Fire Protection Performance and Thermal Behavior of Thin Film Intumescent Coating
Abstract
1. Introduction
2. Experimental
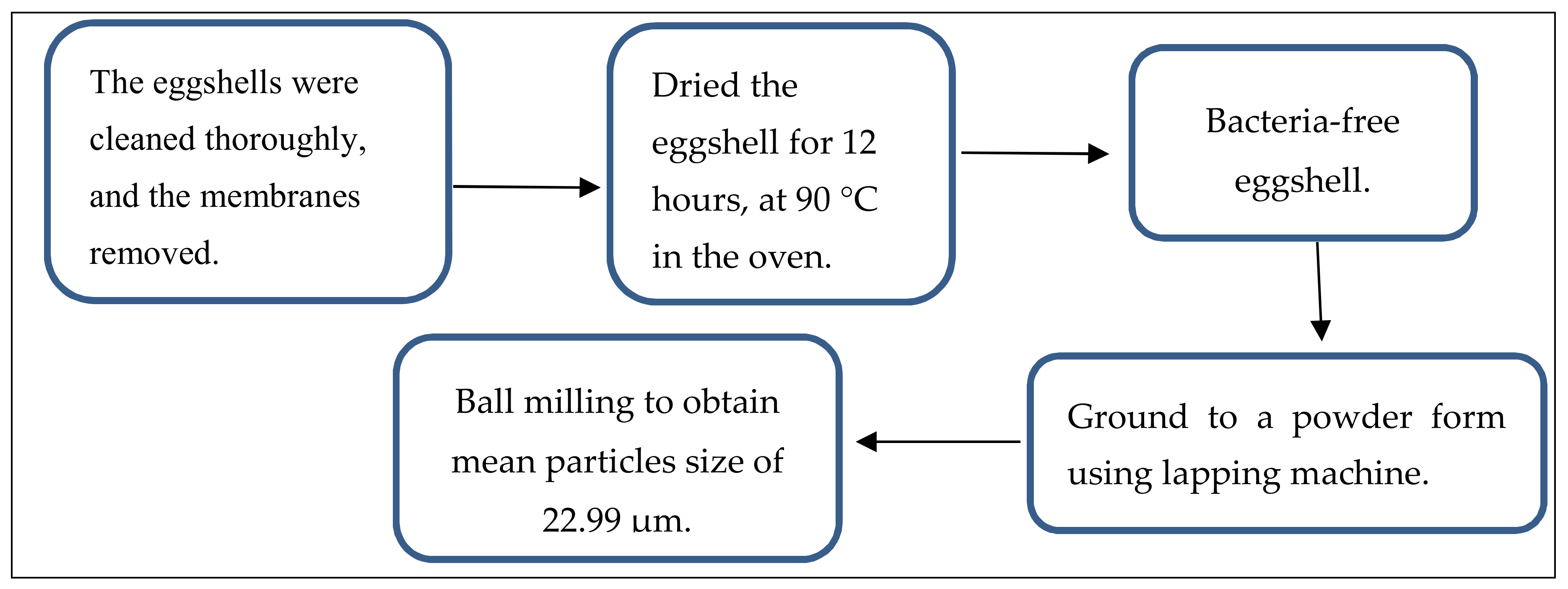
2.1. Materials
2.2. Fire Protective Test
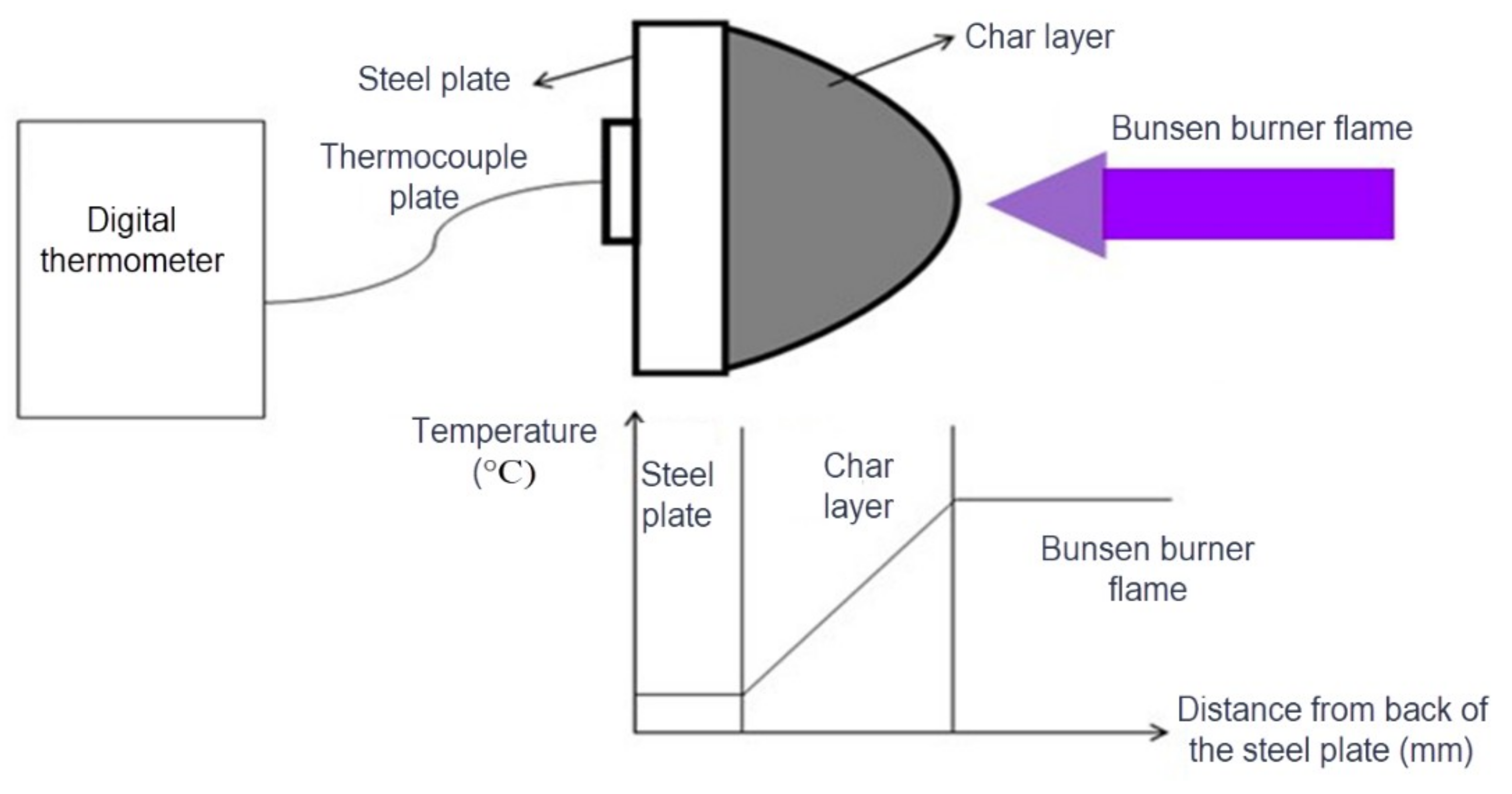
2.3. BS 476: Part 6 Fire Test
- I = index of intumescent coating performance;
- t = time from the beginning at which readings were taken (min);
- θm = temperature of the intumescent coating at time t;
- θc = temperature of the calibration curve at time t.
2.4. Sample Preparation for the Cone Calorimeter Test
Flammability Test
3. Results and Discussion
3.1. Bunsen Burner
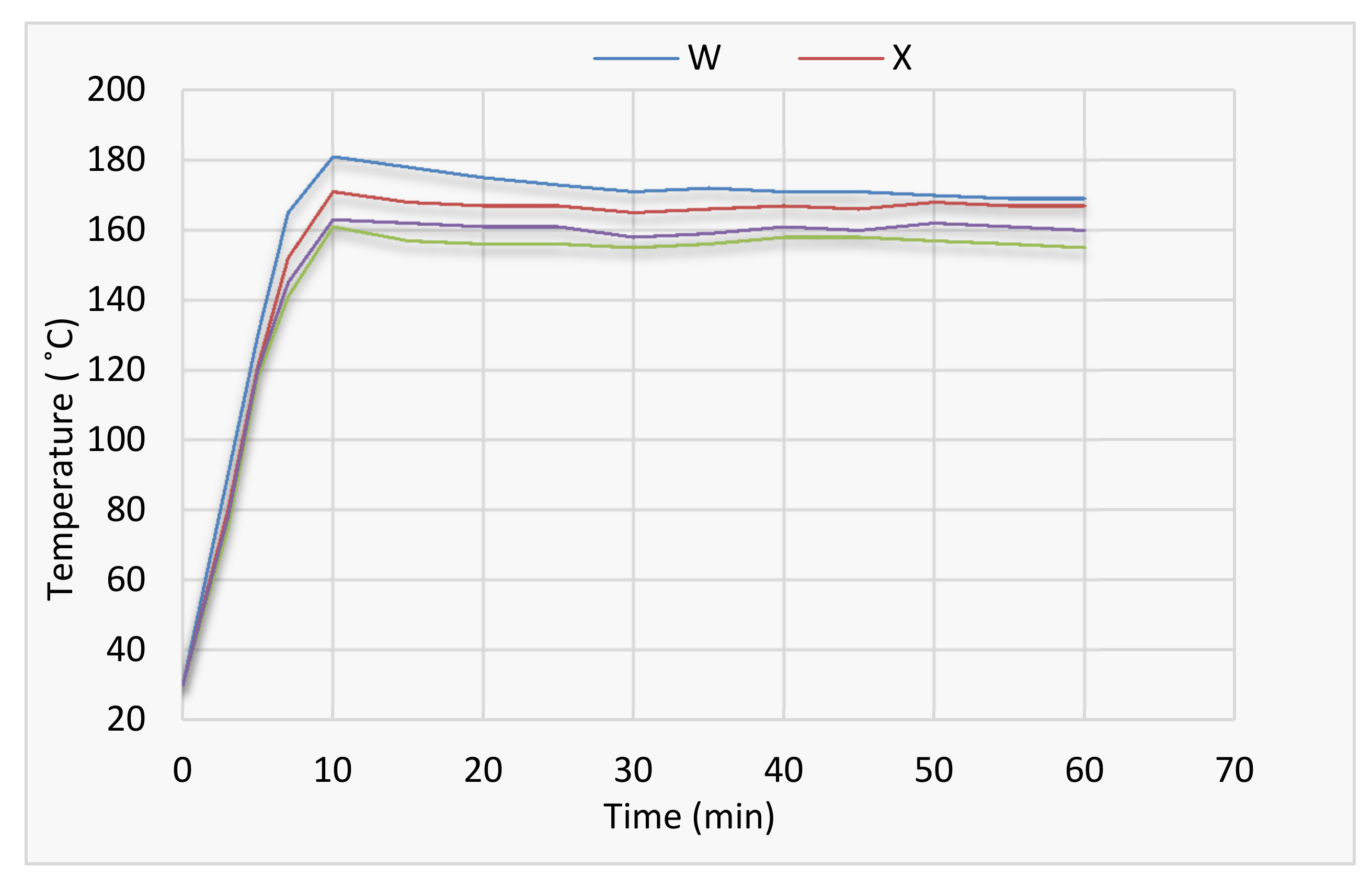
3.2. BS 476: Part 6
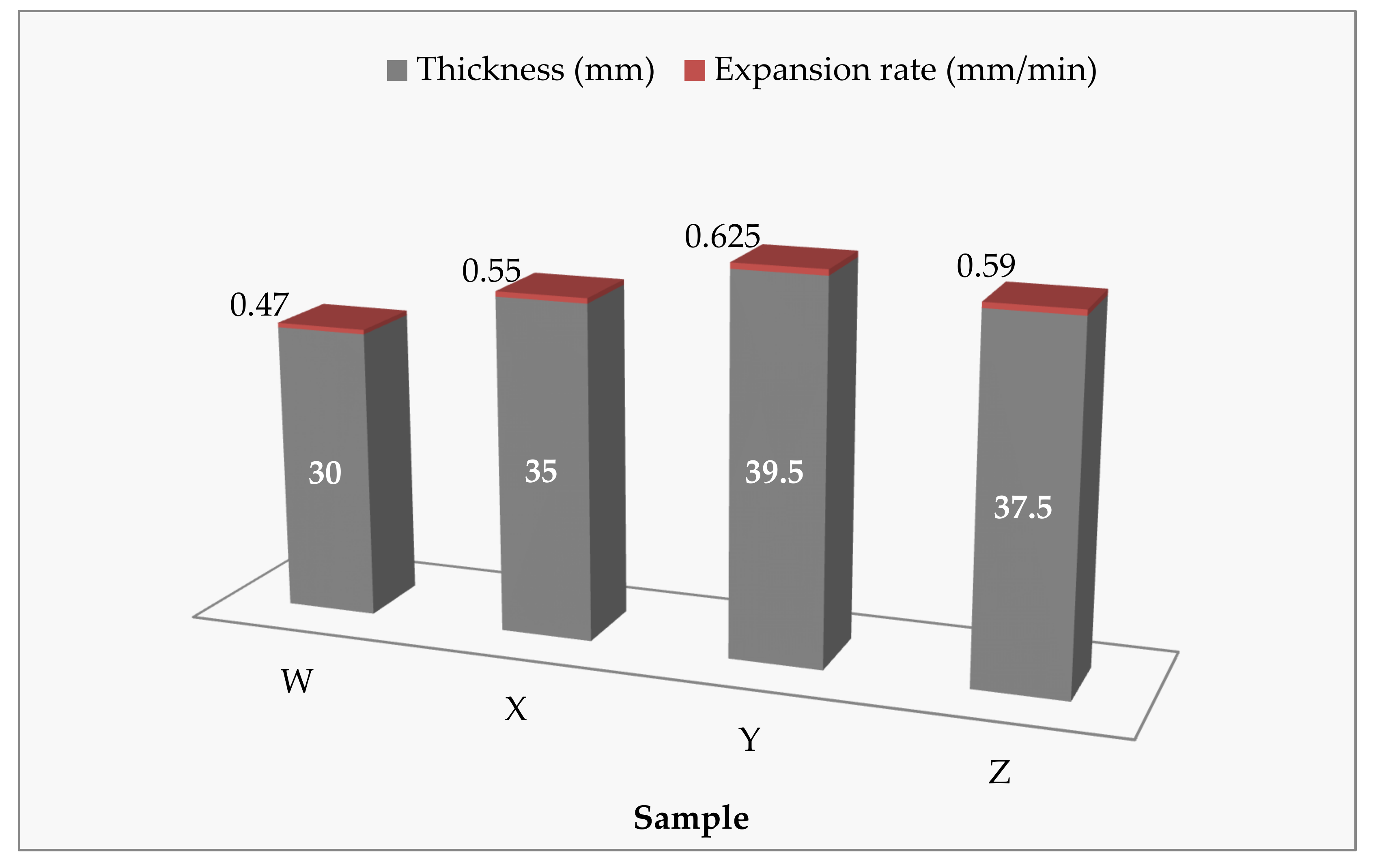
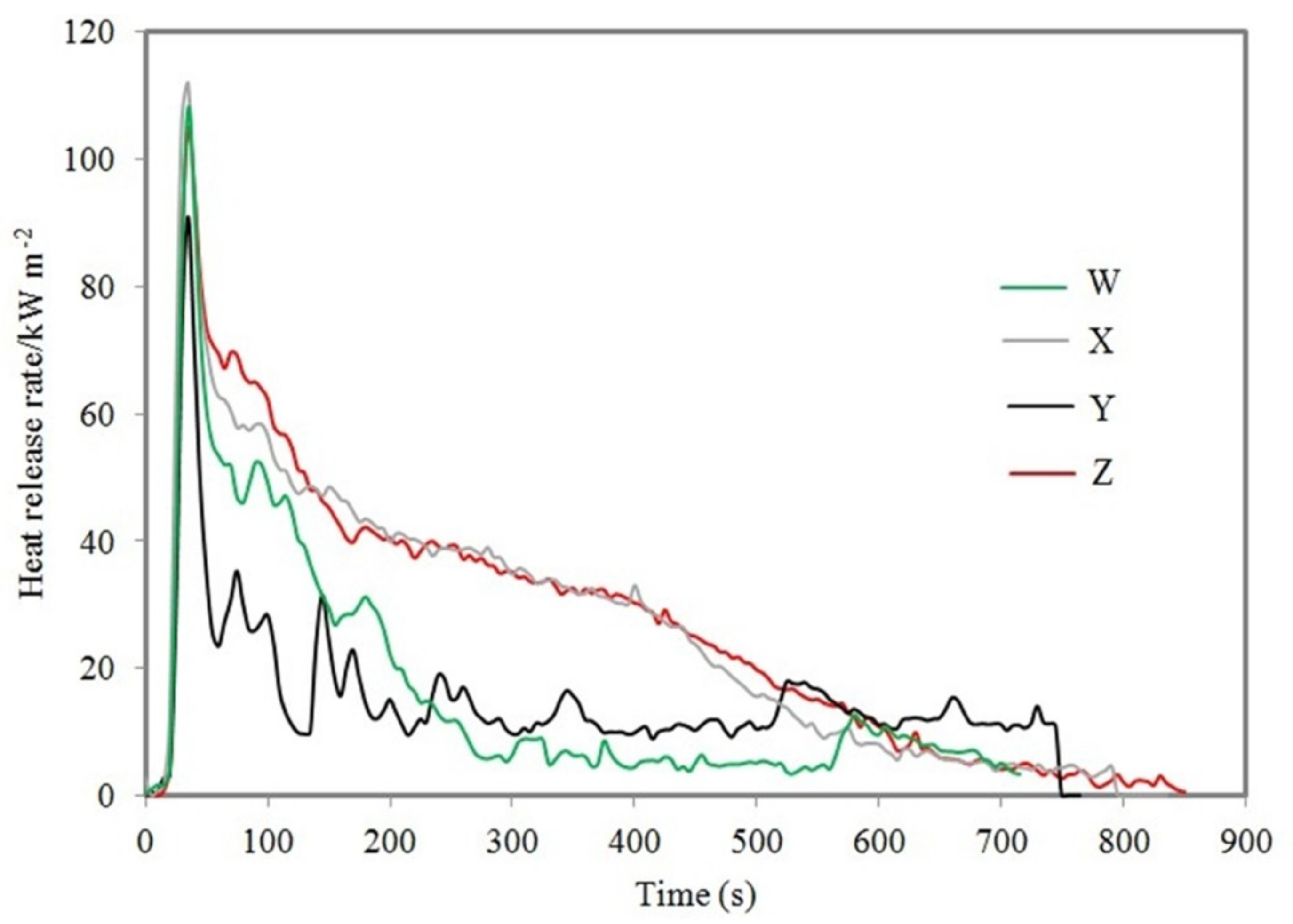
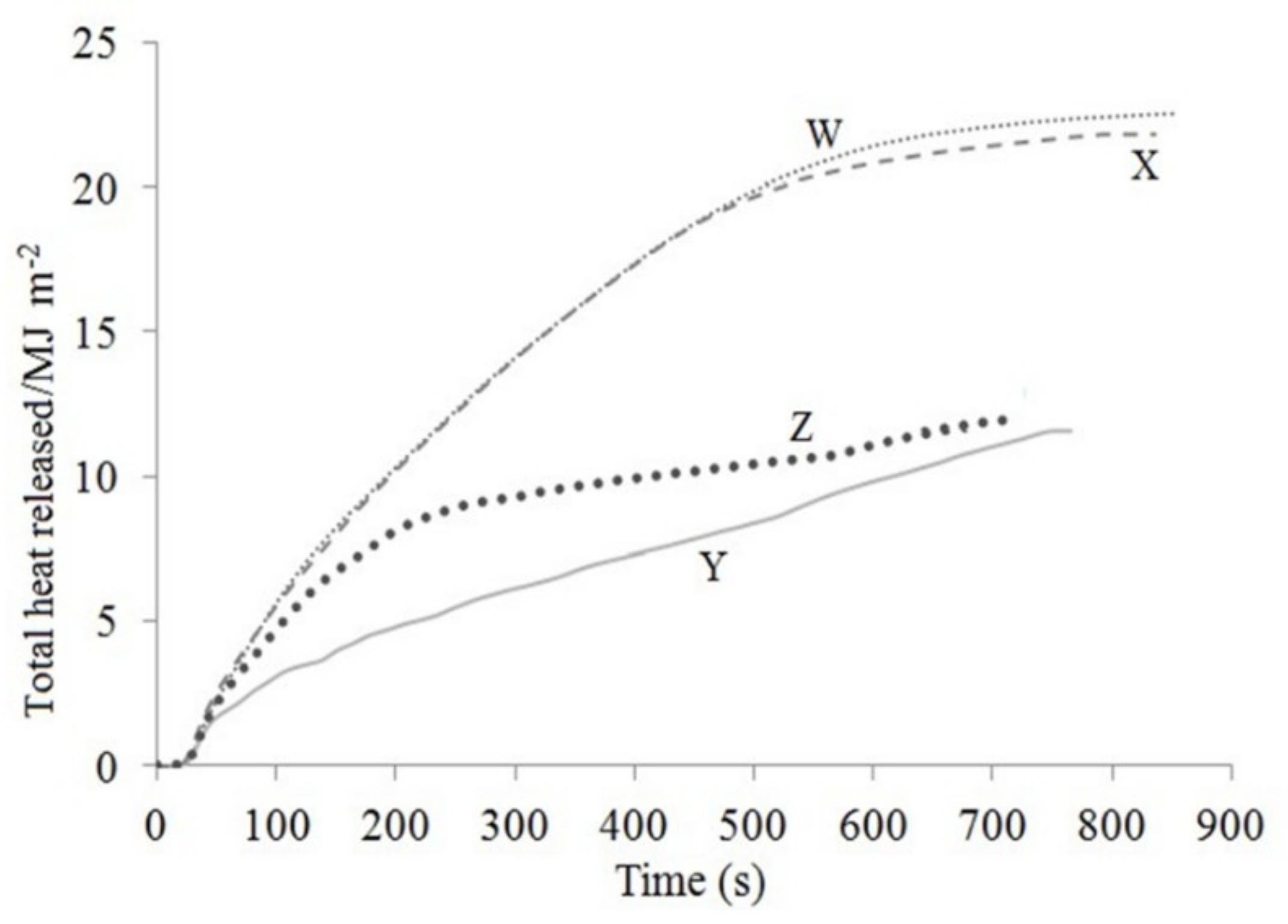
3.3. Cone Calorimeter Test
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mahmoud, A.S.; Sanni-Anibire, M.O.; Hassanain, M.A. Performance-based fire safety evacuation of an auditorium facility using a theoretical calculation method. Int. J. Build. Pathol. Adapt. 2017, 35, 124–138. [Google Scholar] [CrossRef]
