Activity of Temporin A and Short Lipopeptides Combined with Gentamicin against Biofilm Formed by Staphylococcus aureus and Pseudomonas aeruginosa
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains and Culture Conditions
4.2. Antimicrobials
4.3. Minimum Inhibitory Concentration (MIC)
4.4. Fractional Inhibitory Concentration (FIC)
4.5. Minimum Biofilm Eradication Concentration (MBEC)
4.6. Activity of Gentamicin Applied in Combination with AMPs against Biofilms Formed by SA and PA on 96-well PS
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hasan, A.; Waibhaw, G.; Tiwari, S.; Dharmalingam, K.; Shukla, I.; Pandey, L.M. Fabrication and characterization of chitosan, polyvinylpyrrolidone, and cellulose nanowhiskers nanocomposite films for wound healing drug delivery application. J. Biomed. Mater. Res. A 2017, 9, 2391–2404. [Google Scholar] [CrossRef]
- Lau, K.H.; Ren, C.; Park, S.H.; Szleifer, I.; Messersmith, P.B. An experimental-theoretical analysis of protein adsorption on peptidomimetic polymer brushes. Langmuir 2012, 2, 2288–2298. [Google Scholar] [CrossRef] [Green Version]
- Bryers, J.D. Medical biofilms. Biotechnol. Bioeng. 2008, 100, 1–18. Available online: https://onlinelibrary.wiley.com/doi/epdf/10.1002/bit.21838 (accessed on 20 April 2020). [CrossRef]
- Hasan, A.P.S.; Pandey, L.M. Effect of Functional Groups of Self-Assembled Monolayers on Protein Adsorption and Initial Cell Adhesion. ACS Biomater. Sci. Eng. 2018, 4, 3224–3333. [Google Scholar] [CrossRef]
- Potera, C. ANTIBIOTIC RESISTANCE: Biofilm Dispersing Agent Rejuvenates Older Antibiotics. Environ. Health Perspect. 2010, 118, A288. [Google Scholar] [CrossRef]
- Chromek, M.; Slamova, Z.; Bergman, P.; Kovacs, L.; Podracka, L.; Ehren, I.; Hökfelt, T.; Gudmundsson, G.H.; Gallo, R.L.; Agerberth, B. The antimicrobial peptide cathelicidin protects the urinary tract against invasive bacterial infection. Nat. Med. 2006, 6, 636–641. [Google Scholar] [CrossRef] [PubMed]
- Harder, J.; Schroder, J.M. Antimicrobial peptides in human skin. Chem. Immunol. Allergy 2005, 86, 22–41. [Google Scholar] [PubMed]
- McDermott, A.M. The role of antimicrobial peptides at the ocular surface. Ophthalmic Res. 2009, 2, 60–75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gorr, S.U. Antimicrobial peptides in periodontal innate defense. Front. Oral Biol. 2012, 15, 84–98. [Google Scholar]
- Devine, D.A. Antimicrobial peptides in defence of the oral and respiratory tracts. Mol. Immunol. 2003, 7, 431–443. [Google Scholar] [CrossRef]
- Carriel-Gomes, M.C.; Kratz, J.M.; Barracco, M.A.; Bachere, E.; Barardi, C.R.; Simoes, C.M. In vitro antiviral activity of antimicrobial peptides against herpes simplex virus 1, adenovirus, and rotavirus. Mem. Inst. Oswaldo Cruz 2007, 4, 469–572. [Google Scholar] [CrossRef]
- Bahar, A.A.; Ren, D. Antimicrobial peptides. Pharmaceuticals 2013, 12, 1543–1575. [Google Scholar] [CrossRef] [Green Version]
- Steckbeck, J.D.; Deslouches, B.; Montelaro, R.C. Antimicrobial peptides: New drugs for bad bugs? Expert Opin. Biol. 2014, 1, 11–14. [Google Scholar] [CrossRef] [Green Version]
- Matejuk, A.; Leng, Q.; Begum, M.D.; Woodle, M.C.; Scaria, P.; Chou, S.T.; Mixson, A.J. Peptide-based Antifungal Therapies against Emerging Infections. Drugs Future 2010, 3, 197. [Google Scholar] [CrossRef] [Green Version]
- Scott, M.G.; Vreugdenhil, A.C.; Buurman, W.A.; Hancock, R.E.; Gold, M.R. Cutting edge: Cationic antimicrobial peptides block the binding of lipopolysaccharide (LPS) to LPS binding protein. J. Immunol. 2000, 2, 549–553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sorensen, O.E.; Borregaard, N.; Cole, A.M. Antimicrobial peptides in innate immune responses. Contrib. Microbiol. 2008, 15, 61–77. [Google Scholar] [PubMed]
- Nijnik, A.; Hancock, R. Host defence peptides: Antimicrobial and immunomodulatory activity and potential applications for tackling antibiotic-resistant infections. Emerg. Health Threats J. 2009, 2, e1. [Google Scholar] [PubMed]
- Marr, A.K.; Gooderham, W.J.; Hancock, R.E. Antibacterial peptides for therapeutic use: Obstacles and realistic outlook. Curr. Opin. Pharmacol. 2006, 5, 468–472. [Google Scholar] [CrossRef]
- Hancock, R.E.; Sahl, H.G. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat. Biotechnol. 2006, 12, 1551–1557. [Google Scholar] [CrossRef]
- Rabadia, A.K.; Kamat, D.V. Study of synergistic action of cefotaxime and Terminalia chebula on Acinetobacter baumannii using checkerboard Assay. Int. J. Pharm. Pharm. Sci. 2019, 3, 830–832. [Google Scholar]
- Characterization of Genatmycin, Garamycin, Dexamytrex, Genatmycin KRKA, Gentamycin WZF, Bedicort, G. Triderm. Available online: http.//www.pharmindex.pl (accessed on 6 June 2020).
