Measuring Antibiotic Stewardship Programmes and Initiatives: An Umbrella Review in Primary Care Medicine and a Systematic Review of Dentistry
Abstract
:1. Introduction
2. Method
2.1. Protocol and Research Questions
2.2. Search Strategies and Study Selection
2.3. Quality Assessment
2.4. Extraction of Outcome Measures
2.5. Comparing the Outcome Measures
3. Results
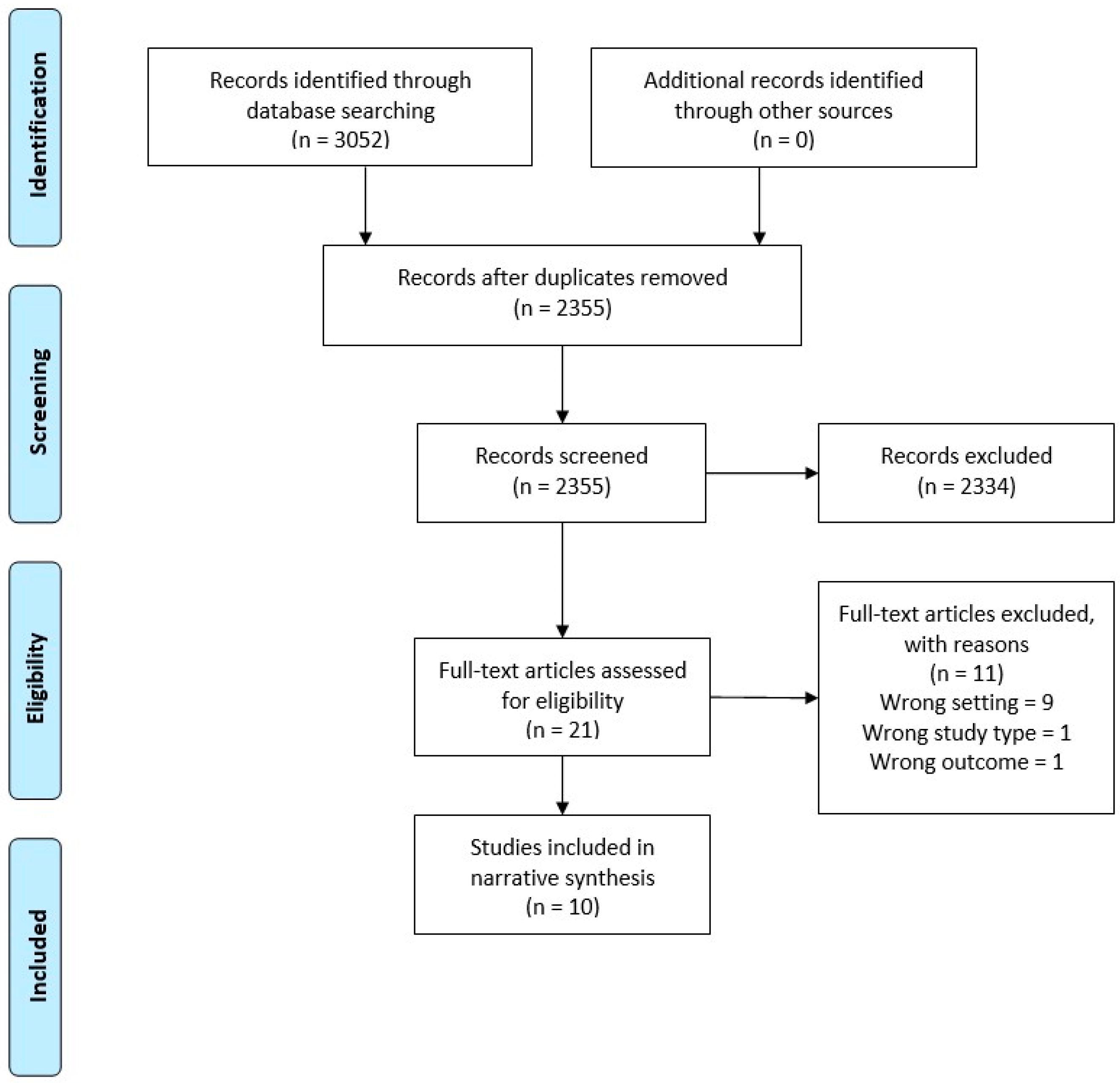
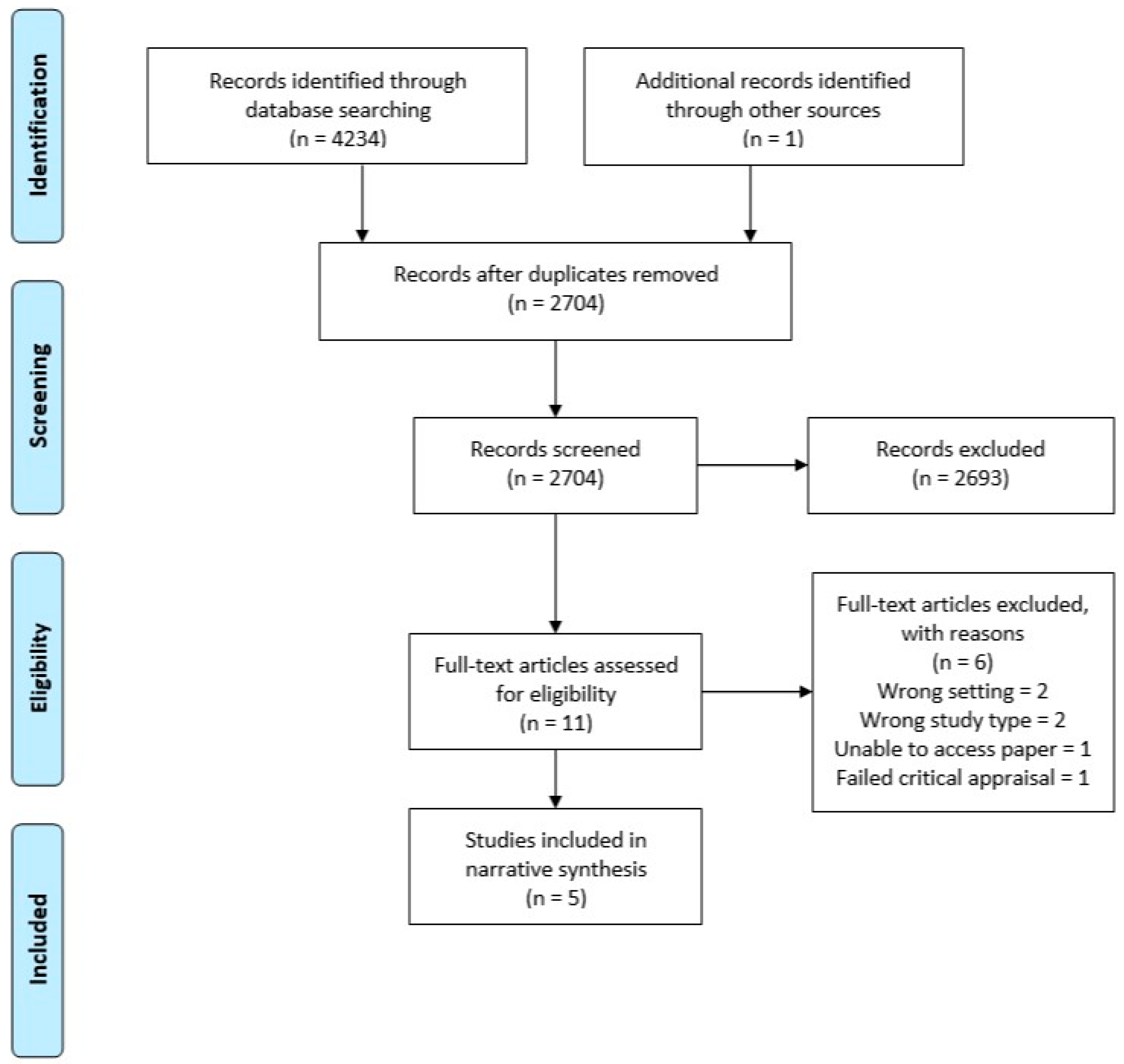
3.1. Study Selection
3.2. Study Characteristics
3.2.1. Umbrella Review across Primary Medical Care
3.2.2. Systematic Review across Primary Dental Care
3.2.3. Outcomes Measured in Primary Medical Care
3.2.4. Outcomes Measured in Primary Dental Care
3.2.5. Comparing the Outcomes Measured between Primary Care Medicine and Dentistry
4. Discussion
4.1. Antibiotic Stewardship and Resistance
4.2. Antibiotic Usage versus Quality of Antibiotic Use
4.3. Clinical and Patient Outcome Measures
4.4. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Economic Forum. Global Risks 2013, 8th ed.; Insight Report; World Economic Forum: Geneva, Switzerland, 2013. [Google Scholar]
- World Health Organisation. Global Action Plan on Antimicrobial Resistance; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- World Health Organisation. Antimicrobial Resistance: A Manual for Developing National Action Plans; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Anderson, M.; Schulze, K.; Cassini, A.; Plachouras, D.; Mossialos, E. A governance framework for development and assessment of national action plans on antimicrobial resistance. Lancet Infect. Dis. 2019, 19, e371–e384. [Google Scholar] [CrossRef] [Green Version]
