Abstract
Honey is considered to be a functional food with health-promoting properties. However, its potential health benefits can be affected by individual composition that varies between honey types. Although studies describing the health benefits of Tualang honey (TH), Kelulut honey (KH), and Sidr honey (SH) are scarce, these honey types showed a comparable therapeutic efficacy to Manuka honey (MH). The purpose of this review is to characterise the physicochemical, biological, and therapeutic properties of TH, KH, and SH. Findings showed that these honeys have antibacterial, antifungal, antiviral, antioxidant, antidiabetic, antiobesity, anticancer, anti-inflammatory and wound-healing properties and effects on the cardiovascular system, nervous system, and respiratory system. The physicochemical characteristics of TH, KH, and SH were compared with MH and discussed, and results showed that they have high-quality contents and excellent biological activity sources. Flavonoids and polyphenols, which act as antioxidants, are two main bioactive molecules present in honey. The activity of honey depends on the type of bee, sources of nectar, and the geographic region where the bees are established. In conclusion, TH, KH, and SH could be considered as natural therapeutic agents for various medicinal purposes compared with MH. Therefore, TH, KH, and SH have a great potential to be developed for modern medicinal use.
1. Introduction
Honey is a natural sweetener with a complex chemical composition and health/promoting properties [1,2]. Bees collect nectar from plants and/or insect excretions and produce honey, which has been revered for centuries for its nutritional and therapeutic properties [3]. Honey has been resurrected as a therapy for burns, gastrointestinal diseases, asthma, infected wounds, and skin ulcers in humans, and in animal medicine [4,5]. Honey contains several constituents of small amounts, such as minerals, free amino acids, proteins, vitamins, enzymes, organic acids, flavonoids, phenolic acids, and other organic acids in addition to other phytochemicals compounds [6,7]. The amount of these components is determined by several factors, including the honey’s geographical origin, floral source, meteorological circumstances, any treatments applied [8], and seasonality [9]. Honey’s composition can be affected by processing, handling, and storage [10]. The quality of honey also depends on floral resources and the treatment of the beekeepers [11]. Honey’s botanical and geographical origins have traditionally been determined by evaluating pollen quality and quantity and organoleptic and physicochemical testing. In addition, data derived from the sensory profile, bioactive components, and novel methods of investigation should be added to this information [7,12,13]. Water content, sugar reduction, sucrose, insoluble matter, ash, free acid, pH, electrical conductivity, specific rotation, and sensory and microbiological properties are the basis for the quality assessment of honey [14,15]. Honey’s components have a variety of beneficial biological actions, such as antibacterial, antiviral, antifungal, antioxidant, antidiabetic, antitumor, anti-inflammatory, and anticancer activity honey [16,17,18] (Figure 1). Various studies have demonstrated that antioxidant activity highly correlates to total phenolic levels [19]. Moreover, darker honey has been reported to have a higher total phenolic content and thus more significant antioxidant activity [20]. Honey’s composition includes various components, including hydrogen peroxide and polyphenols, and is also strongly linked to antibacterial activity [21,22]. The ability of honey to fight different types of microorganisms is determined by various variables, including the kind and natural structure of the nectar and the environmental circumstances in which the bees were raised [23,24]. The phenolic and flavonoid chemicals that make up honey are thought to be responsible for most of its biological activity. According to previous research, the activity of honey is influenced by the bioavailability of different phytochemical components as well as how they are absorbed and metabolized [18,25]. The flavonoids are largely water-soluble natural chemical compounds with low molecular weight. When flavonoids are not linked to sugars, they are referred to as aglycones [26,27,28,29,30,31,32,33]. In general, these chemicals include at least two phenolic groups (OH) and are frequently coupled with sugars (glycosides) [34,35,36,37,38,39,40,41,42,43,44,45]. The term “phenolic acids” refers to compounds that have a phenolic ring and at least one organic carboxylic acid function. They can be classified into three groups based on their structural types: C6–C3 compounds (such as p-coumaric, ferulic, and caffeic acids), C6–C2 compounds (such as acetophenones and phenylacetic acids), and C6–C1 compounds (e.g., syringic, vanillic and gallic acid). Most of these substances are typically linked to the cellulose and lignin that serve as the plant’s structural foundation, as well as to other classes of organic molecules such as glucose, other sugars, and flavonoids [46]. Additionally, some phenolic chemicals found in honey, including acaetin, caffeic acid, quercetin, galangin, and kaempferol, may hold potential as medicines for the treatment of cardiovascular disorders [47]. According to numerous research, flavonoids, which are crucial in reducing oxidative stress, are also necessary for honey’s antioxidant potential [13,48,49,50,51,52,53,54,55,56,57,58,59,60]. An updated study to comprehensively analyse the benefits of TH, KH, and SH is still required compared with other types of honey such as MH which has been extensively reviewed. With the recent growth in the body of literature for these kinds of honey, assessment of MH, TH, KH, and SH is needed to avoid future cross-studies or unnecessary research due to missed reviews of the existing research. In this review, relevant studies related to the medicinal properties, health benefits and physicochemical properties of MH, TH, KH, and SH were identified and critically analysed. The findings showed that most studies reported the health advantages of honey consumption, whereas some reported disadvantages or no significant changes upon consumption. Furthermore, analyses of physiochemical-related research revealed that MH, TH, KH, and SH have high-quality contents and are excellent sources of antioxidants.
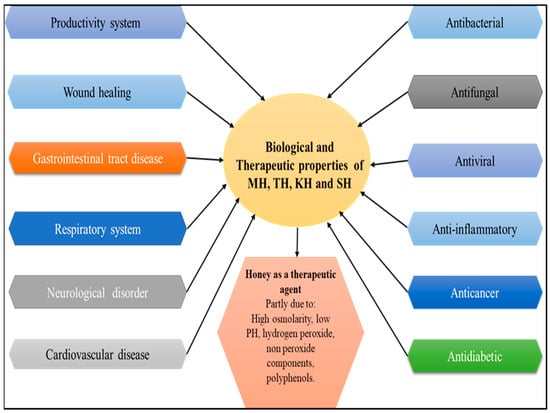
Figure 1.
Schematic representation of the therapeutic effects of honey. Adapted from Nweze et al. (2019) [26], Vazhacharickal et al. (2021) [27], Al-kafaween et al. (2022) [28], and Rao et al. (2016) [61].
2. Methods of Review
A literature search was conducted to identify previous articles illustrating the physiochemical and phytochemical of Manuka honey, Tualang honey, Kelulut honey, and Sidr honey and efficacy in the cure of diseases. Several databases were queried, including Web of Science, Scopus, Science Direct and PubMed. A literature search was performed by combining the following set of keywords: Manuka honey, Tualang honey, Kelulut honey, and Sidr honey. In addition, a literature search was undertaken to identify and map out relevant and pertinent articles related to the physicochemical, biological and therapeutic properties and health benefits of Manuka honey, Tualang honey, Kelulut honey, and Sidr honey. The present review covers a 52-year period which includes publications from 1970 to 2022. Initial searches yielded nearly 320 results. The abstracts of these publications were reviewed to confirm applicability. After further exclusion criteria (non-English language and manuscripts not available in full text), 243 articles remained.
3. Physicochemical Properties and Composition of MH, TH, KH and SH
Honey has a wide range of physicochemical properties depending on its botanical and geographical origins and compositions that subsequently affect its biological capabilities [29,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126,127,128,129,130,131,132,133,134,135,136,137,138,139,140,141,142,143,144,145,146,147]. Honey is defined as the natural sweet material generated by the Apies mellifera bees from the nectar of plants in European Union Council Directive 2001/110/EC [30]. Table 1 shows the physicochemical characterisation of MH TH, KH and SH extracted from various studies. Several studies have been conducted to look at the physical and chemical properties of MH TH, KH, and SH. On the other hand, MH TH, KH, and SH mostly complied with the accepted range by the two most common legislation of honey criteria and standards referred to as the European Honey Legislation and Codex Alimentarius Standards for Honey [29,31,32]. The primary quality indicators for honey include moisture content, sucrose content, reducing sugars content, pH value, electrical conductivity, ash content, free acidity, diastase activity, and hydroxymethylfurfural (HMF) content [36,37]. In terms of honey colour characteristics, MH is categorized as light-colored honey, TH is dark brown honey, KH is amber brown, and SH is dark-colored honey [29,38,43,148]. In this review most of the studies reported that TH and KH contained more than 20% moisture content, thus violating European Honey Legislation and Codex Alimentarius Standards. Nonetheless, honey samples from tropical countries, such as Malaysia, typically have higher moisture content, which could be due to the rainy season all over the year. Therefore, Malaysia’s honey is always first treated by evaporation to reduce the water content, thereby simultaneously increasing the honey quality [11,29,31,35,41,51,55,56]. MH contained 18.7% moisture content [41,43,50] and SH contained 13.5–20.67% moisture content [11,31,55,56]. The low pH of MH is approximately similar to TH and less than to KH and SH. TH is more acidic than KH and SH [41]. Darker honey typically has a higher conductivity, whereas brighter honey typically has a lower conductivity [33,34]. The pH values of TH, KH, and SH were reported to be in the range 3.14–4, 2.76–4.66 and 3.90–5.2, respectively, compared with MH (3.20–4.21) [11,29,31,35,41,42,43,51,55,56,57,59]. The electrical conductivity of TH, KH, and SH reported in this review is in a broad range of 0.75–1.37 mS/cm, 0.26–8.77 mS/cm, and 0.53 mS/cm, respectively, compared with 0.53 mS/cm to MH. Additionally, the four honey types met the sugar content requirements set forth by the Codex Alimentarius Standards and the European Honey Legislation. According to the European Honey Legislation and the Codex Alimentarius Standards, honey moisture should be less than 20%, with glucose and fructose composition of more than 60 g/100 g, sucrose content of not more than 5 g/100 g and electrical conductivity of not more than 0.8 mS/cm. According to the Malaysian Standard Kelulut, the raw honey moisture content must be less than 35 g/100 g, with a pH of less than 3.8 and 5-hydroxymethylfurfural of less than 30 mg/kg [29]. In addition, MH reported to has high protein content (g/kg) in the range 5.02–5.06 (g/kg) compared to 3.6–6.6 (g/kg) TH, 3.9–8.5 (g/kg) KH, and 1.5–4.09 (g/kg) SH [11,29,31,35,41,43,50,51,52,53,54,55,56]. Most bacteria grow in a neutral and mildly alkaline environment, whereas yeasts and moulds could grow in an acidic environment (pH = 4.0–4.5). Conversely, the pH values of honey are neither those needed for bacteria nor yeast growth [38]. This is of great importance during storage, as they influence the texture, stability, and shelf-life of honey [11,29,35,39,43,50,51,57,59,60,62]. The low protein content and high carbon-to-nitrogen ratio of honey are not conducive to microbial growth, nor is the acidity of honey. The low redox potential of honey (which is due to its high content of reducing sugars) discourages growth of molds and aerobic bacteria, whereas the viscosity of honey opposes convection currents and limits the entry of dissolved oxygen. As the osmotic pressure is high, the microbes shrivel as water flows out of their cells into the surrounding honey [29,39]. Various factors, such as storage, time, temperature, water content and concentration of ions and minerals, were reported to contribute to the electrical conductivity of honey [11,35]. A comparison of the physicochemical characteristics of TH, KH, and SH with that of MH is presented in Table 1.

Table 1.
The physicochemical characteristics of TH, KH, SH versus MH.
Honey is mostly composed of fructose (35.6–41.8 g), glucose (25.4–28.1 g), sucrose (0.23–1.21 g) and Maltose (1.8–2.7 g) and other sugars [29,149,150,151,152,153,154,155,156,157,158,159,160,161,162,163,164,165,166,167,168,169,170,171,172,173,174,175,176,177,178,179]. It includes around 180 different compounds, including amino acids, vitamins, minerals, and enzymes. The composition varies according to the floral source and origin [29,179]. The concentration of sucrose (g/100 g) in MH and SH was higher than that in TH and KH [29,55,64,65], and the concentration of glucose (g/100 g) in MH and TH was higher than that in KH and SH [29,55,61,65]. In addition, the concentration of fructose (g/100 g) in MH and TH was higher than that in KH and SH [29,55,61,65], and the concentration of maltose (g/100 g) in TH and KH was higher than that in MH and SH [29,61,65]. Moreover, the protein content of MH (5.02–5.06 (g/kg) was higher than the values reported for TH (3.6–6.6 (g/kg), KH (3.9–8.5 (g/kg), and SH (1.5–4.09 (g/kg) [29,55,64,65]. Among the major and minor elements found in honey, potassium (K) is found in the highest concentrations, followed by sodium (Na), calcium (Ca), and magnesium (Mg). Furthermore, because sugars are its primary constituents, honey’s physical characteristics and behavior are attributed to sugars. Sugar tests will indicate its sweetness due to its high sugar content, with fructose being the most abundant sugar. The concentration of sodium in MH, KH and SH was lower than the values reported for TH and the concentration of potassium and calcium in SH was higher than the values reported for MH, TH, and KH [29,55,61,66]. Additionally, the concentration of magnesium in SH was higher than the values reported for MH, TH, and KH [29,55,61,66]. Minor constituents such as flavor compounds, minerals, acids, pigments, and phenols play a significant role in distinguishing each variety of honey [30,36,40,41]. Honey is a natural source of flavonoids, phenolic acids, and phenolic acid derivatives [44]. MH has higher total phenolic content (429.61 (mg/kg)) [64] than TH (251.7–1103.94 (mg/kg)) [29], KH (477.30–614.7 (mg/kg)) [29], and SH (212.4–520.34 (mg/kg)) [65]. In addition, the total flavonoid content in MH (97.62) [64] was higher than in TH (49.04–185) [29], KH (36.3) [29], and SH (42.5) [67]. A total of seven phenolic acids (caffeic, gallic, syringic, vanillic, p-coumaric, benzoic, and trans-cinnamic acids) and six flavonoids (apigenin, kaempferol, luteolin, naringenin, naringin, and catechin) are found in TH. Additionally, a total of four phenolic acids (syringic, gallic, ferulic and caffeic acids) and eight flavonoids (chrysin, galangin, isorhamnetin, kaempferol, luteolin, pinobanksin, pinocembrin, and quercetin) are found in MH. A total of nine phenolic acids (gallic, syringic, vanillic, 3 4-dihydroxybenzoic, 4-hydroxybenzoic, p-coumaric, cinnamic, salicylic, cis-trans-Abscisic acids) and three flavonoids (luteolin, naringenin, and taxifolin) are found in KH. A total of six phenolic acids (gallic, salicylic, chlorogenic and tannic acids) and five flavonoids (catechin and quercetin) are found in SH [28,29,41]. TH contains more phenolic acids and flavonoids than MH, KH and SH [41]. Some compounds found in TH previously not reported in other honeys include stearic acids, 2-cyclopentene-1,4,-dione, 2[3H]-furanone or dihydro-butyrolactone, gamma-crotonolactone or 2[5H]-furanone, 2-hydroxy-2-cyclopenten1-one, hyacinthin, 2,4-dihydroxy-2,5-dimethyl3[2H]-furan-3-one, and phenylethanol [41,179]. The details of the various compounds present in MH, TH, KH, and SH are summarised in Table 2 and Figure 2.

Table 2.
The most common phenolic compounds (flavonoids and phenolic acids) identified in MH, TH, KH, and SH [18,41,68,69,70].
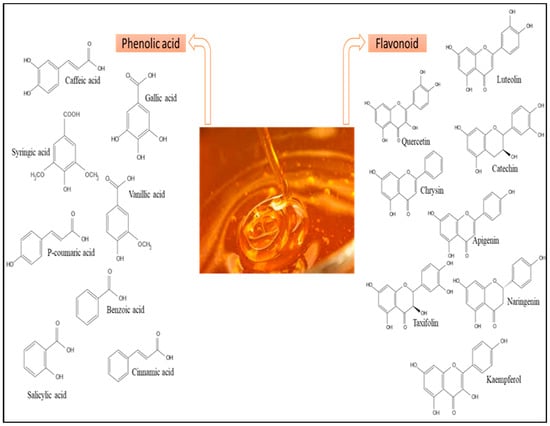
Figure 2.
Most common phenolic compounds identified in honey. Adapted from Cianciosi et al. (2018) [67].
