Novel Aspects of Polynucleotide Phosphorylase Function in Streptomyces
Abstract
1. Introduction
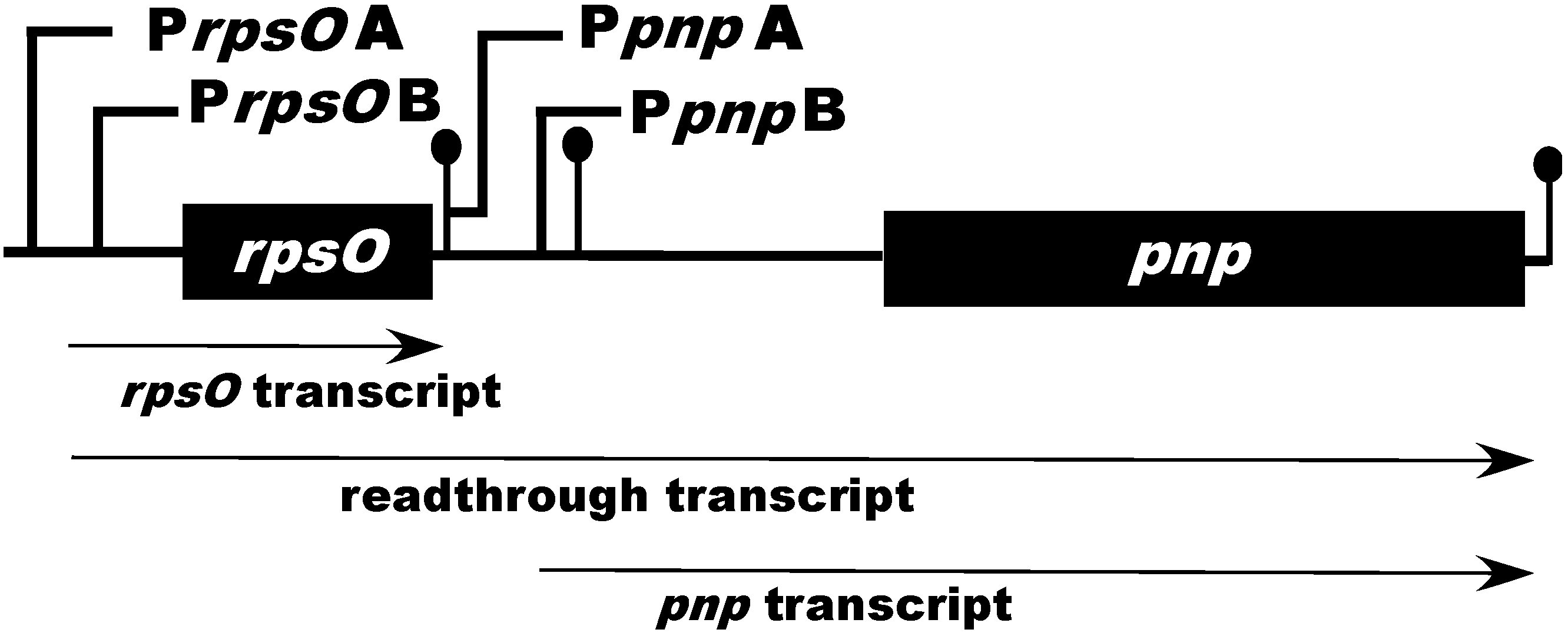
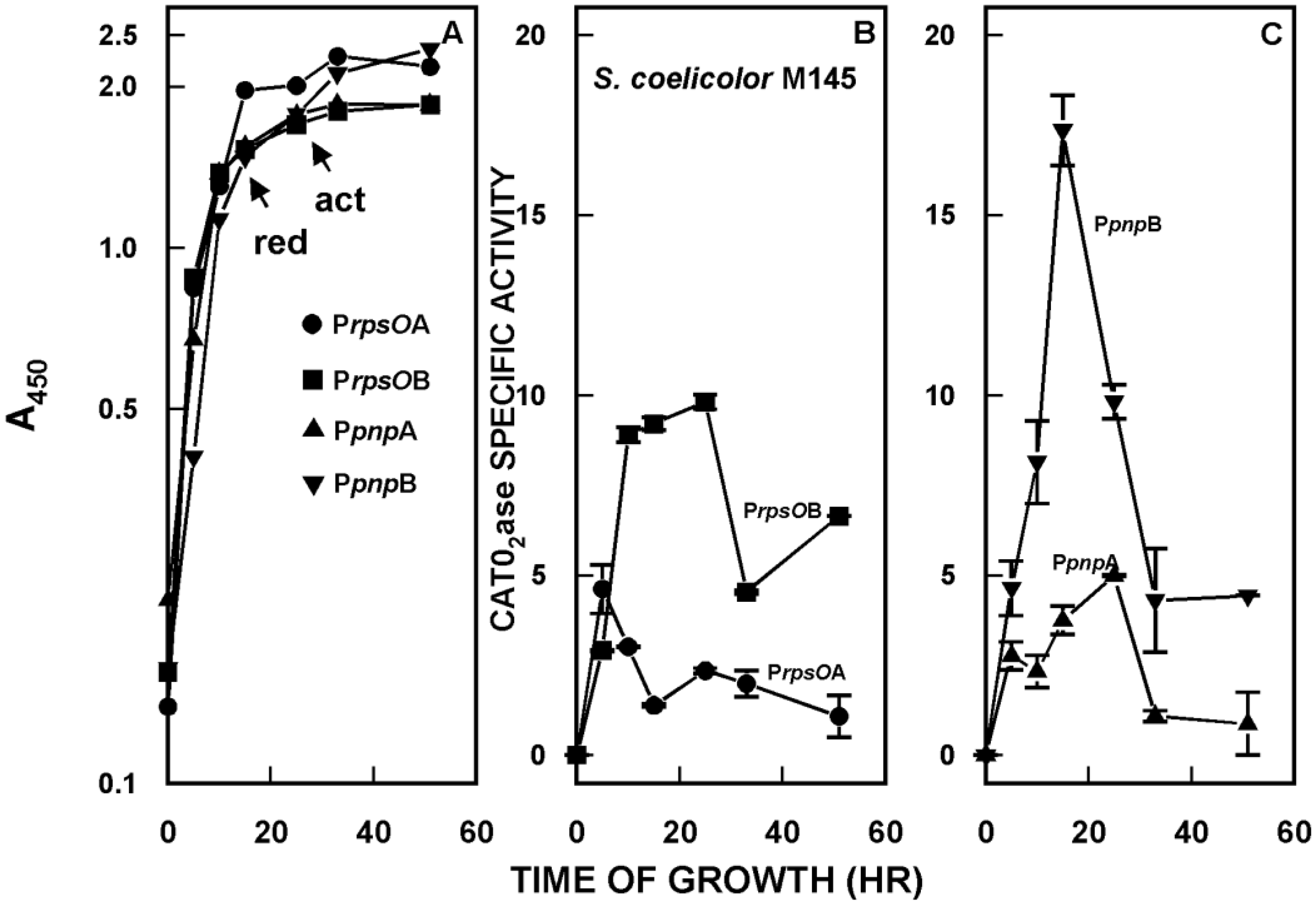
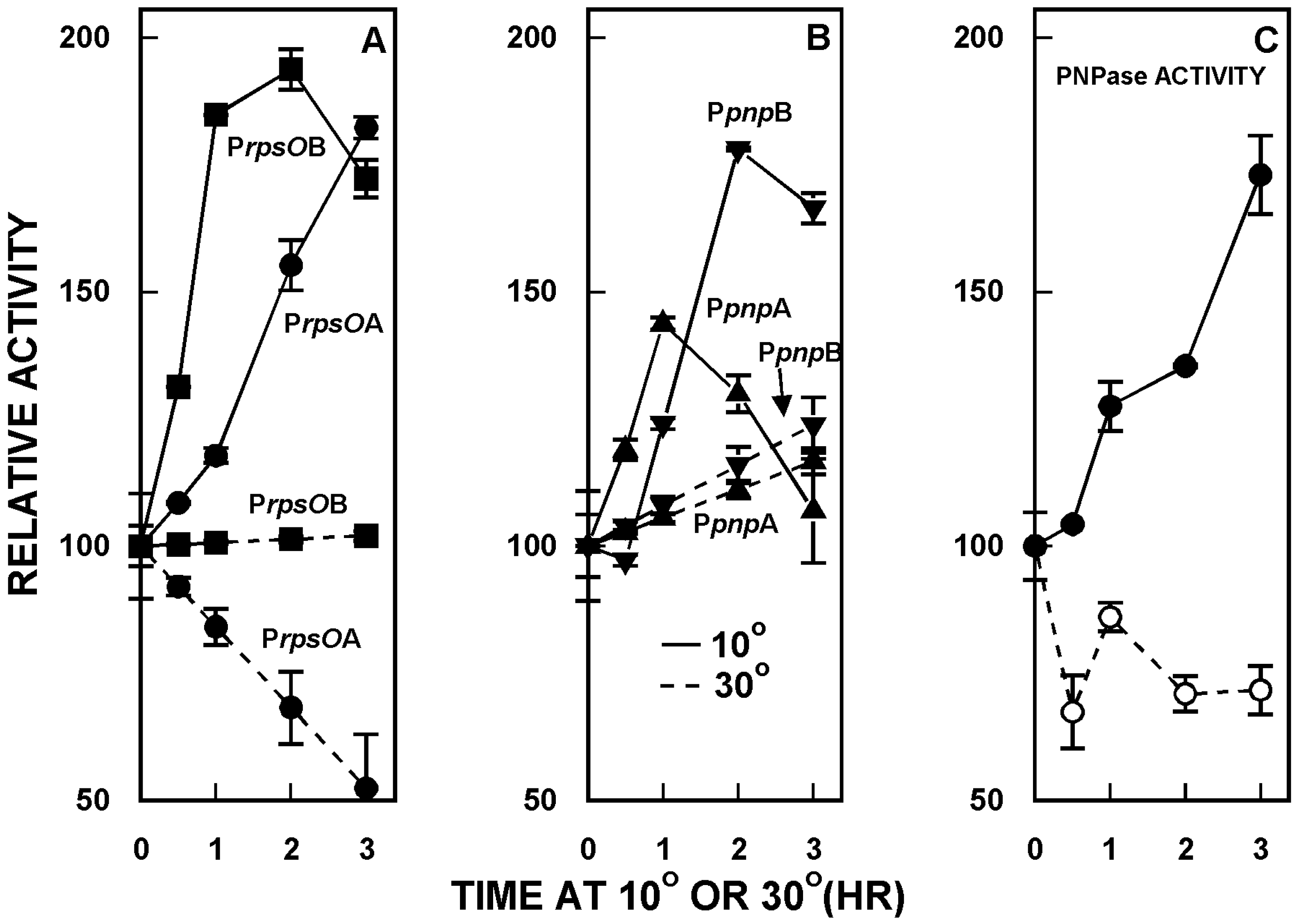
2. Organization and Expression of the PNPase Gene in Streptomyces
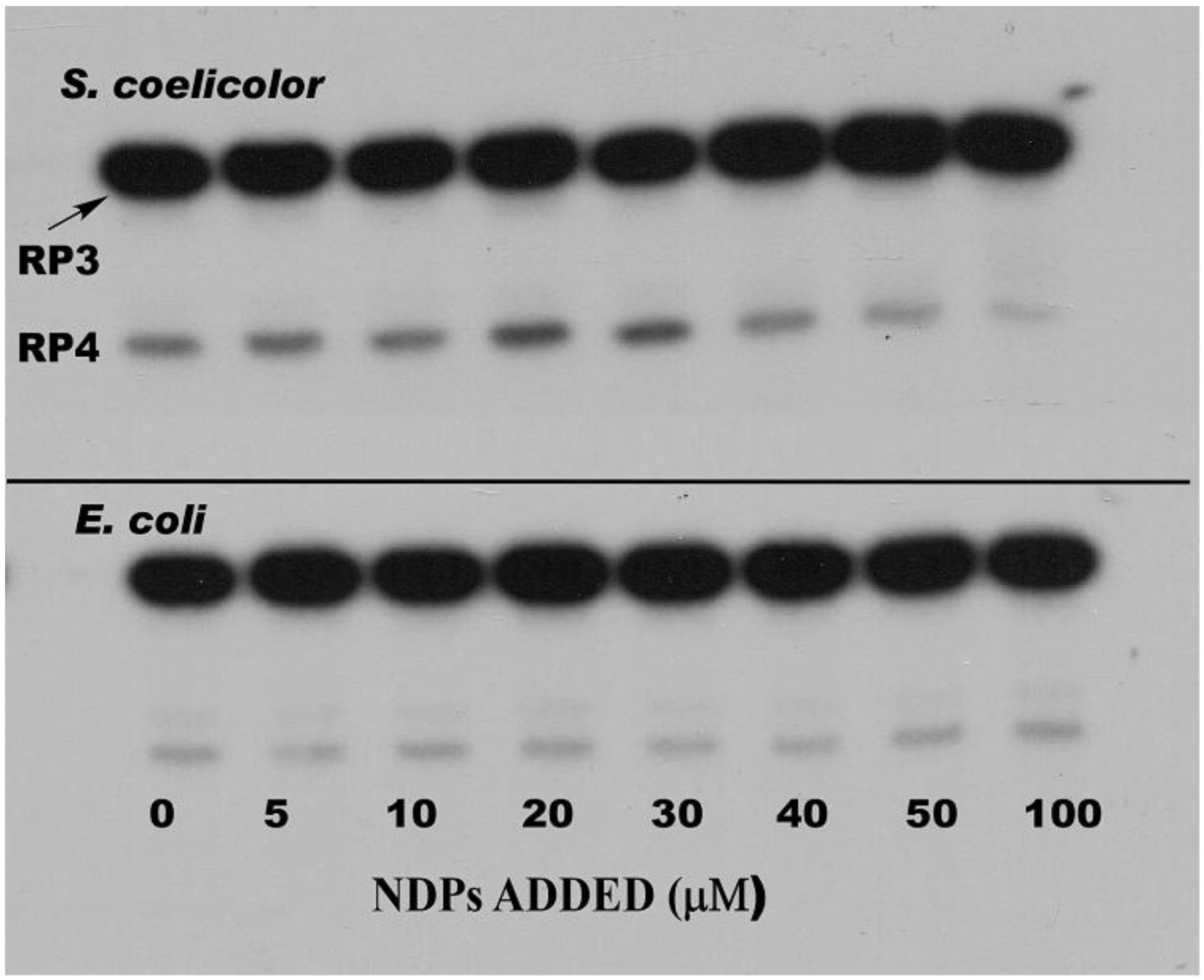
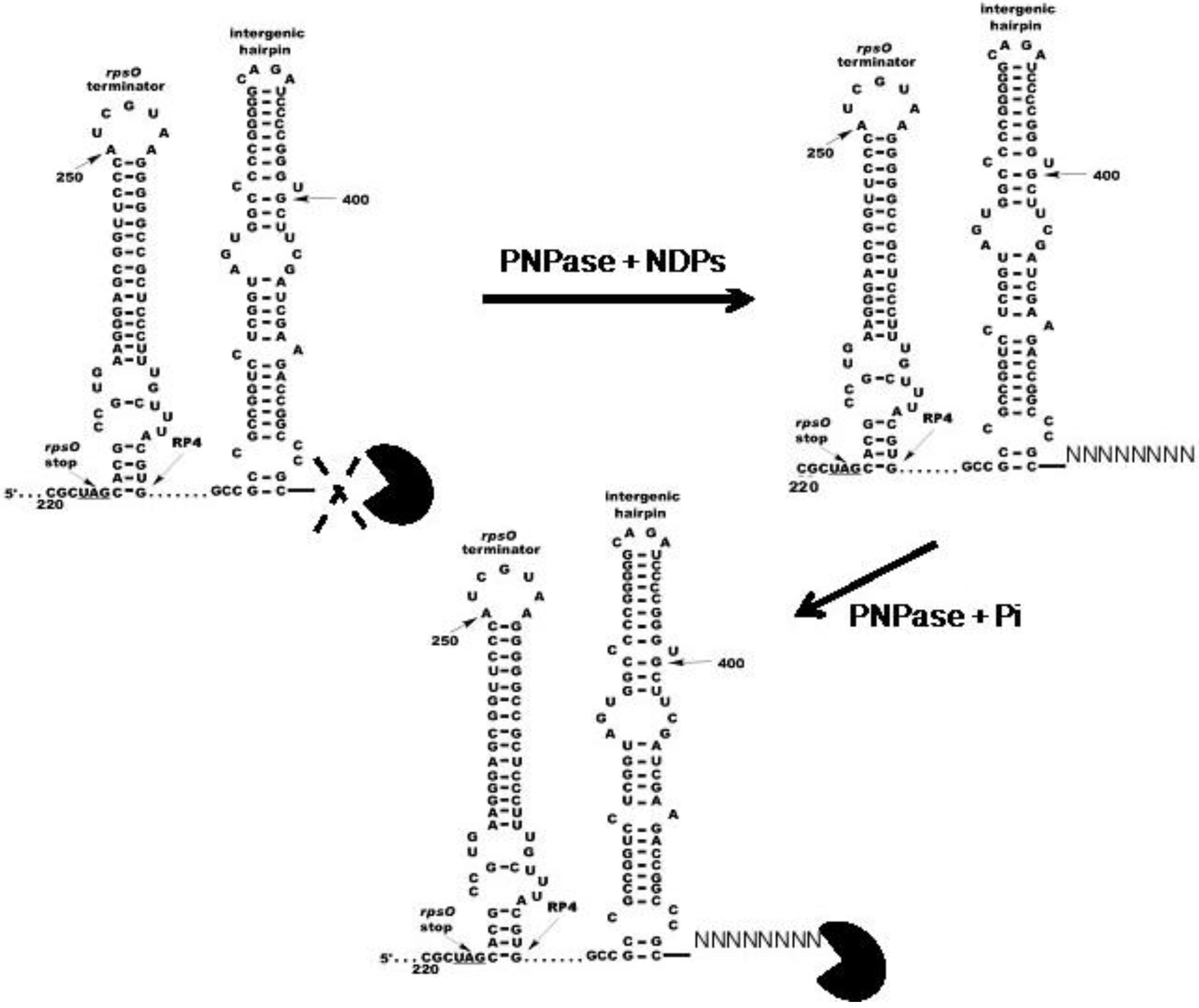
3. PNPase Function as an RNA 3′-Polyribonucleotide Polymerase in Streptomyces
4. Function of PNPase as Both an Exoribonuclease and as an RNA 3′-Polyribonucleotide Polymerase
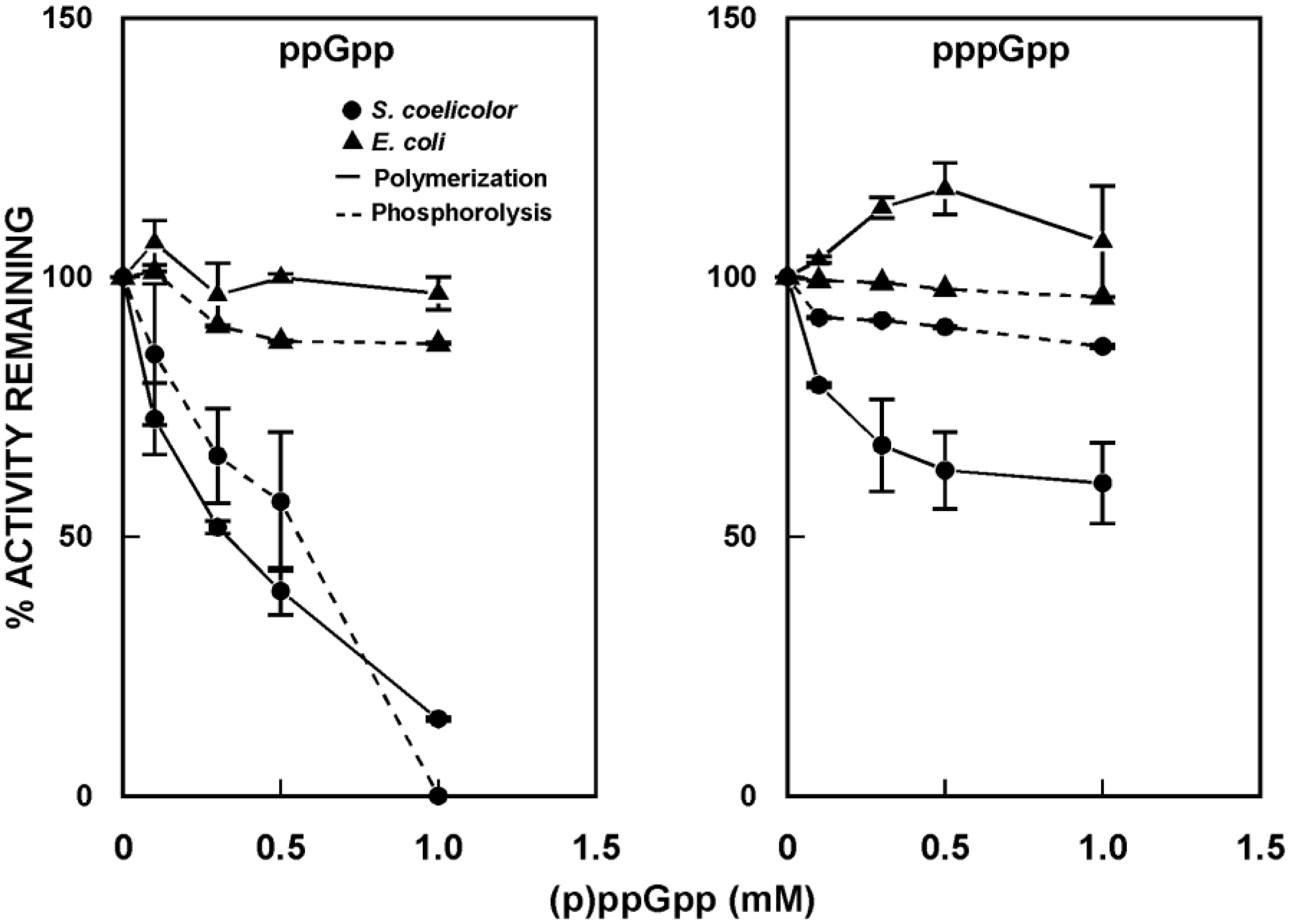
5. (p)ppGpp as a PNPase Effector
6. Conclusions and Unanswered Questions
Acknowledgments
Conflicts of Interest
References
- Grunberg-Manago, M.; Ochoa, S. Enzymatic synthesis and breakdown of polynucleotides: Polynucleotide phosphorylase. J. Am. Chem. Soc. 1955, 77, 3165–3166. [Google Scholar] [CrossRef]
- Godefroy-Colburn, T.; Grunberg-Manago, M. Polynucleotide phosphorylase. Enzymes 1972, 7, 533–574. [Google Scholar]
- Littauer, U.Z.; Soreq, H. Polynucleotide phosphorylase. Enzymes 1982, 15, 517–553. [Google Scholar]
- Lengyel, P.; Speyer, J.F.; Ochoa, S. Synthetic polynucleotides and the amino acid code. Proc. Natl. Acad. Sci. USA 1962, 47, 1936–1942. [Google Scholar] [CrossRef]
- Matthaei, J.H.; Jones, O.W.; Martin, R.G.; Nirenberg, M. Characteristics and composition of RNA coding units. Proc. Natl. Acad. Sci. USA 1962, 48, 666–676. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Y.; Deutscher, M. Exoribonuclease superfamilies: Structural analysis and phylogenetic distribution. Nucleic Acids. Res. 2001, 29, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Lin-Chao, S.; Chiou, N.T.; Schuster, G. The PNPase, exosome and RNA helicases as the building components of evolutionarily-conserved RNA degradation machines. J. Biomed. Sci. 2007, 14, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Donovan, W.P.; Kushner, S.R. Polynucleotide phosphorylase and ribonuclease II are required for cell viability and mRNA turnover in Escherichia coli k-12. Proc. Natl. Acad. Sci. USA 1986, 83, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Clarke, D.J.; Dowds, B.C. The gene coding for polynucleotide phosphorylase in Photorhabdus sp. Strain k122 is induced at low temperatures. J. Bacteriol. 1994, 176, 3775–3784. [Google Scholar] [CrossRef] [PubMed]