- Altarawneh, M.; Saeed, A.; Al-Harahsheh, M.; Dlugogorski, B.Z. Thermal decomposition of brominated flame retardants (BFRs): Products and mechanisms. Prog. Energy Combust. Sci. 2019, 70, 212–259. [Google Scholar] [CrossRef]
- Prabir, B. Biomass Gasification, Pyrolysis and Torrefaction: Practical Design and Theory, 3rd ed.; Academic Press: Cambridge, MA, USA, 2018. [Google Scholar]
- Sigmann, S.B. Playing with fire: Chemical safety expertise required. J. Chem. Educ. 2018, 95, 1736–1746. [Google Scholar] [CrossRef]
- Harada, T. Nuclear flash burns: A review and consideration. Burn. Open 2018, 2, 1–7. [Google Scholar] [CrossRef]
- Duquesne, S.; Magnet, S.; Jama, C.; Delobel, R. Thermoplastic resins for thin film intumescent coatings-towards a better understanding of their effect on intumescent efficiency. Polym. Degrad. Stab. 2005, 88, 63–69. [Google Scholar] [CrossRef]
- Yang, H.; Yu, B.; Song, P.; Maluk, C.; Wang, H. Surface-coating engineering for flame retardant flexible polyurethane foams: A critical review. Compos. Part B Eng. 2019, 176, 107185. [Google Scholar] [CrossRef]
