Temporal Gradients Controlling Embryonic Cell Cycle
Simple Summary
Abstract
1. Introduction
2. Activity Gradient of Cell Cycle Checkpoint
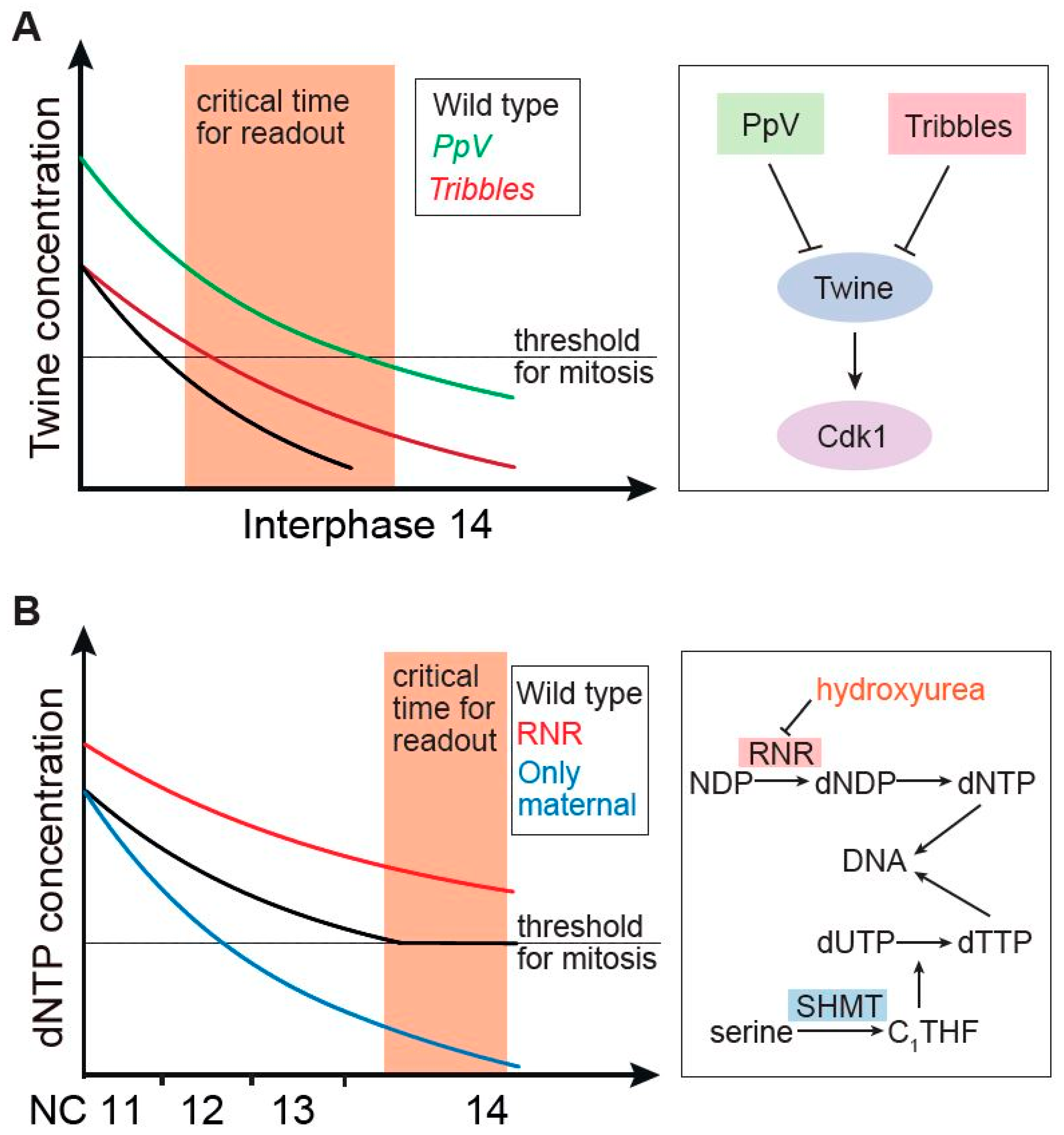
3. Gradient of Cdc25/Twine Decay in Drosophila
4. Temporal Gradient of dNTP Metabolites
5. Temporal Gradient of Free Histone Proteins
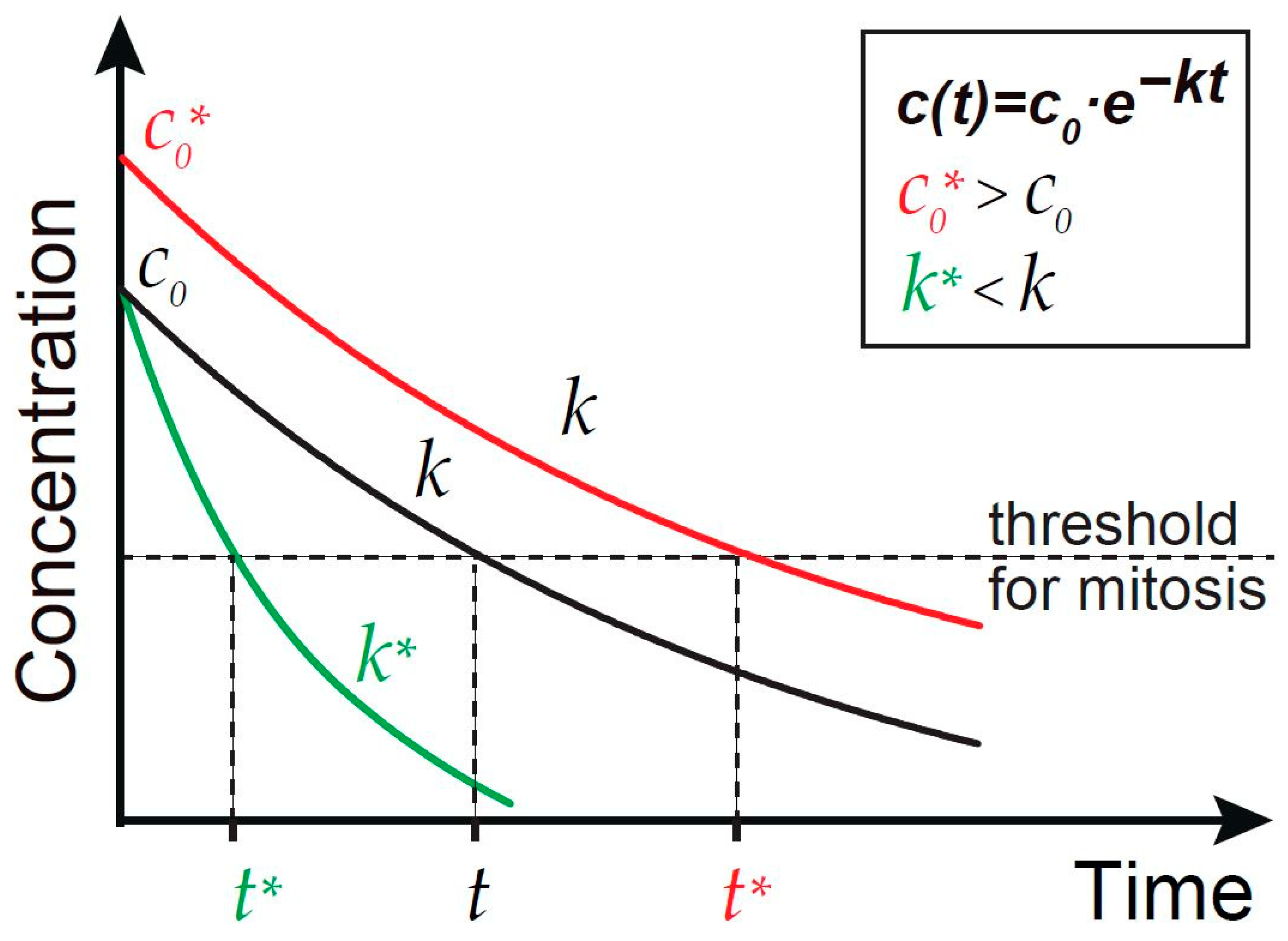
6. Time Scales and Readout of the Gradients
7. Local Response of the Gradients
8. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, B.; Grosshans, J. Link of Zygotic Genome Activation and Cell Cycle Control. Methods Mol. Biol. 2017, 1605, 11–30. [Google Scholar] [CrossRef]
- Vastenhouw, N.L.; Cao, W.X.; Lipshitz, H.D. The maternal-to-zygotic transition revisited. Development 2019, 146, dev161471. [Google Scholar] [CrossRef]
- Farrell, J.A.; O’Farrell, P.H. From egg to gastrula: How the cell cycle is remodeled during the Drosophila mid-blastula transition. Annu. Rev. Genet. 2014, 48, 269–294. [Google Scholar] [CrossRef]
- Liu, B.; Gregor, I.; Muller, H.A.; Grosshans, J. Fluorescence fluctuation analysis reveals PpV dependent Cdc25 protein dynamics in living embryos. PLoS Genet. 2020, 16, e1008735. [Google Scholar] [CrossRef]
- Li, L.; Zhu, S.; Shu, W.; Guo, Y.; Guan, Y.; Zeng, J.; Wang, H.; Han, L.; Zhang, J.; Liu, X.; et al. Characterization of Metabolic Patterns in Mouse Oocytes during Meiotic Maturation. Mol. Cell 2020, 80, 525–540.e9. [Google Scholar] [CrossRef] [PubMed]
- Rodenfels, J.; Neugebauer, K.M.; Howard, J. Heat Oscillations Driven by the Embryonic Cell Cycle Reveal the Energetic Costs of Signaling. Dev. Cell 2020, 53, 492. [Google Scholar] [CrossRef]
- Edgar, B.A.; Kiehle, C.P.; Schubiger, G. Cell cycle control by the nucleo-cytoplasmic ratio in early Drosophila development. Cell 1986, 44, 365–372. [Google Scholar] [CrossRef]
- O’Farrell, P.H.; Stumpff, J.; Su, T.T. Embryonic cleavage cycles: How is a mouse like a fly? Curr. Biol. 2004, 14, R35–R45. [Google Scholar] [CrossRef] [PubMed]
- Blythe, S.A.; Wieschaus, E.F. Coordinating Cell Cycle Remodeling with Transcriptional Activation at the Drosophila MBT. Curr. Top. Dev. Biol. 2015, 113, 113–148. [Google Scholar] [CrossRef] [PubMed]
- Yuan, K.; Seller, C.A.; Shermoen, A.W.; O’Farrell, P.H. Timing the Drosophila Mid-Blastula Transition: A Cell Cycle-Centered View. Trends Genet. 2016, 32, 496–507. [Google Scholar] [CrossRef]
- Kane, D.A.; Kimmel, C.B. The zebrafish midblastula transition. Development 1993, 119, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Newport, J.; Kirschner, M. A major developmental transition in early Xenopus embryos: II. Control of the onset of transcription. Cell 1982, 30, 687–696. [Google Scholar] [CrossRef]
- Newport, J.; Kirschner, M. A major developmental transition in early Xenopus embryos: I. characterization and timing of cellular changes at the midblastula stage. Cell 1982, 30, 675–686. [Google Scholar] [CrossRef]
