- Tangsaengvit, N.; Kitphati, W.; Tadtong, S.; Bunyapraphatsara, N.; Nukoolkarn, V. Neurite outgrowth and neuroprotective effects of quercetin from caesalpinia mimosoides lamk. on CULTURED P19-derived neurons. Evid. Based Compl. Altern. Med. 2013, 2013, 838051. [Google Scholar] [CrossRef]
- Selvakumar, K.; Prabha, R.L.; Saranya, K.; Bavithra, S.; Krishnamoorthy, G.; Arunakaran, J. Polychlorinated biphenyls impair blood-brain barrier integrity via disruption of tight junction proteins in cerebrum, cerebellum and hippocampus of female Wistar rats: Neuropotential role of quercetin. Hum. Exp. Toxicol. 2013, 32, 706–720. [Google Scholar] [CrossRef] [PubMed]
- Annapurna, A.; Ansari, M.A.; Manjunath, P.M. Partial role of multiple pathways in infarct size limiting effect of quercetin and rutin against cerebral ischemia-reperfusion injury in rats. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 491–500. [Google Scholar] [PubMed]
- Sharma, D.R.; Wani, W.Y.; Sunkaria, A.; Kandimalla, R.J.L.; Verma, D.; Cameotra, S.S.; Gill, K.D. Quercetin protects against chronic aluminum-induced oxidative stress and ensuing biochemical, cholinergic, and neurobehavioral impairments in rats. Neurotox. Res. 2013, 23, 336–357. [Google Scholar] [CrossRef]
- Karuppagounder, S.S.; Madathil, S.K.; Pandey, M.; Haobam, R.; Rajamma, U.; Mohanakumar, K.P. Quercetin up-regulates mitochondrial complex-I activity to protect against programmed cell death in rotenone model of Parkinson’s disease in rats. Neuroscience 2013, 236, 136–148. [Google Scholar] [CrossRef]
- Liu, R.; Zhang, T.-T.; Zhou, D.; Bai, X.-Y.; Zhou, W.-L.; Huang, C.; Song, J.-K.; Meng, F.-R.; Wu, C.-X.; Li, L.; et al. Quercetin protects against the Aβ25-35-induced amnesic injury: Involvement of inactivation of RAGE-mediated pathway and conservation of the NVU. Neuropharmacology 2013, 67, 419–431. [Google Scholar] [CrossRef]
- Haleagrahara, N.; Siew, C.J.; Ponnusamy, K. Effect of quercetin and desferrioxamine on 6-hydroxydopamine (6-OHDA) induced neurotoxicity in striatum of rats. J. Toxicol. Sci. 2013, 38, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Denny Joseph, K.M. Muralidhara Enhanced neuroprotective effect of fish oil in combination with quercetin against 3-nitropropionic acid induced oxidative stress in rat brain. Prog. Neuropsychopharmacol. Biol. Psychiatry 2013, 40, 83–92. [Google Scholar] [CrossRef]
- Yang, E.-J.; Kim, G.-S.; Kim, J.; Song, K.-S. Protective effects of onion-derived quercetin on glutamate-mediated hippocampal neuronal cell death. Pharmacogn. Mag. 2013, 9, 302–308. [Google Scholar] [CrossRef]
- Dal Belo, C.A.; Lucho, A.P.D.B.; Vinadé, L.; Rocha, L.; Seibert França, H.; Marangoni, S.; Rodrigues-Simioni, L. In vitro antiophidian mechanisms of Hypericum brasiliense choisy standardized extract: Quercetin-dependent neuroprotection. BioMed Res. Int. 2013, 2013, 943520. [Google Scholar] [CrossRef] [PubMed]
- Selvakumar, K.; Bavithra, S.; Krishnamoorthy, G.; Ganesh, A.; Venkataraman, P.; Arunakaran, J. Impact of quercetin on PCBs (Aroclor-1254)-induced impairment of dopaminergic receptors expression in hippocampus of adult male Wistar rats. Biomed. Prev. Nutr. 2013, 3, 42–52. [Google Scholar] [CrossRef]
- Keddy, P.G.W.; Dunlop, K.; Warford, J.; Samson, M.L.; Jones, Q.R.D.; Rupasinghe, H.P.V.; Robertson, G.S. Neuroprotective and Anti-Inflammatory Effects of the Flavonoid-Enriched Fraction AF4 in a Mouse Model of Hypoxic-Ischemic Brain Injury. PLoS ONE 2012, 7, 12. [Google Scholar] [CrossRef]
- Lopes, C.R.P.; Ferreira, P.E.B.; Zanoni, J.N.; Alves, A.M.P.; Alves, É.P.B.; Buttow, N.C. Neuroprotective effect of quercetin on the duodenum enteric nervous system of streptozotocin-induced diabetic rats. Dig. Dis. Sci. 2012, 57, 3106–3115. [Google Scholar] [CrossRef] [PubMed]