- USP, editor. Gentamicin Sulfate-PRODUCT DATA SHEET. Version 2.0 ed2010. Available online: https://toku-e.com/gentamicin-sulfate-usp/ (accessed on 6 June 2020).
- Wang, L.; Di Luca, M.; Tkhilaishvili, T.; Trampuz, A.; Gonzalez Moreno, M. Synergistic Activity of Fosfomycin, Ciprofloxacin, and Gentamicin Against Escherichia coli and Pseudomonas aeruginosa Biofilms. Front. Microbiol. 2019, 10, 2522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giacometti, A.; Cirioni, O.; Kamysz, W.; D’Amato, G.; Silvestri, C.; Del Prete, M.S.; Licci, A.; Łukasiak, J.; Scalise, G. In vitro activity and killing effect of temporin A on nosocomial isolates of Enterococcus faecalis and interactions with clinically used antibiotics. J. Antimicrob. Chemother. 2005, 2, 272–274. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, Y.; Barra, D.; Simmaco, M.; Shai, Y.; Mangoni, M.L. A synergism between temporins toward Gram-negative bacteria overcomes resistance imposed by the lipopolysaccharide protective layer. J. Biol. Chem. 2006, 39, 28565–28574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avrahami, D.; Shai, Y. A new group of antifungal and antibacterial lipopeptides derived from non-membrane active peptides conjugated to palmitic acid. J. Biol. Chem. 2004, 13, 12277–12285. [Google Scholar] [CrossRef] [Green Version]
- Shai, Y.; Makovitzky, A.; Avrahami, D. Host defense peptides and lipopeptides: Modes of action and potential candidates for the treatment of bacterial and fungal infections. Curr. Protein Pept. Sci. 2006, 6, 479–486. [Google Scholar] [CrossRef]
- Bionda, N.; Fleeman, R.M.; de la Fuente-Nunez, C.; Rodriguez, M.C.; Reffuveille, F.; Shaw, L.N.; Pastar, I.; Davis, S.C.; Hancock, R.E.; Cudic, P. Identification of novel cyclic lipopeptides from a positional scanning combinatorial library with enhanced antibacterial and antibiofilm activities. Eur. J. Med. Chem. 2016, 108, 354–363. [Google Scholar] [CrossRef] [Green Version]
- Costerton, W.; Veeh, R.; Shirtliff, M.; Pasmore, M.; Post, C.; Ehrlich, G. The application of biofilm science to the study and control of chronic bacterial infections. J. Clin. Investig. 2003, 10, 1466–1477. [Google Scholar] [CrossRef] [Green Version]
- Jorge, P.; Grzywacz, D.; Kamysz, W.; Lourenco, A.; Pereira, M.O. Searching for new strategies against biofilm infections: Colistin-AMP combinations against Pseudomonas aeruginosa and Staphylococcus aureus single- and double-species biofilms. PLoS ONE 2017, 3, e0174654. [Google Scholar] [CrossRef] [Green Version]
- Grassi, L.; Maisetta, G.; Esin, S.; Batoni, G. Combination Strategies to Enhance the Efficacy of Antimicrobial Peptides against Bacterial Biofilms. Front. Microbiol. 2017, 8, 2409. [Google Scholar] [CrossRef]
- Galdiero, E.; Lombardi, L.; Falanga, A.; Libralato, G.; Guida, M.; Carotenuto, R. Biofilms: Novel Strategies Based on Antimicrobial Peptides. Pharmaceutics 2019, 11, 322. [Google Scholar] [CrossRef] [Green Version]
- Park, S.C.; Park, Y.; Hahm, K.S. The role of antimicrobial peptides in preventing multidrug-resistant bacterial infections and biofilm formation. Int. J. Mol. Sci. 2011, 12, 5971–5992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pletzer, D.; Coleman, S.R.; Hancock, R.E. Anti-biofilm peptides as a new weapon in antimicrobial warfare. Curr. Opin. Microbiol. 2016, 33, 35–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de la Fuente-Nunez, C.; Cardoso, M.H.; de Souza Candido, E.; Franco, O.L.; Hancock, R.E. Synthetic antibiofilm peptides. Biochim. Biophys. Acta 2016, 5, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Di Luca, M.; Maccari, G.; Nifosi, R. Treatment of microbial biofilms in the post-antibiotic era: Prophylactic and therapeutic use of antimicrobial peptides and their design by bioinformatics tools. Pathog. Dis. 2014, 3, 257–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dawgul, M.; Baranska-Rybak, W.; Kamysz, E.; Karafova, A.; Nowicki, R.; Kamysz, W. Activity of short lipopeptides and conventional antimicrobials against planktonic cells and biofilms formed by clinical strains of Staphylococcus aureus. Future Med. Chem. 2012, 12, 541–551. [Google Scholar]