- Di Talia, S.; She, R.; Blythe, S.A.; Lu, X.; Zhang, Q.F.; Wieschaus, E.F. Posttranslational control of Cdc25 degradation terminates Drosophila’s early cell-cycle program. Curr. Biol. 2013, 23, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Blythe, S.A.; Wieschaus, E.F. Zygotic genome activation triggers the DNA replication checkpoint at the midblastula transition. Cell 2015, 160, 1169–1181. [Google Scholar] [CrossRef]
- Lu, X.; Drocco, J.; Wieschaus, E.F. Cell cycle regulation via inter-nuclear communication during the early embryonic development of Drosophila melanogaster. Cell Cycle 2010, 9, 2908–2910. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Murphy, C.M.; Michael, W.M. Control of DNA replication by the nucleus/cytoplasm ratio in Xenopus. J. Biol. Chem. 2013, 288, 29382–29393. [Google Scholar] [CrossRef]
- Jevtic, P.; Levy, D.L. Nuclear size scaling during Xenopus early development contributes to midblastula transition timing. Curr. Biol. 2015, 25, 45–52. [Google Scholar] [CrossRef]
- Reisser, M.; Palmer, A.; Popp, A.P.; Jahn, C.; Weidinger, G.; Gebhardt, J.C.M. Single-molecule imaging correlates decreasing nuclear volume with increasing TF-chromatin associations during zebrafish development. Nat. Commun. 2018, 9, 5218. [Google Scholar] [CrossRef]
- Chen, H.; Einstein, L.C.; Little, S.C.; Good, M.C. Spatiotemporal Patterning of Zygotic Genome Activation in a Model Vertebrate Embryo. Dev. Cell 2019, 49, 852–866.e7. [Google Scholar] [CrossRef]
- Gentsch, G.E.; Owens, N.D.L.; Smith, J.C. The Spatiotemporal Control of Zygotic Genome Activation. iScience 2019, 16, 485–498. [Google Scholar] [CrossRef]
- Sung, H.W.; Spangenberg, S.; Vogt, N.; Grosshans, J. Number of nuclear divisions in the Drosophila blastoderm controlled by onset of zygotic transcription. Curr. Biol. 2013, 23, 133–138. [Google Scholar] [CrossRef]
- Syed, S.; Wilky, H.; Raimundo, J.; Lim, B.; Amodeo, A.A. The nuclear to cytoplasmic ratio directly regulates zygotic transcription in Drosophila through multiple modalities. Proc. Natl. Acad. Sci. USA 2021, 118, e2010210118. [Google Scholar] [CrossRef]
- Strong, I.J.T.; Lei, X.; Chen, F.; Yuan, K.; O’Farrell, P.H. Interphase-arrested Drosophila embryos activate zygotic gene expression and initiate mid-blastula transition events at a low nuclear-cytoplasmic ratio. PLoS Biol. 2020, 18, e3000891. [Google Scholar] [CrossRef]
- Shermoen, A.W.; McCleland, M.L.; O’Farrell, P.H. Developmental control of late replication and S phase length. Curr. Biol. 2010, 20, 2067–2077. [Google Scholar] [CrossRef]
- Rothe, M.; Pehl, M.; Taubert, H.; Jackle, H. Loss of gene function through rapid mitotic cycles in the Drosophila embryo. Nature 1992, 359, 156–159. [Google Scholar] [CrossRef]
