Antibacterial Activities, Phytochemical Screening and Metal Analysis of Medicinal Plants: Traditional Recipes Used against Diarrhea
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Collection of Medicinal Plants Parts and Their Identification
4.2. Plants Grinding and Recipe Formulation
4.3. Preparation of Plant Extracts
4.4. Qualitative Phytochemical Screening
4.5. Quantitative Phytochemical Screening
4.6. Heavy Metal Analysis of Medicinal Plants and Their Recipes
4.7. Antibacterial Activity
4.8. Determination of Minimum Inhibitory Concentrations (MICs) by Broth Dilution Method
4.9. Statistical Analysis of the Data
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yadav, R.; Agarwala, M. Phytochemical analysis of some medicinal plants. J. Phytol. 2011. Available online: https://updatepublishing.com/journal/index.php/jp/article/view/2737 (accessed on 5 April 2019).
- Babu-Kasimala, M.; Tukue, M.; Ermias, R. Phytochemical screening and antibacterial activity of two common terresterial medicinal plants Rutachalepensis and Rumexnervosus. Available online: https://ojs.unud.ac.id/index.php/bmj/article/view/21605/14295 (accessed on 20 August 2019).
- Ndam, L.; Mih, A.; Fongod, A.; Tening, A.; Tonjock, R.; Enang, J.; Fujii, Y. Phytochemical screening of the bioactive compounds in twenty (20) Cameroonian medicinal plants. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 768–778. [Google Scholar]
- Almeida, C.E.; Karnikowski, M.G.; Foleto, R.; Baldisserotto, B. Analysis of antidiarrhoeic effect of plants used in popular medicine. Rev. Saude Publica 1989, 29, 428. [Google Scholar] [CrossRef] [PubMed]
- Atta, A.H.; Mouneir, S.M. Antidiarrhoeal activity of some Egyptian medicinal plant extracts. J. Ethnopharmacol. 2004, 92, 303. [Google Scholar] [CrossRef] [PubMed]
- Mamoon, S.A.; Azam, M.G. Preliminary phytochemical screening and antidiarrhoeal activity of Derris trifoliata Lour. Int. J. Pharm. Sci. Res. 2012, 3, 97. [Google Scholar]
- Das, P.R.; Akter, S.; Islam, M.T.; Kabir, M.H.; Haque, M.M.; Khatun, Z.; Nurunnabi, M.; Khatun, Z.; Lee, Y.; Jahan, R.; et al. A selection of medicinal plants used for treatment of diarrhea by folk medicinal practitioners of Bangladesh. Am. Eurasian J. Sustain. Agric. 2012, 6, 153. [Google Scholar]
- Samant, S.S.; Dhar, U.; Palni, L.M.S. Medicinal Plants of Indian Himalaya: Diversity, Distribution Potential Values. Nainital Gyanodaya Prakashan 1998, 163. Available online: https://www.scirp.org/(S(i43dyn45teexjx455qlt3d2q))/reference/ReferencesPapers.aspx?ReferenceID=222461 (accessed on 23 October 2019).
- Sharma, V.; Joshi, B.D. Traditional medicines used for dental health Care amongst the local people of Almora district of Central Himalaya in India. Asian J. Tradit. Med. 2010, 5, 117. [Google Scholar]
- Sunil, K.; Rana, A.C. Herbal Approach for Diarrhea. Int. Res. J. Pharm. 2013, 4, 31. [Google Scholar]
- Hussain, I.; Khan, F.; Khan, I.; Khan, L.; Ullah, W. Determination of heavy metals in medicinal plants. J. Chem. Soc. Pak. 2006, 28, 347. [Google Scholar]
- Gawel, J.E.; Ahner, B.A.; Friedland, A.J.; Morel, F.M. Role for heavy metals in forest decline indicated by phytochelatin measurements. Nature 1996, 381, 64. [Google Scholar] [CrossRef]
- Khan, I.; Ali, J.; Tullah, H. Heavy metals determination in medicinal plant Withaniasomnifera growing in various areas of peshawar, NWFP, Pakistan. J. Soc. Pak. 2008, 30, 69. [Google Scholar]
- Abdel-Salam, A. Functional foods: Hopefulness to good health. Am. J. Food Technol. 2010, 2, 86–99. [Google Scholar]
- Thomson, I.F.A.; Ripa, M. Haque and IJ Bulbul. Pak. J. Bio. Sci. 2010, 13, 22–27. [Google Scholar]
- Patsilinakos, A.; Artini, M.; Papa, R.; Saatino, M.; Bozovic, M.; Garzoli, S.; Vrenna, G.; Buzzi, R.; Manfredini, S.; Selan, L.; et al. Machine learning analyses on data including essential oil chemical composition and in vitro experimental antibiofilm activities against staphylococcus species. Molecules 2019, 24, 890. [Google Scholar] [CrossRef]
