Surface Modification of the Ti-6Al-4V Alloy by Anodic Oxidation and Its Effect on Osteoarticular Cell Proliferation
Abstract
1. Introduction
2. Materials and Methods
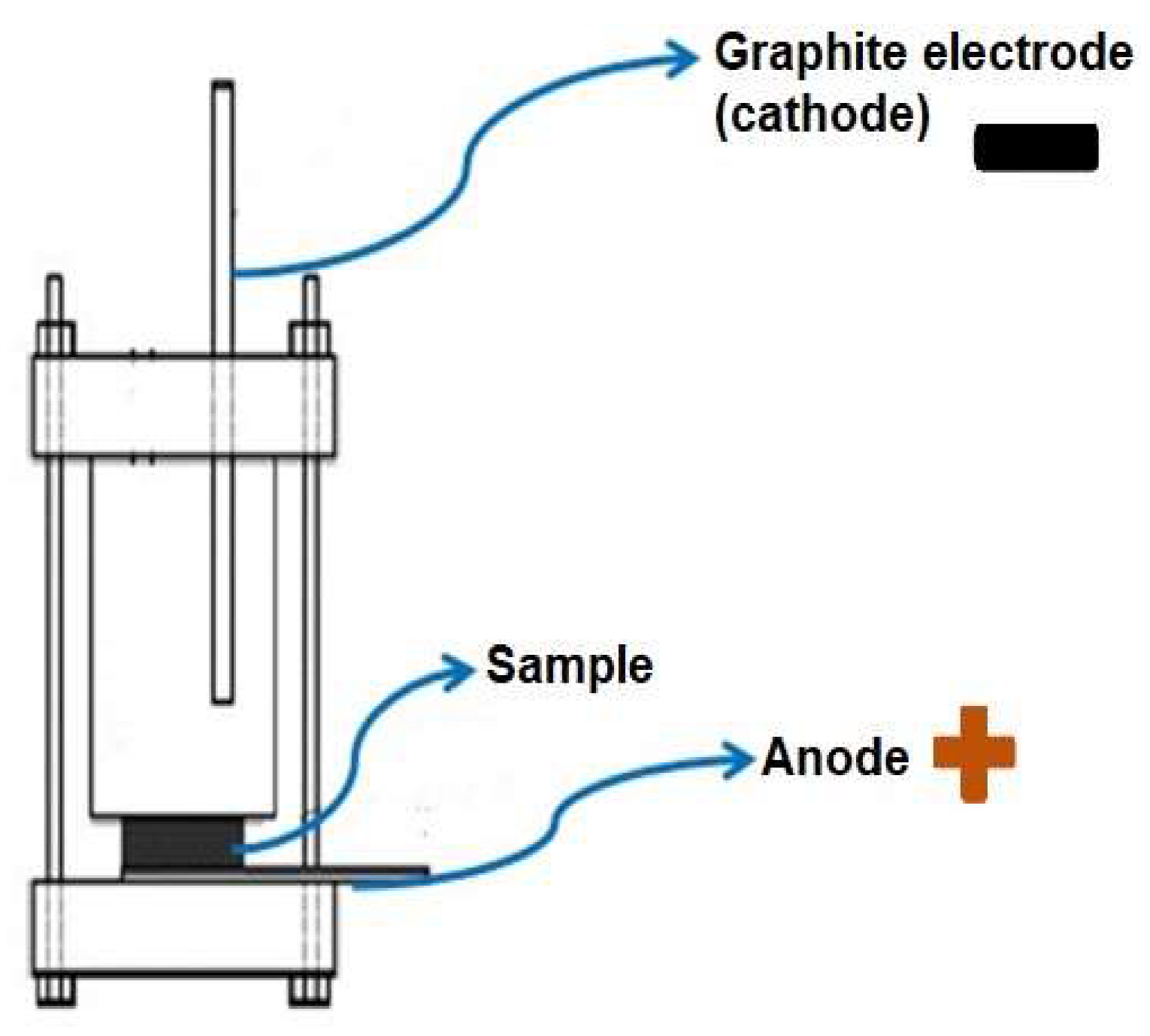
2.1. Anodizing Treatment
2.2. Surface Characterization
2.3. Wettability of the Surfaces
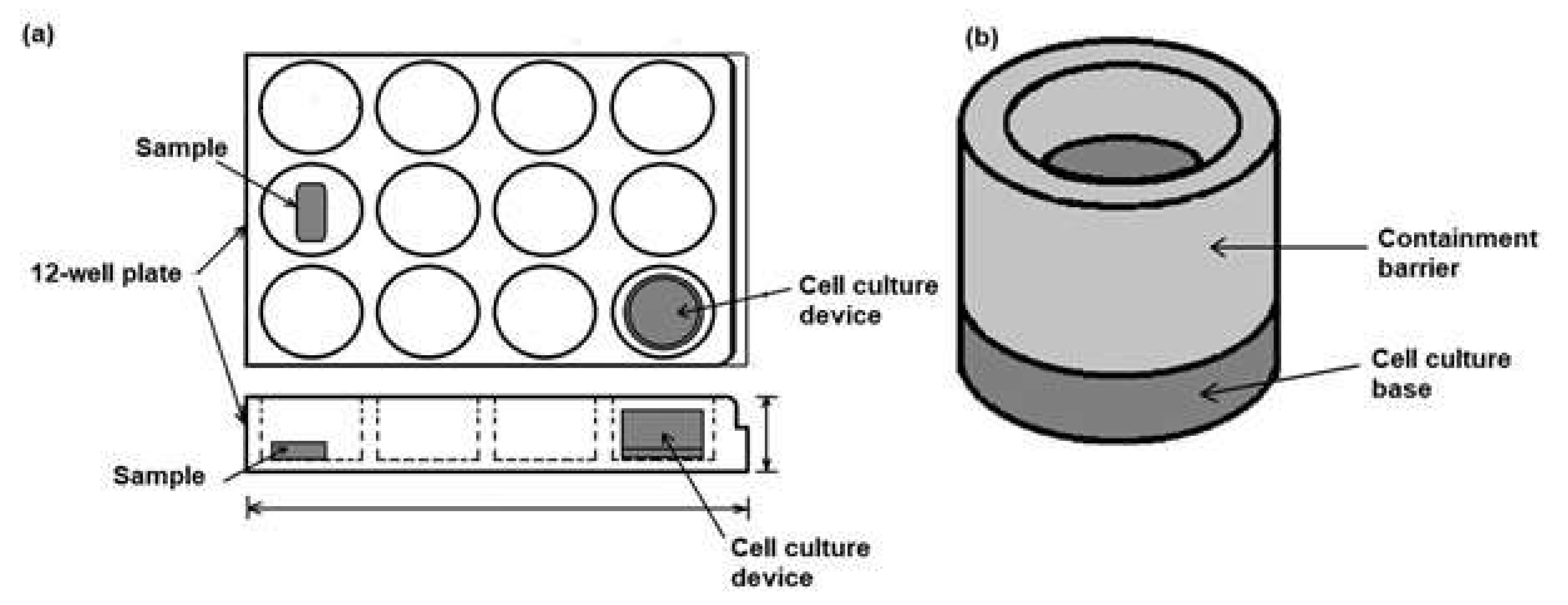
2.4. Design of the Direct Cell Culture Device
2.5. Cell Culture of Osteoarticular Cells
2.6. Cytotoxicity and Proliferation Assays in Osteoarticular Cells
3. Results and Discussions
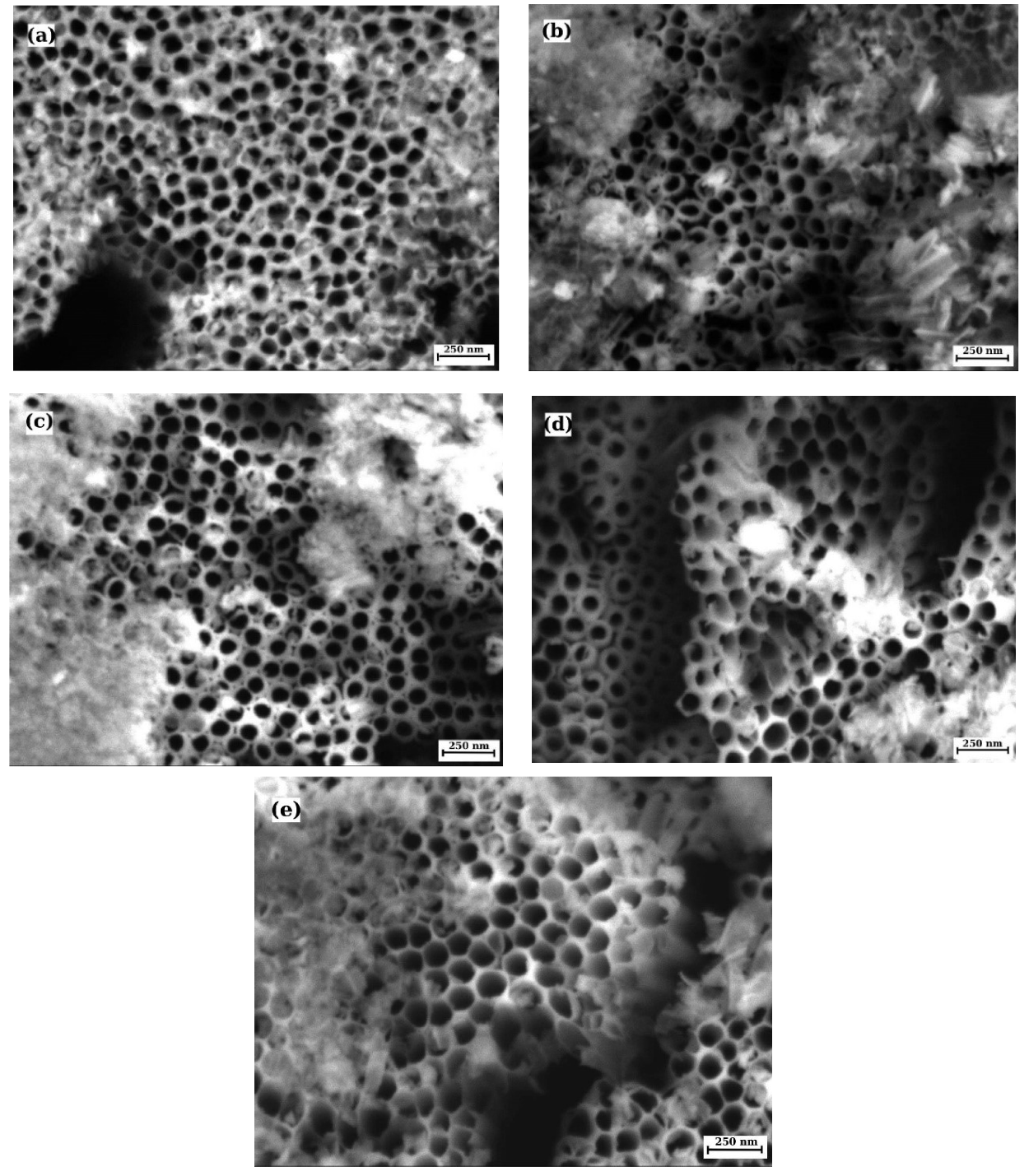
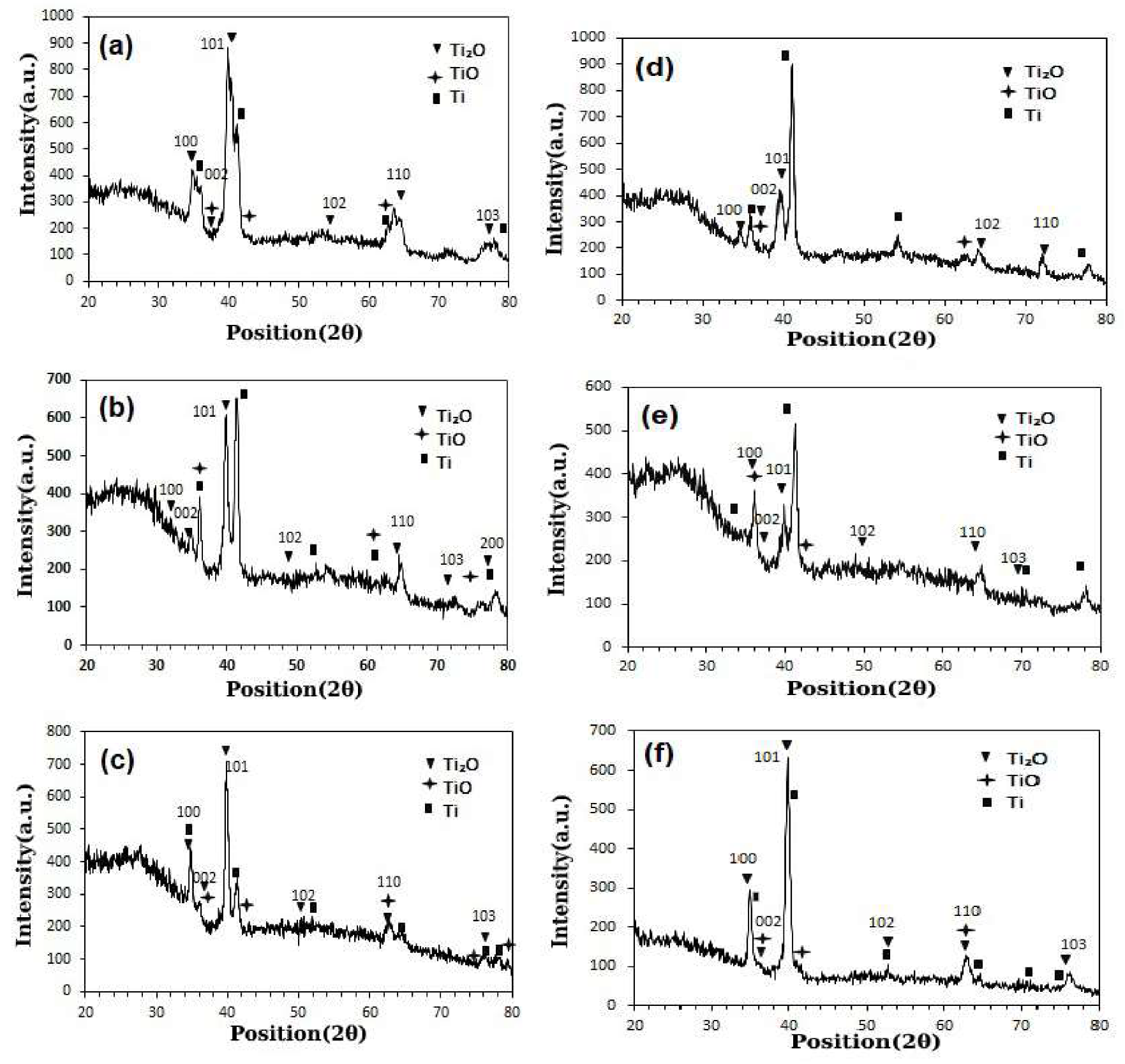
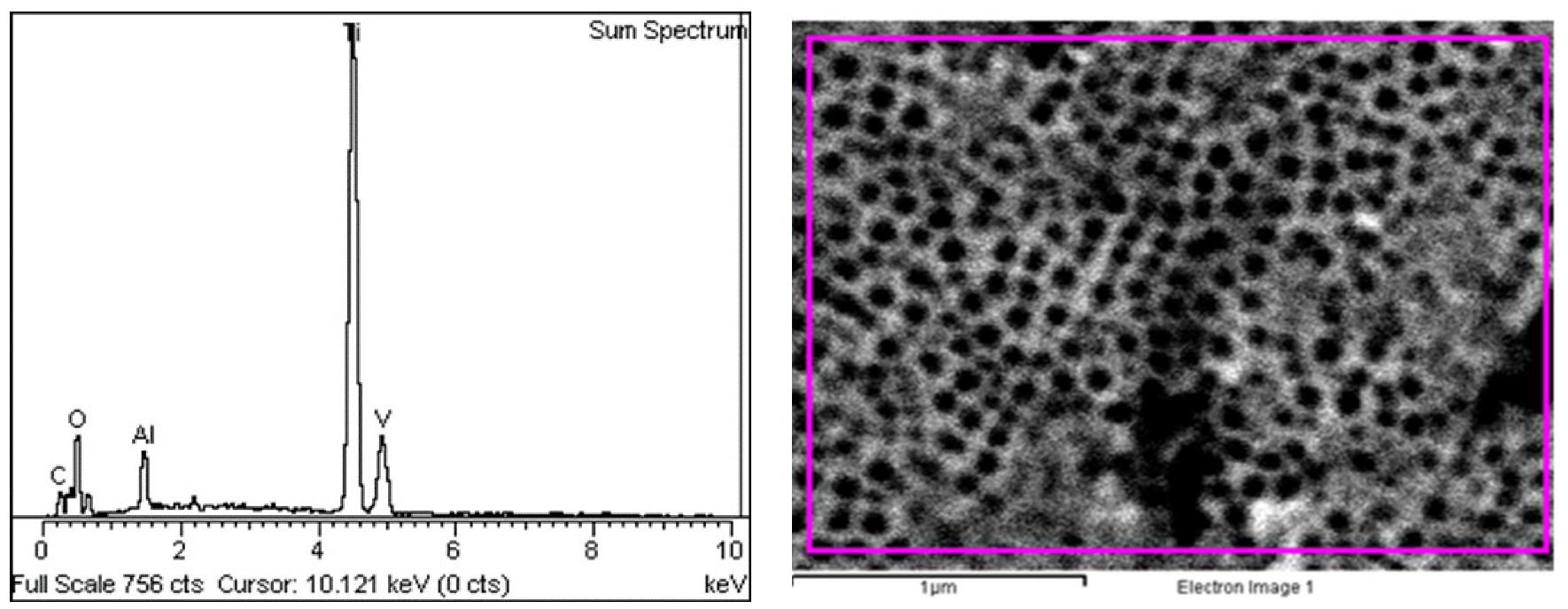
3.1. Surface Characterization
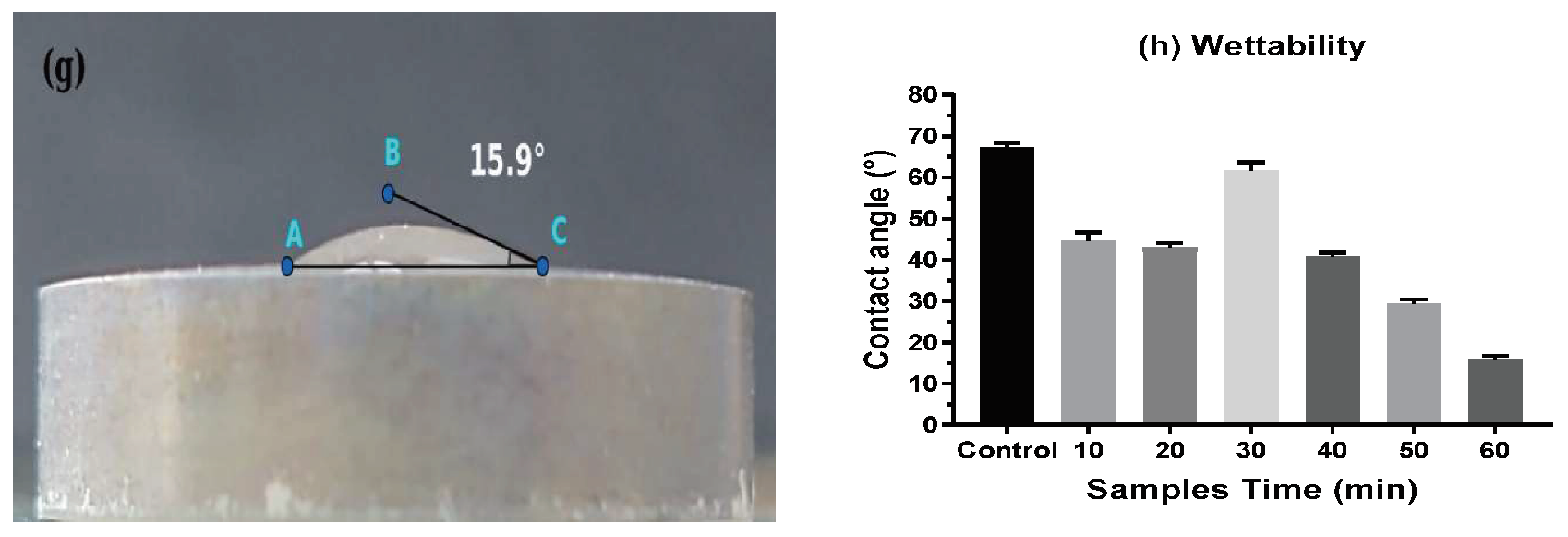
3.2. Wettability of the Surface
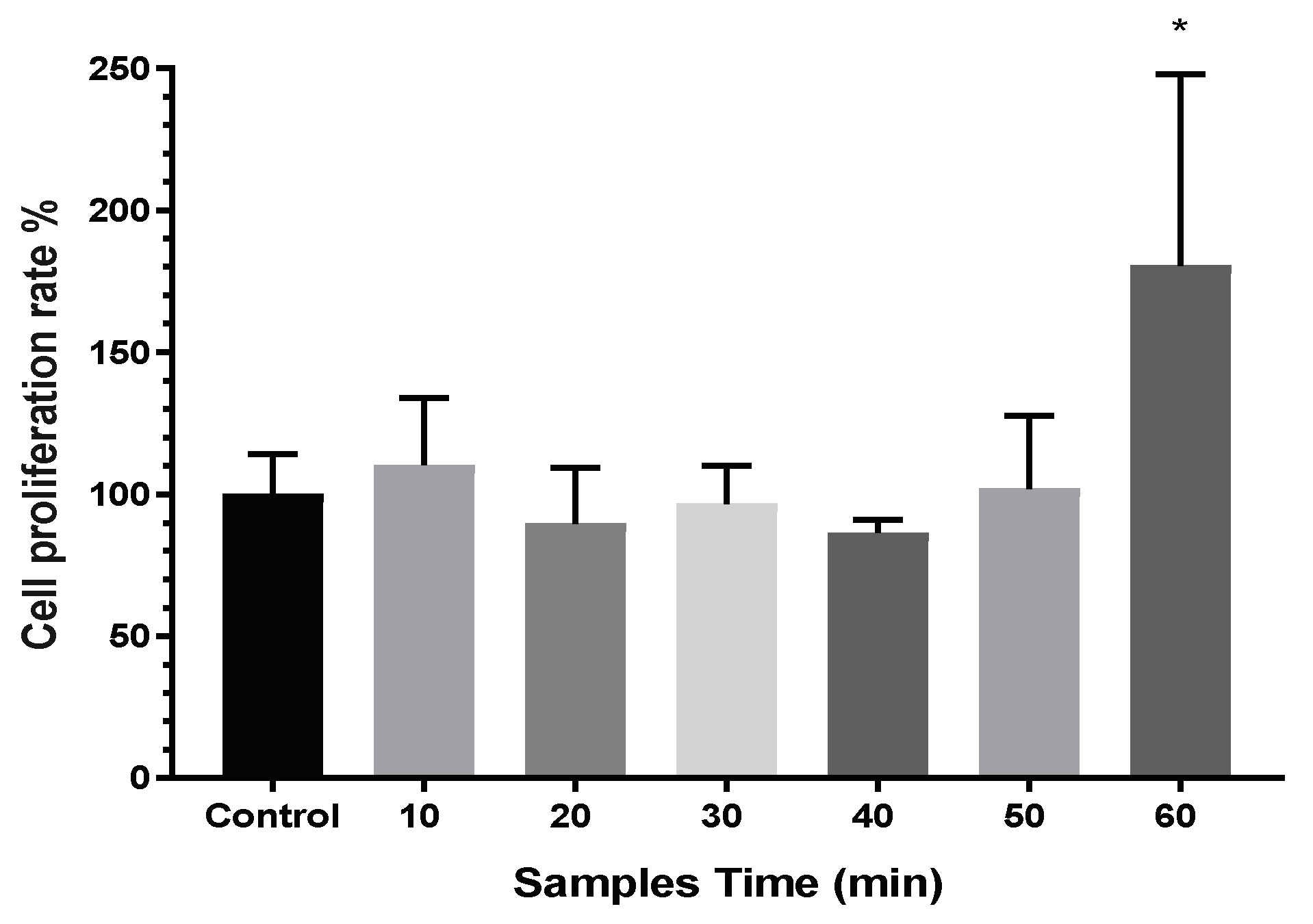
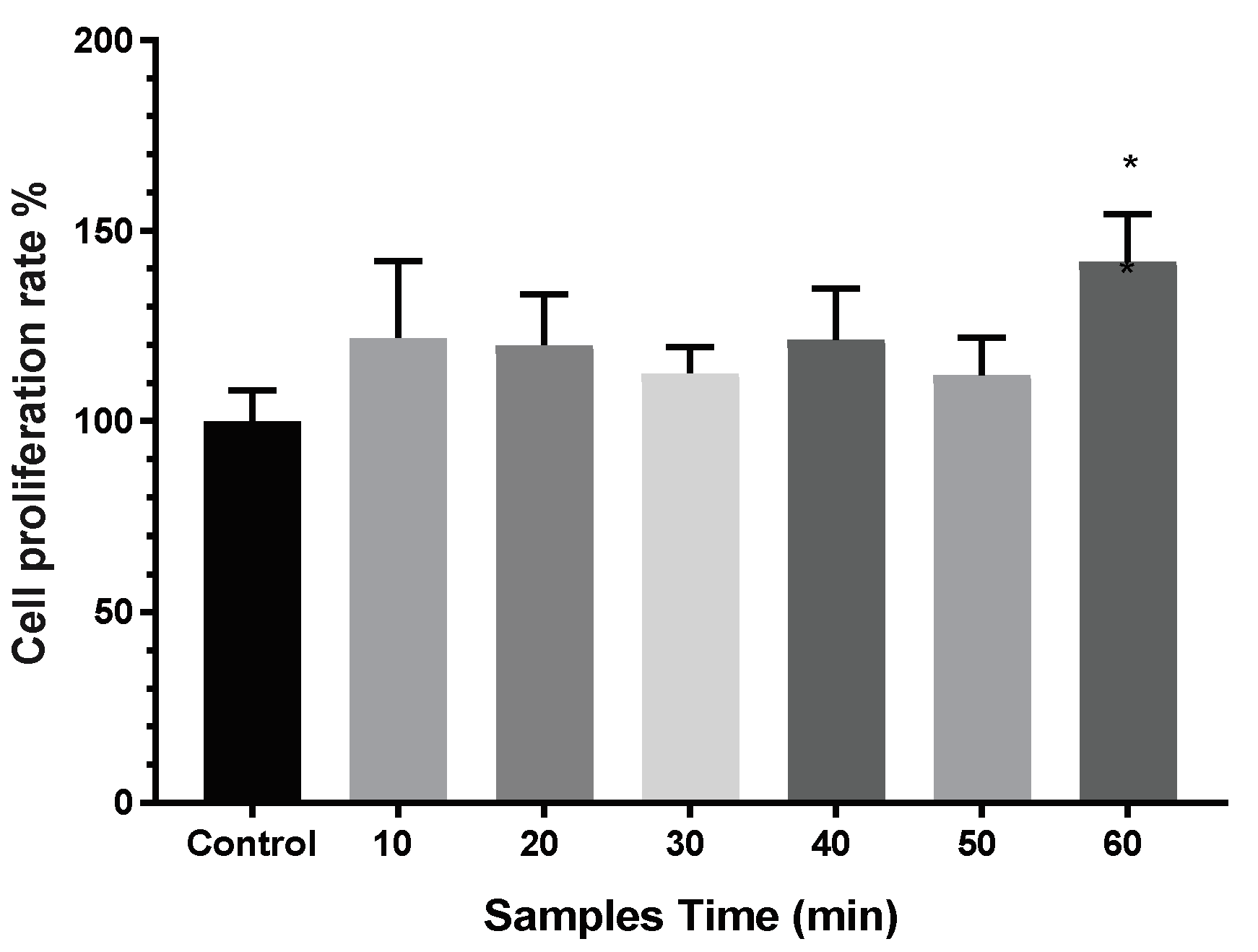
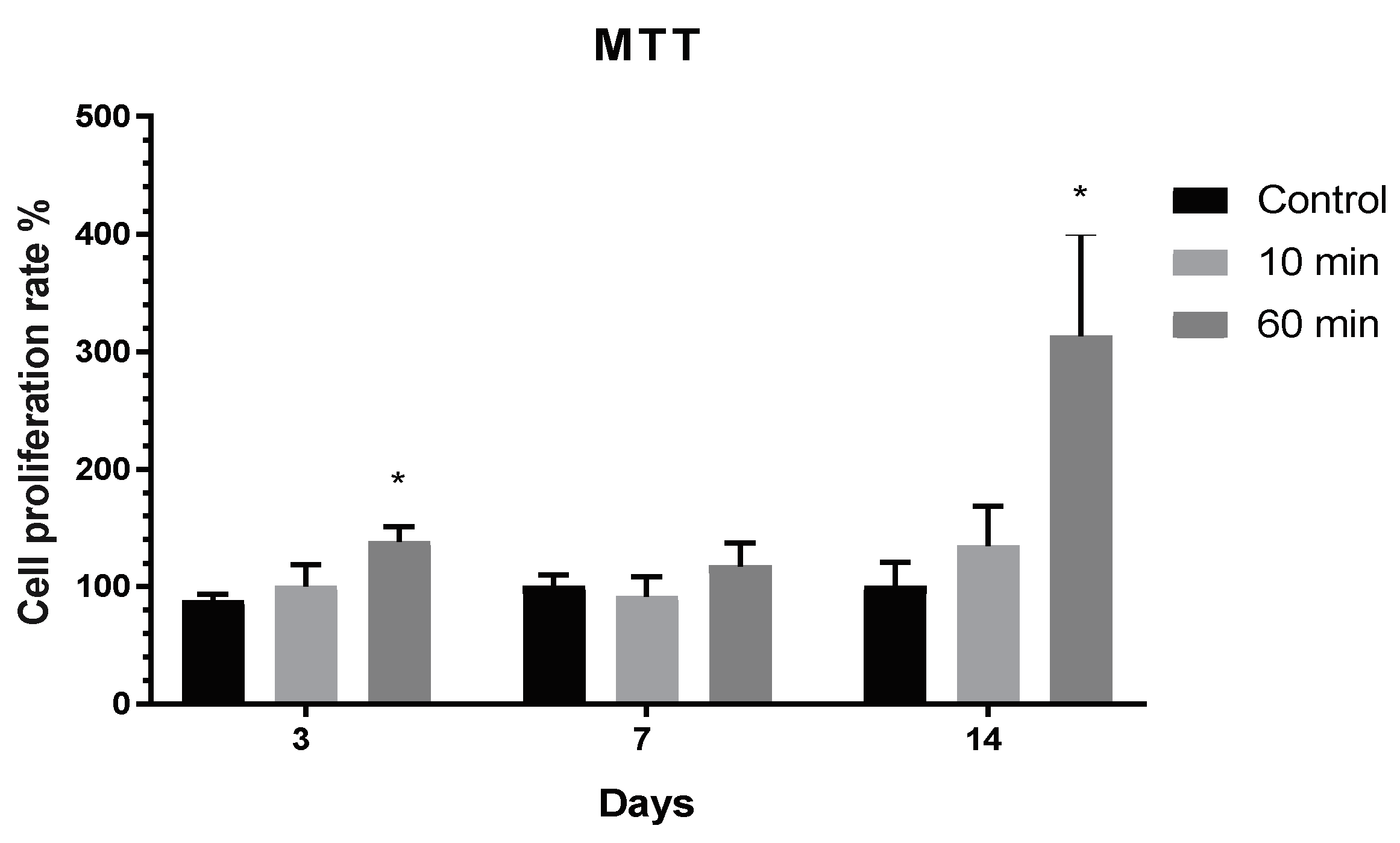
3.3. Cytotoxicity and Proliferation of TNT Determined in the Direct Cell Culture Device Culture
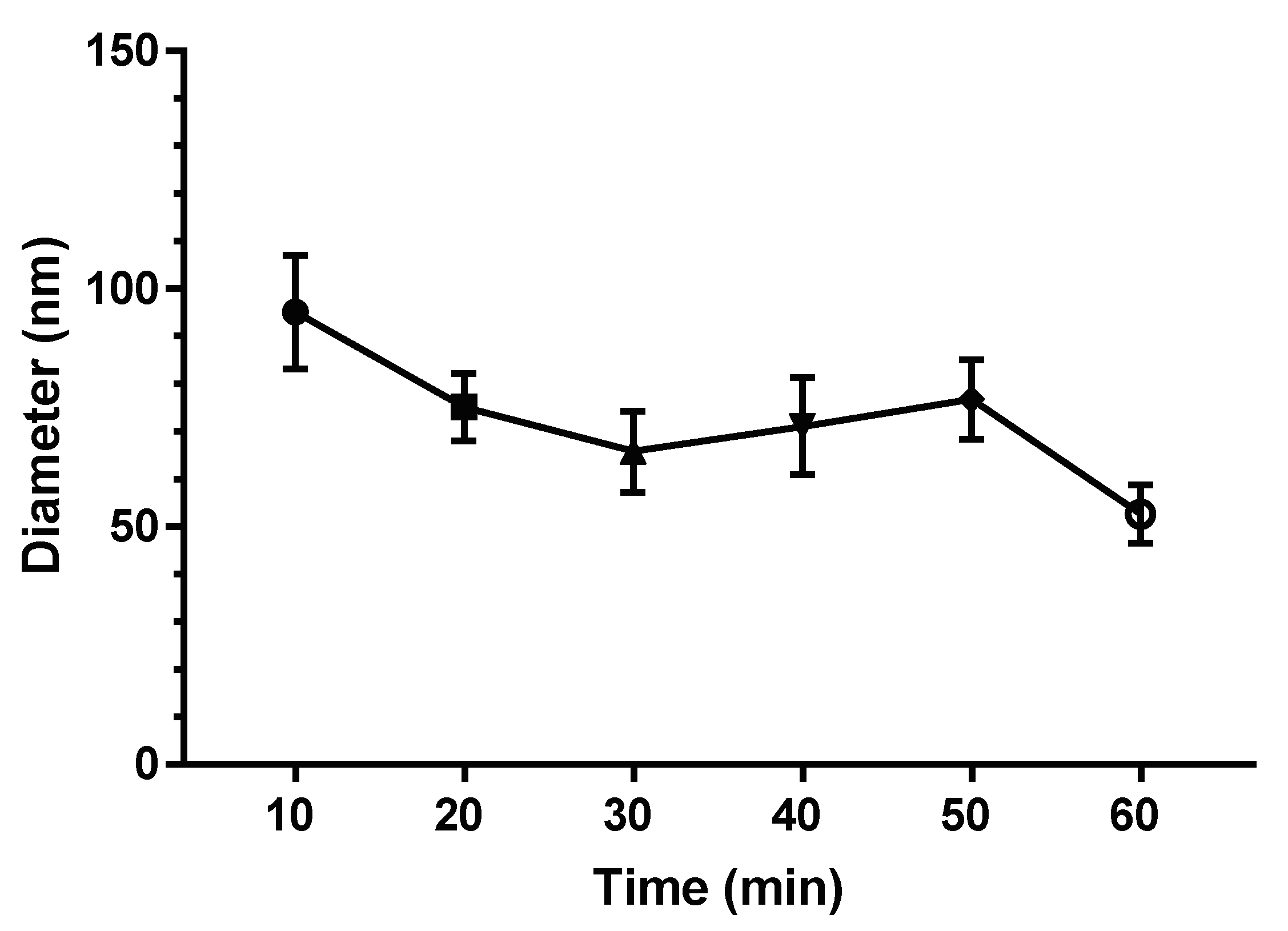
3.4. Osteoblast Morphology at Different Times
4. Conclusions
5. Patents
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baoe, L.; Ying, L.; Jun, L.; Xiaolong, F. Improvement of biological properties of titanium by anodic oxidation and ultraviolet irradiation. Appl. Surf. Sci. 2014, 307, 202–208. [Google Scholar]
- Katić, J.; Šarić, A.; Despotović, I.; Matijaković, N.; Petković, M.; Petrović, Ž. Bioactive Coating on Titanium Dental Implants for Improved Anticorrosion Protection: A Combined Experimental and Theoretical Study. Coatings 2019, 9, 612. [Google Scholar] [CrossRef]