Generally, honey is rich in phenolic compounds, which act as natural antioxidants and are becoming increasingly popular because of their potential role in contributing to human health [29,43,47,68,179]. In this review, Table 3 shows some of phenolic compounds with their different potential health benefits found in honey. A wide range of phenolic constituents are present in honey, including quercetin, caffeic acid, gallic acid, catechin, apigenin and kaempferol, which have promising effect in the treatment of cardiovascular diseases [29,43,47,68,179]. Many epidemiological studies have shown that regular intake of phenolic compounds is associated with reduced risk of heart diseases. In coronary heart disease, the protective effects of phenolic compounds include being antithrombotic, anti-ischemic, anti-oxidant, and vasorelaxant [29,43,47,68,179]. It is suggested that flavonoids decrease the risk of coronary heart disease by three major actions: improving coronary vasodilatation, decreasing the ability of platelets in the blood to clot, and preventing low-density lipoproteins (LDLs) from oxidizing [29,43,47,68,179]. Cell viability of fibroblast-like synoviocytes (FLS) from patients with rheumatoid arthritis (RA) was significantly decreased by treatment with 10 or more μM of gallic acid. Treatment with 0.1 and 1 μM of gallic acid also showed in a significant increase in caspase-3 activity and regulated the production of Bcl-2, Bax, p53, and pAkt. The mRNA expression levels of pro-inflammatory cytokines (IL-1β, IL-6), chemokines (CCL-2/MCP-1, CCL-7/MCP-3), cyclooxygenase-2, and matrix metalloproteinase-9 from RA FLS were suppressed by the gallic acid treatment in a dose-dependent manner [29,43,47,68,179]. The phenolic compounds in honey such as p-hydroxibenzoic acid, cinnamic acid, naringenin, pinocembrin, and chrysin showed antimicrobial activity [29,43,47,68,179]. Additionally, caffeic acid exhibits a significant potential as an antidiabetic agent by suppressing a progression of type 2 diabetic states that is suggested by an attenuation of hepatic glucose output and enhancement of adipocyte glucose uptake, insulin secretion, and antioxidant capacity [29,43,47,68,179]. In addition, protocatechuic and p-hydroxybenzoic acid exhibit significant antioxidant, anticancer and antiatherogenic activities [29,43,47,68,179]. The chrysin decreased lipid peroxide, reduced the increased activities of superoxide dismutase, and attenuated the decreased activities of glutathione peroxidase in 2VO rats and Quercetin-3-O-rhamnoside showed moderate antitumor activity [29,43,47,68,179].

Table 3.
Some of phenolic compounds with their potential health benefits found in MH, TH, KH, and SH.
4. Therapeutic Proprieties of MH, TH, KH and SH
The therapeutic benefits of MH, TH, KH, and SH are discussed in this section based on different properties. Additionally, literature on the therapeutic benefits of MH, TH, KH, and SH were obtained. These articles covered in vitro, in vivo, and ex vivo studies, and human clinical trials.
4.1. Oxidative Stress, Antioxidant and Anti-Inflammatory Properties
Oxidative stress (OS) is characterized as an imbalance in favor of oxidants over antioxidants. The OS causes oxidative damage, which can impair a variety of physiological activities. OS is the basis of structural and functional damage to the main biomolecules such as nucleic acids, lipids, and proteins. In fact, these injuries lead to the development of many diseases, such as cancer, metabolic disorders, and cardiovascular dysfunctions. The imbalance created between the production of free radicals and antioxidant defense can occur not only in pathological situations, but also in some physiological conditions such as intense physical activity [179]. The primary oxidants in biological systems are free radicals and reactive oxygen species (ROS). They are also implicated in aging and the beginning of many diseases [71]. ROS and free radicals are physiologically formed in several cellular biochemical events that occur in the body, such as in mitochondria for aerobic oxygen synthesis, fatty acid metabolism, medication metabolization, and immune system activity [72,73,74]. Free radicals, on the other hand, can be created by external sources such as pollution, poor lifestyle choices, UV rays, ionizing radiation, and psychophysical stress from strenuous physical exercise [75]. Antioxidants are compounds that may transfer an electron to free radicals, thus neutralizing, reducing, or eliminating their capacity to harm cells and key macromolecules including nucleic acids, proteins, and lipids [76]. Oxidative stress is more likely to cause chronic or degenerative disorders. The antioxidant properties of honey may help in reducing oxidative damage and improving brain cell structure and integrity [77,78]. Honey generally contains various kinds of phytochemical with high phenolic and flavonoid content, thus contributing to its high antioxidant activity [28,29]. Many studies have indicated that honey’s antioxidant ability is significantly related to the concentration of its phenolic component [64,68,79]. The present review also found that TH, KH, and SH have a high content of phenolics and flavonoids compared with MH. Amongst the reviewed studies comparing the antioxidant properties of TH and KH, one study showed that KH has the highest antioxidant properties in ranking after MH [57]. A previous study found that TH has higher antioxidant properties than MH and KH [29]. In addition, MH appears to play a protective function against oxidative damage in an in vivo model, lowering DNA damage, the level of malondialdehyde, and glutathione peroxidase activity in the livers of both young and middle-aged groups of rats. However, the glutathione peroxidase activity was increased in the erythrocytes of middle-aged rats given MH supplementation. The catalase activity was reduced in the liver and erythrocytes of both young and middle-aged rats given supplementation [80]. A previous study reported that the pancreases of diabetic control rats showed significantly increased levels of malondialdehyde (MDA) and up-regulation of superoxide dismutase (SOD) and glutathione peroxidase (GPx) activities and catalase (CAT) activity was significantly reduced, whereas glutathione-S-transferase (GST) and glutathione reductase (GR) activities remained unchanged in the pancreases of diabetic rats after being treated with TH (honey-treated diabetic rats had significantly (p < 0.05) reduced blood glucose levels (8.8 (5.8) mmol/L; median (interquartile range)) compared with the diabetic control rats (17.9 (2.6) mmol/L]) [62]. The DPPH radical scavenging activity was expressed in EC50 (mg/mL), where the minimum amount needed to scavenge 50% of the DPPH free radical [51,87]. A previous study showed that MH has higher antioxidant potential compared with TH, KH, and other types of honey (MH showed the lowest EC50 value (116.05 mg/mL), followed by TH (341.25 mg/mL) and KH (329.89 mg/mL) [51]. A study showed that TH increases the effectiveness of glibenclamide and metformin in preventing oxidative stress and damage in the pancreases of diabetic rats and reduces oxidative stress in diabetic rats’ kidneys [81,82]. In addition, TH enhanced the migration of human corneal epithelial progenitor cells and cellular resistance to H2O2-induced oxidative stress, according to an in vitro study [83]. In contrast, there was no discernible difference between the TH treated group and the group that received conventional treatment in terms of inflammatory feature or antioxidant status, according to a study looking at the anti-inflammatory and antioxidant effects of TH in alkali injury on rabbit eyes [84]. A previous study reported that KH reduced oxidative stress by reducing lipid peroxidation and increasing SOD, and also maintained bone structure, increased the number of osteoblasts, and reduced the number of osteoclasts. Therefore, the study suggested that KH could be used as a prophylactic agent to prevent glucocorticoid-induced osteoporosis [85]. Additionally, KH supplementation protected sperm and testicular oxidative damage in streptozotocin-induced diabetic rats [86]. A study by Saeed et al. (2021) demonstrated that SH exhibited considerable variations with reference to their level of total phenolic content (TPC) (98.2–432.91 mg/kg), total flavonoid content (TFC) (49.9–202.0 mg/kg), radical-scavenging activity (7.6–72.6 mg/mL), and ascorbic acid (28.1–161.2 mg/kg), as well as total carotenoid content (TCC) (13.2–36.2 mg/kg). A strong significant correlation between biochemical parameters and radical scavenging activity was found, and the study indicated that SH can be considered a good source of antioxidant and biochemical compounds [87]. A previous study showed that TH decreased neuroinflammation by lowering the elevation of TNF-, IL-1, glial fibrillary acidic protein, allograft inflammatory factor 1, and COX-2 in the rat cerebral cortex, cerebellum, and brainstem in kainic acid (KA)-induced status epilepticus rats [103]. Another study also reported a reduction in TNF-α, IL6, and IFN-γ in the brain homogenates of a TH-treated chronic stress rat model [88]. However, the anti-inflammatory properties of TH could not be translated to humans as a randomised controlled study demonstrated that TH supplementation has opposite effects on TNFα and highly sensitive C-reactive protein, indicating the inconclusive effect of honey on inflammation amongst chronic smokers; thus, further studies are needed on other inflammatory markers or other study populations [89]. One study showed that MH provided protection against trinitro-benzo-sulphonic acid induced colonic damage, reduced colonic inflammation also restored lipid peroxidation and improvement of antioxidant parameters [90]. Minden et al. (2020) reported that 0.5% of MH solution significantly increased the release of CXCL8/IL-8, CCL2/MCP-1, CCL4/MIP-1β, CCL20/MIP-3α, IL-4, IL-1ra, and FGF-13 while reducing Proteinase 3 release in the anti-inflammatory-stimulated models and 3% of MH solution significantly increased the release of TNF-α and CXCL8/IL-8 while reducing the release of all other analytes. These findings demonstrated the variable effects of MH on the release of cytokines, chemokines, and matrix-degrading enzymes of this model of neutrophil anti-inflammatory activity [91]. Another study showed that pre-treatment with MH markedly inhibited LPS induced ROS and nitrite accumulation and increased the protection against cellular biomolecules such as lipids, proteins, and DNA and stimulation by LPS suppressed both antioxidant enzyme activities and expressions, and Keap1-Nrf2 signaling pathway which was significantly increased in the presence of MH also after MH treatment the expression of pro-inflammatory cytokines, such as TNF-α, IL-1β, and IL-6, and other inflammatory mediators (iNOS) were suppressed [92]. Moreover, MH also inhibited the expression of TLR4/NF-кB via IкB phosphorylation in LPS-stressed RAW 264.7 macrophages [92]. KH has been proven to possess various pharmacological properties such as antioxidant and anti-inflammatory [93,94]. Since KH has strong antioxidant activities, it could be one of the potential chemopreventive agents from natural resources [93]. According to multiple studies, the quantity of phenolic and flavonoid groups affects the antioxidant activity of various types of honey produced from various nations [32,95,96]. MH acted as a natural agent for preventing oxidative and inflammatory-related diseases more than TH and SH.
4.2. Antibacterial, Antiviral and Antifungal Properties
Honey has been used as an antibacterial agent since ancient times. Table 4 describes the microorganisms that are sensitive to MH, TH KH and SH. It seems to act on both Gram-positive (G+) and Gram-negative (G−) bacteria, although the first are more sensitive. All the studies, summarized in Table 4, used the agar disk-diffusion and Broth dilution tests to determine the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of various types of honey with diverse bacterial agents. Numerous studies and research have been conducted in laboratories on the biological properties of honey such as antibacterial, antifungal, antiviral, and antiprotozoal [97,98,99]. The biological activity of honey is one of the most important features that distinguish it from other natural products, which makes it important in the medical and therapeutic fields [100,101,102]. Nowadays, honey is effective against more than 60 bacterial species, and its antibacterial activity is dependent on the type of honey. Numerous studies have reported that honey has antibacterial and antibiofilm activity against a wide range of both Gram-positive and Gram-negative bacteria, including; Streptococcus pyogenes, Mycobacterium, Escherichia coli, Pseudomonas aeruginosa, Salmonella typhi, Salmonella paratyphi, Salmonella enterocolitis, Shigella dysenteriae, Pseudomonas aeruginosa, Mycobacterium tuberculosis, methicillin-resistant Staphylococcus aureus, Streptococcus pneumonia, Streptococcus agalactiae, and Shigella flexneri. Among the activity of all types of honey, the inhibiting effect of MH, at low concentrations (2–6%), on the planktonic and biofilm of both Gram-positive and Gram-negative bacteria, was greater than other types of honey, including TH, KH, and SH; moreover, the large number of studies of MH has shown that it has broad spectrum activity against pathogenic bacteria [58,103,104,105,106,107,108,109,110]. TH was the most comparable to MH in terms of antibacterial potency, followed by KH and SH [24,31,104,107,111,112,113,114]. A previous study revealed that when MH and TH dressings were tested against Gram-negative bacteria on the burn wound, the results of two comparative tests on antibacterial potency were equivalent, but TH was less effective than standard care products as silver-based dressings or medical grade and MH dressings against Gram-positive bacteria [115]. TH, KH, and SH exhibited variable activities against different microorganisms, but they were within the same range as those for MH, suggesting that TH could potentially be used as an alternative therapeutic agent against certain microorganisms, particularly Stenotrophomonas maltophilia and Acinetobacter baumannii [31,107,116]. Honey has also been found in some studies to have antiviral action [29]. Küçük et al. (2007) and Mohd et al. (2021) demonstrated that honey has been known to reduce viral load since the 19th century [29,117]. It was evaluated that the in vitro effect of MH and Clover honey in human malignant melanoma cells (MeWo)-infected with varicella Zoster virus (VZV) isolated from a Zoster vesicle. The results showed a reduction of the viral plaques after the treatment of the cells with both types of honey [118]. A similar effect has also been proven in Madin-Darby canine kidney (MDCK) cells infected with influenza virus (H1N1), treated with different types of honey (MH, Renge honey, and Acacia honey). The plaque inhibition assay has been carried out showing a higher antiviral activity of Manuka honey, compared with the other types of honey, and the synergistic effects with some antiviral drugs [119,120]. Another study reported that among the five honey types (Manuka honey (MH), clover honey, acacia honey, rosemary honey, and milk vetch honey), the anti-HIV-1 RT activity of MH was the strongest and was associated with its constituents, 2-methoxybenzoic acid (2-MBA) and methylglyoxal (MGO) and MH inhibited the activity of HIV-1 RT in a dose-dependent manner and the IC50 value was approximately 14.8 mg/mL and the study revealed that the inhibitory effect of MH on the HIV-1 RT activity is mediated by multiple constituents with different physical and chemical properties as mentioned above [121]. It has also been demonstrated an antifungal activity of honey towards different kinds of Candida infections (Candida albicans, Candida glabrata, Candida dubliniensis, Candida tropicalis, Candida krusei, and Candida parapsilosis), and on Rhodotorula sp., evaluating the MIC and using the agar disk-diffusion test [122,123]. Study by Guttentag et al. (2021) indicated that jarrah honey has unique antifungal attributes that work to inhibit and kill dermatophytes, making it a potentially promising candidate for the treatment of tineas [124]. This variation was linked to the botanic origin of the honey, with multi-floral honey having greater phenolic concentrations than monofloral honey [125]. The antibacterial, antiviral, antifungal, antioxidant, and anti-inflammatory activities of honey are noteworthy due to phenolic compounds, especially flavonoids, with the quality of polyphenols being more important than their quantity [18,28,32,63,126]. In addition, honey’s potent antibacterial properties are related to its high osmolarity, acidity, H2O2 content, and non-peroxide component composition [127]. H2O2 is produced when glucose oxidase hydrolyzes glucose, which can only happen when honey is diluted. Relative glucose oxidase levels produced by bees and catalase derived from flowers are used to calculate H2O2 levels [128]. The antibacterial activities of honey at this stage are due to strong osmotic pressures and high acidity in pure honey [127]. MH possess high non-peroxide antibacterial activity that could retain the antibacterial activity even after treatment with catalase [29,129]. They are known as active non-peroxide honey, containing various non-peroxide components that possess antibacterial actions [130,131]. These components include phenolic acids, flavonoids, methylglyoxal, and methyl syringate [29,127]. Finally, honey’s strong antibacterial ability is connected to an improvement in gut microbial balance due to its high number of oligosaccharides, which function as a substrate for the growth of prebiotic microorganisms. One research found that the addition of different types of honey increased the vitality and development rate of Lactobacilli and Bifidobacteria in the gut microbiota balance [132].

Table 4.
List of micro-organisms that have been found to be sensitive to MH, TH, KH, and SH.