- Goverde, R.L.J.; Huis in’t Veld, J.H.J.; Kusters, J.G.; Mooi, F.R. The psychrotropic bacterium Yersinia enterocolitica requires expression of pnp, the gene for polynucleotide phosphorylase, for growth at low termperature (5 °C). Mol. Microbiol. 1998, 28, 555–569. [Google Scholar] [CrossRef] [PubMed]
- Zangrossi, S.; Briani, F.; Ghisotti, D.; Regonesi, M.E.; Tortora, P.; Deho, G. Transcriptional and post-transcriptional control of polynucleotide phosphorylase during cold acclimation in Escherichia coli. Mol. Microbiol. 2000, 36, 1470–1480. [Google Scholar] [CrossRef] [PubMed]
- Carzaniga, T.; Antoniani, D.; Deho, G.; Briani, F.; Landini, P. The RNA processing enzyme polynucleotide phosphorylase negatively controls biofilm formation by repressing poly-N-acetylglucosamine (PNAG) production in Escherichia coli C. BMC Microbiol. 2012, 12, 270. [Google Scholar] [CrossRef] [PubMed]
- Pobre, V.; Arraiano, C.M. Next generation sequencing analysis reveals that the ribonucleases RNAse II, RNAse R and PNPase affect bacterial motility and biofilm formation in E. coli. BMC Genomics 2015, 16, 72. [Google Scholar] [CrossRef] [PubMed]
- Rosenzweig, J.A.; Chopra, A.K. The exoribonuclease polynucleotide phosphorylase influences the virulence and stress responses of Yersiniae and many other pathogens. Front. Cell. Infect. Microbiol. 2013, 3, 81. [Google Scholar] [CrossRef] [PubMed]
- Engman, J.; Negrea, A.; Sigurlasdottir, S.; Georg, M.; Eriksson, J.; Eriksson, O.S.; Kuwae, A.; Sjolinder, H.; Jonsson, A.B. Neisseria meningitidis polynucleotide phosphorylase affects aggregation, adhesion, and virulence. Infect. Immun. 2016, 84, 1501–1513. [Google Scholar] [CrossRef] [PubMed]
- Del Favero, M.; Mazzantini, E.; Briani, F.; Zangrossi, S.; Tortora, P.; Deho, G. Regulation of Escherichia coli polynucleotide phosphorylase by ATP. J. Biol. Chem. 2008, 283, 27355–27359. [Google Scholar] [CrossRef] [PubMed]