- Artini, M.; Patsilinakos, A.; Papa, R.; Bozovic, M.; Sabatino, M.; Garzoli, S.; Vrenna, G.; Tilotta, M.; Pepi, F.; Rango, R.; et al. Antimicrobial and antibiofilm activity and machine learning classification analysis of essential oils from different mediterranean plants against pseudomonas aeruginosa. Molecules 2018, 23, 482. [Google Scholar] [CrossRef]
- Vikas, K.; Sharma, N.; Sourirajan, A.; Khosla, P.K.; Dev, K. Comparative evaluation of antimicrobial and antioxidant potential of ethanolic extract and its fractions of bark and leaves of Terminalia arjuna from north-western Himalayas, India. J. Tradit. Complement. Med. 2018, 8, 100–106. [Google Scholar]
- Bikash, M.; Paudel, K.R.; Sharma, B.; Karki, R. Phytochemical profile and pharmacological activity of Aegle marmelos Linn. J. Integr. Med. 2018, 16, 153–163. [Google Scholar]
- Rathinamoorthy, R.; Thilagavathi, G. Terminalia chebula-review on pharmacological and biochemical studies. Int. J. Pharm Technol. Res. 2014, 6, 97–116. [Google Scholar]
- Ahmed, M.; Khan, M.A.; Zafar, M.; Sultana, S. Treatment of common ailments by plant-based remedies among the people of district Attock (Punjab) of Northern Pakistan. Afr. J. Tradit. Altern. Med. 2007, 4, 112–120. [Google Scholar]
- Pfundstein, B.; El Desouky, S.K.; Hull, W.E.; Haubner, R.; Erben, G.; Owen, R.W. Polyphenolic compounds in the fruits of Egyptian medicinal plants (Terminalia bellerica, Terminalia chebula and Terminalia horrida): Characterization, quantitation and determination of antioxidant capacities. Phytochemistry 2010, 71, 1132–1148. [Google Scholar] [CrossRef] [PubMed]
- Kakar, S.A.; Tareen, R.B.; Kakar, M.A.; Jabeen, H.; Kakar, S.U.R.; Al-Kahraman, Y.S.M.A.; Shafee, M. Screening of antibacterial activity of four medicinal plants of Balochistan-Pakistan. Pak. J Bot. 2012, 44, 245–250. [Google Scholar]
- World Health Organization. Quality Control Methods for Medicinal Plant Materials. 1998. Available online: https://apps.who.int/iris/handle/10665/44479 (accessed on 15 August 2019).
- Shah, A.; Niaz, A.; Ullah, N.; Rehman, A.; Akhlaq, M.; Zakir, M.; Suleman Khan, M.; Comparative study of heavy metals in soil and selected medicinal plants. J. Chem. 2013. Available online: https://www.hindawi.com/journals/jchem/2013/621265/ (accessed on 15 August 2019).
- Hill, A.F. Economic Botany, A Textbook of Useful Plants and Plant Products, 2nd ed.; McGraw Hill Book Company Inc.: New York, NY, USA, 1952; Volume 205. [Google Scholar]
- Ferguson, P.J.; Kurowska, E.; Freeman, D.J.; Chambers, A.F.; Koropatnick, D.J. A flavonoid fraction from cranberry extract inhibits proliferation of human tumor cell lines. J. Nutr. 2004, 134, 1529–1535. [Google Scholar] [CrossRef]
- Kanadaswami, C.; Lee, L.T.; Lee, P.P.H.; Hwang, J.J.; Ke, F.C.; Huang, Y.T.; Lee, M.T. The antitumor activities of flavonoids. In Vivo 2005, 19, 895–909. [Google Scholar]
- Akinpelu, D.; Onakoya, T. Antimicrobial activities of medicinal plants used in folklore remedies in south-western. Afr. J. Biotechnol. 2006, 5, 11. [Google Scholar]
- Nguyi, A. Tannins of some Nigerian flora. Niger. J. Biotechnol. 1988, 6, 221–226. [Google Scholar]
- Khosit, P.; Hiriote, W.; Soonthornchareonnon, N.; Jongsakul, K.; Sireeratawong, S.; Tor-Udom, S. In vitro and in vivo antiplasmodial activity and cytotoxicity of water extracts of Phyllanthusemblica, Terminalia chebula, and Terminalia bellerica. J. Med. Assoc. Thail. 2011, 93, 120. [Google Scholar]
- Mansour, S.; Mahmoud, M.F.; Hasan, R.A.; Abdelfattah, M.A.O.; Osman, S.; Rashid, H.; El-Shazly, A.M.; Wink, M. Chemical composition, antioxidant and hepatoprotective activities of methanol extracts from leaves of Terminalia bellirica and Terminalia sericea (Combretaceae). PeerJ 2019, 7, e6322. [Google Scholar]
- Hasan, Z.; Anwar, Z.; Khattak, K.U.; Islam, M.; Khan, R.U.; Khattak, J.Z.K. Civic pollution and its effect on water quality of river Toi at district Kohat, NWFP. Res. J. Environ. Earth Sci. 2012, 4, 334–339. [Google Scholar]
- McGrath, S. Chromium and Nickel, Heavy Metals in Soils. 1990; pp. 125–150. Available online: https://trove.nla.gov.au/work/17304135?q&versionId=45617592 (accessed on 23 October 2019).
