In Situ Plasma Monitoring of PECVD nc-Si:H Films and the Influence of Dilution Ratio on Structural Evolution
Abstract
1. Introduction
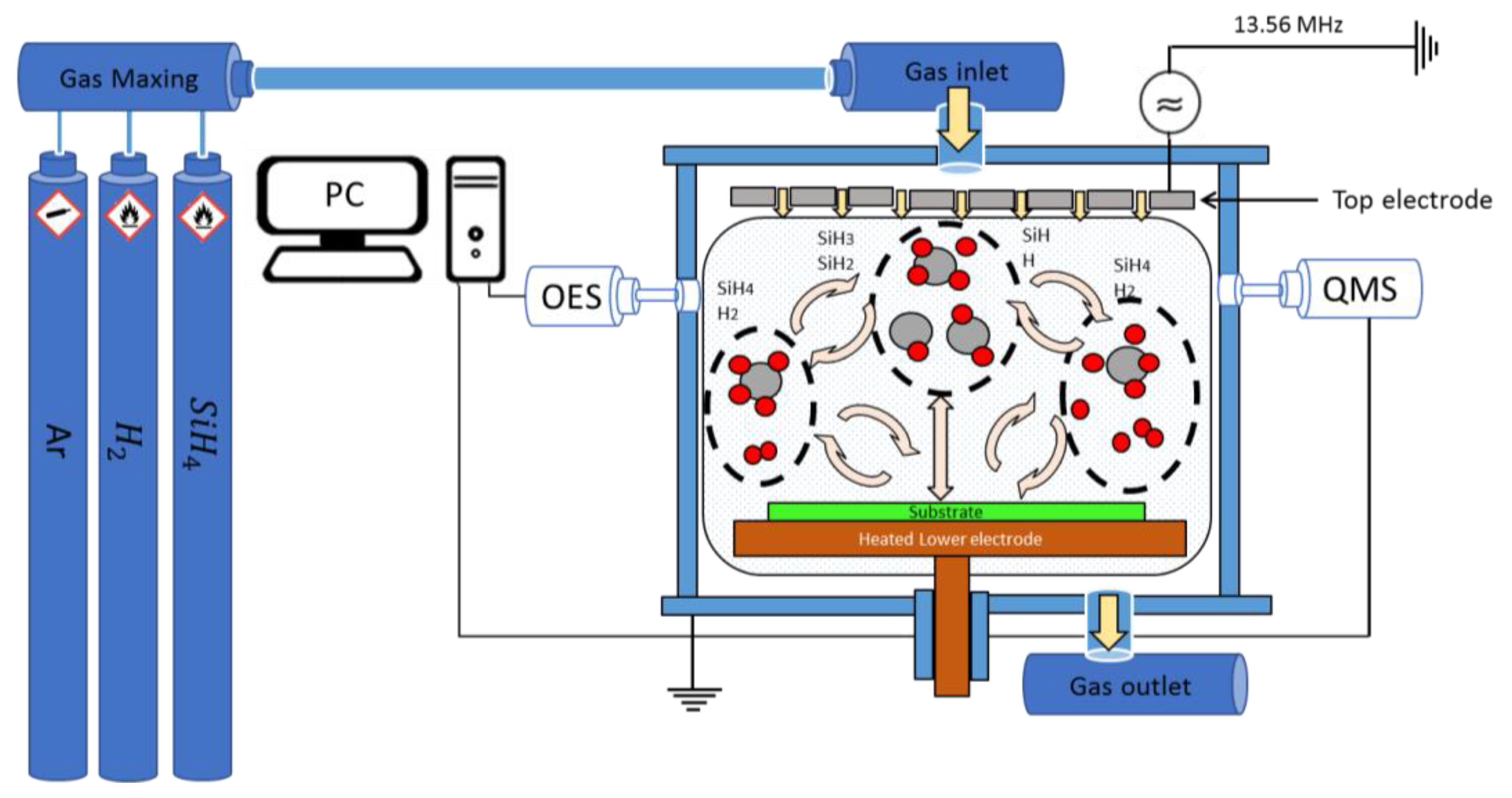
2. Experimental
3. Results and Discussion
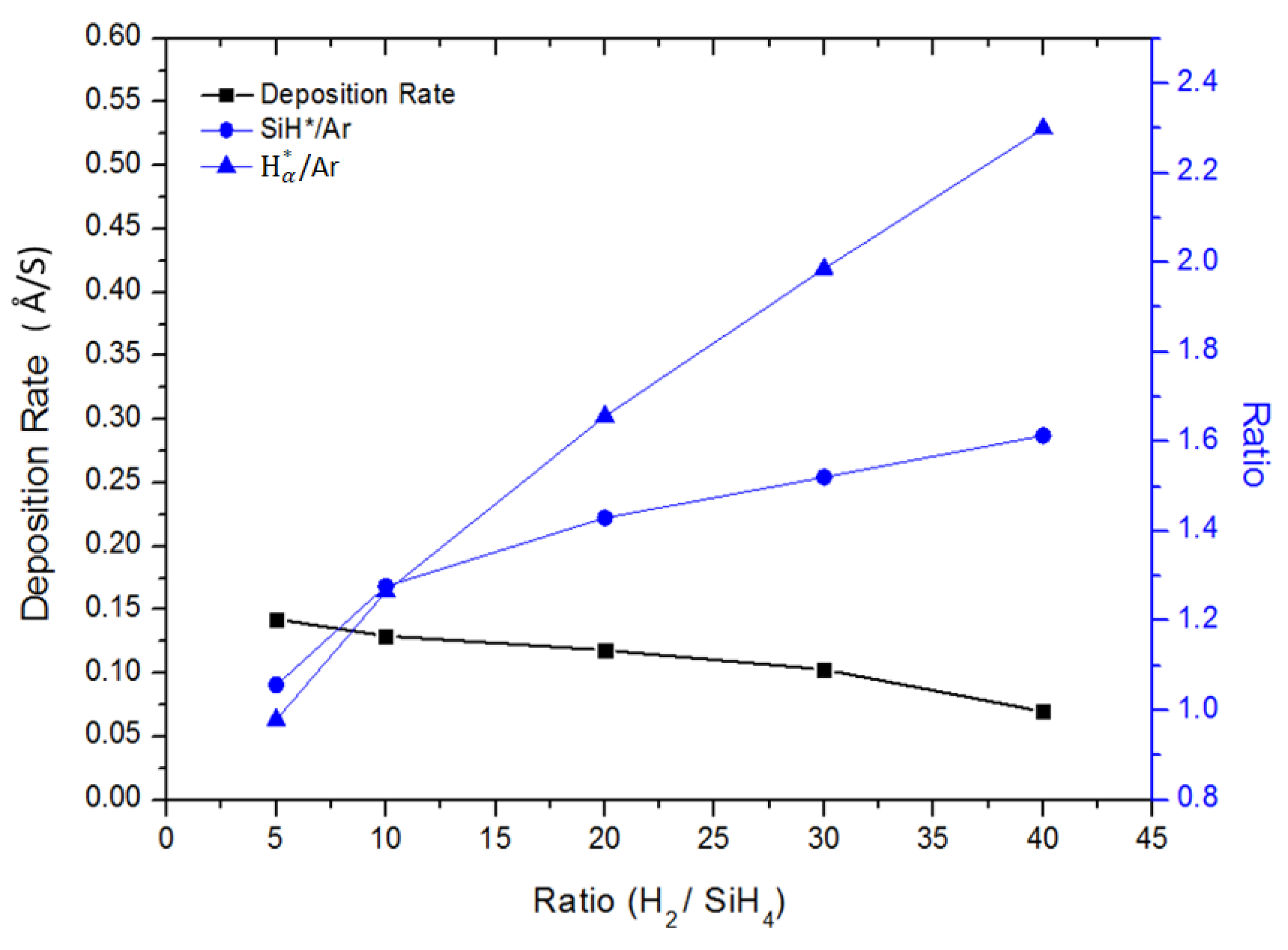
3.1. Effect of the Hydrogen Dilution Ratio on the Deposition Rate
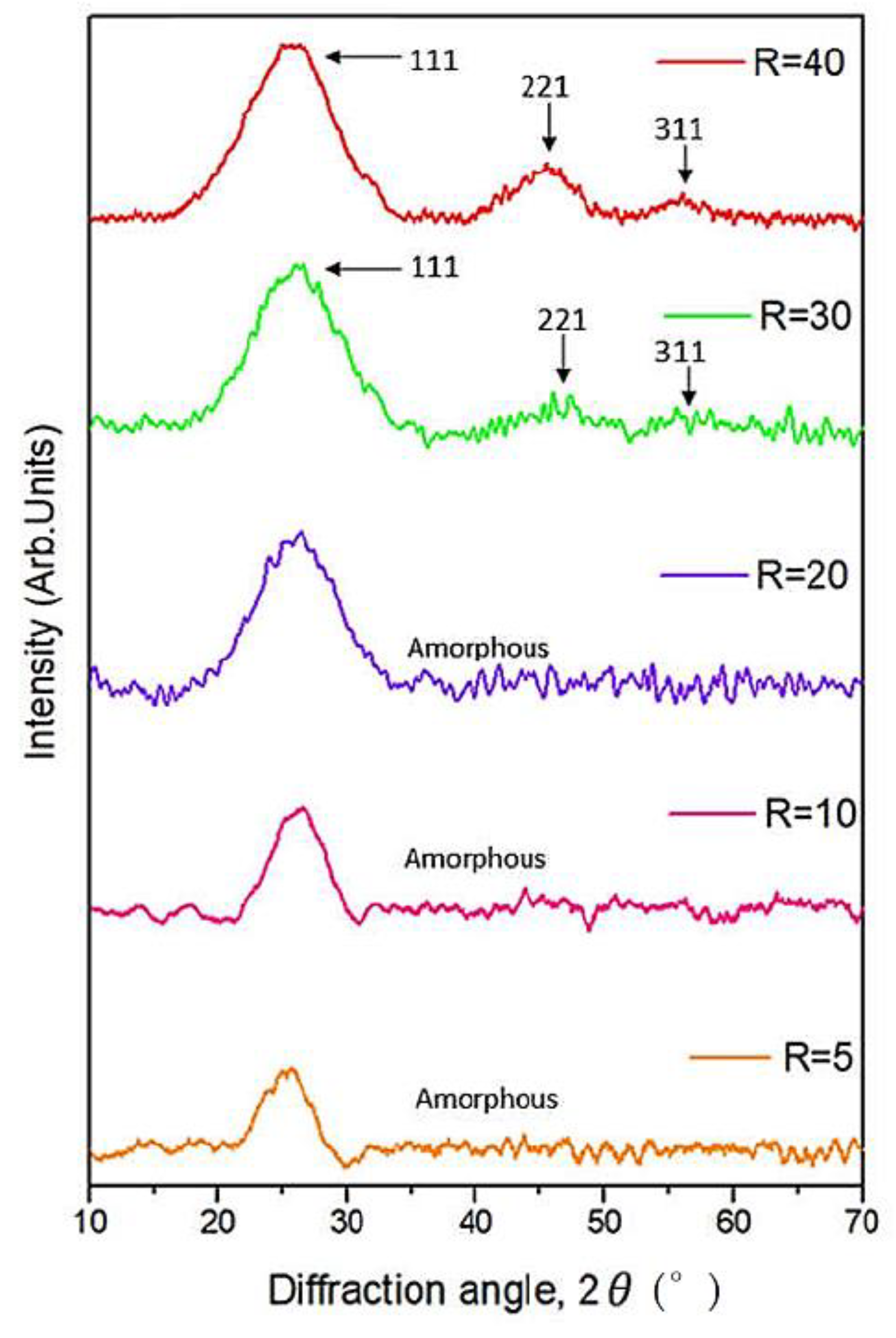
3.2. Low Angle XRD Analysis
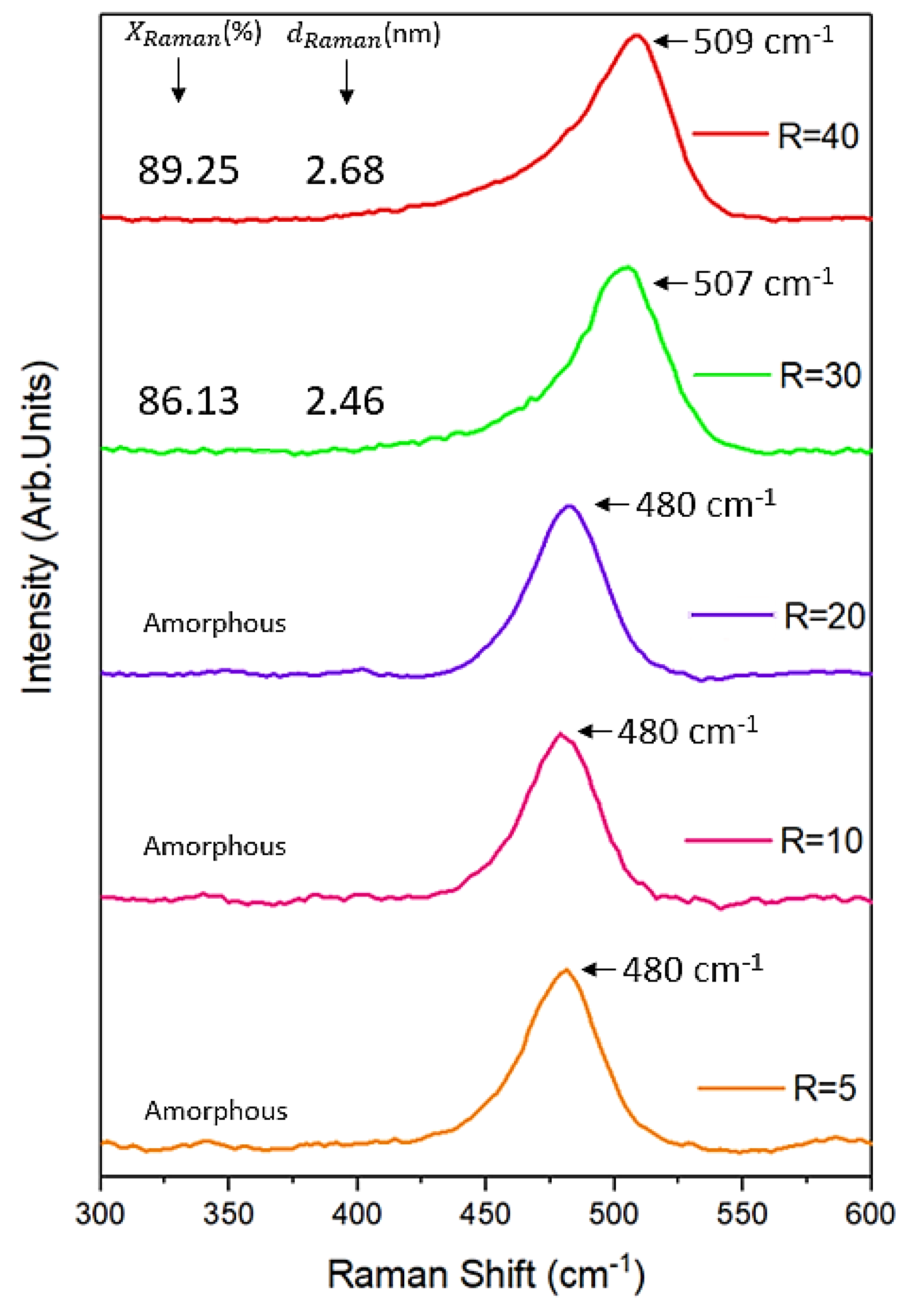
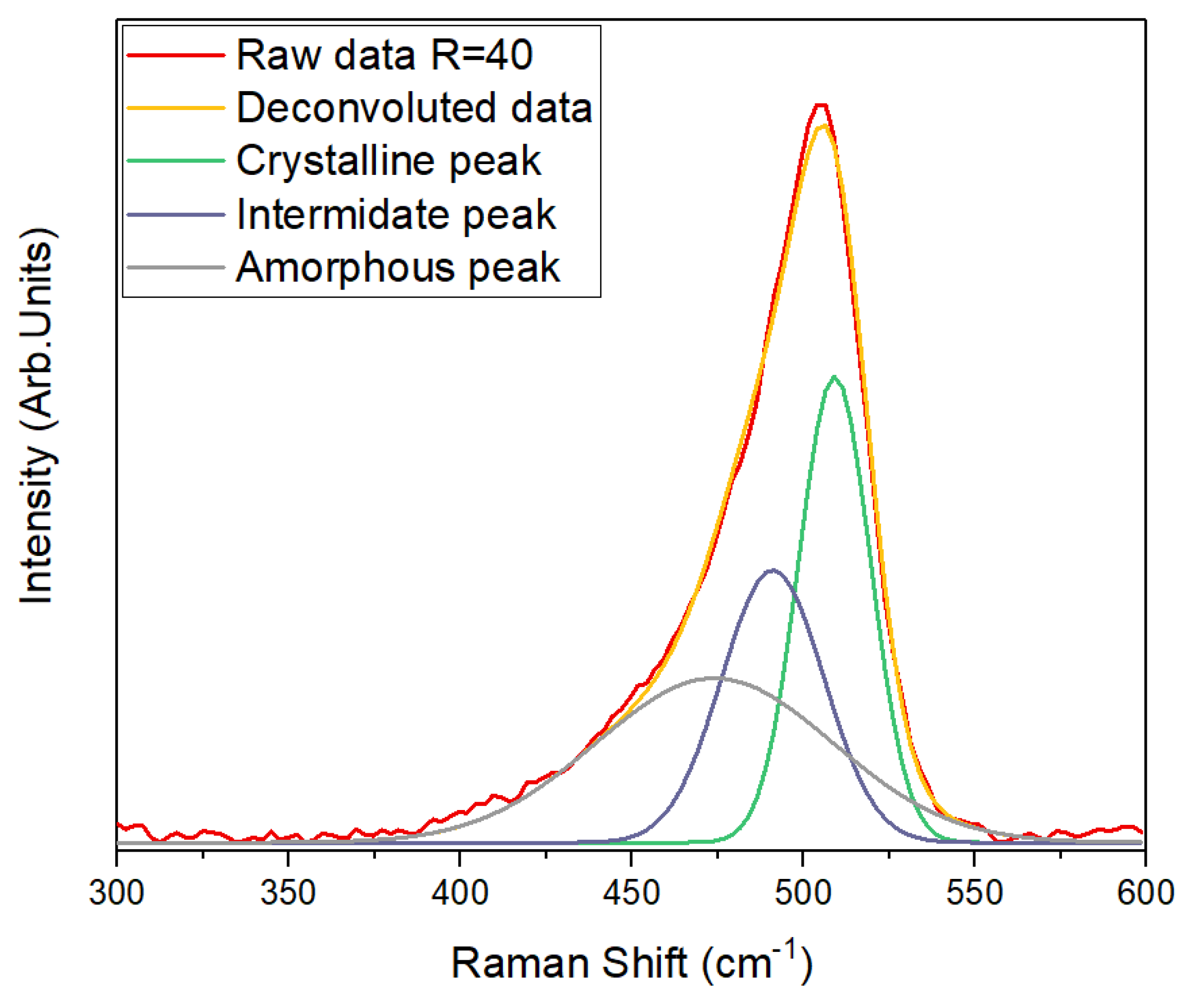
3.3. Raman Spectroscopy Analysis
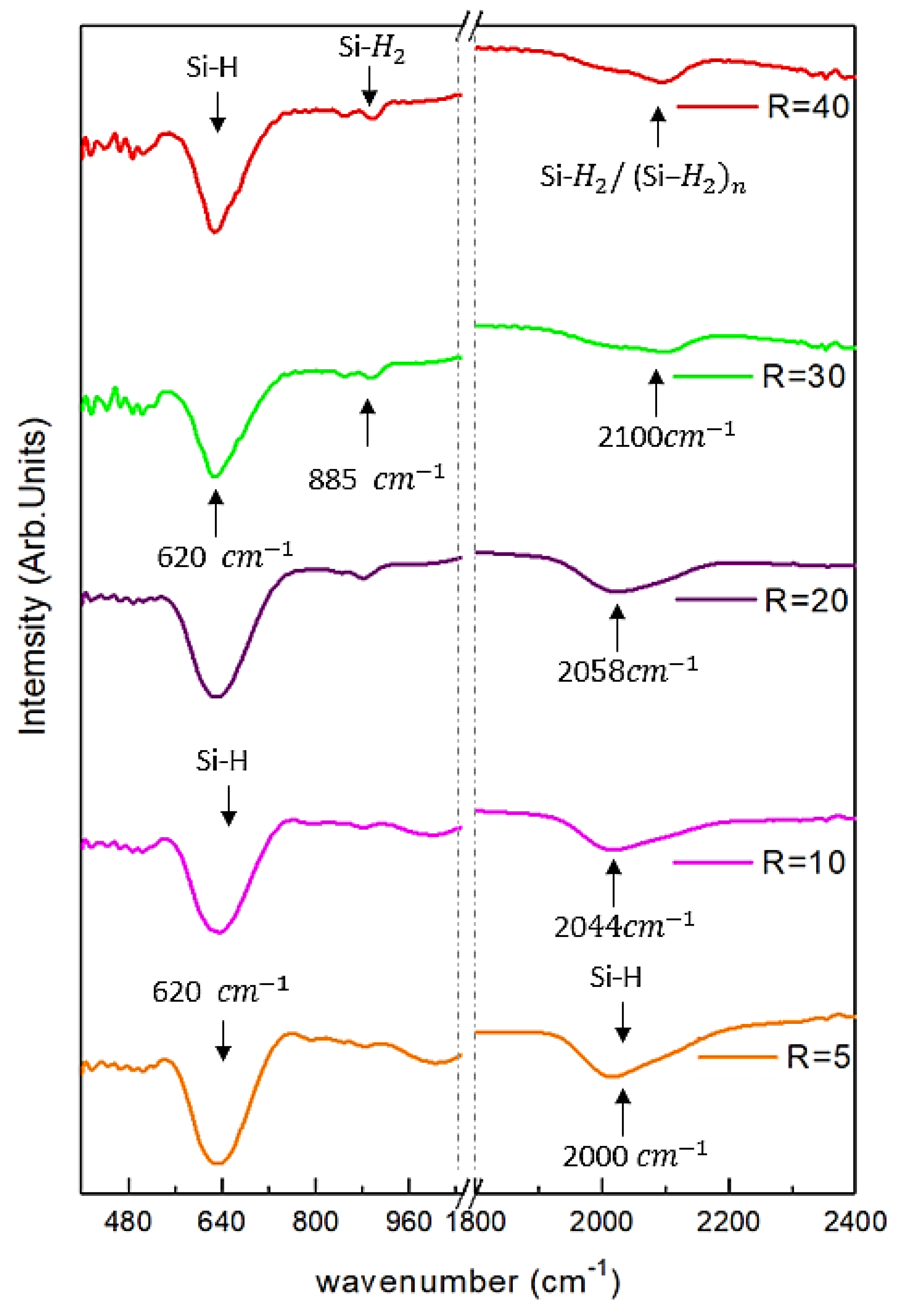
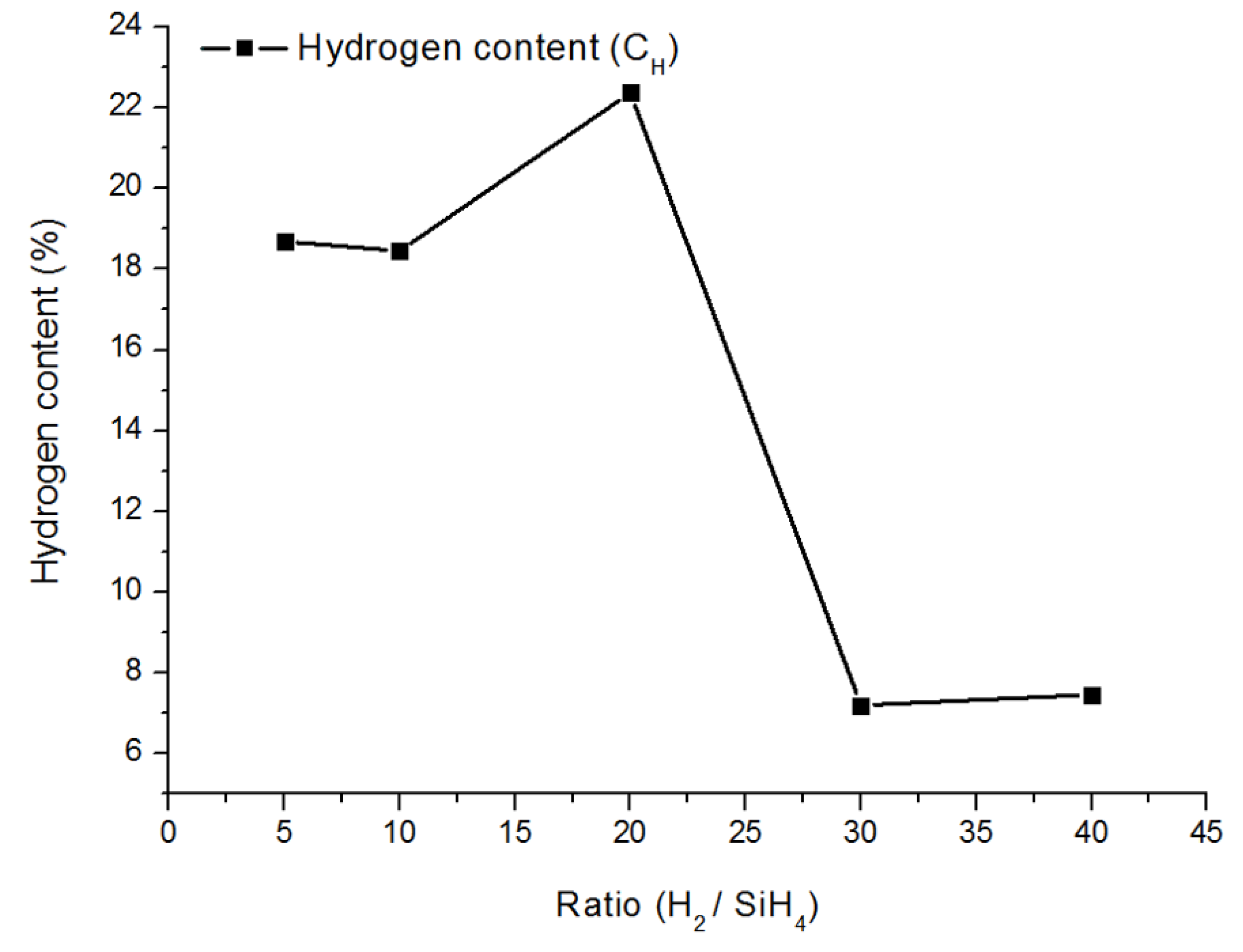
3.4. Fourier Transform Infra-Red (FTIR) Spectroscopy Analysis
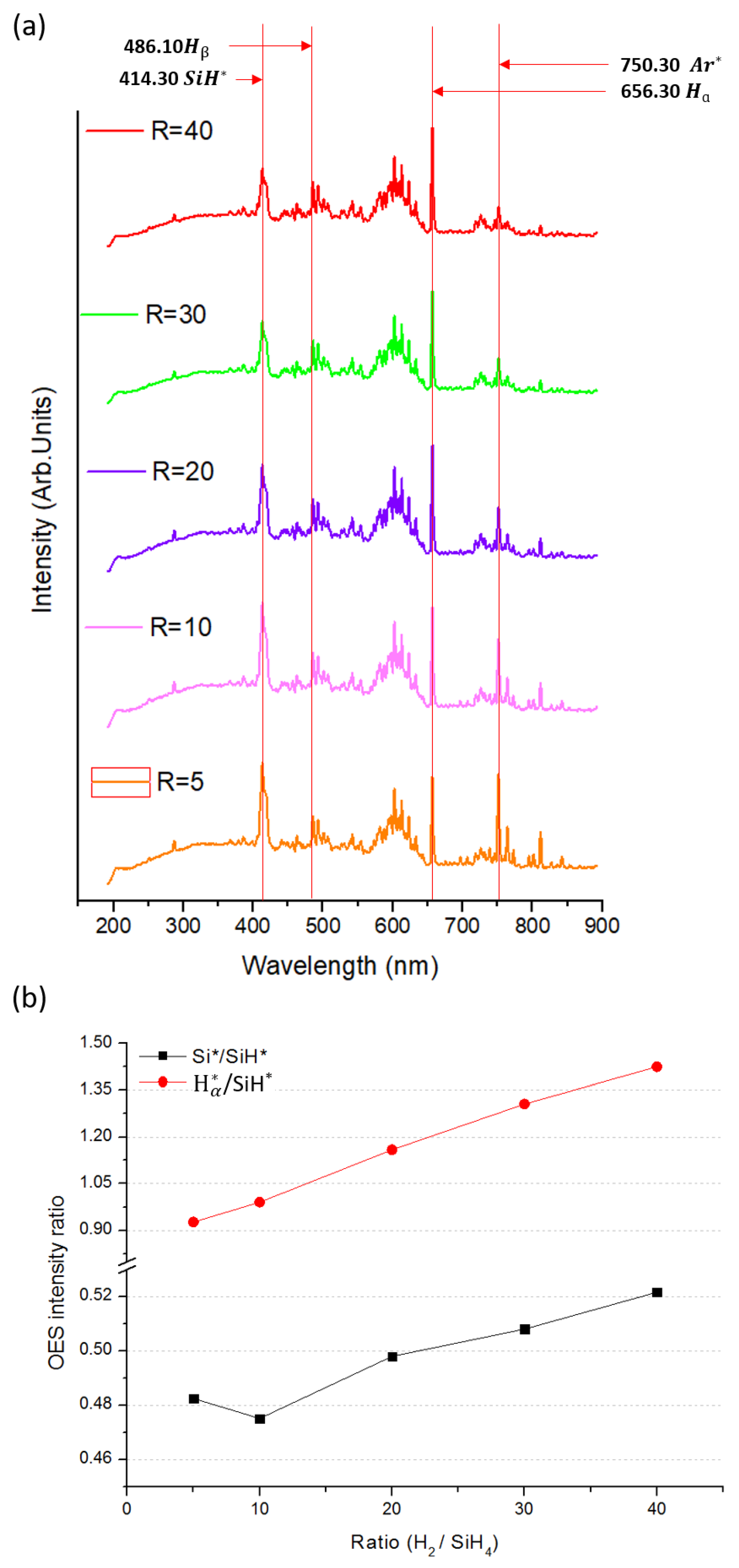
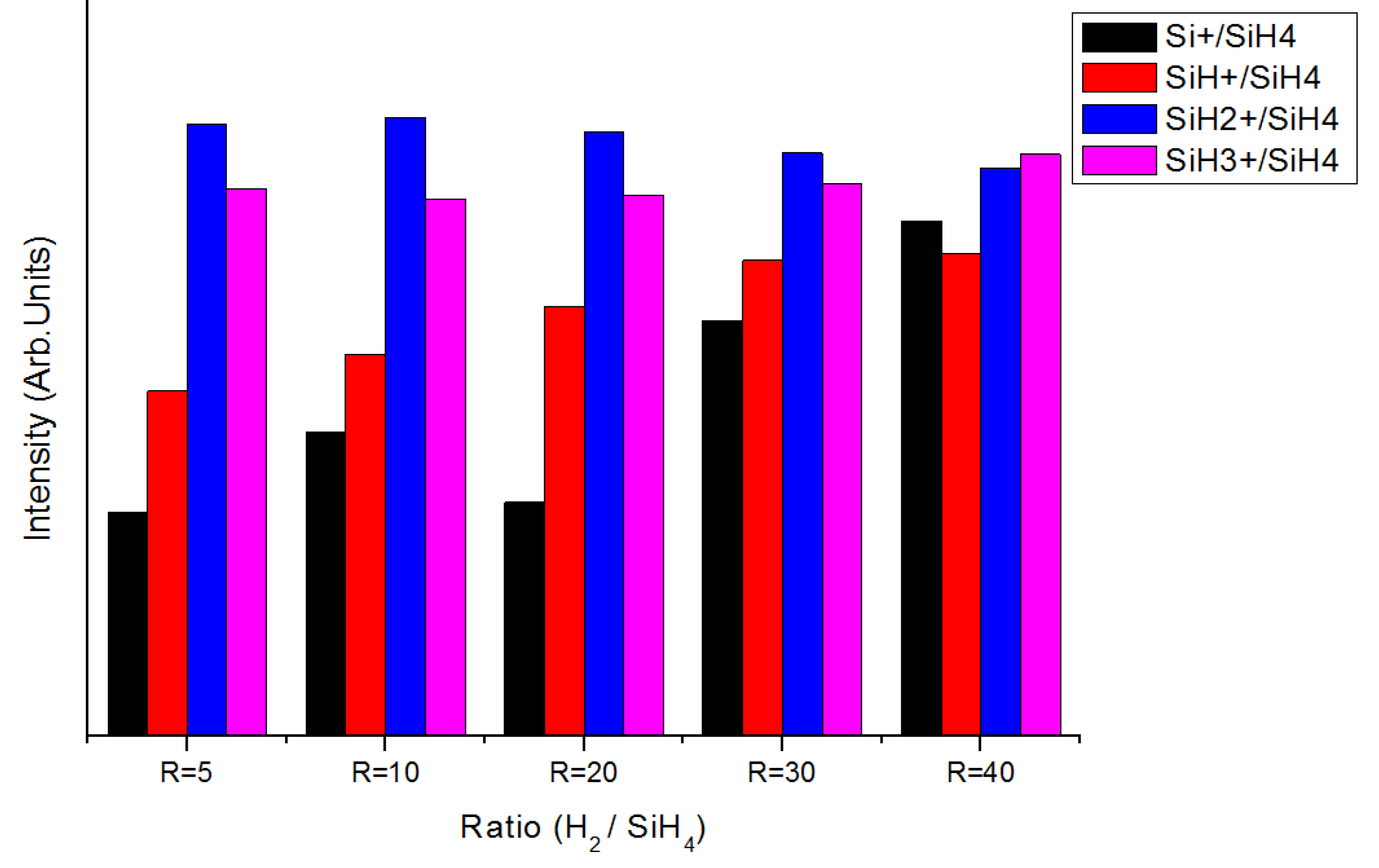
3.5. Optical Emission Spectra (OES) Spectroscopy Analysis
3.6. Quadruple Mass Spectra (QMS) Spectroscopy Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhou, H.P.; Wei, D.Y.; Xu, S.; Xiao, S.Q.; Xu, L.X.; Huang, S.Y.; Guo, Y.N.; Yan, W.S.; Xu, M. Crystalline silicon surface passivation by intrinsic silicon thin films deposited by low-frequency inductively coupled plasma. J. Appl. Phys. 2012, 112, 013708. [Google Scholar] [CrossRef]
- Tong, G.B.; Aspanut, Z.; Muhamad, M.R.; Rahman, S.A. Optical properties and crystallinity of hydrogenated nanocrystalline silicon (nc-Si: H) thin films deposited by RF-PECVD. Vacuum 2012, 86, 1195–1202. [Google Scholar] [CrossRef]
- Dutta, P.; Paul, S.; Galipeau, D.; Bommisetty, V. Effect of hydrogen plasma treatment on the surface morphology, microstructure and electronic transport properties of nc-Si:H. Thin Solid Films 2010, 518, 6811–6817. [Google Scholar] [CrossRef]