- Nurmohamed, S.; Vincent, H.A.; Titman, C.M.; Chandran, V.; Pears, M.R.; Du, D.; Griffin, J.L.; Callaghan, A.J.; Luisi, B.F. Polynucleotide phosphorylase activity may be modulated by metabolites in Escherichia coli. J. Biol. Chem. 2011, 286, 14315–14323. [Google Scholar] [CrossRef] [PubMed]
- Tuckerman, J.R.; Gonzalez, G.; Gilles-Gonzalez, M.A. Cyclic di-GMP activation of polynucleotide phosphorylase signal-dependent RNA processing. J. Mol. Biol. 2011, 407, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Stone, C.M.; Butt, L.E.; Bufton, J.C.; Lourenco, D.C.; Gowers, D.M.; Pickford, A.R.; Cox, P.A.; Vincent, H.A.; Callaghan, A.J. Inhibition of homologous phosphorolytic ribonucleases by citrate may represent an evolutionarily conserved communicative link between RNA degradation and central metabolism. Nucleic Acids Res. 2017, 45, 4655–4666. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Chater, K.F.; Chandra, G.; Niu, G.; Tan, H. Molecular regulation of antibiotic biosynthesis in Streptomyces. Microbiol. Mol. Biol. Rev. 2013, 77, 112–143. [Google Scholar] [CrossRef] [PubMed]
- Chandra, G.; Chater, K.F. Developmental biology of Streptomyces from the perspective of 100 actinobacterial genome sequences. FEMS Microbiol. Rev. 2014, 38, 345–379. [Google Scholar] [CrossRef] [PubMed]
- Berdy, J. Recent advances in and prospects of antibiotic research. Process. Biochem. 1980, 15, 28–35. [Google Scholar]
- Littauer, U.Z. From polynucleotide phosphorylase to neurobiology. J. Biol. Chem. 2005, 280, 38889–38897. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Briani, F.; Carzaniga, T.; Deho, G. Regulation and functions of bacterial PNPase. Wiley Interdiscip. Rev. RNA 2016, 7, 241–258. [Google Scholar] [CrossRef] [PubMed]
- Régnier, P.; Portier, C. Initiation, attenuation and RNase III processing of transcripts from the Escherichia coli operon encoding ribosomal protein S15 and polynucleotide phosphorylase. J. Mol. Biol. 1986, 187, 23–32. [Google Scholar] [CrossRef]
- Luttinger, A.; Hahn, J.; Dubnau, D. Polynucleotide phosphorylase is necessary for competence development in Bacillus subtilis. Mol. Microbiol. 1996, 19, 343–356. [Google Scholar] [CrossRef] [PubMed]
