Hydrophobic and Icephobic Behaviour of Polyurethane-Based Nanocomposite Coatings
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation and Deposition of Silica/Polyurethane (PUR) Nanocomposite Coatings
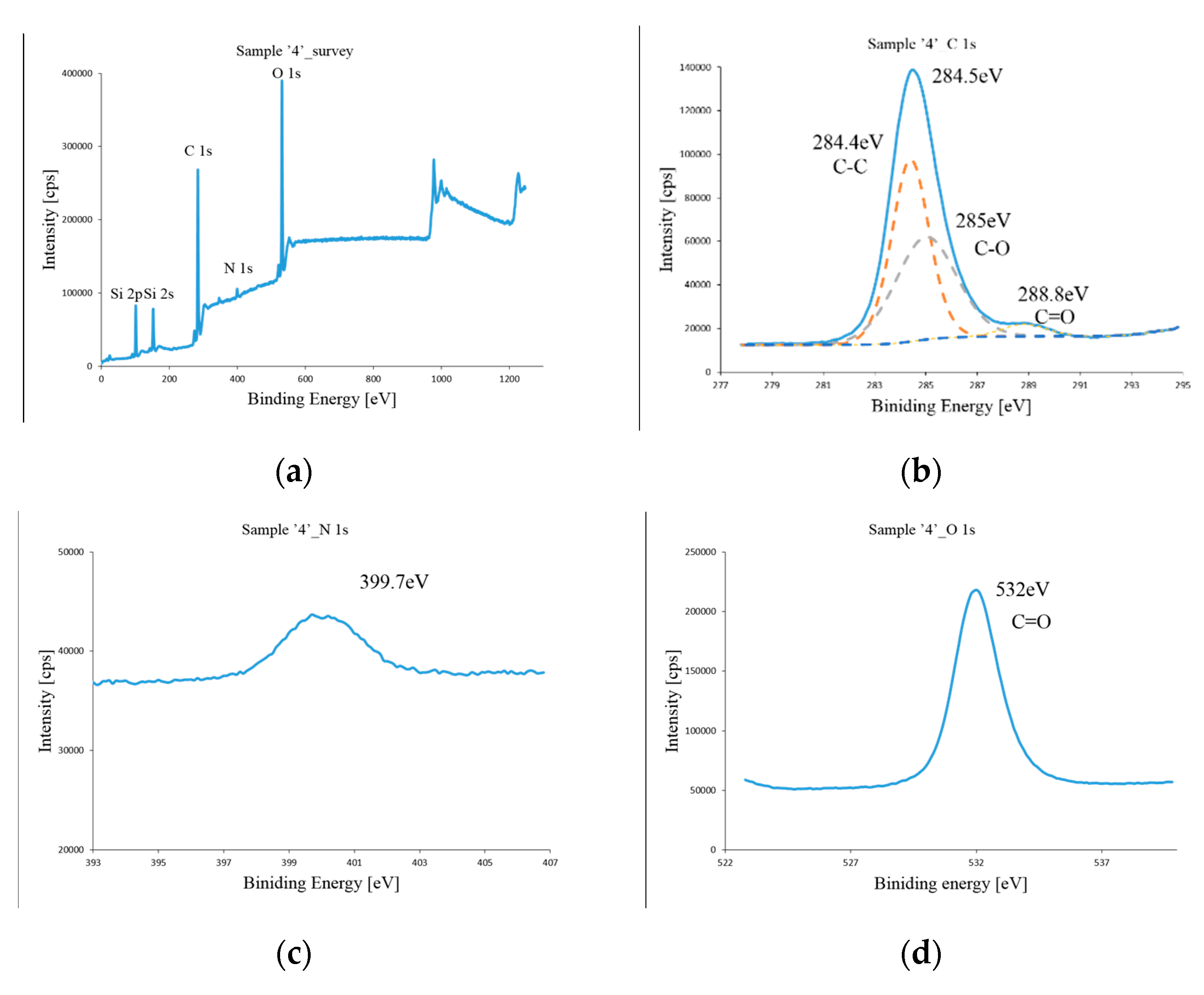
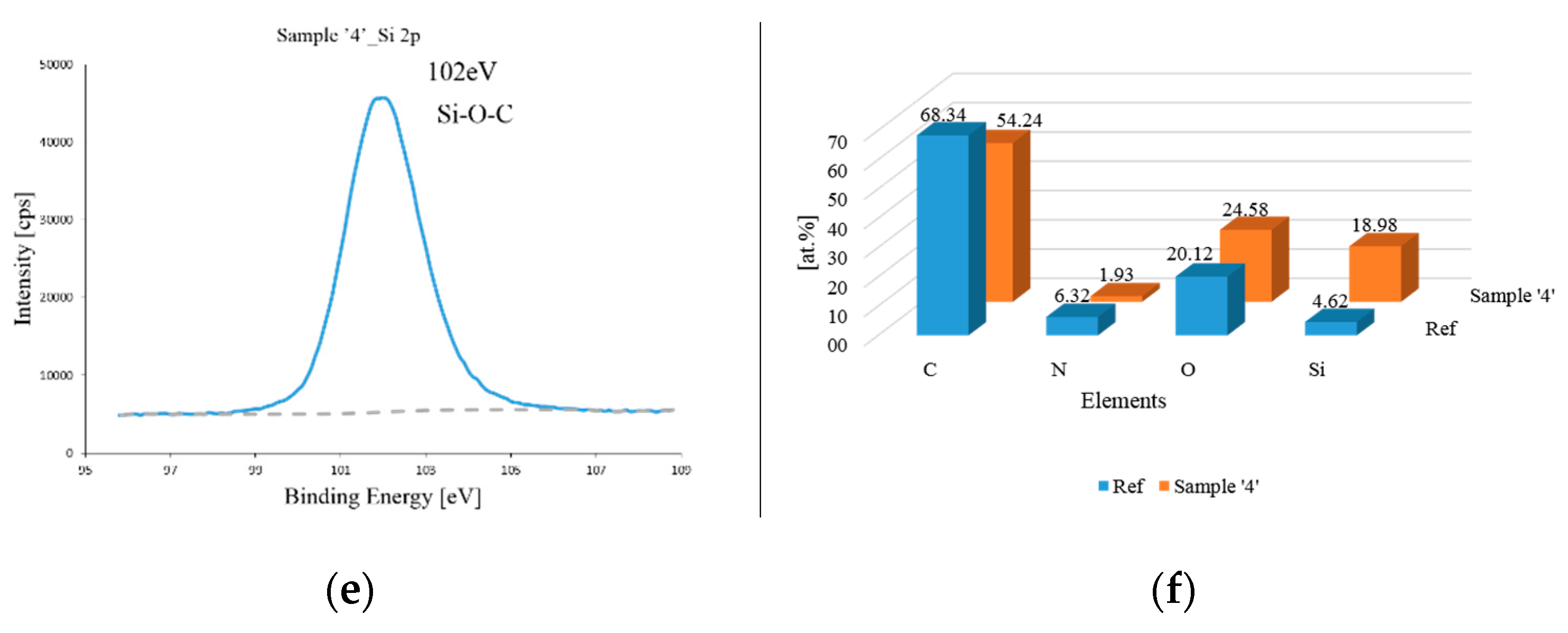
2.3. Characterization
3. Results
3.1. Thickness and Roughness
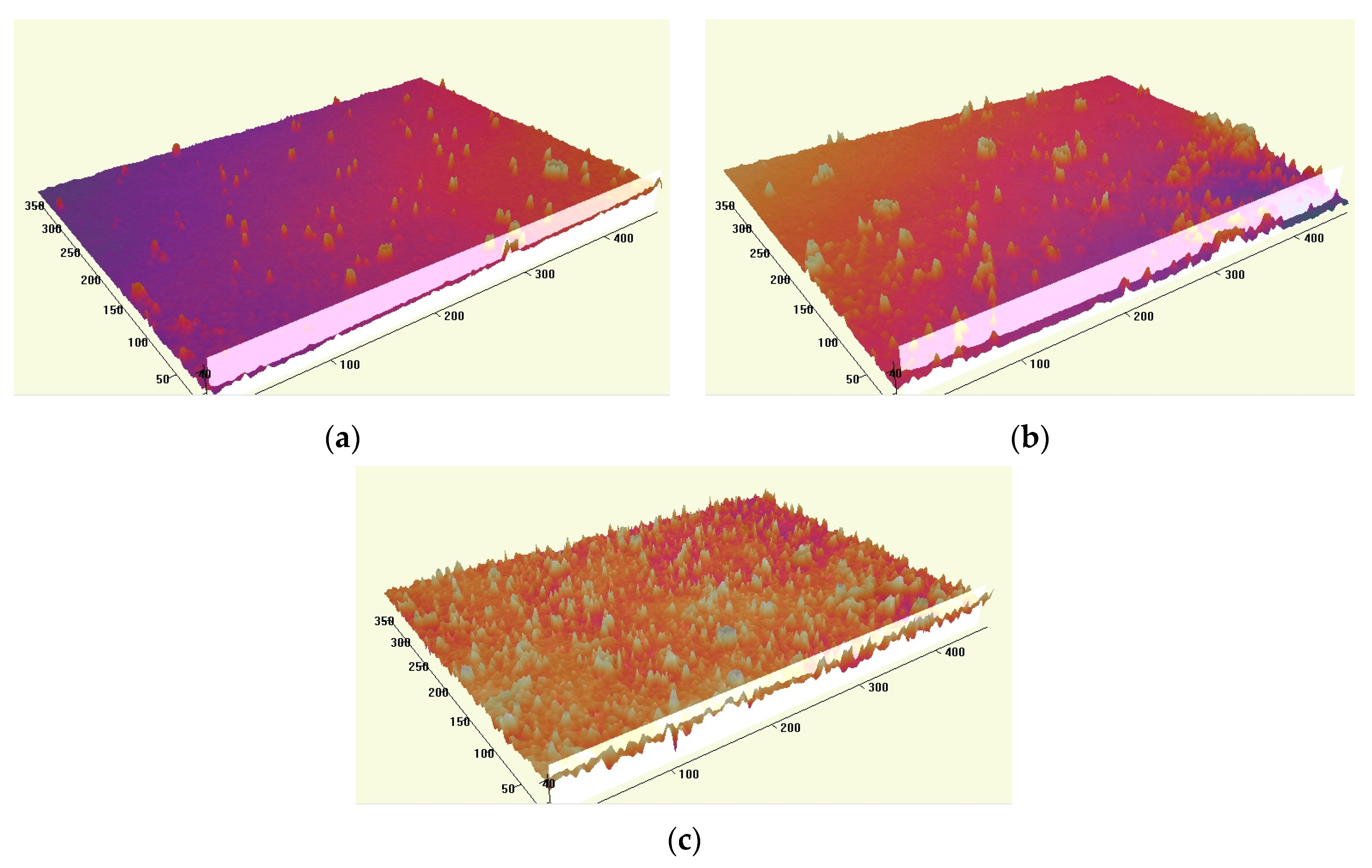
3.2. Surface Morphology
3.3. Wettability Behaviour
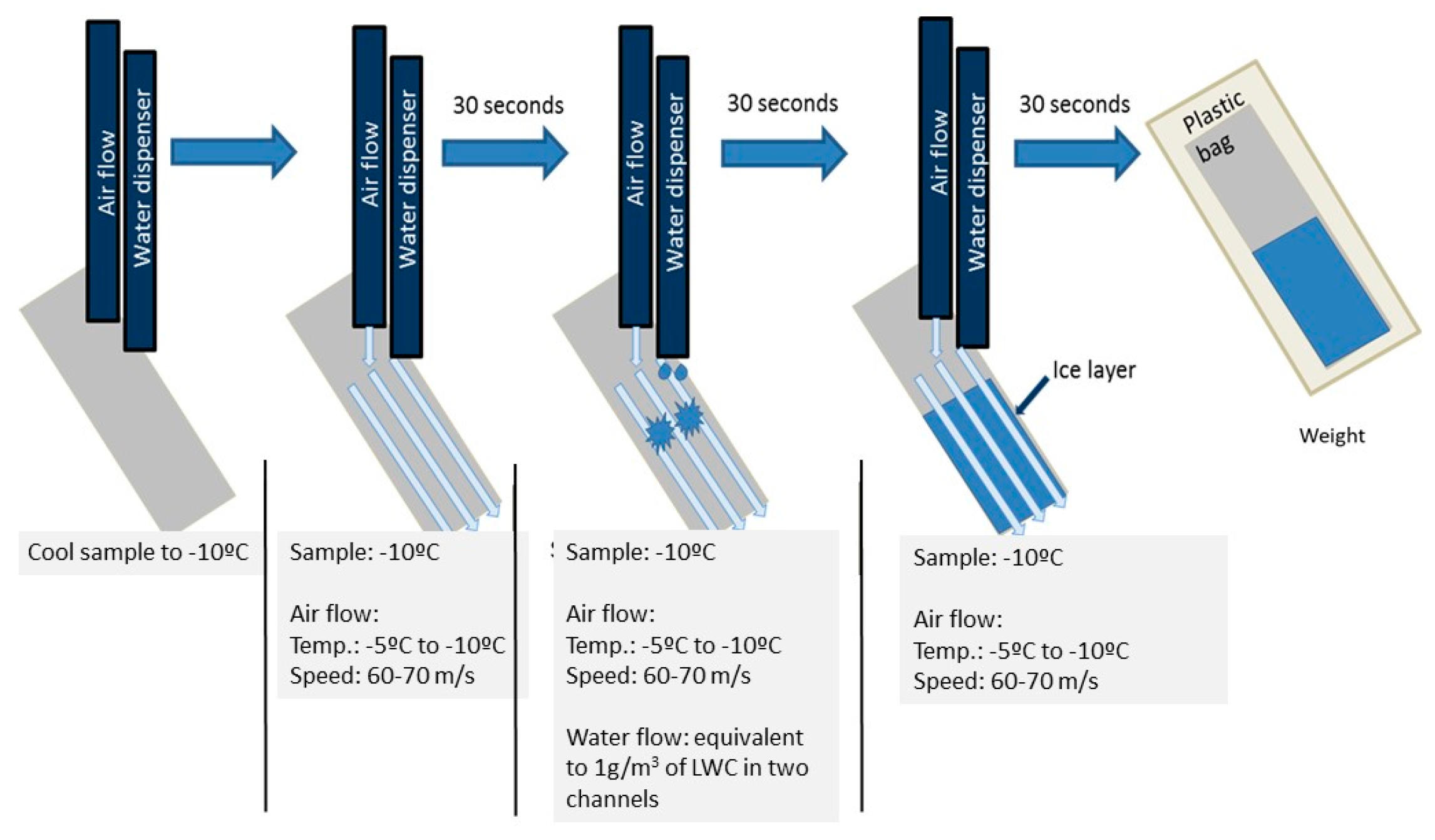
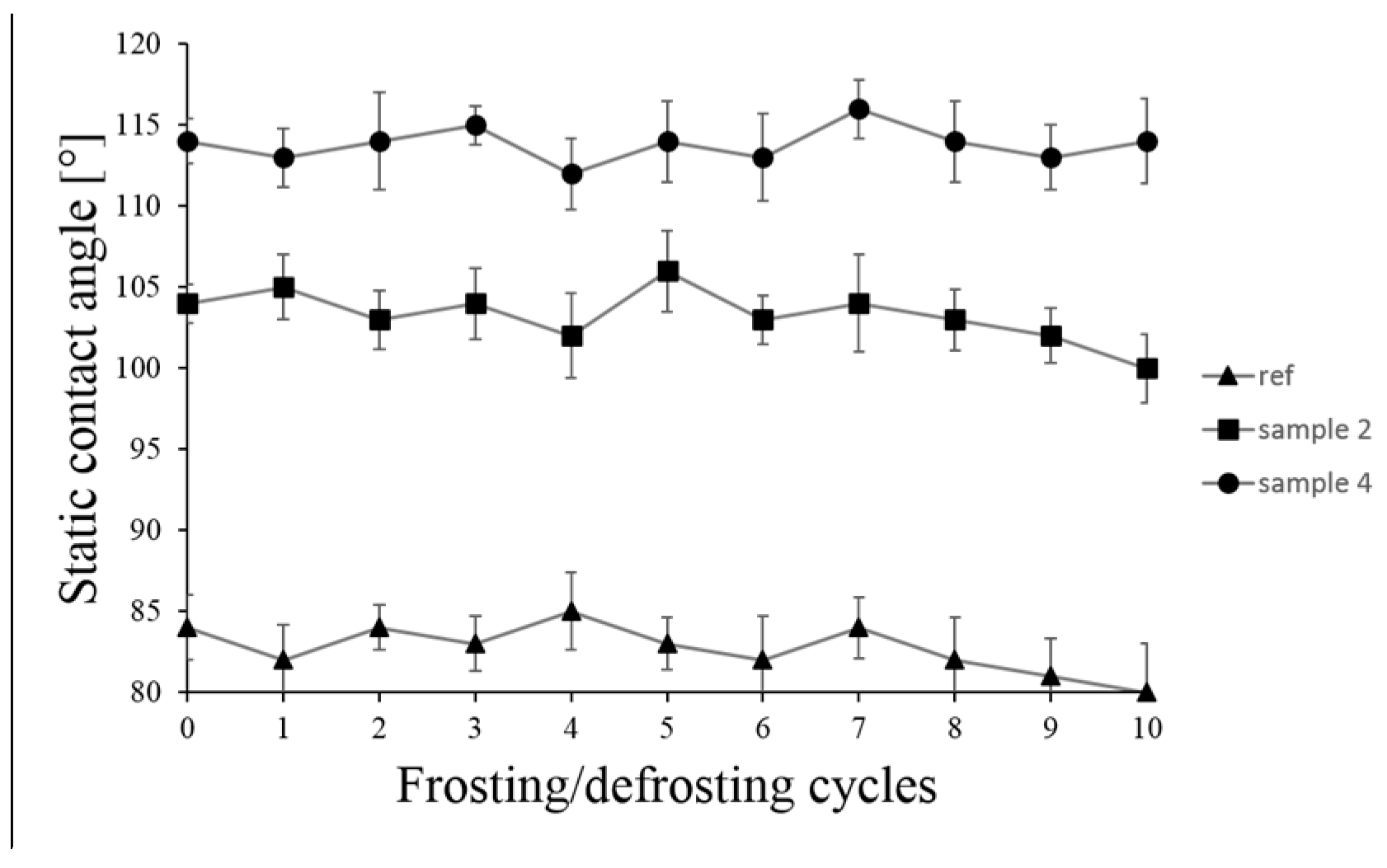
3.4. Frosting/Defrosting Cycles
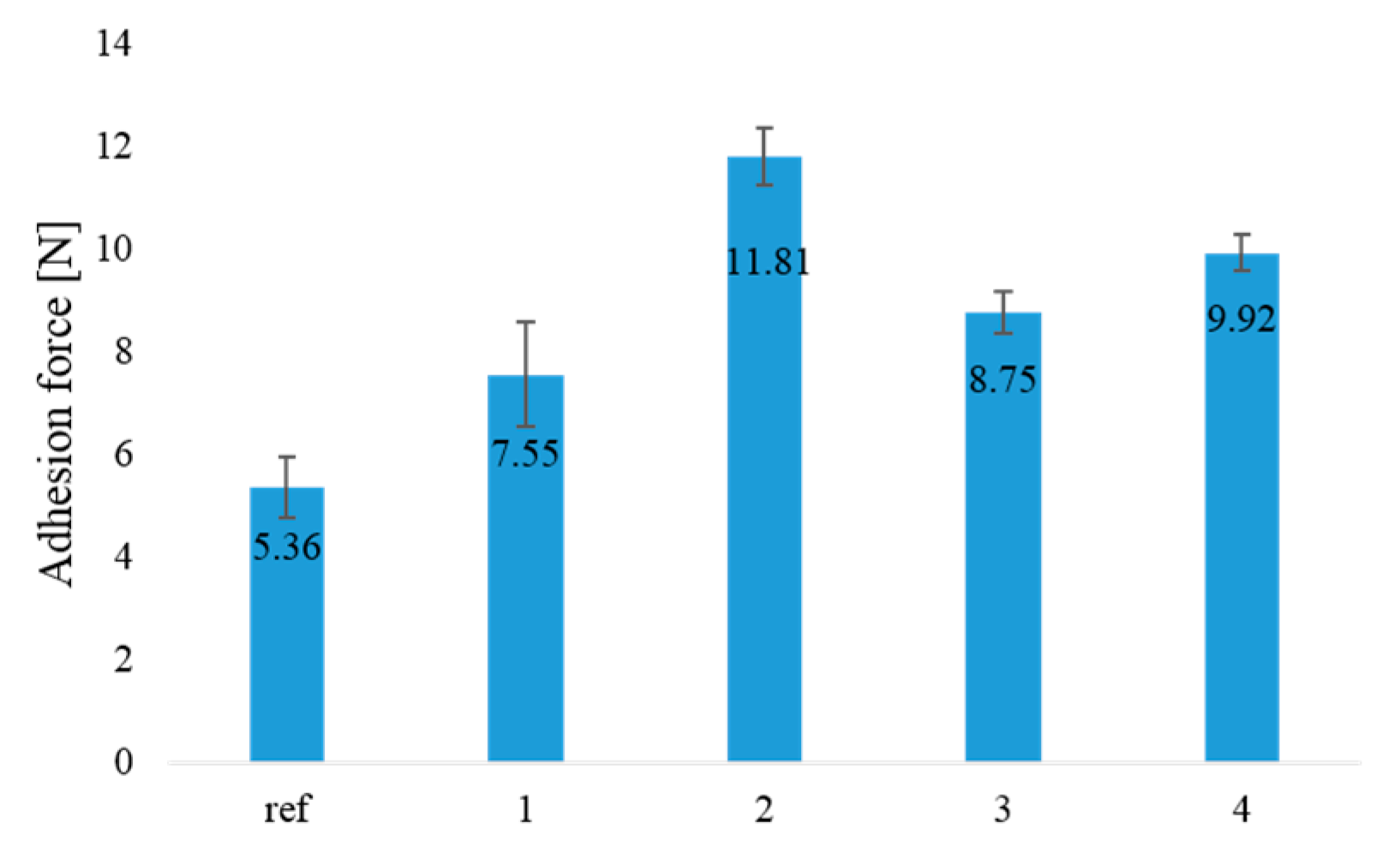
3.5. Coating Adhesion
3.6. Ice Accretion Test
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Antonini, C.; Innocenti, M.; Horn, T.; Marengo, M.; Amirfazli, A. Understanding the effect of superhydrophobic coatings on energy reduction in anti-icing systems. Cold Reg. Sci. Technol. 2011, 67, 58–67. [Google Scholar] [CrossRef]
- Boreyko, J.B.; Collier, C.P. Delayed frost growth on jumping-drop superhydrophobic surfaces. ACS Nano 2013, 7, 1618–1627. [Google Scholar] [CrossRef] [PubMed]
- Brouwers, E.W.; Peterson, A.A.; Palacios, J.L.; Centolanza, L.R. Ice adhesion strength measurements for rotor blade leading edge materials. In Proceedings of the 67th American Helicopter Society International Annual Forum, Virginia Beach, VA, USA, 3–5 May 2011. [Google Scholar]
