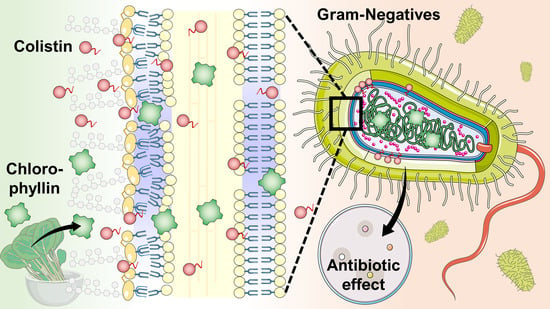
Using Colistin as a Trojan Horse: Inactivation of Gram-Negative Bacteria with Chlorophyllin
Abstract
1. Introduction
2. Results
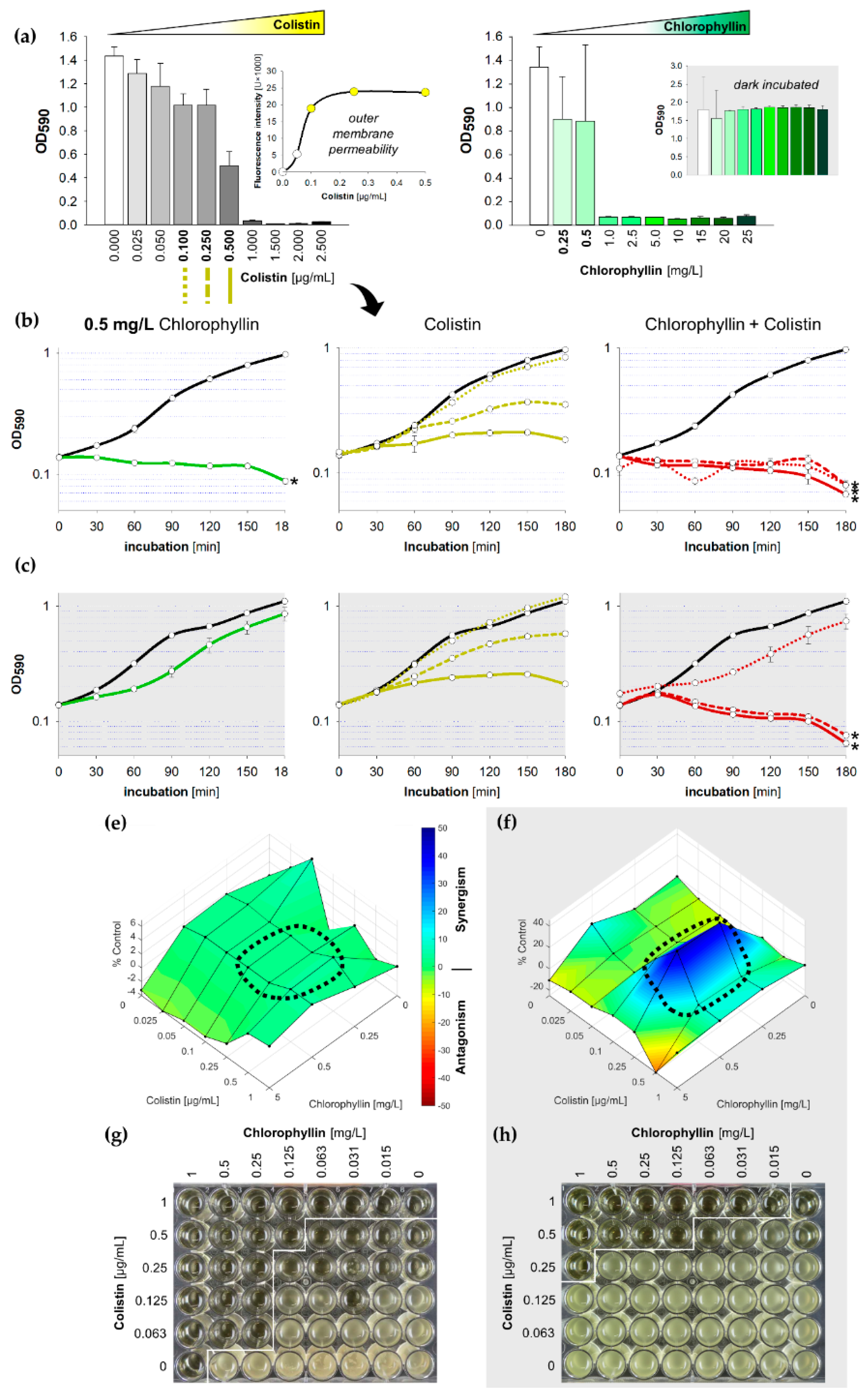
2.1. Synergistic Effects of Chlorophyllin in Combination with Colistin on the Growth of E. coli
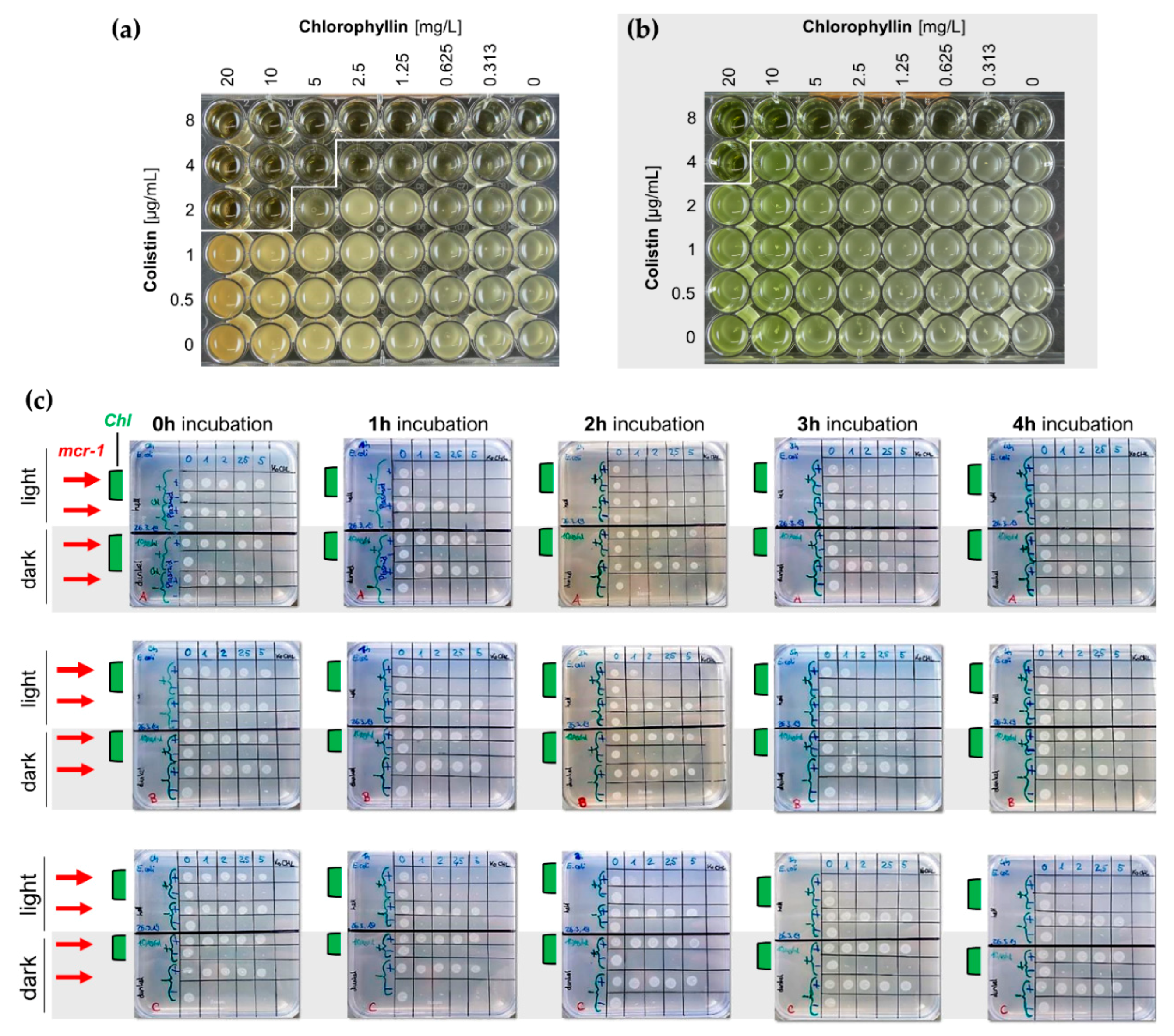
2.2. Synergistic Effects of Chlorophyllin in Combination with Colistin on the Growth of S. Typhimurium
2.3. Synergistic Effects of Chlorophyllin in Combination with Colistin on mcr-1-Positive E. coli
2.4. Confirmation of Chlorophyllin Uptake
3. Discussion
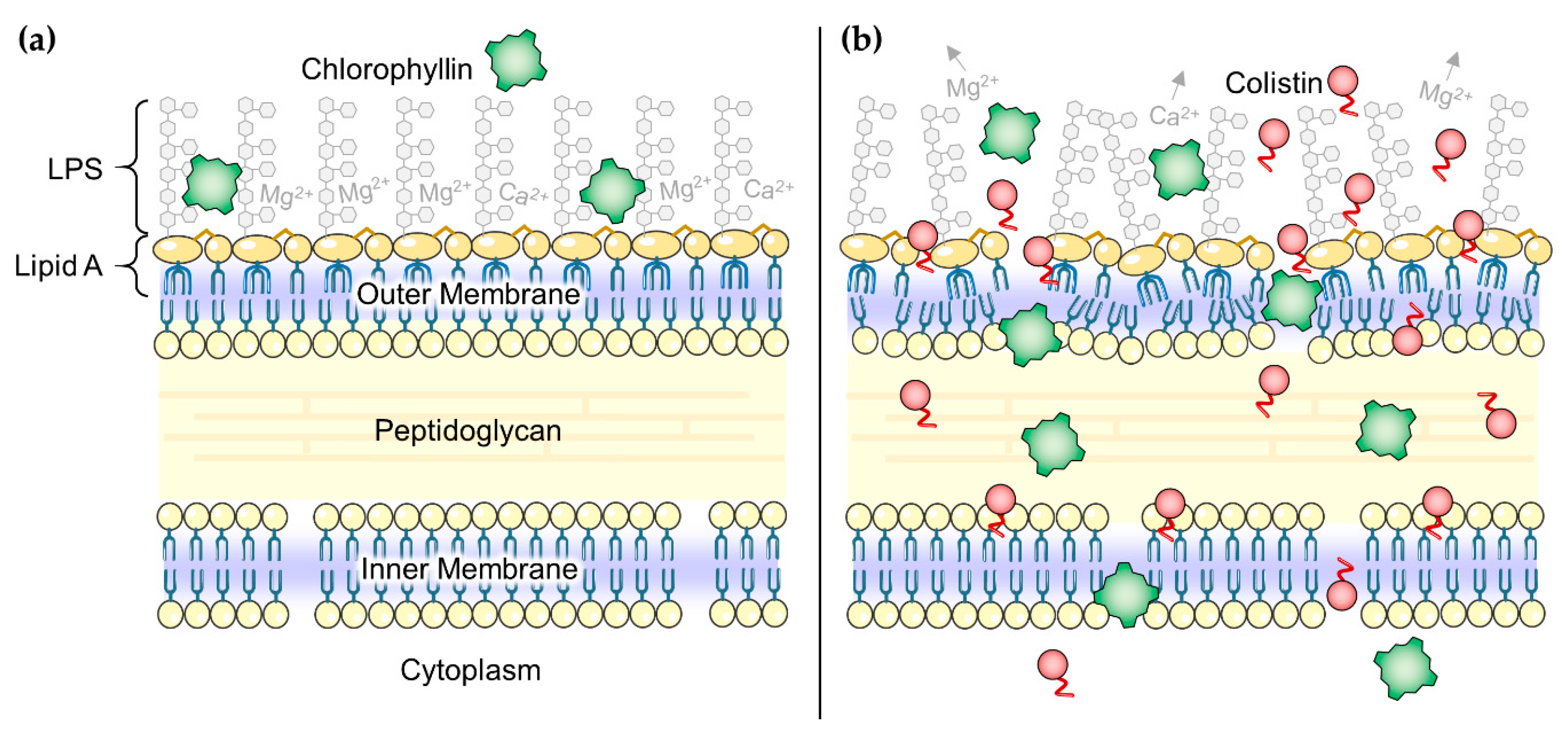
3.1. Strategies to Inactivate Gram-Negative Bacteria with Chlorophyllin
3.2. Overcoming Colistin Resistance of E. coli Using Colistin in Combination with Chlorophyllin
4. Materials and Methods
4.1. Bacteria Strains and Growth
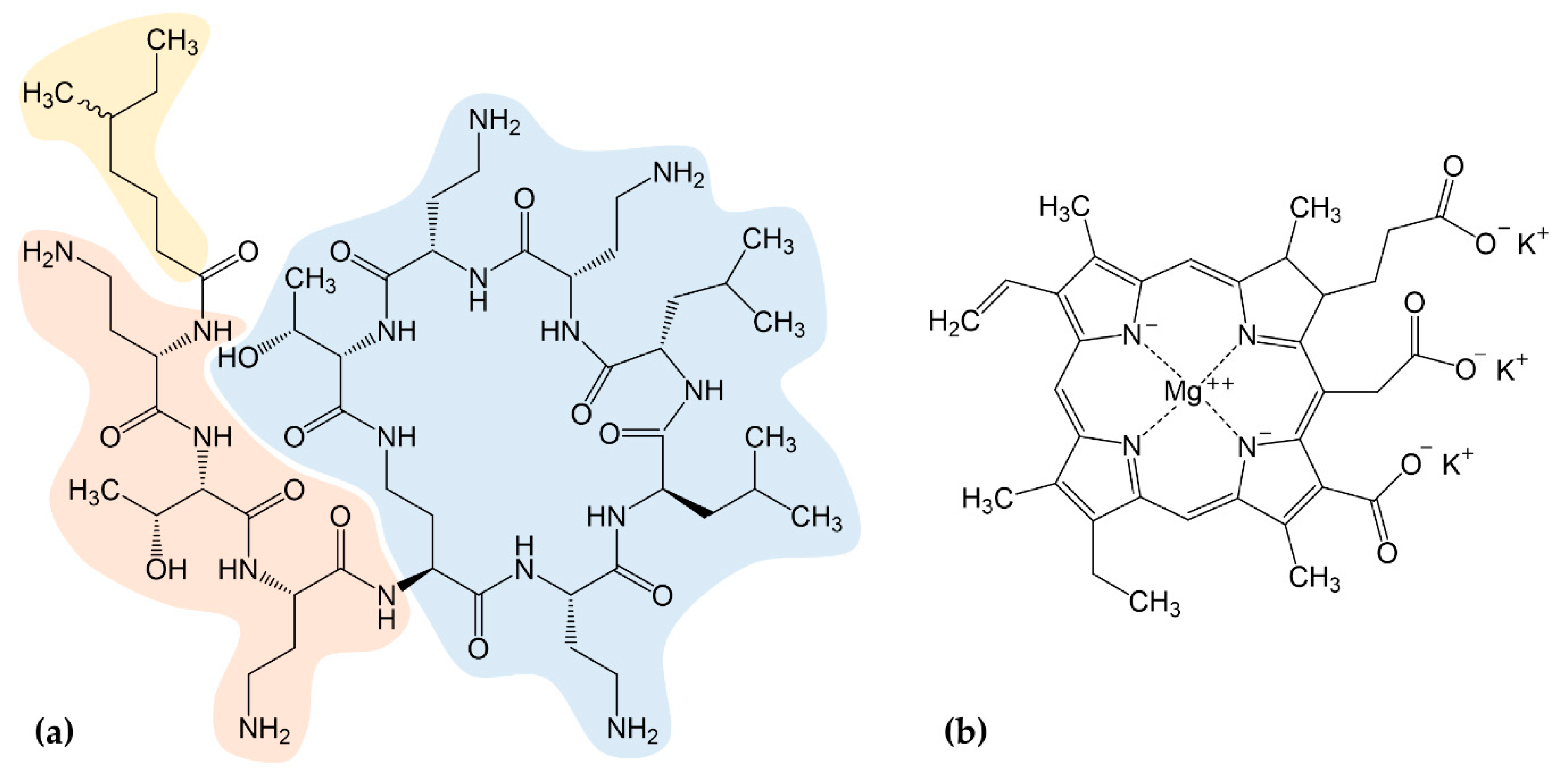
4.2. Preparation of Chlorophyllin
4.3. Growth Experiments
4.4. Determination of Minimal Inhibitory Concentrations (MICs)
4.5. NPN Uptake Assay
4.6. Fluorescence Microscopy
4.7. Data Evaluation and Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Powers, J.H. Antimicrobial drug development—The past, the present, and the future. Clin. Microbiol. Infect. 2004, 10, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Coates, A.; Hu, Y.; Bax, R.; Page, C. The future challenges facing the development of new antimicrobial drugs. Nat. Rev. Drug Discov. 2002, 1, 895. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.; Davies, D. Origins and Evolution of Antibiotic Resistance. Microbiol. Mol. Boil. Rev. 2010, 74, 417–433. [Google Scholar] [CrossRef] [PubMed]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. 2015, 40, 277. [Google Scholar] [PubMed]
- Rice, L.B. Federal Funding for the Study of Antimicrobial Resistance in Nosocomial Pathogens: No ESKAPE. J. Infect. Dis. 2008, 197, 1079–1081. [Google Scholar] [CrossRef] [PubMed]
- Katchanov, J.; Asar, L.; Klupp, E.-M.; Both, A.; Rothe, C.; König, C.; Rohde, H.; Kluge, S.; Maurer, F.P. Carbapenem-resistant Gram-negative pathogens in a German university medical center: Prevalence, clinical implications and the role of novel β-lactam/β-lactamase inhibitor combinations. PLoS ONE 2018, 13, e0195757. [Google Scholar] [CrossRef] [PubMed]
- Grundmann, H.; Glasner, C.; Albiger, B.; Aanensen, D.M.; Tomlinson, C.T.; Andrasević, A.T.; Cantón, R.; Friedrich, A.W.; Giske, C.G.; Gniadkowski, M.; et al. Occurrence of carbapenemase-producing Klebsiella pneumoniae and Escherichia coli in the European survey of carbapenemase-producing Enterobacteriaceae (EuSCAPE): A prospective, multinational study. Lancet Infect. Dis. 2017, 17, 153–163. [Google Scholar] [CrossRef]
- Ayukekbong, J.A.; Ntemgwa, M.; Atabe, A.N. The threat of antimicrobial resistance in developing countries: Causes and control strategies. Antimicrob. Resist. Infect. Control 2017, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Reardon, S. Antibiotic resistance sweeping developing world. Nat. News 2014, 509, 141. [Google Scholar] [CrossRef] [PubMed]
- Founou, L.L.; Founou, R.C.; Essack, S.Y. Antibiotic Resistance in the Food Chain: A Developing Country-Perspective. Front. Microbiol. 2016, 7, 775. [Google Scholar] [CrossRef] [PubMed]
- Tangcharoensathien, V.; Chanvatik, S.; Sommanustweechai, A. Complex determinants of inappropriate use of antibiotics. Bull. World Health Organ. 2018, 96, 141. [Google Scholar] [CrossRef] [PubMed]
- Sharland, M.; Pulcini, C.; Harbarth, S.; Zeng, M.; Gandra, S.; Mathur, S.; Magrini, N. Classifying antibiotics in the WHO Essential Medicines List for optimal use—Be AWaRe. Lancet Infect. Dis. 2018, 18, 18–20. [Google Scholar] [CrossRef]