- Zhang, L.; Shen, H.L.; Jiang, X.F.; Qian, B.; Han, Z.D.; Hou, H.H. Influence of annealing temperature on the properties of polycrystalline silicon films formed by rapid thermal annealing of a-Si:H films. J. Mater. Sci. Mater. Electron. 2013, 24, 4209–4212. [Google Scholar] [CrossRef]
- Adikaari, A.A.D.T.; Mudugamuwa, N.K.; Silva, S.R.P. Nanocrystalline silicon solar cells from excimer laser crystallization of amorphous silicon. Sol. Energy Mater. Sol. Cells 2008, 92, 634–638. [Google Scholar] [CrossRef]
- Shim, J.H.; Im, S.; Kim, Y.J.; Cho, N.H. Nanostructural and optical features of hydrogenated nanocrystalline silicon films prepared by aluminium-induced crystallization. Thin Solid Films 2006, 503, 55–59. [Google Scholar] [CrossRef]
- Kaneko, T.; Wakagi, M.; Onisawa, K.; Minemura, T. Change in crystalline morphologies of polycrystalline silicon films prepared by radio-frequency plasma-enhanced chemical vapor deposition using SiF4 + H2 gas mixture at 350 °C. Appl. Phys. Lett. 1994, 64, 1865. [Google Scholar] [CrossRef]
- He, Y.; Yin, C.; Cheng, G.; Wang, L.; Liu, X.; Hu, G.Y. The structure and properties of nanosize crystalline silicon films. J. Appl. Phys. 1994, 75, 797. [Google Scholar] [CrossRef]
- Banerjee, A.; Liu, F.S.; Beglau, D.; Tining, S.; Pietka, G.; Yang, J.; Guha, S. 12.0% Efficiency on Large-Area, Encapsulated, Multijunction nc-Si:H-Based Solar Cells. IEEE J. Photovolt. 2012, 2, 104–108. [Google Scholar] [CrossRef]