- Djabrayan, N.J.; Smits, C.M.; Krajnc, M.; Stern, T.; Yamada, S.; Lemon, W.C.; Keller, P.J.; Rushlow, C.A.; Shvartsman, S.Y. Metabolic Regulation of Developmental Cell Cycles and Zygotic Transcription. Curr. Biol. 2019, 29, 1193–1198.e5. [Google Scholar] [CrossRef]
- Blythe, S.A.; Wieschaus, E.F. Establishment and maintenance of heritable chromatin structure during early Drosophila embryogenesis. eLife 2016, 5, e20148. [Google Scholar] [CrossRef] [PubMed]
- Hug, C.B.; Grimaldi, A.G.; Kruse, K.; Vaquerizas, J.M. Chromatin Architecture Emerges during Zygotic Genome Activation Independent of Transcription. Cell 2017, 169, 216–228.e19. [Google Scholar] [CrossRef] [PubMed]
- McCleland, M.L.; O’Farrell, P.H. RNAi of mitotic cyclins in Drosophila uncouples the nuclear and centrosome cycle. Curr. Biol. 2008, 18, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Lehner, C.F.; O’Farrell, P.H. The roles of Drosophila cyclins A and B in mitotic control. Cell 1990, 61, 535–547. [Google Scholar] [CrossRef]
- Edgar, B.A.; O’Farrell, P.H. The three postblastoderm cell cycles of Drosophila embryogenesis are regulated in G2 by string. Cell 1990, 62, 469–480. [Google Scholar] [CrossRef]
- Lee, L.A.; Orr-Weaver, T.L. Regulation of cell cycles in Drosophila development: Intrinsic and extrinsic cues. Annu. Rev. Genet. 2003, 37, 545–578. [Google Scholar] [CrossRef]
- Edgar, B.A.; Sprenger, F.; Duronio, R.J.; Leopold, P.; O’Farrell, P.H. Distinct molecular mechanism regulate cell cycle timing at successive stages of Drosophila embryogenesis. Genes Dev. 1994, 8, 440–452. [Google Scholar] [CrossRef] [PubMed]
- Yuan, K.; O’Farrell, P.H. Cyclin B3 is a mitotic cyclin that promotes the metaphase-anaphase transition. Curr. Biol. 2015, 25, 811–816. [Google Scholar] [CrossRef]
- Glotzer, M.; Murray, A.W.; Kirschner, M.W. Cyclin is degraded by the ubiquitin pathway. Nature 1991, 349, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Stumpff, J.; Duncan, T.; Homola, E.; Campbell, S.D.; Su, T.T. Drosophila Wee1 kinase regulates Cdk1 and mitotic entry during embryogenesis. Curr. Biol. 2004, 14, 2143–2148. [Google Scholar] [CrossRef]
- Edgar, B.A.; O’Farrell, P.H. Genetic control of cell division patterns in the Drosophila embryo. Cell 1989, 57, 177–187. [Google Scholar] [CrossRef]
- Price, D.; Rabinovitch, S.; O’Farrell, P.H.; Campbell, S.D. Drosophila wee1 has an essential role in the nuclear divisions of early embryogenesis. Genetics 2000, 155, 159–166. [Google Scholar] [CrossRef]
- Morgan, D.O. The Cell Cycle: Principles of Control; New Science Press in Association with Oxford University Press: London, UK, 2006; p. 297. [Google Scholar]
- Collart, C.; Allen, G.E.; Bradshaw, C.R.; Smith, J.C.; Zegerman, P. Titration of four replication factors is essential for the Xenopus laevis midblastula transition. Science 2013, 341, 893–896. [Google Scholar] [CrossRef]