- Yong, D.; Toleman, M.A.; Giske, C.G.; Cho, H.S.; Sundman, K.; Lee, K.; Walsh, T.R. Characterization of a New Metallo-β-Lactamase Gene, blaNDM-1, and a Novel Erythromycin Esterase Gene Carried on a Unique Genetic Structure in Klebsiella pneumoniae Sequence Type 14 from India. Antimicrob. Agents Chemother. 2009, 53, 5046–5054. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-Y.; Wang, Y.; Walsh, T.R.; Yi, L.-X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X.; et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: A microbiological and molecular biological study. Lancet Infect. Dis. 2016, 16, 161–168. [Google Scholar] [CrossRef]
- Suzuki, T.; Hayashi, K.; Fujikawa, K. Studies on the Chemical Structure of Colistin. J. Biochem. 1963, 54, 412–418. [Google Scholar] [CrossRef]
- Falagas, M.E.; Grammatikos, A.P.; Michalopoulos, A. Potential of old-generation antibiotics to address current need for new antibiotics. Expert Rev. Anti-Infect. Ther. 2008, 6, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Falagas, M.E.; Lourida, P.; Poulikakos, P.; Rafailidis, P.I.; Tansarli, G.S. Antibiotic treatment of infections due to carbapenem-resistant Enterobacteriaceae: Systematic evaluation of the available evidence. Antimicrob. Agents Chemother. 2014, 58, 654–663. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, L.A.; Garcia-Curiel, A.; Pachón-Ibáñez, M.E.; Llanos, A.C.; Ruiz, M.; Pachón, J.; Aznar, J. Reliability of the E-Test Method for Detection of Colistin Resistance in Clinical Isolates of Acinetobacter baumannii. J. Clin. Microbiol. 2005, 43, 903–905. [Google Scholar] [CrossRef]
- Charretier, Y.; Diene, S.M.; Baud, D.; Chatellier, S.; Santiago-Allexant, E.; Van Belkum, A.; Guigon, G.; Schrenzel, J. Colistin Heteroresistance and Involvement of the PmrAB Regulatory System in Acinetobacter baumannii. Antimicrob. Agents Chemother. 2018, 62, e00788-18. [Google Scholar] [CrossRef]
- Tzampaz, E.; Sianou, E.; Tzavaras, I.; Sofianou, D.; Meletis, G. Colistin heteroresistance in carbapenemase-producing Klebsiella pneumoniae. J. Antimicrob. Chemother. 2011, 66, 946–947. [Google Scholar]
- Napier, B.A.; Burd, E.M.; Satola, S.W.; Cagle, S.M.; Ray, S.M.; McGann, P.; Pohl, J.; Lesho, E.P.; Weiss, D.S. Clinical Use of Colistin Induces Cross-Resistance to Host Antimicrobials in Acinetobacter baumannii. Mbio 2013, 4, e00021-13. [Google Scholar] [CrossRef] [PubMed]
- Poudyal, A.; Howden, B.P.; Bell, J.M.; Gao, W.; Owen, R.J.; Turnidge, J.D.; Nation, R.L.; Li, J. In vitro pharmacodynamics of colistin against multidrug-resistant Klebsiella pneumoniae. J. Antimicrob. Chemother. 2008, 62, 1311–1318. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Rayner, C.R.; Nation, R.L.; Owen, R.J.; Spelman, D.; Tan, K.E.; Liolios, L. Heteroresistance to Colistin in Multidrug-Resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 2006, 50, 2946–2950. [Google Scholar] [CrossRef] [PubMed]
- Nicoloff, H.; Hjort, K.; Levin, B.R.; Andersson, D.I. The high prevalence of antibiotic heteroresistance in pathogenic bacteria is mainly caused by gene amplification. Nat. Microbiol. 2019, 4, 504. [Google Scholar] [CrossRef] [PubMed]
- Carroll, L.M.; Gaballa, A.; Guldimann, C.; Sullivan, G.; Henderson, L.O.; Wiedmann, M.; Siragusa, G.; White, D. Identification of Novel Mobilized Colistin Resistance Gene mcr-9 in a Multidrug-Resistant, Colistin-Susceptible Salmonella enterica Serotype Typhimurium Isolate. MBio 2019, 10, e00853-19. [Google Scholar] [CrossRef]
- Johnson, A.P.; Woodford, N. Global spread of antibiotic resistance: The example of New Delhi metallo-β-lactamase (NDM)-mediated carbapenem resistance. J. Med. Microbiol. 2013, 62, 499–513. [Google Scholar] [CrossRef] [PubMed]