- Bakr, N.A.; Funde, A.M.; Waman, V.S.; Kamble, M.M.; Hawaldar, R.R.; Amalnerkar, D.P.; Sathe, V.G.; Gosavi, S.W.; Jadkar, S.R. Influence of deposition pressure on structural, optical and electrical properties of nc-Si:H films deposited by HW-CVD. J. Phys. Chem. Solids 2011, 72, 685–691. [Google Scholar] [CrossRef]
- Goh, B.T.; Wah, C.K.; Aspanut, Z.; Rahman, S.A. Structural and optical properties of nc-Si:H thin films deposited by layer-by-layer technique. J. Mater. Sci. Mater. Electron. 2014, 25, 286–296. [Google Scholar] [CrossRef]
- He, H.; Ye, C.; Wang, X.; Huang, F.; Liu, Y. Effect of driving frequency on growth and structure of silicon films deposited by radio-frequency and very-High-frequency magnetron sputtering. ECS J. Solid State Sci. Technol. 2014, 3, Q74–Q78. [Google Scholar] [CrossRef]
- Lee, S.E.; Park, Y.C. Highly-conductive B-doped nc-Si:H thin films deposited at room temperature by using SLAN ECR-PECVD. J. Korean Phys. Soc. 2014, 65, 651–656. [Google Scholar] [CrossRef]
- Peng, S.; Wang, D.; Yang, F.; Wang, Z.; Ma, F. Grown Low-Temperature Microcrystalline Silicon Thin Film by VHF PECVD for Thin Films Solar Cell. J. Nanomater. 2015, 2015, 327596. [Google Scholar] [CrossRef]
- Brodsky, M.; Cardona, M.; Cuomo, J.J. Infrared and Raman spectra of the silicon-hydrogen bonds in amorphous silicon prepared by glow discharge and sputtering. Phys. Rev. B 1977, 16, 3556. [Google Scholar] [CrossRef]
- Tauc, J. Absorption edge and internal electric fields in amorphous semiconductors. Mater. Res. Bull. 1970, 5, 721–729. [Google Scholar] [CrossRef]