- Raygude, K.S.; Kandhare, A.D.; Ghosh, P.; Ghule, A.E.; Bodhankar, S.L. Evaluation of ameliorative effect of quercetin in experimental model of alcoholic neuropathy in rats. Inflammopharmacology 2012, 20, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Yao, R.-Q.; Qi, D.-S.; Yu, H.-L.; Liu, J.; Yang, L.-H.; Wu, X.-X. Quercetin attenuates cell apoptosis in focal cerebral ischemia rat brain via activation of BDNF-TrkB-PI3K/Akt signaling pathway. Neurochem. Res. 2012, 37, 2777–2786. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, S.F.; Nabavi, S.M.; Latifi, A.M.; Mirzaei, M.; Habtemariam, S.; Moghaddam, A.H. Mitigating role of quercetin against sodium fluoride-induced oxidative stress in the rat brain. Pharm. Biol. 2012, 50, 1380–1383. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Angeline, M.S.; Anand, K.; Ambasta, R.K.; Kumar, P. Naringenin and Quercetin reverse the effect of hypobaric hypoxia and elicit neuroprotective response in the murine model. Brain Res. 2012, 1481, 59–70. [Google Scholar] [CrossRef]
- Kandhare, A.D.; Raygude, K.S.; Shiva Kumar, V.; Rajmane, A.R.; Visnagri, A.; Ghule, A.E.; Ghosh, P.; Badole, S.L.; Bodhankar, S.L. Ameliorative effects quercetin against impaired motor nerve function, inflammatory mediators and apoptosis in neonatal streptozotocin-induced diabetic neuropathy in rats. Biomed. Aging Pathol. 2012, 2, 173–186. [Google Scholar] [CrossRef]
- Park, J.H.; Jung, J.W.; Ahn, Y.-J.; Kwon, H.W. Neuroprotective properties of phytochemicals against paraquat-induced oxidative stress and neurotoxicity in Drosophila melanogaster. Pestic. Biochem. Physiol. 2012, 104, 118–125. [Google Scholar] [CrossRef]
- Mansoorali, K.P.; Prakash, T.; Kotresha, D.; Prabhu, K.; Rama Rao, N. Cerebroprotective effect of Eclipta alba against global model of cerebral ischemia induced oxidative stress in rats. Phytomedicine 2012, 19, 1108–1116. [Google Scholar] [CrossRef]
- Bournival, J.; Plouffe, M.; Renaud, J.; Provencher, C.; Martinoli, M.-G. Quercetin and sesamin protect dopaminergic cells from MPP+-induced neuroinflammation in a microglial (N9)-neuronal (PC12) coculture system. Oxid. Med. Cell. Longev. 2012, 2012, 921941. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Verma, S.; Bhattacharya, P.; Paul, S.; Mishra, A.; Patnaik, R. An in-silico strategy to explore neuroprotection by quercetin in cerebral ischemia: A novel hypothesis based on inhibition of matrix metalloproteinase (MMPs) and acid sensing ion channel 1a (ASIC1a). Med. Hypotheses 2012, 79, 76–81. [Google Scholar] [CrossRef]
- Jazvinšćak Jembrek, M.; Vuković, L.; Puhović, J.; Erhardt, J.; Oršolić, N. Neuroprotective effect of quercetin against hydrogen peroxide-induced oxidative injury in P19 neurons. J. Mol. Neurosci. 2012, 47, 286–299. [Google Scholar] [CrossRef]
- Yang, E.-J.; Kim, S.-I.; Park, S.-Y.; Bang, H.-Y.; Jeong, J.H.; So, J.-H.; Rhee, I.-K.; Song, K.-S. Fermentation enhances the in vitro antioxidative effect of onion (Allium cepa) via an increase in quercetin content. Food Chem. Toxicol. 2012, 50, 2042–2048. [Google Scholar] [CrossRef] [PubMed]
- Cho, N.; Choi, J.H.; Yang, H.; Jeong, E.J.; Lee, K.Y.; Kim, Y.C.; Sung, S.H. Neuroprotective and anti-inflammatory effects of flavonoids isolated from Rhus verniciflua in neuronal HT22 and microglial BV2 cell lines. Food Chem. Toxicol. 2012, 50, 1940–1945. [Google Scholar] [CrossRef] [PubMed]
- Selvakumar, K.; Bavithra, S.; Suganthi, M.; Benson, C.S.; Elumalai, P.; Arunkumar, R.; Krishnamoorthy, G.; Venkataraman, P.; Arunakaran, J. Protective role of quercetin on PCBs-induced oxidative stress and apoptosis in hippocampus of adult rats. Neurochem. Res. 2012, 37, 708–721. [Google Scholar] [CrossRef] [PubMed]