- Liu, B.; Winkler, F.; Herde, M.; Witte, C.P.; Grosshans, J. A Link between Deoxyribonucleotide Metabolites and Embryonic Cell-Cycle Control. Curr. Biol. 2019, 29, 1187–1192.e3. [Google Scholar] [CrossRef]
- Sibon, O.C.; Stevenson, V.A.; Theurkauf, W.E. DNA-replication checkpoint control at the Drosophila midblastula transition. Nature 1997, 388, 93–97. [Google Scholar] [CrossRef]
- Shindo, Y.; Amodeo, A.A. Excess histone H3 is a competitive Chk1 inhibitor that controls cell-cycle remodeling in the early Drosophila embryo. Curr. Biol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Foe, V.E.; Alberts, B.M. Studies of nuclear and cytoplasmic behaviour during the five mitotic cycles that precede gastrulation in Drosophila embryogenesis. J. Cell Sci. 1983, 61, 31–70. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, A.B.; Kriegstein, H.J.; Hogness, D.S. The units of DNA replication in Drosophila melanogaster chromosomes. Cold Spring Harb. Symp. Quant. Biol. 1974, 38, 205–223. [Google Scholar] [CrossRef]
- Fogarty, P.; Campbell, S.D.; Abu-Shumays, R.; Phalle, B.S.; Yu, K.R.; Uy, G.L.; Goldberg, M.L.; Sullivan, W. The Drosophila grapes gene is related to checkpoint gene chk1/rad27 and is required for late syncytial division fidelity. Curr. Biol. 1997, 7, 418–426. [Google Scholar] [CrossRef]
- Deneke, V.E.; Melbinger, A.; Vergassola, M.; Di Talia, S. Waves of Cdk1 Activity in S Phase Synchronize the Cell Cycle in Drosophila Embryos. Dev. Cell 2016, 38, 399–412. [Google Scholar] [CrossRef] [PubMed]
- Edgar, B.A.; Datar, S.A. Zygotic degradation of two maternal Cdc25 mRNAs terminates Drosophila’s early cell cycle program. Genes Dev. 1996, 10, 1966–1977. [Google Scholar] [CrossRef] [PubMed]
- Hamm, D.C.; Bondra, E.R.; Harrison, M.M. Transcriptional activation is a conserved feature of the early embryonic factor Zelda that requires a cluster of four zinc fingers for DNA binding and a low-complexity activation domain. J. Biol. Chem. 2015, 290, 3508–3518. [Google Scholar] [CrossRef] [PubMed]
- Staudt, N.; Fellert, S.; Chung, H.R.; Jackle, H.; Vorbruggen, G. Mutations of the Drosophila zinc finger-encoding gene vielfaltig impair mitotic cell divisions and cause improper chromosome segregation. Mol. Biol. Cell 2006, 17, 2356–2365. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Johnston, J.; Shao, W.; Meier, S.; Staber, C.; Zeitlinger, J. A global change in RNA polymerase II pausing during the Drosophila midblastula transition. eLife 2013, 2, e00861. [Google Scholar] [CrossRef]
- Kimelman, D.; Kirschner, M.; Scherson, T. The events of the midblastula transition in Xenopus are regulated by changes in the cell cycle. Cell 1987, 48, 399–407. [Google Scholar] [CrossRef]
