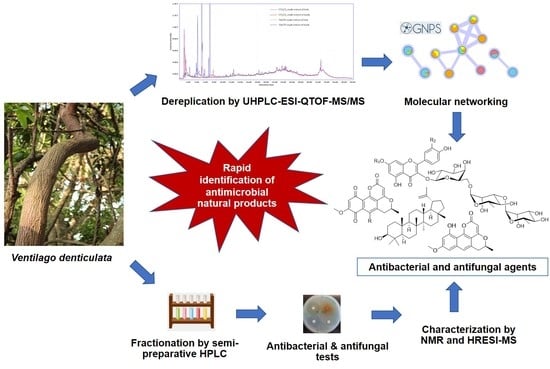
UHPLC-ESI-QTOF-MS/MS-Based Molecular Networking Guided Isolation and Dereplication of Antibacterial and Antifungal Constituents of Ventilago denticulata
Abstract
1. Introduction
2. Results and Discussions
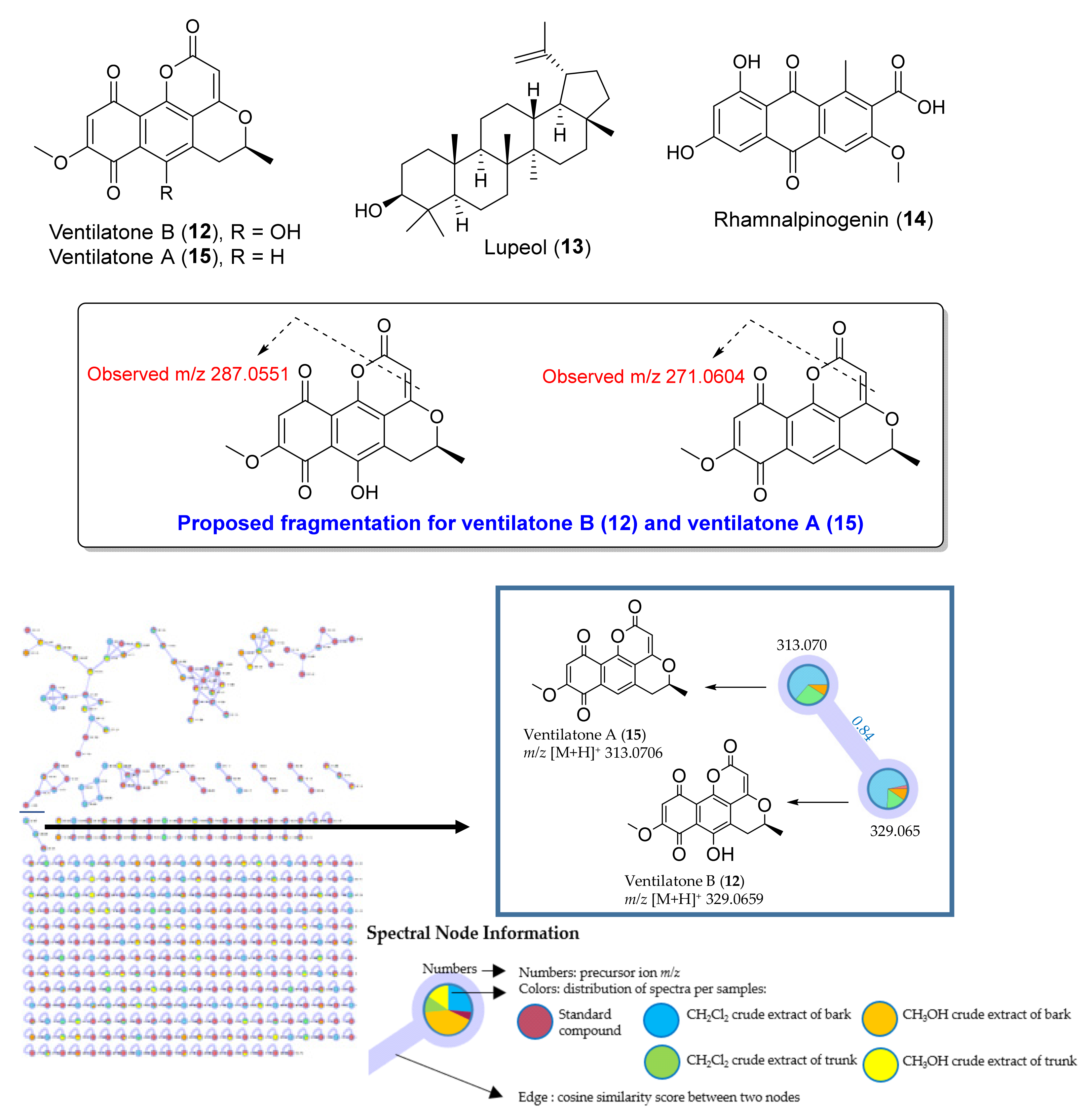
2.1. Dereplication of Compounds from Crude Extracts of V. denticulata and Guided Isolation by UHPLC-ESI-QTOF-MS/MS-Based Molecular Networking
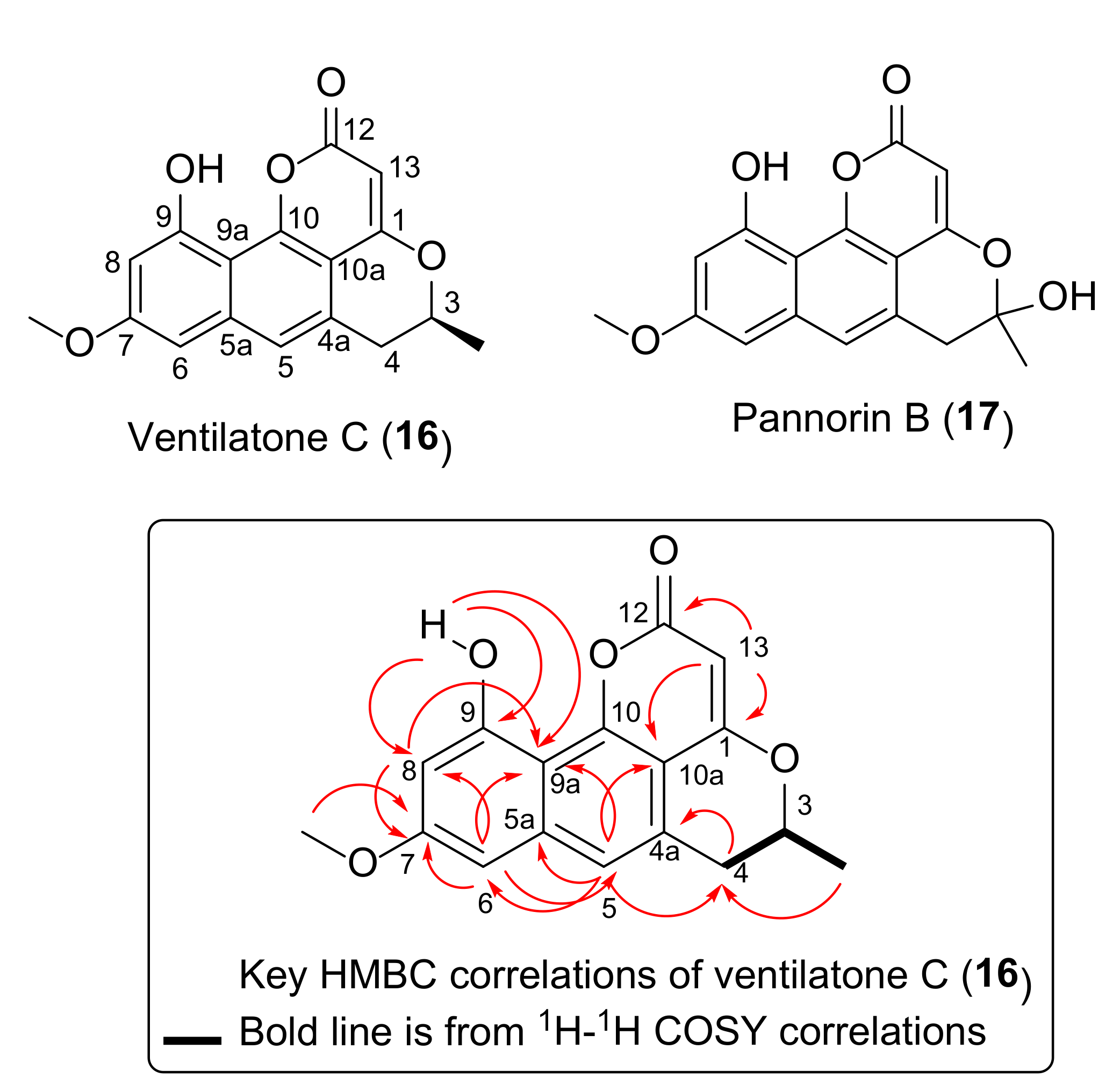
2.2. Structure Elucidation of Ventilatone C (16)
2.3. Antibacterial and Antifungal Activities of Crude Extracts, Fractions and Isolated Compounds
2.4. Dereplication of Antibacterial and Antifungal Constituents from HPLC Fractions of V. denticulata
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Plant Materials and Extraction of Plant
3.3. Crude Extract and Preparation of Standard Compounds for LC-MS/MS Analysis
3.4. UHPLC-ESI-QTOF-MS/MS Conditions
3.5. Molecular Networking
3.5.1. Converting MS/MS Data
- 32-Bit was selected for binary encoding precision and zip compression was unchecked.
- Peak picking was set as a filter to make the output data become centroid.
- MS-Levels 1 and 2 should be checked.
