Cost-Effective and High-Throughput Plasmonic Interference Coupled Nanostructures by Using Quasi-Uniform Anodic Aluminum Oxide
Abstract
1. Introduction
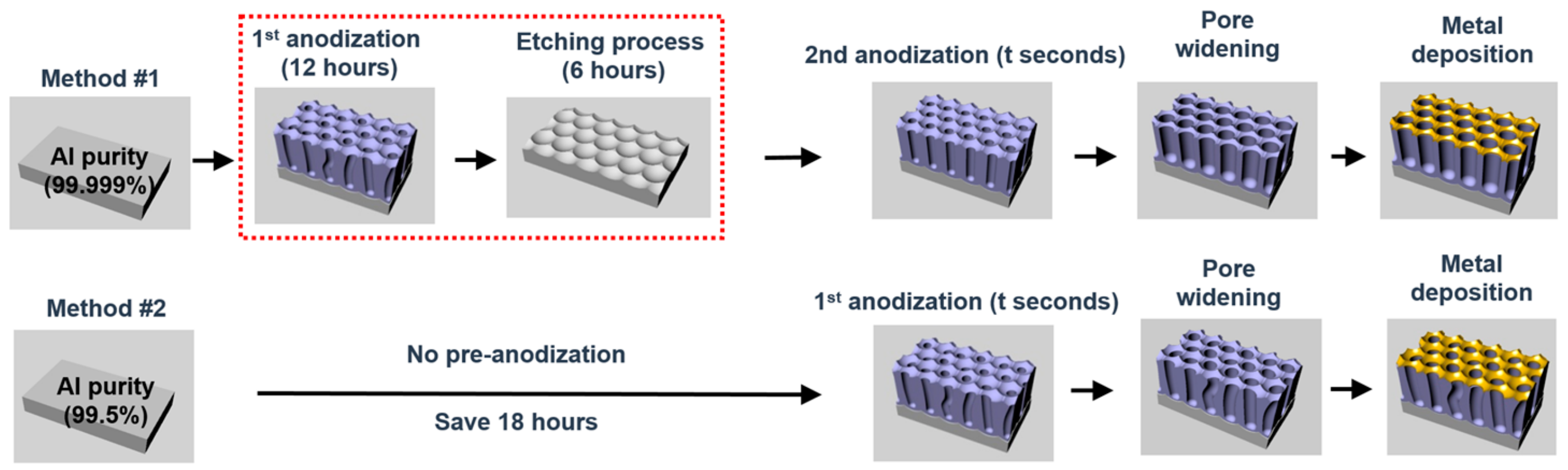
2. Materials and Methods
2.1. Fabrication of AAO Nanostructures
2.2. Characterization of Surface Morphology and Optical Property
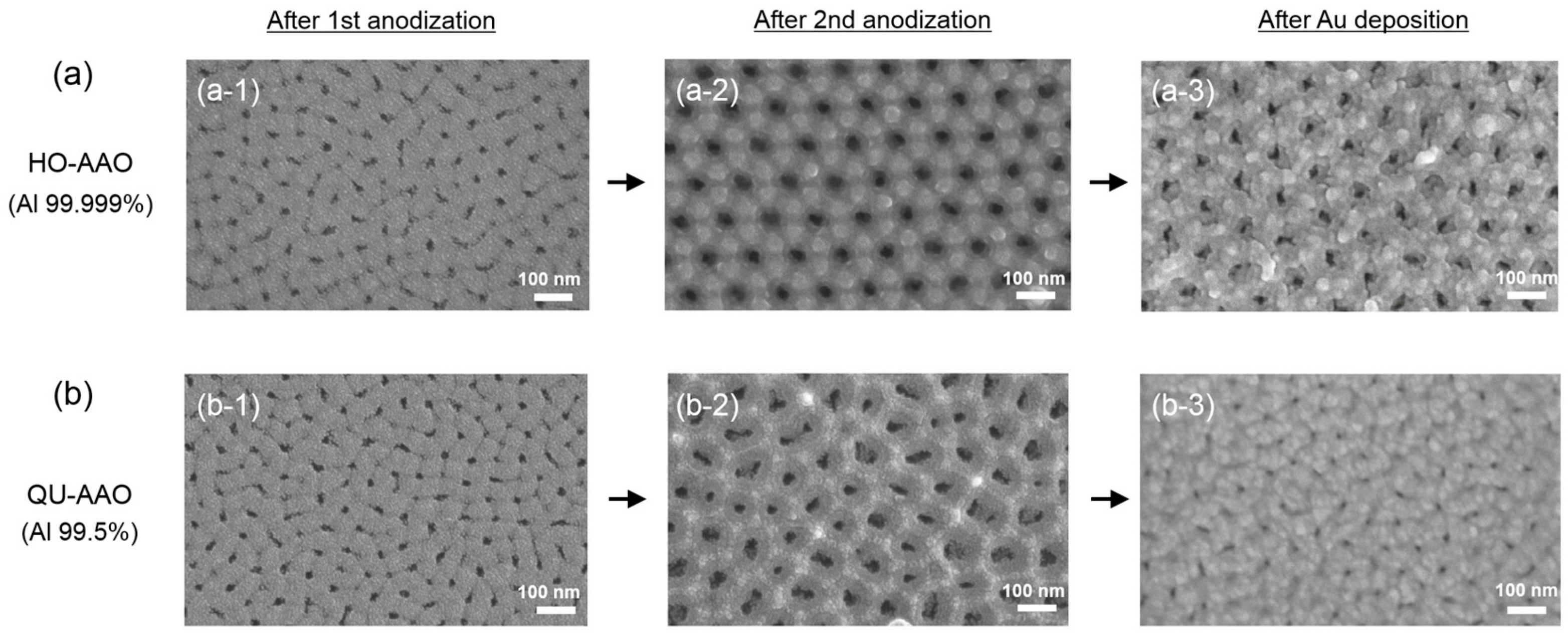
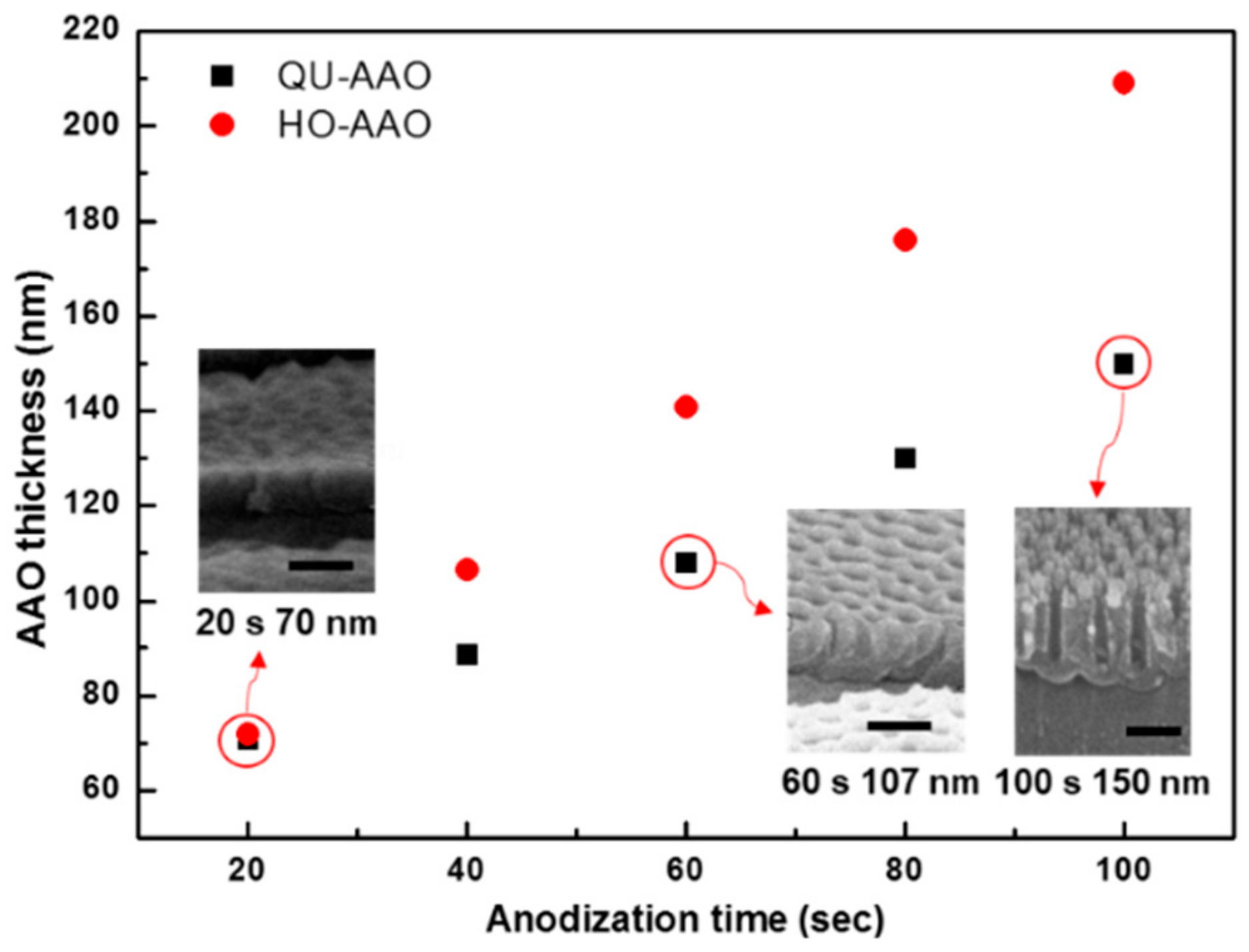
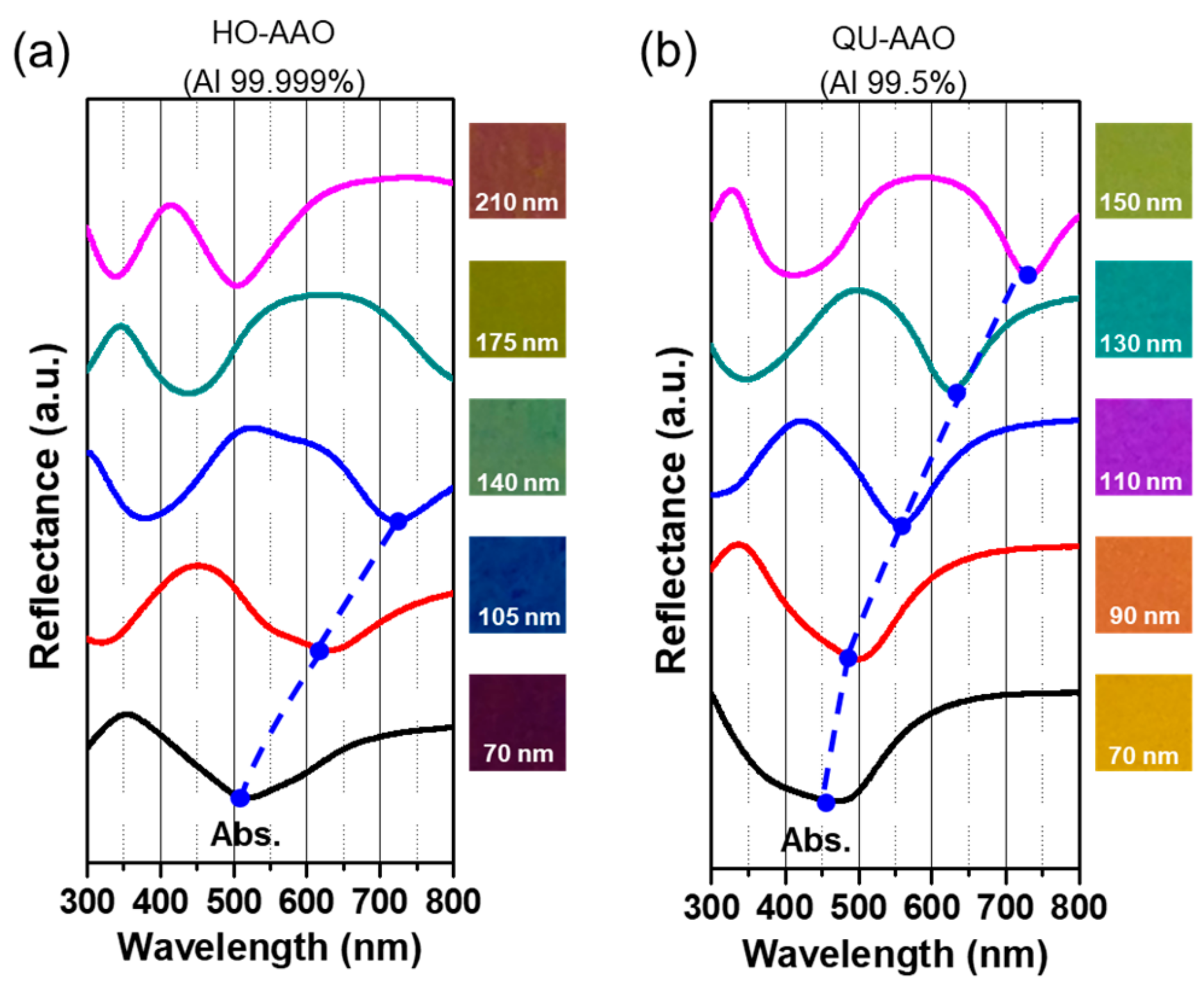
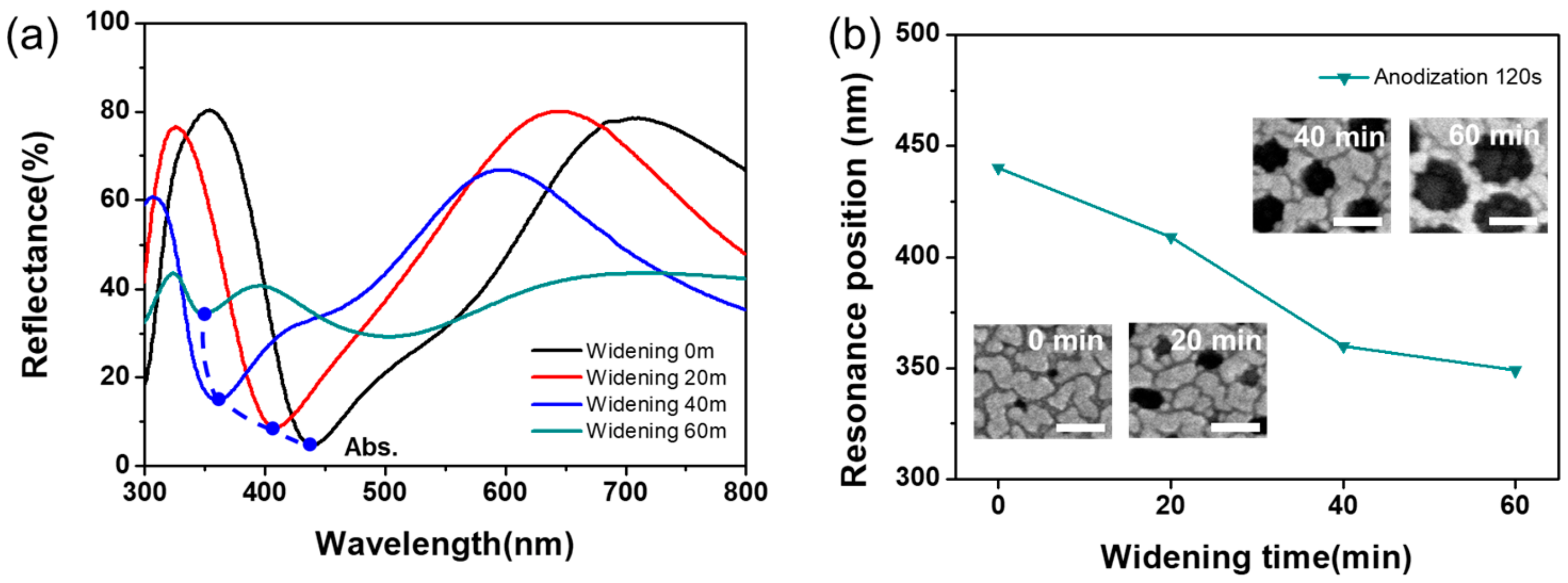
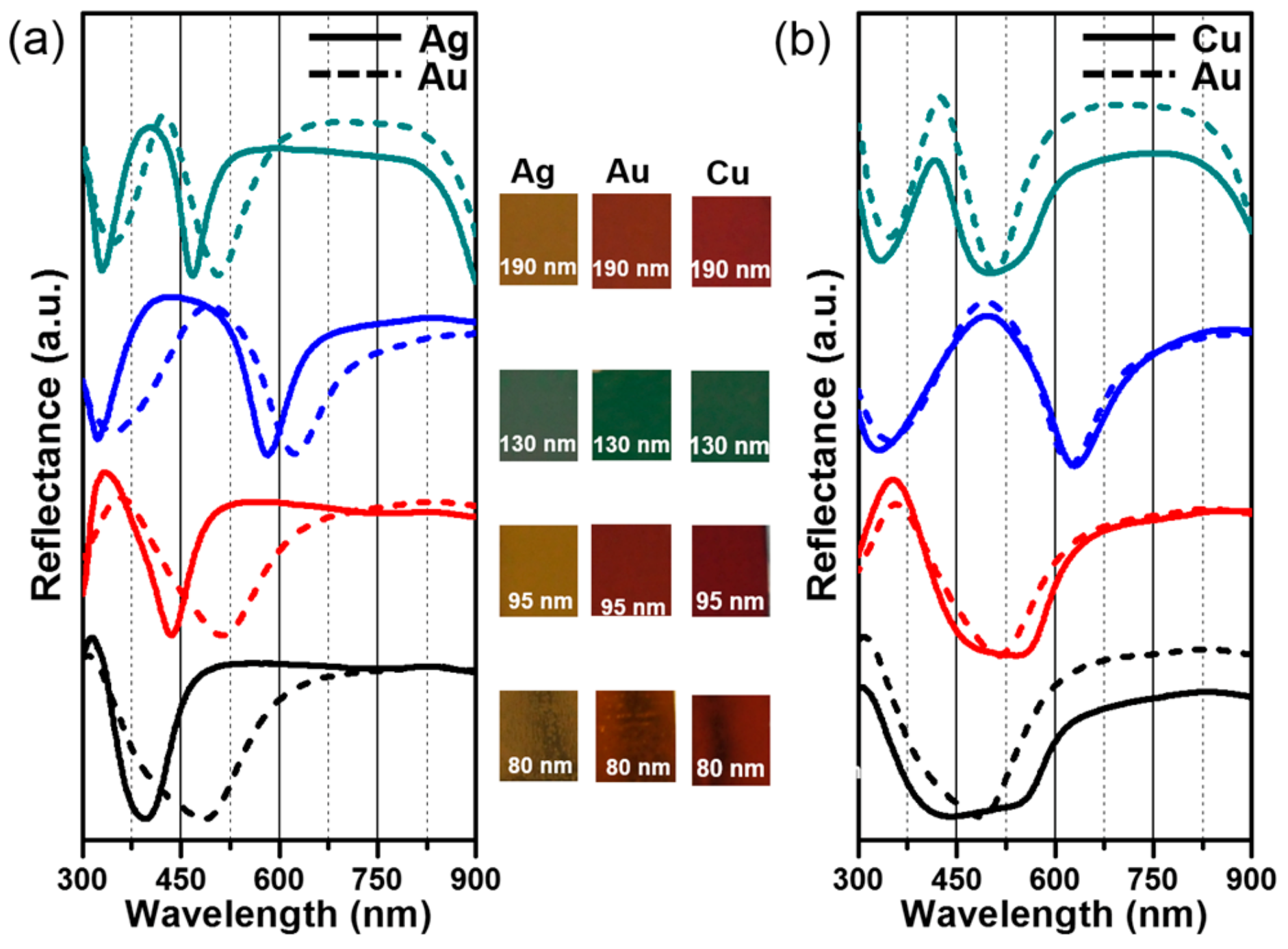
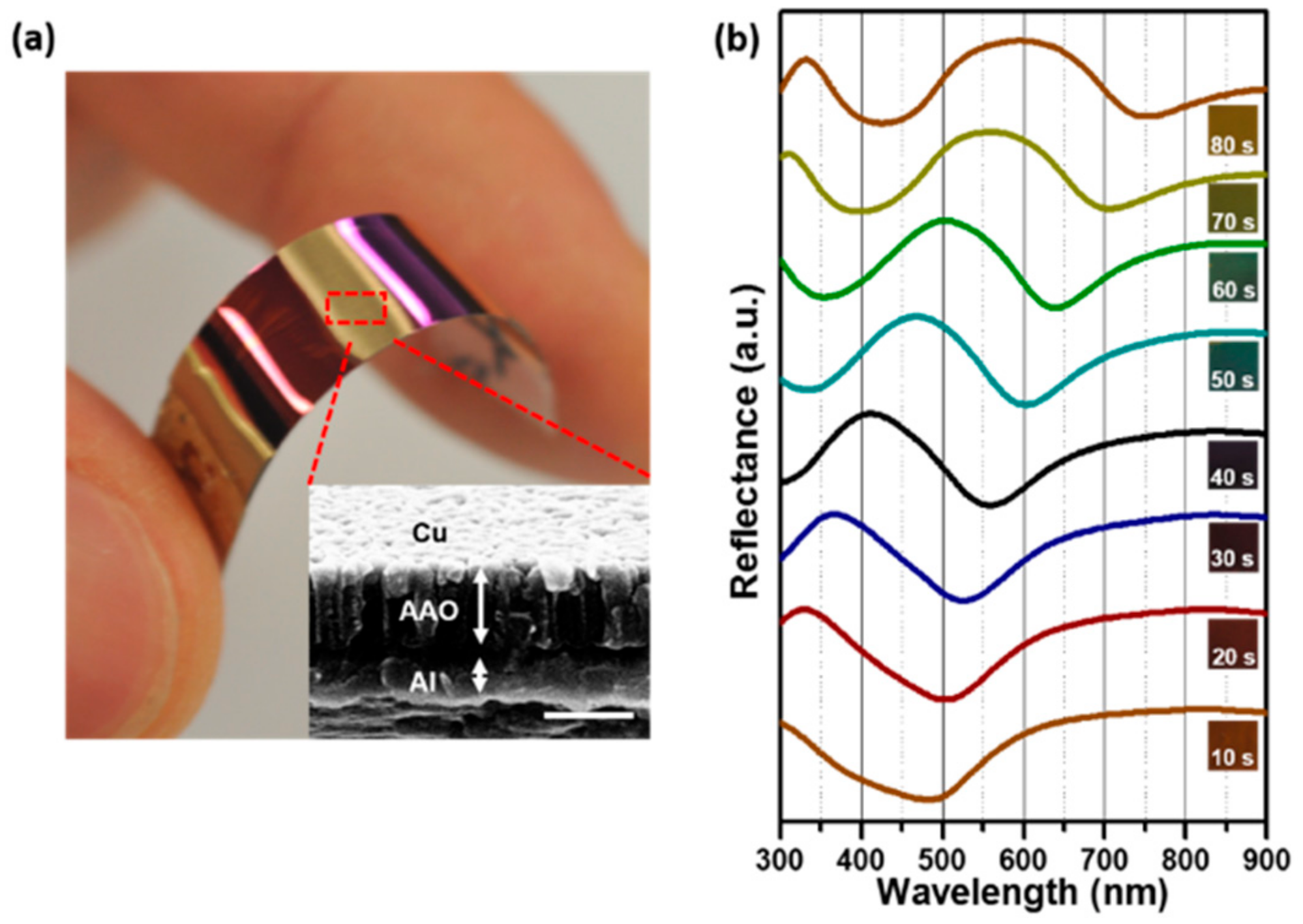
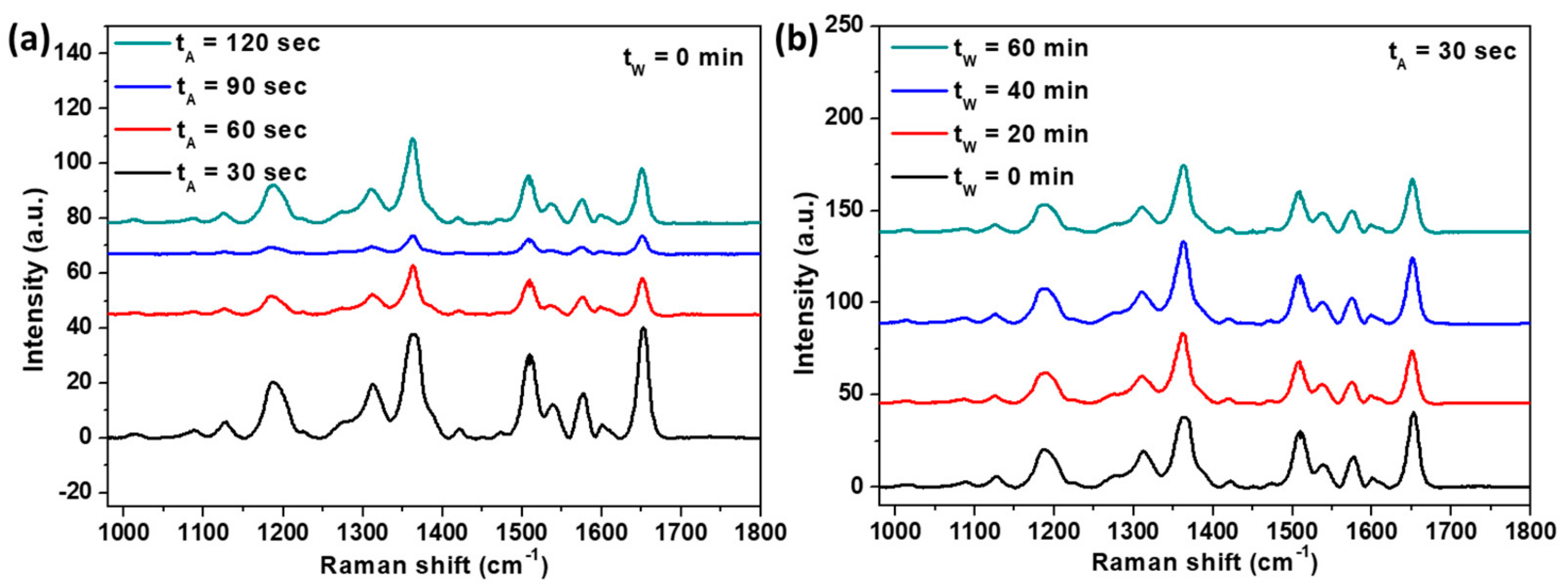
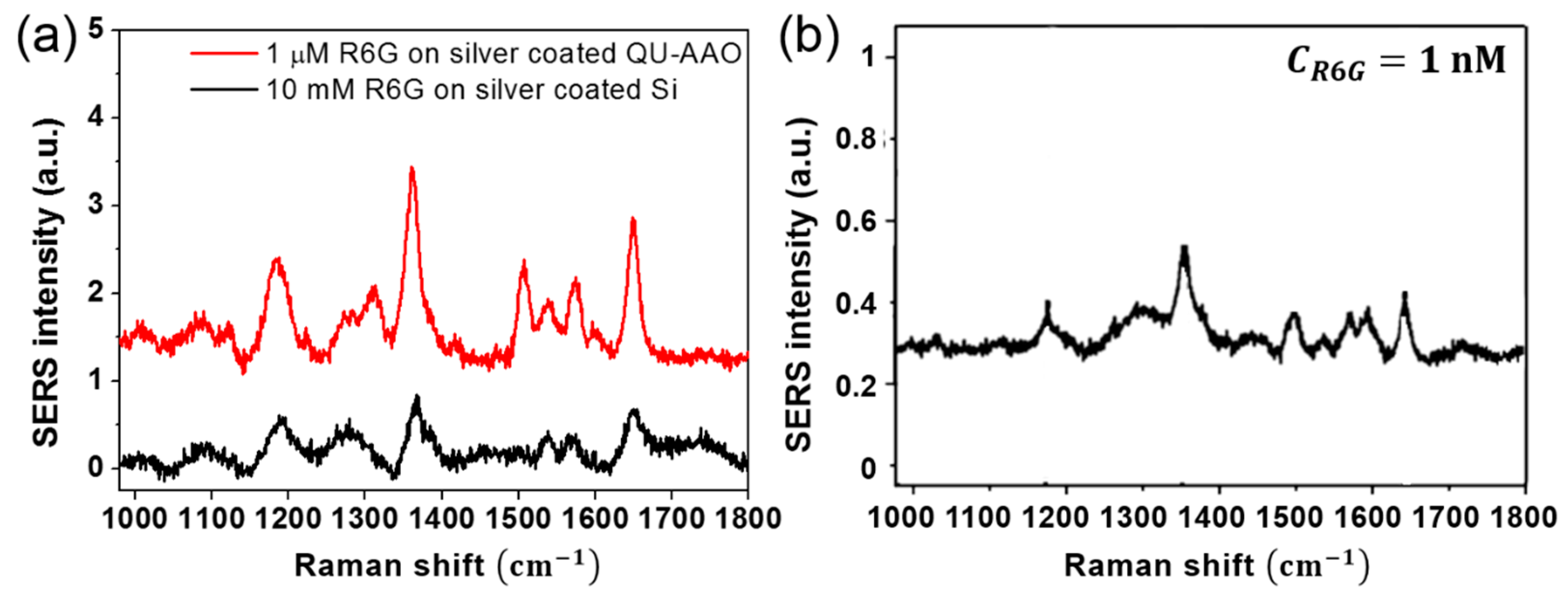
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Barbillon, G. Fabrication and SERS performances of metal/Si and metal/ZnO nanosensors: A review. Coatings 2019, 9, 86. [Google Scholar] [CrossRef]
- Maier, S.A.; Brongersma, M.L.; Kik, P.G.; Meltzer, S.; Requicha, A.A.G.; Atwater, H.A. Plasmonics-a route to nanoscale optical devices. Adv. Mater. 2001, 13, 1501–1505. [Google Scholar] [CrossRef]
- Zhang, N.M.Y.; Li, K.; Shum, P.P.; Yu, X.; Zeng, S.; Wu, Z.; Wang, Q.J.; Yong, K.T.; Wei, L. Hybrid graphene/gold plasmonic fiber-optic biosensor. Adv. Mater. Technol. 2017, 2, 1600185. [Google Scholar] [CrossRef]
- Ren, K.; Gan, Y.X.; Young, T.J.; Moutassem, Z.M.; Zhang, L. Photoelectrochemical responses of doped and coated titanium dioxide composite nanotube anodes. Compos. Part B Eng. 2013, 52, 292–302. [Google Scholar] [CrossRef]