- Newport, J.; Dasso, M. On the coupling between DNA replication and mitosis. J. Cell Sci. Suppl. 1989, 12, 149–160. [Google Scholar] [CrossRef]
- Gerhart, J.; Wu, M.; Kirschner, M. Cell cycle dynamics of an M-phase-specific cytoplasmic factor in Xenopus laevis oocytes and eggs. J. Cell Biol. 1984, 98, 1247–1255. [Google Scholar] [CrossRef]
- Newport, J.W.; Kirschner, M.W. Regulation of the cell cycle during early Xenopus development. Cell 1984, 37, 731–742. [Google Scholar] [CrossRef]
- Frederick, D.L.; Andrews, M.T. Cell cycle remodeling requires cell-cell interactions in developing Xenopus embryos. J. Exp. Zool. 1994, 270, 410–416. [Google Scholar] [CrossRef]
- Petrus, M.J.; Wilhelm, D.E.; Murakami, M.; Kappas, N.C.; Carter, A.D.; Wroble, B.N.; Sible, J.C. Altered expression of Chk1 disrupts cell cycle remodeling at the midblastula transition in Xenopus laevis embryos. Cell Cycle 2004, 3, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Zamir, E.; Kam, Z.; Yarden, A. Transcription-dependent induction of G1 phase during the zebra fish midblastula transition. Mol. Cell. Biol. 1997, 17, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Kothari, P.; Mullins, M.; Lampson, M.A. Regulation of zygotic genome activation and DNA damage checkpoint acquisition at the mid-blastula transition. Cell Cycle 2014, 13, 3828–3838. [Google Scholar] [CrossRef] [PubMed]
- Fulka, J., Jr.; First, N.L.; Fulka, J.; Moor, R.M. Checkpoint control of the G2/M phase transition during the first mitotic cycle in mammalian eggs. Hum. Reprod. 1999, 14, 1582–1587. [Google Scholar] [CrossRef][Green Version]
- Kubiak, J.Z.; Ciemerych, M.A. Cell cycle regulation in early mouse embryos. Novartis Found. Symp. 2001, 237, 79–89, discussion 89–99. [Google Scholar] [CrossRef]
- Alphey, L.; Jimenez, J.; White-Cooper, H.; Dawson, I.; Nurse, P.; Glover, D.M. twine, a cdc25 homolog that functions in the male and female germline of Drosophila. Cell 1992, 69, 977–988. [Google Scholar] [CrossRef]
- Farrell, J.A.; O’Farrell, P.H. Mechanism and regulation of Cdc25/Twine protein destruction in embryonic cell-cycle remodeling. Curr. Biol. 2013, 23, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Farrell, J.A.; Shermoen, A.W.; Yuan, K.; O’Farrell, P.H. Embryonic onset of late replication requires Cdc25 down-regulation. Genes Dev. 2012, 26, 714–725. [Google Scholar] [CrossRef] [PubMed]
- Di Talia, S.; Wieschaus, E.F. Short-term integration of Cdc25 dynamics controls mitotic entry during Drosophila gastrulation. Dev. Cell 2012, 22, 763–774. [Google Scholar] [CrossRef]
- Liu, B.; Sung, H.W.; Grosshans, J. Multiple Functions of the Essential Gene PpV in Drosophila Early Development. G3 (Bethesda) 2019, 9, 3583–3593. [Google Scholar] [CrossRef]
- Grosshans, J.; Wieschaus, E. A genetic link between morphogenesis and cell division during formation of the ventral furrow in Drosophila. Cell 2000, 101, 523–531. [Google Scholar] [CrossRef]