- Dawgul, M.; Maciejewska, M.; Jaskiewicz, M.; Karafova, A.; Kamysz, W. Antimicrobial peptides as potential tool to fight bacterial biofilm. Acta Pol. Pharm. 2014, 1, 39–47. [Google Scholar]
- Shapiro, J.A.; Nguyen, V.L.; Chamberlain, N.R. Evidence for persisters in Staphylococcus epidermidis RP62a planktonic cultures and biofilms. J. Med. Microbiol. 2011, 60, 950–960. [Google Scholar] [CrossRef]
- Paduszynska, M.A.; Maciejewska, M.; Greber, K.E.; Sawicki, W.; Kamysz, W. Antibacterial Activities of Lipopeptide (C10)(2)-KKKK-NH(2) Applied Alone and in Combination with Lens Liquids to Fight Biofilms Formed on Polystyrene Surfaces and Contact Lenses. Int. J. Mol. Sci. 2019, 2, 393. [Google Scholar] [CrossRef] [Green Version]
- Paduszynska, M.A.; Maciejewska, M.; Neubauer, D.; Golacki, K.; Szymukowicz, M.; Bauer, M.; Kamysz, W. Influence of Short Cationic Lipopeptides with Fatty Acids of Different Chain Lengths on Bacterial Biofilms Formed on Polystyrene and Hydrogel Surfaces. Pharmaceutics 2019, 11, 506. [Google Scholar] [CrossRef] [Green Version]
- Baranska-Rybak, W.; Cirioni, O.; Dawgul, M.; Sokolowska-Wojdylo, M.; Naumiuk, L.; Szczerkowska-Dobosz, A.; Nowicki, R.; Roszkiewicz, J.; Kamysz, W. Activity of Antimicrobial Peptides and Conventional Antibiotics against Superantigen Positive Staphylococcus aureus Isolated from the Patients with Neoplastic and Inflammatory Erythrodermia. Chemother. Res. Pract. 2011, 2011, 270932. [Google Scholar] [CrossRef]
- Humphries, R.M.; Pollett, S.; Sakoulas, G. A current perspective on daptomycin for the clinical microbiologist. Clin. Microbiol. Rev. 2013, 4, 759–780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dawgul, M.; Baranska-Rybak, W.; Piechowicz, L.; Bauer, M.; Neubauer, D.; Nowicki, R.; Kamysz, W. The Antistaphylococcal Activity of Citropin 1.1 and Temporin A against Planktonic Cells and Biofilms Formed by Isolates from Patients with Atopic Dermatitis: An Assessment of Their Potential to Induce Microbial Resistance Compared to Conventional Antimicrobials. Pharmaceuticals 2016, 2, 30. [Google Scholar]
- Baranska-Rybak, W.; Pikula, M.; Dawgul, M.; Kamysz, W.; Trzonkowski, P.; Roszkiewicz, J. Safety profile of antimicrobial peptides: Camel, citropin, protegrin, temporin a and lipopeptide on HaCaT keratinocytes. Acta Pol. Pharm. 2013, 5, 795–801. [Google Scholar]
- Kamysz, W.; Silvestri, C.; Cirioni, O.; Giacometti, A.; Licci, A.; Della Vittoria, A.; Okroj, M.; Scalise, G. In vitro activities of the lipopeptides palmitoyl (Pal)-Lys-Lys-NH(2) and Pal-Lys-Lys alone and in combination with antimicrobial agents against multiresistant gram-positive cocci. Antimicrob. Agents Chemother. 2007, 1, 354–358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidt, N.W.; Deshayes, S.; Hawker, S.; Blacker, A.; Kasko, A.M.; Wong, G.C. Engineering persister-specific antibiotics with synergistic antimicrobial functions. ACS Nano 2014, 9, 8786–8793. [Google Scholar] [CrossRef] [Green Version]
- Shai, Y.A.D. No. 8445636, inventor Antimicrobial and Anticancer Lipopeptides2017. Available online: https://patentscope.wipo.int/search/en/detail.jsf?docId=US43767459 (accessed on 14 April 2020).