- Xu, S.; Cao, Y.; Zhou, J.; Wang, X.; Xu, W. Plasmonic enhancement of fluorescence on silver nanoparticle films. Nanotechnology 2011, 22, 275715. [Google Scholar] [CrossRef] [PubMed]
- Ahmadivand, A.; Gerislioglu, B. Directional toroidal dipoles driven by oblique poloidal and loop current flows in plasmonic meta-atoms. J. Phys. Chem. C 2018, 122, 24304–24308. [Google Scholar] [CrossRef]
- Fleischman, D.; Fountaine, K.T.; Bukowsky, C.R.; Tagliabue, G.; Sweatlock, L.A.; Atwater, H.A. High spectral resolution plasmonic color filters with subwavelength dimensions. ACS Photon. 2019, 6, 332–338. [Google Scholar] [CrossRef]
- Yun, J.; Lee, H.; Mun, C.; Jahng, J.; Morrison, W.A.; Nowak, D.B.; Song, J.-H.; Kim, D.-K.; Bae, T.-S.; Kim, H.M.; et al. Fabrication and near-field visualization of a wafer-scale dense plasmonic nanostructured array. RSC Adv. 2018, 8, 6444–6451. [Google Scholar] [CrossRef]
- Brongersma, M.L.; Halas, N.J.; Nordlander, P. Plasmon-induced hot carrier science and technology. Nat. Nanotechnol. 2015, 10, 25–34. [Google Scholar] [CrossRef]
- Zhou, Q.; Meng, G.; Liu, J.; Huang, Z.; Han, F.; Zhu, C.; Kim, D.-J.; Kim, T.; Wu, N. A hierarchical nanostructure-based surface-enhanced Raman scattering sensor for preconcentration and detection of antibiotic pollutants. Adv. Mater. Technol. 2017, 2, 1700028. [Google Scholar] [CrossRef]