4.3. Anticancer Properties
The potential effects on cancer have been studied in terms of prevention, progression, and therapy. The majority of the investigations are in vitro and have been conducted on various cell lines and types of honey. Some studies have also been carried out in vivo on mice/rats, inducing or transplanting the tumor [157]. Honey acts at different stages of cancer, on the initiation, proliferation, and progression. Its antitumoral effects are generally attributed to different mechanisms, such as the induction of apoptosis, cell cycle arrest, the modulation of oxidative stress, the amelioration of inflammation, the induction of mitochondrial outer membrane permeabilization (MOMP), and the inhibition of angiogenesis (Figure 3) [18,29]. The majority of the research on MH, TH, KH, and SH concentrated on their anticancer effects in a variety of models and pathways. Investigations involved a variety of subjects, including cell culture, animal, and human studies. A summary of the anticancer properties for MH, TH, KH, and SH is presented in Table 5, whereas their anticancer pathway is illustrated in Figure 3. The impact of MH and TH on breast cancer has also been examined in an in vivo investigation. It was shown that there was a decrease in tumor grade, estrogenic activity, and hematological parameters. Additionally, it has been shown that the expression of certain proteins involved in the inflammatory pathway, such as TNF- and COX-2, as well as pro-apoptotic proteins such as Caspase 9 and p53, has increased [158]. In another study, the activity of MH against HCT-116, human colon cancer cells, and the LoVo metastatic cell line was examined. On both cell lines, they observed an increase in intracellular ROS generation as well as an inhibitory effect on cell proliferation. These results show that the cytotoxic impact of MH may be connected to the quantity of polyphenols present in this matrix as the cytotoxic effect of STH was emphasized as being more pronounced, which exhibited a higher number of phytochemicals and antioxidant capabilities [18,159]. In one vivo study, MH induced a strong proapoptotic activity in a dose- and time-dependent way after being intravenously delivered to mice with murine melanoma tumor cells (B16F1), decreasing the final tumor volume. Additionally, mice which received MH in addition to the chemotherapy drug (Taxol) lived longer than mice that simply received the chemotherapeutic agent [160]. Another study demonstrated that MH has anticancer activity against breast cancer MCF-7 cells [161]. Three in-vivo studies using a breast cancer model showed that TH had anticancer potential [158,162,163]. These studies have shown that TH’s anticancer efficacy was demonstrated by modulating five aspects: tumor development, tumor grading, and haematologic, oestrogenic, and apoptotic activities. In a rat breast cancer model, TH-treated rats had a lower growth rate, tumor size, weight, and tumor multiplicity than untreated controls. [158,162,163]. Histologically, breast cancer rats treated with TH were mostly graded I and II compared with the untreated control [158,162]. Moreover, TH increased proapoptotic protein expression (Apaf-1, caspase-9, IFN-γ, IFNGR1, and p53), whereas it decreased antiapoptotic protein expression (ESR1, TNF-α, COX-2, and Bcl-xL) [158,162,163]. Ahmed et al. (2017) reported that TH treatment was effective on haematological parameters, such as haemoglobin (Hb), red blood cells (RBCs), packed cell volume (PCV), mean corpuscular volume (MCV), red cell distribution width (RDW), mean corpuscular hemoglobin concentration MCHC, polymorphs and lymphocytes values [158]. In an animal breast cancer model, MH was found to have the same anticancer activities as TH [163]. The anticancer effect of TH was successfully demonstrated in another three further in vitro studies utilizing the human breast cancer cell line [164,165,166]. These studies have found that TH treatment triggered caspase-3, caspase-7, and caspase-9 and decreased mitochondrial membrane potential in all tested cancer cells [164,165]. Moreover, TH was found to be cytotoxic to breast cancer cell line (MCF-7); it also protected non-tumorigenic epithelial breast cell line (MCF-10A) from the toxic effects of tamoxifen active metabolite 4-hydroxytamoxifen by increasing the efficiency of the DNA repair mechanism in these cells, as evidenced by the increment in DNA repair proteins Ku70 and Ku80 [166]. In a clinical trial, the combination of TH and anastrozole was more successful than anastrozole alone in decreasing breast background parenchymal enhancement in breast cancer patients [167]. In an animal model, TH revealed chemopreventive properties by decreasing cancer cell proliferation and activity, as seen by a reduction in the expression of cancer-related genes such as CCND1, EGFR, and COX-2. Furthermore, TH also reduced the expression of TWIST1 and RAC1, which are the genes representing epithelial-to-mesenchymal transition (EMT), and overexpressing β-catenin and E-cadherin [168]. Another study found that TH inhibits the growth of oral squamous cell carcinoma and osteosarcoma cell lines by causing early apoptosis [169]. The in vitro studies have revealed further anticancer potentials of TH, including anticancer activity against cervical cancer cell lines; this activity has the same mechanism as the in vitro research on breast cancer cell lines detailed above [164]. Previous research found that TH has antileukemic properties due to its potential to induce apoptosis in acute and chronic myeloid leukemia cell lines [170]. Another study showed that TH protected keratinocytes against UV radiation-induced inflammation and DNA damage by modulating the early biomarkers of photocarcinogenesis, thus providing significant protection from the adverse effects of ultraviolet B (UVB) radiation and a suggestion for its photochemopreventive potential [171]. Ramasamy et al. (2019) showed that after four and eight weeks of treatment with TH or Vitamin C, the fatigue level for experimental group was better than in the control group and no significant changes were detected between the vitamin C and TH groups for the white cell count and C-reactive protein [172]. Another study revealed that TH has anticancer capabilities; increasing the concentration of the extract reduces the viability of cancer cells [173]. Anticancer properties were also exhibited by KH. A previous study demonstrated that KH possesses chemopreventive properties in colorectal cancer-induced rats and also was found not toxic towards the animals [93]. Previous research indicated that KH was not cytotoxic to Human Gingival Fibroblast Cell Line (HGF-1 cell line) [174]. Salim et al. (2019) demonstrated how KH acts as a potential anticancer agent against human lung adenocarcinoma epithelial cell line (A549) and KH was capable of inducing cell cycle arrest at the G2/M phase [175]. A previous study showed that SH has anticancer activity and inhibited HepG2 cancer cell line proliferation [150]. Almeer et al. (2018) reported that the anticancer effects of SH correspond to their ability to modulate gene expression of MMPs and TIMPs in human breast adenocarcinoma (MDA-MB-231) cell lines [176]. SH was shown to have potential antitumor activity in studies on cancer cell growth inhibition [177]. Another study has shown that treatment with SH was able to inhibit proliferation, and induce apoptosis in breast cancer cell lines (MDA-MB-231 and MCF-7) and cervical cancer cell lines (Hela) [178]. Honey has a variety of phytochemicals with significant phenolic and flavonoid content, which contributes to its antioxidant properties [179]. Because free radicals cause oxidative stress, which leads to cancer formation, a drug with significant antioxidant capabilities might potentially prevent cancer [180]. Furthermore, a different type of polyphenol present in honey has been shown to have anticancer capabilities against a variety of cancers via various mechanisms. These polyphenols including, caffeic acid, caffeic acid phenyl esters, chrysin, galangin, quercetin, kaempferol, acacetin, pinocembrin, pinobanksin and apigenin [181]. Honey is a natural immune booster, antibacterial agent, anti-inflammatory agent, promoter of chronic ulcers and wound healing, and antioxidant; all of these characteristics contribute to its anticancer properties.
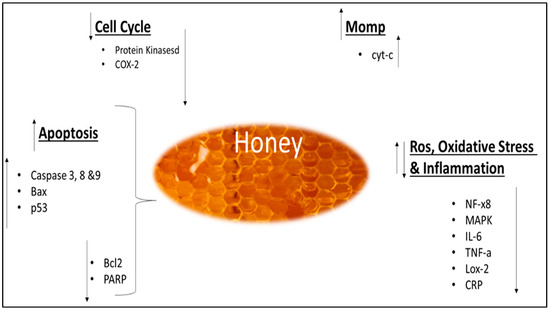
Figure 3.
Molecular mechanism involved in anticancer effect of honey. Adapted from Cianciosi et al. (2018) [18] and Mohd et al. (2021)[29].

Table 5.
List of anticancer properties of MH, TH, KH, and SH.
4.4. Antidiabetic Properties
There is evidence that honey has a beneficial effect on type 1 and type 2 diabetes mellitus. The measurement of fructosamine, glycosylated hemoglobin, and glucose is prevalent and important in the practice of glycemic control in patients with diabetes mellitus [183]. Previous studies showed that MH promoted the complete healing of diabetic foot ulcers, and decreasing the rate of minor amputations [184]. Studies using TH demonstrated a mild hypoglycemic impact, an enhanced liver enzyme profile, and a synergistic benefit on glycemic and metabolic profiles when taken in combination with metformin or glibenclamide [81,185,186]. Robert et al. (2009) reported that TH was found to have intermediary glycemic index values of 65 ± 7 [187]. Another study by Hussain et al. (2012) demonstrated that supplementation of twenty gram per day of TH for 4 months in healthy postmenopausal women caused a significant increase in fasting blood sugar (FBS)[188]. However, extending the therapy to 12 months resulted in a reduction in FBS levels in both healthy and diabetic postmenopausal women [189]. TH showed that the presence of flavonoid and phenolic compounds in honey from different botanical origins have strong α-amylase and α-glucosidase inhibition abilities as their inhibition percentages were more than 70.00% at 100 µg/mL and also recommended the uses of stingless bee honey in diabetes treatment [29,43,52,183]. Previous research revealed that honey had a varied effect on glucose metabolism for short- and long-term consumption [189]. Rashid et al. (2019) found that consuming 30 g KH for 30 days had no significant influence on fasting glucose and fasting lipid profile in patients with impaired fasting blood glucose [59]. The authors suggested that the small effect was due to short-term use of KH. In contrast, one in vivo study used forty male Wistar rats (divided into 5 groups; 8 weeks (C8) and 16 weeks control groups (C16), groups that received High-Carbohydrate High Fructose (HCHF) diet for 8 weeks (MS8) and 16 weeks (MS16), and a group that received HCHF for 16 weeks with KH supplemented for the last 35 days (KH) found that KH has the ability to normalise blood glucose and serum fasting blood glucose decreased in the KH group compared to the MS16 group [190]. Alharbi et al. (2022) reported that a combination of L. plantarum-fermented oats or fermented oats with SH exhibited potential antidiabetic effects and served as a potential approach for controlling glucose levels and lipid profiles, as well as protecting against oxidative stress [191]. Another study revealed that MH did not affect the weight in diabetic mice, whereas the SH-treated mice showed a reduction (7.7 ± 0.41 g) in body weight (Initial body weight: 18 ± 2.107, After treatment: 10.3 ± 2.517 and change: ↓7.7 ± 0.410) [192]. Clinical research has indicated that, unlike other sweeteners, honey consumption lowers the postprandial glycemic response in diabetic and non-diabetic volunteers, reducing the glucose blood concentration in patients with type 1 and type 2 diabetes [193]. Numerous studies attribute honey’s antidiabetic and hypoglycemic properties to its antioxidant capacity in response to dosage; indeed, the etiology of diabetes mellitus, particularly type 2 appears to be intimately linked to the presence of oxidative stress and ROS in numerous organs and tissues [194]. Increased glucose absorption by adipose tissue and muscles increases ROS generation, which contributes to oxidative stress, a process that regulates glycogen synthesis and glucose uptake. Furthermore, oxidative stress can promote insulin resistance by impairing the insulin signaling system, which can be reversed by honey therapy [195] Even in pancreatic -cells, oxidative stress has a role in affecting their functioning, resulting in inappropriate insulin secretion and an increase in -cell death. In general, it has been demonstrated that honey’s scavenger function reduces pancreatic oxidative stress [196]. Diabetes mellitus also compromises lipid metabolism, resulting in a significant presence of low-density lipoproteins (LDLs), which are oxidized and glycated in oxLDLs, causing endothelial damage. Even in this situation, honey’s antioxidant activity aids in the prevention of lipid oxidative metabolism in individuals with type 2 diabetes [96]. However, the antidiabetic efficacy of TH, KH, and SH remained unclear. Future research should extend understanding on the antidiabetic capabilities of TH, KH, and SH on diabetic patients to support the antidiabetic ability of TH, KH, and SH, as described in the animal studies above.
4.5. Antiobesity Properties
Honey provides various nutritional benefits and also honey has demonstrated the antiobesity effects in some studies [197]. Only KH has been shown in animal experiments to have antiobesity effects [198,199,200]. Previous study showed that supplementation of KH yielded a higher reduction in body mass index, the percentage of body weight gain, adiposity index, and relative organ weight in a high-fat diet-induced obese rat model than that of orlistat, an antiobesity drug [198]. Another study found that KH might prevent metabolic-syndrome-induced changes in omental fat mass, serum triglycerides, systolic blood pressure, diastolic blood pressure, adipocyte area, and adipocyte perimeter in rats [200]. However, honey showed better effects than orlistat, as the orlistat group showed hepatotoxicity effects. As of now, MH and TH have not been demonstrated to have an antiobesity effect. MH and TH have not yet been shown to have an antiobesity impact. Meanwhile, KH has not been studied for its antiobesity potential in humans.
4.6. Wound-Healing Properties
The importance of honey in the field of wound treatments has been well known since ancient times [95,139,201]. This practice was rooted primarily in tradition and folklore when investigators began to explore its medicinal potential [29,202]. There have been reports of significant therapeutic benefits of honey from various geographic regions on chronic wounds, ulcers, and burns [203,204]. Honey is reported to have excellent effects compared with traditional treatments for acute wounds, superficial partial-thickness burns, and infected wounds after surgery [205,206]. This healing ability is related to honey’s antioxidant and antibacterial properties, which help to keep wounds moist, as well as to honey’s high viscosity, which forms a barrier of protection over the wound and deters microbial infection. Its immunological activity has implications for healing wounds as well, acting both pro- and anti-inflammatory [103,139,207]. Normal wound healing is a difficult process made up of a number of overlapping activities (coagulation, inflammation, cell proliferation, tissue remodeling), in which the damaged tissue is eventually eliminated and replaced by reparative tissues [208]. While typical inflammation decreases when the neutrophil count falls within 1–2 days, the aggregation of these cells at the site of the wound causes an unbalanced network of regulatory cytokines, which results in a prolonged state of inflammation [209]. The majority of the bacteria in these chronic wounds exist as biofilms, which are a matrix of polysaccharides and other substances that prevent the use of antibiotics for wound healing [210,211,212]. Additionally, the challenge of treating chronic wound biofilm has worsened due to the increase in bacterial resistance to several antibiotics [213,214]. MH, TH, KH and SH have demonstrated to have wound-healing properties, as listed in Table 6. A study reported that MH eradicated methicillin-resistant S. aureus (MRSA) from colonized wounds and to inhibit MRSA in vitro by interrupting cell division. Additionally, MH restores MRSA susceptibility to oxacillin; molecular investigation revealed that it also affects the control of the mecR1 gene, presumably explaining the recovered susceptibility [215]. Another study showed that a synergistic effect between rifampicin and MH was demonstrated on clinical S. aureus isolates, including MRSA strains. Unlike with rifampicin alone [216]. Therefore, it appears that MH has a potential to provide new synergistic combinations with antibiotics for treating wound infections caused by multidrug-resistant (MDR) bacteria. It is important to note that the antibiotics that have demonstrated a synergistic with MH belong to various antibiotic classes that inhibit various targets, including the 30S ribosome, RNA polymerase, membranes, and penicillin binding proteins. This supports the idea that honey is a complex substance perhaps with multiple active components that affect more than one cellular target site [215]. MH is also known to have a relatively low pH (3.5–4.5), which, in addition to preventing microbial development, induces macrophages to engage in bactericidal activity, and, in chronic wounds, decreases protease activity while increasing fibroblast activity and oxygenation [139,203]. Previous studies found that the topical application of TH on burn wounds contaminated with P. aeruginosa and A. baumannii provided a faster healing rate than chitosan gel or hydrofibre silver treatment [217]. Another study found that TH treatment improved wound contraction and P. aeruginosa growth control better than hydrofibre and hydrofibre silver treatment [218]. In the meantime, a different study found that administration of TH via oral administration improved anastomotic wound healing in rats with large bowel anastomosis by raising the proportion of fibroblasts and lowering the proportion of inflammatory cells, enhancing wound strength [219]. In a clinical trial looking into TH effect on promoting the post-tonsillectomy healing process has been conducted by comparing sultamicillin-treated group and sultamicillin + TH-treated group. During surgery, TH was applied topically, and for seven days later, 4 mL of it was taken orally three times each day. According to the results obtained showed that TH + sultamicillin-treated group’s healing process was much faster [220]. Based on the several research stated above, TH promotes wound healing more effectively than the standard antibiotic or topical treatment, which may support its usage as a wound dressing. Keloid scar formation is related to the healing of wounds. The excessive stimulation of EMT by TGFβ in keratinocytes results in the production of keloid scars. A study found that treating with KH reduced the TGFβ induced EMT in human primary keratinocytes, demonstrating the therapeutic potential of KH in decreasing the development of keloid scars [221]. Pretreatment with KH significantly reduced (p < 0.05) both the total area of ulcer and the ulcer index compared with the positive control group, and the percentage of ulcer inhibition in the KH pre-treated group was 65.56% compared with the positive control group. The treatment, KH, exhibited antiulcer properties against ethanol-induced gastric ulcer [222]. The study by Hananeh et al. (2015) showed the beneficial effect of SH on second-intention healing of full-thickness contaminated skin wounds in dogs. Another study reported that wounds treated with SH healed as fast as those wounds treated with iodine [223]. Another study demonstrated the activity of SH to induce burns in rabbits, and the areas of the wounds caused by the burns reduced from 3.38 mm2 to 2.36 mm2 after exposure to SH [224]. Honey is generally reported to have remarkable effects compared with conventional treatments for acute wounds, superficial partial-thickness burns and infected post-operative wounds [205,206].