- Boinovich, L.B.; Emelyanenko, A.M.; Emelyanenko, K.A.; Modin, E.B. Modus operandi of protective and anti-icing mechanisms underlying the design of longstanding outdoor icephobic coatings. ACS Nano 2019, 13, 4335–4346. [Google Scholar] [CrossRef] [PubMed]
- Tarquini, S.; Antonini, C.; Amirfazli, A.; Marengo, M.; Palacios, J. Investigation of ice shedding properties of superhydrophobic coatings on helicopter blades. Cold Reg. Sci. Technol. 2014, 100, 50–58. [Google Scholar] [CrossRef]
- Ky, P. Annual Safety Review 2017; EASA: Cologne, Germany, 2017; p. 12.
- Coffman, H.J. Helicopter Rotor Icing Protection Methods. J. Am. Helicopter Soc. 1987, 32, 34–39. [Google Scholar] [CrossRef]
- Itagaki, K. Self-Shedding from High-Speed Rotors. In Mechanical Ice Release Processes; US Army Corps of Engineers, Cold Regions Research & Engineering Laboratory: Hanover, NH, USA, 1983. [Google Scholar]
- Mishchenko, L.; Hatton, B.; Bahadur, V.; Taylor, J.A.; Krupenkin, T.; Aizenberg, J. Design of ice-free nanostructured surfaces based on repulsion of impacting water droplets. ACS Nano 2010, 4, 7699–7707. [Google Scholar] [CrossRef]