- Hasman, H.; Hammerum, A.M.; Hansen, F.; Hendriksen, R.S.; Olesen, B.; Agersø, Y.; Zankari, E.; Leekitcharoenphon, P.; Stegger, M.; Kaas, R.S.; et al. Detection of mcr-1 encoding plasmid-mediated colistin-resistant Escherichia coli isolates from human bloodstream infection and imported chicken meat, Denmark 2015. Eurosurveillance 2015, 20, 1–5. [Google Scholar] [CrossRef]
- Wang, Y.; Tian, G.-B.; Zhang, R.; Shen, Y.; Tyrrell, J.M.; Huang, X.; Zhou, H.; Lei, L.; Li, H.-Y.; Doi, Y.; et al. Prevalence, risk factors, outcomes, and molecular epidemiology of mcr-1-positive Enterobacteriaceae in patients and healthy adults from China: An epidemiological and clinical study. Lancet Infect. Dis. 2017, 17, 390–399. [Google Scholar] [CrossRef]
- Yi, L.; Wang, J.; Gao, Y.; Liu, Y.; Doi, Y.; Wu, R.; Zeng, Z.; Liang, Z.; Liu, J.-H. mcr-1−Harboring Salmonella enterica Serovar Typhimurium Sequence Type 34 in Pigs, China. Emerg. Infect. Dis. 2017, 23, 291–295. [Google Scholar] [CrossRef]
- Luo, J.; Yao, X.; Lv, L.; Doi, Y.; Huang, X.; Huang, S.; Liu, J.-H. Emergence of mcr-1 in Raoultella ornithinolytica and Escherichia coli Isolates from Retail Vegetables in China. Antimicrob. Agents Chemother. 2017, 61, e01139-17. [Google Scholar] [CrossRef]
- Mendes, A.C.; Novais, Â.; Campos, J.; Rodrigues, C.; Santos, C.; Antunes, P.; Ramos, H.; Peixe, L. mcr-1 in Carbapenemase-Producing Klebsiella pneumoniae with Hospitalized Patients, Portugal, 2016–2017. Emerg. Infect. Dis. 2018, 24, 762. [Google Scholar] [CrossRef] [PubMed]
- Principe, L.; Piazza, A.; Mauri, C.; Anesi, A.; Bracco, S.; Brigante, G.; Casari, E.; Agrappi, C.; Caltagirone, M.; Novazzi, F.; et al. Multicenter prospective study on the prevalence of colistin resistance in Escherichia coli: Relevance of mcr-1-positive clinical isolates in Lombardy, Northern Italy. Infect. Drug Resist. 2018, 11, 377. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.-Q.; Li, Y.-X.; Lei, C.-W.; Zhang, A.-Y.; Wang, H.-N. Novel plasmid-mediated colistin resistance gene mcr-7.1 in Klebsiella pneumoniae. J. Antimicrob. Chemother. 2018, 73, 1791–1795. [Google Scholar] [CrossRef] [PubMed]
- Xavier, B.B.; Lammens, C.; Ruhal, R.; Kumar-Singh, S.; Butaye, P.; Goossens, H.; Malhotra-Kumar, S. Identification of a novel plasmid-mediated colistin-resistance gene, mcr-2, in Escherichia coli, belgium, june 2016. EuroSurveill Mon. 2016, 21, 30280. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Graells, C.; De Keersmaecker, S.C.J.; Vanneste, K.; Pochet, B.; Vermeersch, K.; Roosens, N.; Dierick, K.; Botteldoorn, N. Detection of plasmid-mediated colistin resistance, mcr-1 and mcr-2 genes, in Salmonella spp. Isolated from food at retail in Belgium from 2012 to 2015. Foodborne Pathog. Dis. 2018, 15, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Yin, W.; Li, H.; Shen, Y.; Liu, Z.; Wang, S.; Shen, Z.; Zhang, R.; Walsh, T.R.; Shen, J.; Wang, Y. Novel Plasmid-Mediated Colistin Resistance Gene mcr-3 in Escherichia coli. MBio 2017, 8, e00543-17. [Google Scholar] [CrossRef] [PubMed]
- Carattoli, A.; Villa, L.; Feudi, C.; Curcio, L.; Orsini, S.; Luppi, A.; Pezzotti, G.; Magistrali, C.F. Novel plasmid-mediated colistin resistance mcr-4 gene in salmonella and Escherichia coli, italy 2013, spain and belgium, 2015 to 2016. EuroSurveillance 2017, 22, 30589. [Google Scholar] [CrossRef] [PubMed]
- Borowiak, M.; Fischer, J.; Szabo, I.; Malorny, B.; A Hammerl, J.; Hendriksen, R.S. Identification of a novel transposon-associated phosphoethanolamine transferase gene, mcr-5, conferring colistin resistance in d-tartrate fermenting Salmonella enterica subsp. enterica serovar Paratyphi B. J. Antimicrob. Chemother. 2017, 72, 3317–3324. [Google Scholar] [CrossRef]