- Ullah, R.; Khader, J.A.; Hussain, I.; Talha, N.M.A.; Khan, N. Investigation of macro and micro-nutrients in selected medicinal plants. Afr. J. Pharm. Pharmacol. 2012, 6, 1829–1832. [Google Scholar]
- Rehman, A.; Ullah, H.; Khan, R.U.; Ahmad, I. Population based study of heavy metals in medicinal plant Capparis decidua. Int. J. Pharm. Pharmacol. Sci. 2013, 5, 108–113. [Google Scholar]
- Adelekan, B.; Abegunde, K. Heavy metals contamination of soil and groundwater at automobile mechanic villages in Ibadan, Nigeria. Int. J. Phys. Sci. 2011, 5, 1045–1058. [Google Scholar]
- Gupta, U. Copper in the Environment; Nariago, J.O., Ed.; John Wiley and Sons: New York, NY, USA, 1975; Volume 255. [Google Scholar]
- Chishti, K.A.; Khan, F.A.; Shah, S.M.H.; AsifKhan, M.; Khan, J.; Shah, S.M.M.; Hussain, I. Estimation of heavy metals in the seeds of blue and white capitulum’s of silybummarianum grown in various districts of pakistan. J. Basic Appl. Sci. 2011, 7. Available online: https://search.proquest.com/openview/4cc7dfa78ca921cb079d285fe6cf6626/1.pdf?pq-origsite=gscholar&cbl=616537 (accessed on 23 October 2019).
- Adnan, M.; Ullah, I.; Tariq, A.; Murad, W.; Azizullah, A.; Khan, A.L.; Ali, N. Ethnomedicine use in the war affected region of northwest Pakistan. J. Ethnobiol. Ethnomed. 2014, 10, 16. [Google Scholar] [CrossRef]
- Murad, W.; Azizullah, A.; Adnan, M.; Tariq, A.; Khan, K.U.; Waheed, S.; Ahmad, A. Ethnobotanical assessment of plant resources of Banda Daud Shah, District Karak, Pakistan. J. Ethnobiol. Ethnomed. 2013, 9, 77. [Google Scholar] [CrossRef]
- Amjad, M.S.; faisal Qaeem, M.; Ahmad, I.; Khan, S.U.; Chaudhari, S.K.; Malik, N.Z.; Shaheen, H.; Khan, A.M. Descriptive study of plant resources in the context of the ethnomedicinal relevance of indigenous flora: A case study from Toli Peer National Park, Azad Jammu and Kashmir, Pakistan. PLoS ONE 2017, 12, e0171896. [Google Scholar] [CrossRef]
- Mattila, P.; Hellström, J. Phenolic acids in potatoes, vegetables, and some of their products. J. Food. Compos. Anal. 2007, 20, 152–160. [Google Scholar] [CrossRef]
- Krishnaiah, D.; Devi, T.; Bono, A.; Sarbatly, R. Studies on phytochemical constituents of six Malaysian medicinal plants. J. Med. Plants Res. 2009, 3, 67–72. [Google Scholar]
- Tyler, V.E. Phytomedicines in Western Europe: Potential impact on herbal medicine in the United States. ACS Publ. 1993. Available online: https://pubs.acs.org/doi/10.1021/bk-1993-0534.ch003 (accessed on 23 October 2019).
- Harborne, J. Phytochemical methods: London Chapman and hill Ltd. 1973; pp. 49–188. Available online: https://trove.nla.gov.au/version/45450216 (accessed on 23 October 2019).
- Obadoni, B.; Ochuko, P. Phytochemical studies and comparative efficacy of the crude extracts of some haemostatic plants in Edo and Delta States of Nigeria. Glob. J. Pure Appl. Sci. 2002, 8, 203–208. [Google Scholar] [CrossRef]
- Boham, B.; Kocipai-Abuyazan, R. Flavonoids and condensed tannins from the leaves of Vaccinumraticulatum and Vaccinumcalycinium. Pac. Sci. 1994, 48, 458–463. [Google Scholar]
- Khan, S.A.; Khan, L.; Hussain, I.; Marwat, K.B.; Akhtar, N. Profile of heavy metals in selected medicinal plants. Pak. J. Weed Sci. Res. 2008, 14, 101–110. [Google Scholar]
- Kirby, W.; Yoshihara, G.; Sundsted, K.; Warren, J. Clinical usefulness of a single disc method for antibiotic sensitivity testing. Antibiot. Annu. 1956, 892. Available online: https://www.asm.org/getattachment/2594ce26-bd44-47f6-8287-0657aa9185ad/Kirby-Bauer-Disk-Diffusion-Susceptibility-Test-Protocol-pdf.pdf (accessed on 23 October 2019).