- Hsieh, Y.L.; Lee, C.C.; Lu, C.C.; Fuh, Y.K.; Chang, J.Y.; Lee, J.Y.; Li, T.T. Structural and electrical investigations of a-Si:H(i) and a-Si:H(n+) stacked layers for improving the interface and passivation qualities. J. Photonics Energy 2017, 7, 035503. [Google Scholar] [CrossRef]
- Dushaq, G.; Nayfeh, A.; Rasras, M. Tuning the optical properties of RF-PECVD grown μc-Si:H thin films using different hydrogen flow rate. Superlattice Microstruct. 2017, 107, 172–177. [Google Scholar] [CrossRef]
- Mataras, D.; Cavadias, S.; Rapakoulias, D. Spatial profiles of reactive intermediates in RF silane discharges. J. Appl. Phys. 1989, 66, 119. [Google Scholar] [CrossRef]
- Dingemans, G.; van den Donker, M.N.; Gordijn, A.; Kessels, W.M.M.; van de Sanden, M.C.M. Probing the phase composition of silicon films in situ by etch product detection. Appl. Phys. Lett. 2007, 91, 161902. [Google Scholar] [CrossRef]
- Fantz, U. Spectroscopic diagnostics and modelling of silane microwave plasmas. Plasma Phys. Control. Fusion 1998, 40, 1035. [Google Scholar] [CrossRef]
- Lisovskiy, V.; Booth, J.-P.; Landry, K.; Douai, D.; Cassagne, V.; Yegorenkov, V. RF discharge dissociative mode in NF3 and SiH4. Appl. Phys. D 2007, 40, 6631. [Google Scholar] [CrossRef]
- Nunomura, S.; Yoshida, I.; Kondo, M. Time-dependent gas phase kinetics in a hydrogen diluted silane plasma. Appl. Phys. Lett. 2009, 94, 071502. [Google Scholar] [CrossRef]
