Transparent, Adherent, and Photocatalytic SiO2-TiO2 Coatings on Polycarbonate for Self-Cleaning Applications
Abstract
:1. Introduction
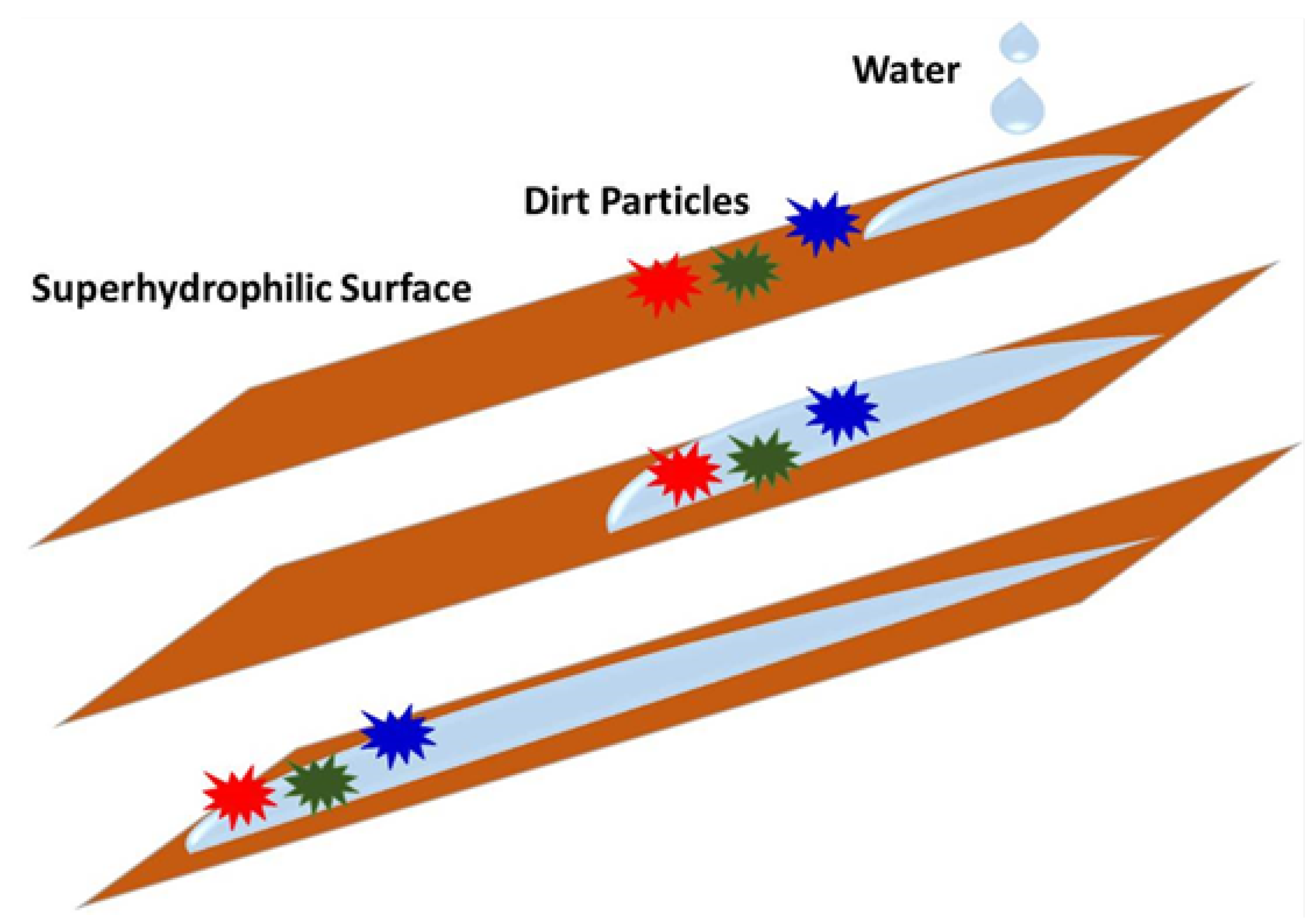
2. Results and Discussion
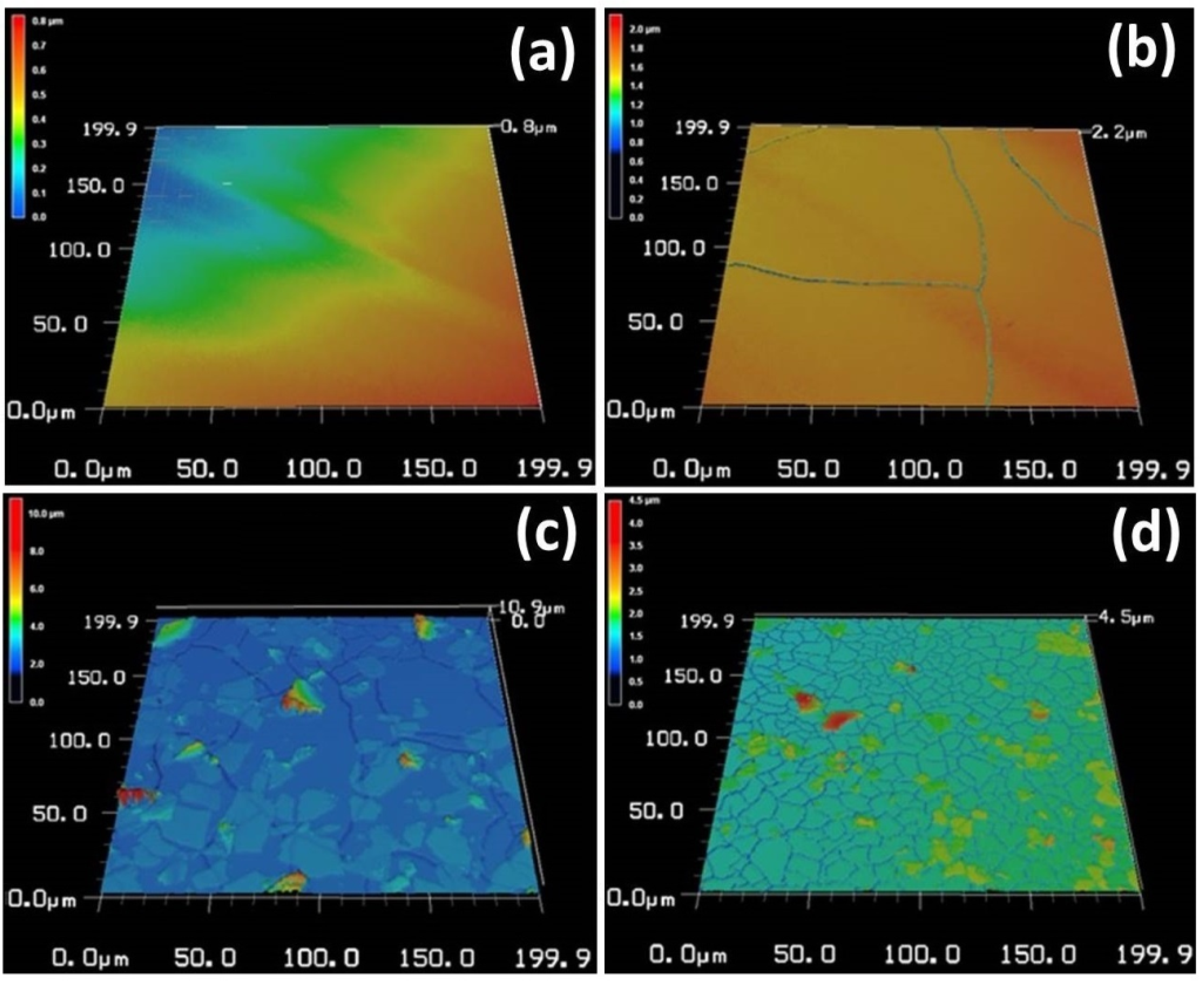
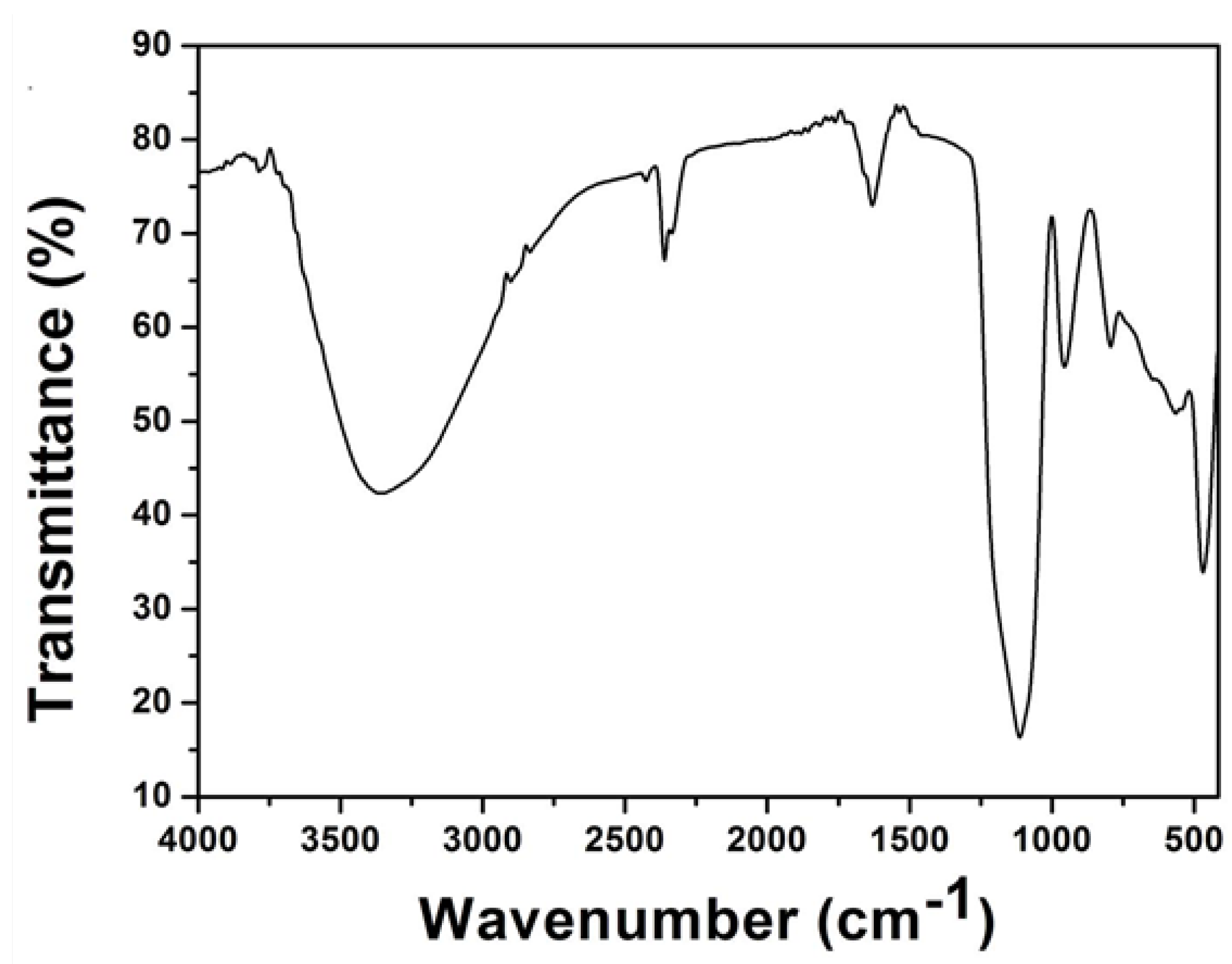
2.1. Surface Microstructure, Roughness and Chemical Composition of the Coatings

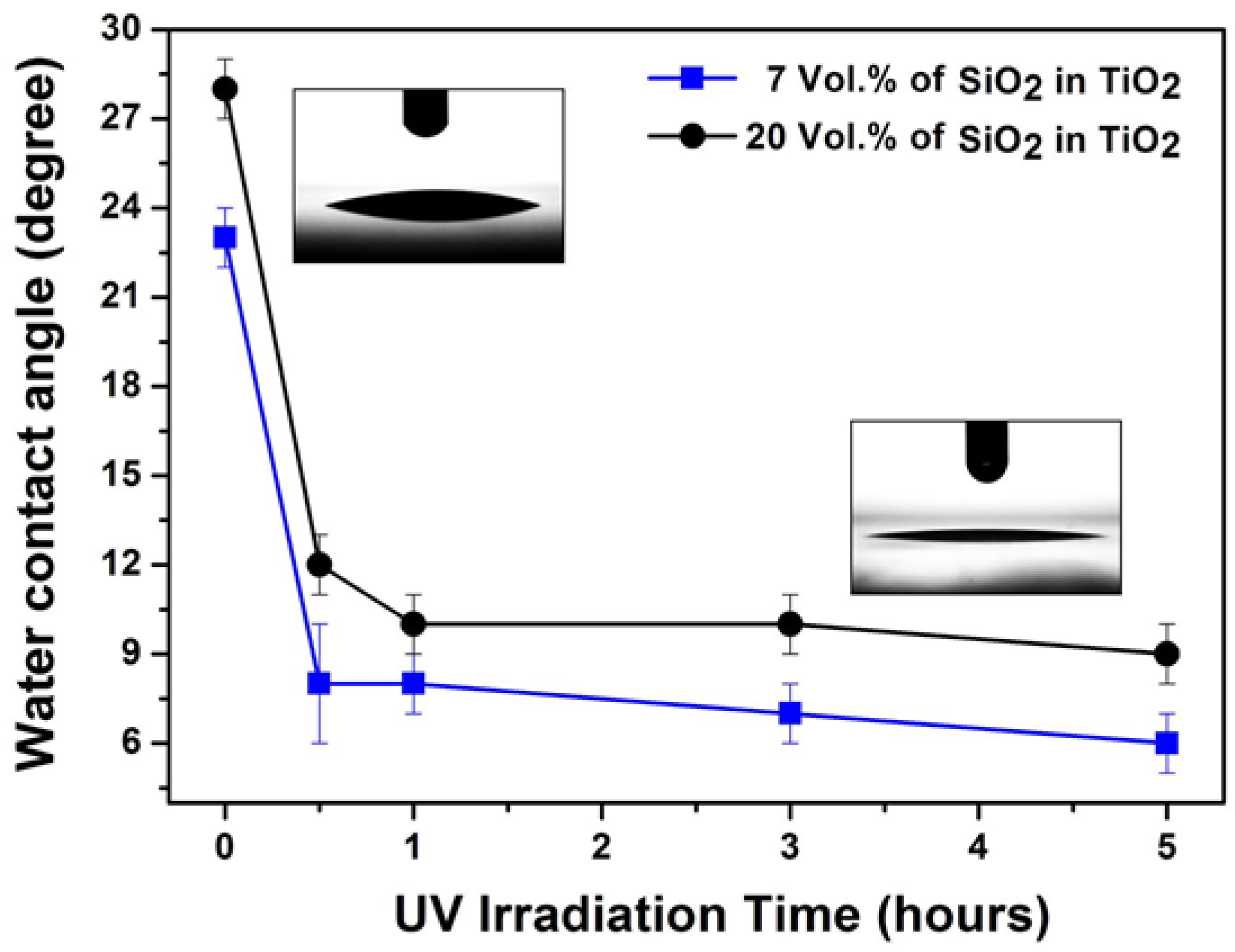
2.2. Superhydrophilic, Photocatalytic and Optical Properties of the Coatings

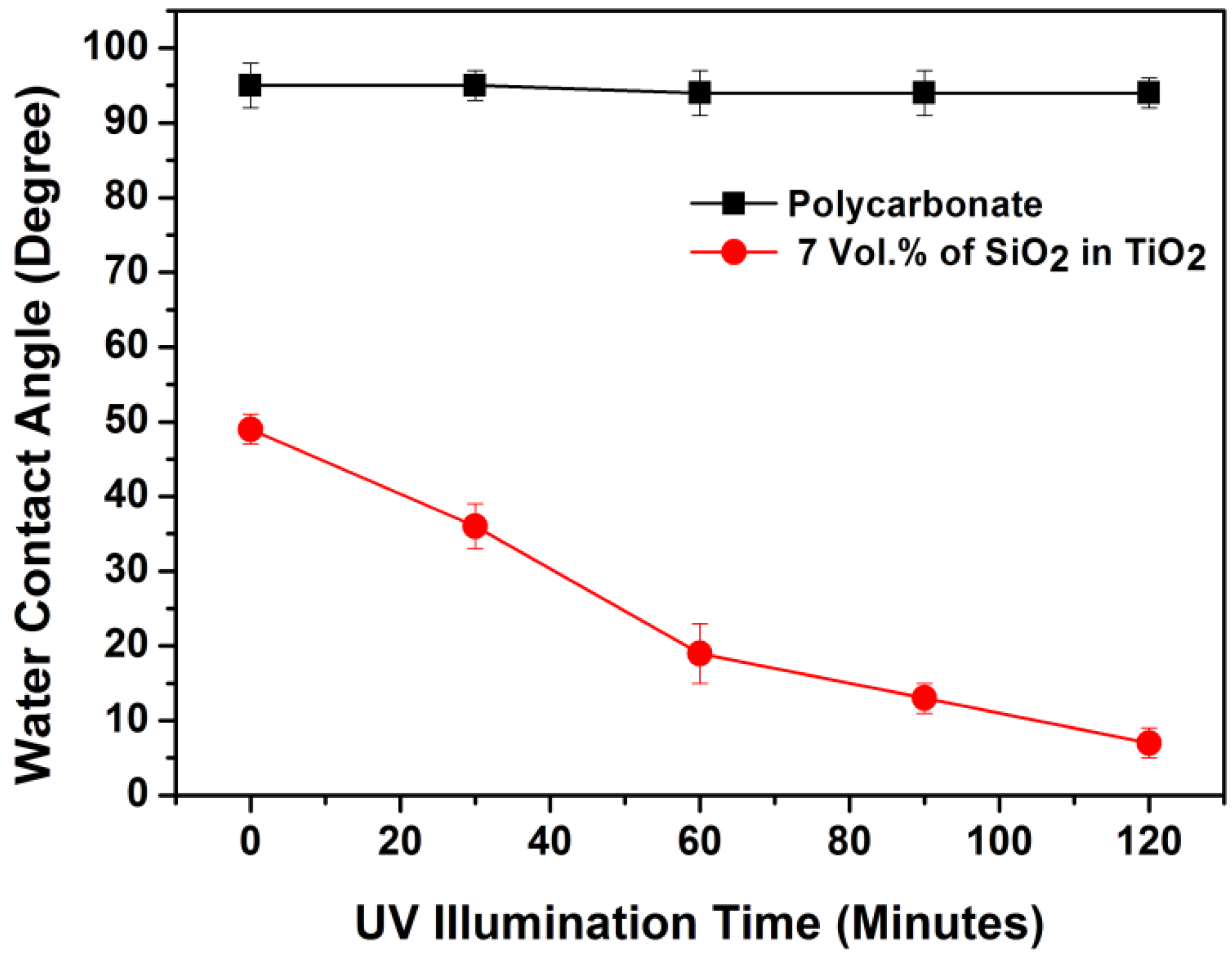
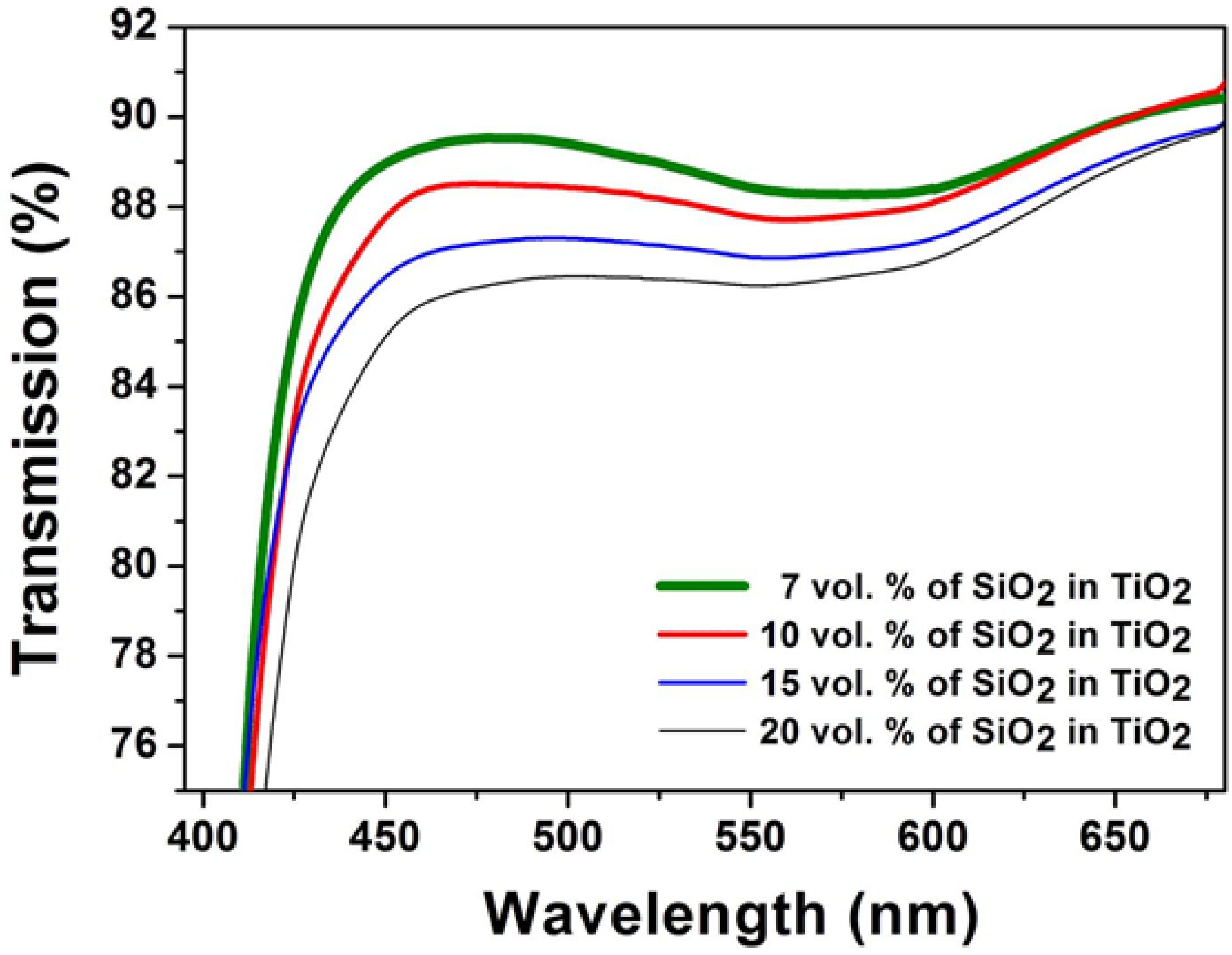
2.3. Mechanical Properties of the Coatings

3. Experimental Section
3.1. Materials
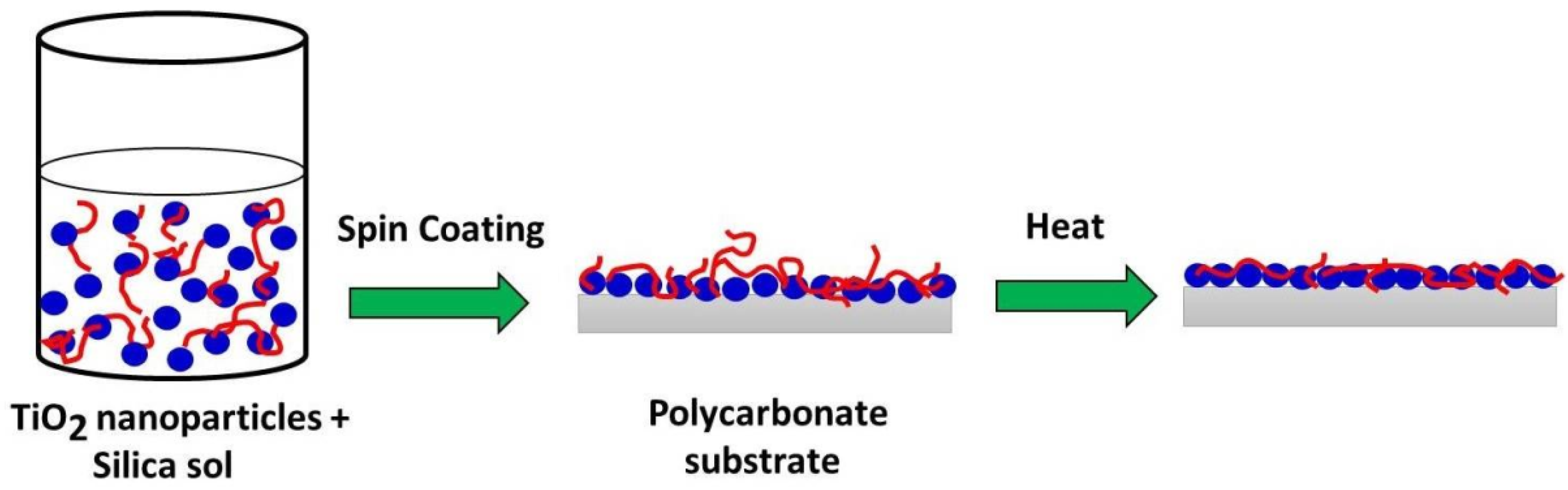
3.2. Self-Cleaning Coating on PC Substrates
3.3. Characterization Techniques