- Karmouch, R.; Coude, S.; Abel, G.; Ross, G.G. Icephobic PTFE coatings for wind turbines operating in cold climate conditions. In Proceedings of the 2009 IEEE Electrical Power & Energy Conference, Montreal, QC, Canada, 22–23 October 2009. [Google Scholar]
- Kulinich, S.A.; Farzaneh, M. Ice adhesion on super-hydrophobic surfaces. Appl. Surf. Sci. 2009, 255, 8153–8157. [Google Scholar] [CrossRef]
- Seyfi, J.; Jahari, S.H.; Khonakdar, H.A.; Sadeghi, G.M.M.; Zohuri, G.; Hejazi, I.; Simon, F. Fabrication of robust and thermally stable superhydrophobic nanocomposite coatings based on thermoplastic polyurethane and silica nanoparticles. Appl. Surf. Sci. 2015, 347, 224–230. [Google Scholar] [CrossRef]
- Menini, R.; Farzaneh, M. Advanced Icephobic Coatings. J. Adhes. Sci. Technol. 2011, 25, 971–992. [Google Scholar] [CrossRef]
- Work, A.; Lian, Y. A critical review of the measurement of ice adhesion to solid substrates. Prog. Aerosp. Sci. 2018, 98, 1–26. [Google Scholar] [CrossRef]
- Koch, K.; Bhushan, B.; Jung, Y.C.; Barthlott, W. Fabrication of artificial Lotus leaves and significance of hierarchical structure for superhydrophobicity and low adhesion. Soft Matter 2009, 5, 1386–1393. [Google Scholar] [CrossRef]
- Koch, K.; Bhushan, B.; Barthlott, W. Diversity of structure, morphology and wetting of plant surfaces. Soft Matter 2008, 4, 1943–1963. [Google Scholar] [CrossRef]