- Suda, K.J.; Hicks, L.A.; Roberts, R.M.; Hunkler, R.J.; Matusiak, L.M.; Schumock, G.T. Antibiotic Expenditures by Medication, Class, and Healthcare Setting in the United States, 2010–2015. Clin. Infect. Dis. 2018, 66, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Drekonja, D.M.; Filice, G.A.; Greer, N.; Olson, A.; MacDonald, R.; Rutks, I.; Wilt, T.J. Antimicrobial stewardship in outpatient settings: A systematic review. Infect. Control Hosp. Epidemiol. 2015, 36, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Hicks, L.A.; Bartoces, M.G.; Roberts, R.M.; Suda, K.J.; Hunkler, R.J.; Taylor, T.H., Jr.; Schrag, S.J. US outpatient antibiotic prescribing variation according to geography, patient population, and provider specialty in 2011. Clin. Infect. Dis. 2015, 60, 1308–1316. [Google Scholar] [CrossRef] [Green Version]
- FDI World Dental Federation. Antibiotic Stewardship in Dentistry; General Assembly: San Francisco, CA, USA, 2019. [Google Scholar]
- Fiore, D.C.; Fettic, L.P.; Wright, S.D.; Ferrara, B.R. Antibiotic overprescribing: Still a major concern. J. Fam. Pract. 2017, 66, 730–736. [Google Scholar]
- Suda, K.J.; Calip, G.S.; Zhou, J.; Rowan, S.; Gross, A.E.; Hershow, R.C.; Perez, R.I.; McGregor, J.C.; Evans, C.T. Assessment of the Appropriateness of Antibiotic Prescriptions for Infection Prophylaxis Before Dental Procedures, 2011 to 2015. JAMA Netw. Open 2019, 2, e193909. [Google Scholar] [CrossRef] [Green Version]
- Teoh, L.; Marino, R.J.; Stewart, K.; McCullough, M.J. A survey of prescribing practices by general dentists in Australia. BMC Oral Health 2019, 19, 193. [Google Scholar] [CrossRef] [Green Version]
- Kudiyirickal, M.G.; Hollinshead, F. Antimicrobial prescribing practice by dentists: A study from two primary care centres in UK. Minerva Stomatol. 2011, 60, 495–500. [Google Scholar]