- Teo, J.W.P.; Kalisvar, M.; Venkatachalam, I.; Ng, O.T.; Lin, R.T.P.; Octavia, S. mcr-3 and mcr-4 Variants in Carbapenemase-Producing Clinical Enterobacteriaceae Do Not Confer Phenotypic Polymyxin Resistance. J. Clin. Microbiol. 2018, 56, e01562-17. [Google Scholar] [CrossRef]
- Fukuda, A.; Sato, T.; Shinagawa, M.; Takahashi, S.; Asai, T.; Yokota, S.-I.; Usui, M.; Tamura, Y. High prevalence of mcr-1, mcr-3 and mcr-5 in Escherichia coli derived from diseased pigs in Japan. Int. J. Antimicrob. Agents 2018, 51, 163–164. [Google Scholar] [CrossRef]
- Hammerl, J.A.; Borowiak, M.; Schmoger, S.; Shamoun, D.; Grobbel, M.; Malorny, B.; Tenhagen, B.A.; Kasbohrer, A. Mcr-5 and a novel mcr-5.2 variant in Escherichia coli isolates from food and food-producing animals, germany, 2010 to 2017. J. Antimicrob. Chemother. 2018, 73, 1433–1435. [Google Scholar] [CrossRef] [PubMed]
- Uchil, R.R.; Kohli, G.S.; Katekhaye, V.M.; Swami, O.C. Strategies to Combat Antimicrobial Resistance. J. Clin. Diagn. Res. 2014, 8, ME01. [Google Scholar] [PubMed]
- Coates, A.R.; Halls, G.; Hu, Y. Novel classes of antibiotics or more of the same? Br. J. Pharmacol. 2011, 163, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Aponiene, K.; Luksiene, Z. Effective combination of LED-based visible light, photosensitizer and photocatalyst to combat Gram (−) bacteria. J. Photochem. Photobiol. B Boil. 2015, 142, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Kreitner, M.; Wagner, K.-H.; Alth, G.; Ebermann, R.; Foißy, H.; Elmadfa, I. Haematoporphyrin- and sodium chlorophyllin-induced phototoxicity towards bacteria and yeasts—A new approach for safe foods. Food Control 2001, 12, 529–533. [Google Scholar] [CrossRef]
- Luksiene, Z.; Buchovec, I.; Paskeviciute, E. Inactivation of several strains of Listeria monocytogenes attached to the surface of packaging material by Na-Chlorophyllin-based photosensitization. J. Photochem. Photobiol. B Boil. 2010, 101, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Luksiene, Z.; Buchovec, I.; Paskeviciute, E. Inactivation of Bacillus cereus by Na-chlorophyllin-based photosensitization on the surface of packaging. J. Appl. Microbiol. 2010, 109, 1540–1548. [Google Scholar] [CrossRef]
- Luksiene, Z.; Paskeviciute, E. Microbial control of food-related surfaces: Na-Chlorophyllin-based photosensitization. J. Photochem. Photobiol. B Boil. 2011, 105, 69–74. [Google Scholar] [CrossRef]
- Luksiene, Z.; Paskeviciute, E. Novel approach to the microbial decontamination of strawberries: Chlorophyllin-based photosensitization. J. Appl. Microbiol. 2011, 110, 1274–1283. [Google Scholar] [CrossRef]
- Wohllebe, S.; Richter, R.; Richter, P.; Häder, D.P. Photodynamic control of human pathogenic parasites in aquatic ecosystems using chlorophyllin and pheophorbid as photodynamic substances. Parasitol. Res. 2009, 104, 593–600. [Google Scholar] [CrossRef]
- Häder, D.-P.; Schmidl, J.; Hilbig, R.; Oberle, M.; Wedekind, H.; Richter, P. Fighting fish parasites with photodynamically active chlorophyllin. Parasitol. Res. 2016, 115, 2277–2283. [Google Scholar] [CrossRef] [PubMed]
- Elhadad, H.A.; El-Habet, B.A.; Azab, R.M.; Abu El Einin, H.M.; Lotfy, W.M.; Atef, H.A. Effect of chlorophyllin on Biomphalaria alexandrina snails and Schistosoma mansoni larvae. Int. J. Curr. Microbiol. Appl. Sci. 2018, 7, 3725–3736. [Google Scholar] [CrossRef]
- Singh, D.K.; Singh, D.J. Photoactivated chlorophyllin and acetylcholinesterase/cytochrome oxidase activity in Fasciola gigantica cercaria larvae. Pharmacogn. J. 2018, 10, 768–772. [Google Scholar] [CrossRef]