- Hejazi, I.; Sadeghi, G.M.M.; Seyfi, J.; Jafari, S.H.; Khonakdar, H.A. Self-cleaning behavior in polyurethane/silica coatings via formation of a hierarchical packed morphology of nanoparticles. Appl. Surf. Sci. 2016, 368, 216–223. [Google Scholar] [CrossRef]
- El Dessouky, W.I.; Abbas, R.; Sadik, W.A.; El Demerdash, A.G.M.; Hefnawy, A. Improved adhesion of superhydrophobic layer on metal surfaces via one step spraying method. Arab. J. Chem. 2017, 10, 368–377. [Google Scholar] [CrossRef]
- Lazauskas, A.; Guobiene, A.; Prosycevas, I.; Baltrusaitis, V.; Grigaliunas, V.; Narmontas, P.; Baltrusaitis, J. Water droplet behavior on superhydrophobic SiO2 nanocomposite films during icing/deicing cycles. Mater. Charact. 2013, 82, 9–16. [Google Scholar] [CrossRef]
- Król, P.; Król, B.; Kozakiewicz, J.; Zapotoczny, S.; Pilch-Pitera, B.; Kozdra, S. Composites prepared from polyurethanes modified with silicone-acrylic nanopowders. Prog. Org. Coat. 2015, 81, 72–79. [Google Scholar] [CrossRef]
- Verma, G.; Kaushik, A.; Ghosh, A.K. Comparative assessment of nano-morphology and properties of spray coated clear polyurethane coatings reinforced with different organoclays. Prog. Org. Coat. 2013, 76, 1046–1056. [Google Scholar] [CrossRef]