- Seher, T.C.; Leptin, M. Tribbles, a cell-cycle brake that coordinates proliferation and morphogenesis during Drosophila gastrulation. Curr. Biol. 2000, 10, 623–629. [Google Scholar] [CrossRef]
- Lu, X.; Li, J.M.; Elemento, O.; Tavazoie, S.; Wieschaus, E.F. Coupling of zygotic transcription to mitotic control at the Drosophila mid-blastula transition. Development 2009, 136, 2101–2110. [Google Scholar] [CrossRef]
- Grosshans, J.; Muller, H.A.; Wieschaus, E. Control of cleavage cycles in Drosophila embryos by fruhstart. Dev. Cell 2003, 5, 285–294. [Google Scholar] [CrossRef]
- Gawlinski, P.; Nikolay, R.; Goursot, C.; Lawo, S.; Chaurasia, B.; Herz, H.M.; Kussler-Schneider, Y.; Ruppert, T.; Mayer, M.; Grosshans, J. The Drosophila mitotic inhibitor Fruhstart specifically binds to the hydrophobic patch of cyclins. EMBO Rep. 2007, 8, 490–496. [Google Scholar] [CrossRef]
- Seller, C.A.; O’Farrell, P.H. Rif1 prolongs the embryonic S phase at the Drosophila mid-blastula transition. PLoS Biol. 2018, 16, e2005687. [Google Scholar] [CrossRef]
- Collart, C.; Smith, J.C.; Zegerman, P. Chk1 Inhibition of the Replication Factor Drf1 Guarantees Cell-Cycle Elongation at the Xenopus laevis Mid-blastula Transition. Dev. Cell 2017, 42, 82–96.e3. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Grosshans, J. The role of dNTP metabolites in control of the embryonic cell cycle. Cell Cycle 2019, 18, 2817–2827. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Marmion, R.A.; Park, J.O.; Biswas, D.; Rabinowitz, J.D.; Shvartsman, S.Y. Dynamic Control of dNTP Synthesis in Early Embryos. Dev. Cell 2017, 42, 301–308.e3. [Google Scholar] [CrossRef] [PubMed]
- An, P.N.; Yamaguchi, M.; Bamba, T.; Fukusaki, E. Metabolome analysis of Drosophila melanogaster during embryogenesis. PLoS ONE 2014, 9, e99519. [Google Scholar] [CrossRef] [PubMed]
- Woodland, H.R.; Pestell, R.Q. Determination of the nucleoside triphosphate contents of eggs and oocytes of Xenopus laevis. Biochem. J. 1972, 127, 597–605. [Google Scholar] [CrossRef]
- Vastag, L.; Jorgensen, P.; Peshkin, L.; Wei, R.; Rabinowitz, J.D.; Kirschner, M.W. Remodeling of the metabolome during early frog development. PLoS ONE 2011, 6, e16881. [Google Scholar] [CrossRef]
- Winkler, F.; Kriebel, M.; Clever, M.; Groning, S.; Grosshans, J. Essential Function of the Serine Hydroxymethyl Transferase (SHMT) Gene During Rapid Syncytial Cell Cycles in Drosophila. G3 (Bethesda) 2017, 7, 2305–2314. [Google Scholar] [CrossRef]
- Conn, C.W.; Lewellyn, A.L.; Maller, J.L. The DNA damage checkpoint in embryonic cell cycles is dependent on the DNA-to-cytoplasmic ratio. Dev. Cell 2004, 7, 275–281. [Google Scholar] [CrossRef]
- Song, Y.; Park, J.O.; Tanner, L.; Nagano, Y.; Rabinowitz, J.D.; Shvartsman, S.Y. Energy budget of Drosophila embryogenesis. Curr. Biol. 2019, 29, R566–R567. [Google Scholar] [CrossRef] [PubMed]