- Xodo, L.E.; Rapozzi, V.; Zacchigna, M.; Drioli, S.; Zorzet, S. The chlorophyll catabolite pheophorbide a as a photosensitizer for the photodynamic therapy. Curr. Med. Chem. 2012, 19, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Roomi, M.W.; Bhanap, B.; Niedzwiecki, A.; Rath, M. Chlorophyllin suppresses growth, mmp secretion, invasion and cell migration of fibrosarcoma cell line ht-1080. Med. Res. Arch. 2018, 6. [Google Scholar] [CrossRef]
- Chaturvedi, D.; Singh, K.; Singh, V. Therapeutic and pharmacological aspects of photodynamic product chlorophyllin. Eur. J. Biol. Res. 2019, 9, 64–76. [Google Scholar] [CrossRef]
- Wainwright, M. Photodynamic antimicrobial chemotherapy (PACT). J. Antimicrob. Chemother. 1998, 42, 13–28. [Google Scholar] [CrossRef] [PubMed]
- Wainwright, M.; Maisch, T.; Nonell, S.; Plaetzer, K.; Almeida, A.; Tegos, G.P.; Hamblin, M.R. Photoantimicrobials-are we afraid of the light? Lancet Infect. Dis. 2017, 17, e49–e55. [Google Scholar] [CrossRef]
- Cieplik, F.; Deng, D.; Crielaard, W.; Buchalla, W.; Hellwig, E.; Al-Ahmad, A.; Maisch, T. Antimicrobial photodynamic therapy—What we know and what we don’t. Crit. Rev. Microbiol. 2018, 44, 571–589. [Google Scholar] [CrossRef]
- Krüger, M.; Richter, P.; Strauch, S.M.; Nasir, A.; Burkovski, A.; Antunes, C.A.; Meißgeier, T.; Schlücker, E.; Schwab, S.; Lebert, M. What an Escherichia coli Mutant Can Teach Us About the Antibacterial Effect of Chlorophyllin. Microorganisms 2019, 7, 59. [Google Scholar] [CrossRef] [PubMed]
- Nikaido, H. Prevention of drug access to bacterial targets: Permeability barriers and active efflux. Science 1994, 264, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Sperandio, F.F.; Huang, Y.-Y.; Hamblin, M.R. Antimicrobial Photodynamic Therapy to Kill Gram-negative Bacteria. Recent Pat. Anti-Infect. Drug Discov. 2013, 8, 108–120. [Google Scholar] [CrossRef]
- Yahav, D.; Farbman, L.; Leibovici, L.; Paul, M. Colistin: New lessons on an old antibiotic. Clin. Microbiol. Infect. 2012, 18, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Govil, D.; Gupta, S.; Malhotra, A.; Kakar, P.N.; Arora, D.; Das, S.; Govil, P.; Prakash, O. Colistin and polymyxin B: A re-emergence. Indian J. Crit. Care Med. 2009, 13, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.E. Peptide antibiotics. Lancet 1997, 349, 418–422. [Google Scholar] [CrossRef]
- Elimam, M.M.; Shantier, S.W.; Gadkariem, E.A.; Mohamed, M.A. Derivative Spectrophotometric Methods for the Analysis and Stability Studies of Colistin Sulphate. J. Chem. 2015, 2015, 1–5. [Google Scholar] [CrossRef]
- Whitfield, C.; Roberts, I.S. Structure, assembly and regulation of expression of capsules in Escherichia coli. Mol. Microbiol. 1999, 31, 1307–1319. [Google Scholar] [CrossRef] [PubMed]
- Campos, M.A.; Vargas, M.A.; Regueiro, V.; Llompart, C.M.; Albertí, S.; Bengoechea, J.A. Capsule polysaccharide mediates bacterial resistance to antimicrobial peptides. Infect. Immun. 2004, 72, 7107–7114. [Google Scholar] [CrossRef]
- Pereira, M.A.; Faustino, M.A.; Tome, J.P.; Neves, M.G.; Tome, A.C.; Cavaleiro, J.A.; Cunha, A.; Almeida, A. Influence of external bacterial structures on the efficiency of photodynamic inactivation by a cationic porphyrin. Photochem. Photobiol. Sci. 2014, 13, 680–690. [Google Scholar] [CrossRef]
- Pages, J.-M.; James, C.E.; Winterhalter, M. The porin and the permeating antibiotic: A selective diffusion barrier in Gram-negative bacteria. Nat. Rev. Genet. 2008, 6, 893. [Google Scholar] [CrossRef]
- Stavenger, R.A.; Winterhalter, M. TRANSLOCATION Project: How to Get Good Drugs into Bad Bugs. Sci. Transl. Med. 2014, 6, 228. [Google Scholar] [CrossRef]