- Gil, F.J.; Ginebra, M.P.; Planell, J.A. Metales y aleaciones para la substitución de tejidos duros. Biomecánica 1999, 13, 73–78. [Google Scholar]
- Castillea, T.; Paniagua, L.; Segura, J.; Seral, B.; Seral, E. Interfase patológica por fricción metal-metal y metal-polietileno Diferencias microscópicas. Revista Española de Cirujía Osteoarticular 1996, 31, 171–176. [Google Scholar]
- Zhao, L.; Mei, S.; Chu, P.K.; Zhang, Y.; Wu, Z. The influence of hierarchical hybrid micro/nano-textured titanium surface with titania nanotubes on osteoblast functions. Biomaterials 2010, 31, 5072. [Google Scholar] [CrossRef]
- Schwartz, Z.; Olivares-Navarrete, R.; Wieland, M.; Cochran, D.L.; Boyan, B.D. Mechanisms regulating increased production of osteoprotegerin by osteoblasts cultured on microstructured titanium surfaces. Biomaterials 2009, 30, 3390. [Google Scholar] [CrossRef]
- Kim, S.E.; Lee, S.B.; Kwak, S.W.; Kim, C.K.; Kim, K.N. Nanoporous anodic oxidation titanium enhances cell proliferation and differentiation of immortalized mouse embryonic cells. Surf. Coat. Technol. 2013, 228, S37–S40. [Google Scholar] [CrossRef]
- Wang, J.; Lin, Z. Anodic Formation of Ordered TiO2 Nanotube Arrays: Effects of Electrolyte Temperature and Anodization Potential. J. Phys. Chem. C 2009, 113, 4026–4030. [Google Scholar] [CrossRef]