- Prakash, R.; Jayashree, V.; Pranavswarup, G. Neuroprotective effect of flavonoids in global cerebral ischemic conditions. Res. J. Pharm. Biol. Chem. Sci. 2012, 3, 1161–1176. [Google Scholar]
- Selvakumar, K.; Bavithra, S.; Krishnamoorthy, G.; Venkataraman, P.; Arunakaran, J. Polychlorinated biphenyls-induced oxidative stress on rat hippocampus: A neuroprotective role of quercetin. Sci. World J. 2012, 2012, 980314. [Google Scholar] [CrossRef] [PubMed]
- Bavithra, S.; Selvakumar, K.; Pratheepa Kumari, R.; Krishnamoorthy, G.; Venkataraman, P.; Arunakaran, J. Polychlorinated Biphenyl (PCBs)-induced oxidative stress plays a critical role on cerebellar dopaminergic receptor expression: Ameliorative role of quercetin. Neurotox. Res. 2012, 21, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Barreira, J.C.M.; Arraibi, A.A.; Ferreira, I.C.F.R. Bioactive and functional compounds in apple pomace from juice and cider manufacturing: Potential use in dermal formulations. Trends Food Sci. Technol. 2019, 90, 76–87. [Google Scholar] [CrossRef]
- Diñeiro García, Y.; Valles, B.S.; Picinelli Lobo, A. Phenolic and antioxidant composition of by-products from the cider industry: Apple pomace. Food Chem. 2009, 117, 731–738. [Google Scholar] [CrossRef]
- Sójka, M.; Kołodziejczyk, K.; Milala, J.; Abadias, M.; Viñas, I.; Guyot, S.; Baron, A. Composition and properties of the polyphenolic extracts obtained from industrial plum pomaces. J. Funct. Foods 2015, 12, 168–178. [Google Scholar] [CrossRef]
- Marina, Z.; Noriham, A. Quantification of total phenolic compound and in vitro antioxidant potential of fruit peel extracts. Int. Food Res. J. 2014, 21, 1925–1929. [Google Scholar]
- Vu, H.T.; Scarlett, C.J.; Vuong, Q.V. Phenolic compounds within banana peel and their potential uses: A review. J. Funct. Foods 2018, 40, 238–248. [Google Scholar] [CrossRef]
- Wang, Y.C.; Chuang, Y.C.; Hsu, H.W. The flavonoid, carotenoid and pectin content in peels of citrus cultivated in Taiwan. Food Chem. 2008, 106, 277–284. [Google Scholar] [CrossRef]
- Zeyada, N.N.; Zeitoum, M.A.M.; Barbary, O.M. Utilization of some vegetables and fruit waste as natural antioxidants. Alex. J. Food Sci. Technol. 2008, 5, 1–11. [Google Scholar]
- Benítez, V.; Mollá, E.; Martín-Cabrejas, M.A.; Aguilera, Y.; López-Andréu, F.J.; Cools, K.; Terry, L.A.; Esteban, R.M. Characterization of industrial onion wastes (Allium cepa L.): Dietary fibre and bioactive compounds. Plant Foods Hum. Nutr. 2011, 66, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.A.; Kim, K.T.; Kim, H.J.; Chung, M.S.; Chang, P.S.; Park, H.; Pai, H.D. Antioxidant activities of onion (Allium cepa L.) peel extracts produced by ethanol, hot water and subcritical water extraction. Food Sci. Biotechnol. 2014, 23, 615–621. [Google Scholar] [CrossRef]
- Marrelli, M.; Amodeo, V.; Statti, G.; Conforti, F. Biological properties and bioactive components of L.: Focus on potential benefits in the treatment of obesity and related comorbidities. Molecules 2019, 24, 119. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.; Mahato, N.; Lee, Y.R. Extraction, characterization and biological activity of citrus flavonoids. Rev. Chem. Eng. 2019, 35, 265–284. [Google Scholar] [CrossRef]
- Farías-Campomanes, A.M.; A Meireles, M.A. Pisco bagasse as a potential source of bioactive compounds—A review. Recent Pat. Eng. 2013, 7, 41–50. [Google Scholar] [CrossRef]