- Strahm, B.; Howling, A.A.; Sansonnens, L.; Hollenstein, C. Plasma silane concentration as a determining factor for the transition from amorphous to microcrystalline silicon in SiH4/H2 discharges. Plasma Sources Sci. Technol. 2007, 16, 80. [Google Scholar] [CrossRef]
- Howling, A.; Strahm, B.; Hollenstein, C. Non-intrusive plasma diagnostics for the deposition of large area thin film silicon. Thin Solid Films 2009, 517, 6218–6224. [Google Scholar] [CrossRef]
- Rath, J.K. Low temperature polycrystalline silicon: A review on deposition, physical properties and solar cell applications. Sol. Energy Mater. Sol. Cells 2003, 76, 431–487. [Google Scholar] [CrossRef]
- Jellison, J.G.E.; Modine, F.A. Parameterization of the optical functions of amorphous materials in the interband region. Appl. Phys. Lett. 1996, 69, 371. [Google Scholar] [CrossRef]
- Lin, L.J.H.; Chiou, Y.-P. Improving thin-film crystalline silicon solar cell efficiency with back surface field layer and blaze diffractive grating. Sol. Energy 2012, 86, 1485–1490. [Google Scholar] [CrossRef]
- Matsuda, A. Growth mechanism of microcrystalline silicon obtained from reactive plasmas. Thin Solid Films 1999, 337, 1–6. [Google Scholar] [CrossRef]
- Jadhavar, A.; Pawbake, A.; Waykar, R.; Waman, V.I.; Rondiya, S.; Shinde, O.; Kulkarni, R.; Rokade, A.; Bhorde, A.; Funde, A.; et al. Influence of RF power on structural optical and electrical properties of hydrogenated nano-crystalline silicon (nc-Si:H) thin films deposited by PE-CVD. J. Mater. Sci. Mater. Electron. 2016, 27, 12365–12373. [Google Scholar] [CrossRef]
