Magnetite-Accelerated Corrosion of SA508 Tubesheet Material and Its Effect on Steam Generator Tube Denting
Abstract
:1. Introduction
2. Experimental
2.1. Test Materials and Solutions
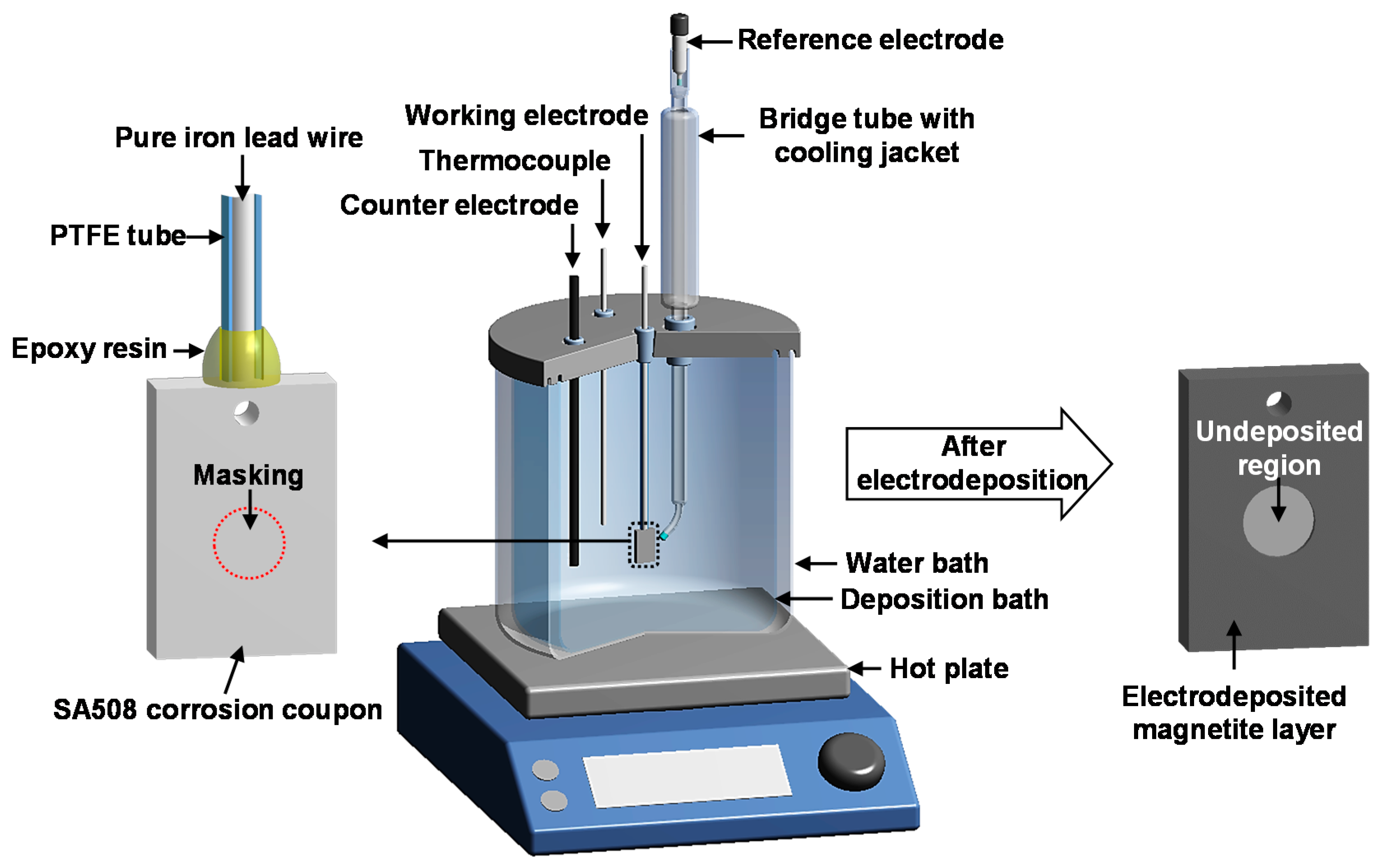
2.2. Magnetite-Deposited Corrosion Coupon
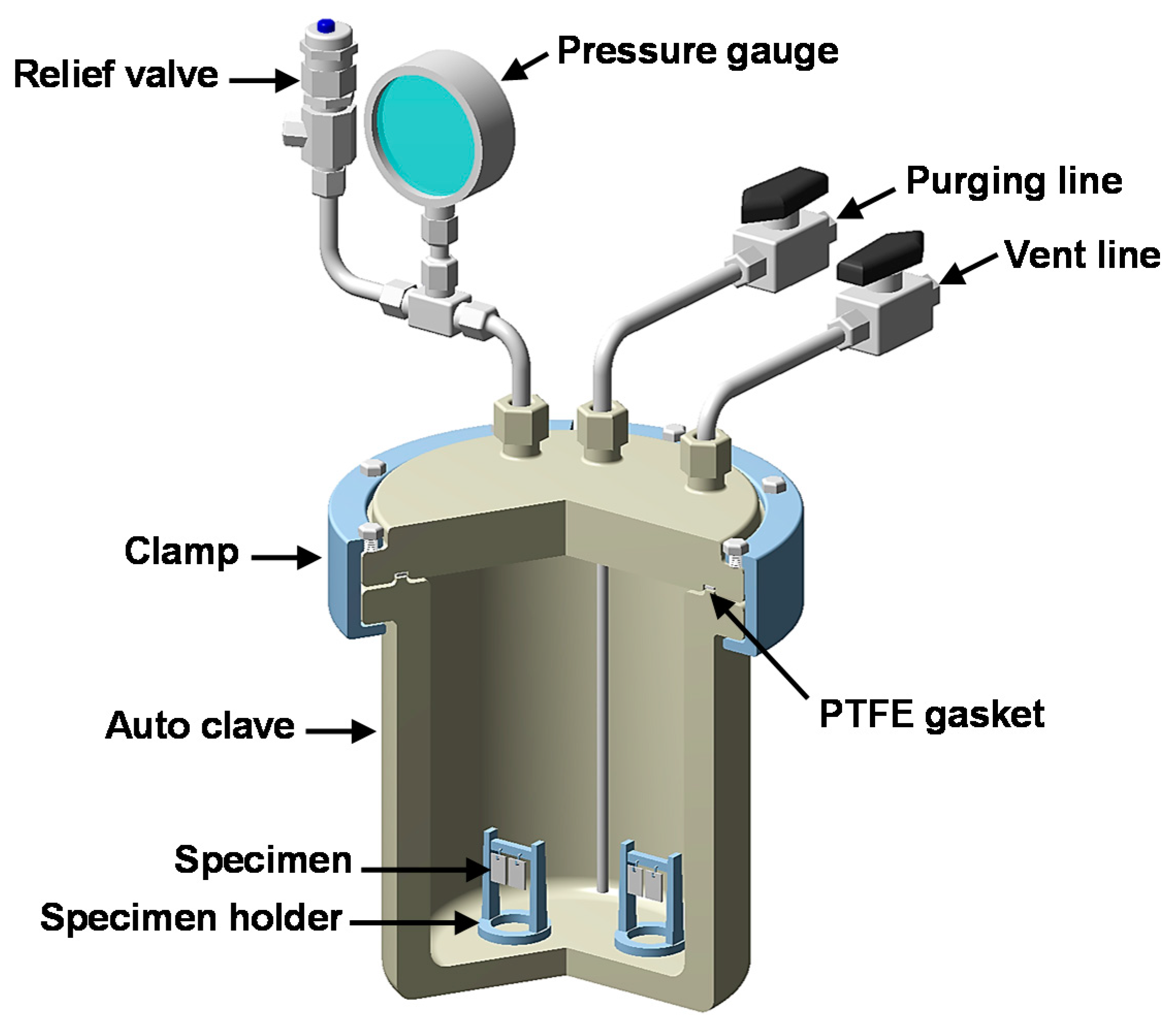
2.3. Immersion Corrosion Tests
2.4. Potentiodynamic Polarizaion Tests
3. Results and Discussion
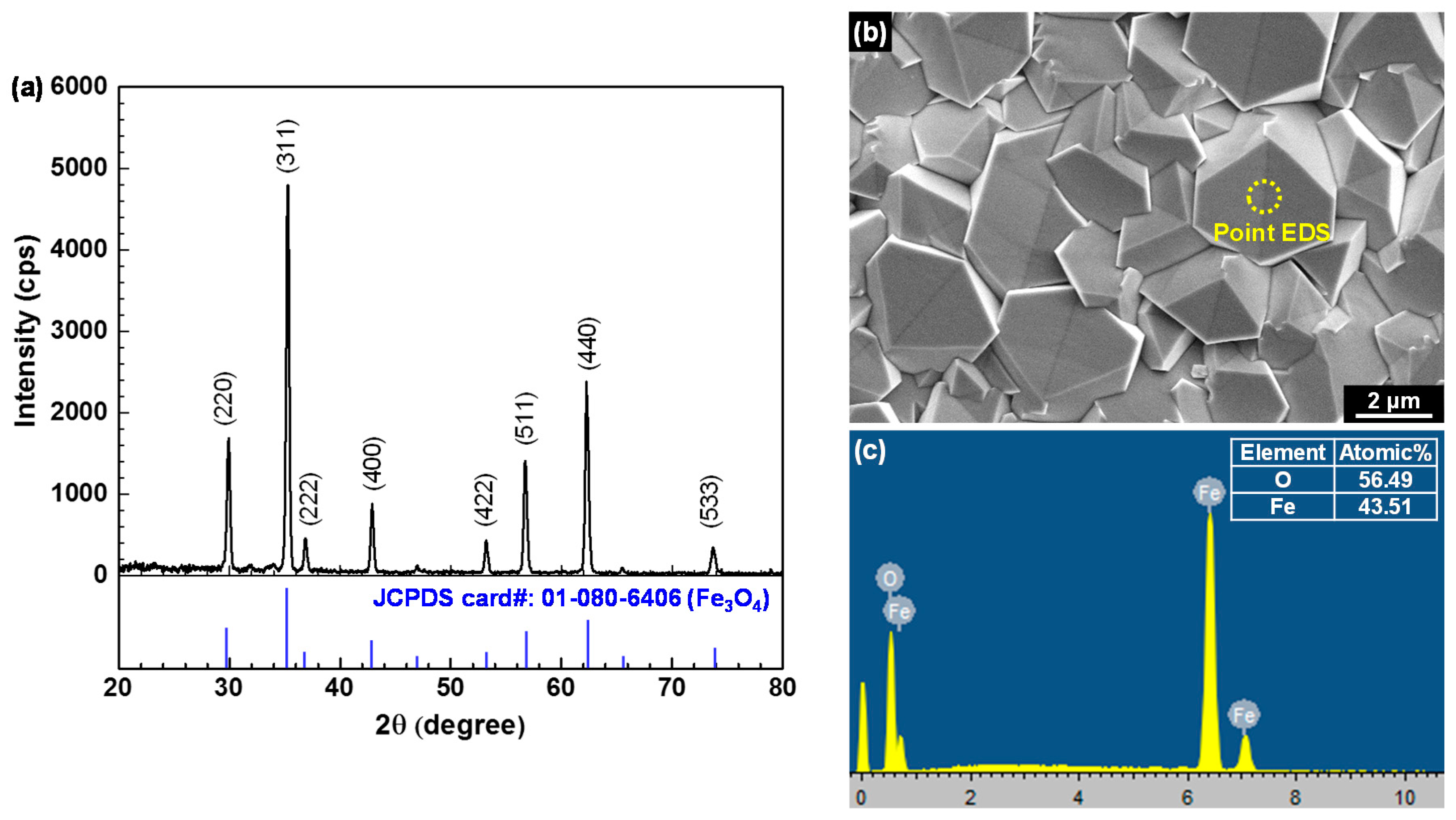
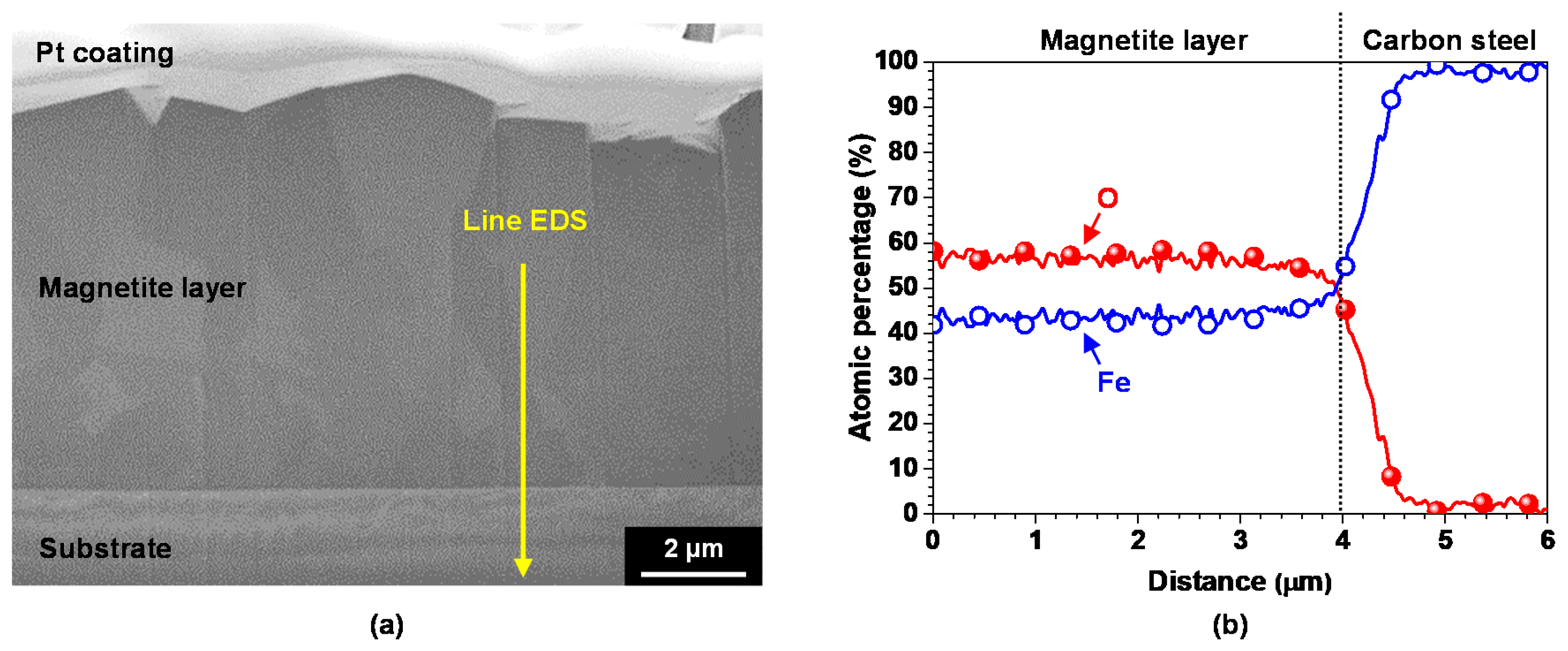
3.1. Electrodeposited Magnetite Layer on SA508 Substrate
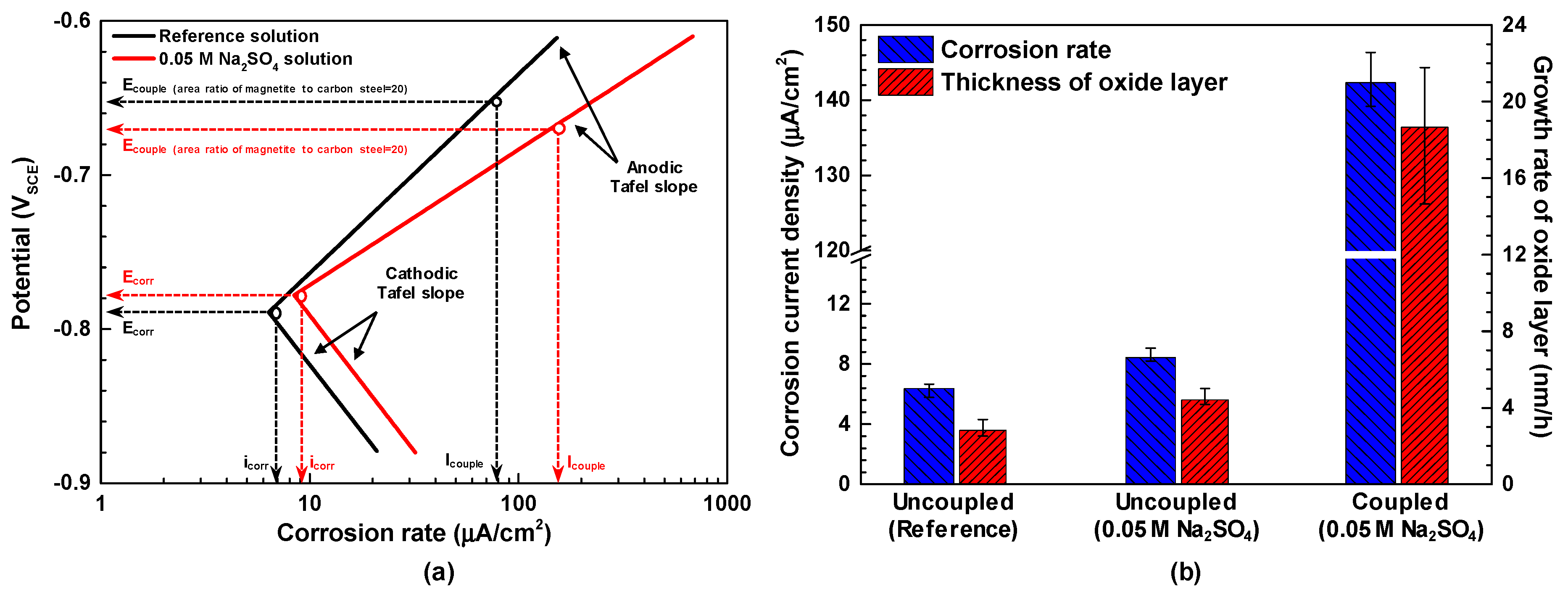
3.2. Immersion Corrosion Behavior
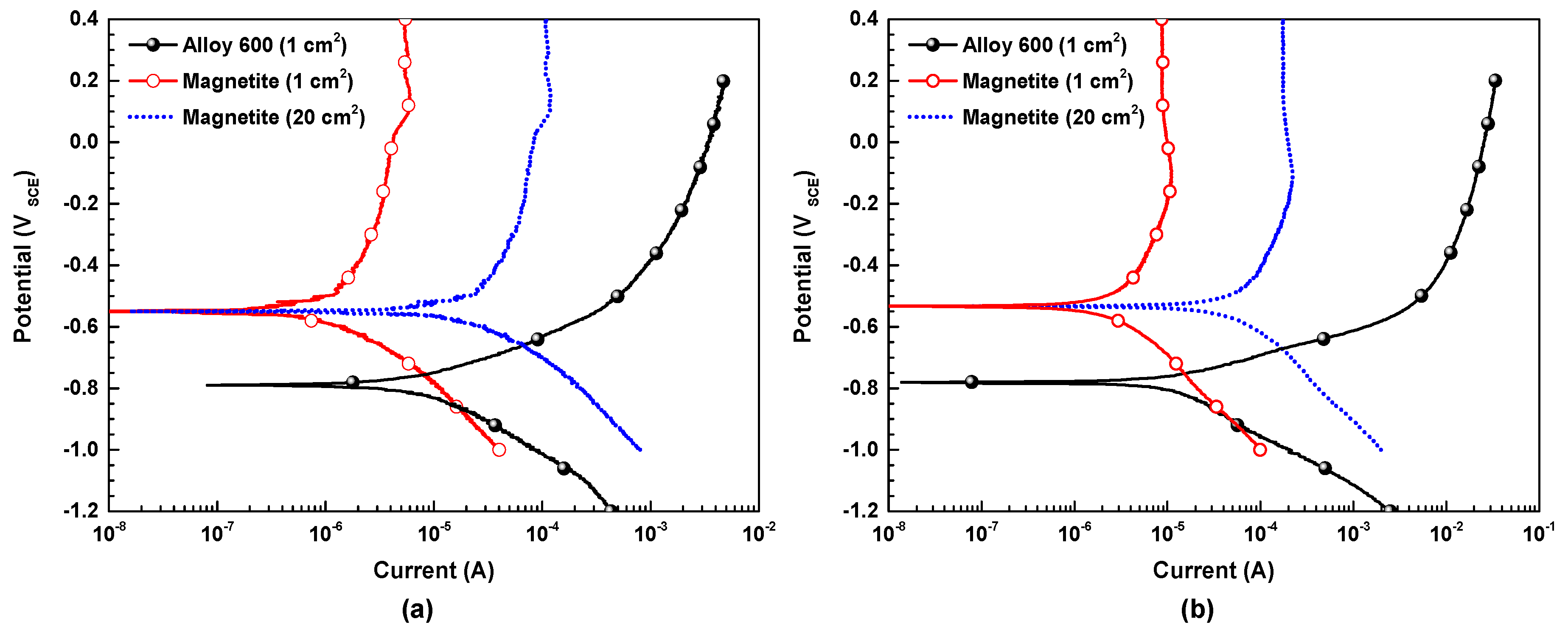
3.3. Potentiodynamic Polarization Curves
4. Conclusions
- Magnetite in contact with SA508 induced the fast corrosion of SA508. This accelerated corrosion of SA508 by magnetite was more severe in a test solution with sulfate ions.