- Clifton, P.M. Effect of grape seed extract and quercetin on cardiovascular and endothelial parameters in high-risk subjects. J. Biomed. Biotechnol. 2004, 2004, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Castro-Vargas, H.I.; Baumann, W.; Ferreira, S.R.; Parada-Alfonso, F. Valorization of papaya (Carica papaya L.) agroindustrial waste through the recovery of phenolic antioxidants by supercritical fluid extraction. J. Food Sci. Technol. 2019, 56, 3055–3066. [Google Scholar] [CrossRef]
- Yang, H.; Protiva, P.; Cui, B.; Ma, C.; Baggett, S.; Hequet, V.; Mori, S.; Weinstein, I.B.; Kennelly, E.J. New bioactive polyphenols from Theobroma grandiflorum (“cupuaçu”). J. Nat. Prod. 2003, 66, 1501–1504. [Google Scholar] [CrossRef]
- Ballesteros-Vivas, D.; Álvarez-Rivera, G.; Morantes, S.J.; Sánchez-Camargo, A.P.; Ibáñez, E.; Parada-Alfonso, F.; Cifuentes, A. An integrated approach for the valorization of mango seed kernel: Efficient extraction solvent selection, phytochemical profiling and antiproliferative activity assessment. Food Res. Int. 2019, 126, 108616. [Google Scholar] [CrossRef]
- Yang, L.; Li, X.-F.; Zhang, Y.-O.; Cai, G.-P. Suppressive effects of quercetin-3-O-(6″-feruloyl)-β-D-galactopyranoside on adipogenesis in 3T3-L1 preadipocytes through down-regulation of PPARγ and C/EBPα expression. Phytother. Res. 2012, 26, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Teleszko, M.; Wojdyło, A. Comparison of phenolic compounds and antioxidant potential between selected edible fruits and their leaves. J. Funct. Foods 2015, 14, 736–746. [Google Scholar] [CrossRef]
- Sagar, N.A.; Pareek, S.; Sharma, S.; Yahia, E.M.; Lobo, M.G. Fruit and vegetable waste: Bioactive compounds, their extraction, and possible utilization. Compr. Rev. Food Sci. Food Saf. 2018, 17, 512–531. [Google Scholar] [CrossRef] [PubMed]
- Benzarti, S.; Hamdi, H.; Lahmayer, I.; Toumi, W.; Kerkeni, A.; Belkadhi, K.; Sebei, H. Total phenolic compounds and antioxidant potential of quince (Cydonia oblonga Miller) leaf methanol extract. Int. J. Innov. Appl. Stud. 2015, 13, 518. [Google Scholar]
- Silva, P.; Ferreira, S.; Nunes, F.M. Elderberry (Sambucus nigra L.) by-products a source of anthocyanins and antioxidant polyphenols. Ind. Crop Prod. 2017, 95, 227–234. [Google Scholar] [CrossRef]
- Thomas, M.; Badr, A.; Desjardins, Y.; Gosselin, A.; Angers, P. Characterization of industrial broccoli discards (Brassica oleracea var. italica) for their glucosinolate, polyphenol and flavonoid contents using UPLC MS/MS and spectrophotometric methods. Food Chem. 2018, 245, 4–1211. [Google Scholar] [CrossRef]
- Gonzales, G.B.; Raes, K.; Coelus, S.; Struijs, K.; Smagghe, G.; Van Camp, J. Ultra(high)-pressure liquid chromatography-ele trospray ionization-time-of-flight-ion mobility-high definition mass spectrometry for the rapid identification and structural characterization of flavonoid glycosides from cauliflower waste. J. Chromatogr. A 2014, 1323, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Huynh, N.T.; Smagghe, G.; Gonzales, G.B.; Camp, J.-V.; Raes, K. Enzyme-Assisted Extraction Enhancing the Phenolic Release from Cauliflower (Brassica oleracea L. var. botrytis) Outer Leaves. J. Agric. Food Chem. 2014, 62, 7468–7476. [Google Scholar] [CrossRef]
- Zheng, R.; Su, S.; Zhou, H.; Yan, H.; Ye, J.; Zhao, Z.; You, L.; Fu, X. Antioxidant/antihyperglycemic activity of phenolics from sugarcane (Saccharum officinarum L.) bagasse and identification by UHPLC-HR TOFMS. Ind. Crops. Prod. 2017, 101, 104–114. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, M.; Zhao, Z.; Yu, S. The antibiotic activity and mechanisms of sugarcane (Saccharum officinarum L.) bagasse extract against food-borne pathogens. Food Chem. 2015, 185, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Marques, T.R.; Caetano, A.A.; Simão, A.A.; de O. Castro, F.C.; de Oliveira, R.V.; Corrêa, A.D. Methanolic extract of Malpighia emarginata bagasse: Phenolic Compounds and inhibitory potential on digestive enzymes. Braz. J. Pharmacog. 2016, 26, 191–196. [Google Scholar] [CrossRef]
