Finding a Balance in the Vaginal Microbiome: How Do We Treat and Prevent the Occurrence of Bacterial Vaginosis?
Abstract
1. Introduction
2. The Development of the Vaginal Microbiome: How Does It Change throughout a Woman’s Life?
3. Epidemiology of Bacterial Vaginosis (BV)
4. Pathogenesis of BV and the Identification of Causative Agents
5. Diagnostics Methods of BV
6. Eradication and Management of BV-Associated Pathogens via Antibiotics
7. Reinstate Balance in the Vaginal Microbiome via Probiotics Supplement
7.1. Outcomes of Probiotics with Antibiotics
| Reference | Sample Size | Treatment | Mode of Treatment | Single/Multi-Strain | Probiotics Strains | Observations |
|---|---|---|---|---|---|---|
| [252] | 190 | 7 days of clindamycin (2 × 300 mg) + probiotics or placebo | Vaginal capsule (1 week) | Single strain (109 CFU) | L. casei var. rhamnosus (Lcr35) |
|
| [253] | 84 | 7 days of oral metronidazole (2 × 500 mg/d) + probiotic or placebo | Vaginal tablet (once a week for 2 months) | Single strain (>40,000 CFU) | L. rhamnosus |
|
| [254] | 49 | 7 days of oral metronidazole (2 × 500 mg/d + probiotic | Vaginal capsule (once a week for 6 months) | Single strain (>40,000 CFU) | L. rhamnosus |
|
| [73] | 78 | 7 days of oral metronidazole (1 g/d) + probiotic or placebo | Vaginal capsule (56 days) | Single strain (109 CFU) | L. crispatus IP 174178 |
|
| [255] | 24 | 5 days of vaginal metronidazole 0.75% gel + probiotic/placebo | Vaginal applicator once daily for 5 days followed by once weekly for 2 weeks | Single strain (109 CFU) | L. crispatus CTV-05 |
|
| [256] | 228 | 5 days of vaginal metronidazole (0.75% gel/d) + probiotic or placebo | Vaginal (powder form; administered using prefilled vaginal applicator) | Single strain (2 × 109 CFU) | L. crispatus CTV-05 |
|
| [260] | 40 | 5 days of Ofloxacin–Ornidazole (200–500 mg) per day + 3 days of co-kimaxazol vaginal peccaries + probiotic/control | Oral capsules (90 days) | Single strain (109 CFU) | B. coagulans Unique IS-2 |
|
| [261] | 64 | Tinidazole (2 g) one dose + probiotic or placebo | Oral capsule (2 capsules for 4 weeks) | Multi-strain (2 strains 109 CFU) | L. rhamnosus GR-1, L. reuteri RC-14 |
|
| [262] | 578 | 7 days of oral metronidazole (2 × 500 mg/d) + probiotic or placebo | Oral capsule (10 days every month for 3 months) | Multi-strain (3 strains-108 CFU) | L. fermentum 57A, L. plantarum 57B, L. gasseri 57C |
|
| [263] | 125 | 7 days of oral metronidazole (2 × 500 mg/d) + probiotic or placebo | Oral capsule (30 days) | Multi-strain (2 strains-109 CFU) | L. rhamnosus GR-1, L. reuteri RC -14 |
|
| [264] | 65 | 10 days of oral metronidazole (2 × 400 mg/d) + probiotic or placebo | Oral capsule (twice daily for 25 weeks) | Multi-strain (2 strains-2 × 109 CFU) | L. rhamnosus GR-1, L. reuteri RC-14 |
|
| [265] | 100 | 7 days of 2% clindamycin cream + probiotic or placebo | vaginal capsule (gelatin; 10 days for 3 cycles) | Multi-strain (2 strains-109 CFU) | L. gasseri, L. rhamnosus (EcoVag®) |
|
| [266] | 187 | 3 days of vaginal clindamycin ovules (Dalacin 100 mg) + probiotic or placebo | Vaginal (tampon) (2 menstruation periods) | Multi-strain (4 strains-106 CFU) | L. fermentum, L. caseivar, L. rhamnosus, and L. gasseri |
|
| [267] | 36 | 7 days of oral metronidazole (2 × 500 mg/d) + twice daily probiotic or placebo | Yogurt (4 weeks) | Multi-strain (4 strains-107 CFU/mL) | L. crispatus, L. gasseri, L. rhamnosus, L. jensenii |
|
| [268] | 68 | 7 days of oral metronidazole (2 × 500 mg/d) + probiotic Ecologic Femi (EF+)/Gynophilus LP | Vaginal capsule | Multi-strain (109 CFU) EF+ (6 strains) Gynophilus LP (2 strains) | EF+ (Bifidobacterium bifidum W28, Lactobacillus acidophilus W70, L. helveticus W74, L. brevis W63, L. plantarum W21, L. salivarus W23) Gynophilus LP (Lcr regenerans, L. rhamnosus 35) |
|
| [269] | 39 | Cefixime + doxycycline + metronidazole) + probiotic or no additional treatment | Vaginal capsule | Multi-strain (2 strains-108 CFU) | L. rhamnosus DSM 14870, L. gasseri DSM 14869 |
|
| [270] | 60 | 5 days of vaginal metronidazole (0.75% gel/d) + probiotic or no additional treatment | Oral capsule/vaginal spray (15 days: 5 days of oral capsule followed by 10 days of oral capsules along with vaginal spray) | Multi-strain (≥2 × 109 CFU) | L. acidophilus, L. rhamnosus GG, B. bifidum and B. longum |
|
| [271] | 95 | Clindamycin ovules (100 mg) ± clotrimazole vaginal tablets (200 mg) for 3 nights + probiotic/placebo | Vaginal capsule (5 days) | Multi-strain (108–1010 CFU) | L. gasseri LN40, L. fermentum LN99, L. casei subsp. rhamnosus LN113 and P. acidilactici LN23 |
|
| Reference | Type of Study | Sample Size | Treatment | Mode of Treatment | Probiotics Strains | Observations |
|---|---|---|---|---|---|---|
| [259] | Observational prospective study | 124 | Probiotic (+systematic antibiotic use for other bacterial infections) | Vaginal capsule (4 months) | L. plantarum P 17630 (108 CFU) | May reduce recurrent BV infection, but findings were not statistically significant |
| [258] | Clinical cohort trial | 121 | Metronidazole/probiotic | Vaginal suppository (30 days) | L. delbrueckii subsp. lactis DM8909 (109 CFU) | Significant cure rate in probiotic group at day 30 (p = 0.013) |
| [257] | Prospective case-control study | 250 | Metronidazole (2 × 500 mg for 7 days) + probiotic/no additional treatment | Vaginal tablet (7 months) | L. rhamnosus BMX 54 (NORMOGIN) (104 CFU) | Significant reduction vaginal pH, symptoms, and rate of recurrence (p < 0.001) for 9 months |
7.2. Outcomes with Probiotics Only