- He, J.; Villa, N.S.; Luo, Z.; An, S.; Shen, Q.; Tao, P.; Song, C.; Wu, J.; Deng, T.; Shang, W. Integrating plasmonic nanostructures with natural photonic architectures in Pd-modified morpho butterfly wings for sensitive hydrogen gas sensing. RSC Adv. 2018, 8, 32395–32400. [Google Scholar] [CrossRef]
- Lee, S.; Lee, M.H.; Shin, H.J.; Choi, D. Control of density and LSPR of Au nanoparticles on graphene. Nanotechnology 2013, 24, 275702–275708. [Google Scholar] [CrossRef]
- Gerislioglu, B.; Ahmadivand, A.; Pala, N. Functional quadrumer clusters for switching between Fano and charge transfer plasmons. IEEE Photon. Technol. Lett. 2017, 29, 2226–2229. [Google Scholar] [CrossRef]
- Naik, G.V.; Shalaev, V.M.; Boltasseva, A. Alternative plasmonic materials: beyond gold and silver. Adv. Mater. 2013, 25, 3264–3294. [Google Scholar] [CrossRef]
- Guler, U.; Shalaev, V.M.; Boltasseva, A. Nanoparticle plasmonics: Going practical with transition metal nitirdes. Mater. Today 2015, 18, 227–237. [Google Scholar] [CrossRef]
- Kim, S.; Kim, J.M.; Park, J.E.; Nam, J.M. Nonnoble-metal-based plasmonic nanomaterials: Recent advances and future perspectives. Adv. Mater. 2018, 30, 1704528. [Google Scholar] [CrossRef]
- Mueller, A.D.; Tobing, L.Y.M.; Zhang, D.H. Reliable fabrication of high aspect ratio plasmonic nanostructures based on seedless pulsed electrodeposition. Adv. Mater. Technol. 2018, 4, 1800364. [Google Scholar] [CrossRef]