- Zhang, F.; Yang, Y.; Su, P.; Guo, Z. Microwave-assisted Extraction of Rutin and Quercetin from the Stalks of Euonymus alatus (Thunb.) Sieb. Phytochem. Anal. 2009, 20, 33–37. [Google Scholar] [CrossRef]
- Balaraman, H.B.; Sivasubramaniyam, A.; Rathnasamy, S.K. High selective purification of Quercetin from Peanut hull using protic deep eutectic mixture based liquid–liquid microextraction. Microchem. J. 2020, 152, 104444. [Google Scholar] [CrossRef]
- Flórez-Montes, C.; Mosquera-Martínez, Ó.M.; Rojas-González, A.F. Prospective use of fruit byproducts in Colombia according to their antioxidant capacity [Uso prospectivo de subproductos de frutas en Colombia según su capacidad antioxidante]. Rev. Acad. Colomb. Cienc. Exact. Fis. Nat. 2020, 44, 1113–1125. [Google Scholar] [CrossRef]
- da Silva-Maia, J.K.; Batista, A.G.; Correa, L.C.; Lima, G.C.; Bogusz Junior, S.; Maróstica Junior, M.R. Aqueous extract of berry (Plinia jaboticaba) byproduct modulates gut microbiota and maintains the balance on antioxidant defense system in rats. J. Food Biochem. 2019, 4, e12705. [Google Scholar] [CrossRef]
- Huang, H.; Belwal, T.; Jiang, L.; Hu, J.; Limwachiranon, J.; Li, L.; Ren, G.; Zhang, X.; Luo, Z. Valorization of lotus byproduct (Receptaculum Nelumbinis) under green extraction condition. Food Bioprod. Process. 2019, 115, 110–117. [Google Scholar] [CrossRef]
- Campone, L.; Celano, R.; Piccinelli, A.L.; Pagano, I.; Carabetta, S.; Sanzo, R.D.; Russo, M.; Ibañez, E.; Cifuentes, A.; Rastrelli, L. Response surface methodology to optimize supercritical carbon dioxide/co-solvent extraction of brown onion skin by-product as source of nutraceutical compounds. Food Chem. 2018, 269, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Frum, A.; Georgescu, C.; Gligor, F.G.; Lengyel, E.; Stegarus, D.I.; Dobrea, C.M.; Tita, O. Identification and quantification of phenolic compounds from red grape pomace. Sci. Stud. Res. Chem. ChemEng. Biotech. Food Ind. 2018, 19, 45–52. [Google Scholar]
- Valadez-Carmona, L.; Plazola-Jacinto, C.P.; Hernández-Ortega, M.; Hernández-Navarro, M.D.; Villarreal, F.; Necoechea-Mondragón, H.; Ortiz-Moreno, A.; Ceballos-Reyes, G. Effects of microwaves, hot air and freeze-drying on the phenolic compounds, antioxidant capacity, enzyme activity and microstructure of cacao pod husks (Theobroma cacao L.). Innov. Food Sci. Emerg. Technol. 2017, 41, 378–386. [Google Scholar] [CrossRef]
- Jelley, R.E.; Herbst-Johnstone, M.; Klaere, S.; Pilkington, L.I.; Grose, C.; Martin, D.; Barker, D.; Fedrizzi, B. Optimization of Ecofriendly Extraction of Bioactive Monomeric Phenolics and Useful Flavor Precursors from Grape Waste. ACS Sustain. Chem. Eng. 2016, 4, 5060–5067. [Google Scholar] [CrossRef]
- Ribeiro, L.F.; Ribani, R.H.; Francisco, T.M.G.; Soares, A.A.; Pontarolo, R.; Haminiuk, C.W.I. Profile of bioactive compounds from grape pomace (Vitis vinifera and Vitis labrusca) by spectrophotometric, chromatographic and spectral analyses. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2015, 1007, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Ekman, A.; Campos, M.; Lindahl, S.; Co, M.; Börjesson, P.; Karlsson, E.N.; Turner, C. Bioresource utilisation by sustainable technologies in new value-added biorefinery concepts—Two case studies from food and forest industry. J. Cleaner Prod. 2013, 57, 46–58. [Google Scholar] [CrossRef]
- Bakowska-Barczak, A.M.; Schieber, A.; Kolodziejczyk, P. Characterization of Canadian black currant (Ribes nigrum L.) seed oils and residues. J. Agr. Food Chem. 2009, 57, 11528–11536. [Google Scholar] [CrossRef] [PubMed]