- Portier, C.; Regnier, P. Expression of the rpsO and pnp genes: Structural analysis of a DNA fragment carrying their control regions. Nucleic Acids Res. 1984, 12, 6091–6102. [Google Scholar] [CrossRef] [PubMed]
- Robert-Le Meur, M.; Portier, C. E. coli polynucleotide phosphorylase expression is autoregulated through an RNase III-dependent mechanism. EMBO J. 1992, 11, 2633–2641. [Google Scholar] [PubMed]
- Robert-Le Meur, M.; Portier, C. Polynucleotide phosphorylase of Escherichia coli induces degradation of its RNase III processed messenger by preventing its translation. Nucleic Acids Res. 1994, 22, 397–403. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jarrige, A.-C.; Mathy, N.; Portier, C. Pnpase autocontrols its expression by degrading a double-stranded structure in the pnp mRNA leader. EMBO J. 2001, 20, 6845–6855. [Google Scholar] [CrossRef] [PubMed]
- Bralley, P.; Gatewood, M.L.; Jones, G.H. Transcription of the rpsO-pnp operon of Streptomyces coelicolor involves four temporally regulated, stress responsive promoters. Gene 2014, 536, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Strohl, W.R. Compilation and analysis of DNA sequences associated with apparent streptomycete promoters. Nucleic Acids Res. 1992, 20, 961–974. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, H.; Ohnishi, Y.; Horinouchi, S. Transcriptional switch on of ssgA by a-factor, which is essential for spore septum formation in Streptomyces griseus. J. Bacteriol. 2003, 185, 1273–1283. [Google Scholar] [CrossRef] [PubMed]
- Seghezzi, N.; Amar, P.; Koebmann, B.; Jensen, P.R.; Virolle, M.J. The construction of a library of synthetic promoters revealed some specific features of strong Streptomyces promoters. Appl. Microbiol. Biotechnol. 2011, 90, 615–623. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.H. Integrative, xyle-based promoter probe vectors for use in Streptomyces. Plasmid 2011, 65, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Liu, C.; Zhu, J.; Liu, W. Connecting metabolic pathways: Sigma factors in Streptomyces spp. Front. Microbiol. 2017, 8, 2546. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, B.K.; Kushner, S.R. Bacterial/archaeal/organellar polyadenylation. Wiley Interdiscip. Rev. RNA 2011, 2, 256–276. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.J.; Sarkar, N. Identification of the gene for an Escherichia coli poly(A) polymerase. Proc. Natl. Acad. Sci. USA 1992, 89, 10380–10384. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.J.; Kalapos, M.P.; Sarkar, N. Polyadenylated mrna in Escherichia coli: Modulation of poly(A) RNA levels by polynucleotide phosphorylase and ribonuclease II. Biochimie 1997, 79, 211–220. [Google Scholar] [CrossRef]
- Mohanty, B.K.; Kushner, S.R. Polynucleotide phosphorylase functions both as a 3′-5′ exonuclease and a poly(A) polymerase in Escherichia coli. Proc. Natl. Acad. Sci. USA 2000, 97, 11966–11971. [Google Scholar] [CrossRef] [PubMed]
- Bralley, P.; Jones, G.H. cDNA cloning confirms the polyadenylation of RNA decay intermediates in Streptomyces coelicolor. Microbiology 2002, 148, 1421–1425. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bralley, P.; Gust, B.; Chang, S.A.; Chater, K.F.; Jones, G.H. RNA 3′-tail synthesis in streptomyces: In vitro and in vivo activities of RNase PH, the SCO3896 gene product and pnpase. Microbiology 2006, 152, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Yehudai-Resheff, S.; Hirsh, M.; Schuster, G. Polynucleotide phosphorylase functions as both an exonuclease and a poly(A) polymerase in spinach chloroplasts. Mol. Cell Biol. 2001, 21, 5408–5416. [Google Scholar] [CrossRef] [PubMed]
- Rott, R.; Zipor, G.; Portnoy, V.; Liveanu, V.; Schuster, G. RNA polyadenylation and degradation in cyanobacteria are similar to the chloroplast but different from E. coli. J. Biol. Chem. 2003, 278, 15771–15777. [Google Scholar] [CrossRef] [PubMed]
- Sohlberg, B.; Huang, J.; Cohen, S.N. The Streptomyces coelicolor polynucleotide phosphorylase homologue, and not the putative poly(A) polymerase can polyadenylate RNA. J. Bacteriol. 2003, 185, 7273–7278. [Google Scholar] [CrossRef] [PubMed]