- Delannoy, M.; Agrinier, N.; Charmillon, A.; Degand, N.; Dellamonica, J.; Leone, M.; Pulcini, C.; Novy, E. Implementation of antibiotic stewardship programmes in French ICUs in 2018: A nationwide cross-sectional survey. J. Antimicrob. Chemother. 2019, 74, 2106–2114. [Google Scholar] [CrossRef]
- Thompson, W.; Tonkin-Crine, S.; Pavitt, S.H.; McEachan, R.R.C.; Douglas, G.V.A.; Aggarwal, V.R.; Sandoe, J.A.T. Factors associated with antibiotic prescribing for adults with acute conditions: An umbrella review across primary care and a systematic review focusing on primary dental care. J. Antimicrob. Chemother. 2019, 74, 2139–2152. [Google Scholar] [CrossRef] [Green Version]
- Braithwaite, R.S.; Caplan, A. Who is watching the watchmen: Is quality reporting ever harmful? SAGE Open Med. 2014, 2, 2050312114523425. [Google Scholar] [CrossRef] [PubMed]
- Versporten, A.; Gyssens, I.C.; Pulcini, C.; Monnier, A.A.; Schouten, J.; Milanic, R.; Stanic Benic, M.; Tebano, G.; Le Marechal, M.; Zanichelli, V.; et al. Metrics to assess the quantity of antibiotic use in the outpatient setting: A systematic review followed by an international multidisciplinary consensus procedure. J. Antimicrob. Chemother. 2018, 73 (Suppl. 6), vi59–vi66. [Google Scholar] [CrossRef] [PubMed]
- Teoh, L.M.M.; Thompson, W. PROSPERO International Prospective Register of Systematic Reviews. Available online: https://www.crd.york.ac.uk/PROSPERO/display_record.php?RecordID=186364 (accessed on 13 August 2020).
- National Health Service. NHS Primary Care. Available online: https://www.england.nhs.uk/primary-care/ (accessed on 6 July 2020).
- Oxford Centre for Triple Value Healthcare, Critical Appraisal Skills Programme Checklist. Oxford, England. Available online: https://casp-uk.net/wp-content/uploads/2018/01/CASP-Systematic-Review-Checklist_2018.pdf (accessed on 30 June 2020).