- European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID). Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution. Clin. Microbiol. Infect. 2003, 9, 1–7. [Google Scholar]
| S. No | Plant Parts | Alkaloids | Flavonoids | Saponins | Tannins | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Meth ext | Aqe Ext | Chlor Ext | n-hex ext | Meth Ext | Aqe Ext | Chlo ext | n-hex ext | Meth ext | Aqe ext | Chlor ext | n-hex ext | Meth Ext | Aqe ext | Chlor ext | n-hex ext | ||
| 1 | T. chebula (fruits) | + | + | + | + | + | − | − | − | + | + | + | + | + | + | + | − |
| 2 | A. marmelos (fruits) | + | + | + | − | + | + | + | − | + | + | + | + | + | + | + | − |
| 3 | C. longa (rhizomes) | + | − | + | + | − | − | − | − | + | + | + | + | − | − | − | − |
| 4 | S. aromaticum | + | + | + | + | + | − | − | + | + | + | + | + | + | + | + | + |
| 5 | P. nigrum (seeds) | + | + | + | + | − | − | − | − | + | + | + | + | + | + | − | − |
| 6 | C. cassia (barks) | + | + | + | + | − | − | − | − | + | + | + | + | + | − | − | − |
| 7 | Recipe (1) | + | + | + | + | + | + | + | − | + | + | + | + | + | + | + | − |
| 8 | Recipe (2) | + | + | + | + | + | − | − | + | + | + | + | + | + | + | + | + |
| S. NO | Recipes/Plant Parts | Alkaloids % | Flavonoids % | Saponins % |
|---|---|---|---|---|
| 1 | Recipe (1) | 19.66 | 28.13 | 1.19 |
| 2 | T. chebula (fruits) | 27.84 | 61.21 | 6.32 |
| 3 | A. marmelos (fruits) | 2.54 | 23.81 | 0.24 |
| 4 | C. longa (rhizomes) | 2.66 | 6.82 | 0.36 |
| 5 | Recipe (2) | 17.78 | 14.25 | 0.83 |
| 6 | S. aromaticum | 11.88 | 18.6 | 1.10 |
| 7 | P. nigrum (seeds) | 5.06 | 9 | 0.19 |