- Heesterbeek, D.A.C.; Martin, N.I.; Velthuizen, A.; Duijst, M.; Ruyken, M.; Wubbolts, R.; Rooijakkers, S.H.M.; Bardoel, B.W. Complement-dependent outer membrane perturbation sensitizes Gram-negative bacteria to Gram-positive specific antibiotics. Sci. Rep. 2019, 9, 3074. [Google Scholar] [CrossRef]
- Delcour, A.H. Outer membrane permeability and antibiotic resistance. Biochim. Biophys. Acta 2009, 1794, 808–816. [Google Scholar] [CrossRef]
- Vaara, M. Agents that increase the permeability of the outer membrane. Microbiol. Rev. 1992, 56, 395–411. [Google Scholar]
- O’Driscoll, N.H.; Cushnie, T.P.T.; Matthews, K.H.; Lamb, A.J. Colistin causes profound morphological alteration but minimal cytoplasmic membrane perforation in populations of Escherichia coli and Pseudomonas aeruginosa. Arch. Microbiol. 2018, 200, 793–802. [Google Scholar] [CrossRef]
- He, Y.; Huang, Y.-Y.; Xi, L.; Gelfand, J.A.; Hamblin, M.R. Tetracyclines function as dual-action light-activated antibiotics. PLoS ONE 2018, 13, e0196485. [Google Scholar] [CrossRef]
- Greenstein, M.; Monji, T.; Yeung, R.; Maiese, W.M.; White, R.J. Light-dependent activity of the antitumor antibiotics ravidomycin and desacetylravidomycin. Antimicrob. Agents Chemother. 1986, 29, 861–866. [Google Scholar] [CrossRef]
- Helander, I.; Nurmiaho-Lassila, E.-L.; Ahvenainen, R.; Rhoades, J.; Roller, S. Chitosan disrupts the barrier properties of the outer membrane of Gram-negative bacteria. Int. J. Food Microbiol. 2001, 71, 235–244. [Google Scholar] [CrossRef]
- Buchovec, I.; Lukseviciūtė, V.; Kokstaite, R.; Labeikyte, D.; Kaziukonyte, L.; Luksiene, Z. Inactivation of Gram (−) bacteria Salmonella enterica by chlorophyllin-based photosensitization: Mechanism of action and new strategies to enhance the inactivation efficiency. J. Photochem. Photobiol. B Boil. 2017, 172, 1–10. [Google Scholar] [CrossRef]
- Lukseviciute, V.; Buchovec, I.; Marsalka, A.; Reklaitis, I.; Luksiene, Z. Effective photosensitization-based inactivation of Gram (−) food pathogens and molds using the chlorophyllin–chitosan complex: Towards photoactive edible coatings to preserve strawberries. Photochem. Photobiol. Sci. 2016, 15, 506–516. [Google Scholar]
- Alakomi, H.-L.; Skyttä, E.; Saarela, M.; Mattila-Sandholm, T.; Latva-Kala, K.; Helander, I.M. Lactic Acid Permeabilizes Gram-Negative Bacteria by Disrupting the Outer Membrane. Appl. Environ. Microbiol. 2000, 66, 2001–2005. [Google Scholar] [CrossRef]
- Helander, I.; Von Wright, A.; Mattila-Sandholm, T.-M. Potential of lactic acid bacteria and novel antimicrobials against Gram-negative bacteria. Trends Food Sci. Technol. 1997, 8, 146–150. [Google Scholar] [CrossRef]
- Volzing, K.; Borrero, J.; Sadowsky, M.J.; Kaznessis, Y.N. Antimicrobial Peptides Targeting Gram-negative Pathogens, Produced and Delivered by Lactic Acid Bacteria. ACS Synth. Boil. 2013, 2, 643–650. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, C.; Peng, P.; Hossain, M.; Jiang, T.; Fu, W.; Liao, Y.; Su, M. Visible light mediated killing of multidrug-resistant bacteria using photoacids. J. Mater. Chem. B 2013, 1, 997–1001. [Google Scholar] [CrossRef]
- Nitzan, Y.; Gutterman, M.; Malik, Z.; Ehrenberg, B. inactivation of gram-negative bacteria by photosensitized porphyrins. Photochem. Photobiol. 1992, 55, 89–96. [Google Scholar] [CrossRef]
- Le Guern, F.; Sol, V.; Ouk, C.; Arnoux, P.; Frochot, C.; Ouk, T.-S. Enhanced Photobactericidal and Targeting Properties of a Cationic Porphyrin following the Attachment of Polymyxin B. Bioconjugate Chem. 2017, 28, 2493–2506. [Google Scholar] [CrossRef]
- Alakomi, H.-L.; Paananen, A.; Suihko, M.-L.; Helander, I.M.; Saarela, M. Weakening Effect of Cell Permeabilizers on Gram-Negative Bacteria Causing Biodeterioration. Appl. Environ. Microbiol. 2006, 72, 4695–4703. [Google Scholar] [CrossRef]