3.5.2. Molecular Networking by GNPS (Global Natural Products Social Molecular Networking)
3.5.3. Visualization of Molecular Networking Using Cytoscape
3.6. Isolation of (+)-(R)-Ventilagolin (1), Flavonoid Glycosides (7–11), Ventilatone B (12), Lupeol (13), Ventilatone A (15) and Ventilatone C (16)
3.7. Spectroscopic Data of a New Compound, Ventilatone C (16)
3.8. Structure Elucidation of the Isolated Compounds
3.9. HPLC Fractionation of V. denticulata Extracts
3.10. ESI-HRMS Analysis for the Identification of Compounds in HPLC Fractions
3.11. In-Vitro Antibacterial and Antifungal Assays
3.11.1. Preparation of Bacteria and Fungi for Bioassay
3.11.2. Disk Diffusion Method for Antibacterial and Antifungal Assays
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Over, B.; Wetzel, S.; Grütter, C.; Nakai, Y.; Renner, S.; Rauh, D.; Waldmann, H. Natural-product-derived fragments for fragment-based ligand discovery. Nat. Chem. 2013, 5, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed]
- Patridge, E.; Gareiss, P.; Kinch, M.S.; Hoyer, D. An analysis of FDA-approved drugs: Natural products and their derivatives. Drug Discov. Today 2016, 21, 204–207. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta 2013, 1830, 3670–3695. [Google Scholar] [CrossRef]
- Wibowo, M.; Forster, P.I.; Guymer, G.P.; Hofmann, A.; Davis, R.A. Using UHPLC-MS profiling for the discovery of new dihydro-beta-agarofurans from Australian Celastraceae plant extracts. Molecules 2019, 24, 859. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.Y.; Sanchez, L.M.; Rath, C.M.; Liu, X.; Boudreau, P.D.; Bruns, N.; Glukhov, E.; Wodtke, A.; de Felicio, R.; Fenner, A.; et al. Molecular networking as a dereplication strategy. J. Nat. Prod. 2013, 76, 1686–1699. [Google Scholar] [CrossRef]
- Fox Ramos, A.E.; Evanno, L.; Poupon, E.; Champy, P.; Beniddir, M.A. Natural products targeting strategies involving molecular networking: Different manners, one goal. Nat. Prod. Rep. 2019, 36, 960–980. [Google Scholar] [CrossRef]
- Zani, C.L.; Carroll, A.R. Database for rapid dereplication of known natural products using data from MS and fast NMR experiments. J. Nat. Prod. 2017, 80, 1758–1766. [Google Scholar] [CrossRef]
- Mohimani, H.; Gurevich, A.; Shlemov, A.; Mikheenko, A.; Korobeynikov, A.; Cao, L.; Shcherbin, E.; Nothias, L.F.; Dorrestein, P.C.; Pevzner, P.A. Dereplication of microbial metabolites through database search of mass spectra. Nat. Commun. 2018, 9, 4035. [Google Scholar] [CrossRef]
- Quinn, R.A.; Nothias, L.F.; Vining, O.; Meehan, M.; Esquenazi, E.; Dorrestein, P.C. Molecular networking as a drug discovery, drug metabolism, and precision medicine strategy. Trends Pharmacol. Sci. 2017, 38, 143–154. [Google Scholar] [CrossRef]
- Wang, M.; Carver, J.J.; Phelan, V.V.; Sanchez, L.M.; Garg, N.; Peng, Y.; Nguyen, D.D.; Watrous, J.; Kapono, C.A.; Luzzatto-Knaan, T.; et al. Sharing and community curation of mass spectrometry data with global natural products social molecular networking. Nat. Biotechnol. 2016, 34, 828–837. [Google Scholar] [CrossRef] [PubMed]
- Zang, Y.; Gong, Y.; Gong, J.; Liu, J.; Chen, C.; Gu, L.; Zhou, Y.; Wang, J.; Zhu, H.; Zhang, Y. Fungal polyketides with three distinctive ring skeletons from the fungus Penicillium canescens uncovered by OSMAC and molecular networking strategies. J. Org. Chem. 2020, 85, 4973–4980. [Google Scholar] [CrossRef]
- Woo, S.; Kang, K.B.; Kim, J.; Sung, S.H. Molecular networking reveals the chemical diversity of selaginellin derivatives, natural phosphodiesterase-4 inhibitors from Selaginella tamariscina. J. Nat. Prod. 2019, 82, 1820–1830. [Google Scholar] [CrossRef] [PubMed]
- Gross, M. The race against antibiotics resistance. Curr. Biol. 2019, 29, R859–R861. [Google Scholar] [CrossRef]
- Kadchumsang, S.; Sirisa-Ard, P.; Sookkhee, S.; Chansakaow, S. Antibacterial and antioxidant activities of lanna medicinal plants used in mahoog formula. Int. J. Pharm. Pharm. Res. 2015, 7, 366–370. [Google Scholar]
- Bessa, L.J.; Fazii, P.; Di Giulio, M.; Cellini, L. Bacterial isolates from infected wounds and their antibiotic susceptibility pattern: Some remarks about wound infection. Int. Wound J. 2015, 12, 47–52. [Google Scholar] [CrossRef]
- Michelotti, F.; Bodansky, H.J. Bacillus cereus causing widespread necrotising skin infection in a diabetic person. Pract. Diabetes 2015, 32, 169–170a. [Google Scholar] [CrossRef]
- Sfeir, M.; Youssef, P.; Mokhbat, J.E. Salmonella typhi sternal wound infection. Am. J. Infect. Control 2013, 41, e123–e124. [Google Scholar] [CrossRef]
- Kalan, L.; Grice, E.A. Fungi in the wound microbiome. Adv. Wound Care (New Rochelle) 2018, 7, 247–255. [Google Scholar] [CrossRef]
- Tambe, A.B.; Dhage, O.L.; Bhalerao, P.B.; Bhalerao, S.S.; Thokal, S.H.; Shende, V.S.; Bansode, A.S.; Devhadrao, N.V. In-vitro anti-fungal activity and phytochemical screening of stem bark extracts from Ventilago denticulata. J. Drug Deliv. Ther. 2019, 9, 359–362. [Google Scholar] [CrossRef]
- Venkata, S.P.; Murali, M.C.; da Silva, J.A.T.; Raju, B.A.; Sravani, R. Screening the antimicrobial and antioxidant potential of Ventilago denticulata, Scolopia crenata and Rivea hypocrateriformis from maredumilli forest, india. Med. Aromat. Plant Sci. Biotechnol. 2012, 6, 58–62. [Google Scholar]
- Molee, W.; Phanumartwiwath, A.; Kesornpun, C.; Sureram, S.; Ngamrojanavanich, N.; Ingkaninan, K.; Mahidol, C.; Ruchirawat, S.; Kittakoop, P. Naphthalene derivatives and quinones from Ventilago denticulata and their nitric oxide radical scavenging, antioxidant, cytotoxic, antibacterial, and phosphodiesterase inhibitory activities. Chem. Biodivers. 2018, 15, e1700537. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.Y.; Shin, H.; Lim, J.W.; Ahn, J.H.; Jo, Y.H.; Lee, K.Y.; Hwang, B.Y.; Jung, S.J.; Kang, S.Y.; Lee, M.K. Comparison of antibacterial activity and phenolic constituents of bark, lignum, leaves and fruit of Rhus verniciflua. PLoS ONE 2018, 13, e0200257. [Google Scholar] [CrossRef] [PubMed]
- Ruan, J.; Yan, J.; Zheng, D.; Sun, F.; Wang, J.; Han, L.; Zhang, Y.; Wang, T. Comprehensive chemical profiling in the ethanol extract of Pluchea indica aerial parts by liquid chromatography/mass spectrometry analysis of its silica gel column chromatography fractions. Molecules 2019, 24, 2784. [Google Scholar] [CrossRef] [PubMed]
- Kachlicki, P.; Piasecka, A.; Stobiecki, M.; Marczak, Ł. Structural characterization of flavonoid glycoconjugates and their derivatives with mass spectrometric techniques. Molecules 2016, 21, 1494. [Google Scholar] [CrossRef]
- Medentsev, A.G.; Arinbasarova, A.Y.; Akimenko, V.K. Biosynthesis of naphthoquinone pigments by fungi of the genus Fusarium. Appl. Biochem. Microbiol. 2005, 41, 503–507. [Google Scholar] [CrossRef]
- Adorisio, S.; Fierabracci, A.; Muscari, I.; Liberati, A.M.; Cannarile, L.; Thuy, T.T.; Sung, T.V.; Sohrab, H.; Hasan, C.M.; Ayroldi, E.; et al. Fusarubin and anhydrofusarubin isolated from a Cladosporium species inhibit cell growth in human cancer cell lines. Toxins 2019, 11, 503. [Google Scholar] [CrossRef]