- Sirriyeh, R.; Lawton, R.; Gardner, P.; Armitage, G. Reviewing studies with diverse designs: The development and evaluation of a new tool. J. Eval. Clin. Pract. 2012, 18, 746–752. [Google Scholar] [CrossRef] [PubMed]
- Arroll, B.; Kenealy, T.; Kerse, N. Do delayed prescriptions reduce antibiotic use in respiratory tract infections? A systematic review. Br. J. Gen. Pract. 2003, 53, 871–877. [Google Scholar]
- De Bont, E.; Alink, M.; Falkenberg, F.C.J.; Dinant, G.J.; Cals, J.W.L. Patient information leaflets to reduce antibiotic use and reconsultation rates in general practice: A systematic review. BMJ Open 2015, 5, e007612. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.; Chen, R.; Wu, T.; Wei, X.; Guo, A. Association between point-of-care CRP testing and antibiotic prescribing in respiratory tract infections: A systematic review and meta-analysis of primary care studies. Br. J. Gen. Pract. 2013, 63, e787–e794. [Google Scholar] [CrossRef] [Green Version]
- Holstiege, J.; Mathes, T.; Pieper, D. Effects of computer-aided clinical decision support systems in improving antibiotic prescribing by primary care providers: A systematic review. J. Am. Med. Inform. Assoc. 2015, 22, 236–242. [Google Scholar] [CrossRef]
- Hu, Y.H.; Walley, J.; Chou, R.; Tucker, J.D.; Harwell, J.I.; Wu, X.Y.; Yin, J.; Zou, G.Y.; Wei, X.L. Interventions to reduce childhood antibiotic prescribing for upper respiratory infections: Systematic review and meta-analysis. J. Epidemiol. Community Health 2016, 70, 1162–1170. [Google Scholar] [CrossRef] [Green Version]
- Kochling, A.; Loffler, C.; Reinsch, S.; Hornung, A.; Bohmer, F.; Altiner, A.; Chenot, J.F. Reduction of antibiotic prescriptions for acute respiratory tract infections in primary care: A systematic review. Implement. Sci. 2018, 13, 47. [Google Scholar] [CrossRef]
- Lane, I.; Bryce, A.; Ingle, S.M.; Hay, A.D. Does locally relevant, real-time infection epidemiological data improve clinician management and antimicrobial prescribing in primary care? A systematic review. Fam. Pract. 2018, 35, 542–550. [Google Scholar] [CrossRef]
- O’Sullivan, J.W.; Harvey, R.T.; Glasziou, P.P.; McCullough, A. Written information for patients (or parents of child patients) to reduce the use of antibiotics for acute upper respiratory tract infections in primary care. Cochrane Database Syst. Rev. 2016, 11, CD011360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saha, S.K.; Hawes, L.; Mazza, D. Effectiveness of interventions involving pharmacists on antibiotic prescribing by general practitioners: A systematic review and meta-analysis. J. Antimicrob. Chemother. 2019, 74, 1173–1181. [Google Scholar] [CrossRef] [PubMed]
- Vodicka, T.A.; Thompson, M.; Lucas, P.; Heneghan, C.; Blair, P.S.; Buckley, D.I.; Redmond, N.; Hay, A.D. Reducing antibiotic prescribing for children with respiratory tract infections in primary care: A systematic review. Br. J. Gen. Pract. 2013, 63, e445–e454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chate, R.A.; White, S.; Hale, L.R.; Howat, A.P.; Bottomley, J.; Barnet-Lamb, J.; Lindsay, J.; Davies, T.I.; Heath, J.M. The impact of clinical audit on antibiotic prescribing in general dental practice. Br. Dent. J. 2006, 201, 635–641. [Google Scholar] [CrossRef]
- Elouafkaoui, P.; Young, L.; Newlands, R.; Duncan, E.M.; Elders, A.; Clarkson, J.E.; Ramsay, C.R. An Audit and Feedback Intervention for Reducing Antibiotic Prescribing in General Dental Practice: The RAPiD Cluster Randomised Controlled Trial. PLoS Med. 2016, 13, e1002115. [Google Scholar] [CrossRef] [Green Version]
- Palmer, N.A.; Dailey, Y.M.; Martin, M.V. Can audit improve antibiotic prescribing in general dental practice? Br. Dent. J. 2001, 191, 253–255. [Google Scholar] [CrossRef]
- Seager, J.M.; Howell-Jones, R.S.; Dunstan, F.D.; Lewis, M.A.; Richmond, S.; Thomas, D.W. A randomised controlled trial of clinical outreach education to rationalise antibiotic prescribing for acute dental pain in the primary care setting. Br. Dent. J. 2006, 201, 217–222. [Google Scholar] [CrossRef] [Green Version]
- Teoh, L.; Stewart, K.; Marino, R.J.; McCullough, M.J. Improvement of dental prescribing practices using education and a prescribing tool: A pilot intervention study. Br. J. Clin. Pharmacol. 2020. [Google Scholar] [CrossRef]
- Kathy, T. What’s Needed in Second National Action Plan for Combating Superbugs. Available online: https://www.pewtrusts.org/en/research-and-analysis/articles/2020/03/06/whats-needed-in-second-national-action-plan-for-combating-superbugs (accessed on 6 July 2020).
- National Action. U.S. National Action Plan for Combating Antibiotic-Resistant Bacteria (National Action Plan). Available online: https://www.cdc.gov/drugresistance/us-activities/national-action-plan.html (accessed on 6 July 2020).