- Spickler, C.; Mackie, G.A. Action of RNase II and polynucleotide phosphorylase against RNAs containing stem-loops of defined structure. J. Bacteriol. 2000, 182, 2422–2427. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.A.; Cozad, M.; Mackie, G.A.; Jones, G.H. Kinetics of polynucleotide phosphorylase: Comparison of enzymes from Streptomyces and Escherichia coli and effects of nucleoside diphosphates. J. Bacteriol. 2008, 190, 98–106. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Symmons, M.; Jones, G.H.; Luisi, B. A duplicated fold is the structural basis for polynucleotide phosphorylase catalytic activity, processivity and regulation. Structure 2000, 8, 1215–1226. [Google Scholar] [CrossRef]
- Jones, G.H.; Mackie, G.A. Streptomyces coelicolor polynucleotide phosphorylase can polymerize nucleoside diphosphates under phosphorolysis conditions, with implications for the degradation of structured rnas. J. Bacteriol. 2013, 195, 5151–5159. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Potrykus, K.; Cashel, M. (p)ppgpp: Still magical? Annu. Rev. Microbiol. 2008, 62, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Srivatsan, A.; Wang, J.D. Control of bacterial transcription, translation and replication by (p)ppgpp. Curr. Opin. Microbiol. 2008, 11, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Petersen, F.S.; Kjelgaard, N.O. Analysis of the relA gene product of Escherichia coli. Eur. J. Biochem. 1977, 76, 91–97. [Google Scholar] [CrossRef]
- Chakraburtty, R.; White, J.; Takano, E.; Bibb, M.J. Cloning and characterization and disruption of a (p)ppgpp synthetase gene (relA) of Streptomyces coelicolor a3(2). Mol. Microbiol. 1996, 19, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Chakraburtty, R.; Bibb, M.J. The ppgpp synthetase gene (relA) of Streptomyces coelicolor a3(2) plays a conditional role in antibiotic production and morphological differentiation. J. Bacteriol. 1997, 179, 5854–5864. [Google Scholar] [CrossRef] [PubMed]
- Hoyt, S.; Jones, G.H. RelA is required for actinomycin production in Streptomyces antibioticus. J. Bacteriol. 1999, 181, 3824–3829. [Google Scholar] [PubMed]