- Riess-Maurer, I.; Wagner, H. Struktur und synthese von flavonol-triosiden aus rhamnus-arten. Tetrahedron 1982, 38, 1269–1278. [Google Scholar] [CrossRef]
- Dutton, G.G.S.; Merrifield, E.H.; Laffite, C.; Pratviel-Sosa, F.; Wylde, R. Comparative NMR study of rhamnobioses-applications. Org. Magn. Reson. 1982, 20, 154–158. [Google Scholar] [CrossRef]
- Wang, J.; Kasai, R.; Sakimori, M.; Miyakoshi, M.; Tanaka, O.; Jia, M.-R.; Ling, Y.-K. Flavonol glycosides from the fruits of Rhamus leptophylla. Phytochemistry 1988, 27, 3995–3996. [Google Scholar] [CrossRef]
- Riess-Maurer, I.; Wagner, H.; Lipták, A. Synthese und 13C-NMR-untersuchung von flavonol-3-O-(dirhamnosyl)-galactosiden der xanthorhamnin-, catharticin- und alaterin-reihe. Tetrahedron Lett. 1979, 20, 3695–3698. [Google Scholar] [CrossRef]
- Zheng, Y.F.; Qi, L.W.; Zhou, J.L.; Li, P. Structural characterization and identification of oleanane-type triterpene saponins in Glycyrrhiza uralensis Fischer by rapid-resolution liquid chromatography coupled with time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 2010, 24, 3261–3270. [Google Scholar] [CrossRef] [PubMed]
- Peterman, S.M.; Mulholland, J.J. A novel approach for identification and characterization of glycoproteins using a hybrid linear ion trap/FT-ICR mass spectrometer. J. Am. Soc. Mass Spectrom. 2006, 17, 168–179. [Google Scholar] [CrossRef] [PubMed]
- Halim, A.; Westerlind, U.; Pett, C.; Schorlemer, M.; Rüetschi, U.; Brinkmalm, G.; Sihlbom, C.; Lengqvist, J.; Larson, G.; Nilsson, J. Assignment of saccharide identities through analysis of oxonium ion fragmentation profiles in LC–MS/MS of glycopeptides. J. Proteome Res. 2014, 13, 6024–6032. [Google Scholar] [CrossRef]
- Rath, C.B.; Schirmeister, F.; Figl, R.; Seeberger, P.H.; Schaffer, C.; Kolarich, D. Flagellin glycoproteomics of the periodontitis associated pathogen Selenomonas sputigena reveals previously not described O-glycans and rhamnose fragment rearrangement occurring on the glycopeptides. Mol. Cell. Proteom. 2018, 17, 721–736. [Google Scholar] [CrossRef] [PubMed]
- Hanumaiah, T.; Marshall, D.S.; Rao, B.K.; Rao, J.U.M.; Rao, K.V.J.; Thomson, R.H. Naphthoquinone-lactones and extended quinones from Ventilago calyculata. Phytochemistry 1985, 24, 2669–2672. [Google Scholar] [CrossRef]
- Asha, R.; Gayathri Devi, V.; Abraham, A. Lupeol, a pentacyclic triterpenoid isolated from Vernonia cinerea attenuate selenite induced cataract formation in Sprague Dawley rat pups. Chem. Biol. Interact. 2016, 245, 20–29. [Google Scholar] [CrossRef]
- Pongjanta, A.; Pangjit, K.; Srichairatanakool, S. Antioxidant activity and cytotoxic effect of Ventilago denticulata Willd leaves extracts. J. Med. Assoc. Thai. 2016, 99 (Suppl. 1), S51–S57. [Google Scholar]
- Pepalla, S.B.; Jammula, S.R.; Rao, K.V.J.; Thomson, R.H. Quinones and tetracosanolide in Ventilago bombaiensis. Phytochemistry 1992, 31, 2103–2104. [Google Scholar] [CrossRef]
- Kaur, A.; Raja, H.A.; Deep, G.; Agarwal, R.; Oberlies, N.H. Pannorin B, a new naphthopyrone from an endophytic fungal isolate of Penicillium sp. Magn. Reson. Chem. 2016, 54, 164–167. [Google Scholar] [CrossRef]
- Vagstad, A.L.; Newman, A.G.; Storm, P.A.; Belecki, K.; Crawford, J.M.; Townsend, C.A. Combinatorial domain swaps provide insights into the rules of fungal polyketide synthase programming and the rational synthesis of non-native aromatic products. Angew. Chem. Int. Ed. Engl. 2013, 52, 1718–1721. [Google Scholar] [CrossRef] [PubMed]
- Chaudhri, A.A.; Nadeem, M.; Rahman, A.U.; Alam, T.; Sajjad, W.; Hasan, F.; Badshah, M.; Khan, S.; Rehman, F.; Shah, A.A. Antioxidative and radioprotective properties of glycosylated flavonoid, xanthorhamnin from radio-resistant bacterium Bacillus indicus strain TMC-6. Curr. Microbiol. 2020, 77, 1245–1253. [Google Scholar] [CrossRef]
- Ammar, R.B.; Bhouri, W.; Sghaier, M.B.; Boubaker, J.; Skandrani, I.; Neffati, A.; Bouhlel, I.; Kilani, S.; Mariotte, A.-M.; Chekir-Ghedira, L.; et al. Antioxidant and free radical-scavenging properties of three flavonoids isolated from the leaves of Rhamnus alaternus L. (Rhamnaceae): A structure-activity relationship study. Food Chem. 2009, 116, 258–264. [Google Scholar] [CrossRef]
- Otsuka, N.; Liu, M.H.; Shiota, S.; Ogawa, W.; Kuroda, T.; Hatano, T.; Tsuchiya, T. Anti-methicillin resistant Staphylococcus aureus (MRSA) compounds isolated from Laurus nobilis. Biol. Pharm. Bull. 2008, 31, 1794–1797. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Zang, X.; He, M.; Pan, S.; Xu, X. Structure–activity relationship of flavonoids on their anti-Escherichia coli activity and inhibition of DNA gyrase. J. Agric. Food Chem. 2013, 61, 8185–8190. [Google Scholar] [CrossRef]
- Ramos, F.A.; Takaishi, Y.; Shirotori, M.; Kawaguchi, Y.; Tsuchiya, K.; Shibata, H.; Higuti, T.; Tadokoro, T.; Takeuchi, M. Antibacterial and antioxidant activities of quercetin oxidation products from yellow onion (Allium cepa) skin. J. Agric. Food Chem. 2006, 54, 3551–3557. [Google Scholar] [CrossRef]
- Wu, D.; Kong, Y.; Han, C.; Chen, J.; Hu, L.; Jiang, H.; Shen, X. D-Alanine:D-alanine ligase as a new target for the flavonoids quercetin and apigenin. Int. J. Antimicrob. Agents 2008, 32, 421–426. [Google Scholar] [CrossRef]
- Hossion, A.M.L.; Zamami, Y.; Kandahary, R.K.; Tsuchiya, T.; Ogawa, W.; Iwado, A.; Sasaki, K. Quercetin diacylglycoside analogues showing dual inhibition of DNA gyrase and topoisomerase IV as novel antibacterial agents. J. Med. Chem. 2011, 54, 3686–3703. [Google Scholar] [CrossRef]
- Obey, J.K.; von Wright, A.; Orjala, J.; Kauhanen, J.; Tikkanen-Kaukanen, C. Antimicrobial activity of Croton macrostachyus stem bark extracts against several human pathogenic bacteria. J. Pathog. 2016, 2016, 1453428. [Google Scholar] [CrossRef]
- del Valle, P.; García-Armesto, M.R.; de Arriaga, D.; González-Donquiles, C.; Rodríguez-Fernández, P.; Rúa, J. Antimicrobial activity of kaempferol and resveratrol in binary combinations with parabens or propyl gallate against Enterococcus faecalis. Food Control 2016, 61, 213–220. [Google Scholar] [CrossRef]
- Bashyal, P.; Parajuli, P.; Pandey, R.P.; Sohng, J.K. Microbial biosynthesis of antibacterial chrysoeriol in recombinant Escherichia coli and bioactivity assessment. Catalysts 2019, 9, 112. [Google Scholar] [CrossRef]
- Nascimento, P.L.; Nascimento, T.C.; Ramos, N.S.; Silva, G.R.; Gomes, J.E.; Falcao, R.E.; Moreira, K.A.; Porto, A.L.; Silva, T.M. Quantification, antioxidant and antimicrobial activity of phenolics isolated from different extracts of Capsicum frutescens (Pimenta Malagueta). Molecules 2014, 19, 5434–5447. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.A. In-vitro antibacterial, antifungal, antibiofilm, and antioxidant potentials of isopimpinellin recovered from Citrullus colocynthis. Int. J. Pharm. Pharm. Sci. 2016, 8, 117–122. [Google Scholar]