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sakai, N.; Fukuda, K.; Shibata, T.; Ebina, Y.; Takada, K.; Sasaki, T. Photoinduced hydrophilic conversion properties of titania nanosheets. J. Phys. Chem. B 2006, 110, 6198–6203. [Google Scholar] [CrossRef] [PubMed]
- Blossey, R. Self-cleaning surfaces—virtual realities. Nat. Mater. 2003, 2, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Weng, K.-W.; Huang, Y.-P. Preparation of TiO2 thin films on glass surfaces with self-cleaning characteristics for solar concentrators. Surf. Coat. Technol. 2013, 231, 201–204. [Google Scholar] [CrossRef]
- Xi, B.; Verma, L.K.; Li, J.; Bhatia, C.S.; Danner, A.J.; Yang, H.; Zeng, H.C. TiO2 thin films prepared via adsorptive self-assembly for self-cleaning applications. ACS Appl. Mater. Interfaces 2012, 4, 1093–1102. [Google Scholar] [CrossRef] [PubMed]
- Euvananont, C.; Junin, C.; Inpor, K.; Limthongkul, P.; Thanachayanont, C. TiO2 optical coating layers for self-cleaning applications. Ceram. Int. 2008, 34, 1067–1071. [Google Scholar] [CrossRef]
- Lai, Y.; Tang, Y.; Gong, J.; Gong, D.; Chi, L.; Lin, C.; Chen, Z. Transparent superhydrophobic/superhydrophilic TiO2-based coatings for self-cleaning and anti-fogging. J. Mater. Chem. 2012, 22, 7420–7426. [Google Scholar] [CrossRef]
- Fateh, R.; Dillert, R.; Bahnemann, D. Preparation and characterization of transparent hydrophilic photocatalytic TiO2/SiO2 thin films on polycarbonate. Langmuir 2013, 29, 3730–3739. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.K.; Moon, J.H.; Shul, Y.G.; Jung, K.T.; Kim, D.H.; Lee, D.W. Scratch resistant and transparent UV-protective coating on polycarbonate. J. Sol Gel Sci. Technol. 2003, 26, 783–787. [Google Scholar] [CrossRef]
- Lam, S.; Soetanto, A.; Amal, R. Self-cleaning performance of polycarbonate surfaces coated with titania nanoparticles. J. Nanopart. Res. 2009, 11, 1971–1979. [Google Scholar] [CrossRef]
- Fateh, R.; Ismail, A.A.; Dillert, R.; Bahnemann, D.W. Highly active crystalline mesoporous TiO2 films coated onto polycarbonate substrates for self-cleaning applications. J. Phys. Chem. C 2011, 115, 10405–10411. [Google Scholar] [CrossRef]
- Fateh, R.; Dillert, R.; Bahnemann, D. Self-cleaning properties, mechanical stability, and adhesion strength of transparent photocatalytic TiO2-ZnO coatings on polycarbonate. ACS Appl. Mater. Interfaces 2014, 6, 2270–2278. [Google Scholar] [CrossRef] [PubMed]
- Hatton, B.; Mishchenko, L.; Davis, S.; Sandhage, K.H.; Aizenberg, J. Assembly of large-area, highly ordered, crack-free inverse opal films. Proc. Natl. Acad. Sci. USA 2010, 107, 10354–10359. [Google Scholar] [CrossRef] [PubMed]
- Khanna, P.K.; Singh, N.; Charan, S. Synthesis of nano-particles of anatase-TiO2 and preparation of its optically transparent film in PVA. Mater. Lett. 2007, 61, 4725–4730. [Google Scholar] [CrossRef]
- Kumar, D.A.; Shyla, J.M.; Xavier, F.P. Synthesis and characterization of TiO2/SiO2 nano composites for solar cell applications. Appl. Nanosci. 2012, 2, 429–436. [Google Scholar] [CrossRef]