- Agapov, R.L.; Srijanto, B.; Fowler, C.; Briggs, D.; Lavrik, N.V.; Sepaniak, M.J. Lithography-free approach to highly efficient, scalable SERS substrates based on disordered clusters of disc-on-pillar structures. Nanotechnology 2013, 24, 505302. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Nam, W.; Zhou, W. Scalable high-performance nanolaminated SERS substrates based on multistack vertically oriented plasmonic nanogaps. Adv. Mater. Technol. 2019, 4, 1800689. [Google Scholar] [CrossRef]
- Ahmadivand, A.; Semmlinger, M.; Dong, L.; Gerislioglu, B.; Nordlander, P.; Halas, N.J. Toroidal dipole-enhanced third harmonic generation of deep ultraviolet light using plasmonic meta-atoms. Nano Lett. 2019, 19, 605–611. [Google Scholar] [CrossRef]
- Lee, W.; Park, S.J. Porous anodic aluminum oxide: Anodization and templated synthesis of functional nanostructures. Chem. Rev. 2014, 114, 7487–7556. [Google Scholar] [CrossRef]
- Zhou, L.; Tan, Y.; Wang, J.; Xu, W.; Yuan, Y.; Cai, W.; Zhu, S.; Zhu, J. 3D self-assembly of aluminum nanoparticles for plasmon-enhanced solar desalination. Nat. Photon. 2016, 10, 393–398. [Google Scholar] [CrossRef]
- Sohn, A.; Gwon, M.; Choi, D.; Kim, D.W. Plasmonic coupling in three-dimensional Au nanoparticle assemblies fabricated by anodic aluminum oxide templates. J. Nanomater. 2013, 2013, 1–6. [Google Scholar] [CrossRef]
- Li, J.; Yu, W.; Zheng, D.; Zhao, X.; Choi, C.H.; Sun, G. Hot embossing for whole Teflon superhydrophobic surface. Coatings 2018, 8, 227. [Google Scholar] [CrossRef]