- Fawe, A.; Abou-Zaid, M.; Menzies, J.G.; Belanger, R.R. Silicon-mediated accumulation of flavonoid phytoalexins in cucumber. Phytopathology 1998, 88, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Báidez, A.G.; Gómez, P.; Del Río, J.A.; Ortuño, A. Antifungal capacity of major phenolic compounds of Olea europaea L. against Phytophthora megasperma Drechsler and Cylindrocarpon destructans (Zinssm.) Scholten. Physiol. Mol. Plant Pathol. 2006, 69, 224–229. [Google Scholar] [CrossRef]
- Basile, A.; Giordano, S.; Lopez-Saez, J.A.; Cobianchi, R.C. Antibacterial activity of pure flavonoids isolated from mosses. Phytochemistry 1999, 52, 1479–1482. [Google Scholar] [CrossRef]
- Mori, A.; Nishino, C.; Enoki, N.; Tawata, S. Antibacterial activity and mode of action of plant flavonoids against Proteus vulgaris and Staphylococcus aureus. Phytochemistry 1987, 26, 2231–2234. [Google Scholar] [CrossRef]
- Lv, P.C.; Li, H.Q.; Xue, J.Y.; Shi, L.; Zhu, H.L. Synthesis and biological evaluation of novel luteolin derivatives as antibacterial agents. Eur. J. Med. Chem. 2009, 44, 908–914. [Google Scholar] [CrossRef]
- Parmar, V.S.; Vardhan, A.; Nagarajan, G.R.; Jain, R. Dihydroflavonols from Prunus domestica. Phytochemistry 1992, 31, 2185–2186. [Google Scholar] [CrossRef]
- Li, L.; Song, X.; Yin, Z.; Jia, R.; Li, Z.; Zhou, X.; Zou, Y.; Li, L.; Yin, L.; Yue, G.; et al. The antibacterial activity and action mechanism of emodin from Polygonum cuspidatum against Haemophilus parasuis in vitro. Microbiol. Res. 2016, 186–187, 139–145. [Google Scholar] [CrossRef]
- Martini, N.D.; Katerere, D.R.; Eloff, J.N. Biological activity of five antibacterial flavonoids from Combretum erythrophyllum (Combretaceae). J. Ethnopharmacol. 2004, 93, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Hu, J.R.; Zhan, W.Q.; Liu, X. Screening for fractions of Oxytropis falcata Bunge with antibacterial activity. Nat. Prod. Res. 2009, 23, 953–959. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.-S.; Kobayashi, A.; Kajiyama, S.-I.; Kawazu, K.; Kanzaki, H.; Kim, C.-M. Antimicrobial constituents of Angelica dahurica roots. Phytochemistry 1997, 44, 887–889. [Google Scholar] [CrossRef]
- Dorrestein, P.; Wang, M.; Bandeira, N.; Truman, A.W.; Schmid, R.; Jarmusch, A.K.; Aksenov, A.A.; Ndlovu, N.; Tugizimana, F.; Keyzers, R.A.; et al. Reproducible molecular networking of untargeted mass spectrometry data using GNPS. Nat. Protoc. 2020, 15, 1954–1991. [Google Scholar] [CrossRef]


| No. | tR (min) | Compounds | Molecular Formula | Mass | Adduct Ions | Observed m/z | Calculated m/z | Δ (ppm) | Fragment Ions (m/z) | Found in Extracts a |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1.086 | Unidentified | C18 H3N O14 | 487.1895 | (M-H)- | 486.1826 | 486.1828 | 0.50 | 341.1082, 179.0561, 144.0663, 119.0346, 101.0242 | DT |
| 2 | 1.091 | Unidentified | C37H36N2 O11 | 684.2316 | (M-H)- | 683.2244 | 683.2246 | 0.40 | 341.1086, 179.0556, 119.0346 | MB, MT |
| 3 | 1.357 | 2′-Methoxy-3-(2,4-dihydroxyphenyl)-1,2-propanediol 4′-glucoside [M, H] | C16 H24 O9 | 360.1420 | (M+Na)+ | 383.1313 | 383.1313 | −0.01 | 306.9908, 248.9974, 207.0666, 185.0403, 102.0900 | MB, MT |
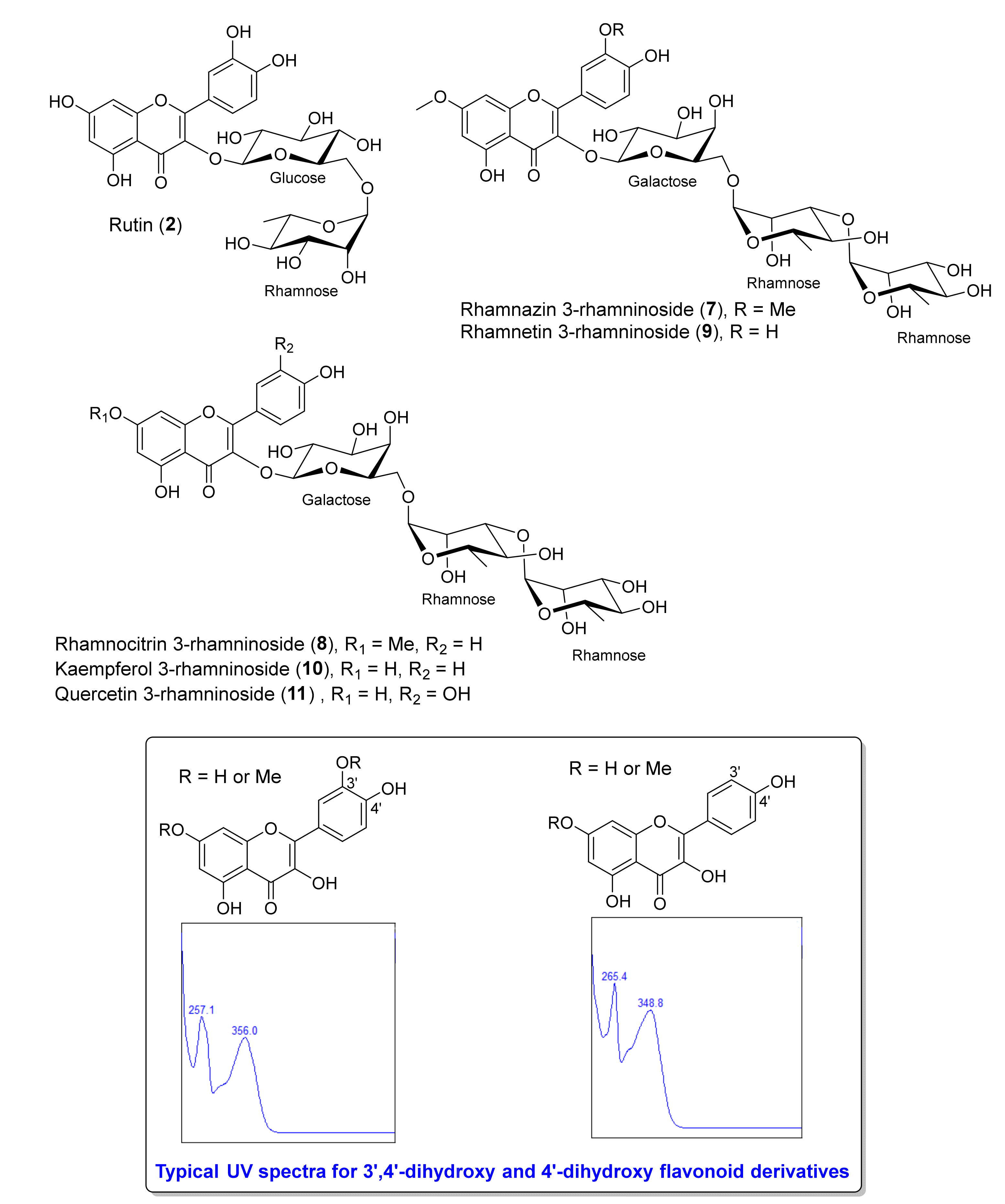
| 4 | 1.371 | Kaempferol-3-rhamninoside [M] | C33 H40 O19 | 740.2162 | (M+H)+ | 741.2233 | 741.2237 | 0.50 | 595.1677, 449.1072, 346.0867, 287.0557, 147.0649 | MB |
| (M-H)- | 739.2080 | 739.2091 | 1.49 | 285.0396, 255.0315 | MB, MT | |||||
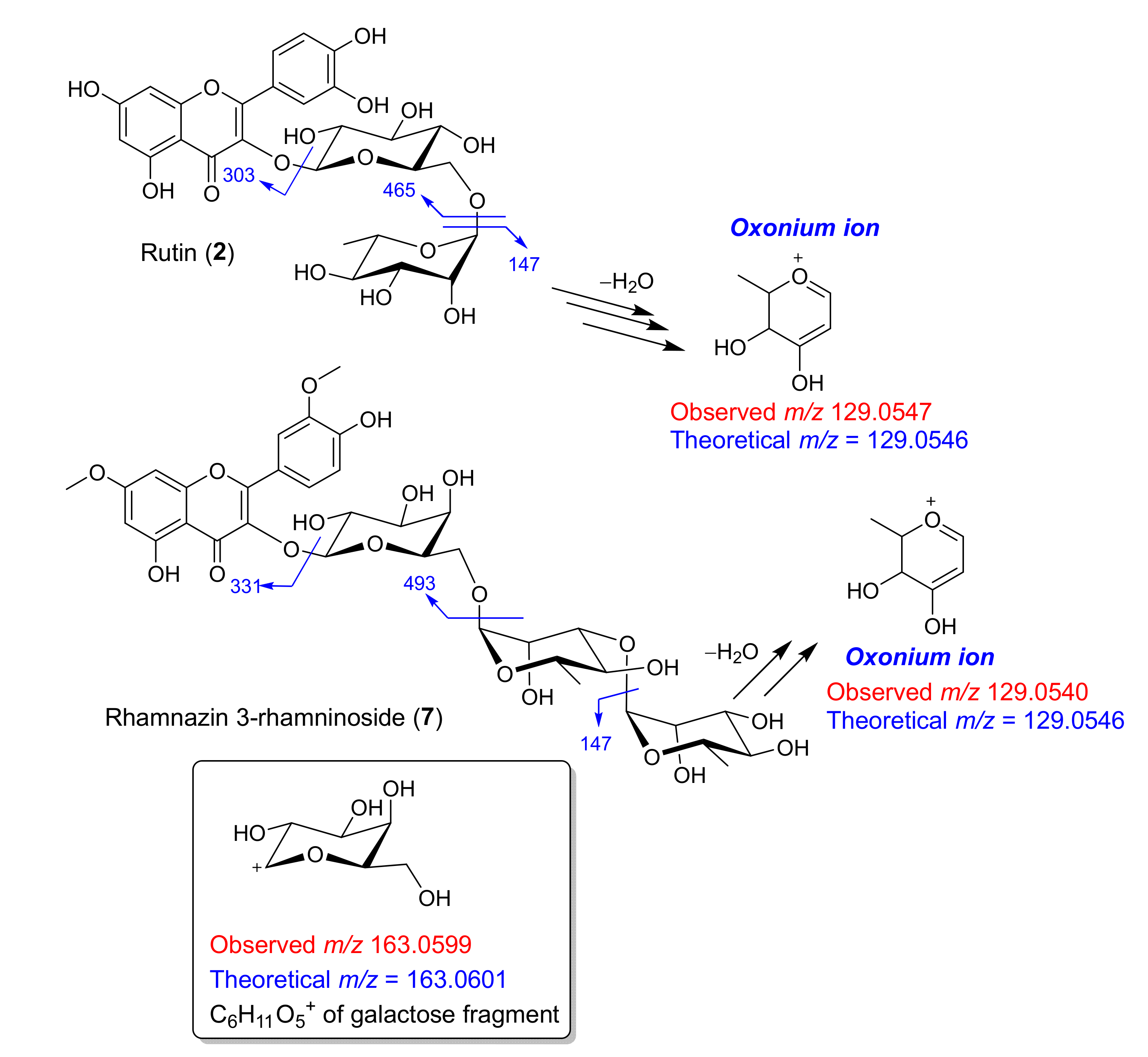
| 5 | 1.405 | Rhamnetin 3-rhamninoside [M] | C34 H42 O20 | 770.2269 | (M+H)+ | 771.2343 | 771.2342 | −0.09 | 479.1186, 317.0657, 239.0928, 163.0602, 147.0653, 129.0548 | MB, MT |
| (M-H)- | 769.2192 | 769.2197 | 0.66 | 315.0505, 299.0186 | MB, MT, DT | |||||
| 6 | 1.414 | 1,2,10-Trihydroxydihydro-trans-linalyl oxide 7-O-β-D-glucopyranoside [M, H] | C16 H30 O10 | 382.1841 | (M+Na)+ | 405.1733 | 405.1731 | −0.54 | 355, 0125, 273.1298, 129.0543 | MB |
| (M-H)- | 381.1762 | 381.1766 | 1.15 | 322.0691, 249.1343, 205.3362, 161.0450, 113.0235, 101.0243 | MB, DB | |||||
| 7 | 1.493 | Rhamnocitrin 3- rhamninoside [M] | C34 H42 O19 | 754.2320 | (M+H)+ | 755.2394 | 755.2393 | −0.19 | 463.1233, 301.0709, 163.0600, 147.0651, 129.0543 | MB, MT, DT |
| (M-H)- | 753.2239 | 753.2248 | 1.12 | 557.2233, 299.0554, 283.0236 | MB, MT, DT | |||||
| 8 | 1.499 | Unidentified | C27 H34 N7 O21 | 792.1803 | (M+2H)+2 | 397.0973 | 397.0977 | 0.97 | 647.1279, 575.1043, 545.1010, 501.0683, 399.0395, 339.0179, 201.0041, 121.0495 | MB, MT |
| 9 | 1.535 | Furocoumarinic acid glucoside [M, H] | C17H18 O9 | 366.0955 | (M+H)+ | 367.1024 | 367.1024 | −0.21 | 349.0928, 331.0806, 307.0803, 289.0703, 275.0556, 263.0559, 217.0494, 161.0594 | DB |
| (M-H)- | 365.0871 | 365.0878 | 1.96 | 350.0639, 306.0746, 289.0707, 274.0482, 246.0522, 161.0181 | DB | |||||
| 10 | 1.540 | Unidentified | C27 H50 Cl2 N9 O8 S | 762.2601 | (M+Na)+ | 785.2493 | 785.2493 | −0.03 | 493.1342, 331.0815, 147.0664 | DB |
| 11 | 1.547 | Unidentified | C29 H30 N16 O11 | 778.2272 | (M+2H)+2 | 412.1027 | 412.1032 | 1.32 | 677.1394, 575.1073, 429.0485, 369.0279, 266.0451, 201.0073, 129.0543 | MB |
| 12 | 1.558 | 3,3′,4′-Trihydroxyflavone 3-O-[α-L-rhamnopyranosyl-(1→2)[α-L-rhamnopyranosyl-(1→6)]-β-D-glucopyranoside] [M, H] | C33 H40 O18 | 724.2206 | (M+CH3COO)- | 783.2345 | 783.2353 | 1.06 | 453.1600, 329.0657, 314.0425, 145.0503, 101.0246 | MB, MT, DB, DT |
| 13 | 1.564 | 5,7,8-Trihydroxyflavanone 7-glucoside [M, H] | C21H22 O10 | 434.1212 | (M-H)- | 433.1138 | 433.1140 | 0.59 | 313.0719, 271.0556, 270.0528, 231.0611, 139.0402 | MB, DT |
| 14 | 1.575 | Rhamnazin 3-rhamninoside [M] | C35 H44 O20 | 784.2430 | (M+H)+ | 785.2503 | 785.2503 | 0.00 | 493.1341, 331.0814, 163.0599, 147. 0653, 129.054 | MB, MT, DB, DT |
| (M-H)- | 783.2349 | 783.2348 | −0.13 | 537.1992, 453.1584, 329.0664, 234.1049, 145.0490 | MB, MT, DB, DT | |||||
| 15 | 1.626 | Astragalin [M, H] | C21 H20 O11 | 448.1009 | (M+H)+ | 449.1081 | 449.1078 | −0.58 | 317.0661, 287.0553, 269.0444, 195.0657 | MB, MT, DT |
| 16 | 1.629 | Unidentified | C23 H30 N7 O8 | 532.2155 | (M+Na)+ | 555.2043 | 555.2048 | 0.88 | 381.1307, 286.0742, 207.0619, 147.0433 | MB |
| 17 | 1.634 | Kaempferol 5-glucoside [M, H] | C21 H20 O11 | 448.0996 | (M+HCOO)- | 493.0979 | 493.0988 | 1.76 | 346.8297, 327.0481, 298.0487, 285.0402, 240.0460 | MB, MT, DT |
| 18 | 1.635 | Naringenin 4′-O-glucuronide [M, H] | C21 H20 O11 | 448.1002 | (M+Na)+ | 471.0895 | 471.0898 | 0.57 | 339.0471, 309.0368, 294.0188, 249.1094, 161.9958 | MB, MT |
| 19 | 1.708 | Aloesol [M, H] | C13 H14 O4 | 234.0891 | (M+H)+ | 235.0964 | 235.0965 | 0.45 | 217.0860, 191.0705, 163.0754, 151.0385, 135.0804, 107.0847 | MB, MT, DT |
| (M-H)- | 233.0818 | 233.1819 | 0.60 | 189.0552, 161.0593, 149.0251 | MB, MT, DB | |||||
| 20 | 1.765 | Zingerone glucoside [M, H] | C17 H24 O8 | 356.1468 | (M+Na)+ | 379.1361 | 379.1363 | 0.63 | 323.9212, 278.3414, 235. 8741, 217.0847, 111.0775 | MB, DB |