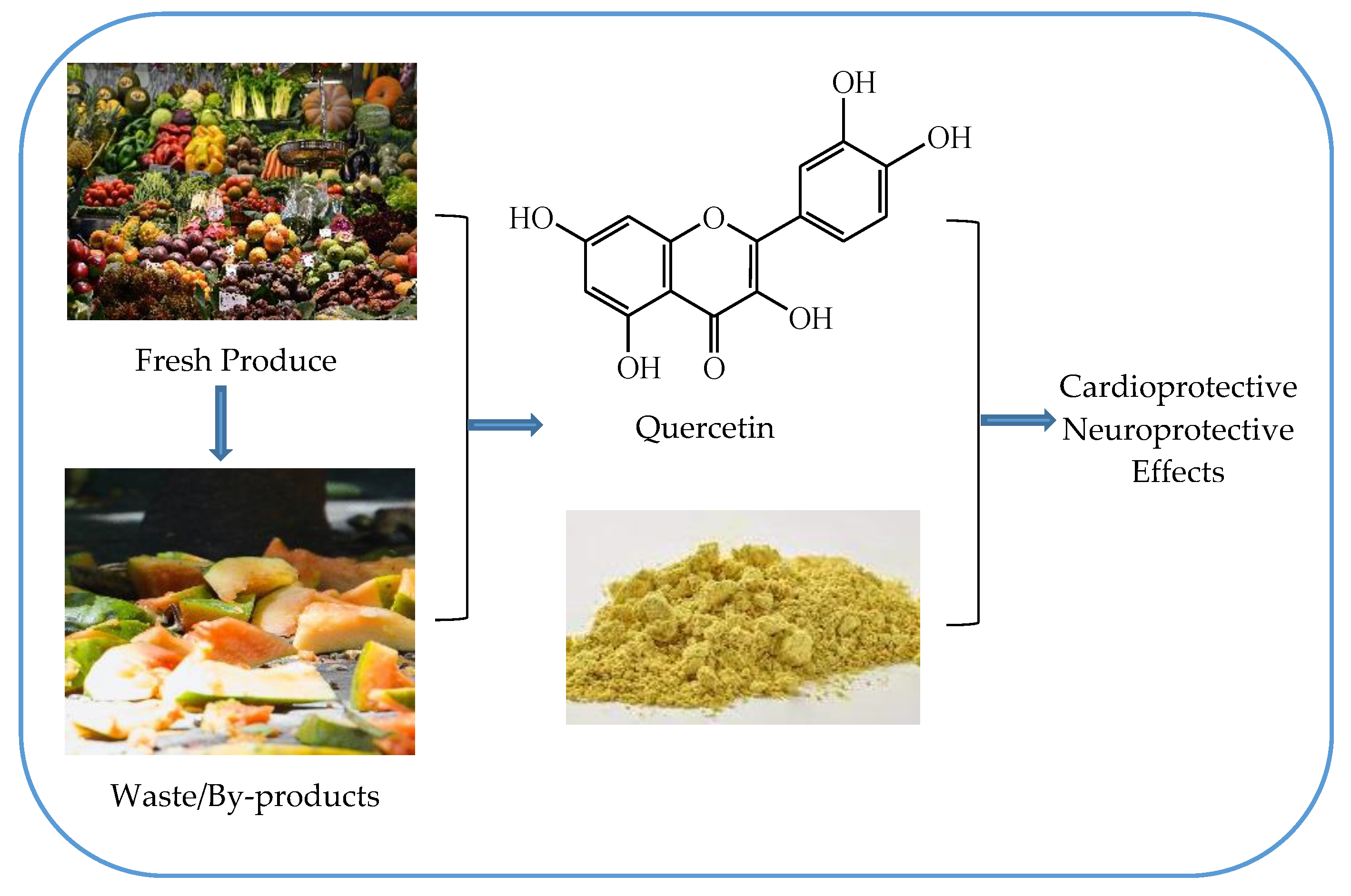
| Role | Mode | Reference |
|---|---|---|
| Translocation of NFκB, and expression of TGF-β1, CTGF, and BNP | In vivo | [43] |
| Cardioprotective effects restoring plasma thyroid hormone levels and attenuating oxidative stress in the heart | In vivo | [44] |
| Inhibition of JNK and p38 mitogen-activated protein kinase signaling pathways | In vivo | [45] |
| Inhibits angiotensin-converting enzyme activity, improves vascular relaxation, and decreases oxidative stress and gene expression | In vitro, in vivo | [46] |
| Post-ischemic recovery of left ventricular developed pressure, as well as recovery of markers of contraction and relaxation, respectively | In vivo | [47] |
| Cardioprotective | In vivo | [48] |
| Myocardial infarction Release of creatine kinase-MB (CK-MB), lactate dehydrogenase (LDH) level in coronary effluent and reduced myocardial infarct size | In vivo | [49] |
| Myocardial ischemia-reperfusion injury via suppressing the NF-κB pathway | In vivo and in vitro | [50] |
| Protein disulfide isomerase (PDI) inhibition for cardiovascular benefits | In vitro | [51] |
| Protects cardiomyocytes against oxidative toxicity and the regulation of stress-sensitive protein kinase cascades and transcription factors. | In vitro | [52] |
| Lowers ABP in patients with hypertension | In vivo | [53] |
| Cardioprotective control | In vitro | [54] |
| Endothelial function and reducing inflammation, vascular function, and cardiometabolic health | In vivo | [55,56] |
| Improve cyclosporine-induced cardiotoxicity, as it has antioxidant and anti-inflammatory enzyme activities | In vivo | [57] |
| Atherogenic and cardioprotective indices | In vivo | [58] |
| Reduction in cardiac and renal markers of oxidative stress | In vivo | [59] |
| Antichronic doxorubicin cardiotoxicity via antioxidant and anti-inflammatory properties | In vivo | [60] |
| Antioxidative cardiotoxicity and dyslipidemia | In vivo | [61] |
| Cardiac weight index and myocardial enzyme activity Antioxidative stress, inhibition of the renin–angiotensin–aldosterone system | In vivo | [62] |
| Ischemia/reperfusion injury in cardiomyocytes | In vitro | [63] |
| Anti-doxorubicin-induced cardiomyopathy in H9c2 cell; myocardial ischemia/reperfusion injury in rats through the PI3K/Akt pathway | In vitro | [64,65] |
| Induce activation of AMPK and eNOS in human aortic endothelial cells | In vivo | [66] |
| Cardioprotective effect of GSK-3b inhibitors | In vivo | [67] |
| Reduction of the serum CK-MB, LDH, and SGPT level enzymes | In vivo | [68] |
| Protects rat hearts from oxidative stress by its antioxidant potential | In vivo | [69] |
| Decrease in doxorubicin-induced cytotoxicity and promoting the cell repair system in cardiomyocyte H9C2 cells | In vitro | [70] |
| Role | Mode | Reference |
|---|---|---|
| Antioxidant and AChE inhibitory activity | In vitro and in vivo | [203] |
| To prevent the increase in AChE activity in the brain, improve the memory and anxiety-like behavior | In vivo | [204] |
| Proteasome activities | In vitro | [205] |
| Against oxidation-induced neuronal necrotic-such as cell death | In vitro | [206] |
| Modulation of neuroinflammation and the cholinergic system | In vivo | [207] |
| Neuronal autophagy and brain injury model by activation of PI3K/Akt signaling pathway | In vivo | [208] |
| Endoplasmic reticulum stress and neuronal cells | In vitro | [209] |
| Reduced oxidative/nitrative damage to DNA, lipids, and proteins of neuroblastoma cell line (SH-SY5Y) cell | In vitro | [210] |
| Improves ischemia/reperfusion-induced cognitive deficits. Inhibition of ASK1/JNK3/caspase-3 Akt signaling pathway | In vitro | [211] |