| Reference | Sample Size | Mode of Treatment | Single/Multi-Strain (CFU) | Probiotics Strains | Observations |
|---|---|---|---|---|---|
| [272] | 57 | Vaginal suppository (2× daily for 6 days) | Single strain (108−9 CFU) | L. acidophilus | Improvement in vaginal wet smear |
| [282] | 64 | Yogurt (vaginal application-2 weeks) | Single strain (108 CFU/mL) | L. acidophilus | Significant cure rate after 1 month (p = 0.04) |
| [276] | 544 | Oral capsule (6 weeks) | Multi-strain (2 strains-109 CFU) | L. rhamnosus GR-1, L. reuteri RC-14 | Significant decrease in recurrence (p < 0.001) |
| [275] | 40 | Oral capsule (1 week + 2-week washout period (vice versa)) | Multi-strain (3 sub-strains-109 CFU/dose) | L. crispatus | Significant reduction in Nugent score (p = 0.002) |
| [277] | 310 | Oral yogurt | Multi-strain (4 strains-107 CFU) | L. acidophilus, L. bulgaris, Streptococcus thermophilus, and Bifidobacterium lactis | Significant reduction in vaginal pH (p < 0.0001) |
| [281] | 34 | Vaginal tablets (4 weeks) | Multi-strain (2 strains-400 million live cells/dose) | L. fermentum and L. plantarum | Significant reduction of Nugent score in probiotic group (p < 0.001) in 4th week |
| [280] | 120 | Vaginal capsule (14 days) | Multi-strain (3 strains–8 billion CFU) | L. rhamnosus, L. acidophilus, and Strep. thermophilus | Lower recurrence rates for 11 months after treatment (p < 0.001) |
| [279] | 159 | Vaginal tablet (8 days) | Multi-strain (3 strains-109 CFU) | L. brevis (CD2), L. salivarius subsp. salicinius (FV2), L. plantarum (FV9) | Significant reduction in (IL-1β, IL-6) pro-inflammatory cytokines (p < 0.001) |
| [278] | 39 | Vaginal tablet (1 week) | Multi-strain (3 strains-109 CFU) | L. brevis CD2, L. salivarius subsp. salicinius (FV2), and L. plantarum (FV9) | Significant reduction of symptoms and recurrence (p < 0.05) |
8. Can Prebiotics or Synbiotics Help to Relieve BV as Well?
9. Vaginal Microbiota Transplant: A More Innovative Approach?
10. Conclusions and Future Recommendations
Author Contributions
Funding
Conflicts of Interest
References
- Dominguez-Bello, M.G.; Costello, E.K.; Contreras, M.; Magris, M.; Hidalgo, G.; Fierer, N.; Knight, R. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl. Acad. Sci. USA 2010, 107, 11971–11975. [Google Scholar] [CrossRef]
- Lau, A.W.Y.; Tan, L.T.-H.; Ab Mutalib, N.-S.; Wong, S.H.; Letchumanan, V.; Lee, L.-H. The chemistry of gut microbiome in health and diseases. Prog. Microbes Mol. Biol. 2021, 4, a0000175. [Google Scholar] [CrossRef]
- Lee, L.-H.; Wong, S.H.; Chin, S.-F.; Singh, V.; Ab Mutalib, N.-S. Human Microbiome: Symbiosis to Pathogenesis. Front. Microbiol. 2021, 12, 605783. [Google Scholar] [CrossRef]
- Farage, M.; Maibach, H. Lifetime changes in the vulva and vagina. Arch. Gynecol. Obstet. 2006, 273, 195–202. [Google Scholar] [CrossRef]
- Hickey, R.J.; Zhou, X.; Pierson, J.D.; Ravel, J.; Forney, L.J. Understanding vaginal microbiome complexity from an ecological perspective. Transl. Res. 2012, 160, 267–282. [Google Scholar] [CrossRef] [PubMed]
- Nunn, K.L.; Forney, L.J. Unraveling the Dynamics of the Human Vaginal Microbiome. Yale J. Biol. Med. 2016, 89, 331–337. [Google Scholar]
- Bilardi, J.E.; Walker, S.; Temple-Smith, M.; McNair, R.; Mooney-Somers, J.; Bellhouse, C.; Fairley, C.K.; Chen, M.Y.; Bradshaw, C. The burden of bacterial vaginosis: Women’s experience of the physical, emotional, sexual and social impact of living with recurrent bacterial vaginosis. PLoS ONE 2013, 8, e74378. [Google Scholar] [CrossRef] [PubMed]
- Adolfsson, A.; Hagander, A.; Mahjoubipour, F.; Larsson, P.-G. How vaginal infections impact women’s everyday life: Women’s lived experiences of bacterial vaginosis and recurrent vulvovaginal candidiasis. Adv. Sex. Med. 2017, 7, 1–19. [Google Scholar] [CrossRef]
- Newton, D.C.; McCabe, M.P. A theoretical discussion of the impact of stigma on psychological adjustment to having a sexually transmissible infection. Sex Health 2005, 2, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, N.; Luby, S. Vaginal discharge: Perceptions and health seeking behavior among Nepalese women. J. Pak. Med. Assoc. 2004, 54, 620–624. [Google Scholar]
- Parker, C.E.; Johnson, F.C. The effect of maternal estrogens on the vaginal epithelium of the newborn. Clin. Pediatr. 1963, 2, 374–377. [Google Scholar] [CrossRef]
- Widdowson, E.M. Changes in body proportions and composition during growth. In Scientific Foundations of Pediatrics; Davis, J., Dobbing, J., Eds.; Heinemann: London, UK, 1974; pp. 153–163. [Google Scholar]
- Huffman, J.W. The structure and bacteriology of the premenarchal vagina. Ann. N. Y. Acad. Sci. 1959, 83, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Brownell, A.D.; Shapiro, R.A.; Hammerschlag, M.R. Caution is required when using non-Food and Drug Administration-cleared assays to diagnose sexually transmitted infections in children. J. Pediatr. 2019, 206, 280–282. [Google Scholar] [CrossRef] [PubMed]
- Gerstner, G.; Grünberger, W.; Boschitsch, E.; Rotter, M. Vaginal organisms in prepubertal children with and without vulvovaginitis. Arch. Gynecol. 1982, 231, 247–252. [Google Scholar] [CrossRef]
- Pettit, H.; Hitchcock, C.H. Normal flora of the prepuberal vagina. J. Infect. Dis. 1933, 53, 372–375. [Google Scholar] [CrossRef]
- Hardy, G.C. Vaginal Flora in Children. Am. J. Dis. Child. 1941, 62, 939–954. [Google Scholar] [CrossRef]
- Anderson, R.A.; Schultz, O.T.; Stein, I.F. A bacteriologic study of vulvovaginitis of children. J. Infect. Dis. 1923, 32, 444–455. [Google Scholar] [CrossRef]