- West, J.O.F.; Critchlow, G.W.; Lake, D.R.; Banks, R. Development of a superhydrophobic polyurethane-based coating from a two-step plasma-fluoroalkyl silane treatment. Int. J. Adhes. Adhes. 2016, 68, 195–204. [Google Scholar] [CrossRef]
- Tang, Y.; Yang, J.; Yin, L.; Chen, B.; Tang, H.; Liu, C.; Li, C. Fabrication of superhydrophobic polyurethane/MoS2 nanocomposite coatings with wear-resistance. Colloids Surf. A Physicochem. Eng. Asp. 2014, 459, 261–266. [Google Scholar] [CrossRef]
- Kosak Söz, C.; Yilgör, E.; Yilgör, I. Influence of the average surface roughness on the formation of superhydrophobic polymer surfaces through spin-coating with hydrophobic fumed silica. Polymer 2015, 62, 118–128. [Google Scholar] [CrossRef]
- Zou, M.; Beckford, S.; Wei, R.; Ellis, C.; Hatton, G.; Miller, M.A. Effects of surface roughness and energy on ice adhesion strength. Appl. Surf. Sci. 2011, 257, 3786–3792. [Google Scholar] [CrossRef]
- Bharathidasan, T.; Kumar, S.V.; Bobji, M.S.; Chakradhar, R.P.S.; Basu, B.J. Effect of wettability and surface roughness on ice-adhesion strength of hydrophilic, hydrophobic and superhydrophobic surfaces. Appl. Surf. Sci. 2014, 314, 241–250. [Google Scholar] [CrossRef]