| 8 | C. cassia (barks) | 5.28 | 4.74 | 0.23 |
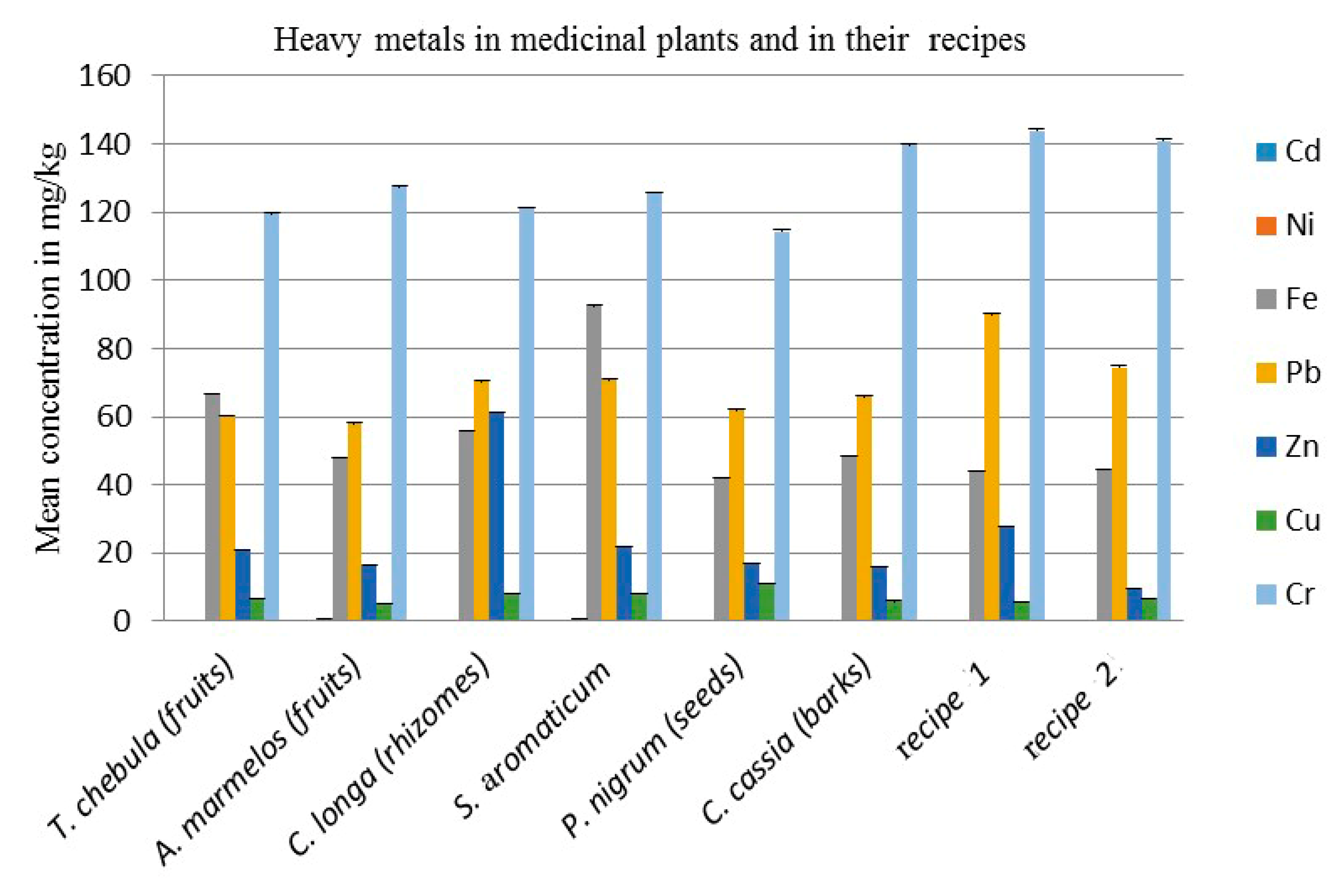
| S. NO | Name of Plants Part | Cd mg/kg | Ni mg/kg | Fe mg/kg | Pb mg/kg | Zn mg/kg | Cu mg/kg | Cr mg/kg |
|---|---|---|---|---|---|---|---|---|
| 1 | T.chebula (fruits) | BDL | BDL | 66.775 ± 0.016 | 60.125 ± 0.250 | 21.075 ± 0.013 | 6.8 ± 0.008 | 119.475 ± 0.430 |
| 2 | A. marmelos (fruits) | BDL | 0.55 ± 0.019 | 48.1 ± 0.002 | 58 ± 0.184 | 16.325 ± 0.004 | 5.175 ± 0.008 | 127.375 ± 0.347 |
| 3 | C. longa (rhizomes) | BDL | 0.05 ± 0.013 | 55.9 ± 0.072 | 70.1 ± 0.348 | 61.375 ± 0.003 | 8.225 ± 0.022 | 121.05 ± 0.099 |
| 4 | S. Aromaticum | BDL | 0.825 ± 0.005 | 92.45 ± 0.039 | 70.675 ± 0.401 | 21.75 ± 0.030 | 8.3 ± 0.003 | 125.6 ± 0.302 |
| 5 | P. nigrum (seeds) | BDL | 0.35 ± 0.045 | 41.975 ± 0.034 | 61.925 ± 0.150 | 16.9 ± 0.006 | 10.9 ± 0.015 | 114.325 ± 0.402 |
| 6 | C. cassia (barks) | BDL | BDL | 48.475 ± 0.075 | 65.875 ± 0.179 | 16.025 ± 0.040 | 5.875 ± 0.064 | 139.65 ± 0.467 |
| 7 | Recipe (1) | BDL | BDL | 43.875 ± 0.045 | 89.9 ± 0.449 | 27.925 ± 0.022 | 5.675 ± 0.020 | 143.9 ± 0.326 |
| 8 | Recipe (2) | BDL | BDL | 44.475 ± 0.077 | 74.45 ± 0.438 | 9.475 ± 0.064 | 6.557 ± 0.016 | 140.926 ± 0.314 |
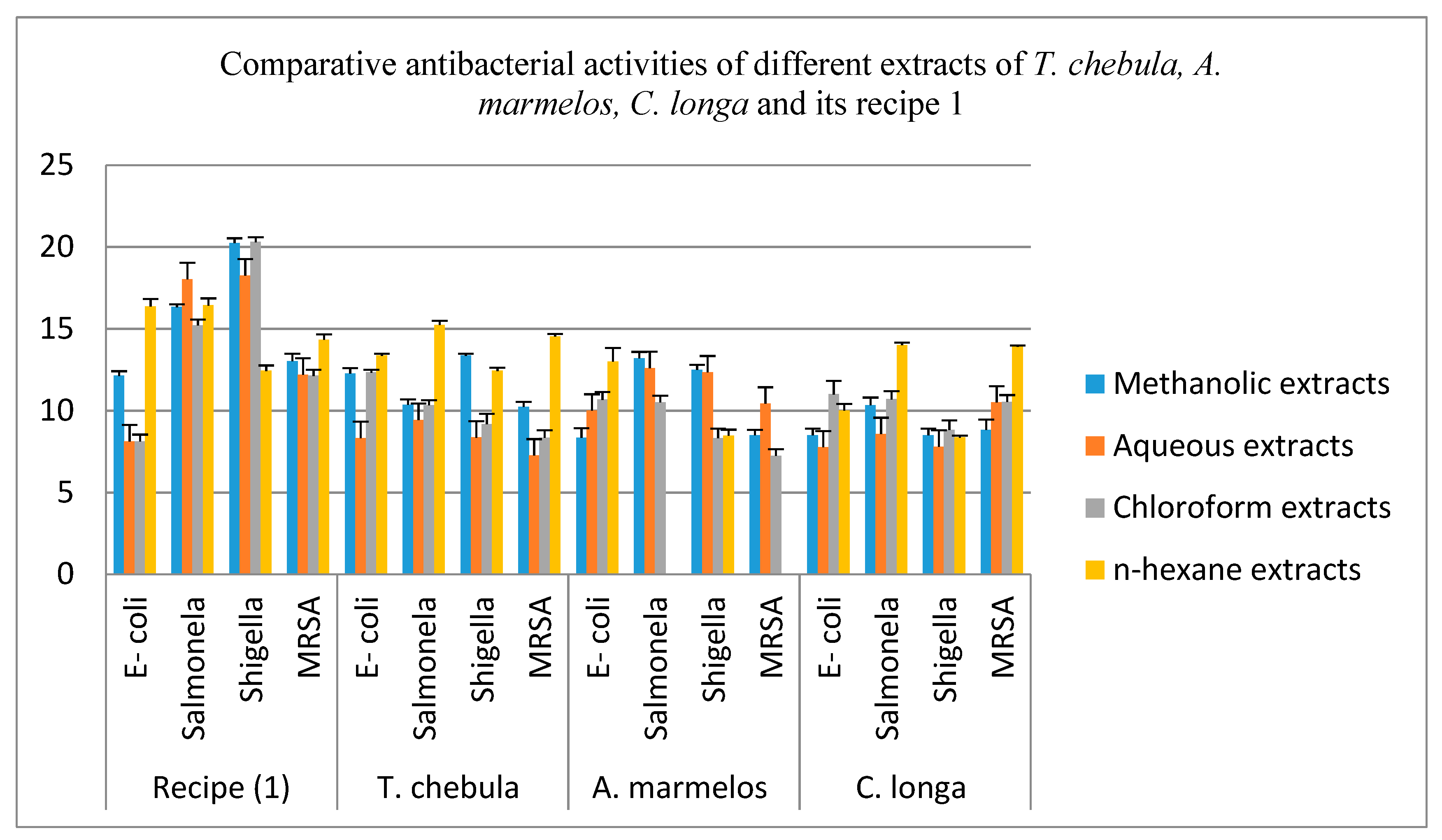
| Recipe/Plant Part | Bacteria | Methanol Extracts | Aqueous Extracts | Chloroform Extracts | n-Hexane Extracts | Cpx | DMSO |
|---|---|---|---|---|---|---|---|
| (mm) | (mm) | (mm) | (mm) | Mm | (mm) | ||
| Recipe (1) | E-coli | 12.13 ± 0.28 | 8.13 ± 0.44 | 8.13 ± 0.41 | 16.36 ± 0.47 | 32 | ̶ |