- Reichert, J.; Epstein, I.; Jung, R.; Colwell, C.A. Infection of the lower part of the genital tract in girls. Am. J. Dis. Child. 1937, 54, 459–495. [Google Scholar] [CrossRef]
- Hammerschlag, M.R.; Alpert, S.; Rosner, I.; Thurston, P.; Semine, D.; McComb, D.; McCormack, W.M. Microbiology of the vagina in children: Normal and potentially pathogenic organisms. Pediatrics 1978, 62, 57–62. [Google Scholar]
- Hammerschlag, M.R.; Alpert, S.; Onderdonk, A.B.; Thurston, P.; Drude, E.; McCormack, W.M.; Bartlett, J.G. Anaerobic microflora of the vagina in children. Am. J. Obstet. Gynecol. 1978, 131, 853–856. [Google Scholar] [CrossRef]
- Yilmaz, A.E.; Celik, N.; Soylu, G.; Donmez, A.; Yuksel, C. Comparison of clinical and microbiological features of vulvovaginitis in prepubertal and pubertal girls. J. Formos. Med. Assoc. 2012, 111, 392–396. [Google Scholar] [CrossRef]
- Thoma, M.E.; Gray, R.H.; Kiwanuka, N.; Aluma, S.; Wang, M.C.; Sewankambo, N.; Wawer, M.J. Longitudinal changes in vaginal microbiota composition assessed by gram stain among never sexually active pre- and postmenarcheal adolescents in Rakai, Uganda. J. Pediatr. Adolesc. Gynecol. 2011, 24, 42–47. [Google Scholar] [CrossRef]
- Hickey, R.J.; Zhou, X.; Settles, M.L.; Erb, J.; Malone, K.; Hansmann, M.A.; Shew, M.L.; Van Der Pol, B.; Fortenberry, J.D.; Forney, L.J. Vaginal microbiota of adolescent girls prior to the onset of menarche resemble those of reproductive-age women. mBio 2015, 6, e00097-15. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, J.G.; Onderdonk, A.B.; Drude, E.; Goldstein, C.; Anderka, M.; Alpert, S.; McCormack, W.M. Quantitative bacteriology of the vaginal flora. J. Infect. Dis. 1977, 136, 271–277. [Google Scholar] [CrossRef]
- Lindner, J.; Plantema, F.; Hoogkamp-Korstanje, J.A. Quantitative studies of the vaginal flora of healthy women and of obstetric and gynaecological patients. J. Med. Microbiol. 1978, 11, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Vásquez, A.; Jakobsson, T.; Ahrné, S.; Forsum, U.; Molin, G. Vaginal Lactobacillus flora of healthy Swedish women. J. Clin. Microbiol. 2002, 40, 2746–2749. [Google Scholar] [CrossRef]
- Ceccarani, C.; Foschi, C.; Parolin, C.; D’Antuono, A.; Gaspari, V.; Consolandi, C.; Laghi, L.; Camboni, T.; Vitali, B.; Severgnini, M. Diversity of vaginal microbiome and metabolome during genital infections. Sci. Rep. 2019, 9, 14095. [Google Scholar] [CrossRef]
- Zhou, X.; Bent, S.J.; Schneider, M.G.; Davis, C.C.; Islam, M.R.; Forney, L.J. Characterization of vaginal microbial communities in adult healthy women using cultivation-independent methods. Microbiology 2004, 150, 2565–2573. [Google Scholar] [CrossRef]
- Hillier, S.L.; Krohn, M.A.; Rabe, L.K.; Klebanoff, S.J.; Eschenbach, D.A. The normal vaginal flora, H2O2-producing lactobacilli, and bacterial vaginosis in pregnant women. Clin. Infect. Dis. 1993, 16, S273–S281. [Google Scholar] [CrossRef]
- Vallor, A.C.; Antonio, M.A.; Hawes, S.E.; Hillier, S.L. Factors associated with acquisition of, or persistent colonization by, vaginal lactobacilli: Role of hydrogen peroxide production. J. Infect. Dis. 2001, 184, 1431–1436. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Niu, X.; Wang, B.; Na, R.; Xiao, B.; Yang, H. Evaluation of the inhibitory effects of Lactobacillus gasseri and Lactobacillus crispatus on the adhesion of seven common lower genital tract infection-causing pathogens to vaginal epithelial cells. Front. Med. 2020, 7, 284. [Google Scholar] [CrossRef]
- Hibbing, M.E.; Fuqua, C.; Parsek, M.R.; Peterson, S.B. Bacterial competition: Surviving and thriving in the microbial jungle. Nat. Rev. Microbiol. 2010, 8, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Voordouw, M.J.; Hill, J.E. Competition among Gardnerella subgroups from the human vaginal microbiome. Front. Cell. Infect. Microbiol. 2019, 9, 374. [Google Scholar] [CrossRef] [PubMed]
- France, M.T.; Mendes-Soares, H.; Forney, L.J. Genomic comparisons of Lactobacillus crispatus and Lactobacillus iners reveal potential ecological drivers of community composition in the vagina. Appl. Environ. Microbiol. 2016, 82, 7063–7073. [Google Scholar] [CrossRef] [PubMed]
- Ravel, J.; Gajer, P.; Abdo, Z.; Schneider, G.M.; Koenig, S.S.; McCulle, S.L.; Karlebach, S.; Gorle, R.; Russell, J.; Tacket, C.O.; et al. Vaginal microbiome of reproductive-age women. Proc. Natl. Acad. Sci. USA 2011, 108, 4680–4687. [Google Scholar] [CrossRef] [PubMed]
- Gajer, P.; Brotman, R.M.; Bai, G.; Sakamoto, J.; Schütte, U.M.; Zhong, X.; Koenig, S.S.; Fu, L.; Ma, Z.S.; Zhou, X.; et al. Temporal dynamics of the human vaginal microbiota. Sci. Transl. Med. 2012, 4, 132ra152. [Google Scholar] [CrossRef]
- Gupta, S.; Kakkar, V.; Bhushan, I. Crosstalk between vaginal microbiome and female health: A review. Microb. Pathog. 2019, 136, 103696. [Google Scholar] [CrossRef]
- Hyman, R.W.; Fukushima, M.; Diamond, L.; Kumm, J.; Giudice, L.C.; Davis, R.W. Microbes on the human vaginal epithelium. Proc. Natl. Acad. Sci. USA 2005, 102, 7952–7957. [Google Scholar] [CrossRef] [PubMed]
- Borgdorff, H.; van der Veer, C.; van Houdt, R.; Alberts, C.J.; de Vries, H.J.; Bruisten, S.M.; Snijder, M.B.; Prins, M.; Geerlings, S.E.; Schim van der Loeff, M.F.; et al. The association between ethnicity and vaginal microbiota composition in Amsterdam, the Netherlands. PLoS ONE 2017, 12, e0181135. [Google Scholar] [CrossRef]
- Zhou, X.; Hansmann, M.A.; Davis, C.C.; Suzuki, H.; Brown, C.J.; Schutte, U.; Pierson, J.D.; Forney, L.J. The vaginal bacterial communities of Japanese women resemble those of women in other racial groups. FEMS Immunol. Med. Microbiol. 2010, 58, 169–181. [Google Scholar] [CrossRef]