- Hyam, R.S.; Choi, D. Effects of titanium foil thickness on TiO2 nanostructures synthesized by anodization. RSC. Adv. 2013, 3, 7057–7063. [Google Scholar] [CrossRef]
- Wang, S.; Liu, H.; Han, J. Comprehensive study of Au nano-mesh as a catalyst in the fabrication of silicon nanowires arrays by metal-assisted chemical etching. Coatings 2019, 9, 149. [Google Scholar] [CrossRef]
- Gan, Y.; Yu, Z.; Gan, J.; Cheng, W.; Li, M. Gold catalyst-assisted metal organic chemical vapor deposition of Bi–Te–Ni–Cu–Au complex thermoelectric materials on anodic aluminum oxide nanoporous template. Coatings 2018, 8, 166. [Google Scholar] [CrossRef]
- Belushkin, A.; Yesilkoy, F.; Altug, H. Nanoparticle-enhanced plasmonic biosensor for digital biomarker detection in a microarray. ACS Nano 2018, 12, 4453–4461. [Google Scholar] [CrossRef]
- Shen, Y.; Zhou, J.; Liu, T.; Tao, Y.; Jiang, R.; Liu, M.; Xiao, G.; Zhu, J.; Zhou, J.-K.; Wang, X.; et al. Plasmonic gold mushroom arrays with refractive index sensing figures of merit approaching the theoretical limit. Nat. Commun. 2013, 4, 2381. [Google Scholar] [CrossRef]
- Jia, P.; Jiang, H.; Sabarinathan, J.; Yang, J. Plasmonic nanohole array sensors fabricated by template transfer with improved optical performance. Nanotechnology 2013, 24, 195501. [Google Scholar] [CrossRef]