- Lebeaux, D.; Ghigo, J.M.; Beloin, C. Biofilm-related infections: Bridging the gap between clinical management and fundamental aspects of recalcitrance toward antibiotics. Microbiol. Mol. Biol. Rev. 2014, 3, 510–543. [Google Scholar] [CrossRef] [Green Version]
- Zheng, X.C.Q.; Cao, Q.; Mao, F.; Li, X.; Zhu, J.; Lan, L.; Li, J. Discovery of synergistic activity of fluoroquinolones in combination with antimicrobial peptides against clinical polymyxin-resistant Pseudomonas aeruginosa DK2. Chin. Chem. Lett. 2020, 2, 413–417. [Google Scholar] [CrossRef]
- Maciejewska, M.; Bauer, M.; Neubauer, D.; Kamysz, W.; Dawgul, M. Influence of Amphibian Antimicrobial Peptides and Short Lipopeptides on Bacterial Biofilms Formed on Contact Lenses. Materials 2016, 9, 873. [Google Scholar] [CrossRef] [Green Version]
- Ruden, S.; Rieder, A.; Chis Ster, I.; Schwartz, T.; Mikut, R.; Hilpert, K. Synergy Pattern of Short Cationic Antimicrobial Peptides Against Multidrug-Resistant Pseudomonas aeruginosa. Front. Microbiol. 2019, 10, 2740. [Google Scholar] [CrossRef] [Green Version]
- Parvez, M.A.K.; Saha, K.; Rahman, J.; Munmun, R.A.; Rahman, M.A.; Dey, S.K.; Rahman, M.S.; Islam, S.; Shariare, M.H. Antibacterial activities of green tea crude extracts and synergistic effects of epigallocatechingallate (EGCG) with gentamicin against MDR pathogens. Heliyon 2019, 7, e02126. [Google Scholar] [CrossRef] [Green Version]
- Kumari, N.; Singh, S.; Kumari, V.; Kumar, S.; Kumar, V.; Kumar, A. Ouabain potentiates the antimicrobial activity of aminoglycosides against Staphylococcus aureus. BMC Complement Altern. Med. 2019, 1, 119. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Cheng, C.; Xu, G.B.; Tang, L.; Chua, K.L.; Yang, Y.Y. Synthesis and antibiofilm evaluation of 3-hydroxy-2,3-dihydroquinazolin-4(1H)-one derivatives against opportunistic pathogen Acinetobacter baumannii. Bioorg. Med. Chem. 2020, 16, 115606. [Google Scholar] [CrossRef] [PubMed]
- Cirioni, O.; Giacometti, A.; Ghiselli, R.; Kamysz, W.; Silvestri, C.; Orlando, F.; Mocchegiani, F.; Della Vittoria, A.; Kamysz, E.; Saba, V.; et al. The lipopeptides Pal-Lys-Lys-NH(2) and Pal-Lys-Lys soaking alone and in combination with intraperitoneal vancomycin prevent vascular graft biofilm in a subcutaneous rat pouch model of staphylococcal infection. Peptides 2007, 6, 1299–1303. [Google Scholar] [CrossRef] [PubMed]
- Cirioni, O.; Giacometti, A.; Ghiselli, R.; Kamysz, W.; Orlando, F.; Mocchegiani, F.; Silvestri, C.; Licci, A.; Chiodi, L.; Łukasiak, J.; et al. Citropin 1.1-treated central venous catheters improve the efficacy of hydrophobic antibiotics in the treatment of experimental staphylococcal catheter-related infection. Peptides 2006, 6, 1210–1216. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.L.; Hsueh, J.Y.; Yip, B.S.; Chih, Y.H.; Peng, K.L.; Cheng, J.W. Antimicrobial Peptides Display Strong Synergy with Vancomycin against Vancomycin-Resistant, E. faecium, S. aureus, and Wild-Type, E. coli. Int. J. Mol. Sci. 2020, 21, 4578. [Google Scholar] [CrossRef]
- Ciandrini, E.; Morroni, G.; Cirioni, O.; Kamysz, W.; Kamysz, E.; Brescini, L.; Baffone, W.; Campana, R. Synergistic combinations of antimicrobial peptides against biofilms of methicillin-resistant Staphylococcus aureus (MRSA) on polystyrene and medical devices. J. Glob. Antimicrob. Resist. 2020, 21, 203–210. [Google Scholar] [CrossRef]
- Dawgul, M.A.; Greber, K.E.; Bartoszewska, S.; Baranska-Rybak, W.; Sawicki, W.; Kamysz, W. In Vitro Evaluation of Cytotoxicity and Permeation Study on Lysine- and Arginine-Based Lipopeptides with Proven Antimicrobial Activity. Molecules 2017, 22, 2173. [Google Scholar] [CrossRef] [Green Version]
- Greber, K.E.; Dawgul, M.; Kamysz, W.; Sawicki, W.; Lukasiak, J. Biological and surface-active properties of double-chain cationic amino acid-based surfactants. Amino Acids 2014, 8, 1893–1898. [Google Scholar] [CrossRef]
- Institute CaLS. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, Approved Standard, 9th ed.; Wayne, PA, USA, 2012; Available online: https://clsi.org/ (accessed on 10 November 2019).