- Marino, L.; Concia, E.; Riccardi, A.; Sciarra, E.; Marone, P.; Bolis, P. Vaginal and endocervical bacterial contamination in IUD users. Clin. Exp. Obstet. Gynecol. 1984, 11, 43–48. [Google Scholar] [PubMed]
- Eschenbach, D.A.; Patton, D.L.; Hooton, T.M.; Meier, A.S.; Stapleton, A.; Aura, J.; Agnew, K. Effects of vaginal intercourse with and without a condom on vaginal flora and vaginal epithelium. J. Infect. Dis. 2001, 183, 913–918. [Google Scholar] [CrossRef]
- Martins, G.G.; Junior, J.E.; Tomaz, T.; de Andrade, C.L.; Campos, W. The risk of genital infections in women using intrauterine device. DST-J bras Doenças Sex Transm 2016, 28, 61–63. [Google Scholar]
- Hardy, L.; Jespers, V.; De Baetselier, I.; Buyze, J.; Mwambarangwe, L.; Musengamana, V.; van de Wijgert, J.; Crucitti, T. Association of vaginal dysbiosis and biofilm with contraceptive vaginal ring biomass in African women. PLoS ONE 2017, 12, e0178324. [Google Scholar] [CrossRef]
- Xiao, B.; Wu, C.; Song, W.; Niu, X.; Qin, N.; Liu, Z.; Xu, Q. Association analysis on recurrence of bacterial vaginosis revealed microbes and clinical variables important for treatment outcome. Front. Cell. Infect. Microbiol. 2019, 9, 189. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Song, N.; Williams, C.J.; Brown, C.J.; Yan, Z.; Xu, C.; Forney, L.J. Effects of low dose estrogen therapy on the vaginal microbiomes of women with atrophic vaginitis. Sci. Rep. 2016, 6, 24380, Erratum in 2016, 6, 34119.. [Google Scholar] [CrossRef]
- Edfeldt, G.; Lajoie, J.; Rohl, M.; Oyugi, J.; Ahlberg, A.; Khalilzadeh-Binicy, B.; Bradley, F.; Mack, M.; Kimani, J.; Omollo, K.; et al. Regular use of depot medroxyprogesterone acetate causes thinning of the superficial lining and apical distribution of HIV target cells in the human ectocervix. J. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Bradley, F.; Birse, K.; Hasselrot, K.; Noel-Romas, L.; Introini, A.; Wefer, H.; Seifert, M.; Engstrand, L.; Tjernlund, A.; Broliden, K.; et al. The vaginal microbiome amplifies sex hormone-associated cyclic changes in cervicovaginal inflammation and epithelial barrier disruption. Am. J. Reprod. Immunol. 2018, 80, e12863. [Google Scholar] [CrossRef]
- Ishida, J.; Matsuoka, T.; Saito-Fujita, T.; Inaba, S.; Kunita, S.; Sugiyama, F.; Yagami, K.; Fukamizu, A. Pregnancy-associated homeostasis and dysregulation: Lessons from genetically modified animal models. J. Biochem. 2011, 150, 5–14. [Google Scholar] [CrossRef]
- Soma-Pillay, P.; Nelson-Piercy, C.; Tolppanen, H.; Mebazaa, A. Physiological changes in pregnancy. Cardiovasc. J. Afr. 2016, 27, 89–94. [Google Scholar] [CrossRef]
- Di Simone, N.; Santamaria Ortiz, A.; Specchia, M.; Tersigni, C.; Villa, P.; Gasbarrini, A.; Scambia, G.; D’Ippolito, S. Recent Insights on the Maternal Microbiota: Impact on Pregnancy Outcomes. Front. Immunol. 2020, 11, 528202. [Google Scholar] [CrossRef] [PubMed]
- Nuriel-Ohayon, M.; Neuman, H.; Koren, O. Microbial changes during pregnancy, birth, and infancy. Front. Microbiol. 2016, 7, 1031. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Herrera, G.G.; Tam, K.K.; Lizarraga, J.S.; Beedle, M.-T.; Winuthayanon, W. Estrogen action in the epithelial cells of the mouse vagina regulates neutrophil infiltration and vaginal tissue integrity. Sci. Rep. 2018, 8, 11247. [Google Scholar] [CrossRef]
- Spear, G.T.; French, A.L.; Gilbert, D.; Zariffard, M.R.; Mirmonsef, P.; Sullivan, T.H.; Spear, W.W.; Landay, A.; Micci, S.; Lee, B.H.; et al. Human alpha-amylase present in lower-genital-tract mucosal fluid processes glycogen to support vaginal colonization by Lactobacillus. J. Infect. Dis. 2014, 210, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Redondo-Lopez, V.; Cook, R.L.; Sobel, J.D. Emerging role of lactobacilli in the control and maintenance of the vaginal bacterial microflora. Rev. Infect. Dis. 1990, 12, 856–872. [Google Scholar] [CrossRef]
- Sobel, J.D. Is there a protective role for vaginal flora? Curr. Infect. Dis. Rep. 1999, 1, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Boris, S.; Barbés, C. Role played by lactobacilli in controlling the population of vaginal pathogens. Microb. Infect. 2000, 2, 543–546. [Google Scholar] [CrossRef]
- Kovachev, S. Vaginal ecosystem. Akusherstvo i Ginekologiia 2011, 50, 41–49. [Google Scholar]
- O’Hanlon, D.E.; Moench, T.R.; Cone, R.A. Vaginal pH and microbicidal lactic acid when lactobacilli dominate the microbiota. PLoS ONE 2013, 8, e80074. [Google Scholar] [CrossRef]
- Romero, R.; Hassan, S.S.; Gajer, P.; Tarca, A.L.; Fadrosh, D.W.; Nikita, L.; Galuppi, M.; Lamont, R.F.; Chaemsaithong, P.; Miranda, J.; et al. The composition and stability of the vaginal microbiota of normal pregnant women is different from that of non-pregnant women. Microbiome 2014, 2, 4, Erratum in 2014, 2, 10. [Google Scholar] [CrossRef]
- Serrano, M.G.; Parikh, H.I.; Brooks, J.P.; Edwards, D.J.; Arodz, T.J.; Edupuganti, L.; Huang, B.; Girerd, P.H.; Bokhari, Y.A.; Bradley, S.P.; et al. Racioethnic diversity in the dynamics of the vaginal microbiome during pregnancy. Nat. Med. 2019, 25, 1001–1011. [Google Scholar] [CrossRef]
- McKinlay, S.M.; Brambilla, D.J.; Posner, J.G. The normal menopause transition. Maturitas 1992, 14, 103–115. [Google Scholar] [CrossRef]
- Luoto, R.; Kaprio, J.; Uutela, A. Age at natural menopause and sociodemographic status in Finland. Am. J. Epidemiol. 1994, 139, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Bian, G.; Zheng, M.; Lu, G.; Chan, W.Y.; Li, W.; Yang, K.; Chen, Z.J.; Du, Y. Fertility factors affect the vaginal microbiome in women of reproductive age. Am. J. Reprod. Immunol. 2020, 83, e13220. [Google Scholar] [CrossRef]