- Bruera, F.A.; Kramer, G.R.; Vera, M.L.; Ares, A.E. Synthesis and morphological characterization of nanoporous aluminum oxide films by using a single anodization step. Coatings 2019, 9, 115. [Google Scholar] [CrossRef]
- Zhou, Q.; Meng, G.; Huang, Q.; Zhu, C.; Tang, H.; Qian, Y.; Chen, B.; Chen, B. Ag-nanoparticles-decorated NiO-nanoflakes grafted Ni-nanorod arrays stuck out of porous AAO as effective SERS substrates. Phys. Chem. Chem. Phys. 2014, 16, 3686–3692. [Google Scholar] [CrossRef] [PubMed]
- Shan, D.; Huang, L.; Li, X.; Zhang, W.; Wang, J.; Cheng, L.; Feng, X.; Liu, Y.; Zhu, J.; Zhang, Y. Surface plasmon resonance and interference coenhanced SERS substrate of AAO/Al-based Ag nanostructure arrays. J. Phys. Chem. C 2014, 118, 23930–23936. [Google Scholar] [CrossRef]
- Le Ru, E.C.; Blackie, E.; Meyer, M.; Etchegoin, P.G. Surface enhanced Raman scattering enhancement factors: A comprehensive study. J. Phys. Chem. C 2007, 111, 13794–13803. [Google Scholar] [CrossRef]
- Yao, Z.; Wang, C.; Li, Y.; Kim, N.Y. AAO-assisted synthesis of highly ordered, large-scale TiO2 nanowire arrays via sputtering and atomic layer deposition. Nanoscale Res. Lett. 2015, 10, 166. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yin, A.J.; Li, J.; Jian, W.; Bennett, A.J.; Xu, J.M. Fabrication of highly ordered metallic nanowire arrays by electrodeposition. Appl. Phys. Lett. 2001, 79, 1039–1041. [Google Scholar] [CrossRef]