| Salmonella | 16.33 ± 0.17 | 18.03 ± 0.70 | 15.2 ± 0.37 | 16.43 ± 0.43 | 33 | ̶ | |
| Shigella | 20.23 ± 0.30 | 18.26 ± 0.32 | 20.3 ± 0.29 | 12.43 ± 0.33 | 31 | ̶̶̶ | |
| MRSA | 13.03 ± 0.45 | 12.2 ± 0.24 | 12.13 ± 0.36 | 14.33 ± 0.32 | 23 | ̶ | |
| T. chebula (fruits) | E-coli | 12.26 ± 0.34 | 8.33 ± 0.48 | 12.33 ± 0.17 | 13.36 ± 0.12 | 32 | ̶ |
| Salmonella | 10.36 ± 0.33 | 9.43 ± 0.24 | 10.33 ± 0.30 | 15.23 ± 0.25 | 33 | ̶ | |
| Shigella | 13.36 ± 0.12 | 8.36 ± 0.16 | 9.17 ± 0.63 | 12.43 ± 0.20 | 31 | ̶̶̶ | |
| MRSA | 10.23 ± 0.30 | 7.26 ± 0.26 | 8.34 ± 0.47 | 14.53 ± 0.16 | 23 | ̶ | |
| A. marmelos (fruits) | E-coli | 8.34 ± 0.58 | 10 ± 0.81 | 10.67 ± 0.48 | 13 ± 0.82 | 32 | ̶ |
| Salmonella | 13.2 ± 0.39 | 12.6 ± 0.43 | 10.5 ± 0.41 | NIL | 33 | ̶ | |
| Shigella | 12.5 ± 0.29 | 12.34 ± 0.47 | 8.33 ± 0.57 | 8.47 ± 0.37 | 31 | ̶̶̶ | |
| MRSA | 8.5 ± 0.33 | 10.43 ± 0.42 | 7.24 ± 0.40 | NIL | 23 | ̶ | |
| C. longa (rhizomes) | E-coli | 8.5 ± 0.40 | 7.76 ± 0.20 | 11 ± 0.81 | 10 ± 0.41 | 32 | ̶ |
| Salmonella | 10.33 ± 0.47 | 8.57 ± 0.32 | 10.7 ± 0.49 | 14 ± 0.16 | 33 | ̶ | |
| Shigella | 8.5 ± 0.40 | 7.8 ± 0.59 | 8.83 ± 0.57 | 8.36 ± 0.12 | 31 | ̶̶̶ | |
| MRSA | 8.83 ± 0.62 | 10.5 ± 0.24 | 10.53 ± 0.42 | 13.9 ± 0.08 | 23 | ̶ |
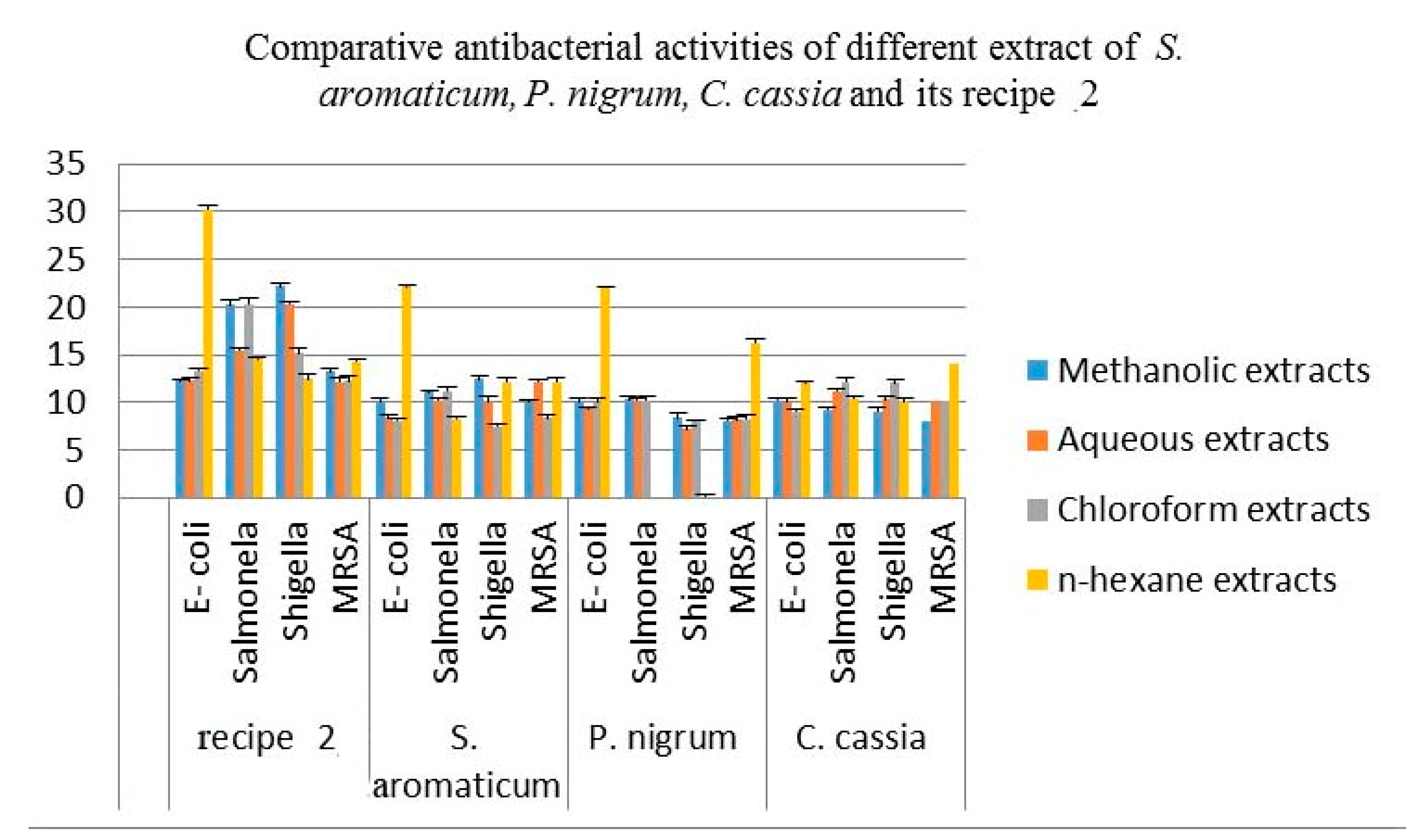
| Recipe/Plant Part | Bacteria | Methanol Extracts | Aqueous Extracts | Chloroform Extracts | n-Hexane Extracts | Cpx | DMSO |
|---|---|---|---|---|---|---|---|
| (mm) | (mm) | (mm) | (mm) | Mm | (mm) | ||
| recipe 2 | E-coli | 12.23 ± 0.49 | 12.26 ± 0.53 | 13.33 ± 0.12 | 24.23 ± 0.12 | 32 | ̶ |