- Mirmonsef, P.; Modur, S.; Burgad, D.; Gilbert, D.; Golub, E.T.; French, A.L.; McCotter, K.; Landay, A.L.; Spear, G.T. An exploratory comparison of vaginal glycogen and Lactobacillus levels in pre-and post-menopausal women. Menopause 2015, 22, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Gliniewicz, K.; Schneider, G.M.; Ridenhour, B.J.; Williams, C.J.; Song, Y.; Farage, M.A.; Miller, K.; Forney, L.J. Comparison of the vaginal microbiomes of premenopausal and postmenopausal women. Front. Microbiol. 2019, 10, 193. [Google Scholar] [CrossRef]
- Burton, J.P.; Reid, G. Evaluation of the bacterial vaginal flora of 20 postmenopausal women by direct (Nugent score) and molecular (polymerase chain reaction and denaturing gradient gel electrophoresis) techniques. J. Infect. Dis. 2002, 186, 1770–1780. [Google Scholar] [CrossRef]
- Shardell, M.; Gravitt, P.E.; Burke, A.E.; Ravel, J.; Brotman, R.M. Association of vaginal microbiota with signs and symptoms of the genitourinary syndrome of menopause across reproductive stages. J. Gerontol. A Biol. Sci. Med. Sci. 2021. [Google Scholar] [CrossRef] [PubMed]
- Brotman, R.M.; Shardell, M.D.; Gajer, P.; Fadrosh, D.; Chang, K.; Silver, M.I.; Viscidi, R.P.; Burke, A.E.; Ravel, J.; Gravitt, P.E. Association between the vaginal microbiota, menopause status, and signs of vulvovaginal atrophy. Menopause 2014, 21, 450–458. [Google Scholar] [CrossRef]
- Koumans, E.H.; Sternberg, M.; Bruce, C.; McQuillan, G.; Kendrick, J.; Sutton, M.; Markowitz, L.E. The prevalence of bacterial vaginosis in the United States, 2001–2004; associations with symptoms, sexual behaviors, and reproductive health. Sex. Transm. Dis. 2007, 34, 864–869. [Google Scholar] [CrossRef]
- Morris, M.; Nicoll, A.; Simms, I.; Wilson, J.; Catchpole, M. Bacterial vaginosis: A public health review. BJOG 2001, 108, 439–450. [Google Scholar] [CrossRef]
- Bohbot, J.; Daraï, E.; Bretelle, F.; Brami, G.; Daniel, C.; Cardot, J. Efficacy and safety of vaginally administered lyophilized Lactobacillus crispatus IP 174178 in the prevention of bacterial vaginosis recurrence. J. Gynecol. Obstet. Hum. Reprod. 2018, 47, 81–86. [Google Scholar] [CrossRef]
- Peebles, K.; Velloza, J.; Balkus, J.E.; McClelland, R.S.; Barnabas, R.V. High global burden and costs of bacterial vaginosis: A systematic review and meta-analysis. Sex. Transm. Dis. 2019, 46, 304–311. [Google Scholar] [CrossRef]
- Kenyon, C.; Colebunders, R.; Crucitti, T. The global epidemiology of bacterial vaginosis: A systematic review. Am. J. Obstet. Gynecol. 2013, 209, 505–523. [Google Scholar] [CrossRef]
- Allsworth, J.E.; Peipert, J.F. Prevalence of bacterial vaginosis: 2001–2004 national health and nutrition examination survey data. Obstet. Gynecol. 2007, 109, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Rivera, L.; Trenado, M.; Valdez, A.; Gonzalez, C. Prevalence of bacterial vaginitis and vaginosis: Association with clinical and laboratory features, and treatment. Ginecol. Obstet. Mex. 1996, 64, 26–35. [Google Scholar]
- Brooks-Smith-Lowe, K.; Rodrigo, S. Prevalence of bacterial vaginosis in Grenadian women of reproductive age. West Indian Med. J. 2013, 62, 599–603. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Garcia, P.J.; Chavez, S.; Feringa, B.; Chiappe, M.; Li, W.; Jansen, K.U.; Carcamo, C.; Holmes, K.K. Reproductive tract infections in rural women from the highlands, jungle, and coastal regions of Peru. Bull. World Health Organ. 2004, 82, 483–492. [Google Scholar] [PubMed]
- Jones, F.R.; Miller, G.; Gadea, N.; Meza, R.; Leon, S.; Perez, J.; Lescano, A.G.; Pajuelo, J.; Caceres, C.F.; Klausner, J.D.; et al. Prevalence of bacterial vaginosis among young women in low-income populations of coastal Peru. Int. J. STD AIDS 2007, 18, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.; Rogers, P.; Kinghorn, G. Is bacterial vaginosis a sexually transmitted infection? Sex. Transm. Infect. 2001, 77, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Akinbiyi, A.; Watson, R.; Feyi-Waboso, P. Prevalence of Candida albicans and bacterial vaginosis in asymptomatic pregnant women in South Yorkshire, United Kingdom. Arch. Gynecol. Obstet. 2008, 278, 463–466. [Google Scholar] [CrossRef]
- Uma, S.; Balakrishnan, P.; Murugavel, K.G.; Srikrishnan, A.K.; Kumarasamy, N.; Anand, S.; Cecelia, J.A.; Celentano, D.; Mayer, K.H.; Thyagarajan, S.P.; et al. Bacterial vaginosis in women of low socioeconomic status living in slum areas in Chennai, India. Sexual Health 2006, 3, 297–298. [Google Scholar] [CrossRef]
- Bhalla, P.; Chawla, R.; Garg, S.; Singh, M.; Raina, U.; Bhalla, R.; Sodhani, P. Prevalence of bacterial vaginosis among women in Delhi, India. Indian J. Med. Res. 2007, 125, 167–172. [Google Scholar] [PubMed]
- Uma, S.; Balakrishnan, P.; Murugavel, K.G.; Srikrishnan, A.K.; Kumarasamy, N.; Cecelia, J.A.; Anand, S.; Mayer, K.H.; Celentano, D.; Thyagarajan, S.P.; et al. Bacterial vaginosis in female sex workers in Chennai, India. Sexual Health 2005, 2, 261–262. [Google Scholar] [CrossRef] [PubMed]
- Nayak, M.; Purohit, P.; Debta, A.; Sinha, S.; Patel, S.; Patel, O.; Das, P.; Sahoo, S. Prevalence of bacterial vaginosis in postmenopausal women in the state of Odisha, India. Al Ameen J. Med. Sci. 2020, 13, 266–272. [Google Scholar]
- Ranjit, E.; Raghubanshi, B.R.; Maskey, S.; Parajuli, P. Prevalence of bacterial vaginosis and its association with risk factors among nonpregnant women: A hospital based study. Int. J. Microbiol. 2018, 2018, 8349601. [Google Scholar] [CrossRef] [PubMed]