- Drew, R.H.; White, R.; MacDougall, C.; Hermsen, E.D.; Owens, R.C., Jr. Insights from the Society of Infectious Diseases Pharmacists on antimicrobial stewardship guidelines from the Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America. Pharmacotherapy 2009, 29, 593–607. [Google Scholar] [CrossRef]
- Khadem, T.M.; Ashley, E.D.; Wrobel, M.J.; Brown, J. Antimicrobial stewardship: A matter of process or outcome? Pharmacotherapy 2012, 32, 688–706. [Google Scholar] [CrossRef]
- Teoh, L.S.K.; Marino, R.; McCullough, M. Antibiotic resistance and relevance to general dental practice in Australia. Aust. Dent. J. 2018, 63, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Costelloe, C.; Metcalfe, C.; Lovering, A.; Mant, D.; Hay, A.D. Effect of antibiotic prescribing in primary care on antimicrobial resistance in individual patients: Systematic review and meta-analysis. BMJ 2010, 340, c2096. [Google Scholar] [CrossRef] [Green Version]
- Seppala, H.; Klaukka, T.; Vuopio-Varkila, J.; Muotiala, A.; Helenius, H.; Lager, K.; Huovinen, P. The effect of changes in the consumption of macrolide antibiotics on erythromycin resistance in group A streptococci in Finland. Finnish Study Group for Antimicrobial Resistance. N. Engl. J. Med. 1997, 337, 441–446. [Google Scholar] [CrossRef]
- Australian Commission on Safety and Quality in Health Care. AURA 2019: Third Australian Report on Antimicrobial Use and Resistance in Human Health; ACSQHC: Sydney, Australia, 2019. [Google Scholar]
- WHO Collaborating Centre for Drug Statistics Methodology. ATC/DDD Index 2017. Available online: https://www.whocc.no/atc_ddd_index/ (accessed on 1 August 2017).
- Pulcini, C.; Beovic, B.; Cavalié, P.; Deptula, A.; Dyar, O.; Gyssens, I.; Kern, W.; Knepper, V.; Kofteridis, D.; Hanberger, H.; et al. Amoxicillin dosing recommendations are very different in European countries: A cross-sectional survey. Clin. Microbiol. Infect. 2017, 23, 414–415. [Google Scholar] [CrossRef] [Green Version]
- England, P.H. English Surveillance Programme for Antimicrobial Utilisation and Resistance (ESPAUR); Report 2018–2019; Public Health England: London, UK, 2019. [Google Scholar]
- Australian Government Department of Health. Australia’s National Antimicrobial Resistance Strategy–2020 and Beyond; Creative Commons Licence Attribution CC BY; The Council of the Australian Governments: Canberra, Australia, 2020.
- Sundararajan, K.; Gopaldas, J.A.; Somehsa, H.; Edwards, S.; Shaw, D.; Sambrook, P. Morbidity and mortality in patients admitted with submandibular space infections to the intensive care unit. Anaesth. Intensive Care 2015, 43, 420–422. [Google Scholar]
- Ramsamy, Y.; Hardcastle, T.C.; Muckart, D.J.J. Surviving Sepsis in the Intensive Care Unit: The Challenge of Antimicrobial Resistance and the Trauma Patient. World J. Surg. 2017, 41, 1165–1169. [Google Scholar] [CrossRef]
- Alves, S.; Stella, L.; Carvalho, I.; Moreira, D. Lemierre’s syndrome: A disguised threat. BMJ Case Rep. 2019, 12, e22839. [Google Scholar] [CrossRef]
- Barlam, T.F.; Cosgrove, S.E.; Abbo, L.M.; MacDougall, C.; Schuetz, A.N.; Septimus, E.J.; Srinivasan, A.; Dellit, T.H.; Falck-Ytter, Y.T.; Fishman, N.O.; et al. Implementing an Antibiotic Stewardship Program: Guidelines by the Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America. Clin. Infect. Dis. 2016, 62, e51–e77. [Google Scholar] [CrossRef]
- Ni Riordain, R.; Glick, M.; Al Mashhadani, S.S.A.; Aravamudhan, K.; Barrow, J.; Cole, D.; Crall, J.J.; Gallagher, J.E.; Gibson, J.; Hegde, S.; et al. Developing a standard set of patient-centred outcomes for adult oral health—An international, cross-disciplinary consensus. Int. Dent. J. 2020. [Google Scholar] [CrossRef]
- Teoh, L.; Thompson, W.; McCullough, M. Questioning dual antimicrobial therapy as first line in recent Australian Therapeutic Guidelines. Aust. Dent. J. 2020. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence 2008. Prophylaxis against Infective Endocarditis: Antimicrobial Prophylaxis against Infective Endocarditis in Adults and Children Undergoing Interventional Procedures. Available online: https://www.nice.org.uk/guidance/cg64/resources/endorsed-resource-antibiotic-prophylaxis-against-infective-endocarditis-6535433773 (accessed on 30 June 2020).