- Regarding the electrochemical behavior, SA508 and magnetite acted as anodic and cathodic members, respectively, in the coupling system.
- In this coupling system, the corrosion current density of SA508 was increased by a galvanic corrosion mechanism with magnetite.
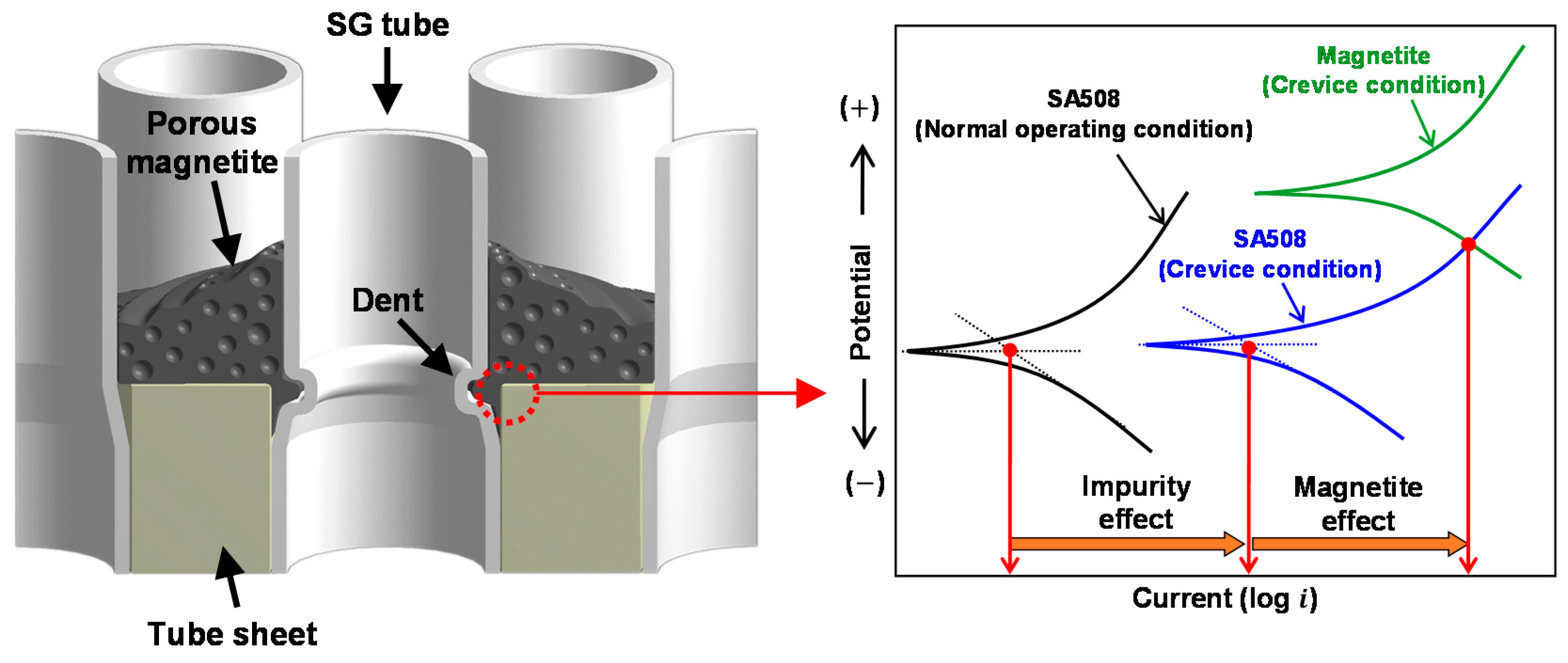
- Based on the experimental results, galvanic corrosion between magnetite and SA508 is proposed as a new acceleration contributor to the denting of SG tubes in the PWR secondary environment.
Author Contributions
Funding
Conflicts of Interest
References
- Scott, P.M. Stress corrosion cracking in pressurized water reactors – interpretation, modeling and remedies. Corrosion 2000, 56, 771–782. [Google Scholar] [CrossRef]