- Jin, W.; Ryu, Y.G.; Kang, S.G.; Kim, S.K.; Saito, N.; Ochi, K.; Lee, S.H.; Lee, K.J. Two relA/spoT homologous genes are involved in the morphological and physiological differentiation of Streptomyces clavuligerus. Microbiology 2004, 150, 1485–1493. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jones, G.H. Purification and properties of atp:Gtp 3′-pyrophosphotransferase (guanosine pentaphosphate synthetase) from Streptomyces antibioticus. J. Bacteriol. 1994, 176, 1475–1481. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jones, G.H. Activation of ATP-GTP 3′-pyrophosphotransferase (guanosine pentaphosphate synthetase) in Streptomyces antibioticus. J. Bacteriol. 1994, 176, 1482–1487. [Google Scholar] [CrossRef] [PubMed]
- Gatewood, M.L.; Jones, G.H. (p)ppgpp inhibits polynucleotide phosphorylase from Streptomyces but not from Escherichia coli and increases the stability of bulk mRNA in Streptomyces coelicolor. J. Bacteriol. 2010, 192, 4275–4280. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.H. RNA synthesis in Streptomyces antibioticus: In vitro effects of actinomycin and transcriptional inhibitors from 48-h cells. Biochemistry 1976, 15, 3331–3341. [Google Scholar] [CrossRef] [PubMed]
- Liras, P.; Villanueva, J.R.; Martin, J.F. Sequential expression of macromolecule biosynthesis and candicidin formation in Streptomyces griseus. J. Gen. Microbiol. 1977, 102, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Kelly, K.S.; Ochi, K.; Jones, G.H. Pleiotropic effects of a relC mutation in Streptomyces antibioticus. J. Bacteriol. 1991, 173, 2297–2300. [Google Scholar] [CrossRef] [PubMed]
- Hesketh, A.; Sun, J.; Bibb, M.J. Induction of ppGpp synthesis in Streptomyces coelicolor A3(2) grown under conditions of nutritional sufficiency elicits actII-ORF4 transcription and actinorhodin biosynthesis. Mol. Microbiol. 2001, 39, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Siculella, L.; Damiano, F.; di Summa, R.; Tredici, S.M.; Alduina, R.; Gnoni, G.V.; Alifano, P. Guanosine 5′-diphosphate 3′-diphosphate (ppgpp) as a negative modulator of polynucleotide phosphorylase activity in a ‘rare’ actinomycete. Mol. Microbiol. 2010, 77, 716–729. [Google Scholar] [CrossRef] [PubMed]