- Oral and Dental Expert Group. Therapeutic Guidelines: Oral and Dental Version 3; Therapeutic Guidelines Ltd.: Melbourne, Australia, 2019. [Google Scholar]
- Wilson, W.; Taubert, K.A.; Gewitz, M.; Lockhart, P.B.; Baddour, L.M.; Levison, M.; Bolger, A.; Cabell, C.H.; Takahashi, M.; Baltimore, R.S.; et al. Prevention of infective endocarditis: Guidelines from the American Heart Association: A guideline from the American Heart Association Rheumatic Fever, Endocarditis and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group. J. Am. Dent. Assoc. (1939) 2008, 139, 3S–24S. [Google Scholar]
- Gabbay, J.; le May, A. Evidence based guidelines or collectively constructed “mindlines?” Ethnographic study of knowledge management in primary care. BMJ 2004, 329, 1013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinnock, H.; Barwick, M.; Carpenter, C.R.; Eldridge, S.; Grandes, G.; Griffiths, C.J.; Rycroft-Malone, J.; Meissner, P.; Murray, E.; Patel, A.; et al. Standards for Reporting Implementation Studies (StaRI): Explanation and elaboration document. BMJ Open 2017, 7, e013318. [Google Scholar] [CrossRef] [PubMed]
| Study | Objectives | Number of Databases Searched | Published Date Range | Participants | Number, Type | Outcomes Measured |
|---|---|---|---|---|---|---|
| Arroll et al., 2003 | To conduct a systematic review of the controlled trials of delayed antibiotic prescription for upper respiratory infections | 3 | 1997–2002 | Adults and children | 5 studies; 4 randomised controlled trials, 1 controlled before-after study | Reduction in prescriptions consumed or collected Antibiotics consumed by delayed group and immediate group Adverse effects in delayed group compared to the immediate group (diarrhoea) Antibiotic use by intervention delayed group compared to the control group Symptoms in delayed group compared to immediate group Patient satisfaction, beliefs about antibiotics Time off school, distress |
| de Bont et al., 2015 | To review the effectiveness of information leaflets used for informing patients about common infections during consultations in general practice and if this reduces antibiotic use | 2 | 1981–2013 | Adult and child patients | 8 studies; 1 non-randomised controlled trial, 1 randomised controlled trial, 2 factorial randomised controlled trials; 2 cluster randomised controlled trials, 2 single blinded randomised controlled trials | Antibiotic use Re-consultation rates Severity of symptoms, duration of symptoms, belief in the effectiveness of antibiotics Recall of information Compliance with the prescribed course of antibiotics Patient behaviour (as measured by patient diaries and telephone interviews regarding specific advice given in the consultation after 1 months and 1–2 weeks; telephone questionnaire with the patient or adult (of child patient) after 10–15 days) |
| Huang et al., 2013 | To study the association between family physician use of point-of-care C-reactive protein testing and antibiotic prescribing for respiratory tract infections in general practice | 2 | 1995–2013 | Patients with upper and lower respiratory tract infections | 13 studies in total: 3 cluster randomised controlled trials, 4 parallel randomised controlled trials, 6 observational studies | Antibiotic prescribing rate Antibiotic prescribing at any time during the 28-day follow-up period Patient satisfaction |
| Holstiege et al., 2015 | To assess the effectiveness of computer-aided clinical decision support systems in improving antibiotic prescribing in primary care | 2 | 2001–2013 | Adult and child patients; a wide range of conditions | 7 studies in total: 3 cluster randomised trials, 4 randomised controlled trials | Antibiotic prescribing rates Optimal duration of antibiotic prescriptions Prescribing according to guidelines Reduction in antibiotic prescribing Appropriateness of antibiotic treatment |
| Hu et al., 2016 | To analyse the effectiveness of different intervention approaches, targeted different groups (clinicians, parents or both) and whether other factors (study setting, study design and study period), influence effectiveness for reducing antibiotic prescribing for childhood upper respiratory infections. | 8 | 2001–2013 | Children aged less than or equal to 18 years diagnosed with any upper respiratory infection | 12 studies; 7 cluster randomised controlled trials, 3 non-randomised controlled trials, 2 individual randomised controlled trials | Percentage prescriptions with antibiotic intervention Rate of antimicrobial prescribing per person year Antibiotics prescribing at the index consultation compared to the control group Broad-spectrum prescriptions in intervention compared to control Antibiotic prescribing rate (defined as the number of children who were prescribed one or more antibiotic classes divided by the total number of children assessed for upper respiratory infections during a designated interval) Adjusted antibiotic prescription rate Change in antibiotic prescription rate in intervention compared to control Percentage of patients deciding to use antibiotics Parents administered antibiotics to their children in intervention compared to control Total number of prescriptions in intervention compared to control |