- Quintero, D.; Galvis, O.; Calderón, J.A.; Castaño, J.G.; Echeverría, F. Effect of electrochemical parameters on the formation of anodic films on commercially pure titanium by plasma electrolytic oxidation. Surf. Coat. Technol. 2014, 258, 1223–1231. [Google Scholar] [CrossRef]
- Diamanti, M.V.; Pedeferri, M.P. EVect of anodic oxidation parameters on the titanium oxides formation. Corros. Sci. 2007, 49, 939–948. [Google Scholar] [CrossRef]
- Chen, J.F.; Lin, J.; Chen, X.F. Self-assembled TiO2 nanotube arrays with U-shaped profile by controlling anodization temperature. J. Nanomater. 2010. [Google Scholar] [CrossRef]
- Ponsonnet, L.; Reybier, K.; Jaffrezic, N.; Comte, V.; Lagneau, C.; Lissac, M.; Martelet, C. Relationship between surface properties (roughness, wettability) of titanium and titanium alloys and cell behavior. Mater. Sci. Eng. C 2003, 23, 551–560. [Google Scholar] [CrossRef]
- Rosales-Leal, J.I.; Rodríguez-Valverde, M.A.; Mazzaglia, G.; Ramón-Torregrosa, P.J.; Díaz-Rodríguez, L.; García-Martínez, O.; Vallecillo-Capilla, M.; Ruiz, C.; Cabrerizo-Vílchez, M.A. Effect of roughness, wettability and morphology of engineered titanium surfaces on osteoblast-like cell adhesion. Colloids Surf. Physicochem. Eng. Asp. 2010, 365, 222–229. [Google Scholar] [CrossRef]
- Voltrova, B.; Hybasek, V.; Blahnova, V.; Sepitka, J.; Lukasova, V.; Vocetkova, K.; Sovkova, V.; Matejka, R.; Fojt, J.; Joska, L.; et al. Different diameters of titanium dioxide nanotubes modulate Saos-2 osteoblast-like cell adhesion and osteogenic differentiation and nanomechanical properties of the surface. RSC Adv. 2019, 9, 11341–11355. [Google Scholar] [CrossRef]