- Staehle, R.W.; Gorman, J.A. Quantitative assessment of submodes of stress corrosion cracking on the secondary side of steam generator tubing in pressurized water reactors: Part1. Corrosion 2003, 59, 931–944. [Google Scholar] [CrossRef]
- Staehle, R.W.; Gorman, J.A. Quantitative assessment of submodes of stress corrosion cracking on the secondary side of steam generator tubing in pressurized water reactors: Part2. Corrosion 2004, 60, 5–63. [Google Scholar] [CrossRef]
- Staehle, R.W.; Gorman, J.A. Quantitative assessment of submodes of stress corrosion cracking on the secondary side of steam generator tubing in pressurized water reactors: Part3. Corrosion 2004, 60, 115–180. [Google Scholar] [CrossRef]
- EPRI. Primary System Corrosion Research Program: EPRI Materials Degradation Matrix, Revision 2; EPRI TR-1020987; Electric Power Research Institute: Palo Alto, CA, USA, 2010. [Google Scholar]
- De Curieres, I. Environmental degradations in PWR steam generators. In Steam Generators for Nuclear Power Plants; Woodhead Publishing: Cambridge, UK, 2017. [Google Scholar]
- Choi, S. PWR steam generator tube denting at top of tubesheet. Paper 10137. In Proceedings of the International Conference on Nuclear Power Chemistry, Sapporo, Japan, 26–31 October 2014. [Google Scholar]
- Pearl, W.L.; Sawochka, S.G. Tube support and tubesheet corrosion. In Steam Generator Reference Book; Paine, J.P.N., Ed.; Electric Power Research Institute: Palo Alto, CA, USA, 1994. [Google Scholar]
- Klodian, X.; Matjaž, F. The corrosion inhibition of AA6082 Aluminium alloy by certain azoles in chloride solution: electrochemistry and surface analysis. Coatings 2019, 9, 380. [Google Scholar]