| Kochling et al., 2018 | To summarise the evidence of the effectiveness of interventions in primary care aiming to reduce antibiotic prescriptions in patients for acute respiratory tract infections | 2 | 2006–2016 | Primary care physicians and patients greater than or equal to 13 years old | 17 studies; 13 cluster randomised controlled studies, 4 randomised controlled trials (at patient level) | Antibiotic prescription rates Absolute number of prescribed antibiotics Absolute reductions of antibiotic prescriptions Antibiotic prescriptions according to guidelines Odds ratios for antibiotic prescriptions Number of adverse events (e.g., hospitalisations, deaths) Number of side effects of Ab therapy Difference in emergency room visits/hospitalisation rates Patient satisfaction Re-consultation rates Return rate visit within 30 days after initial consultation in which no antibiotics were prescribed |
| Lane et al., 2018 | To determine whether locally relevant, real-time syndromic or microbiological infection epidemiology could reduce diagnostic uncertainty and improve antibacterial prescribing. | 4 | 1999–2014 | General practitioners, primary care providers, family practice residents, urgent care clinics and community clinics | 12 studies; 11 observational studies, 1 prospective cluster randomised controlled trial | Antibacterial prescribing rates Percentage of cases of antibiotics prescribed |
| O’Sullivan et al., 2016 | To assess if written information for patients (or parents of child patients) reduces antibiotic use for acute upper respiratory tract infections in primary care | 8 | 2000 and 2009 | Children with upper respiratory tract infections; parents were given written information | 2 studies; 2 randomised controlled trials | Antibiotic use Antibiotics used by patients Antibiotics prescribed by clinicians |
| Saha et al., 2019 | To assess the effectiveness of antibiotic stewardship interventions involving pharmacists at improving prescribing by general practitioners | 8 | 1983–2017 | General practitioners | 35 articles for systematic review; 6 randomised controlled trials, 7 cluster randomised controlled trialss, 19 controlled before-after studies, 2 before-after studies and 1 interrupted time-series | Antibiotic prescribing rate (proportion of all patient visits involving prescription of antibiotics by GPs) Antibiotic adherence rate (proportion of antibiotic prescriptions issued to patients that adhered to guidelines/recommendations) Changes to broad-spectrum antibiotic prescribing rate |
| Vodicka et al., 2013 | To review the effectiveness of educational or behavioural interventions directed to parents, clinicians or both, to reduce antibiotic prescribing for children with respiratory tract infections in primary care | 5 | 1992–2011 | Clinicians and parents | 17 studies; 12 randomised design, 3 pre–post test, 2 non-randomised | Antibiotic prescribing rates Antibiotics filled per consultation Antibiotics per person-year for children Exceptions to care pathway per 1000 episodes of care Antibiotics/index consultation Antibiotics per upper respiratory infection episode of care Change in annual antibiotics per 100 patient years Antibiotic rates for penicillin, macrolide and cephalosporin Antibiotics per child with acute otitis media Records of incorrect use of antibiotics per all antibiotics Antibiotics per upper respiratory infection Change in proportion of antibiotics<10 days Change in frequency of antibiotics Number of visits with antibiotics for otitis media or sinusitis Mean change in proportion of consultations resulting in antibiotics Antibiotics/respiratory tract infection consultation Appropriate antibiotics/consultations Incorrect antibiotic order compared to all antibiotic orders Antibiotic adherence to guidelines compared to total antibiotics Proportion/number of consultations resulting in antibiotics Appropriate antibiotics/consultations Antibiotic in adherence to guidelines Antibiotics/respiratory tract infection consultation Adverse events Re-consultation rates |
| Article | Location | Years Study/Timing | Setting | Study Design | Participants | Objective | Outcomes Measured |
|---|---|---|---|---|---|---|---|
| Chate et al., 2006 | England | 2002–2004 | General dental practices | Pre–post | Dentists | To use the intervention of education and prescribing guidelines to reduce the number of antibiotics inappropriately prescribed by general dental practitioners, and to increase overall prescription accuracy. | Number of antibiotics according to guidelines (dose, frequency and duration) Numbers of antibiotics prescribed for specific clinical indications Numbers of prophylactic antibiotics prescribed for specific medical conditions |
| Elouaflaoui et al., 2016 | Scotland | 2012–2013 | General dental practices | Prospective; 1 control group and 2 intervention groups | Dentists in NHS practices in Scotland | To compare the effectiveness of individualised audit and feedback interventions for the translation into practice of national guidance recommendations on antibiotic prescribing. A secondary objective was to explore dentists’ experiences of and responses to the individualised A&F interventions and to increase understanding of the factors associated dental antibiotic prescribing. | Antibiotic prescribing rate: Primary outcome: Total number of claims Total number and DDD of antibiotics Total number and DDD of amoxicillin 3g sachets Total number and DDD of broad-spectrum antibiotics Secondary outcomes: Total DDD of antibiotics/100 claims Total number of amoxicillin 3g sachets/100 claims Total DDD of amoxicillin 3g sachets/100 claims Total number of broad-spectrum antibiotics/100 claims Total DDD of broad-spectrum antibiotics/100 claims |