- Yew, M.C.; Ramli Sulong, N.H.; Yew, M.K.; Amalina, M.A.; Johan, M.R. Fire propagation performance of intumescent fire protective coatings using eggshells as a novel biofiller. Sci. World J. 2014, 2014, 805094. [Google Scholar] [CrossRef]
- Aziz, H.; Ahmad, F. Effects from nano-titanium oxide on the thermal resistance of an intumescent fire retardant coating for structural applications. Prog. Org. Coat. 2016, 101, 431–439. [Google Scholar] [CrossRef]
- Guru, P.S.; Dash, S. Sorption on eggshell waste—A review on ultrastructure, biomineralization and other applications. Adv. Colloid Interface Sci. 2014, 209, 49–67. [Google Scholar] [CrossRef]
- Siriprom, W.; Sangwaranatee, N.; Hidayat, R.; Kongsriprapan, S.; Teanchai, K.; Chamchoi, N. The physicochemical characteristic of biodegradable methylcellulose film reinforced with chicken eggshells. Mater. Today Proc. 2018, 5, 14836–14839. [Google Scholar] [CrossRef]
- Yew, M.C.; Ramli Sulong, N.H.; Yew, M.K.; Amalina, M.A.; Johan, M.R. The formulation and study of the thermal stability and mechanical properties of an acrylic coating using chicken eggshell as a novel bio-filler. Prog. Org. Coat. 2013, 76, 1549–1555. [Google Scholar] [CrossRef]