Table 6.
Summary of wound-healing properties of MH, TH, KH, and SH.
4.7. Effects on Nervous System
Polyphenols are crucial to honey’s ability to protect the nervous system. Various brain diseases associated with aging can be countered by the scavenger activity against ROS, which are neurotoxic. Additionally, several age-related brain diseases are caused by the buildup of misfolded proteins such beta amyloid. This abnormal buildup can be prevented by polyphenols [225]. Previous studies demonstrated that MH alleviated Aβ-induced paralysis in the Alzheimer’s disease (AD) model CL4176 through HSP-16.2 and SKN-1/Nrf2 pathways [226]. Numerous research has shown that TH enhances memory and reduces depressive-like behavior in both humans and animals. In aged rats with noise-stress-induced memory problems, TH administration improved medial prefrontal cortex and hippocampus morphology, perhaps via lowering brain oxidative stress and increasing brain-derived neurotrophic factor (BDNF) concentration and cholinergic system activity [227]. Azman et al. (2015) reported that TH administration enhanced memory function and reduced depressive-like behavior in rats exposed to loud noise stress [228]. Another study on stressed ovariectomised rats revealed that TH or 17β-estradiol treatment has been shown to demonstrate antidepressive-like effects, possibly via restoration of the hypothalamic–pituitary–adrenal axis and enhancement of the BDNF concentration [229]. Al-Rahbi et al. (2014) showed that TH has an anti-anxiety effect in ovariectomized rats by improving their oxidative stress status [230]. In another study, rats administered with TH or 17β estradiol reported improved short-term and long-term memory and enhanced neuronal proliferation of hippocampal CA2, CA3 and dentate gyrus (DG) regions compared with untreated stressed ovariectomised rats [231]. According to recent study showed that TH pretreatment significantly attenuated an increase in lipid peroxidation level and decreased the total antioxidant status level induced by KA treatment in the rat cerebral cortex [232]. Previous studies showed that TH changed and altered in oxidative stress markers at the level of the spinal cord and neuronal damage in the spinal cord morphology of the offspring of prenatally stressed rats [233]. A study on metabolic-disease-induced rats showed that the KH-treated group exhibited less anxious behavior and demonstrated significant memory retention [190]. However, the underlying mechanism through which KH exhibits this effect has still not discovered to researchers. Within the literature, KH and SH have not been explored for their activity on the nervous system in humans.
4.8. Effects on the Cardiovascular System
Within the literature, only TH was reported to affect the cardiovascular system in three studies [234,235,236]: one combined in vivo/in vitro study [237] and two human studies [188,189]. TH was found to have a beneficial effect on the cardiovascular system in all animal studies; however, human studies showed varying degrees of results. Study by Khalil et al. (2015) found that the pretreatment of ischaemic rats with TH yielded significant protective effects on cardiac troponin, triglycerides and total cholesterol [234]. Two studies revealed that TH supplementation significantly lowered high systolic blood pressure by treating spontaneously hypertensive rats. [235,236]. Previous studies reported that TH could inhibit H2O2-induced vascular hyperpermeability in vitro and in vivo by suppressing adherence junction protein redistribution via calcium and cAMP [237]. A human study evaluated the effects of 20g of TH alone and a honey cocktail (a combination of TH, beebread, and royal jelly) for 12 months. When compared to supplementing with honey cocktail, TH showed a significant impact in lowering diastolic blood pressure and FBS and honey cocktail showed remarkable effects on body mass index. In addition, the results revealed that there are no significant changes in the blood pressure of healthy adults who were on royal jelly supplementation. However, no demonstrable effect of TH on the lipid profiles and anthropometric measurements was found and authors mentioned that further studies are required to formulate the best proportion of each bioactive compound in honey cocktail in order to exclude a possible matrix effect or antagonist effect of honey cocktail and to assess the physicochemical properties of TH and honey cocktail [189]. A randomised controlled trial comparing the effects of TH and hormone replacement therapy for 4 months revealed no demonstrable impacts on blood pressure measurement, BMI, and waist circumference. No significant difference was found in the lipid profile, blood sugar profile and bone density between the two groups [188]. Honey reduced fibrinogen levels and prevented coagulation via the intrinsic, extrinsic, and common coagulation cascades. For all of the reasons mentioned, honey can be considered as being very effective at preventing the development of atherosclerotic plaques, which can result in the onset of cardiac disorders. Lipid peroxidation is a major factor in the pathophysiology of atherosclerotic plaques as well [238]. It is confirmed that the honey phenolic compounds have a preventive and protective effect towards the damaging action of free radicals, counteracting, as explained, lipid peroxidation [239,240].
4.9. Effects on Respiratory System
The majority of scientific research on the protective effects of honey on the respiratory system is focused on asthma. A chronic inflammatory condition called asthma is defined by a generally reversible obstruction of the lower airways, which frequently results from allergen activity [241]. Previous studies reported that honey inhalation is able to reduce inflammation of the lower airways in a rabbit model of ovalbumin-induced chronic asthma [242]. Another study showed that the ingestion of honey in high doses (1 g/kg body weight daily for four weeks) improved the overall symptoms for up to one month after the end of treatment [243].
5. Conclusions
It has been extensively shown that MH, TH, KH, and SH provide a variety of health benefits for many different diseases and systems. Clinical studies, particularly those on wound healing, demonstrated that MH and TH are superior to standard dressings as a wound dressing. Clinical trials using TH, KH, and SH are still required, however, to add to the findings because convincing effects have not yet been finalised. The mechanism of action of these types of honey was also unclear, particularly in human studies, providing the need for additional research. In addition, TH, KH, and SH have been well researched compared with MH. Future research may, therefore, concentrate more on the potential benefits of KH and SH. This study also showed that, in comparison to MH, TH, KH, and SH have excellent preclinical potential in a variety of diseases and physiological systems, and this could guide future research into thoroughly examining honey as an effective and proven superfood that can be optimized for the benefit of humanity.
Author Contributions
Conceptualization, M.A.A.-K., M.A., A.B.M.H. and D.H.A.; methodology, M.A.A.-K. and D.H.A.; validation, M.A.A.-K., M.A., A.B.M.H. and D.H.A.; formal analysis, M.A.A.-K. and A.B.M.H.; investigation, M.A.A.-K. and A.B.M.H.; data curation, M.A.A.-K. and D.H.A.; writing—original draft preparation, M.A.A.-K. and D.H.A.; writing-review and editing, M.A.A.-K., M.A. and A.B.M.H.; visualization, M.A.A.-K., M.A., A.B.M.H. and D.H.A.; supervision, M.A.A.-K. and A.B.M.H.; project administration, M.A.A.-K., M.A., A.B.M.H. and D.H.A.; funding acquisition, M.A.A.-K. and A.B.M.H. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by Universiti Sultan Zainal Abidin (UniSZA/2018/DPU/13: R0034-R013).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors thank all the staff members of the Faculty of Pharmacy at Al-Zaytoonah University of Jordan and the Faculty of Health Sciences at UniSZA for their support and commitment.
Conflicts of Interest
The authors declare that they have no conflict of interest.
References
- Ben Amor, S.; Mekious, S.; Allal Benfekih, L.; Abdellattif, M.H.; Boussebaa, W.; Almalki, F.A.; Ben Hadda, T.; Kawsar, S.M. Phytochemical Characterization and Bioactivity of Different Honey Samples Collected in the Pre-Saharan Region in Algeria. Life 2022, 12, 927. [Google Scholar] [CrossRef]
- Al-kafaween, M.A.; Hilmi, A.B.M.; Jaffar, N.; Al-Jamal, H.A.N.; Zahri, M.K.; Jibril, F.I. Antibacterial and Antibiofilm activities of Malaysian Trigona honey against Pseudomonas aeruginosa ATCC 10145 and Streptococcus pyogenes ATCC 19615. Jordan J. Biol. Sci. 2020, 13, 69–76. [Google Scholar]
- Rana, S.; Mishra, M.; Yadav, D.; Subramani, S.K.; Katare, C.; Prasad, G. Medicinal uses of honey: A review on its benefits to human health. Prog. Nutr. 2018, 20, 5–14. [Google Scholar]
- Nigussie, K.; Subramanian, P.; Mebrahtu, G. Physicochemical analysis of Tigray honey: An attempt to determine major quality markers of honey. Bull. Chem. Soc. Ethiop. 2012, 26, 127–133. [Google Scholar] [CrossRef]
- Bazaid, A.S.; Alamri, A.; Almashjary, M.N.; Qanash, H.; Almishaal, A.A.; Amin, J.; Binsaleh, N.K.; Kraiem, J.; Aldarhami, A.; Alafnan, A. Antioxidant, Anticancer, Antibacterial, Antibiofilm Properties and Gas Chromatography and Mass Spectrometry Analysis of Manuka Honey: A Nature’s Bioactive Honey. Appl. Sci. 2022, 12, 9928. [Google Scholar] [CrossRef]
- Makhloufi, C.; Kerkvliet, J.D.; D’albore, G.R.; Choukri, A.; Samar, R. Characterization of Algerian honeys by palynological and physico-chemical methods. Apidologie 2010, 41, 509–521. [Google Scholar] [CrossRef]
- Al-Kafaween, M.A.; Abu Bakar, M.H.; Al-Jamal, H.A.N.; Elsahoryi, N.A.; Jaffar, N.B.; Zahri, M.K. Pseudomonas aeruginosa and Streptococcus pyogenes exposed to Malaysian trigona honey in vitro demonstrated downregulation of virulence factor. Iran. J. Biotechnol. 2020, 18, 115–123. [Google Scholar]
- Benfekih, L.A.; Bellache, M.; Aoudia, B.; Mahmoudi, A. Impact of insecticides on pollinator populations: Role of phytosanitary performance indicators in tomato crops. AGROFOR Int. J. 2018, 3, 5–13. [Google Scholar] [CrossRef]
- Clearwater, M.J.; Revell, M.; Noe, S.; Manley-Harris, M. Influence of genotype, floral stage, and water stress on floral nectar yield and composition of mānuka (Leptospermum scoparium). Ann. Bot. 2018, 121, 501–512. [Google Scholar] [CrossRef]
- Bertoncelj, J.; Doberšek, U.; Jamnik, M.; Golob, T. Evaluation of the phenolic content, antioxidant activity and colour of Slovenian honey. Food Chem. 2007, 105, 822–828. [Google Scholar] [CrossRef]
- El Sohaimy, S.A.; Masry, S.; Shehata, M. Physicochemical characteristics of honey from different origins. Ann. Agric. Sci. 2015, 60, 279–287. [Google Scholar] [CrossRef]
- Özcan, M.M.; Ölmez, Ç. Some qualitative properties of different monofloral honeys. Food Chem. 2014, 163, 212–218. [Google Scholar] [CrossRef]
- Can, Z.; Yildiz, O.; Sahin, H.; Turumtay, E.A.; Silici, S.; Kolayli, S. An investigation of Turkish honeys: Their physico-chemical properties, antioxidant capacities and phenolic profiles. Food Chem. 2015, 180, 133–141. [Google Scholar] [CrossRef]
- Gomes, S.; Dias, L.G.; Moreira, L.L.; Rodrigues, P.; Estevinho, L. Physicochemical, microbiological and antimicrobial properties of commercial honeys from Portugal. Food Chem. Toxicol. 2010, 48, 544–548. [Google Scholar] [CrossRef]
- Belay, A.; Solomon, W.; Bultossa, G.; Adgaba, N.; Melaku, S. Physicochemical properties of the Harenna forest honey, Bale, Ethiopia. Food Chem. 2013, 141, 3386–3392. [Google Scholar] [CrossRef]
- Stagos, D.; Soulitsiotis, N.; Tsadila, C.; Papaeconomou, S.; Arvanitis, C.; Ntontos, A.; Karkanta, F.; Adamou-Androulaki, S.; Petrotos, K.; Spandidos, D.A. Antibacterial and antioxidant activity of different types of honey derived from Mount Olympus in Greece. Int. J. Mol. Med. 2018, 42, 726–734. [Google Scholar] [CrossRef]
- Pauliuc, D.; Dranca, F.; Oroian, M. Antioxidant activity, total phenolic content, individual phenolics and physicochemical parameters suitability for Romanian honey authentication. Foods 2020, 9, 306. [Google Scholar] [CrossRef]
- Afrin, S.; Giampieri, F.; Gasparrini, M.; Forbes-Hernández, T.Y.; Cianciosi, D.; Reboredo-Rodriguez, P.; Manna, P.P.; Zhang, J.; Quiles, J.L.; Battino, M. The inhibitory effect of Manuka honey on human colon cancer HCT-116 and LoVo cell growth. Part 2: Induction of oxidative stress, alteration of mitochondrial respiration and glycolysis, and suppression of metastatic ability. Food Funct. 2018, 9, 2158–2170. [Google Scholar] [CrossRef]
- Blasa, M.; Candiracci, M.; Accorsi, A.; Piacentini, M.P.; Albertini, M.C.; Piatti, E. Raw Millefiori honey is packed full of antioxidants. Food Chem. 2006, 97, 217–222. [Google Scholar] [CrossRef]
- Beretta, G.; Granata, P.; Ferrero, M.; Orioli, M.; Facino, R.M. Standardization of antioxidant properties of honey by a combination of spectrophotometric/fluorimetric assays and chemometrics. Anal. Chim. Acta 2005, 533, 185–191. [Google Scholar] [CrossRef]