| Prevention of brain damage by acrylamide | In vivo | [212] |
| Inhibition of μ-calpain protein in hypoxia-induced neuronal injury | In vitro | [213] |
| Autophagy-modulating, Parkinson’s diseases | In vivo | [214] |
| Deprivation and restoration of oxygen/glucose, increased the expression of Nrf2 | In vitro | [215] |
| Neuronal death prevention | In vivo | [216] |
| Anti-convulsant | In vivo | [217] |
| Inhibition of monoamine oxidase (MAO), AChE, and BChE activities | In vitro | [218] |
| Antioxidative insult | In vivo | [219] |
| Cerebroprotective action | In vivo | [220] |
| Prevention of okadaic-acid-induced injury by MAPK and PI3K/Akt/GSK3β signaling pathways | In vitro | [221] |
| Reduction of oxaliplatin-induced oxidative stress in brain | In vitro and in vivo | [222] |
| Reduction of immunoreactivity of degenerating neurons | In vivo | [223] |
| Parkinson’s disease | In vivo | [224] |
| Apoptosis on neural cells via PI3K/Akt signal pathway | In vitro | [225] |
| Cerebrovascular disorders | In vivo | [226] |
| Alzheimer’s disease (AD) prevention | In vivo | [227] |
| Defense of oxidative Stress via PKC- ϵ inactivation/ERK1/2 activation | In vivo | [228] |
| Neuropathic pain reliever | In vitro | [229] |
| Inhibiting oxidative stress and inflammation in brain injury | In vivo | [230] |
| Hypoxic–ischemic brain injury | In vivo | [231] |
| Alzheimer’s disease prevention | In vitro | [232] |
| Anti-neuroinflammatory | In vitro | [233] |
| Enhanced neuronal mitochondrial performance | In vitro | [234] |
| Brain therapy, hypoxia | In vivo | [235] |
| Anti-inflammatory, antioxidant, and anti-acetylcholinesterase activities in | In vitro | [236] |
| Reduction in oxidative-stress-mediated neurodegeneration | In vivo | [237] |
| Prevention of Parkinson’s disease by gene expression | In vitro | [238] |
| Anti-brain ischemic/reperfusion injury using Akt pathway | In vivo | [239] |
| In neuron survival | In vitro | [240] |
| Cognitive function | In vivo | [241] |
| Antioxidative stress, neuronal damage, | In vivo | [242] |
| Protection of human brain cells | In vitro | [243] |
| Spatial memory dysfunctions improvement | In vivo | [244] |
| Protection of cognitive and emotional functions | In vivo | [245] |
| Reduction of cell apoptosis of oxidant-stressed neuroblastoma (SK-N-MC) cells | In vitro | [246] |
| Protects the weakening of memory and anxiogenic behavior | In vitro | [247] |
| Locomotor activities, neurotransmission | In vivo | [248] |
| Spinal cord injury treatment | In vitro | [249] |
| Perinatal cerebral hypoxia–ischemia | In vivo | [250] |
| Protection from oxidative stress and brain edema | In vivo | [251] |
| Brain protection | In vivo and in vitro | [252] |
| Retinal neuroprotection | In vivo | [253] |
| Brain injury treatment | In vivo | [254] |
| Neurolemmocytes damage prevention | In vivo | [255] |
| Protection of PC12 neural cells | In vitro | [256] |
| Multiple therapeutic molecular targets of Alzheimer diseases | In vitro | [257] |
| Reduction of neuroinflammatory response, antidepressant | In vivo | [258] |
| Prevention of hippocampal nerve damage, improved memory function | In vivo | [259] |
| Neuron density | In vivo | [260] |
| Prevention of chemical hypoxia | In vitro | [261] |
| Inhibition of glutamate release | In vitro | [262] |
| Anxiolytic effects | In vivo | [263] |
| Ectoenzymes and acetylcholinesterase activities | In vivo | [264] |
| Increases levels of mitochondrial enzyme (PON2) in brain cells | In vivo | [265] |
| Nerve protection via Nrf-2/HO-1 activation and NF-κB inhibition | In vivo | [266] |
| Catalepsy normalization, improvement of neurochemical parameters | In vivo | [267] |