- Zhang, C.; Li, W.; Yu, D.; Wang, Y.; Yin, M.; Wang, H.; Song, Y.; Zhu, X.; Chang, P.; Chen, X.; et al. Wafer-scale highly ordered anodic aluminum oxide by soft nanoimprinting lithography for optoelectronics light management. Adv. Mater. Interfaces 2017, 4, 1601116. [Google Scholar] [CrossRef]
- Choi, D.; Shin, K.S.; Yoon, D.; Chung, D.S.; Jin, Y.W.; Lee, L.P. Plasmonic optical interference. Nano Lett. 2014, 14, 3374–3381. [Google Scholar] [CrossRef]
- Jo, H.; Yoon, D.; Sohn, A.; Kim, D.-W.; Choi, Y.; Kang, T.; Choi, D.; Kim, S.-W.; Lee, L.P. Asymmetrically coupled plasmonic core and nanotriplet satellites. J. Phys. Chem. C 2014, 118, 18659–18667. [Google Scholar] [CrossRef]
- Choi, D.; Choi, Y.; Hong, S.; Kang, T.; Lee, L.P. Self-organized hexagonal-nanopore SERS array. Small 2010, 16, 1741–1744. [Google Scholar] [CrossRef]
- Hu, M.; Chen, J.; Li, Z.Y.; Au, L.; Hartland, G.V.; Li, X.; Marquez, M.; Xia, Y. Gold nanostructures: Engineering their plasmonic properties for biomedical applications. Chem. Soc. Rev. 2006, 35, 1084–1094. [Google Scholar] [CrossRef] [PubMed]
- Kravets, V.G.; Jalil, R.; Kim, Y.J.; Ansell, D.; Aznakayeva, D.E.; Thackray, B.; Britnell, L.; Belle, B.D.; Withers, F.; Radko, I.P.; et al. Graphene-protected copper and silver plasmonics. Sci. Rep. 2014, 4, 5517. [Google Scholar] [CrossRef]
- Guay, J.M.; Killaire, G.; Gordon, P.G.; Barry, S.T.; Berini, P.; Weck, A. Passivation of plasmonic colors on bulk silver by atomic layer deposition of aluminum oxide. Langmuir 2018, 34, 4998–5010. [Google Scholar] [CrossRef] [PubMed]