| 21 | 1.810 | Unidentified | C33 H46 N4 O6 | 594.3419 | (M+H)+ | 595.3492 | 595.349 | −0.35 | 577.3542, 536.2739, 173.1640, 120.0805 | MB |
| 22 | 1.852 | Unidentified | C21 H28 O8 | 408.1771 | (M+Na)+ | 431.1674 | 431.1676 | 0.53 | 317.1031, 275.0908, 205.0465 | DB |
| 23 | 1.909 | Xanthotoxol glucoside [M, H] | C17 H16 O9 | 364.0795 | (M+H)+ | 365.0866 | 365.0867 | 0.36 | 305.0661, 291.0851, 277.0713, 259.0606, 215.0704, 132.0900 | DB |
| (M-H)- | 363.0716 | 363.0722 | 1.51 | 304.0583, 287.0556, 272.0320, 261.0404, 244.0375, 228.0435, 201.0195 | DB | |||||
| 24 | 2.000 | Unidentified | C24 H18 N8 O4 | 482.1448 | (M+Na)+ | 505.1338 | 505.1343 | 0.96 | 419.1317, 343.1048, 257.0809, 127.0393 | MB |
| 25 | 2.006 | Isoliquiritin [M, H] | C21 H22 O9 | 418.1261 | (M+H)+ | 419.1334 | 419.1337 | 0.71 | 335.0877, 257.0804, 239.0703, 191.0702, 127.0390 | MB |
| (M-H)- | 417.1184 | 417.1191 | 1.66 | 297.0764, 255.0643 | MB, MT, DB | |||||
| 26 | 2.042 | 6”-O-Acetyldaidzin [M, H] | C23 H22 O10 | 458.1209 | (M+HCOO)- | 503.1192 | 503.1195 | 0.67 | 418.1190, 297.0765, 255.0690 | MB |
| 27 | 2.190 | Glucoemodin [M, H] | C21 H20 O10 | 432.1051 | (M-H)- | 431.0979 | 431.0984 | 1.07 | 344.8229, 311.0557, 269.0448, 227.1067 | MB, MT, DT |
| 28 | 2.282 | Kievitol [M, H] | C20 H22 O7 | 374.1356 | (M-H)- | 373.1284 | 373.1293 | 2.29 | 359.0953, 246.0522, 193.0504, 179.0714, 164.0475, 149.0600, 134.0368 | DB |
| 29 | 2.291 | Wharangin [M, H] | C17 H12 O8 | 344.0536 | (M+H)+ | 345.0608 | 345.0605 | −0.74 | 303.0497, 327.0487, 299.0544, 275.0543, 261.0401, 195.0290 | DB, DT |
| (M-H)- | 343.0454 | 343.0459 | 1.69 | 330.0381, 301.0348, 287.0196, 273.0040, 158.0608 | DB | |||||
| 30 | 2.314 | 4″-Methyl-6″-(3,4-dihydroxy-E-cinnamoyl)isoorientin [M, H] | C31 H28 O14 | 624.1474 | (M-H)- | 623.1400 | 623.1406 | 1.02 | 517.8187, 458.3673, 375.3759, 298.0471, 295.0808, 285.0416, 241.0516 | MB |
| 31 | 2.376 | Chrysoeriol [M, H] | C16 H12 O6 | 300.0640 | (M+H)+ | 301.0712 | 301.0707 | −1.80 | 273.0397, 260.0310, 255.0651, 245.0442, 227.0698 | DB |
| (M-H)- | 299.0561 | 299.0561 | 0.00 | 270.0168, 258.0166, 255.0661, 240.0428, 227.0346, 214.0269, 151.0033 | DB | |||||
| 32 | 2.377 | 6”-Malonylcosmosiin [M, H] | C24 H22 O13 | 518.1048 | (M-H)- | 517.0975 | 517.0988 | 2.44 | 473.1078, 432.1734, 385.1729, 269.0452, 225.0402 | MB |
| 33 | 2.382 | Cicerin 7-(6-malonylglucoside) [M, H] | C26 H26 O15 | 578.1269 | (M+H)+ | 579.1341 | 579.1344 | 0.64 | 437.0247, 342.9891, 331.0819, 147.0531, 127.0390 | MB |
| 34 | 2.416 | Unidentified | C40 H38 N O5 S | 644.2467 | (M+Na)+ | 667.2356 | 667.2363 | 1.04 | 553.2780, 425.0864, 329.1411, 129.0528 | MB, DT |
| 35 | 2.445 | Quercetin [M, H] | C15 H10 O7 | 302.0422 | (M+H)+ | 303.0494 | 303.0499 | 1.86 | 276.8345, 240.8436, 229.0471, 195.0268, 182.9751, 139.8692 | MB |
| (M-H)- | 301.0351 | 301.0354 | 0.76 | 273.0382, 229.0518, 178.9980, 151.0032, 121.0300, 107.0132 | MB | |||||
| 36 | 2.552 | Unidentified | C18 H40 N5 O18 | 614.2359 | (M+Na)+ | 637.2247 | 637.2261 | 2.19 | 537.1811, 410.0280, 339.1044, 145.0475, 110.0979 | MB, DT |
| 37 | 2.563 | Emodinanthranol [M, H] | C15 H12 O4 | 256.0738 | (M+H)+ | 257.0810 | 257.0808 | −0.66 | 242.0590, 217.0500, 214.0612, 198.9313, 145.0656, 101.0594 | DB |
| (M-H)- | 255.0658 | 255.0663 | 1.84 | 213.0555, 187.0768, 183.0814 | DB | |||||
| 38 | 2.753 | α-Hydrojuglone 4-O-β-D-glucoside [H] | C13 H18 O5 | 338.0995 | (M-H)- | 337.0922 | 337.0929 | 1.98 | 250.0844, 221.081, 163.0765 | MB, DB, DT |
| 39 | 3.039 | Unidentified | C13 H20N3 O8 S | 378.0958 | (M+H)+ | 379.1025 | 379.1044 | 4.86 | 319.0809, 291.0861, 202.0630, 111.0421 | DB |
| 40 | 3.087 | Unidentified | C17 H18 O8 | 350.1003 | (M+H)+ | 351.1075 | 351.1074 | −0.19 | 333.0949, 301.0702, 276.0630, 259.0604, 215.0700 | DB |
| (M-H)- | 349.0924 | 349.0929 | 1.35 | 334.0694, 319.0457, 291.0506, 219.0304 | DB | |||||
| 41 | 3.131 | Unidentified | C28 H24 O12 | 552.1264 | (M-2H)-2 | 275.0558 | 275.0561 | 1.17 | 338.0072, 262.0703, 232.0368, 218.0236, 188.0462 | DB |
| 42 | 3.233 | Isopimpinellin [M, H] | C13 H10 O5 | 246.0522 | (M+CH3COO)- | 305.0660 | 305.0667 | 2.34 | 245.0447, 201.0512, 173.0585, 129.0714 | DB |
| 43 | 3.274 | Kaempferol [M, H] | C15 H10 O6 | 286.0480 | (M+H)+ | 287.0554 | 287.0550 | −1.39 | 227.8855, 165.0174, 153.0172, 121.0271 | MB, MT |
| (M-H)- | 285.0401 | 285.0405 | 1.37 | 257.0426, 241.0493, 229.0487, 211.0396, 151.0029 | MB, MT, DB | |||||
| 44 | 3.291 | Coriandrone C [M, H] | C13 H10 O5 | 246.0536 | (M+H)+ | 247.0609 | 247.0601 | −3.39 | 229.0499, 219.0262, 201.0552, 173.0586, 158.0695, 137.1239 | DB |
| 45 | 3.364 | Eriodictyol [M, H] | C15 H12 O6 | 288.0631 | (M-H)- | 289.0704 | 289.0707 | 0.84 | 271.0589, 259.0603, 257.0465, 231.0641, 229.0488, 173.0582 | DB |
| (M-H)- | 287.0558 | 287.0561 | 0.93 | 259.0604, 243.0653, 201.0582, 177.0550, 151.0041, 125.0243 | MB, MT, DT | |||||
| 46 | 3.583 | Coumesterol [M, H] | C15 H8 O5 | 268.0373 | (M+H)+ | 269.0446 | 269.0444 | −0.46 | 243.1493, 241.0487, 213.0553, 185.0602, 157.0644 | MB, MT |
| 47 | 3.594 | Citreorosein [M, H] | C15 H10 O6 | 286.0482 | (M+H)+ | 287.0555 | 287.0550 | −1.65 | 269.0447, 213.0536, 185.0593 | MB, MT, DT |
| (M-H)- | 285.0404 | 285.0405 | 0.35 | 241.0503, 172.9762 | MB, MT, DB, DT | |||||
| 48 | 3.608 | Physcion [M] | C16 H12 O5 | 284.0688 | (M+H)+ | 285.0761 | 285.0757 | −1.4 | 257.0808, 243.0644, 239.0696, 229.0496, 211.0750 | DB |
| (M-H)- | 283.0612 | 283.0612 | −0.07 | 255.0650, 241.0503, 239.0703, 227.0345, 224.0477 | DB | |||||
| 49 | 3.698 | R-Angolensin [M] | C16 H16 O4 | 272.1051 | (M+H)+ | 273.1124 | 273.1121 | −0.81 | 255.1016, 231.1015, 227.1068, 189.0915, 174.0667, 111.8671 | DB |
| 50 | 3.894 | (±)-Sphaerosin [M, H] | C17 H18 O5 | 302.1153 | (M+H)+ | 303.1225 | 303.1227 | 0.80 | 285.1117, 261.1129, 257.1174, 219.1029, 204.0783, 163.0361 | MB, DB |
| 51 | 3.919 | Unidentified | C34 H36 O10 | 604.2310 | (M+Na)+ | 627.2204 | 627.2201 | −0.58 | 325.1052 | DB |
| 52 | 3.926 | 3-Hydroxyphloretin [M, H] | C15 H14 O6 | 290.0785 | (M+HCOO)- | 335.0767 | 335.0772 | 1.67 | 268.0917, 259.0604, 248.0686, 220.0728, 205.0504, 147.0429 | MB, DB |
| 53 | 3.996 | 3′,7-Dihydroxy-4′,8-dimethoxyisoflavone [H] | C17 H14 O6 | 314.0785 | (M-H)- | 313.0712 | 313.0718 | 1.82 | 300.0246, 269.0808, 254.0571, 239.0326 | MB, DB |
| 54 | 4.062 | Unidentified | C19 H22 O10 | 410.1214 | (M+Na)+ | 433.1106 | 433.1105 | −0.21 | 401.0840, 369.0571, 341.0618, 250.5698 | MB |
| 55 | 4.064 | Unidentified | C37 H32 N3 O15 | 758.1834 | (M-H)- | 757.1760 | 757.1761 | 0.13 | 713.1893, 458.1202, 410.6138, 373.7386, 299.7235, 254.0514, 191.1313 | MB, MT |
| 56 | 4.075 | Unidentified | C25 H30 N8 O7 | 650.1400 | (M+H)+ | 651.1473 | 651.1472 | −0.03 | 337.0683 | DB |
| 57 | 4.132 | 5,6,7,8-Tetrahydroxy-3′,4′-dimethoxyflavone [M, H] | C17 H14 O8 | 346.0681 | (M-H)- | 345.0610 | 345.0616 | 1.85 | 331.0413, 298.0119, 270.0171, 242.0246 | MB |