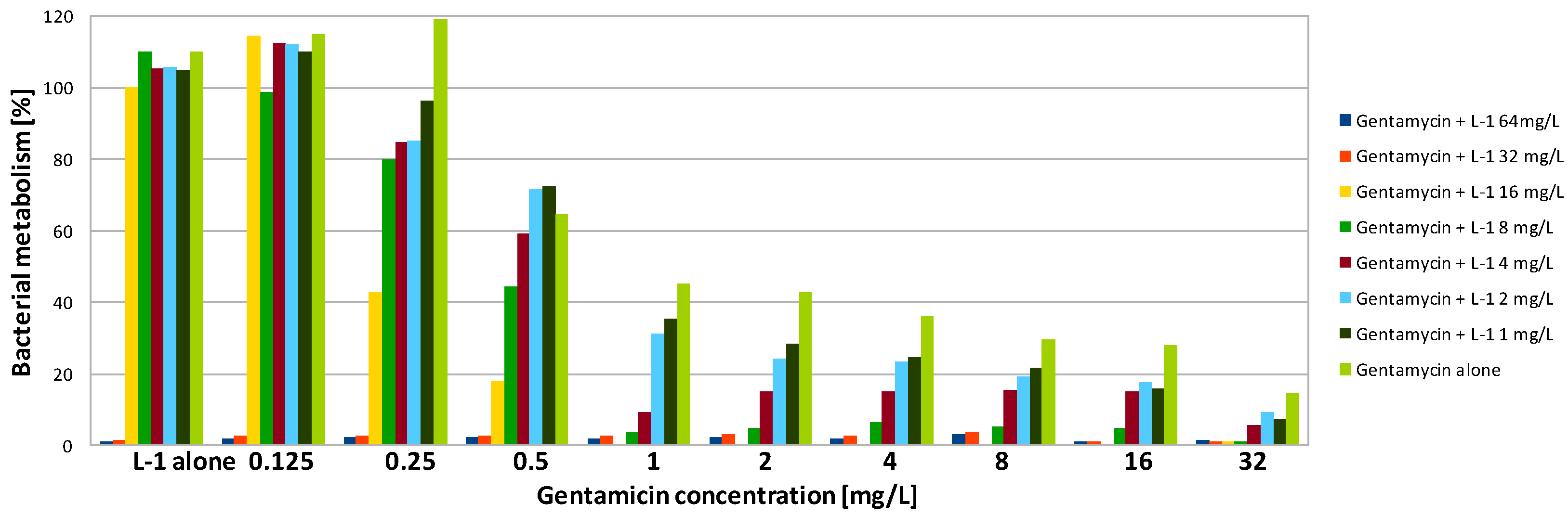
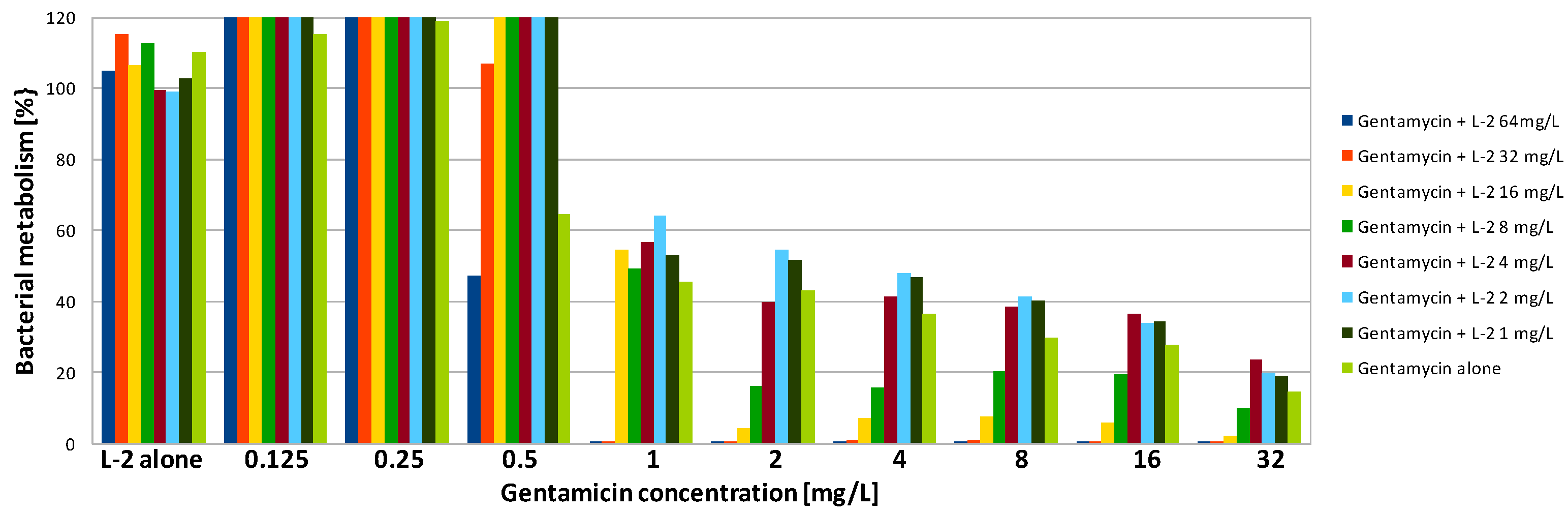
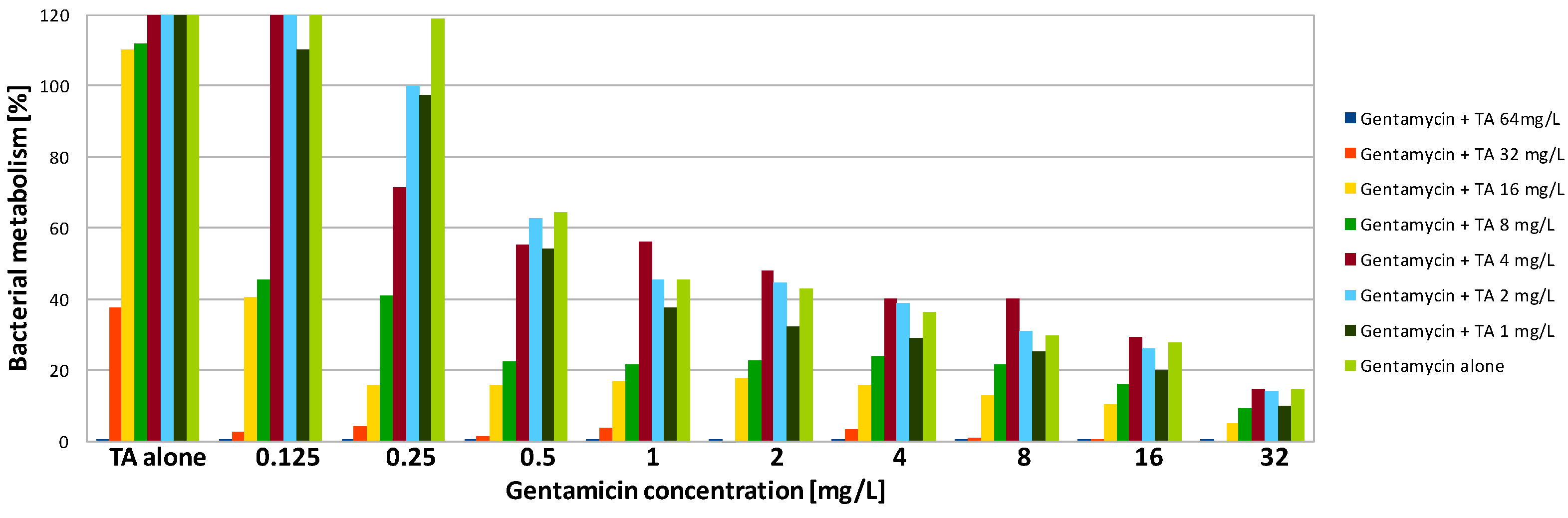
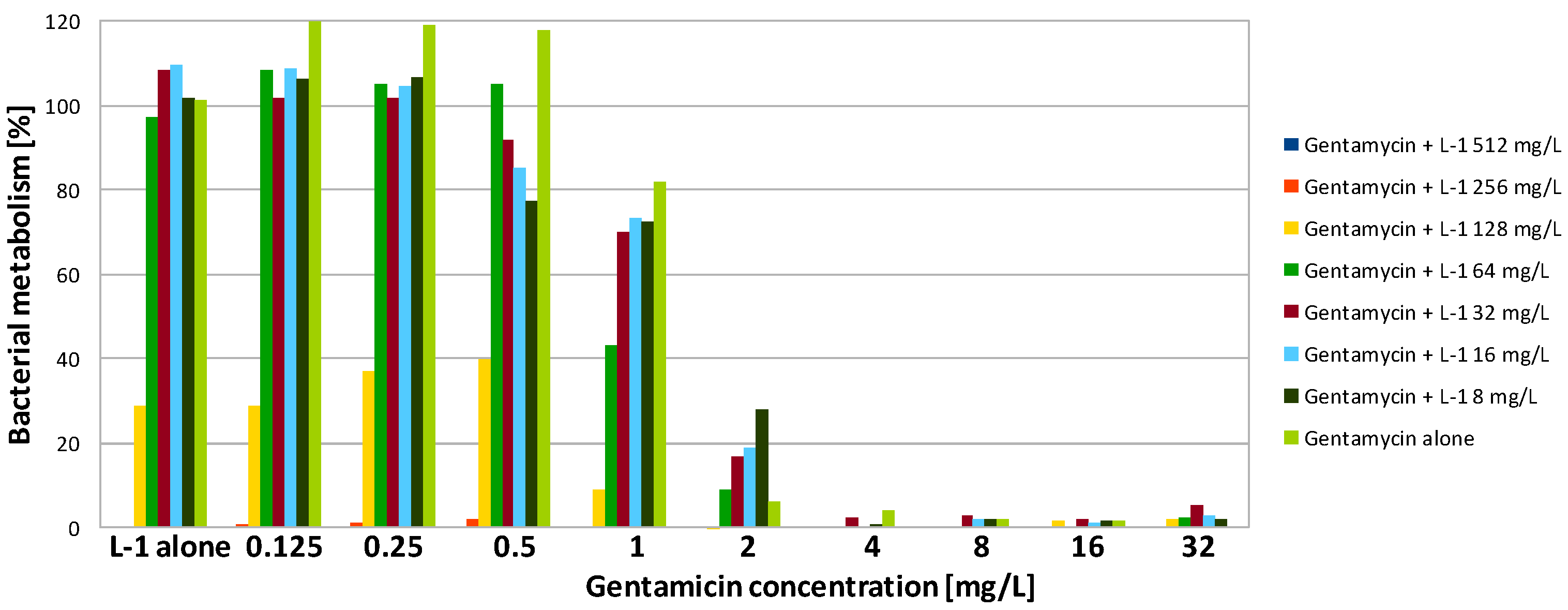
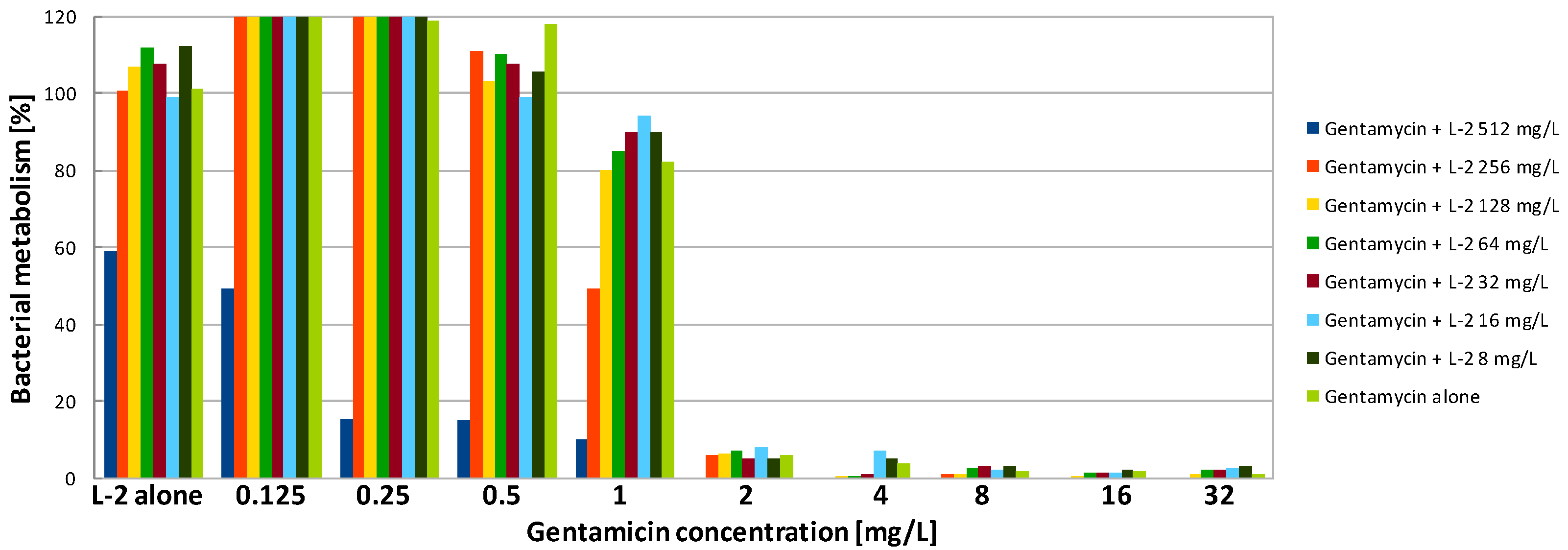
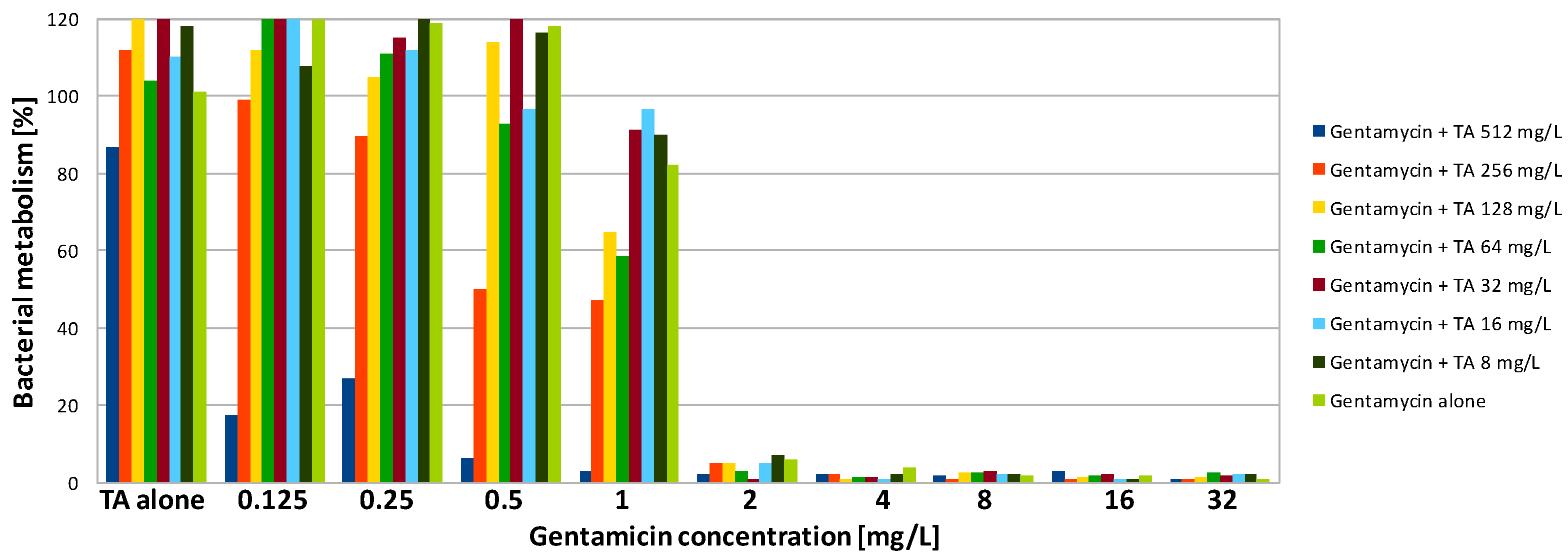
| Compound | SA | PA | ||||
|---|---|---|---|---|---|---|
| MIC | ΣFIC with Gentamicin | Interaction | MIC | ΣFIC with Gentamicin | Interaction | |
| Gentamicin | 0.25 | – | – | 0.5 | – | – |
| Temporin A | 8 | 1 | indifferent | 512 | 1 | indifferent |
| Lipopeptide 1 | 8 | 2 | indifferent | 16 | 2 | indifferent |
| Lipopeptide 2 | 16 | 1 | indifferent | 32 | 2 | indifferent |
| Compound | MBEC (mg/L) | |||||
|---|---|---|---|---|---|---|
| SA | PA | |||||