- Bauer, S.; Park, J.; Faltenbacher, J.; Berger, S.; von der Mark, K.; Schmuki, P. Size selective behavior of mesenchymal stem cells on ZrO2 and TiO2 nanotube array. Integr. Biol. 2009, 1, 525–532. [Google Scholar] [CrossRef]
- Lan, M.Y.; Liu, C.P.; Huang, H.H.; Lee, S.W. Both Enhanced Biocompatibility and Antibacterial Activity in Ag-Decorated TiO2 Nanotubes. PLoS ONE 2013, 8, e75364. [Google Scholar] [CrossRef]
- Rafieerad, A.R.; Bushroa, A.R.; Nasiri-Tabrizi, B.; Baradaran, S.; Amiri, A.; Saber-Samandari, S.; Khanahmadi, S.; Zeimaran, E.; Basirun, W.J.; Kalaiselvam, K. Simultaneous enhanced antibacterial, and osteoblast cytocompatibility performance of Ti6Al7Nb implant by nano-silver/graphene oxide decorated mixed oxide nanotube composite. Surf. Coat. Technol. 2019, 360, 181–195. [Google Scholar] [CrossRef]
- Liu, Q.; Li, W.; Cao, L.; Wang, J.; Qu, Y.; Wang, X.; Qiu, R.; Di, X.; Wang, Z.; Liang, B. Response of MG63 Osteoblast Cells to Surface Modification of Ti-6Al-4V Implant Alloy by Laser Interference Lithography. J. Bionic Eng. 2017, 14, 448–458. [Google Scholar] [CrossRef]
- Kaczmarek, M.; Jurczyk, M.U.; Miklaszewski, A.; Paszel-Jaworska, A.; Romaniuk, A.; Lipińska, N.; Żurawski, J.; Urbaniak, P.; Jurczyk, K. In vitro biocompatibility of titanium after plasma surface alloying with boron. Mater. Sci. Eng. C. 2016, 69, 1240–1247. [Google Scholar] [CrossRef]