- Grecka, K.; Kuś, P.M.; Worobo, R.W.; Szweda, P. Study of the anti-staphylococcal potential of honeys produced in Northern Poland. Molecules 2018, 23, 260. [Google Scholar] [CrossRef]
- Bouacha, M.; Boudiar, I.; Akila, A.; Al-Kafaween, M.A.; Khallef, M. The antimutagenic effect of multifloral honey in salmonella/microsomal assay and its correlation with the total polyphenolic content. J. Microbiol. Biotechnol. Food Sci. 2022, 11, e5557. [Google Scholar] [CrossRef]
- Abd-El Aal, A.; El-Hadidy, M.; El-Mashad, N.; El-Sebaie, A. Antimicrobial effect of bee honey in comparison to antibiotics on organisms isolated from infected burns. Ann. Burn. Fire Disasters 2007, 20, 83. [Google Scholar]
- Al-kafaween, M.A.; Al-Jamal, H.A.N. A comparative study of antibacterial and antivirulence activities of four selected honeys to Manuka honey. Iran. J. Microbiol. 2022, 14, 238–251. [Google Scholar] [CrossRef]
- Bouacha, M.; Besnaci, S.; Boudiar, I.; Al-kafaween, M.A. Impact of Storage on Honey Antibacterial and Antioxidant Activities and their Correlation with Polyphenolic Content: Doi. org/10.26538/tjnpr/v6i1. 7. Trop. J. Nat. Prod. Res. (TJNPR) 2022, 6, 34–39. [Google Scholar]
- Moniruzzaman, M.; Yung An, C.; Rao, P.V.; Hawlader, M.N.I.; Azlan, S.A.B.M.; Sulaiman, S.A.; Gan, S.H. Identification of phenolic acids and flavonoids in monofloral honey from Bangladesh by high performance liquid chromatography: Determination of antioxidant capacity. BioMed Res. Int. 2014, 2014, 737490. [Google Scholar] [CrossRef]
- Padayachee, A.; Netzel, G.; Netzel, M.; Day, L.; Zabaras, D.; Mikkelsen, D.; Gidley, M. Binding of polyphenols to plant cell wall analogues–Part 2: Phenolic acids. Food Chem. 2012, 135, 2287–2292. [Google Scholar] [CrossRef]
- Hossen, M.S.; Ali, M.Y.; Jahurul, M.; Abdel-Daim, M.M.; Gan, S.H.; Khalil, M.I. Beneficial roles of honey polyphenols against some human degenerative diseases: A review. Pharmacol. Rep. 2017, 69, 1194–1205. [Google Scholar] [CrossRef]
- Escriche, I.; Kadar, M.; Juan-Borrás, M.; Domenech, E. Suitability of antioxidant capacity, flavonoids and phenolic acids for floral authentication of honey. Impact of industrial thermal treatment. Food Chem. 2014, 142, 135–143. [Google Scholar] [CrossRef]
- Nweze, A.; Olovo, C.V.; Nweze, E.I.; John, O.O.; Paul, C. Therapeutic properties of honey. Honey Anal. New Adv. Chall 2020, 332, 1–21. [Google Scholar]
- Vazhacharickal, P.J. Biological action and health benefits of honey, propolis and royal jelly: An overview. Pharma Innov. J. 2021, 10, 28–30. [Google Scholar]
- Al-Kafaween, M.A.; Hilmi, A.B.M.; Al-Jamal, H.A.N. Physicochemical and Therapeutic Properties of Malaysian Stingless Bee Kelulut Honey in Comparison with Yemeni Sidr Honey. Anti-Infective Agents. Anti-Infect. Agents 2022, 20, 150–159. [Google Scholar]
- Rao, P.V.; Krishnan, K.T.; Salleh, N.; Gan, S.H. Biological and therapeutic effects of honey produced by honey bees and stingless bees: A comparative review. Rev. Bras. De Farmacogn. 2016, 26, 657–664. [Google Scholar] [CrossRef]
- Mohd Kamal, D.A.; Ibrahim, S.F.; Kamal, H.; Kashim, M.I.A.M.; Mokhtar, M.H. Physicochemical and Medicinal Properties of Tualang, Gelam and Kelulut Honeys: A Comprehensive Review. Nutrients 2021, 13, 197. [Google Scholar] [CrossRef]
- Solayman, M.; Islam, M.A.; Paul, S.; Ali, Y.; Khalil, M.I.; Alam, N.; Gan, S.H. Physicochemical properties, minerals, trace elements, and heavy metals in honey of different origins: A comprehensive review. Compr. Rev. Food Sci. Food Saf. 2016, 15, 219–233. [Google Scholar] [CrossRef]
- Hegazi, A.G.; Al Guthami, F.M.; Al Gethami, A.F.; Abd Allah, F.M.; Saleh, A.A.; Fouad, E.A. Potential antibacterial activity of some Saudi Arabia honey. Vet. World 2017, 10, 233. [Google Scholar] [CrossRef]
- Ahmed, S.; Sulaiman, S.A.; Baig, A.A.; Ibrahim, M.; Liaqat, S.; Fatima, S.; Jabeen, S.; Shamim, N.; Othman, N.H. Honey as a potential natural antioxidant medicine: An insight into its molecular mechanisms of action. Oxid. Med. Cell. Longev. 2018, 2018, 8367846. [Google Scholar] [CrossRef]
- Živkov-Baloš, M.; Popov, N.; Vidaković, S.; Ljubojević-Pelić, D.; Pelić, M.; Mihaljev, Ž.; Jakšić, S. Electrical conductivity and acidity of honey. Arch. Veter Med. 2018, 11, 91–101. [Google Scholar] [CrossRef]
- Kropf, U.; Jamnik, M.; Bertoncelj, J.; Golob, T. Linear regression model of the ash mass fraction and electrical conductivity for Slovenian honey. Food Technol. Biotechnol. 2008, 46, 335–340. [Google Scholar]
- Rahaman, N.L.; Chua, L.S.; Sarmidi, M.R.; Aziz, R. Physicochemical and radical scavenging activities of honey samples from Malaysia. Agric. Sci. 2013, 4, 46. [Google Scholar]
- Chua, L.S.; Abdul-Rahaman, N.-L.; Sarmidi, M.R.; Aziz, R. Multi-elemental composition and physical properties of honey samples from Malaysia. Food Chem. 2012, 135, 880–887. [Google Scholar] [CrossRef] [PubMed]
- Thrasyvoulou, A.; Tananaki, C.; Goras, G.; Karazafiris, E.; Dimou, M.; Liolios, V.; Kanelis, D.; Gounari, S. Legislation of honey criteria and standards. J. Apic. Res. 2018, 57, 88–96. [Google Scholar] [CrossRef]
- Ciulu, M.; Solinas, S.; Floris, I.; Panzanelli, A.; Pilo, M.I.; Piu, P.C.; Spano, N.; Sanna, G. RP-HPLC determination of water-soluble vitamins in honey. Talanta 2011, 83, 924–929. [Google Scholar] [CrossRef] [PubMed]
- Pavlova, T.; Stamatovska, V.; Kalevska, T.; Dimov, I.; Assistant, G.; Nakov, G. Quality characteristics of honey: A review. Proc. Univ. Ruse 2018, 57, 31–37. [Google Scholar]
- Ahmed, S.; Othman, N.H. Review of the medicinal effects of tualang honey and a comparison with manuka honey. Malays. J. Med. Sci. MJMS 2013, 20, 6. [Google Scholar]
- Ariefdjohan, M.W.; Martin, B.R.; Lachcik, P.J.; Weaver, C.M. Acute and chronic effects of honey and its carbohydrate constituents on calcium absorption in rats. J. Agric. Food Chem. 2008, 56, 2649–2654. [Google Scholar] [CrossRef]
- Zulkhairi Amin, F.A.; Sabri, S.; Mohammad, S.M.; Ismail, M.; Chan, K.W.; Ismail, N.; Norhaizan, M.E.; Zawawi, N. Therapeutic properties of stingless bee honey in comparison with european bee honey. Adv. Pharmacol. Sci. 2018, 2018, 6179596. [Google Scholar] [CrossRef]
- Olas, B. Honey and its phenolic compounds as an effective natural medicine for cardiovascular diseases in humans? Nutrients 2020, 12, 283. [Google Scholar] [CrossRef]
- Al-Haik, M.; Al-Haddad, M.; Al-Kaf, G.; Edrees, H. Antimicrobial activities for hadhrami honey on growth of some pathogenic bacteria. Univers. J. Pharm. Res. 2017, 2, 7–12. [Google Scholar] [CrossRef]
- Emmertz, A. Mineral Composition of New Zealand Monofloral Honeys; Sveriges Lantbruksuniversitet: Uppsala, Sweden, 2010. [Google Scholar]
- Moniruzzaman, M.; Khalil, M.I.; Sulaiman, S.A.; Gan, S.H. Physicochemical and antioxidant properties of Malaysian honeys produced by Apis cerana, Apis dorsata and Apis mellifera. BMC Complement. Altern. Med. 2013, 13, 43. [Google Scholar] [CrossRef]
- Lim, D.; Bakar, M.A.; Majid, M. Nutritional composition of stingless bee honey from different botanical origins. IOP Conf. Ser. Earth Environ. Sci. 2019, 269, 012025. [Google Scholar]
- Shamsudin, S.; Selamat, J.; Sanny, M.; AR, S.B.; Jambari, N.N.; Khatib, A. A comparative characterization of physicochemical and antioxidants properties of processed Heterotrigona itama honey from different origins and classification by chemometrics analysis. Molecules 2019, 24, 3898. [Google Scholar] [CrossRef]
- Wong, P.; Ling, H.S.; Chung, K.C.; Yau, T.M.S.; Gindi, S.R.A. Chemical Analysis on the Honey of Heterotrigona itama and Tetrigona binghami from Sarawak, Malaysia. Sains Malays. 2019, 48, 1635–1642. [Google Scholar] [CrossRef]
- Saeed, M.A.; Jayashankar, M. Physico-chemical characteristics of some Indian and Yemeni Honey. J. Bioenergy Food Sci. 2020, 7, 2832019. [Google Scholar] [CrossRef]
- Khadra, Y.; MK, N.S.A.; AA, N.N.; Shukri, R.; Nor-Khaizura, M. Physicochemical and Microbiological Quality of Selected Commercial and Traditional Honey in Klang Valley Market, Malaysia. J. Sci. Technol. 2018, 10, 71–76. [Google Scholar]
- Kek, S.P.; Chin, N.L.; Yusof, Y.A.; Tan, S.W.; Chua, L.S. Classification of entomological origin of honey based on its physicochemical and antioxidant properties. Int. J. Food Prop. 2017, 20, S2723–S2738. [Google Scholar] [CrossRef]
- Morroni, G.; Alvarez-Suarez, J.M.; Brenciani, A.; Simoni, S.; Fioriti, S.; Pugnaloni, A.; Giampieri, F.; Mazzoni, L.; Gasparrini, M.; Marini, E. Comparison of the antimicrobial activities of four honeys from three countries (New Zealand, Cuba, and Kenya). Front. Microbiol. 2018, 9, 1378. [Google Scholar] [CrossRef]
- Rashid, M.R.; Nor Aripin, K.N.; Syed Mohideen, F.B.; Baharom, N.; Omar, K.; Md Taujuddin, N.M.S.; Mohd Yusof, H.H.; Addnan, F.H. The effect of Kelulut honey on fasting blood glucose and metabolic parameters in patients with impaired fasting glucose. J. Nutr. Metab. 2019, 2019, 3176018. [Google Scholar] [CrossRef]
- Stephens, J.M.; Schlothauer, R.C.; Morris, B.D.; Yang, D.; Fearnley, L.; Greenwood, D.R.; Loomes, K.M. Phenolic compounds and methylglyoxal in some New Zealand manuka and kanuka honeys. Food Chem. 2010, 120, 78–86. [Google Scholar] [CrossRef]
- Erejuwa, O.; Sulaiman, S.; Wahab, M.; Sirajudeen, K.; Salleh, M.M.; Gurtu, S. Antioxidant protection of Malaysian tualang honey in pancreas of normal and streptozotocin-induced diabetic rats. Ann. D’endocrinologie 2021, 71, 291–296. [Google Scholar] [CrossRef]
- Hegazi, A.G.; Al Guthami, F.M.; Ramadan, M.F.; Al Gethami, A.F.; Craig, A.M.; Serrano, S. Characterization of Sidr (Ziziphus spp.) Honey from Different Geographical Origins. Appl. Sci. 2022, 12, 9295. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Sulaiman, S.A.; Khalil, M.I.; Gan, S.H. Evaluation of physicochemical and antioxidant properties of sourwood and other Malaysian honeys: A comparison with manuka honey. Chem. Cent. J. 2013, 7, 138. [Google Scholar] [CrossRef] [PubMed]
- Alkhyat, S.H.; Maqtari, M.A.A. Antibacterial Potential and Physicochemical Properties of Selected Yemeni Honeys Against Clinical Wounds Bacteria and Comparative with Standard Bacteria Isolates. Glob. Adv. Res. J. 2014, 3, 49–58. [Google Scholar]
- Moniruzzaman, M.; Chowdhury, M.A.Z.; Rahman, M.A.; Sulaiman, S.A.; Gan, S.H. Determination of mineral, trace element, and pesticide levels in honey samples originating from different regions of Malaysia compared to Manuka honey. BioMed Res. Int. 2014, 2014, 359890. [Google Scholar] [CrossRef]
- Taha, M.M.E.; Abdelwahab, S.I.; Elsanousi, R.; Sheikh, B.Y.; Abdulla, M.A.; Babiker, S.E.; Elraih, H.; Mohamed, E. Effectiveness of Sidr Honey on the prevention of ethanol-induced gatroulcerogenesis: Role of antioxidant and antiapoptotic mechanism. Pharmacogn. J. 2015, 7, 157–164. [Google Scholar] [CrossRef]
- Cianciosi, D.; Forbes-Hernández, T.Y.; Afrin, S.; Gasparrini, M.; Reboredo-Rodriguez, P.; Manna, P.P.; Zhang, J.; Bravo Lamas, L.; Martínez Flórez, S.; Agudo Toyos, P. Phenolic compounds in honey and their associated health benefits: A review. Molecules 2018, 23, 2322. [Google Scholar] [CrossRef]
- Khalil, M.; Alam, N.; Moniruzzaman, M.; Sulaiman, S.; Gan, S. Phenolic acid composition and antioxidant properties of Malaysian honeys. J. Food Sci. 2011, 76, C921–C928. [Google Scholar] [CrossRef]
- da Silva, I.A.A.; da Silva, T.M.S.; Camara, C.A.; Queiroz, N.; Magnani, M.; de Novais, J.S.; Soledade, L.E.B.; de Oliveira Lima, E.; de Souza, A.L.; de Souza, A.G. Phenolic profile, antioxidant activity and palynological analysis of stingless bee honey from Amazonas, Northern Brazil. Food Chem. 2013, 141, 3552–3558. [Google Scholar] [CrossRef]
- Alshammari, G.M.; Ahmed, M.A.; Alsulami, T.; Hakeem, M.J.; Ibraheem, M.A.; Al-Nouri, D.M. Phenolic Compounds, Antioxidant Activity, Ascorbic Acid, and Sugars in Honey from Ingenious Hail Province of Saudi Arabia. Appl. Sci. 2022, 12, 8334. [Google Scholar] [CrossRef]
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative stress, prooxidants, and antioxidants: The interplay. BioMed Res. Int. 2014, 2014, 761264. [Google Scholar] [CrossRef]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.; Rahu, N. Oxidative stress and inflammation: What polyphenols can do for us? Oxid. Med. Cell. Longev. 2016, 2016, 7432797. [Google Scholar] [CrossRef]
- Banerjee, S.; Ghosh, J.; Sil, P. Drug metabolism and oxidative stress: Cellular mechanism and new therapeutic insights. Biochem. Anal. Biochem. 2016, 5, 255. [Google Scholar] [CrossRef]
- Yang, Y.; Bazhin, A.V.; Werner, J.; Karakhanova, S. Reactive oxygen species in the immune system. Int. Rev. Immunol. 2013, 32, 249–270. [Google Scholar] [CrossRef] [PubMed]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. IJBS 2008, 4, 89. [Google Scholar] [PubMed]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118. [Google Scholar] [CrossRef]
- Mohamed, M.; Sirajudeen, K.; Swamy, M.; Yaacob, M.; Sulaiman, S. Studies on the antioxidant properties of Tualang honey of Malaysia. Afr. J. Tradit. Complement. Altern. Med. 2010, 7, 59–63. [Google Scholar] [CrossRef]
- Azman, K.F.; Zakaria, R. Honey as an antioxidant therapy to reduce cognitive ageing. Iran. J. Basic Med. Sci. 2019, 22, 1368. [Google Scholar] [PubMed]