| MBEC90 | MBEC50 | Enhancement of Activity of Gentamicin | MBEC90 | MBEC50 | Enhancement of Activity of Gentamicin | |
| Gentamicin | >32 | 1 | – | 2 | 2 | – |
| Temporin A | 64 | 32 | +++ | >512 | >512 | ++ |
| Lipopeptide 1 | 32 | 32 | +++ | 256 | 128 | + |
| Lipopeptide 2 | >64 | >64 | +++ | >512 | >512 | + |
| Compound | MBEC 90 (mg/L) | |||
|---|---|---|---|---|
| SA | PA | |||
| Gentamicin | Peptide | Gentamicin | Peptide | |
| Gentamicin alone | >32 | – | 2 | – |
| Lipopeptide 1 alone | – | 32 | – | 256 |
| Gentamicin + L-1 | 1 | 4 | 1 | 128 |
| Lipopeptide 2 alone | – | >64 | – | >512 |
| Gentamicin + L-2 | 1 | 32 | 1 | 512 |
| Temporin A alone | – | 64 | – | >512 |
| Gentamycin + TA | 0.125 | 32 | 0.5 | 512 |
| Compound | MBEC 50 (mg/L) | |||
|---|---|---|---|---|
| SA | PA | |||
| Gentamicin | Peptide | Gentamicin | Peptide | |
| Gentamicin alone | 1 | – | 2 | – |
| Lipopeptide 1 alone | – | 32 | – | 128 |
| Gentamicin + L-1 | 0.5 | 8 | 1 | 64 |
| Lipopeptide 2 alone | – | > 64 | – | > 512 |
| Gentamicin + L-2 | 0.5 | 64 | 1 | 256 |
| Temporin A alone | – | 32 | – | > 512 |
| Gentamicin + TA | 0.125 | 8 | 0.5 | 256 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paduszynska, M.A.; Greber, K.E.; Paduszynski, W.; Sawicki, W.; Kamysz, W. Activity of Temporin A and Short Lipopeptides Combined with Gentamicin against Biofilm Formed by Staphylococcus aureus and Pseudomonas aeruginosa. Antibiotics 2020, 9, 566. https://doi.org/10.3390/antibiotics9090566
Paduszynska MA, Greber KE, Paduszynski W, Sawicki W, Kamysz W. Activity of Temporin A and Short Lipopeptides Combined with Gentamicin against Biofilm Formed by Staphylococcus aureus and Pseudomonas aeruginosa. Antibiotics. 2020; 9(9):566. https://doi.org/10.3390/antibiotics9090566
Chicago/Turabian StylePaduszynska, Malgorzata Anna, Katarzyna Ewa Greber, Wojciech Paduszynski, Wieslaw Sawicki, and Wojciech Kamysz. 2020. "Activity of Temporin A and Short Lipopeptides Combined with Gentamicin against Biofilm Formed by Staphylococcus aureus and Pseudomonas aeruginosa" Antibiotics 9, no. 9: 566. https://doi.org/10.3390/antibiotics9090566