- Wang, X.; Qu, Z.; Li, J.; Zhang, E. Comparison study on the solution-based surface biomodification of titanium: Surface characteristics and cell biocompatibility. Surf. Coat. Technol. 2017, 329, 109–119. [Google Scholar] [CrossRef]
- Biesiekierski, A.; Li, Y.; Xiao, Y.; Cuie, W. Assessing the Biocompatibility of Biomaterials: A Critical Review of Current In Vitro Toxicity Assays, Their Advantages and Limitations; LAP LAMBERT Academic Publishing: Mauritius Rīgā, Latvia, 2018. [Google Scholar]
- Webb, K.; Hlady, V.; Tresco, P.A. Relative importance of surface wettability and charged functional groups on NIH 3T3 fibroblast attachment, spreading, and cytoskeletal organization. J. Biomed. Mater. Res. 1998, 41, 422–430. [Google Scholar] [CrossRef]
- Macak, J.M.; Tsuchiya, H.; Ghicov, A.; Yasuda, K.; Hahn, R.; Bauer, S.; Schmuki, P. TiO2 nanotubes: Self-organized electrochemical formation, properties, and applications. Curr. Opin. Solid SMS 2007, 11, 113–118. [Google Scholar] [CrossRef]
- Yin, H.; Liu, H.; Shen, W.Z. The large diameter and fast growth of self-organized TiO2 nanotube arrays achieved via electrochemical anodization. Nanotechnology 2010, 21, 1–7. [Google Scholar] [CrossRef]
- Lan, M.-Y.; Liu, C.-P.; Huang, H.-H.; Chang, J.-K.; Lee, S.-W. Diameter-sensitive biocompatibility of anodic TiO2 nanotubes treated with supercritical CO2 fluid. Nanoscale Res. Lett. 2013, 8, 1–8. [Google Scholar] [CrossRef]
- Xing, J.; Xia, Z.; Hu, J.; Zhang, Y.; Zhong, L. Time dependence of growth and crystallization of anodic titanium oxide films in potentiostatic mode. Corros. Sci. 2013, 75, 212–219. [Google Scholar] [CrossRef]