- Hassan, S.B.; Aigbodion, V.S. Effects of eggshell on the microstructures and properties of Al–Cu–Mg/eggshell particulate composites. J. King Saud Univ. Eng. Sci. 2015, 27, 49–56. [Google Scholar] [CrossRef]
- Arias, J.L.; Fernandez, M.S.; Dennis, J.E.; Caplan, A.I. Collagen of the chicken eggshell membranes. Connect. Tissue. Res. 1991, 26, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Hiremath, P.; Shettar, M.; Shankar, M.G.; Mohan, N.S. Investigation on effect of egg shell powder on mechanical properties of GFRP composites. Mater. Today Proc. 2018, 5, 3014–3018. [Google Scholar] [CrossRef]
- Somdee, P.; Hasook, A. Effect of modified eggshell powder on physical properties of poly(lactic acid) and natural rubber composites. Mater. Today Proc. 2017, 4, 6502–6511. [Google Scholar] [CrossRef]
- Nakano, T.; Ikawa, N.I.; Ozimek, L. Chemical composition of chicken eggshell and shell membranes. Poult. Sci. 2003, 82, 510–514. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.; Zhang, Y.; Xu, X.; Dayo, A.; Li, X.; Wang, S.; Liu, W. Reinforcement of stearic acid treated egg shell particles in epoxy thermosets: structural, thermal, and mechanical characterization. Materials 2018, 11, 1872. [Google Scholar] [CrossRef] [PubMed]