- Mengistie, Z.; Woldeamanuel, Y.; Asrat, D.; Adera, A. Prevalence of bacterial vaginosis among pregnant women attending antenatal care in Tikur Anbessa University Hospital, Addis Ababa, Ethiopia. BMC Res. Notes 2014, 7, 822. [Google Scholar] [CrossRef]
- Bitew, A.; Abebaw, Y.; Bekele, D.; Mihret, A. Prevalence of bacterial vaginosis and associated risk factors among women complaining of genital tract infection. Int. J. Microbiol. 2017, 2017, 4919404. [Google Scholar] [CrossRef]
- Ibrahim, S.; Bukar, M.; Galadima, G.; Audu, B.; Ibrahim, H. Prevalence of bacterial vaginosis in pregnant women in Maiduguri, North-Eastern Nigeria. Niger. J. Clin. Pract. 2014, 17, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Abdullateef, R.M.; Ijaiya, M.A.; Abayomi, F.; Adeniran, A.S.; Idris, H. Bacterial vaginosis: Prevalence and associated risk factors among non-pregnant women of reproductive age attending a Nigerian tertiary hospital. Malawi Med. J. 2017, 29, 290–293. [Google Scholar] [CrossRef]
- Shahgeibi, S.; Seied-Al-Shohadaie, F.; Seied-Al-Shohadaie, A.; Ghaderi, E. Complications of bacterial vaginosis in pregnancy. Pak. J. Med. Sci. Q. 2009, 25, 53–56. [Google Scholar]
- Yzeiraj-Kalemaj, L.; Shpata, V.; Vyshka, G.; Manaj, A. Bacterial vaginosis, educational level of pregnant women, and preterm birth: A case-control study. Int. Sch. Res. 2013, 2013, 980537. [Google Scholar] [CrossRef]
- Cauci, S.; Driussi, S.; De Santo, D.; Penacchioni, P.; Iannicelli, T.; Lanzafame, P.; De Seta, F.; Quadrifoglio, F.; de Aloysio, D.; Guaschino, S. Prevalence of bacterial vaginosis and vaginal flora changes in peri- and postmenopausal women. J. Clin. Microbiol. 2002, 40, 2147–2152. [Google Scholar] [CrossRef]
- Torrone, E.A.; Morrison, C.S.; Chen, P.-L.; Kwok, C.; Francis, S.C.; Hayes, R.J.; Looker, K.J.; McCormack, S.; McGrath, N.; van de Wijgert, J.H.; et al. Prevalence of sexually transmitted infections and bacterial vaginosis among women in sub-Saharan Africa: An individual participant data meta-analysis of 18 HIV prevention studies. PLoS Med. 2018, 15, e1002511. [Google Scholar] [CrossRef]
- Shimaoka, M.; Yo, Y.; Doh, K.; Kotani, Y.; Suzuki, A.; Tsuji, I.; Mandai, M.; Matsumura, N. Association between preterm delivery and bacterial vaginosis with or without treatment. Sci. Rep. 2019, 9, 509. [Google Scholar] [CrossRef]
- Guaschino, S.; De Seta, F.; Piccoli, M.; Maso, G.; Alberico, S. Aetiology of preterm labour: Bacterial vaginosis. BJOG 2006, 113, 46–51. [Google Scholar] [CrossRef]
- Manns-James, L. Bacterial vaginosis and preterm birth. J. Midwifery Women’s Health 2011, 56, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Klebanoff, M.A.; Brotman, R.M. Treatment of bacterial vaginosis to prevent preterm birth. Lancet 2018, 392, 2141–2142. [Google Scholar] [CrossRef]
- Subtil, D.; Brabant, G.; Tilloy, E.; Devos, P.; Canis, F.; Fruchart, A.; Bissinger, M.-C.; Dugimont, J.-C.; Nolf, C.; Hacot, C.; et al. Early clindamycin for bacterial vaginosis in pregnancy (PREMEVA): A multicentre, double-blind, randomised controlled trial. Lancet 2018, 392, 2171–2179. [Google Scholar] [CrossRef]
- Mark, K.S.; Tenorio, B.; Stennett, C.A.; Ghanem, K.G.; Brotman, R.M. Bacterial vaginosis diagnosis and treatment in postmenopausal women: A survey of clinician practices. Menopause 2020, 27, 679–683. [Google Scholar] [CrossRef]
- Hoffmann, J.N.; You, H.M.; Hedberg, E.C.; Jordan, J.A.; McClintock, M.K. Prevalence of bacterial vaginosis and Candida among postmenopausal women in the United States. J. Gerontol. B Psychol. Sci. Soc. Sci. 2014, 69 (Suppl. 2), S205–S214. [Google Scholar] [CrossRef]
- Onderdonk, A.B.; Delaney, M.L.; Fichorova, R.N. The human microbiome during bacterial vaginosis. Clin. Microbiol. Rev. 2016, 29, 223–238. [Google Scholar] [CrossRef] [PubMed]
- Basavaprabhu, H.; Sonu, K.; Prabha, R. Mechanistic insights into the action of probiotics against bacterial vaginosis and its mediated preterm birth: An overview. Microb. Pathog. 2020, 141, 104029. [Google Scholar] [CrossRef]
- Kumar, N.; Behera, B.; Sagiri, S.S.; Pal, K.; Ray, S.S.; Roy, S. Bacterial vaginosis: Etiology and modalities of treatment—A brief note. J. Pharm. Bioallied Sci. 2011, 3, 496–503. [Google Scholar] [CrossRef]
- Jung, H.-S.; Ehlers, M.M.; Lombaard, H.; Redelinghuys, M.J.; Kock, M.M. Etiology of bacterial vaginosis and polymicrobial biofilm formation. Crit. Rev. Microbiol. 2017, 43, 651–667. [Google Scholar] [CrossRef] [PubMed]
- Eschenbach, D.A.; Davick, P.R.; Williams, B.L.; Klebanoff, S.J.; Young-Smith, K.; Critchlow, C.M.; Holmes, K.K. Prevalence of hydrogen peroxide-producing Lactobacillus species in normal women and women with bacterial vaginosis. J. Clin. Microbiol. 1989, 27, 251–256. [Google Scholar] [CrossRef]
- Aleshkin, V.A.; Voropaeva, E.A.; Shenderov, B.A. Vaginal microbiota in healthy women and patients with bacterial vaginosis and nonspecific vaginitis. Microb. Ecol. Health Dis. 2006, 18, 71–74. [Google Scholar] [CrossRef][Green Version]
- Cook, R.L.; Redondo-Lopez, V.; Schmitt, C.; Meriwether, C.; Sobel, J.D. Clinical, microbiological, and biochemical factors in recurrent bacterial vaginosis. J. Clin. Microbiol. 1992, 30, 870–877. [Google Scholar] [CrossRef]
- Houpikian, P.; Raoult, D. Traditional and molecular techniques for the study of emerging bacterial diseases: One laboratory’s perspective. Emerg. Infect. Dis. 2002, 8, 122–131. [Google Scholar] [CrossRef]