- Marquardt, D. An Algorithm for Least-Squares Estimation of Nonlinear Parameters. J. Soc. Ind. Appl. Math. 1963, 11, 431–441. [Google Scholar] [CrossRef]
- Li, Z.; Li, W.; Jiang, Y.; Cai, H.; Gong, Y.; He, J. Raman characterization of the structural evolution in amorphous and partially nanocrystalline hydrogenated silicon thin films prepared by PECVD. J. Raman Spectrosc. 2011, 42, 415–421. [Google Scholar] [CrossRef]
- Tong, G.B.; Muhamad, M.R.; Rahman, S.A. Effects of RF Power on structural properties of nc-Si:H thin films deposited by layer-by-layer (LbL) deposition Technique. Sains Malays. 2012, 41, 993–1000. [Google Scholar]
- Droz, C.; Vallat-Sauvain, E.; Bailat, J.; Feitknecht, L.; Meier, J.; Shah, A. Relationship between Raman crystallinity and open-circuit voltage in microcrystalline silicon solar cells. Sol. Energy Mater. Sol. Cells 2004, 81, 61–71. [Google Scholar] [CrossRef]
- Lucovsky, G. Vibrational spectroscopy of hydrogenated amorphous silicon alloys. Sol. Cells 1980, 2, 431–442. [Google Scholar] [CrossRef]
- Halindintwali, S.; Knoesen, D.; Swanepoel, R.; Julies, B.; Arendse, C.; Muller, T.; Theron, C.; Gordijn, A.; Bronsveld, P.; Rath, J.K.; et al. Improved stability of intrinsic nanocrystalline Si thin films deposited by hot-wire chemical vapour deposition technique. Thin Solid Films 2007, 515, 8040–8044. [Google Scholar] [CrossRef]
- Knights, J.C.; Lucovsky, G.; Nemanich, R.J. Defects in plasma-deposited a-Si:H. J. Non-Cryst. Solids 1979, 32, 393–403. [Google Scholar] [CrossRef]