- Yusuff, A.S. Preparation and characterization of composite anthill-chicken eggshell adsorbent: Optimization study on heavy metals adsorption using response surface methodology. J. Environ. Sci. Technol. 2017, 10, 120–130. [Google Scholar] [CrossRef][Green Version]
- Ashok, B.; Naresh, S.; Obi, R.K.; Madhukar, K.; Cai, J.; Zhang, L.; Varadu, R.A. Tensile and thermal properties of poly(lactic acid)/eggshell powder composite films. Int. J. Polym. Anal. Charact. 2014, 19, 245–255. [Google Scholar] [CrossRef]
- Toro, P.; Quijada, R.; Yazdani-Pedram, M.; Arias, J.L. Eggshell: A new bio-filler for polypropylene composites. Mater. Lett. 2007, 61, 4347–4350. [Google Scholar] [CrossRef]
- Ishikawa, S.I.; Sekine, S.; Miura, N.; Suyama, K.; Arihara, K.; Itoh, M. Removal of selenium and arsenic by animal biopolymers. Biol. Trace. Elem. Res. 2004, 102, 113–127. [Google Scholar] [CrossRef]
- Liu, J.L.; Wu, Q.S.; Ding, Y.P.; Wang, S.Y. Biomimetic synthesis of BaSO4 nanotubes using eggshell membrane as template. J. Mater. Res. 2004, 19, 2803–2806. [Google Scholar] [CrossRef]
- Liu, J.L.; Wu, Q.S.; Ding, Y.P. Controlled synthesis of different morphologies of BaWO4 crystals through biomembrane/organic-addition supramolecule Templates. Cryst. Growth Des. 2005, 5, 445. [Google Scholar] [CrossRef]
- Tsai, W.T.; Yang, J.M.; Lai, C.W.; Cheng, Y.H.; Lin, C.C.; Yeh, C.W. Characterization and adsorption properties of eggshells and eggshell membrane. Bioresour. Technol. 2006, 97, 488–493. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Choi, M.M.F. Aspartame optical biosensor with bienzyme-immobilized eggshell membrane and oxygen-sensitive optode membrane. Anal. Chem. 2002, 74, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Yi, F.; Guo, Z.X.; Zhang, L.X.; Yu, J.; Li, Q. Soluble eggshell membrane protein: Preparation, characterization and biocompatibility. Biomaterials 2004, 25, 4591–4599. [Google Scholar] [CrossRef]
- Yew, M.C.; Ramli Sulong, N.H. Fire-resistive performance of intumescent flame-retardant coatigs for steel. Mater. Des. 2012, 34, 719–724. [Google Scholar] [CrossRef]