- Gruskin, B. Chlorophyll—Its therapeutic place in acute and suppurative disease: Preliminary report of clinical use and rationale. Am. J. Surg. 1940, 49, 49–55. [Google Scholar] [CrossRef]
- Yoshida, A.; Yokono, O.; Oda, T. Therapeutic effect of chlorophyll-a in the treatment of patients with chronic pancreatitis. Gastroenterol. Jpn. 1980, 15, 49–61. [Google Scholar] [CrossRef]
- Ozger, H.S.; Cuhadar, T.; Yildiz, S.S.; Gulmez, Z.D.; Dizbay, M.; Tunccan, O.G.; Kalkanci, A.; Simsek, H.; Unaldi, O. In vitro activity of eravacycline in combination with colistin against carbapenem-resistant A. baumannii isolates. J. Antibiot. 2019, 72, 600–604. [Google Scholar] [CrossRef]
- Cikman, A.; Gulhan, B.; Aydin, M.; Ceylan, M.R.; Parlak, M.; Karakeçili, F.; Karagoz, A. In vitro Activity of Colistin in Combination with Tigecycline against Carbapenem-Resistant Acinetobacter baumannii Strains Isolated from Patients with Ventilator-Associated Pneumonia. Int. J. Med. Sci. 2015, 12, 695–700. [Google Scholar] [CrossRef]
- Fan, B.; Guan, J.; Wang, X.; Cong, Y. Activity of Colistin in Combination with Meropenem, Tigecycline, Fosfomycin, Fusidic Acid, Rifampin or Sulbactam against Extensively Drug-Resistant Acinetobacter baumannii in a Murine Thigh-Infection Model. PLoS ONE 2016, 11, e0157757. [Google Scholar] [CrossRef]
- Tascini, C.; Tagliaferri, E.; Giani, T.; Leonildi, A.; Flammini, S.; Casini, B.; Lewis, R.; Ferranti, S.; Rossolini, G.M.; Menichetti, F. Synergistic Activity of Colistin plus Rifampin against Colistin-Resistant KPC-Producing Klebsiella pneumoniae. Antimicrob. Agents Chemother. 2013, 57, 3990–3993. [Google Scholar] [CrossRef]
- Brennan-Krohn, T.; Pironti, A.; Kirby, J.E. Synergistic Activity of Colistin-Containing Combinations against Colistin-Resistant Enterobacteriaceae. Antimicrob. Agents Chemother. 2018, 62, e00873-18. [Google Scholar] [CrossRef]
- Macnair, C.R.; Stokes, J.M.; Carfrae, L.A.; Fiebig-Comyn, A.A.; Coombes, B.K.; Mulvey, M.R.; Brown, E.D. Overcoming mcr-1 mediated colistin resistance with colistin in combination with other antibiotics. Nat. Commun. 2018, 9, 458. [Google Scholar] [CrossRef]
- Cannatelli, A.; Principato, S.; Colavecchio, O.L.; Pallecchi, L.; Rossolini, G.M. Synergistic Activity of Colistin in Combination with Resveratrol Against Colistin-Resistant Gram-Negative Pathogens. Front. Microbiol. 2018, 9, 1808. [Google Scholar] [CrossRef]
- Ingólfsson, H.I.; Thakur, P.; Herold, K.F.; Hobart, E.A.; Ramsey, N.B.; Periole, X.; De Jong, D.H.; Zwama, M.; Yilmaz, D.; Hall, K.; et al. Phytochemicals Perturb Membranes and Promiscuously Alter Protein Function. ACS Chem. Boil. 2014, 9, 1788–1798. [Google Scholar] [CrossRef]
- Vaara, M. Polymyxin Derivatives that Sensitize Gram-Negative Bacteria to Other Antibiotics. Molecules 2019, 24, 249. [Google Scholar] [CrossRef]
- Vaara, M.; Vaara, T. Sensitization of Gram-negative bacteria to antibiotics and complement by a nontoxic oligopeptide. Nature 1983, 303, 526. [Google Scholar] [CrossRef]
- Stokes, J.M.; Macnair, C.R.; Ilyas, B.; French, S.; Côté, J.-P.; Bouwman, C.; Farha, M.A.; Sieron, A.O.; Whitfield, C.; Coombes, B.K.; et al. Pentamidine sensitizes Gram-negative pathogens to antibiotics and overcomes acquired colistin resistance. Nat. Microbiol. 2017, 2, 17028. [Google Scholar] [CrossRef]
- Trimble, M.J.; Mlynárčik, P.; Kolář, M.; Hancock, R.E.W. Polymyxin: Alternative mechanisms of action and resistance. Cold Spring Harb Perspect. Med. 2016, 6, a025288. [Google Scholar] [CrossRef]
- Pourhajibagher, M.; Kazemian, H.; Chiniforush, N.; Bahador, A. Evaluation of photodynamic therapy effect along with colistin on pandrug-resistant Acinetobacter baumannii. Laser Ther. 2017, 26, 97–103. [Google Scholar] [CrossRef]