- de Tacconi, N.R.; Chenthamarakshan, C.R.; Yogeeswaran, G.; Watcharenwong, A.; de Zoysa, R.S.; Basit, N.A.; Rajeshwar, K. Nanoporous TiO2 and WO3 Films by Anodization of Titanium and Tungsten Substrates: Influence of Process Variables on Morphology and Photoelectrochemical Response. J. Phys. Chem. B 2006, 110, 25347–25355. [Google Scholar] [CrossRef]
- Su, Z.; Zhang, L.; Jiang, F.; Hong, M. Formation ofcrystallineTiO2 by anodicoxidation of titanium. Prog. Nat. Sci. Mater. Int. 2013, 23, 294–301. [Google Scholar] [CrossRef]
- Wittenbrink, I.; Hausmann, A.; Schickle, K.; Lauria, I.; Davtalab, R.; Foss, M.; Fischer, H. Low-aspect ratio nanopatterns on bioinert alumina influence the response and morphology of osteoblast-like cells. Biomaterials 2015, 62, 58–65. [Google Scholar] [CrossRef]
- Rodriguez-Mercado, J.J.; Alvarez-Barrera, L.; Altamirano-Lozano, M.A. Chromosomal damage induced by vanadium oxides in human peripheral lymphocytes. Drug Chem. Toxicol. 2010, 33, 97–102. [Google Scholar] [CrossRef]
- Ivanković, S.; Musić, S.; Gotić, M.; Ljubešić, N. Cytotoxicity of nanosize V2O5 particles to selected fibroblast and tumor cells. Toxicol. Vitro 2006, 20, 286–294. [Google Scholar] [CrossRef]
- Srivastava, S.; Kumar, N.; Roy, P. Role of ERK/NFkB in vanadium (IV) oxide mediated osteoblast differentiation in C3H10t1/2 cells. Biochimie 2014, 101, 132–144. [Google Scholar] [CrossRef] [PubMed]