- Yew, M.C.; Ramli Sulong, N.H. Effect of epoxy binder on fire protection and bonding strength of intumescent fire protective coatings for steel. Adv. Mater. Res. 2010, 168–170, 1228–1232. [Google Scholar] [CrossRef]
- Yew, M.C.; Ramli Sulong, N.H.; Chong, W.T.; Poh, S.C.; Ang, B.C.; Tan, K.H. Integration of thermal insulation coating and moving-air-cavity in a cool roof system for attic temperature reduction. Energy Convers. Manage. 2013, 75, 241–248. [Google Scholar] [CrossRef]
- Yew, M.C.; Yew, M.K.; Saw, L.H.; Ng, T.C.; Rajkumar, D.; Beh, J.H. Influence of nano bio-filler on the fire-resistive and mechanical properties of water-based intumescent coatings. Prog. Org. Coat. 2018, 124, 33–40. [Google Scholar] [CrossRef]
- BS 476 Part 6: 1989+A1: 2009 Fire Tests on Building Materials and Structures. Method of test for fire propagation for products; British Standards Institution: London, UK, 1989.
- ISO 5660-1 Reaction-to-Fire Tests-Heat Release, Smoke Production and Mass Loss Rate-Part 1: Heat Release Rate (Cone Calorimeter Method); International Standard Organization: Geneva, Switzerland, 2002.