- Kishore, R.K.; Halim, A.S.; Syazana, M.N.; Sirajudeen, K. Tualang honey has higher phenolic content and greater radical scavenging activity compared with other honey sources. Nutr. Res. 2011, 31, 322–325. [Google Scholar] [CrossRef] [PubMed]
- Jubri, Z.; Rahim, N.B.A.; Aan, G.J. Manuka honey protects middle-aged rats from oxidative damage. Clinics 2013, 68, 1446–1454. [Google Scholar] [CrossRef]
- Saeed, M.A.; Jayashankar, M.; Tejaswini, H. Evaluation of biochemical and antioxidant properties of Indian and Yemeni honey. J. Apic. Res. 2021, 13, 1–14. [Google Scholar] [CrossRef]
- Omotayo, E.O.; Gurtu, S.; Sulaiman, S.A.; Ab Wahab, M.S.; KNS, S.; Salleh, M.S.M. Hypoglycemic and antioxidant effects of honey supplementation in streptozotocin-induced diabetic rats. Int. J. Vitam. Nutr. Res. 2010, 80, 74–82. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.S.A.; Salam, S.K.N.; Salleh, M.S.M.; Gurtu, S. Comparison of antioxidant effects of honey, glibenclamide, metformin, and their combinations in the kidneys of streptozotocin-induced diabetic rats. Int. J. Mol. Sci. 2011, 12, 829–843. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.J.; Azmi, S.M.; Yong, Y.K.; Cheah, H.L.; Lim, V.; Sandai, D.; Shaharuddin, B. Tualang honey improves human corneal epithelial progenitor cell migration and cellular resistance to oxidative stress in vitro. PLoS ONE 2014, 9, e96800. [Google Scholar] [CrossRef]
- Bashkaran, K.; Zunaina, E.; Bakiah, S.; Sulaiman, S.A.; Sirajudeen, K.; Naik, V. Anti-inflammatory and antioxidant effects of Tualang honey in alkali injury on the eyes of rabbits: Experimental animal study. BMC Complement. Altern. Med. 2011, 11, 90. [Google Scholar] [CrossRef] [PubMed]
- Kamaruzzaman, M.A.; Thanu, A.; Yusof, M.R.; Soelaiman, I.N.; Ramli, E.S. Kelulut honey ameliorates glucocorticoid induced osteoporosis via its antioxidant activity in rats. Asian Pac. J. Trop. Biomed. 2019, 9, 493. [Google Scholar]
- Budin, S.B.; Jubaidi, F.F.; Azam, S.N.F.M.N.; Yusof, N.L.M.; Taib, I.S.; Mohamed, J. Kelulut honey supplementation prevents sperm and testicular oxidative damage in streptozotocin-induced diabetic rats. J. Teknol. 2017, 79, 89–95. [Google Scholar] [CrossRef]
- Asari, M.A.; Zulkaflee, M.H.; Sirajudeen, K.; Yusof, N.A.M.; Sairazi, N.S.M. Tualang honey and DHA-rich fish oil reduce the production of pro-inflammatory cytokines in the rat brain following exposure to chronic stress. J. Taibah Univ. Med. Sci. 2019, 14, 317–323. [Google Scholar] [CrossRef]
- Ghazali, W.S.W.; Romli, A.C.; Mohamed, M. Effects of honey supplementation on inflammatory markers among chronic smokers: A randomized controlled trial. BMC Complement. Altern. Med. 2017, 17, 175. [Google Scholar] [CrossRef]
- Medhi, B.; Prakash, A.; Avti, P.; Saikia, U.; Pandhi, P.; Khanduja, K. Effect of Manuka honey and sulfasalazine in combination to promote antioxidant defense system in experimentally induced ulcerative colitis model in rats. Experiment 2008, 46, 583–590. [Google Scholar]
- Minden-Birkenmaier, B.A.; Meadows, M.B.; Cherukuri, K.; Smeltzer, M.P.; Smith, R.A.; Radic, M.Z.; Bowlin, G.L. Manuka honey modulates the release profile of a dHL-60 neutrophil model under anti-inflammatory stimulation. J. Tissue Viability 2020, 29, 91–99. [Google Scholar] [CrossRef]
- Afrin, S.; Gasparrini, M.; Forbes-Hernández, T.Y.; Cianciosi, D.; Reboredo-Rodriguez, P.; Manna, P.P.; Battino, M.; Giampieri, F. Protective effects of Manuka honey on LPS-treated RAW 264.7 macrophages. Part 1: Enhancement of cellular viability, regulation of cellular apoptosis and improvement of mitochondrial functionality. Food Chem. Toxicol. 2018, 121, 203–213. [Google Scholar] [CrossRef]
- Saiful Yazan, L.; Muhamad Zali, M.F.S.; Mohd Ali, R.; Zainal, N.A.; Esa, N.; Sapuan, S.; Ong, Y.S.; Tor, Y.S.; Gopalsamy, B.; Voon, F.L. Chemopreventive properties and toxicity of Kelulut honey in Sprague Dawley rats induced with Azoxymethane. BioMed Res. Int. 2016, 2016, 4036926. [Google Scholar] [CrossRef]
- Kek, S.P.; Chin, N.L.; Yusof, Y.A.; Tan, S.W.; Chua, L.S. Total phenolic contents and colour intensity of Malaysian honeys from the Apis spp. and Trigona spp. bees. Agric. Agric. Sci. Procedia 2014, 2, 150–155. [Google Scholar] [CrossRef]
- Kuropatnicki, A.K.; Kłósek, M.; Kucharzewski, M. Honey as medicine: Historical perspectives. J. Apic. Res. 2018, 57, 113–118. [Google Scholar] [CrossRef]
- Al-kafaween, M.A.; Mohd Hilmi, A.B.; Jaffar, N.; Nagi Al-Jamal, H.A.; Zahri, M.K.; Amonov, M.; Mabrouka, B.; Elsahoryi, N.A. Effects of Trigona honey on the Gene Expression Profile of Pseudomonas aeruginosa ATCC 10145 and Streptococcus pyogenes ATCC 19615. Jordan J. Biol. Sci. 2020, 13, 133–138. [Google Scholar]
- Rani, G.N.; Budumuru, R.; Bandaru, N.R. Antimicrobial activity of honey with special reference to methicillin resistant Staphylococcus aureus (MRSA) and methicillin sensitive Staphylococcus aureus (MSSA). J. Clin. Diagn. Res. JCDR 2017, 11, DC05. [Google Scholar] [CrossRef]
- Mohammed, S.E.A.; Kabbashi, A.S.; Koko, W.S.; Ansari, M.J.; Adgaba, N.; Al-Ghamdi, A. In vitro activity of some natural honeys against Entamoeba histolytica and Giardia lamblia trophozoites. Saudi J. Biol. Sci. 2019, 26, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Al-kafaween, M.A.; Hilmi, A.B.M.; Al-Jamal, H.A.N. The Beneficial Effects of Stingless Bee Kelulut Honey against Pseudomonas aeruginosa and Streptococcus pyogenes Planktonic and Biofilm: Doi. org/10.26538/tjnpr/v5i10. 15. Trop. J. Nat. Prod. Res. (TJNPR) 2021, 5, 1788–1796. [Google Scholar]
- Ibarguren, C.; Raya, R.R.; Apella, M.C.; Audisio, M.C. Enterococcus faecium isolated from honey synthesized bacteriocin-like substances active against different Listeria monocytogenes strains. J. Microbiol. 2010, 48, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Aween, M.M.; Hassan, Z.; Muhialdin, B.J.; Noor, H.M.; Eljamel, Y.A. Evaluation on antibacterial activity of Lactobacillus acidophilus strains isolated from honey. Am. J. Appl. Sci. 2012, 9, 807–817. [Google Scholar] [CrossRef]
- Mathialagan, M.; Edward, Y.; David, P.; Senthilkumar, M.; Srinivasan, M.; Mohankumar, S. Isolation, characterization and identification of probiotic lactic acid bacteria (LAB) from honey bees. Int. J Curr. Microbiol Appl. Sci. 2018, 7, 849–906. [Google Scholar] [CrossRef]
- Mandal, M.D.; Mandal, S. Honey: Its medicinal property and antibacterial activity. Asian Pac. J. Trop. Biomed. 2011, 1, 154–160. [Google Scholar] [CrossRef]
- Johnston, M.; McBride, M.; Dahiya, D.; Owusu-Apenten, R.; Nigam, P.S. Antibacterial activity of Manuka honey and its components: An overview. AIMS Microbiol. 2018, 4, 655. [Google Scholar] [CrossRef]
- Gośliński, M.; Nowak, D.; Kłębukowska, L. Antioxidant properties and antimicrobial activity of manuka honey versus Polish honeys. J. Food Sci. Technol. 2020, 57, 1269–1277. [Google Scholar] [CrossRef]
- Girma, A.; Seo, W.; She, R.C. Antibacterial activity of varying UMF-graded Manuka honeys. PLoS ONE 2019, 14, e0224495. [Google Scholar] [CrossRef]
- Almasaudi, S.B.; Al-Nahari, A.A.; El Sayed, M.; Barbour, E.; Al Muhayawi, S.M.; Al-Jaouni, S.; Azhar, E.; Qari, M.; Qari, Y.A.; Harakeh, S. Antimicrobial effect of different types of honey on Staphylococcus aureus. Saudi J. Biol. Sci. 2017, 24, 1255–1261. [Google Scholar] [CrossRef]
- Feknous, N.; Boumendjel, M. Natural bioactive compounds of honey and their antimicrobial activity. Czech J. Food Sci. 2022, 40, 163–178. [Google Scholar] [CrossRef]
- Shenoy, V.P.; Ballal, M.; Shivananda, P.; Bairy, I. Honey as an antimicrobial agent against Pseudomonas aeruginosa isolated from infected wounds. J. Glob. Infect. Dis. 2012, 4, 102. [Google Scholar]
- Ahmadi–Motamayel, F.; Hendi, S.S.; Alikhani, M.Y.; Khamverdi, Z. Antibacterial activity of honey on cariogenic bacteria. J. Dent. 2013, 10, 10–15. [Google Scholar]
- McLoone, P.; Warnock, M.; Fyfe, L. Honey: A realistic antimicrobial for disorders of the skin. J. Microbiol. Immunol. Infect. 2016, 49, 161–167. [Google Scholar] [CrossRef]
- Mohd-Aspar, M.A.S.; Edros, R.Z.; Hamzah, N.A. Antibacterial Properties of Kelulut, Tualang and Acacia Honey against Wound-Infecting Bacteria. Pertanika J. Trop. Agric. Sci. 2019, 42, 1185–1208. [Google Scholar]
- Omar, S.; Mat-Kamir, N.F.; Sanny, M. Antibacterial activity of Malaysian produced stingless-bee honey on wound pathogens. J. Sustain. Sci. Manag. 2019, 14, 67–79. [Google Scholar]
- Ng, W.-J.; Sit, N.-W.; Ooi, P.A.-C.; Ee, K.-Y.; Lim, T.-M. The antibacterial potential of honeydew honey produced by stingless bee (Heterotrigona itama) against antibiotic resistant bacteria. Antibiotics 2020, 9, 871. [Google Scholar] [CrossRef]
- Nasir, N.-A.M.; Halim, A.S.; Singh, K.-K.B.; Dorai, A.A.; Haneef, M.-N.M. Antibacterial properties of tualang honey and its effect in burn wound management: A comparative study. BMC Complement. Altern. Med. 2010, 10, 31. [Google Scholar] [CrossRef]
- Tan, H.T.; Rahman, R.A.; Gan, S.H.; Halim, A.S.; Hassan, S.A.; Sulaiman, S.A.; BS, K.-K. The antibacterial properties of Malaysian tualang honey against wound and enteric microorganisms in comparison to manuka honey. BMC Complement. Altern. Med. 2009, 9, 34. [Google Scholar] [CrossRef]
- Küçük, M.; Kolaylı, S.; Karaoğlu, Ş.; Ulusoy, E.; Baltacı, C.; Candan, F. Biological activities and chemical composition of three honeys of different types from Anatolia. Food Chem. 2007, 100, 526–534. [Google Scholar] [CrossRef]
- Shahzad, A. In vitro antiviral activity of honey against varicella zoster virus (VZV): A translational medicine study for potential remedy for shingles. Transl. Biomed. 2012, 3, 2. [Google Scholar]
- Watanabe, K.; Rahmasari, R.; Matsunaga, A.; Haruyama, T.; Kobayashi, N. Anti-influenza viral effects of honey in vitro: Potent high activity of manuka honey. Arch. Med. Res. 2014, 45, 359–365. [Google Scholar] [CrossRef]
- Behbahani, M. Anti-HIV-1 activity of eight monofloral Iranian honey types. PLoS ONE 2014, 9, e108195. [Google Scholar] [CrossRef]
- Obossou, E.K.; Shikamoto, Y.; Hoshino, Y.; Kohno, H.; Ishibasi, Y.; Kozasa, T.; Taguchi, M.; Sakakibara, I.; Tonooka, K.; Shinozuka, T. Effect of manuka honey on human immunodeficiency virus type 1 reverse transcriptase activity. Nat. Prod. Res. 2022, 36, 1552–1557. [Google Scholar] [CrossRef]
- Irish, J.; Carter, D.A.; Shokohi, T.; Blair, S.E. Honey has an antifungal effect against Candida species. Med. Mycol. 2006, 44, 289–291. [Google Scholar] [CrossRef]
- Moussa, A.; Noureddine, D.; Saad, A.; Abdelmelek, M.; Abdelkader, B. Antifungal activity of four honeys of different types from Algeria against pathogenic yeast: Candida albicans and Rhodotorula sp. Asian Pac. J. Trop. Biomed. 2012, 2, 554–557. [Google Scholar] [CrossRef] [PubMed]
- Guttentag, A.; Krishnakumar, K.; Cokcetin, N.; Hainsworth, S.; Harry, E.; Carter, D. Inhibition of dermatophyte fungi by Australian jarrah honey. Pathogens 2021, 10, 194. [Google Scholar] [CrossRef] [PubMed]
- Perna, A.; Intaglietta, I.; Simonetti, A.; Gambacorta, E. A comparative study on phenolic profile, vitamin C content and antioxidant activity of Italian honeys of different botanical origin. Int. J. Food Sci. Technol. 2013, 48, 1899–1908. [Google Scholar] [CrossRef]
- Yap, P.; Abu Bakar, M.F.; Lim, H.; Carrier, D. Antibacterial activity of polyphenol-rich extract of selected wild honey collected in Sabah, Malaysia. J. Apic. Res. 2015, 54, 163–172. [Google Scholar] [CrossRef]
- Albaridi, N.A. Antibacterial potency of honey. Int. J. Microbiol. 2019, 2019, 2464507. [Google Scholar] [CrossRef]
- Weston, R.J. The contribution of catalase and other natural products to the antibacterial activity of honey: A review. Food Chem. 2000, 71, 235–239. [Google Scholar] [CrossRef]
- Allen, K.L.; Molan, P.C.; Reid, G. A survey of the antibacterial activity of some New Zealand honeys. J. Pharm. Pharmacol. 1991, 43, 817–822. [Google Scholar] [CrossRef]
- Zainol, M.I.; Mohd Yusoff, K.; Mohd Yusof, M.Y. Antibacterial activity of selected Malaysian honey. BMC Complement. Altern. Med. 2013, 13, 129. [Google Scholar] [CrossRef] [PubMed]
- Alkafaween, M.A.; Kafaween, H.; Al-Groom, R.M. A Comparative Study of Antibacterial Activity of Citrus and Jabali Honeys with Manuka Honey. Appl. Environ. Biotechnol. 2022, 7, 28–37. [Google Scholar] [CrossRef]
- Mohan, A.; Quek, S.-Y.; Gutierrez-Maddox, N.; Gao, Y.; Shu, Q. Effect of honey in improving the gut microbial balance. Food Qual. Saf. 2017, 1, 107–115. [Google Scholar] [CrossRef]
- Mullai, V.; Menon, T. Bactericidal activity of different types of honey against clinical and environmental isolates of Pseudomonas aeruginosa. J. Altern. Complement. Med. 2007, 13, 439–442. [Google Scholar] [CrossRef]
- Henriques, A.; Jenkins, R.; Burton, N.; Cooper, R. The intracellular effects of manuka honey on Staphylococcus aureus. Eur. J. Clin. Microbiol. Infect. Dis. 2010, 29, 45–50. [Google Scholar] [CrossRef]
- Henriques, A.; Jenkins, R.; Burton, N.; Cooper, R. The effect of manuka honey on the structure of Pseudomonas aeruginosa. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 167–171. [Google Scholar] [CrossRef]