- Smeets, R.; Stadlinger, B.; Schwarz, F.; Beck-Broichsitter, B.; Jung, O.; Precht, C.; Ebker, T. Impact of Dental Implant Surface Modifications on Osseointegration. BioMed Res. Int. 2016, 20, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Das, K.; Bose, S.; Bandyopadhyay, A. Surface modifications and cell-materials interactions with anodized Ti. Acta Biomater. 2007, 3, 573–585. [Google Scholar] [CrossRef]
- Toita, R.; Tsuru, K.; Ishikawa, K. Ozone-Gas-Mediated Surface Hydrophilization Enhances the Cell Responses to Titanium. Mater. Lett. 2020, 261, 127168. [Google Scholar]
- Brammer, K.S.; Oh, S.; Cobb, C.J.; Bjursten, L.M.; van der Heyde, H.; Jin, S. Improved bone-forming functionality on diameter-controlled TiO2 nanotube surface. Acta Biomater. 2009, 5, 3215–3223. [Google Scholar] [CrossRef]
- Moravec, H.; Vandrovcova, M.; Chotova, K.; Fojt, J.; Pruchova, E.; Joska, L.; Bacakova, L. Cell interaction with modified nanotubes formed on titanium alloy Ti-6Al-4V. Mater. Sci. Eng. C 2016, 65, 313–322. [Google Scholar] [CrossRef]
- Oh, S.; Brammer, K.S.; Li, Y.S.J.; Teng, D.; Engler, A.J.; Chien, S.; Jin, S. Stem cell fate dictated solely by altered nanotube dimension. Proc. Natl. Acad. Sci. USA 2009, 106, 2130–2135. [Google Scholar] [CrossRef]



| C | Fe (Max) | H (Max) | N (Max) | O (Max) | V | Al | Ti |
|---|---|---|---|---|---|---|---|
| 0.08 | 0.30 | 0.015 | 0.05 | 0.2 | 3.5–4.5 | 5.5–6.75 | Balance |
| C (wt.%) | O (wt.%) | F (wt.%) | Al (wt.%) | V (wt.%) | Ti (wt.%) |
|---|---|---|---|---|---|