- Gu, J.W.; Zhang, G.C.; Dong, S.I.; Zhang, Q.Y.; Kong, J. Study on preparation and fire-retardant mechanism analysis of intumescent flame-retardant coatings. Surf. Coat. Technol. 2007, 201, 7835–7841. [Google Scholar] [CrossRef]
- Lim, M.; Li, B.; Li, Q.; Li, S.; Zhang, S. Synergistic effect of metal oxides on the flame retardancy and thermal degradation of novel intumescent flame-retardant thermoplastic polyurethanes. J. Appl. Polym. Sci. 2011, 121, 1951–1960. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, J.Q. A new kind of halogen-free fire retardant coatings made from expandable graphite. J. Beijing Inst. Tech. 2001, 21, 649–652. [Google Scholar]
- Wang, Z.Y.; Han, E.H.; Ke, W. Influence of expandable graphiteon fire resistance and water resistance of flame retardant coatings. Corros. Sci. 2007, 49, 2237–2253. [Google Scholar] [CrossRef]
- Kartal, S.N.; Green, F. Decay and termite resistance of medium density fiberboard (MDF) made from different wood species. Int. Biodeterior. Biodegrad. 2003, 51, 29–35. [Google Scholar] [CrossRef]
- Santos, P.S.; Santos, H.S.; Toledo, S.P. Standard transition aluminas. Electron microscopy studies. Mater. Res. 2000, 3, 104–114. [Google Scholar] [CrossRef]
- Hornsby, P.R.; Watson, C.L. A study of the mechanism of flame retardance and smoke suppression in polymers filled with magnesium hydroxide. Polym. Degrad. Stab. 1990, 30, 73–87. [Google Scholar] [CrossRef]

| Ingredients | Parts by Weight for Formulations | |||
|---|---|---|---|---|
| W | X | Y | Z | |
| APP | 20 | 20 | 20 | 20 |
| MEL | 10 | 10 | 10 | 10 |
| PER | 10 | 10 | 10 | 10 |
| TiO2 | 5.00 | 3.40 | 3.20 | 3.00 |
| Al(OH)3 | – | 2.80 | 3.30 | 2.75 |
| Mg(OH)2 | 5.00 | 3.80 | – | 1.75 |
| ES | – | – | 3.50 | 2.50 |
| Polymer Binder | W | X | Y | Z |
| Acrylic resin | 50 | 50 | 50 | 50 |
| Weight * (g) | 26.70 | 24.80 | 25.80 | 25.90 |
| Thickness (mm) | 1.50 | 1.50 | 1.50 | 1.50 |
| Density (g/cm3) | 1.780 | 1.653 | 1.720 | 1.727 |
| Time (min) | Calibration, Temperature (°C) | Coating W (°C) | Coating X (°C) | Coating Y (°C) | Coating Z (°C) |
|---|---|---|---|---|---|
| 0.5 | 14 | 18 | 14 | 12 | 11 |
| 1 | 18 | 21 | 18 | 16 | 15 |
| 1.5 | 23 | 26 | 23 | 19 | 19 |
| 2 | 27 | 30 | 27 | 23 | 22 |
| 2.5 | 30 | 34 | 31 | 26 | 26 |
| 3 | 34 | 38 | 34 | 30 | 29 |
| 4 | 72 | 122 | 55 | 54 | 60 |
| 5 | 108 | 212 | 133 | 129 | 150 |
| 6 | 129 | 274 | 169 | 181 | 179 |
| 7 | 148 | 321 | 213 | 202 | 208 |
| 8 | 166 | 364 | 227 | 219 | 226 |
| 9 | 182 | 378 | 272 | 234 | 236 |
| 10 | 192 | 405 | 290 | 244 | 241 |
| 12 | 214 | 417 | 295 | 249 | 246 |
| 14 | 230 | 418 | 302 | 253 | 250 |
| 16 | 238 | 416 | 304 | 258 | 253 |
| 18 | 246 | 403 | 306 | 260 | 253 |
| 20 | 257 | 385 | 308 | 263 | 259 |
| Sub index 1 (I1) | 1.6 | 0.2 | 0.1 | 0 | |
| Sub index 2 (I2) | 15 | 5.1 | 4.6 | 4.4 | |
| Sub index 3 (I3) | 5.7 | 1.5 | 1.2 | 0.7 | |
| Index of Performance (I) | 22.3 | 6.8 | 5.9 | 5.1 | |
| Sample | Peak of Heat Released Rate (kW/m2) | Total Heat Released (MJ/m2) | Thickness of Char Layer (mm) | Time to Ignition (s) | Residual Weight (wt.%) |
|---|---|---|---|---|---|
| W | 106.03 | 22.4 | 21.0 | 9 | 43.85 |
| X | 111.86 | 21.6 | 30.0 | 8 | 46.12 |
| Y | 91.00 | 11.5 | 35.5 | 10 | 61.81 |
| Z | 99.98 | 12.0 | 34.0 | 10 | 58.48 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beh, J.H.; Yew, M.C.; Yew, M.K.; Saw, L.H. Fire Protection Performance and Thermal Behavior of Thin Film Intumescent Coating. Coatings 2019, 9, 483. https://doi.org/10.3390/coatings9080483
Beh JH, Yew MC, Yew MK, Saw LH. Fire Protection Performance and Thermal Behavior of Thin Film Intumescent Coating. Coatings. 2019; 9(8):483. https://doi.org/10.3390/coatings9080483
Chicago/Turabian StyleBeh, Jing Han, Ming Chian Yew, Ming Kun Yew, and Lip Huat Saw. 2019. "Fire Protection Performance and Thermal Behavior of Thin Film Intumescent Coating" Coatings 9, no. 8: 483. https://doi.org/10.3390/coatings9080483
APA StyleBeh, J. H., Yew, M. C., Yew, M. K., & Saw, L. H. (2019). Fire Protection Performance and Thermal Behavior of Thin Film Intumescent Coating. Coatings, 9(8), 483. https://doi.org/10.3390/coatings9080483