- Campeau, M.E.; Patel, R. Antibiofilm activity of Manuka honey in combination with antibiotics. Int. J. Bacteriol. 2014, 2014, 795281. [Google Scholar] [CrossRef] [PubMed]
- Roberts, A.E.; Maddocks, S.E.; Cooper, R.A. Manuka honey reduces the motility of Pseudomonas aeruginosa by suppression of flagella-associated genes. J. Antimicrob. Chemother. 2015, 70, 716–725. [Google Scholar] [CrossRef]
- Abbas, H.A. Comparative antibacterial and antibiofilm activities of manuka honey and Egyptian clover honey. Asian J. Appl. Sci. 2014, 2, 110–115. [Google Scholar]
- Alvarez-Suarez, J.M.; Gasparrini, M.; Forbes-Hernández, T.Y.; Mazzoni, L.; Giampieri, F. The composition and biological activity of honey: A focus on Manuka honey. Foods 2014, 3, 420–432. [Google Scholar] [CrossRef]
- Halawani, E.M.; Shohayeb, M.M. Shaoka and Sidr honeys surpass in their antibacterial activity local and imported honeys available in Saudi markets against pathogenic and food spoilage bacteria. Aust. J. Basic Appl. Sci. 2011, 5, 187–191. [Google Scholar]
- Ng, W.; Ken, K.-W.; Kumar, R.-V.; Gunasagaran, H.; Chandramogan, V.; Lee, Y.-Y. In-vitro screening of Malaysian honey from different floral sources for antibacterial activity on human pathogenic bacteria. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 315–318. [Google Scholar] [CrossRef]
- Tumin, N.; Halim, N.; Shahjahan, M.; Noor Izani, N.; Sattar, M.A.; Khan, A.H.; Mohsin, S. Antibacterial activity of local Malaysian honey. Malays. J. Pharm. Sci. 2005, 3, 1–10. [Google Scholar]
- Aween, M.M.; Hassan, Z.; Muhialdin, B.J.; Eljamel, Y.A.; Al-Mabrok, A.S.W.; Lani, M.N. Antibacterial activity of Lactobacillus acidophilus strains isolated from honey marketed in malaysia against selected multiple antibiotic resistant (mar) gram-positive bacteria. J. Food Sci. 2012, 77, M364–M371. [Google Scholar] [CrossRef] [PubMed]
- Al Kafaween, M.A.; Hilmi, A.B.M.; Khan, R.S.; Bouacha, M.; Amonov, M. Effect of Trigona honey on Escherichia coli cell culture growth: In vitro study. J. Apitherapy 2019, 5, 10–17. [Google Scholar] [CrossRef]
- Al-kafaween, M.A.; Mohd Hilmi, A.B.; Nagi Al-Jamal, H.A.; Jaffar, N.; Al-Sayyed, H.; Zahri, M.K. Effects of Selected Malaysian Kelulut Honey on Biofilm Formation and the Gene Expression Profile of Staphylococcus aureus, Pseudomonas aeruginosa and Escherichia coli. Jordan J. Pharm. Sci. 2021, 14, 9–26. [Google Scholar]
- Ng, W.J.; Chan, Y.J.; Lau, Z.K.; Lye, P.Y.; Ee, K.Y. Antioxidant properties and inhibitory effects of trigona honey against Staphylococcus aureus planktonic and biofilm cultures. GEOMATE J. 2017, 13, 28–33. [Google Scholar]
- Shahjahan, M.; Halim, N.A.B.A.; NJ, N.I. Antimicrobial Properties of ’Kelulut’ (Trigona spp.) Honey. Malays. J. Med. Sci. 2007, 14, 107–108. [Google Scholar]
- Al-kafaween, M.A.; Hilmi, A.B.M.; Al-Jamal, H.A.N.; Al-Groom, R.M.; Elsahoryi, N.A.; Al-Sayyed, H. Potential Antibacterial Activity of Yemeni Sidr Honey Against Pseudomonas aeruginosa and Streptococcus pyogenes. Anti-Infect. Agents 2021, 19, 51–65. [Google Scholar] [CrossRef]
- Othman, A.S. Original Research Article Antibacterial Activity of Bee and Yemeni Sidr Honey Against Some Pathogenic Bacterial Species. Int. J. Curr. Microbiol. App. Sci 2014, 3, 1015–1025. [Google Scholar]
- Ghramh, H.A.; Ibrahim, E.H.; Kilany, M. Study of anticancer, antimicrobial, immunomodulatory, and silver nanoparticles production by Sidr honey from three different sources. Food Sci. Nutr. 2020, 8, 445–455. [Google Scholar] [CrossRef]
- Owayss, A.A.; Elbanna, K.; Iqbal, J.; Abulreesh, H.H.; Organji, S.R.; Raweh, H.S.; Alqarni, A.S. In vitro antimicrobial activities of Saudi honeys originating from Ziziphus spina-christi L. and Acacia gerrardii Benth. trees. Food Sci. Nutr. 2020, 8, 390–401. [Google Scholar] [CrossRef]
- Othman, A.S. Determination of the antibacterial effect of some natural products against some gram-positive and gram-negative bacteria. Egypt. Pharm. J. 2016, 15, 10. [Google Scholar] [CrossRef]
- Enany, M.E.; Algammal, A.M.; Shagar, G.I.; Hanora, A.M.; Elfeil, W.K.; Elshaffy, N.M. Molecular typing and evaluation of Sidr honey inhibitory effect on virulence genes of MRSA strains isolated from catfish in Egypt. Pak. J. Pharm. Sci. 2018, 31, 1865–1870. [Google Scholar] [PubMed]
- Noori, A.; Al Ghamdi, A.; Ansari, M.J.; Al-Attal, Y.; Al-Mubarak, A.; Salom, K. Differences in composition of honey samples and their impact on the antimicrobial activities against drug multiresistant bacteria and pathogenic fungi. Arch. Med. Res. 2013, 44, 307–316. [Google Scholar]
- Bazaid, A.S.; Aldarhami, A.; Gattan, H.; Aljuhani, B. Saudi Honey: A Promising Therapeutic Agent for Treating Wound Infections. Cureus 2021, 13, e18882. [Google Scholar] [CrossRef]
- Al-Sayaghi, A.M.; Al-Kabsi, A.M.; Abduh, M.S.; Saghir, S.A.M.; Alshawsh, M.A. Antibacterial Mechanism of Action of Two Types of Honey against Escherichia coli through Interfering with Bacterial Membrane Permeability, Inhibiting Proteins, and Inducing Bacterial DNA Damage. Antibiotics 2022, 11, 1182. [Google Scholar]
- Miguel, M.; Antunes, M.D.; Faleiro, M.L. Honey as a complementary medicine. Integr. Med. Insights 2017, 12, 1178633717702869. [Google Scholar] [CrossRef]
- Ahmed, S.; Othman, N.H. The anti-cancer effects of Tualang honey in modulating breast carcinogenesis: An experimental animal study. BMC Complement. Altern. Med. 2017, 17, 208. [Google Scholar]
- Afrin, S.; Giampieri, F.; Gasparrini, M.; Forbes-Hernández, T.Y.; Cianciosi, D.; Reboredo-Rodriguez, P.; Amici, A.; Quiles, J.L.; Battino, M. The inhibitory effect of Manuka honey on human colon cancer HCT-116 and LoVo cell growth. Part 1: The suppression of cell proliferation, promotion of apoptosis and arrest of the cell cycle. Food Funct. 2018, 9, 2145–2157. [Google Scholar]
- Fernandez-Cabezudo, M.J.; El-Kharrag, R.; Torab, F.; Bashir, G.; George, J.A.; El-Taji, H.; Al-Ramadi, B.K. Intravenous administration of manuka honey inhibits tumor growth and improves host survival when used in combination with chemotherapy in a melanoma mouse model. PLoS ONE 2013, 8, e55993. [Google Scholar]
- Portokalakis, I.; Yusof, H.M.; Ghanotakis, D.; Nigam, P.S.; Owusu-Apenten, R. Manuka honey-induced cytotoxicity against MCF7 breast cancer cells is correlated to total phenol content and antioxidant power. J. Adv. Biol. Biotechnol. 2016, 8, 1–10. [Google Scholar] [CrossRef]
- Kadir, E.A.; Sulaiman, S.A.; Yahya, N.K.; Othman, N.H. Inhibitory effects of tualang honey on experimental breast cancer in rats: A preliminary study. Asian Pac. J. Cancer Prev. 2013, 14, 2249–2254. [Google Scholar]
- Ahmed, S.; Sulaiman, S.A.; Othman, N.H. Oral administration of Tualang and Manuka honeys modulates breast cancer progression in Sprague-Dawley rats model. Evid. Based Complement. Altern. Med. 2017, 2017, 5904361. [Google Scholar] [CrossRef] [PubMed]
- Fauzi, A.N.; Norazmi, M.N.; Yaacob, N.S. Tualang honey induces apoptosis and disrupts the mitochondrial membrane potential of human breast and cervical cancer cell lines. Food Chem. Toxicol. 2011, 49, 871–878. [Google Scholar] [CrossRef] [PubMed]
- Yaacob, N.S.; Nengsih, A.; Norazmi, M. Tualang honey promotes apoptotic cell death induced by tamoxifen in breast cancer cell lines. Evid. Based Complement. Altern. Med. 2013, 2013, 5904361. [Google Scholar] [CrossRef]
- Yaacob, N.S.; Ismail, N.F. Comparison of cytotoxicity and genotoxicity of 4-hydroxytamoxifen in combination with Tualang honey in MCF-7 and MCF-10A cells. BMC Complement. Altern. Med. 2014, 14, 106. [Google Scholar] [CrossRef]
- Hizan, N.S.; Hassan, N.H.M.; Haron, J.; Abubakar, M.B.; Mahdi, N.M.N.; Gan, S.H. Tualang honey adjunct with anastrozole improve parenchyma enhancement of breast tissue in breast cancer patients: A randomized controlled trial. Integr. Med. Res. 2018, 7, 322–327. [Google Scholar] [CrossRef]
- Al-Koshab, M.; Alabsi, A.M.; Bakri, M.M.; Naicker, M.S.; Seyedan, A. Chemopreventive activity of Tualang honey against oral squamous cell carcinoma—In vivo. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2020, 129, 484–492. [Google Scholar] [CrossRef]
- Ghashm, A.A.; Othman, N.H.; Khattak, M.N.; Ismail, N.M.; Saini, R. Antiproliferative effect of Tualang honey on oral squamous cell carcinoma and osteosarcoma cell lines. BMC Complement. Altern. Med. 2010, 10, 49. [Google Scholar] [CrossRef]
- Nik Man, N.M.K.; Hassan, R.; Ang, C.Y.; Abdullah, A.D.; Mohd Radzi, M.A.R.; Sulaiman, S.A. Antileukemic effect of tualang honey on acute and chronic leukemia cell lines. BioMed Res. Int. 2015, 2015, 307094. [Google Scholar] [CrossRef]
- Ahmad, I.; Jimenez, H.; Yaacob, N.S.; Yusuf, N. Tualang honey protects keratinocytes from ultraviolet radiation-induced inflammation and DNA damage. Photochem. Photobiol. 2012, 88, 1198–1204. [Google Scholar] [CrossRef]
- Ramasamy, V.; Abdullah, B.; Singh, A. Effects of Tualang Honey on Cancer Related Fatigue: A Multicenter Open-label Trial of H&N Cancer Patients. Gulf J. Oncol. 2019, 1, 43–51. [Google Scholar]
- Jamil, M.M.A.; Abd Rahman, N.A.; Adon, M.N.; Ambar, R.; Saidin, S. Local Honey (Tualang & Kelulut) Effect on Breast Cancer Cell (MCF-7). In Proceedings of the 2022 IEEE 12th International Conference on Control System, Computing and Engineering (ICCSCE), Penang, Malaysia, 21–22 October 2022; pp. 209–213. [Google Scholar]
- Yun, C.Z.; Nor, N.H.M.; Berahim, Z.; Ponnuraj, K.T. Cytotoxic Evaluation of Malaysian Kelulut Honey on Human Gingival Fibroblast Cell Line using MTT Assay. J. Dent. Indones. 2021, 28, 88–93. [Google Scholar]
- Salim, S.N.M.; Ramakreshnan, L.; Fong, C.S.; Wahab, R.A.; Rasad, M.S.B.A. In-vitro cytotoxicity of Trigona itama honey against human lung adenocarcinoma epithelial cell line (A549). Eur. J. Integr. Med. 2019, 30, 100955. [Google Scholar] [CrossRef]
- Almeer, R.; Alqarni, A.; Alqattan, S.; Abdi, S.; Alarifi, S.; Hassan, Z.; Semlali, A. Effect of honey in improving breast cancer treatment and gene expression modulation of MMPs and TIMPs in Triple-Negative Breast Cancer Cells. Pak. J. Zool. 2018, 50, 1999–2007. [Google Scholar] [CrossRef]
- Kamaratos, A.V.; Tzirogiannis, K.N.; Iraklianou, S.A.; Panoutsopoulos, G.I.; Kanellos, I.E.; Melidonis, A.I. Manuka honey-impregnated dressings in the treatment of neuropathic diabetic foot ulcers. Int. Wound J. 2014, 11, 259–263. [Google Scholar] [CrossRef]
- Almnayan, D. Immune-Modulatory Effects of Sidr Honey: Implications for Anti-Proliferative Effects on Cancer Cells. Ph.D. Thesis, Laurentian University of Sudbury, Sudbury, ON, Canada, 2020. [Google Scholar]
- Bogdanov, S.; Jurendic, T.; Sieber, R.; Gallmann, P. Honey for nutrition and health: A review. J. Am. Coll. Nutr. 2008, 27, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Jaganathan, S.K.; Mandal, M. Antiproliferative effects of honey and of its polyphenols: A review. J. Biomed. Biotechnol. 2009, 2009, 830616. [Google Scholar] [CrossRef]
- Patel, S.; Cichello, S. Manuka honey: An emerging natural food with medicinal use. Nat. Prod. Bioprospect. 2013, 3, 121–128. [Google Scholar] [CrossRef]
- Rohlfing, C.L.; Wiedmeyer, H.-M.; Little, R.R.; England, J.D.; Tennill, A.; Goldstein, D.E. Defining the relationship between plasma glucose and HbA1c: Analysis of glucose profiles and HbA1c in the Diabetes Control and Complications Trial. Diabetes Care 2002, 25, 275–278. [Google Scholar] [CrossRef]
- Al Saeed, M. Therapeutic efficacy of conventional treatment combined with Manuka honey in the treatment of patients with diabetic foot ulcers: A randomized controlled study. Egypt. J. Hosp. Med. 2013, 53, 1064–1071. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.; Sirajudeen, K.; Salleh, M.; Gurtu, S. Hepatoprotective effect of tualang honey supplementation in streptozotocin-induced diabetic rats. Int. J. Appl. Res. Nat. Prod 2012, 4, 37–41. [Google Scholar]
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S.; Sirajudeen, K.N.S.; Salleh, M.S.M.; Gurtu, S. Glibenclamide or metformin combined with honey improves glycemic control in streptozotocin-induced diabetic rats. Int. J. Biol. Sci. 2011, 7, 244. [Google Scholar] [CrossRef]
- Robert, S.D.; Ismail, A. Two varieties of honey that are available in Malaysia gave intermediate glycemic index values when tested among healthy individuals. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc. Czech Repub. 2009, 153, 145–147. [Google Scholar] [CrossRef]
- Hussain, N.H.N.; Sulaiman, S.A.; Hassan, I.I.; Kadir, A.A.; Nor, N.M.; Ismail, S.B.; Yaacob, L.H.; Zakaria, R.; Shafie, N.S.; Haron, J. Randomized controlled trial on the effects of tualang honey and hormonal replacement therapy (HRT) on cardiovascular risk factors, hormonal profiles and bone density among postmenopausal women: A pilot study. J. Food Res. 2012, 1, 171. [Google Scholar] [CrossRef]
- Ab Wahab, S.Z.; Hussain, N.H.N.; Zakaria, R.; Kadir, A.A.; Mohamed, N.; Tohit, N.M.; Norhayati, M.N.; Hassan, I.I. Long-term effects of honey on cardiovascular parameters and anthropometric measurements of postmenopausal women. Complement. Ther. Med. 2018, 41, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Arshad, N.A.; Lin, T.S.; Yahaya, M.F. Stingless bee honey reduces anxiety and improves memory of the metabolic disease-induced rats. CNS Neurol. Disord. Drug Targets 2020, 19, 115–126. [Google Scholar] [CrossRef]