| 58 | 4.134 | 5-Hydroxy-4′,7,8-trimethoxyflavone [M, H] | C18 H16 O6 | 328.0939 | (M-H)- | 327.0867 | 327.0874 | 2.06 | 312.0620, 286.0477, 271.0240, 268.0732, 253.0500, 225.0558 | DB |
| 59 | 4.178 | Unidentified | C12 H8 N5 O6 S | 350.0196 | (M+H)+ | 351.0265 | 351.0268 | 0.77 | 297.3586, 261.9442, 245.8488, 222.0035, 181.0472, 135.0783 | MB |
| 60 | 4.203 | Unidentified | C34 H24 O12 | 624.1270 | (M+Na)+ | 647.1162 | 647.1160 | −0.38 | 335.053 | DB |
| 61 | 4.251 | Aloe emodin w-acetate [M, H]; or Ventilatone A (isolation) | C17 H12 O6 | 312.0636 | (M+H)+ | 313.0706 | 313.0707 | 0.29 | 285.0759, 271.0604, 243.0659, 215.0685, 167.8890 | MB, DB |
| (M-H)- | 311.0559 | 311.0561 | 0.72 | 297.0393, 269.0438, 268.0373, 253.0140, 224.0472 | DB | |||||
| 62 | 4.532 | Cartorimine [M, H] | C15 H14 O6 | 290.0794 | (M+Na)+ | 313.0686 | 313.0683 | −1.23 | 276.9105, 212.8751, 123.1149 | MB, MT |
| (M-H)- | 289.0712 | 289.01718 | 1.81 | 273.0402, 259.0239, 245.0457, 201.0550, | MB, MT, DB | |||||
| 63 | 4.619 | Rhamnetin [M] | C16 H12 O7 | 316.0581 | (M+H)+ | 317.0654 | 317.0654 | 0.56 | 271.0590, 243.0679, 167.0342, 121.0279 | MB |
| (M-H)- | 315.0505 | 315.0510 | 1.57 | 300.0261, 166.0221, 121.0293, 112.9849 | MB, DB | |||||
| 64 | 4.721 | Luteolin [M, H] | C15 H10 O6 | 286.0473 | (M-H)- | 285.0401 | 285.04005 | 1.34 | 270.0163, 257.0450, 241.0499, 213.0526, 151.9236 | MB, MT, DB, DT |
| 65 | 4.752 | Unidentified | C18 H14 O7 | 342.0744 | (M+Na)+ | 365.0636 | 365.0632 | −1.14 | 321.0373, 305.0419, 156.0637 | DB |
| 66 | 4.874 | 5,4′-Dihydroxy-3,3′-dimethoxy-6:7-methylenedioxyflavone [M, H] | C18H14O8 | 358.0688 | (M+Na)+ | 381.0579 | 381.0581 | 0.39 | 349.0312, 333.4380, 328.4933, 273.3009, 243.5325, 189.0203 | DB |
| 67 | 4.999 | 1,3,5-Trihydroxy-6,7-dimethoxy-2-methylantraquinone [H] | C16 H10 O7 | 330.0734 | (M-H)- | 329.0661 | 329.0667 | 1.74 | 314.0427, 299.0207, 288.0280, 285.077, 273.0031, 270.0525, 258.0168 | MB, TB, DB, DT |
| 68 | 5.007 | Ventilagodenin A; or 5-De-O-methyltoddanol [M, H] | C15 H16 O5 | 276.1000 | (M+H)+ | 277.1074 | 277.1071 | −1.21 | 259.0957, 244.0731, 235.0973, 199.0748, 171.0804 | MB, TB, DB, DT |
| (M-H)- | 275.0922 | 275.0925 | 1.14 | 259.0609, 245.0447, 231.0661, 192.6885, 175.0355 | MB, TB, DB | |||||
| 69 | 5.097 | Unidentified | C18 H10 N O4 | 304.0612 | (M+Na)+ | 327.0504 | 327.0502 | −0.46 | 287.0555, 259.0604, 255.0288, 245.0422, 167.0345 | DB |
| 70 | 5.129 | Rhamnalpinogenin [M, H]; or Ventilatone B (isolation) | C17 H12 O7 | 328.0589 | (M+H)+ | 329.0659 | 329.0656 | −1.09 | 311.0551, 287.0551, 259.0607, 167.0345 | MB, TB, DB, DT |
| (M-H)- | 327.0508 | 327.0510 | 0.63 | 312.0273, 284.0326, 269.0092, 256.0378, 185.0239 | DB | |||||
| 71 | 5.135 | Unidentified | C12 H8 N5 O7 S | 366.0142 | (M+H)+ | 367.0212 | 367.0217 | 1.49 | 352.3162, 309.0637, 277.0991, 235.8736, 186.9023, 123.1163 | MB |
| 72 | 5.152 | 3,5,7-Trihydroxy-4′,6-dimethoxyflavanone [M, H] | C17 H16 O7 | 332.0891 | (M+HCOO)- | 377.0873 | 377.0878 | 1.30 | 317.0660, 306.0738, 259.0245, 174.9557, 130.9658 | MB, DB |
| 73 | 5.265 | Mukurozidiol (M, H) | C17 H18 O7 | 334.1051 | (M+H)+ | 335.1123 | 335.1125 | 0.77 | 303.0866, 285.0752, 275.0914, 261.0750, 245.0448, 233.0425 | MB, DB, DT |
| (M+HCOO)- | 379.1026 | 379.1035 | 2.23 | 308.0893, 305.0640, 277.0688, 262.0477, 174.9575 | MB, DB | |||||
| 74 | 5.288 | Unidentified | C13 H20 N3 O8 S | 378.0957 | (M+H)+ | 379.1026 | 379.1044 | 4.68 | 364.0528, 291.0863, 215.0331, 115.0550 | MB |
| 75 | 5.296 | Unidentified | C19 H22 O10 | 410.1214 | (M+Na)+ | 433.1106 | 433.1105 | −0.13 | 373.0897, 342.0707, 327.0475 | MB |
| 76 | 5.420 | Genistin [M, H] | C21 H20 O10 | 432.1036 | (M+H)+ | 433.1109 | 433.1129 | 4.76 | 401.0843, 373.0894, 369.0579, 342.0711, 327.0470 | DB |
| 77 | 5.463 | 6′-Hydroxyangolensin [M, H] | C16 H16 O5 | 288.1000 | (M+H)+ | 289.1073 | 289.1071 | −0.92 | 271.0967, 247.0966, 243.1013, 229.0856, 205.0864 | DB |
| (M-H)- | 287.0920 | 287.0925 | 1.70 | 269.0821, 254.0605, 245.0823, 203.0702 | DB | |||||
| 78 | 5.578 | (S)-Rutaretin [M,H] | C14 H14 O5 | 262.0835 | (M-H)- | 261.0761 | 261.0768 | 2.71 | 246.0527, 231.0291, 218.0561, 203.0352 | DB |
| 79 | 5.611 | Unidentified | C35 H30 O11 | 626.1773 | (M+HCOO)- | 671.1753 | 671.177 | 2.60 | 509.1242, 416.1098, 254.0577 | TB, TD |
| 80 | 5.650 | Pratenol A [M,H] | C14 H12 O5 | 260.0687 | (M+H)+ | 261.0759 | 261.0757 | −0.49 | 243.0656, 215.0705, 200.0470, 187.0749, 159.0439 | DB |
| 81 | 5.743 | Gingerenone C [M, H] | C20 H22 O4 | 326.1521 | (M+H)+ | 327.1592 | 327.1591 | −0.46 | 203.1049, 171.0802, 151.0758, 148.1110, 137.0600 | DB |
| 82 | 5.848 | Unidentified | C53 H26 N3 O2 | 736.2027 | (M+Na)+ | 759.1920 | 759.1917 | −0.37 | 664.0398, 504.1286, 418.1196, 299.0856, 256.0729 | MB |
| 83 | 6.222 | Afzelechin [M, H] | C15 H14 O5 | 274.0841 | (M-H)- | 273.0768 | 273.0768 | 0.29 | 229.0501, 202.026 | MB, DB |
| 84 | 6.342 | Ducunolide E [M, H] | C26 H28 O9 | 484.1724 | (M-H)- | 483.1650 | 483.1661 | 2.19 | 468.1412, 439.1389, 424.1156, 409.0887 | DB |
| 85 | 6.472 | Rhamnocitrin [M] | C16 H12 O6 | 300.0637 | (M+H)+ | 301.0711 | 301.0707 | −1.35 | 286.0458, 179.03331, 167.0344, 121.0286 | MB, DB, DT |
| (M-H)- | 299.0556 | 299.0561 | 1.78 | 284.0310, 271.0605, 240.0420, 178.0257, 165.0189 | MB, DB, DT | |||||
| 86 | 6.607 | 7-Hydroxy-3,4′,8-trimethoxyflavone [M, H] | C18 H16 O6 | 328.0949 | (M+H)+ | 329.1023 | 329.1020 | −1.04 | 314.0786, 313.0702, 285.0766, 198.0922, 121.1025 | DB |
| 87 | 6.698 | Acerosin [M, H] | C18 H16 O8 | 360.0834 | (M-H)- | 359.0761 | 359.0772 | 3.11 | 344.0538, 297.0054, 269.0084, 171.2585 | MB, DB |
| 88 | 6.732 | Unidentified | C13 H13 N6 O7 | 365.0841 | (M+2Na)+2 | 205.5309 | 205.5315 | 2.91 | 320.7446, 254.9948, 205.1755, 155.0088, 141. 5110, 112.4964 | MB |
| 89 | 6.766 | Alfalone [M, H] | C17 H14 O5 | 298.0841 | (M+H)+ | 299.0916 | 299.0914 | −0.54 | 271.3851, 213.8909, 189.0528, 112.7128 | DB, DT |
| 90 | 6.775 | Rhamnazin [M, H] | C17 H14 O7 | 330.0743 | (M+H)+ | 331.0816 | 331.0812 | −1.22 | 316.0577, 299.0542, 288,0634, 179.0327, 167.0338 | MB, DB |
| (M-H)- | 329.0664 | 329.0667 | 0.74 | 315.0457, 314.0424, 286.0478, 254.0217, 241.051, 170.0353 | MB, DB | |||||
| 91 | 6.924 | Xanthoxyletin [M, H] | C15 H14 O4 | 258.0894 | (M+H)+ | 259.0967 | 259.0965 | −0.73 | 244.0734, 241.0863, 226.0628, 217.0862, 213.0906, 195.0799, 167.0879 | DB |
| 92 | 6.981 | Barpisoflavone A [M, H] | C16 H12 O6 | 300.0636 | (M+H)+ | 301.0708 | 301.0707 | −0.43 | 287.0570, 269.0441, 236.9047, 185.0603, 127.0056 | MB, TB, DT |
| (M-H)- | 299.0560 | 299.0561 | 0.24 | 267.0297, 240.0422, 212.0476 | MB, TB, DB | |||||
| 93 | 7.015 | (+)-(R)-Ventilagolin [S] | C17 H16 O7 | 332.0897 | (M+H)+ | 333.0971 | 333.0969 | 0.60 | 318.0736, 301.0710, 276.0630, 259.0606, 213.0544, 185.0596 | MB, DB, DT |
| (M+HCOO)- | 377.0873 | 377.0878 | 1.30 | 317.066, 306.0738, 303.0506, 259.0245, 174.9557 | MB, DB, DT | |||||