- Zhao, H.; Hao, T.H.; Hu, G.H.; Shi, D.; Huang, D.; Jiang, T.; Zhang, Q.C. Preparation and characterization of polyurethanes with cross-linked siloxane in the side chain by sol-gel reactions. Materials 2017, 10, 247. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Hong, R.Y.; Xie, H.D.; Feng, W.G. One-step synthesis of functional silica nanoparticles for reinforcement of polyurethane coatings. Powder Technol. 2012, 218, 23–30. [Google Scholar] [CrossRef]
- Shirtcliffe, N.J.; McHale, G.; Atherton, S.; Newton, M.I. An introduction to superhydrophobicity. Adv. Colloid Interface Sci. 2010, 161, 124–138. [Google Scholar] [CrossRef]
- Furstner, R.; Barthlott, W.; Neinhuis, C.; Walzel, P. Wetting and self-cleaning properties of artificial superhydrophobic surfaces. Langmuir 2005, 21, 956–961. [Google Scholar] [CrossRef]
- Hang, T.T.X.; Dung, N.T.; Truc, T.A.; Duong, N.T.; Truoc, B.V.; Vu, P.G.; Hoang, T.; Thanh, D.T.M.; Olivier, M.G. Effect of silane modified nano ZnO on UV degradation of polyurethane coatings. Prog. Org. Coat. 2015, 79, 68–74. [Google Scholar] [CrossRef]
- Farhadi, S.; Farzaneh, M.; Simard, S. On stability and ice-releasing performance of nanostructured fluoro-alkylsilane-based superhydrophobic Al alloy2024 surfaces. Int. J. Theor. Appl. Nanotechnol. 2012, 1, 38–45. [Google Scholar] [CrossRef][Green Version]