- Jeon, S.H.; Hong, S.; Kwon, H.C.; Hur, D.H. Characteristics of steam generator tube deposits in an operating pressurized water reactor. J. Nucl. Mater. 2018, 507, 371–380. [Google Scholar] [CrossRef]
- Tapping, R.L.; Turner, C.W.; Thompson, R.H. Steam generator deposits-A detailed analysis and some inferences. Corrosion 1991, 47, 489–495. [Google Scholar] [CrossRef]
- Manahan, M.P. Mechanical behaviour of magnetite from the Oconee-2 and TMI-1 steam generators using miniaturized specimen technology. J. Mater. Sci. 1990, 25, 3415–3423. [Google Scholar] [CrossRef]
- Fan, Z.D.; Du, J.S.; Zhang, Z.B.; Ma, Y.C.; Cao, S.Y.; Niu, K.; Liu, C.X. Internal leakage of plate heat exchangers caused by cooperation of pitting, crevice corrosion, and fretting. Eng. Fail. Anal. 2019, 96, 340–347. [Google Scholar] [CrossRef]
- Tsuda, N.; Nasu, K.; Fujimori, A.; Siratori, K. Electrical Conduction in Oxides; Springer: Berlin, Germany, 2000. [Google Scholar]
- Cornell, R.M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurrences and Uses, 2nd ed.; Wiley: Hoboken, NJ, USA, 2003. [Google Scholar]
- Goujon, C.; Pauporte, T.; Mansour, C.; Delaunary, S.; Bretelle, J.L. Fouling of steam generator tubes in nuclear power plants: Laboratory tests to reproduce oxides deposition. In Proceedings of the International Conference on Heat Exchanger Fouling and Cleaning, Budapest, Hungary, 9–14 June 2013. [Google Scholar]
- Kothari, H.M.; Kulp, E.A.; Limmer, S.J.; Poizot, P.; Bohannan, E.W.; Switzer, J.A. Electrochemical deposition and characterization of Fe3O4 films produced by the reduction of Fe(III)-triethanolamine. J. Mater. Res. 2006, 21, 293–301. [Google Scholar] [CrossRef]