| 94 | 7.123 | Caryatin [M, H] | C17 H14 O7 | 330.0741 | (M+H)+ | 331.0813 | 331.0812 | −0.12 | 299.0551, 276.0625, 259.0611, 211.3641, 167.0181 | MB, DB |
| (M-H)- | 329.0660 | 329.0667 | 2.00 | 314.0423, 299.0194, 286.0488, 271.0240, 165.0184 | MB, MT, DB, DT | |||||
| 95 | 7.349 | Kanzonol O [M, H] | C22 H22 O6 | 382.1418 | (M+Na)+ | 405.1310 | 405.1309 | −0.29 | 335.0526, 270.0508, 143.0333 | DB |
| 96 | 7.548 | Unidentified | C12 H24 Cl2 N2 O8 S | 426.0635 | (M+Na)+ | 449.0527 | 449.0523 | −0.91 | 408.2483, 388.7627, 287.1038 | MB |
| 97 | 7.887 | Unidentified | C34 H30 N3 O11 | 656.1874 | (M-H)- | 655.1800 | 655.1808 | 1.18 | 557.9872, 254.0580 | MB, MT |
| 98 | 8.003 | Unidentified | C33 H28 N3 O11 | 642.1727 | (M-H)- | 641.1655 | 641.1651 | −0.57 | 509.1224, 491.1100, 254.0579 | MB, MT |
| 99 | 8.120 | Unidentified | C16 H11 N O | 233.0844 | (M+Na)+ | 256.0734 | 256.0733 | −0.36 | 240.0926, 210.0659, 1821.0653, 157.0646, 140.9164 | MB |
| 100 | 8.507 | Dihydromorelloflavone [M, H] | C30 H22 O11 | 558.1161 | (M+H)+ | 559.1236 | 559.1235 | −0.15 | 541.1141, 523.0991, 517.1109, 513.1141, 499.1013, 313.0354, 257.0795 | DB |
| (M-H)- | 557.1085 | 557.1089 | 0.80 | 539.0915, 526.0836, 359.8609, 155.1055 | DB | |||||
| 101 | 8.938 | Emodin [M, H, S] | C15 H10 O5 | 270.0528 | (M+H)+ | 271.0601 | 271.0601 | −0.16 | 229.0509, 225.0560, 201.0539, 197.0590, 140.0222 | MB, MT, DB, DT |
| (M-H)- | 269.0452 | 269.0455 | 1.33 | 241.0511, 225.0562, 210.0316, 195.0415, 135.0911 | MB, MT, DB, DT | |||||
| 102 | 9.187 | Formononetin [M, H] | C16 H12 O4 | 268.0740 | (M+H)+ | 269.0813 | 269.0808 | −1.60 | 254.0572, 239.0708, 226.0618, 151.0543 | DB |
| 103 | 9.383 | 6α-Hydroxymaackiain [M, H] | C16 H12 O6 | 300.0637 | (M+H)+ | 301.0709 | 301.0707 | −0.76 | 255.0638, 117.0696 | MB, DB |
| 104 | 9.986 | Unidentified | C22 H18N7O3 | 428.1473 | (M+Na)+ | 451.1363 | 451.1363 | −0.01 | 319.0570, 292.0353, 133.0864 | DB |
| 105 | 9.991 | Artonin L [M, H] | C22 H20 O7 | 396.1213 | (M+H)+ | 397.1283 | 397.1282 | −0.36 | 379.1160, 366.1054, 337.1045, 327.1201, 295.0939, 287.0557 | DB |
| 106 | 10.699 | Muscomin [M, H] | C18 H18 O7 | 346.1053 | (M+H)+ | 347.1125 | 347.1125 | −0.01 | 332.0896, 315.0864, 290.0781, 273.0764, 227.0696 | DB |
| 107 | 10.824 | Unidentified | C15 H11 O4 | 255.0658 | (M+H)+ | 256.0731 | 256.073 | −0.41 | 241.0502, 238.0625, 210.0683, 198.9302, 182.0727 | MB, MT, DB |
| 108 | 11.423 | 2′,3,5-Trihydroxy-5′,7-dimethoxyflavanone [M, H] | C19 H20 O9 | 332.0885 | (M+CH3COO)- | 391.1024 | 391.1035 | 2.64 | 317.0658, 302.0387, 242.6421, 209.8790, 130.2329 | DB |
| 109 | 11.796 | Palmidin A [M, H] | C30 H22 O8 | 510.1312 | (M+H)+ | 511.1387 | 511.1387 | 0.16 | 256.0733, 133.0854 | MB, MT, DB, DT |
| (M-H)- | 509.1238 | 509.1242 | 0.80 | 254.0583 | MB, MT, DB, DT | |||||
| 110 | 12.237 | 1,3,5,8-Tetrahydroxy-6-methoxy-2- | C16 H12 O7 | 316.0585 | (M+H)+ | 317.0658 | 317.0656 | −0.56 | 299.0575, 254.8649, 193.0125, 135.1168, 127.0534 | MB, DB |
| methylanthraquinone [M, H] | (M-H)- | 315.0505 | 315.0510 | 1.57 | 300.0261, 272.0305, 216.9344, 163.1615, 112.9849 | MB | ||||
| 111 | 12.742 | Khelmarin D [M, H] | C28 H24 O8 | 488.1460 | (M+CH3COO)- | 547.1599 | 547.1610 | 2.00 | 457.0900 | DB |
| 112 | 12.798 | Amentoflavone [M, H] | C30 H18 O10 | 538.0889 | (M-H)- | 537.0814 | 537.0827 | 2.44 | 469.0870, 400.8285, 333.5261, 173.9422, 107.5508 | MB, MT, DB |
| 113 | 12.837 | Isophysalin G [M, H] | C28 H30 O10 | 526.1860 | (M+Na)+ | 549.1752 | 549.1731 | −3.87 | 517.1481, 475.1364, 246.0893 | DB |
| 114 | 13.326 | Yuccaol C [M, H] | C30 H22 O10 | 542.1201 | (M-H)- | 541.1126 | 541.1140 | 2.6 | 523.0998, 511.0683, 493.0539, 308.0347, 231.1206 | DB |
| 115 | 13.632 | Ephedrannin A [M, H] | C30 H20 O11 | 556.0997 | (M+CH3COO)- | 615.1134 | 615.1144 | 1.59 | 299.0208, 289.0709 | DB |
| 116 | 14.355 | Unidentified | C29 H 22 N3 O7 | 524.1462 | (M-H)- | 523.1387 | 523.1385 | −0.47 | 254.0580 | MB |
| 117 | 14.718 | Unidentified | C16 H13 O4 | 269.0814 | (M+H)+ | 270.0885 | 270.0887 | 0.78 | 227.07006, 179.0025, 151.9915, 105.0345 | MB |
| 118 | 14.748 | Palmidin B [M, H] | C30 H22 O7 | 494.1349 | (M-H)- | 493.1279 | 493.1293 | 2.74 | 386.1758, 340.4709, 254.0581, 224.0460, 213.0023, 161.4482 | DB, DT |
| 119 | 16.060 | Murrayazolinine [M, H] | C23 H27 N O2 | 349.2042 | (M+NH4)+ | 367.2390 | 367.2380 | −2.81 | 323.2308, 268.2613, 172.1157, 156.1387, 116.0538 | MB, MT, DB. DT |
| 120 | 16.060 | Unidentified | C32 H28 N2 S3 | 536.1416 | (M+Na)+ | 559.1318 | 559.1307 | −1.94 | 521.0807, 466.7954, 409.8348, 401.2433 | MB |
| 121 | 17.810 | Rheidin B [M, H] | C30 H20 O8 | 508.1146 | (M-H)- | 507.1074 | 507.1085 | 2.24 | 479.1105, 304.9145 | MB, MT, DT |
| 122 | 18.371 | Copalic acid [M, H] | C20 H32 O2 | 304.2407 | (M+H)+ | 305.2479 | 305.2475 | −1.40 | 259.2411, 149.1327, 137.1326, 123.1165, 109.1010 | MB |
| 123 | 20.707 | γ-Pinacene [M, H] | C20 H32 | 272.2506 | (M+H)+ | 273.2578 | 273.2577 | −0.45 | 231.2105, 175.1484, 163.1482, 149.1327, 135.1169, 121.1014, 109.1013, 107.0856 | DB |
| 124 | 20.750 | Pipericine [M, H] | C22 H41 N O | 335.3190 | (M+H)+ | 336.3264 | 336.3261 | −0.90 | 240.2341, 184.1702, 142.1230, 170.1534, 100.0761 | MB, MT, DB, DT |
| 125 | 22.976 | Araliacerebroside [M, H] | C40 H77 N O10 | 731.5543 | (M+Na)+ | 754.5435 | 754.5440 | 0.63 | ND | MB, MT, DT |
| (M-H)- | 730.5462 | 730.5475 | 1.71 | 568.4923, 416.3272, 326.2700, 271.2258, 179.0551, 131.0328, 119.0354 | MB, MT | |||||
| 126 | 23.282 | Unidentified | C26 H51 N13 | 545.4386 | (M+Na)+ | 568.4274 | 568.4283 | 1.52 | 476.3663, 371.2275, 250.1754, 185.1303, 133.0845 | MB |
| 127 | 24.262 | Unidentified | C26 H45 N4 | 413.3639 | (M+H)+ | 414.3710 | 414.3717 | 1.59 | 112.0989 | MB |
| 128 | 24.301 | Unidentified | C26 H49 N O | 391.3819 | (M+H)+ | 392.3893 | 392.3887 | −1.51 | 282.2781, 198.1852, 156.1385, 130.1590 | MB, MT |
| 129 | 24.466 | Unidentified | C36 H38 N4 O5 | 606.2843 | (M+H)+ | 607.2917 | 607.2915 | −0.35 | 547.27 | MB |
| 130 | 24.500 | Clerosterol 3-glucoside [M, H] | C35 H58 O6 | 574.4219 | (M+CH3COO)- | 633.4359 | 633.4372 | 2.00 | 559.3987, 541.3890, 383.3517, 175.0401, 133.0300 | DB |
| 131 | 24.755 | Unidentified | C24 H25 N9 O2 S2 | 535.1579 | (M+H)+ | 536.1658 | 536.1645 | −2.30 | 503.1070, 415.0364, 341.0176, 221.0841, 147.0655 | MB, TB |
| 132 | 24.913 | Unidentified | C37 H38 N5 O2 | 584.3020 | (M+Na)+ | 607.2911 | 607.2918 | 1.17 | 547.2713, 460.2258, 367.0213, 280.2360, 167.1421, 107.0840 | MB |
| 133 | 25.117 | AS 1-5 [M, H] | C40 H77 N O9 | 715.5597 | (M+Na)+ | 738.5489 | 738.5489 | 0.15 | ND | MB, MT, DT |
| (M+HCOO)- | 760.5560 | 760.5580 | 2.69 | 655.7664, 552.4965, 534.4872, 299.4631, 179.0584, 101.0237 | MB, MT | |||||
| 134 | 25.474 | 3-Dehydroteasterone [M, H] | C28 H46 O4 | 446.3401 | (M+Na)+ | 469.3293 | 469.3288 | -0.93 | 385.1727, 329.1716, 189.0170, 171.0054, 113.1314 | MB, MT, DB, DT |