- Rao, A.V.; Latthe, S.S.; Nadargi, D.Y.; Hirashima, H.; Ganesan, V. Preparation of MTMS based transparent superhydrophobic silica films by sol–gel method. J. Colloid Interface Sci. 2009, 332, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Shultz, A.N.; Jang, W.; Hetherington, W.M., III; Baer, D.R.; Wang, L.-Q.; Engelhard, M.H. Comparative second harmonic generation and X-ray photoelectron spectroscopy studies of the UV creation and O2 healing of Ti3+ defects on (110) rutile TiO2 surfaces. Surf. Sci. 1995, 339, 114–124. [Google Scholar]
- Wang, R.; Hashimoto, K.; Fujishima, A.; Chikuni, M.; Kojima, E.; Kitamura, A.; Shimohigoshi, M.; Watanabe, T. Light-induced amphiphilic surfaces. Nature 1997, 388, 431–432. [Google Scholar] [CrossRef]
- Anandan, S.; Rao, T. N.; Sathish, M.; Rangappa, D.; Honma, I.; Miyauchi, M. Superhydrophilic graphene-loaded TiO2 thin film for self-cleaning applications. ACS Appl. Mater. Interfaces 2012, 5, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Aslan, K.; Holley, P.; Geddes, C.D. Metal-enhanced fluorescence from silver nanoparticle-deposited polycarbonate substrates. J. Mater. Chem. 2006, 16, 2846–2852. [Google Scholar] [CrossRef]
- Rivaton, A. Recent advances in bisphenol—A polycarbonate photodegradation. Polym. Degrad. Stab. 1995, 49, 163–179. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Latthe, S.S.; Liu, S.; Terashima, C.; Nakata, K.; Fujishima, A. Transparent, Adherent, and Photocatalytic SiO2-TiO2 Coatings on Polycarbonate for Self-Cleaning Applications. Coatings 2014, 4, 497-507. https://doi.org/10.3390/coatings4030497
Latthe SS, Liu S, Terashima C, Nakata K, Fujishima A. Transparent, Adherent, and Photocatalytic SiO2-TiO2 Coatings on Polycarbonate for Self-Cleaning Applications. Coatings. 2014; 4(3):497-507. https://doi.org/10.3390/coatings4030497
Chicago/Turabian StyleLatthe, Sanjay S., Shanhu Liu, Chiaki Terashima, Kazuya Nakata, and Akira Fujishima. 2014. "Transparent, Adherent, and Photocatalytic SiO2-TiO2 Coatings on Polycarbonate for Self-Cleaning Applications" Coatings 4, no. 3: 497-507. https://doi.org/10.3390/coatings4030497
APA StyleLatthe, S. S., Liu, S., Terashima, C., Nakata, K., & Fujishima, A. (2014). Transparent, Adherent, and Photocatalytic SiO2-TiO2 Coatings on Polycarbonate for Self-Cleaning Applications. Coatings, 4(3), 497-507. https://doi.org/10.3390/coatings4030497