| 4.9 | 30.15 | 10.99 | 2.93 | 1.68 | 49.34 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres-Avila, I.P.; Padilla-Martínez, I.I.; Pérez-Hernández, N.; Bañuelos-Hernández, A.E.; Velázquez, J.C.; Castrejón-Flores, J.L.; Hernández-Sánchez, E. Surface Modification of the Ti-6Al-4V Alloy by Anodic Oxidation and Its Effect on Osteoarticular Cell Proliferation. Coatings 2020, 10, 491. https://doi.org/10.3390/coatings10050491
Torres-Avila IP, Padilla-Martínez II, Pérez-Hernández N, Bañuelos-Hernández AE, Velázquez JC, Castrejón-Flores JL, Hernández-Sánchez E. Surface Modification of the Ti-6Al-4V Alloy by Anodic Oxidation and Its Effect on Osteoarticular Cell Proliferation. Coatings. 2020; 10(5):491. https://doi.org/10.3390/coatings10050491
Chicago/Turabian StyleTorres-Avila, Itzel P., Itzia I. Padilla-Martínez, Nury Pérez-Hernández, Angel E. Bañuelos-Hernández, Julio C. Velázquez, José L. Castrejón-Flores, and Enrique Hernández-Sánchez. 2020. "Surface Modification of the Ti-6Al-4V Alloy by Anodic Oxidation and Its Effect on Osteoarticular Cell Proliferation" Coatings 10, no. 5: 491. https://doi.org/10.3390/coatings10050491
APA StyleTorres-Avila, I. P., Padilla-Martínez, I. I., Pérez-Hernández, N., Bañuelos-Hernández, A. E., Velázquez, J. C., Castrejón-Flores, J. L., & Hernández-Sánchez, E. (2020). Surface Modification of the Ti-6Al-4V Alloy by Anodic Oxidation and Its Effect on Osteoarticular Cell Proliferation. Coatings, 10(5), 491. https://doi.org/10.3390/coatings10050491