| Salmonella | 20.23 ± 0.20 | 15.43 ± 0.33 | 20.33 ± 0.30 | 14.53 ± 0.44 | 33 | ̶ | |
| Shigella | 22.16 ± 0.47 | 20.33 ± 0.24 | 15.16 ± 0.54 | 12.46 ± 0.26 | 31 | ̶̶̶ | |
| MRSA | 13.16 ± 0.41 | 12.13 ± 0.28 | 12.23 ± 0.46 | 14.23 ± 0.45 | 23 | ̶ | |
| S. aromaticum (flower buds) | E-coli | 10.06 ± 0.41 | 8.3 ± 0.37 | 8.03 ± 0.45 | 22.2 ± 0.20 | 32 | ̶ |
| Salmonella | 11.03 ± 0.36 | 10.2 ± 0.33 | 11.13 ± 0.25 | 8.23 ± 0.12 | 33 | ̶ | |
| Shigella | 12.46 ± 0.25 | 10.1 ± 0.29 | 7.5 ± 0.40 | 12.1 ± 0.29 | 31 | ̶̶̶ | |
| MRSA | 10.03 ± 0.28 | 12.13 ± 0.49 | 8.36 ± 0.20 | 12.1 ± 0.53 | 23 | ̶ | |
| P. nigrum (seeds) | E-coli | 10.13 ± 0.28 | 9.3 ± 0.29 | 10.03 ± 0.37 | 22.16 ± 0.41 | 32 | ̶ |
| Salmonella | 10.33 ± 0.25 | 10.2 ± 0.24 | 10.16 ± 0.34 | NIL | 33 | ̶ | |
| Shigella | 8.36 ± 0.32 | 7.2 ± 0.16 | 8.03 ± 0.41 | NIL | 31 | ̶̶̶ | |
| MRSA | 8.03 ± 0.45 | 8.13 ± 0.37 | 8.2 ± 0.16 | 16.2 ± 0.24 | 23 | ̶ | |
| C. cassia (barks) | E-coli | 10.23 ± 0.20 | 10.03 ± 0.38 | 9.06 ± 0.41 | 12.03 ± 0.45 | 32 | ̶ |
| Salmonella | 9.2 ± 0.24 | 11.13 ± 0.36 | 12.13 ± 0.28 | 10.43 ± 0.24 | 33 | ̶ | |
| Shigella | 9.06 ± 0.32 | 10.26 ± 0.34 | 12.03 ± 0.36 | 10.13 ± 0.28 | 31 | ̶̶̶ | |
| MRSA | 8.03 ± 0.45 | 10.06 ± 0.41 | 10.13 ± 0.32 | 14.06 ± 0.32 | 23 | ̶ |
| S. No | Bacteria | Methanol Extracts (mg/L) | Aqueous Extracts (mg/L) | Chloroform Extracts (mg/L) | n-Hexane Extracts (mg/L) |
|---|---|---|---|---|---|
| 1 | E-coli | 11,000 | 12,500 | 14,000 | 15,000 |
| 2 | Salmonella | 12,000 | 11,500 | 13,000 | 14,000 |
| 3 | Shigella | 11,500 | 12,000 | 14,000 | 14,500 |
| 4 | MRSA | 14,000 | 14,500 | 15,000 | 15,000 |