- Kalra, A.; Palcu, C.T.; Sobel, J.D.; Akins, R. Bacterial vaginosis: Culture-and PCR-based characterizations of a complex polymicrobial disease’s pathobiology. Curr. Infect. Dis. Rep. 2007, 9, 485–500. [Google Scholar] [CrossRef] [PubMed]
- Tamrakar, R.; Yamada, T.; Furuta, I.; Cho, K.; Morikawa, M.; Yamada, H.; Sakuragi, N.; Minakami, H. Association between Lactobacillus species and bacterial vaginosis-related bacteria, and bacterial vaginosis scores in pregnant Japanese women. BMC Infect. Dis. 2007, 7, 128. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, P.K.; Agrawal, S.; Shrivastava, R. Modern molecular approaches for analyzing microbial diversity from mushroom compost ecosystem. 3 Biotech 2015, 5, 853–866. [Google Scholar] [CrossRef] [PubMed]
- Younus, N.K.; Gopinath, R.; Jegasothy, R.; Nordin, S.A.; van Belkum, A.; Mary, N.; Neela, V.K. An update on Gardneralla vaginalis associated bacterial vaginosis in Malaysia. Asian Pac. J. Trop. Biomed. 2017, 7, 831–835. [Google Scholar] [CrossRef]
- Lamont, R.F.; Sobel, J.D.; Akins, R.A.; Hassan, S.S.; Chaiworapongsa, T.; Kusanovic, J.P.; Romero, R. The vaginal microbiome: New information about genital tract flora using molecular based techniques. BJOG 2011, 118, 533–549. [Google Scholar] [CrossRef]
- Van De Wijgert, J.H.; Borgdorff, H.; Verhelst, R.; Crucitti, T.; Francis, S.; Verstraelen, H.; Jespers, V. The vaginal microbiota: What have we learned after a decade of molecular characterization? PLoS ONE 2014, 9, e105998. [Google Scholar] [CrossRef]
- Leopold, S. Heretofore undescribed organism isolated from the genitourinary system. U. S. Armed Forces Med. J. 1953, 4, 263–266. [Google Scholar]
- Gardner, H.; Dukes, C. Haemophilus vaginalis vaginitis: A newly defined specific infection previously classified non-specific vaginitis. Am. J. Obstet. Gynecol. 1955, 69, 962–976. [Google Scholar] [CrossRef]
- Dukes, C.D.; Gardner, H.L. Identification of Haemophilus vaginalis. J. Bacteriol. 1961, 81, 277–283. [Google Scholar] [CrossRef]
- Janulaitiene, M.; Paliulyte, V.; Grinceviciene, S.; Zakareviciene, J.; Vladisauskiene, A.; Marcinkute, A.; Pleckaityte, M. Prevalence and distribution of Gardnerella vaginalis subgroups in women with and without bacterial vaginosis. BMC Infect. Dis. 2017, 17, 394. [Google Scholar] [CrossRef]
- Patterson, J.L.; Stull-Lane, A.; Girerd, P.H.; Jefferson, K.K. Analysis of adherence, biofilm formation and cytotoxicity suggests a greater virulence potential of Gardnerella vaginalis relative to other bacterial-vaginosis-associated anaerobes. Microbiology 2010, 156, 392–399. [Google Scholar] [CrossRef]
- Swidsinski, A.; Mendling, W.; Loening-Baucke, V.; Swidsinski, S.; Dörffel, Y.; Scholze, J.; Lochs, H.; Verstraelen, H. An adherent Gardnerella vaginalis biofilm persists on the vaginal epithelium after standard therapy with oral metronidazole. Am. J. Obstet. Gynecol. 2008, 198, 97.e1–97.e6. [Google Scholar] [CrossRef] [PubMed]
- Lewis, W.G.; Robinson, L.S.; Gilbert, N.M.; Perry, J.C.; Lewis, A.L. Degradation, foraging, and depletion of mucus sialoglycans by the vagina-adapted Actinobacterium Gardnerella vaginalis. J. Biol. Chem. 2013, 288, 12067–12079. [Google Scholar] [CrossRef]
- Machado, A.; Jefferson, K.K.; Cerca, N. Interactions between Lactobacillus crispatus and bacterial vaginosis (BV)-associated bacterial species in initial attachment and biofilm formation. Int. J. Mol. Sci. 2013, 14, 12004–12012. [Google Scholar] [CrossRef] [PubMed]
- Verstraelen, H.; Swidsinski, A. The biofilm in bacterial vaginosis: Implications for epidemiology, diagnosis and treatment. Curr. Opin. Infect. Dis. 2013, 26, 86–89. [Google Scholar] [CrossRef]
- Castro, J.; Machado, D.; Cerca, N. Unveiling the role of Gardnerella vaginalis in polymicrobial bacterial vaginosis biofilms: The impact of other vaginal pathogens living as neighbors. ISME J. 2019, 13, 1306–1317. [Google Scholar] [CrossRef] [PubMed]
- Morrill, S.; Gilbert, N.M.; Lewis, A.L. Gardnerella vaginalis as a Cause of Bacterial Vaginosis: Appraisal of the Evidence From in vivo Models. Front. Cell. Infect. Microbiol. 2020, 10, 168. [Google Scholar] [CrossRef]
- Schwebke, J.R.; Lawing, L.F. Prevalence of Mobiluncus spp among women with and without bacterial vaginosis as detected by polymerase chain reaction. Sex. Transm. Dis. 2001, 28, 195–199. [Google Scholar] [CrossRef]
- Schwebke, J.R.; Muzny, C.A.; Josey, W.E. Role of Gardnerella vaginalis in the pathogenesis of bacterial vaginosis: A conceptual model. J. Infect. Dis. 2014, 210, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Muzny, C.A.; Łaniewski, P.; Schwebke, J.R.; Herbst-Kralovetz, M.M. Host-vaginal microbiota interactions in the pathogenesis of bacterial vaginosis. Curr. Opin. Infect. Dis. 2020, 33, 59–65. [Google Scholar] [CrossRef]
- Holm, J.B.; France, M.T.; Ma, B.; McComb, E.; Robinson, C.K.; Mehta, A.; Tallon, L.J.; Brotman, R.M.; Ravel, J. Comparative metagenome-assembled genome analysis of “Candidatus Lachnocurva vaginae”, formerly known as Bacterial Vaginosis-Associated Bacterium-1 (BVAB1). Front. Cell. Infect. Microbiol. 2020, 10, 117. [Google Scholar] [CrossRef] [PubMed]
- Austin, M.N.; Rabe, L.K.; Srinivasan, S.; Fredricks, D.N.; Wiesenfeld, H.C.; Hillier, S.L. Mageeibacillus indolicus gen. nov., sp. nov.: A novel bacterium isolated from the female genital tract. Anaerobe 2015, 32, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Pybus, V.; Onderdonk, A.B. Evidence for a commensal, symbiotic relationship between Gardnerella vaginalis and Prevotella bivia involving ammonia: Potential significance for bacterial vaginosis. J. Infect. Dis. 1997, 175, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, S.; Hoffman, N.G.; Morgan, M.T.; Matsen, F.A.; Fiedler, T.L.; Hall, R.W.; Ross, F.J.; McCoy, C.O.; Bumgarner, R.; Marrazzo, J.M.; et al. Bacterial communities in women with bacterial vaginosis: High resolution phylogenetic analyses reveal relationships of microbiota to clinical criteria. PLoS ONE 2012, 7, e37818. [Google Scholar] [CrossRef]