| 135 | 25.552 | Unidentified | C42 H74 N6 O10 | 822.5471 | (M-H)+ | 821.5396 | 821.5394 | -0.32 | 775.5344, 613.0880, 523.3704, 339.4486, 277.2172, 261.1697, 175.6021, 103.9958 | MB |
| 136 | 25.644 | Unidentified | C29 H41 N2 O2 S5 | 609.1765 | (M+H)+ | 610.1843 | 610.1844 | 0.30 | 489.0548, 355.0700, 281.0509, 221.0844, 147.0659 | MB |
| 137 | 26.561 | Secasterone [M, H] | C28 H46 O4 | 446.3395 | (M+Na)+ | 469.3286 | 469.3286 | 0.44 | 329.1732, 284.1760, 268.0679, 109.1008 | MB, MT |
| 138 | 26.727 | Unidentified | C42 H76 N6 O10 | 824.5628 | (M-H)- | 823.5555 | 823.5550 | −0.60 | 778.5514, 713.2510, 657.5735, 579.3840, 513.3079, 456.2245, 388.2563, 277.2178 | MB |
| 139 | 26.799 | Unidentified | C36 H76 N9 O7 S | 778.5595 | (M+Na)+ | 801.5482 | 801.5482 | −0.14 | 519.2919, 121.1020 | MB |
| 140 | 27.173 | Unidentified | C37 H67 N13 O3 | 741.5491 | (M+Na)+ | 764.5381 | 764.5382 | 0.15 | 102.0913 | MB, MT |
| 141 | 29.246 | Unidentified | C22 H48 Cl2 N5 O2 S | 516.2900 | (M+H)+ | 517.2957 | 517.2979 | 4.07 | 312.0957, 244.0374, 175.9745 | MB, MT |
| 142 | 29.348 | Unidentified | C34 H68 | 476.5322 | (M+NH4)+ | 494.5662 | 494.5659 | −0.53 | 453.3644, 271.3170, 151.1298 | MB, MT |
| 143 | 30.655 | Lansiol [M, H] | C33 H56 O | 468.4326 | (M+CH3COO)- | 527.4465 | 527.4470 | 0.91 | 478.6391, 447.7013, 413.8984, 365.2430, 305.1114, 258.1590, 192.0016 | MB |
| 144 | 32.152 | Unidentified | C6 H12 N6 O3 | 216.0977 | (M+H)+ | 217.1049 | 217.1044 | −2.52 | 204.0959, 161.0979, 134.0842, 107.0513 | MB |
| Position | Ventilatone C (16) | |||
|---|---|---|---|---|
| NMR Data in CDCl3 | NMR Data in Acetone-d6 | |||
| δH, Multiplicity (J in Hz) | δC, Type | δH, Multiplicity (J in Hz) | δC, Type | |
| 1 | - | 166.18, C | - | 167.05, C |
| 3 | 4.53, ddq (10.5, 6.3, 3.4) | 74.97, CH | 4.65, ddq (10.6, 6.3, 3.4) | 76.06, CH |
| 4 | 3.00, ddd (16.3, 10.6, 1.4) 3.15, ddd (16.4, 3.3, 0.8) | 34.23, CH2 | 3.03, ddd (16.5, 10.7, 1.6) 3.25, ddd (16.5, 3.1, 0.8) | 34.50, CH2 |
| 4a | - | 127.70, C | - | 129.66, C |
| 5 | 7.24, s | 120.60, CH | 7.43, s | 121.32, CH |
| 5a | - | 138.35, C | - | 139.79, C |
| 6 | 6.72, s | 99.96, CH | 6.94, d (2.3) | 100.52, CH |
| 7 | - | 161.13, C, | - | 161.07, C |
| 8 | 6.72, s | 103.11, CH | 6.64, d (2.3) | 103.44, CH |
| 9 | - | 156.05, C | - | 156.74, C |
| 9a | - | 107.00, C | - | 107.90, C |
| 10 | - | 152.72, C | - | 153.48, C |
| 10a | - | 104.37, C | - | 105.33, C |
| 12 | - | 161.72, C | - | 162.44, C |
| 13 | 5.74, s | 90.81, CH | 5.63, s | 90.82, CH |
| 3-Me | 1.56, d (6.4) | 20.79, CH3 | 1.54, d (6.3) | 20.83, CH3 |
| 7-OMe | 3.91, s | 55.50, CH3 | 3.92, s | 55.88, CH3 |
| 9-OH | 8.81, br s | - | 8.94, s | - |
| Crude Extracts/ Fractions/ Compounds | Zone of Inhibition (mm) | |||||
|---|---|---|---|---|---|---|
| Bacteria/Fungus | ||||||
| B. cereus | S. aureus | E. coli | S. enterica | P. aeruginosa | C. albicans | |
| MB a | 13 | 15 | 8 | 14 | 10 | 13 |
| DB a | 21 | 18 | 9 | 19 | 8 | 16 |
| MT a | 7 | 14 | 8 | 7 | 7 | 8 |
| DT a | 0 | 13 | 0 | 0 | 0 | 0 |
| FM1 b | 11 | 14 | 9 | 0 | 10 | 0 |
| FM2 b | 11 | 12 | 9 | 8 | 8 | 10 |
| FM3 b | 9 | 7 | 12 | 10 | 8 | 0 |
| FM4 b | 0 | 0 | 0 | 14 | 0 | 0 |
| FM5 b | 0 | 0 | 0 | 0 | 0 | 0 |
| FM6 b | 0 | 0 | 9 | 9 | 0 | 0 |
| FD1 c | 18 | 15 | 14 | 18 | 11 | 17 |
| FD2 c | 19 | 22 | 17 | 23 | 9 | 0 |
| FD3 c | 26 | 25 | 25 | 30 | 12 | 0 |
| FD4 c | 17 | 12 | 13 | 24 | 12 | 0 |
| FD5 c | 14 | 15 | 16 | 14 | 9 | 0 |
| FD6 c | 11 | 17 | 11 | 16 | 10 | 9 |
| 7 | 0 | 11 | 0 | 0 | 0 | 8 |
| 8 | 10 | 0 | 11 | 0 | 0 | 12 |
| 9 | 9 | 9 | 0 | 0 | 13 | 6 |
| 10 | 9 | 11 | 12 | 0 | 0 | 0 |
| 11 | 9 | 14 | 10 | 0 | 0 | 0 |
| 12 | 11 | 11 | 0 | 18 | 0 | 12 |
| 13 | ND | 7 | ND | ND | 0 | 0 |
| 15 | 13 | 17 | ND | 18 | 0 | 0 |
| 16 | 13 | 13 | ND | 14 | 0 | 0 |
| Chloramphenicol d | 44 | 37 | 50 | 50 | 28 | ND |
| Tetracycline d | 40 | 39 | 40 | 44 | 29 | ND |
| Amphotericin B e | ND | ND | ND | ND | ND | 23 |
| Fraction | Compounds in Fractions |
|---|---|
| FM1 a | Kaempferol (285.0391 [M-H]−), chrysoeriol (299.0589 [M-H]−), unidentified C13H20N3O8S (377.0851 [M-H]−), kaempferol 3-rhamninoside (739.2091 [M-H]−), isopimpinellin (305.0657 [M+CH3COO]−), 3-hydroxyphloretin (335.0760 [M+HCOO]−), rhamnocitrin 3-rhamninoside (377.0851 [M-H]−), unidentified C37H32N3O15 (757.1768 [M-H]−), rhamnetin 3-rhamninoside (769.2162 [M-H]−), rhamnazin 3-rhamninoside (783.2310 [M-H]−) |
| FM2 a | Rhamnetin (315.0475 [M-H]−), luteolin (285.0391 [M-H]−), 3,5,7-trihydroxy-4′,6-dimethoxyflavanone (377.0846 [M+CH3COO]−) |
| FM3 a | Emodin (269.0445 [M-H]−), rhamnocitrin (299.0563 [M-H]−), palmidin A (509.1218 [M-H]−), unidentified (523.1351 [M-H]−) |
| FD1 b | Eriodyctiol (287.0554 [M-H]−), cartorimine (289.0705 [M-H]−), chrysoeriol (299.0558 [M-H]−), rhamnetin (315.0507 [M-H]−), 3-hydroxyphloretin (335.0771 [M+HCOO]−), xanthotoxol glucoside (363.0709 [M-H]−), furocoumarinic acid glucoside (365.0875 [M-H]−) |
| FD2 b | Ventilagodenin A (275.0846 [M-H]−), physcion (283.0650 [M-H]−), rhamnocitrin (299.0613 [M-H]−), ventilatone A (311.0602 [M-H]−), 3′,7-dihydroxy-4′,8-dimethoxyisoflavone (313.0761 [M-H]−), rhamnazin (329.0726 [M-H]−), 3,5,7-trihydroxy-4′,6-dimethoxyflavanone (331.0803 [M-H]−), ventilatone B (327.0556 [M-H]−), unidentified C17H18O8 (349.0965 [M-H]−) |
| FD3 b | Afzelechin (273.0727 [M-H]−), (+)-(R)-ventilagolin (331.0827 [M-H]−), mukurozidiol (333.0968 [M-H]−) |
| FD4 b | Emodin (269.0450 [M-H]−), 6α-hydroxymaackiain (299.0550 [M-H]−), 2′,3,5-trihydroxy-5′,7-dimethoxyflavanone (331.0816 [M-H]−), palmidin A (509.1261 [M-H]−), unidentified C15H11O4 (254.0592 [M-H]−), unidentified C29H22N3O7 (523.1416 [M-H]−), |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azizah, M.; Pripdeevech, P.; Thongkongkaew, T.; Mahidol, C.; Ruchirawat, S.; Kittakoop, P. UHPLC-ESI-QTOF-MS/MS-Based Molecular Networking Guided Isolation and Dereplication of Antibacterial and Antifungal Constituents of Ventilago denticulata. Antibiotics 2020, 9, 606. https://doi.org/10.3390/antibiotics9090606
Azizah M, Pripdeevech P, Thongkongkaew T, Mahidol C, Ruchirawat S, Kittakoop P. UHPLC-ESI-QTOF-MS/MS-Based Molecular Networking Guided Isolation and Dereplication of Antibacterial and Antifungal Constituents of Ventilago denticulata. Antibiotics. 2020; 9(9):606. https://doi.org/10.3390/antibiotics9090606
Chicago/Turabian StyleAzizah, Muhaiminatul, Patcharee Pripdeevech, Tawatchai Thongkongkaew, Chulabhorn Mahidol, Somsak Ruchirawat, and Prasat Kittakoop. 2020. "UHPLC-ESI-QTOF-MS/MS-Based Molecular Networking Guided Isolation and Dereplication of Antibacterial and Antifungal Constituents of Ventilago denticulata" Antibiotics 9, no. 9: 606. https://doi.org/10.3390/antibiotics9090606
APA StyleAzizah, M., Pripdeevech, P., Thongkongkaew, T., Mahidol, C., Ruchirawat, S., & Kittakoop, P. (2020). UHPLC-ESI-QTOF-MS/MS-Based Molecular Networking Guided Isolation and Dereplication of Antibacterial and Antifungal Constituents of Ventilago denticulata. Antibiotics, 9(9), 606. https://doi.org/10.3390/antibiotics9090606