- Wes, P.R.; Ishii, S.; Naik, G.V.; Emani, N.K.; Shalaev, V.M.; Boltasseva, A. Searching for better plasmonic materials. Laser Photon. Rev. 2010, 4, 795–808. [Google Scholar] [CrossRef]
- Hildebrandt, P.; Stockburger, M. Surface-enhanced resonance Raman spectroscopy of Rhodamine 6G adsorbed on colloidal silver. J. Phys. Chem. 1984, 88, 5395–5944. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bae, Y.; Yu, J.; Jung, Y.; Lee, D.; Choi, D. Cost-Effective and High-Throughput Plasmonic Interference Coupled Nanostructures by Using Quasi-Uniform Anodic Aluminum Oxide. Coatings 2019, 9, 420. https://doi.org/10.3390/coatings9070420
Bae Y, Yu J, Jung Y, Lee D, Choi D. Cost-Effective and High-Throughput Plasmonic Interference Coupled Nanostructures by Using Quasi-Uniform Anodic Aluminum Oxide. Coatings. 2019; 9(7):420. https://doi.org/10.3390/coatings9070420
Chicago/Turabian StyleBae, Yoonsu, Jiseop Yu, Yeonseok Jung, Donghun Lee, and Dukhyun Choi. 2019. "Cost-Effective and High-Throughput Plasmonic Interference Coupled Nanostructures by Using Quasi-Uniform Anodic Aluminum Oxide" Coatings 9, no. 7: 420. https://doi.org/10.3390/coatings9070420
APA StyleBae, Y., Yu, J., Jung, Y., Lee, D., & Choi, D. (2019). Cost-Effective and High-Throughput Plasmonic Interference Coupled Nanostructures by Using Quasi-Uniform Anodic Aluminum Oxide. Coatings, 9(7), 420. https://doi.org/10.3390/coatings9070420