- Tsu, D.; Lucovsky, G.; Dadison, B. Effects of the nearest neighbors and the alloy matrix on SiH stretching vibrations in the amorphous SiOr:H (0 < r < 2) alloy system. Phys. Rev. B 1989, 40, 1795–1805. [Google Scholar]
- Jones, A.; Ahmed, W.; Hassan, I.; Rego, C.; Sein, H.; Amar, M.; Jackson, M. The impact of inert gases on the structure, properties and growth of nanocrystalline diamond. J. Phys. 2003, 15, S2969. [Google Scholar] [CrossRef]
- Chen, C.Z.; Qiu, S.H.; Liu, C.Q.; Dan, W.Y.; Li, P.; Ying, Y.C.; Lin, X. Low temperature fast growth of nanocrystalline silicon films by RF-PECVD from SiH4/H2 gases: Microstructural characterization. J. Phys. D Appl. Phys. 2008, 41, 195413. [Google Scholar] [CrossRef]
- Lin, K.X.; Lin, X.Y.; Yu, Y.P.; Wang, H.; Chen, J.Y. Measurements in silane radio frequency glow discharges using a tuned and heated Langmuir probe. J. Appl. Phys. 1993, 74, 4899. [Google Scholar] [CrossRef]
- Kondo, M.; Fukawa, M.; Guo, L.; Matsuda, A. High rate growth of microcrystalline silicon at low temperatures. J. Non-Cryst. Solids 2000, 266–269, 84–89. [Google Scholar] [CrossRef]
- Das, C.; Ray, S. Power density in RF-PECVD: A factor for deposition of amorphous silicon thin films and successive solid phase crystallization. J. Phys. D Appl. Phys. 2002, 35, 2211. [Google Scholar] [CrossRef]
- Kirner, S.; Gabriel, O.; Stannowski, B.; Rech, B.; Schlatmann, R. The growth of microcrystalline silicon oxide thin films studied by in situ plasma diagnostics. Appl. Phys. Lett. 2013, 102, 051906. [Google Scholar] [CrossRef]