- Nagaraj, R.; Sharpley, M.S.; Chi, F.; Braas, D.; Zhou, Y.; Kim, R.; Clark, A.T.; Banerjee, U. Nuclear Localization of Mitochondrial TCA Cycle Enzymes as a Critical Step in Mammalian Zygotic Genome Activation. Cell 2017, 168, 210–223.e11. [Google Scholar] [CrossRef]
- Rahman, M.M.; Rosu, S.; Joseph-Strauss, D.; Cohen-Fix, O. Down-regulation of tricarboxylic acid (TCA) cycle genes blocks progression through the first mitotic division in Caenorhabditis elegans embryos. Proc. Natl. Acad. Sci. USA 2014, 111, 2602–2607. [Google Scholar] [CrossRef] [PubMed]
- Marzluff, W.F.; Wagner, E.J.; Duronio, R.J. Metabolism and regulation of canonical histone mRNAs: Life without a poly(A) tail. Nat. Rev. Genet. 2008, 9, 843–854. [Google Scholar] [CrossRef]
- Palfy, M.; Joseph, S.R.; Vastenhouw, N.L. The timing of zygotic genome activation. Curr. Opin. Genet. Dev. 2017, 43, 53–60. [Google Scholar] [CrossRef]
- Almouzni, G.; Wolffe, A.P. Constraints on transcriptional activator function contribute to transcriptional quiescence during early Xenopus embryogenesis. EMBO J. 1995, 14, 1752–1765. [Google Scholar] [CrossRef] [PubMed]
- Lindeman, L.C.; Andersen, I.S.; Reiner, A.H.; Li, N.; Aanes, H.; Ostrup, O.; Winata, C.; Mathavan, S.; Muller, F.; Alestrom, P.; et al. Prepatterning of developmental gene expression by modified histones before zygotic genome activation. Dev. Cell 2011, 21, 993–1004. [Google Scholar] [CrossRef]
- Amodeo, A.A.; Jukam, D.; Straight, A.F.; Skotheim, J.M. Histone titration against the genome sets the DNA-to-cytoplasm threshold for the Xenopus midblastula transition. Proc. Natl. Acad. Sci. USA 2015, 112, E1086–E1095. [Google Scholar] [CrossRef]
- Joseph, S.R.; Palfy, M.; Hilbert, L.; Kumar, M.; Karschau, J.; Zaburdaev, V.; Shevchenko, A.; Vastenhouw, N.L. Competition between histone and transcription factor binding regulates the onset of transcription in zebrafish embryos. eLife 2017, 6, e23326. [Google Scholar] [CrossRef]
- Abrams, E.W.; Fuentes, R.; Marlow, F.L.; Kobayashi, M.; Zhang, H.; Lu, S.; Kapp, L.; Joseph, S.R.; Kugath, A.; Gupta, T.; et al. Molecular genetics of maternally-controlled cell divisions. PLoS Genet. 2020, 16, e1008652. [Google Scholar] [CrossRef]
- Li, Z.; Thiel, K.; Thul, P.J.; Beller, M.; Kuhnlein, R.P.; Welte, M.A. Lipid droplets control the maternal histone supply of Drosophila embryos. Curr. Biol. 2012, 22, 2104–2113. [Google Scholar] [CrossRef]
- Gunesdogan, U.; Jackle, H.; Herzig, A. Histone supply regulates S phase timing and cell cycle progression. eLife 2014, 3, e02443. [Google Scholar] [CrossRef] [PubMed]
- Chari, S.; Wilky, H.; Govindan, J.; Amodeo, A.A. Histone concentration regulates the cell cycle and transcription in early development. Development 2019, 146, dev177402. [Google Scholar] [CrossRef] [PubMed]