- Sun, J.; Zhang, H.; Liu, Y.-H.; Feng, Y. Towards Understanding MCR-like Colistin Resistance. Trends Microbiol. 2018, 26, 794–808. [Google Scholar] [CrossRef]
- Matuschek, E.; Åhman, J.; Webster, C.; Kahlmeter, G. Antimicrobial susceptibility testing of colistin—Evaluation of seven commercial MIC products against standard broth microdilution for Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, and Acinetobacter spp. Clin. Microbiol. Infect. 2018, 24, 865–870. [Google Scholar] [CrossRef]
- Poirel, L.; Jayol, A.; Nordmann, P. Polymyxins: Antibacterial Activity, Susceptibility Testing, and Resistance Mechanisms Encoded by Plasmids or Chromosomes. Clin. Microbiol. Rev. 2017, 30, 557–596. [Google Scholar] [CrossRef]
- Ziegler, R.; Egle, K. Zur quantitativen Analyse der Chloroplastenpigmente I. Beitr. Biol. Pflanzen. 1965, 41, 11–37. [Google Scholar]
- Vadakedath, N.; Halami, P.M. Characterization and mode of action of a potent bio-preservative from food-grade Bacillus licheniformis MCC 2016. Prep. Biochem. Biotechnol. 2019, 49, 334–343. [Google Scholar] [CrossRef]
- Helander, I.M.; Mattila-Sandholm, T. Fluorometric assessment of Gram-negative bacterial permeabilization. J. Appl. Microbiol. 2000, 88, 213–219. [Google Scholar] [CrossRef]
- Di Veroli, G.Y.; Fornari, C.; Wang, D.; Mollard, S.; Bramhall, J.L.; Richards, F.M.; Jodrell, D.I. Combenefit: An interactive platform for the analysis and visualization of drug combinations. Bioinformatics 2016, 32, 2866–2868. [Google Scholar] [CrossRef]




| Strain | Illuminated with 12 mW/cm2 | Dark-Incubated |
|---|---|---|
| E. coli DH5α | 5 mg/L chlorophyllin + 0.125 µg/mL colistin or 2.5 mg/L chlorophyllin + 0.25 µg/mL colistin or 1.25 mg/L chlorophyllin + 0.5 µg/mL colistin | 10 mg/L chlorophyllin + 0.25 µg/mL colistin or 2.5 mg/L chlorophyllin + 0.5 µg/mL colistin |
| E. coli DH5α pGDP2:mcr-1 | 10 mg/L chlorophyllin + 2 mg/L colistin or 5 mg/L chlorophyllin + 4 µg/mL colistin | no inactivation at low chlorophyllin concentrations; observed inactivation with 20 mg/L chlorophyllin + 4 µg/mL colistin |
| S. Typhimurium Ames9274 | 1 mg/L chlorophyllin 1 or 0.063 chlorophyllin + 0.25 µg/mL colistin or 0.125 chlorophyllin + 0.5 µg/mL colistin | 1 mg/L chlorophyllin + 0.25 µg/mL colistin or 0.125 mg/L chlorophyllin + 0.5 µg/mL colistin |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richter, P.; Krüger, M.; Prasad, B.; Gastiger, S.; Bodenschatz, M.; Wieder, F.; Burkovski, A.; Geißdörfer, W.; Lebert, M.; Strauch, S.M. Using Colistin as a Trojan Horse: Inactivation of Gram-Negative Bacteria with Chlorophyllin. Antibiotics 2019, 8, 158. https://doi.org/10.3390/antibiotics8040158
Richter P, Krüger M, Prasad B, Gastiger S, Bodenschatz M, Wieder F, Burkovski A, Geißdörfer W, Lebert M, Strauch SM. Using Colistin as a Trojan Horse: Inactivation of Gram-Negative Bacteria with Chlorophyllin. Antibiotics. 2019; 8(4):158. https://doi.org/10.3390/antibiotics8040158
Chicago/Turabian StyleRichter, Peter, Marcus Krüger, Binod Prasad, Susanne Gastiger, Mona Bodenschatz, Florian Wieder, Andreas Burkovski, Walter Geißdörfer, Michael Lebert, and Sebastian M. Strauch. 2019. "Using Colistin as a Trojan Horse: Inactivation of Gram-Negative Bacteria with Chlorophyllin" Antibiotics 8, no. 4: 158. https://doi.org/10.3390/antibiotics8040158
APA StyleRichter, P., Krüger, M., Prasad, B., Gastiger, S., Bodenschatz, M., Wieder, F., Burkovski, A., Geißdörfer, W., Lebert, M., & Strauch, S. M. (2019). Using Colistin as a Trojan Horse: Inactivation of Gram-Negative Bacteria with Chlorophyllin. Antibiotics, 8(4), 158. https://doi.org/10.3390/antibiotics8040158