- Alharbi, H.F.; Algonaiman, R.; Barakat, H. Ameliorative and Antioxidative Potential of Lactobacillus plantarum-Fermented Oat (Avena sativa) and Fermented Oat Supplemented with Sidr Honey against Streptozotocin-Induced Type 2 Diabetes in Rats. Antioxidants 2022, 11, 1122. [Google Scholar] [CrossRef]
- Salla, H.R.; Al Habsi, F.S.; Al Sharji, W.H. A comparative study on the role of Omani honey with various food supplements on diabetes and wound healing. J. King Saud Univ. Sci. 2020, 32, 2122–2128. [Google Scholar] [CrossRef]
- Shambaugh, P.; Worthington, V.; Herbert, J. Differential effects of honey, sucrose, and fructose on blood sugar levels. J. Manip. Physiol. Ther. 1990, 13, 322–325. [Google Scholar]
- Folli, F.; Corradi, D.; Fanti, P.; Davalli, A.; Paez, A.; Giaccari, A.; Perego, C.; Muscogiuri, G. The role of oxidative stress in the pathogenesis of type 2 diabetes mellitus micro-and macrovascular complications: Avenues for a mechanistic-based therapeutic approach. Curr. Diabetes Rev. 2011, 7, 313–324. [Google Scholar] [CrossRef]
- Kim, J.S.; Saengsirisuwan, V.; Sloniger, J.A.; Teachey, M.K.; Henriksen, E.J. Oxidant stress and skeletal muscle glucose transport: Roles of insulin signaling and p38 MAPK. Free. Radic. Biol. Med. 2006, 41, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.S.A.; Salam, S.K.N.; Salleh, M.S.M.; Gurtu, S. Antioxidant protective effect of glibenclamide and metformin in combination with honey in pancreas of streptozotocin-induced diabetic rats. Int. J. Mol. Sci. 2010, 11, 2056–2066. [Google Scholar] [CrossRef] [PubMed]
- Ugusman, A.; Shahrin, S.A.S.; Azizan, N.H.; Pillai, S.B.; Krishnan, K.; Salamt, N.; Aminuddin, A.; Hamid, A.A.; Kumar, J.; Mokhtar, M.H. Role of Honey in Obesity Management: A Systematic Review. Front. Nutr. 2022, 9, 924097. [Google Scholar] [CrossRef]
- Mohd Rafie, A.Z.; Syahir, A.; Wan Ahmad, W.A.N.; Mustafa, M.Z.; Mariatulqabtiah, A.R. Supplementation of stingless bee honey from Heterotrigona itama improves antiobesity parameters in high-fat diet induced obese rat model. Evid. Based Complement. Altern. Med. 2018, 2018, 6371582. [Google Scholar] [CrossRef]
- Samat, S.; Kanyan Enchang, F.; Nor Hussein, F.; Wan Ismail, W.I. Four-week consumption of Malaysian honey reduces excess weight gain and improves obesity-related parameters in high fat diet induced obese rats. Evid. Based Complement. Altern. Med. 2017, 2017, 1342150. [Google Scholar] [CrossRef] [PubMed]
- Ramli, N.Z.; Chin, K.-Y.; Zarkasi, K.A.; Ahmad, F. The beneficial effects of stingless bee honey from Heterotrigona itama against metabolic changes in rats fed with high-carbohydrate and high-fat diet. Int. J. Environ. Res. Public Health 2019, 16, 4987. [Google Scholar] [CrossRef]
- Lee, D.S.; Sinno, S.; Khachemoune, A. Honey and wound healing. Am. J. Clin. Dermatol. 2011, 12, 181–190. [Google Scholar] [CrossRef]
- Zumla, A.; Lulat, A. Honey-a remedy rediscovered. J. R. Soc. Med. 1989, 82, 384–385. [Google Scholar] [CrossRef]
- Al-Waili, N.; Salom, K.; Al-Ghamdi, A.A. Honey for wound healing, ulcers, and burns; data supporting its use in clinical practice. Sci. World J. 2011, 11, 766–787. [Google Scholar] [CrossRef]
- Saikaly, S.K.; Khachemoune, A. Honey and wound healing: An update. Am. J. Clin. Dermatol. 2017, 18, 237–251. [Google Scholar] [CrossRef]
- Jull, A.B.; Cullum, N.; Dumville, J.C.; Westby, M.J.; Deshpande, S.; Walker, N. Honey as a topical treatment for wounds. Cochrane Database Syst. Rev. 2015, 3, 6–19. [Google Scholar] [CrossRef]
- Yaghoobi, R.; Kazerouni, A. Evidence for clinical use of honey in wound healing as an anti-bacterial, anti-inflammatory anti-oxidant and anti-viral agent: A review. Jundishapur J. Nat. Pharm. Prod. 2013, 8, 100. [Google Scholar] [CrossRef] [PubMed]
- Majtán, J.; Kováčová, E.; Bíliková, K.; Šimúth, J. The immunostimulatory effect of the recombinant apalbumin 1–major honeybee royal jelly protein–on TNFα release. Int. Immunopharmacol. 2006, 6, 269–278. [Google Scholar] [CrossRef]
- Falanga, V. Wound healing and its impairment in the diabetic foot. Lancet 2005, 366, 1736–1743. [Google Scholar] [CrossRef] [PubMed]
- Sell, S.A.; Wolfe, P.S.; Spence, A.J.; Rodriguez, I.A.; McCool, J.M.; Petrella, R.L.; Garg, K.; Ericksen, J.J.; Bowlin, G.L. A preliminary study on the potential of manuka honey and platelet-rich plasma in wound healing. Int. J. Biomater. 2012, 2012, 313781. [Google Scholar] [CrossRef]
- Tarawneh, O.; Alwahsh, W.; Abul-Futouh, H.; Al-Samad, L.A.; Hamadneh, L.; Abu Mahfouz, H.; Fadhil Abed, A. Determination of Antimicrobial and Antibiofilm Activity of Combined LVX and AMP Impregnated in p (HEMA) Hydrogel. Appl. Sci. 2021, 11, 8345. [Google Scholar] [CrossRef]
- Al-Bakri, A.G.; Mahmoud, N.N. Photothermal-induced antibacterial activity of gold nanorods loaded into polymeric hydrogel against Pseudomonas aeruginosa biofilm. Molecules 2019, 24, 2661. [Google Scholar] [CrossRef]
- Huwaitat, R.; Coulter, S.M.; Porter, S.L.; Pentlavalli, S.; Laverty, G. Antibacterial and antibiofilm efficacy of synthetic polymyxin-mimetic lipopeptides. Pept. Sci. 2021, 113, e24188. [Google Scholar] [CrossRef]
- Engemann, J.J.; Carmeli, Y.; Cosgrove, S.E.; Fowler, V.G.; Bronstein, M.Z.; Trivette, S.L.; Briggs, J.P.; Sexton, D.J.; Kaye, K.S. Adverse clinical and economic outcomes attributable to methicillin resistance among patients with Staphylococcus aureus surgical site infection. Clin. Infect. Dis. 2003, 36, 592–598. [Google Scholar] [CrossRef]
- Tarawneh, O.; Hamadneh, I.; Huwaitat, R.; Al-Assi, A.R.; El Madani, A. Characterization of chlorhexidine-impregnated cellulose-based hydrogel films intended for the treatment of periodontitis. BioMed Res. Int. 2021, 2021, 9853977. [Google Scholar] [CrossRef]
- Jenkins, R.; Cooper, R. Improving antibiotic activity against wound pathogens with manuka honey in vitro. PLoS ONE 2012, 7, e45600. [Google Scholar] [CrossRef]
- Müller, P.; Alber, D.G.; Turnbull, L.; Schlothauer, R.C.; Carter, D.A.; Whitchurch, C.B.; Harry, E.J. Synergism between Medihoney and rifampicin against methicillin-resistant Staphylococcus aureus (MRSA). PLoS ONE 2013, 8, e57679. [Google Scholar] [CrossRef]
- Sukur, S.M.; Halim, A.S.; Singh, K.K.B. Evaluations of bacterial contaminated full thickness burn wound healing in Sprague Dawley rats Treated with Tualang honey. Indian J. Plast. Surg. 2011, 44, 112–117. [Google Scholar] [CrossRef]
- Khoo, Y.-T.; Halim, A.S.; Singh, K.-K.B.; Mohamad, N.-A. Wound contraction effects and antibacterial properties of Tualang honey on full-thickness burn wounds in rats in comparison to hydrofibre. BMC Complement. Altern. Med. 2010, 10, 48. [Google Scholar] [CrossRef]
- Aznan, M.I.; Khan, O.H.; Unar, A.O.; Tuan Sharif, S.E.; Khan, A.H.; Zakaria, A.D. Effect of Tualang honey on the anastomotic wound healing in large bowel anastomosis in rats-A randomized controlled trial. BMC Complement. Altern. Med. 2015, 16, 28. [Google Scholar] [CrossRef] [PubMed]
- Lazim, N.M.; Abdullah, B.; Salim, R. The effect of Tualang honey in enhancing post tonsillectomy healing process. An open labelled prospective clinical trial. Int. J. Pediatr. Otorhinolaryngol. 2013, 77, 457–461. [Google Scholar] [CrossRef] [PubMed]
- Nordin, A.; Chowdhury, S.R.; Saim, A.B.; Idrus, R.B.H. Effect of kelulut honey on the cellular dynamics of tgfβ-induced epithelial to mesenchymal transition in primary human keratinocytes. Int. J. Environ. Res. Public Health 2020, 17, 3229. [Google Scholar] [CrossRef]
- Yazan, L.S.; Zainal, N.A.; Ali, R.M.; Zali, M.; Shyfiq, M.F.; Sze, O.Y.; Sim, T.Y.; Gopalsamy, B.; Ling, V.F.; Sapuan, S. Antiulcer Properties of Kelulut Honey against Ethanol-Induced Gastric Ulcer. Pertanika J. Sci. Technol. 2018, 26, 121–132. [Google Scholar]
- Hananeh, W.M.; Ismail, Z.B.; Alshehabat, M.A.; Abeeleh, M.A. Effects of Sidr honey on second-intention healing of contaminated full-thickness skin wounds in healthy dogs. J. Vet. Res. 2015, 59, 433–439. [Google Scholar] [CrossRef]
- Hwisa, N.T.; Katakam, P.; Chandu, B.R.; Abadi, E.G.; Shefha, E.M. Comparative in vivo evaluation of three types of honey on topical wound healing activity in rabbits. J. Appl. Pharm. Sci. 2013, 3, 139–143. [Google Scholar]
- Syarifah-Noratiqah, S.-B.; Naina-Mohamed, I.; Zulfarina, M.S.; Qodriyah, H. Natural polyphenols in the treatment of Alzheimer’s disease. Curr. Drug Targets 2018, 19, 927–937. [Google Scholar] [CrossRef]
- Navarro-Hortal, M.D.; Romero-Márquez, J.M.; Muñoz-Ollero, P.; Jiménez-Trigo, V.; Esteban-Muñoz, A.; Tutusaus, K.; Giampieri, F.; Battino, M.; Sánchez-González, C.; Rivas-García, L. Amyloid β-but not Tau-induced neurotoxicity is suppressed by Manuka honey via HSP-16.2 and SKN-1/Nrf2 pathways in an in vivo model of Alzheimer’s disease. Food Funct. 2022, 13, 11185–11199. [Google Scholar] [CrossRef]
- Azman, K.F.; Zakaria, R.; Abdul Aziz, C.B.; Othman, Z. Tualang honey attenuates noise stress-induced memory deficits in aged rats. Oxid. Med. Cell. Longev. 2016, 2016, 1549158. [Google Scholar] [CrossRef]
- Azman, K.F.; Zakaria, R.; AbdAziz, C.; Othman, Z.; Al-Rahbi, B. Tualang honey improves memory performance and decreases depressive-like behavior in rats exposed to loud noise stress. Noise Health 2015, 17, 83. [Google Scholar] [CrossRef]
- Al-Rahbi, B.; Zakaria, R.; Othman, Z.; Hassan, A.; Ahmad, A.H. Enhancement of BDNF concentration and restoration of the hypothalamic-pituitary-adrenal axis accompany reduced depressive-like behaviour in stressed ovariectomised rats treated with either Tualang honey or estrogen. Sci. World J. 2014, 2014, 310821. [Google Scholar] [CrossRef]
- Al-Rahbi, B.; Zakaria, R.; Othman, Z.; Hassan, A.; Ahmad, A.H. Protective effects of Tualang honey against oxidative stress and anxiety-like behaviour in stressed ovariectomized rats. Int. Sch. Res. Not. 2014, 2014, 521065. [Google Scholar] [CrossRef]
- Al-Rahbi, B.; Zakaria, R.; Othman, Z.; Hassan, A.; Ismail, Z.I.M.; Muthuraju, S. Tualang honey supplement improves memory performance and hippocampal morphology in stressed ovariectomized rats. Acta Histochem. 2014, 116, 79–88. [Google Scholar] [CrossRef]
- Mohd Sairazi, N.S.; KNS, S.; Asari, M.A.; Mummedy, S.; Muzaimi, M.; Sulaiman, S.A. Effect of tualang honey against KA-induced oxidative stress and neurodegeneration in the cortex of rats. BMC Complement. Altern. Med. 2017, 17, 31. [Google Scholar] [CrossRef]
- Abd Aziz, C.B.; Suhaimi, S.Q.A.; Hasim, H.; Ahmad, A.H.; Long, I.; Zakaria, R. Effects of Tualang honey in modulating nociceptive responses at the spinal cord in offspring of prenatally stressed rats. J. Integr. Med. 2019, 17, 66–70. [Google Scholar] [CrossRef]
- Khalil, M.; Tanvir, E.; Afroz, R.; Sulaiman, S.A.; Gan, S.H. Cardioprotective effects of tualang honey: Amelioration of cholesterol and cardiac enzymes levels. BioMed Res. Int. 2015, 2015, 286051. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S.; Sirajudeen, K.N.; Salleh, S.; Gurtu, S. Honey supplementation in spontaneously hypertensive rats elicits antihypertensive effect via amelioration of renal oxidative stress. Oxid. Med. Cell. Longev. 2012, 2012, 374037. [Google Scholar] [CrossRef] [PubMed]
- Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.S.A.; Sirajudeen, K.N.; Salleh, M.S.M.; Gurtu, S. Differential responses to blood pressure and oxidative stress in streptozotocin-induced diabetic wistar-kyoto rats and spontaneously hypertensive rats: Effects of antioxidant (Honey) treatment. Int. J. Mol. Sci. 2011, 12, 1888–1907. [Google Scholar] [CrossRef] [PubMed]
- Devasvaran, K.; Tan, J.J.; Ng, C.T.; Fong, L.Y.; Yong, Y.K. Malaysian tualang honey inhibits hydrogen peroxide-induced endothelial hyperpermeability. Oxid. Med. Cell. Longev. 2019, 2019, 1202676. [Google Scholar] [CrossRef]
- Ahmed, A.; Khan, R.A.; Azim, M.K.; Saeed, S.A.; Mesaik, M.A.; Ahmed, S.; Imran, I. Effect of natural honey on human platelets and blood coagulation proteins. Pak. J. Pharm. Sci. 2011, 24, 389–397. [Google Scholar]
- Makedou, K.; Iliadis, S.; Kara, E.; Gogou, M.; Feslikidis, T.; Papageorgiou, G. Honey and its protective role against oxidation of human low density lipoproteins and total serum lipoproteins. Hippokratia 2012, 16, 287. [Google Scholar] [PubMed]
- Hegazi, A.G.; El-Hady, A.; Faten, K. Influence of honey on the suppression of human low density lipoprotein (LDL) peroxidation (in vitro). Evid. Based Complement. Altern. Med. 2009, 6, 113–121. [Google Scholar] [CrossRef]
- Bâcvarov, V. Treatment of chronic bronchitis and bronchial asthma with honey. Ther. Der Ggw. 1970, 109, 260–268. [Google Scholar]
- Kamaruzaman, N.A.; Sulaiman, S.A.; Kaur, G.; Yahaya, B. Inhalation of honey reduces airway inflammation and histopathological changes in a rabbit model of ovalbumin-induced chronic asthma. BMC Complement. Altern. Med. 2014, 14, 176. [Google Scholar] [CrossRef]
- Asha’ari, Z.A.; Ahmad, M.Z.; Din, W.S.J.W.; Hussin, C.M.C.; Leman, I. Ingestion of honey improves the symptoms of allergic rhinitis: Evidence from a randomized placebo-controlled trial in the East coast of Peninsular Malaysia. Ann. Saudi Med. 2013, 33, 469–475. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).