- Gatewood, M.L.; Bralley, P.; Jones, G.H. RNase III-dependent expression of the rpsO-pnp Operon of Streptomyces coelicolor. J. Bacteriol. 2011, 193, 4371–4379. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bralley, P.; Aseem, M.; Jones, G.H. SCO5745, a bifunctional RNase J Ortholog, affects antibiotic production in Streptomyces coelicolor. J. Bacteriol. 2014, 196, 1197–1205. [Google Scholar] [CrossRef] [PubMed]
- Pei, X.Y.; Bralley, P.; Jones, G.H.; Luisi, B.F. Linkage of catalysis and 5′ end recognition in ribonuclease RNase J. Nucleic Acids Res. 2015, 43, 8066–8076. [Google Scholar] [CrossRef] [PubMed]
- Py, B.; Causton, C.F.; Mudd, E.A.; Higgins, C.F. A protein complex that mediates mRNA degradation in Escherichia coli. Mol. Microbiol. 1994, 14, 717–729. [Google Scholar] [CrossRef] [PubMed]
- Ait-Bara, S.; Carpousis, A.J. RNA degradosomes in bacteria and chloroplasts: Classification, distribution and evolution of RNase E homologs. Mol. Microbiol. 2015, 97, 1021–1135. [Google Scholar] [CrossRef] [PubMed]
- Vanzo, N.E.; Li, Y.S.; Py, B.; Blum, E.; Higgins, C.F.; Raynal, L.C.; Krisch, H.M.; Carpousis, A.J. Ribonuclease E organizes the protein interactions in the Escherichia coli degradosome. Genes Dev. 1998, 12, 2770–2781. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Cohen, S.N. A Streptomyces coelicolor functional orthologue of Escherichia coli RNAse e shows shuffling of catalytic and PNPase-binding domains. Mol. Microbiol. 2003, 48, 349–360. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jones, G.H. Novel Aspects of Polynucleotide Phosphorylase Function in Streptomyces. Antibiotics 2018, 7, 25. https://doi.org/10.3390/antibiotics7010025
Jones GH. Novel Aspects of Polynucleotide Phosphorylase Function in Streptomyces. Antibiotics. 2018; 7(1):25. https://doi.org/10.3390/antibiotics7010025
Chicago/Turabian StyleJones, George H. 2018. "Novel Aspects of Polynucleotide Phosphorylase Function in Streptomyces" Antibiotics 7, no. 1: 25. https://doi.org/10.3390/antibiotics7010025
APA StyleJones, G. H. (2018). Novel Aspects of Polynucleotide Phosphorylase Function in Streptomyces. Antibiotics, 7(1), 25. https://doi.org/10.3390/antibiotics7010025