- Strahm, B.; Howling, A.A.; Sansonnens, L.; Hollenstein, C. Optimization of the microcrystalline silicon deposition efficiency. J. Vac. Sci. Technol. A 2007, 25, 1198. [Google Scholar] [CrossRef]
| Source Gases | Pressure | Power | Gas Flow Ratio | Deposition Time | Distance | Substrate Temperature | ||
|---|---|---|---|---|---|---|---|---|
| (1) Ar | (2) SiH4 | (3) H2 | ||||||
| Ar, SiH4, H2 | 300 mTorr | 100 W | 3 sccm | 5 sccm | 25, 50, 100, 150, 200 sccm | 60 min | 25 mm | 210 °C |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsieh, Y.-L.; Kau, L.-H.; Huang, H.-J.; Lee, C.-C.; Fuh, Y.-K.; Li, T.T. In Situ Plasma Monitoring of PECVD nc-Si:H Films and the Influence of Dilution Ratio on Structural Evolution. Coatings 2018, 8, 238. https://doi.org/10.3390/coatings8070238
Hsieh Y-L, Kau L-H, Huang H-J, Lee C-C, Fuh Y-K, Li TT. In Situ Plasma Monitoring of PECVD nc-Si:H Films and the Influence of Dilution Ratio on Structural Evolution. Coatings. 2018; 8(7):238. https://doi.org/10.3390/coatings8070238
Chicago/Turabian StyleHsieh, Yu-Lin, Li-Han Kau, Hung-Jui Huang, Chien-Chieh Lee, Yiin-Kuen Fuh, and Tomi T. Li. 2018. "In Situ Plasma Monitoring of PECVD nc-Si:H Films and the Influence of Dilution Ratio on Structural Evolution" Coatings 8, no. 7: 238. https://doi.org/10.3390/coatings8070238
APA StyleHsieh, Y.-L., Kau, L.-H., Huang, H.-J., Lee, C.-C., Fuh, Y.-K., & Li, T. T. (2018). In Situ Plasma Monitoring of PECVD nc-Si:H Films and the Influence of Dilution Ratio on Structural Evolution. Coatings, 8(7), 238. https://doi.org/10.3390/coatings8070238