- Kulp, E.A.; Kothari, H.M.; Limmer, S.J.; Yang, J.; Gudavarthy, R.V.; Bohannan, E.W.; Switzer, J.A. Electrodeposition of epitaxial magnetite films and ferrihydrite nanoribbons on single-crystal gold. Chem. Mater. 2009, 21, 5022–5031. [Google Scholar] [CrossRef]
- Jeon, S.-H.; Song, G.D.; Hur, D.H. Effects of deposition potentials on the morphology and structure of iron-based films on carbon steel substrate in an alkaline solution. Adv. Mater. Sci. Eng. 2016, 2016, 9038478. [Google Scholar] [CrossRef]
- Jeon, S.-H.; Choi, W.-I.; Song, G.D.; Son, Y.-H.; Hur, D.H. Influence of surface roughness and agitation on the morphology of magnetite films electrodeposited on carbon steel substrates. Coatings 2016, 6, 62. [Google Scholar] [CrossRef]


| C | Si | Mn | P | S | Ni | Cr | Mo | Fe |
|---|---|---|---|---|---|---|---|---|
| 0.199 | 0.051 | 1.52 | 0.005 | 0.006 | 0.987 | 0.232 | 0.582 | Bal. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, G.D.; Han, J.; Jeon, S.-H.; Hur, D.H. Magnetite-Accelerated Corrosion of SA508 Tubesheet Material and Its Effect on Steam Generator Tube Denting. Coatings 2019, 9, 575. https://doi.org/10.3390/coatings9090575
Song GD, Han J, Jeon S-H, Hur DH. Magnetite-Accelerated Corrosion of SA508 Tubesheet Material and Its Effect on Steam Generator Tube Denting. Coatings. 2019; 9(9):575. https://doi.org/10.3390/coatings9090575
Chicago/Turabian StyleSong, Geun Dong, Jeoh Han, Soon-Hyeok Jeon, and Do Haeng Hur. 2019. "Magnetite-Accelerated Corrosion of SA508 Tubesheet Material and Its Effect on Steam Generator Tube Denting" Coatings 9, no. 9: 575. https://doi.org/10.3390/coatings9090575