- Farhadi, S.; Farzaneh, M.; Kulinich, S.A. Anti-icing performance of superhydrophobic surfaces. Appl. Surf. Sci. 2011, 257, 6264–6269. [Google Scholar] [CrossRef]
- Liao, R.; Zuo, Z.; Guo, C.; Zhuang, A.; Zhao, X.; Yuan, Y. Anti-icing performance in glaze ice of nanostructured film prepared by RF magnetron sputtering. Appl. Surf. Sci. 2015, 356, 539–545. [Google Scholar] [CrossRef]



| Sample | SiO2 Content [wt %] | DC88 Content [wt %] |
|---|---|---|
| ref | - | - |
| 1 | 1 | - |
| 2 | 5 | - |
| 3 | - | 5 |
| 4 | 5 | 5 |
| Sample | Ra [µm] | Std. Dev. | Rz [µm] | Std. Dev. |
|---|---|---|---|---|
| ref | 0.087 | 0.0064 | 0.489 | 0.0362 |
| 1 | 1.49 | 0.1941 | 8.48 | 0.5923 |
| 2 | 3.20 | 0.2175 | 17.06 | 1.3392 |
| 3 | 0.091 | 0.0073 | 0.461 | 0.0427 |
| 4 | 3.36 | 0.2966 | 17.38 | 1.5284 |
| Sample | SCA [°] | Std. Dev. | ACA [°] | Std. Dev. | RCA [°] | Std. Dev. | CAH [°] | SA [°] | Std. Dev. |
|---|---|---|---|---|---|---|---|---|---|
| ref | 84 | 1.48 | 86 | 0.75 | 47 | 0.83 | 39 | 87 | 1.21 |
| 1 | 94 | 0.67 | 98 | 0.93 | 63 | 1.21 | 35 | 67 | 1.35 |
| 2 | 104 | 1.34 | 107 | 0.61 | 76 | 0.43 | 31 | 62 | 0.98 |
| 3 | 105 | 0.71 | 108 | 1.03 | 79 | 1.09 | 29 | 39 | 1.52 |