- Shindo, Y.; Amodeo, A.A. Dynamics of Free and Chromatin-Bound Histone H3 during Early Embryogenesis. Curr. Biol. 2019, 29, 359–366.e4. [Google Scholar] [CrossRef] [PubMed]
- Ffrench-Constant, C. Developmental timers. How do embryonic cells measure time? Curr. Biol. 1994, 4, 415–419. [Google Scholar] [CrossRef]
- Tsai, T.Y.; Theriot, J.A.; Ferrell, J.E., Jr. Changes in oscillatory dynamics in the cell cycle of early Xenopus laevis embryos. PLoS Biol. 2014, 12, e1001788. [Google Scholar] [CrossRef]
- Adar-Levor, S.; Nachmias, D.; Gal-Oz, S.T.; Jahn, Y.M.; Peyrieras, N.; Zaritsky, A.; Birnbaum, R.Y.; Elia, N. Cytokinetic abscission is part of the midblastula transition in early zebrafish embryogenesis. Proc. Natl. Acad. Sci. USA 2021, 118, e2021210118. [Google Scholar] [CrossRef]
- Anderson, G.A.; Gelens, L.; Baker, J.C.; Ferrell, J.E., Jr. Desynchronizing Embryonic Cell Division Waves Reveals the Robustness of Xenopus laevis Development. Cell Rep. 2017, 21, 37–46. [Google Scholar] [CrossRef]
- Deneke, V.E.; Puliafito, A.; Krueger, D.; Narla, A.V.; De Simone, A.; Primo, L.; Vergassola, M.; De Renzis, S.; Di Talia, S. Self-Organized Nuclear Positioning Synchronizes the Cell Cycle in Drosophila Embryos. Cell 2019, 177, 925–941.e17. [Google Scholar] [CrossRef]
- Lv, Z.; Rosenbaum, J.; Mohr, S.; Zhang, X.; Kong, D.; Preiss, H.; Kruss, S.; Alim, K.; Aspelmeier, T.; Grosshans, J. The Emergent Yo-yo Movement of Nuclei Driven by Cytoskeletal Remodeling in Pseudo-synchronous Mitotic Cycles. Curr. Biol. 2020, 30, 2564–2573.e5. [Google Scholar] [CrossRef]
- Onjiko, R.M.; Morris, S.E.; Moody, S.A.; Nemes, P. Single-cell mass spectrometry with multi-solvent extraction identifies metabolic differences between left and right blastomeres in the 8-cell frog (Xenopus) embryo. Analyst 2016, 141, 3648–3656. [Google Scholar] [CrossRef] [PubMed]
- Howe, J.A.; Newport, J.W. A developmental timer regulates degradation of cyclin E1 at the midblastula transition during Xenopus embryogenesis. Proc. Natl. Acad. Sci. USA 1996, 93, 2060–2064. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, B.; Zhao, H.; Wu, K.; Großhans, J. Temporal Gradients Controlling Embryonic Cell Cycle. Biology 2021, 10, 513. https://doi.org/10.3390/biology10060513
Liu B, Zhao H, Wu K, Großhans J. Temporal Gradients Controlling Embryonic Cell Cycle. Biology. 2021; 10(6):513. https://doi.org/10.3390/biology10060513
Chicago/Turabian StyleLiu, Boyang, Han Zhao, Keliang Wu, and Jörg Großhans. 2021. "Temporal Gradients Controlling Embryonic Cell Cycle" Biology 10, no. 6: 513. https://doi.org/10.3390/biology10060513
APA StyleLiu, B., Zhao, H., Wu, K., & Großhans, J. (2021). Temporal Gradients Controlling Embryonic Cell Cycle. Biology, 10(6), 513. https://doi.org/10.3390/biology10060513