| Suppression of cellular acetylcholinesterase (AChE), protection against oxidative stress | In vitro | [268] |
| Preventive medicine for polychlorinated biphenyls (PCBs)-induced neurotoxicity | In vivo | [269] |
| Cerebral ischemia–reperfusion injury treatment | In vivo | [270] |
| Protection against induced neurobehavioral impairments | In vivo | [271] |
| Neuroprotection in mitochondrial neurotoxin-induced Parkinson diseases | In vivo | [272] |
| Neurovascular coupling protection, decrease in neurovascular oxidation | In vivo | [273] |
| Neuronal protection | In vivo | [274] |
| Neuroprotection against brain oxidative stress | In vivo | [275] |
| Against neuron death | In vivo | [276] |
| Against neurotoxic venoms | In vitro | [277] |
| Neurodegeneration protection via production of ROS scavenging | In vivo | [278] |
| Neuroprotection in ypoxic–ischemic brain injury | In vivo | [279] |
| Neuroprotection in duodenum enteric nervous system | In vivo | [280] |
| Alcoholic neuropathy protection | In vivo | [281] |
| Protection in cerebral ischemia through activation of BDNF-TrkB-PI3K/Akt signaling pathway | In vivo | [282] |
| Prevention of oxidative stress in brain | In vivo | [283] |
| Reversal of hypobaric hypoxia, neuroprotective response stimulant | In vivo | [284] |
| Diabetic neuropathy prevention | In vivo | [285] |
| Prevention of oxidative damage by induced neurotoxicity | In vitro | [286] |
| Prevention of cerebral ischemia-induced oxidative stress | In vivo | [287] |
| Neuroinflammation prevention | In vitro | [288] |
| Cerebral ischemia protection | In vitro | [289] |
| Protection Oxidative injury P19 neurons | In vitro | [290] |
| Lutamate-induced neurotoxicity protection in HT22 cells | In vitro | [291] |
| Neuron cell protection | In vitro | [292] |
| Oxidative stress | In vivo | [293] |
| Decreases the neuronal damage and scavenged free radicals | In vivo | [294] |
| Protection for cerebral ischemic conditions | In vivo | [295] |
| Neurodegeneration protection | In vivo | [296] |
| Waste/By-Products | Quantity (mg/g) * | Reference |
|---|---|---|
| Tomato peels | 9.97 ± 0.27 | [326] |
| Berry peel | 0.0001 ± 0.00 | [327] |
| Lotus byproducts | (Only detected not quantified) | [328] |
| Coppery onion outer dry layers | 52.84 ± 0.12 | [329] |
| Red grape pomace | 0.05 ± 0.00 | [330] |
| Cacao beans pod husk | 0.6018 ± 0.0112 | [331] |
| Grape pomace | 0.03189 ± 0.00277 | [332] |
| Grape pomace | 0.24923 ± 0.00114 | [333] |
| Onion waste | (A case study of industrial scale, output yield is in Kgs) | [334] |
| Black currant residue with quercetin glycoside (based on the place of cultivation) | 34.6 ± 5.7 | [335] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhat, I.U.H.; Bhat, R. Quercetin: A Bioactive Compound Imparting Cardiovascular and Neuroprotective Benefits: Scope for Exploring Fresh Produce, Their Wastes, and By-Products. Biology 2021, 10, 586. https://doi.org/10.3390/biology10070586
Bhat IUH, Bhat R. Quercetin: A Bioactive Compound Imparting Cardiovascular and Neuroprotective Benefits: Scope for Exploring Fresh Produce, Their Wastes, and By-Products. Biology. 2021; 10(7):586. https://doi.org/10.3390/biology10070586
Chicago/Turabian StyleBhat, Irshad Ul Haq, and Rajeev Bhat. 2021. "Quercetin: A Bioactive Compound Imparting Cardiovascular and Neuroprotective Benefits: Scope for Exploring Fresh Produce, Their Wastes, and By-Products" Biology 10, no. 7: 586. https://doi.org/10.3390/biology10070586
APA StyleBhat, I. U. H., & Bhat, R. (2021). Quercetin: A Bioactive Compound Imparting Cardiovascular and Neuroprotective Benefits: Scope for Exploring Fresh Produce, Their Wastes, and By-Products. Biology, 10(7), 586. https://doi.org/10.3390/biology10070586