| S. No | Bacteria | Methanol Extracts (mg/L) | Aqueous Extracts (mg/L) | Chloroform Extracts (mg/L) | n-Hexane Extracts (mg/L) |
|---|---|---|---|---|---|
| 1 | E-coli | 12,000 | 11,000 | 13,000 | 14,000 |
| 2 | Salmonella | 10,500 | 12,000 | 12,500 | 13,000 |
| 3 | Shigella | 11,500 | 12,500 | 13,000 | 14,000 |
| 4 | MRSA | 15,000 | 14,000 | 15,000 | 15,000 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahmood, N.; Nazir, R.; Khan, M.; Khaliq, A.; Adnan, M.; Ullah, M.; Yang, H. Antibacterial Activities, Phytochemical Screening and Metal Analysis of Medicinal Plants: Traditional Recipes Used against Diarrhea. Antibiotics 2019, 8, 194. https://doi.org/10.3390/antibiotics8040194
Mahmood N, Nazir R, Khan M, Khaliq A, Adnan M, Ullah M, Yang H. Antibacterial Activities, Phytochemical Screening and Metal Analysis of Medicinal Plants: Traditional Recipes Used against Diarrhea. Antibiotics. 2019; 8(4):194. https://doi.org/10.3390/antibiotics8040194
Chicago/Turabian StyleMahmood, Nasir, Ruqia Nazir, Muslim Khan, Abdul Khaliq, Mohammad Adnan, Mohib Ullah, and Hongyi Yang. 2019. "Antibacterial Activities, Phytochemical Screening and Metal Analysis of Medicinal Plants: Traditional Recipes Used against Diarrhea" Antibiotics 8, no. 4: 194. https://doi.org/10.3390/antibiotics8040194
APA StyleMahmood, N., Nazir, R., Khan, M., Khaliq, A., Adnan, M., Ullah, M., & Yang, H. (2019). Antibacterial Activities, Phytochemical Screening and Metal Analysis of Medicinal Plants: Traditional Recipes Used against Diarrhea. Antibiotics, 8(4), 194. https://doi.org/10.3390/antibiotics8040194