| Palmer et al., 2001 | England | Could not find date | General dental practices | Pre–post | Dentists | To investigate if clinical audit, with an intervention of education and guidelines, can improve general dental practitioners’ antibiotic prescribing | Total numbers of antibiotics Numbers of antibiotic divided by type Numbers of antibiotics prescribed for specific medical conditions Numbers of antibiotics prescribed for specific clinical indications Number of antibiotics according to guidelines (dose, frequency and duration) |
| Seager et al., 2006 | Wales | Could not find date | Primary care general dental practices | Randomised controlled trial | Dentists | To investigate if guidelines, or guidelines and education will improve antibiotic prescribing compared to a control group. | Number and percentage of antibiotic prescriptions for patients with dental pain Number and percentage of inappropriate antibiotic prescriptions (defined as the provision of an antibiotic to a patient who did not present with a symptom indicative of spreading infection) |
| Teoh et al., 2020 | Melbourne, Australia | 2019 | General dental practices | Pre–post | Dentists | The aim of this pilot study was to evaluate a multimodal intervention to improve dental prescribing. The intervention comprised two parts: Targeted education about drug use in dentistry and an online prescribing tool. | Number of antibiotic prescriptions before and after the intervention Number of inappropriate indications for antibiotic prescription before and after the intervention Accuracy of the prescriptions according to the Australian therapeutic guidelines before and after the intervention Confidence and attitude of practitioners towards the online dental prescribing tool. |
| Outcome Measures | Primary Medical Care | Primary Dental Care |
|---|---|---|
| Quantity of antibiotic use | Absolute numbers of prescribed antibiotics (in intervention compared to control groups) Antibiotics used by patients Antibiotic use (odds ratios) Percentage of patients deciding to use antibiotics Reduction in prescribing/prescriptions consumed or collected | Numbers of antibiotics (total, type) Number of antibiotic prescriptions before and after the intervention |
| Rate of antibiotic use | Antibiotic prescribing rate (total, type, by consultation, by patient, per person-year) Antibiotic prescribing at the index consultation compared to the control group Change in antibiotic prescription rate in intervention compared to control group Changes to broad-spectrum antibiotic prescribing rate Change in proportion/frequency of antibiotics | Antibiotic prescribing rate using/100 claims Number/percentage of antibiotics per dental pain visit Total number of claims/100 claims Total DDD of antibiotics/100 claims (total, type) Total number of broad-spectrum antibiotics/100 claims Total DDD of broad-spectrum antibiotics/100 claims |
| Quality of antibiotic use (appropriateness, appropriate indications and accuracy of prescriptions according to guidelines) | Antibiotics prescribed in adherence to guidelines Antibiotic adherence rate/patient compliance (proportion of antibiotic prescriptions that adhered to guidelines/recommendations) Appropriateness of antibiotic treatment (indications) Broad-spectrum prescriptions in intervention compared to control Optimal duration of antibiotic prescriptions Records of incorrect use of antibiotics per all antibiotics | Number of antibiotics according to guidelines (dose, frequency and duration) Number/percentage of inappropriate indications for antibiotic prescription Numbers of antibiotics prescribed for specific medical/clinical indications |
| Confidence of clinicians towards prescribing | Confidence and attitude of practitioners towards the online dental prescribing tool. | |
| Consultation/Re-consultation rates | Appropriate antibiotics/consultations (proportion, number) Antibiotic prescribing at any time during a specified follow-up period Number of visits with antibiotics for specific clinical conditions Mean change in proportion of consultations resulting in antibiotics Re-consultation rates/return rate visit | |
| Adverse effects | Number of adverse effects relating to antibiotic use Number of adverse events (e.g., emergency room visits, hospitalisations, deaths) Time off school, distress | |
| Severity of symptoms | Presence, severity and duration of symptoms, belief in the effectiveness of antibiotics | |
| Other patient outcomes | Patient satisfaction Patient behaviour |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teoh, L.; Sloan, A.J.; McCullough, M.J.; Thompson, W. Measuring Antibiotic Stewardship Programmes and Initiatives: An Umbrella Review in Primary Care Medicine and a Systematic Review of Dentistry. Antibiotics 2020, 9, 607. https://doi.org/10.3390/antibiotics9090607
Teoh L, Sloan AJ, McCullough MJ, Thompson W. Measuring Antibiotic Stewardship Programmes and Initiatives: An Umbrella Review in Primary Care Medicine and a Systematic Review of Dentistry. Antibiotics. 2020; 9(9):607. https://doi.org/10.3390/antibiotics9090607
Chicago/Turabian StyleTeoh, Leanne, Alastair J Sloan, Michael J McCullough, and Wendy Thompson. 2020. "Measuring Antibiotic Stewardship Programmes and Initiatives: An Umbrella Review in Primary Care Medicine and a Systematic Review of Dentistry" Antibiotics 9, no. 9: 607. https://doi.org/10.3390/antibiotics9090607
APA StyleTeoh, L., Sloan, A. J., McCullough, M. J., & Thompson, W. (2020). Measuring Antibiotic Stewardship Programmes and Initiatives: An Umbrella Review in Primary Care Medicine and a Systematic Review of Dentistry. Antibiotics, 9(9), 607. https://doi.org/10.3390/antibiotics9090607