| 4 | 114 | 1.23 | 118 | 0.77 | 93 | 0.84 | 25 | 35 | 0.87 |
| Sample | Accreted Ice [%] | Std. Dev. |
|---|---|---|
| Bare Al | 100 | - |
| ref | 85 | 1.34 |
| 1 | 88 | 0.68 |
| 2 | 92 | 1.21 |
| 3 | 83 | 1.55 |
| 4 | 77 | 1.62 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Przybyszewski, B.; Boczkowska, A.; Kozera, R.; Mora, J.; Garcia, P.; Aguero, A.; Borras, A. Hydrophobic and Icephobic Behaviour of Polyurethane-Based Nanocomposite Coatings. Coatings 2019, 9, 811. https://doi.org/10.3390/coatings9120811
Przybyszewski B, Boczkowska A, Kozera R, Mora J, Garcia P, Aguero A, Borras A. Hydrophobic and Icephobic Behaviour of Polyurethane-Based Nanocomposite Coatings. Coatings. 2019; 9(12):811. https://doi.org/10.3390/coatings9120811
Chicago/Turabian StylePrzybyszewski, Bartlomiej, Anna Boczkowska, Rafal Kozera, Julio Mora, Paloma Garcia, Alina Aguero, and Ana Borras. 2019. "Hydrophobic and Icephobic Behaviour of Polyurethane-Based Nanocomposite Coatings" Coatings 9, no. 12: 811. https://doi.org/10.3390/coatings9120811
APA StylePrzybyszewski, B., Boczkowska, A., Kozera, R., Mora, J., Garcia, P., Aguero, A., & Borras, A. (2019). Hydrophobic and Icephobic Behaviour of Polyurethane-Based Nanocomposite Coatings. Coatings, 9(12), 811. https://doi.org/10.3390/coatings9120811