- Machado, A.; Cerca, N. Influence of biofilm formation by Gardnerella vaginalis and other anaerobes on bacterial vaginosis. J. Infect. Dis. 2015, 212, 1856–1861. [Google Scholar] [CrossRef]
- Pino, A.; Giunta, G.; Randazzo, C.L.; Caruso, S.; Caggia, C.; Cianci, A. Bacterial biota of women with bacterial vaginosis treated with lactoferrin: An open prospective randomized trial. Microb. Ecol. Health Dis. 2017, 28, 1357417. [Google Scholar] [CrossRef]
- Gilbert, N.M.; Lewis, W.G.; Li, G.; Sojka, D.K.; Lubin, J.B.; Lewis, A.L. Gardnerella vaginalis and Prevotella bivia trigger distinct and overlapping phenotypes in a mouse model of bacterial vaginosis. J. Infect. Dis. 2019, 220, 1099–1108. [Google Scholar] [CrossRef]
- Doust, R.H.; Mobarez, A.M. Collagenase activity in Prevotella Bivius isolated from patients with premature rupture of membranes. Med. J. Islam. Repub. Iran 2004, 18, 61–66. [Google Scholar]
- Africa, C.W.; Nel, J.; Stemmet, M. Anaerobes and bacterial vaginosis in pregnancy: Virulence factors contributing to vaginal colonisation. Int. J. Environ. Res. Public Health 2014, 11, 6979–7000. [Google Scholar] [CrossRef]
- Pybus, V.; Onderdonk, A.B. A commensal symbiosis between Prevotella bivia and Peptostreptococcus anaerobius involves amino acids: Potential significance to the pathogenesis of bacterial vaginosis. FEMS Immunol. Med. Microbiol. 1998, 22, 317–327. [Google Scholar] [CrossRef]
- Machado, A.; Salgueiro, D.; Rodrigues, L.; Cerca, N.; Jefferson, K.K. Social interaction by BV anaerobes in initial adhesion and biofilm assays. In Proceedings of the Biofilms 5—International Conference, Paris, France, 10–12 December 2012; pp. 87–88. [Google Scholar]
- Jovita, M.R.; Collins, M.D.; Sjödén, B.; Falsen, E. Characterization of a novel Atopobium isolate from the human vagina: Description of Atopobium vaginae sp. nov. Int. J. Syst. Evol. Microbiol. 1999, 49, 1573–1576. [Google Scholar] [CrossRef] [PubMed]
- Ferris, M.J.; Masztal, A.; Martin, D.H. Use of species-directed 16S rRNA gene PCR primers for detection of Atopobium vaginae in patients with bacterial vaginosis. J. Clin. Microbiol. 2004, 42, 5892–5894. [Google Scholar] [CrossRef] [PubMed]
- Ferris, M.J.; Masztal, A.; Aldridge, K.E.; Fortenberry, J.D.; Fidel, P.L.; Martin, D.H. Association of Atopobium vaginae, a recently described metronidazole resistant anaerobe, with bacterial vaginosis. BMC Infect. Dis. 2004, 4, 5. [Google Scholar] [CrossRef]
- Menard, J.-P.; Fenollar, F.; Henry, M.; Bretelle, F.; Raoult, D. Molecular quantification of Gardnerella vaginalis and Atopobium vaginae loads to predict bacterial vaginosis. Clin. Infect. Dis. 2008, 47, 33–43. [Google Scholar] [CrossRef]
- Polatti, F. Bacterial vaginosis, Atopobium vaginae and nifuratel. Curr. Clin. Pharmacol. 2012, 7, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Mendling, W.; Palmeira-de-Oliveira, A.; Biber, S.; Prasauskas, V. An update on the role of Atopobium vaginae in bacterial vaginosis: What to consider when choosing a treatment? A mini review. Arch. Gynecol. Obstet. 2019, 300, 1–6. [Google Scholar] [CrossRef]
- Bunyan, I.A.; Gatea, A.K.; Hameed, A.K. Molecular identification and genotyping of Atopobium vaginae, 16s rRNA gene from bacterial vaginosis miscarriage women in AL-Hillah city. Int. J. Pharm. Qual. Assur. 2020, 11, 124–130. [Google Scholar] [CrossRef]
- Castro, J.; Rosca, A.S.; Cools, P.; Vaneechoutte, M.; Cerca, N. Gardnerella vaginalis enhances Atopobium vaginae viability in an in vitro model. Front. Cell. Infect. Microbiol. 2020, 10, 83. [Google Scholar] [CrossRef]
- Libby, E.K.; Pascal, K.E.; Mordechai, E.; Adelson, M.E.; Trama, J.P. Atopobium vaginae triggers an innate immune response in an in vitro model of bacterial vaginosis. Microb. Infect. 2008, 10, 439–446. [Google Scholar] [CrossRef]
- Hardy, L.; Jespers, V.; Dahchour, N.; Mwambarangwe, L.; Musengamana, V.; Vaneechoutte, M.; Crucitti, T. Unravelling the bacterial vaginosis-associated biofilm: A multiplex Gardnerella vaginalis and Atopobium vaginae fluorescence in situ hybridization assay using peptide nucleic acid probes. PLoS ONE 2015, 10, e0136658. [Google Scholar] [CrossRef]
- Bradshaw, C.S.; Tabrizi, S.; Fairley, C.K.; Morton, A.N.; Rudland, E.; Garland, S.M. The association of Atopobium vaginae and Gardnerella vaginalis with bacterial vaginosis and recurrence after oral metronidazole therapy. J. Infect. Dis. 2006, 194, 828–836. [Google Scholar] [CrossRef] [PubMed]
- Hardy, L.; Jespers, V.; Abdellati, S.; De Baetselier, I.; Mwambarangwe, L.; Musengamana, V.; van de Wijgert, J.; Vaneechoutte, M.; Crucitti, T. A fruitful alliance: The synergy between Atopobium vaginae and Gardnerella vaginalis in bacterial vaginosis-associated biofilm. Sex. Transm. Infect. 2016, 92, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Sobel, J.D. Antibiotic consideration in bacterial vaginosis. Curr. Infect. Dis. Rep. 2009, 11, 471–475. [Google Scholar] [CrossRef] [PubMed]
- De Backer, E.; Verhelst, R.; Verstraelen, H.; Claeys, G.; Verschraegen, G.; Temmerman, M.; Vaneechoutte, M. Antibiotic susceptibility of Atopobium vaginae. BMC Infect. Dis. 2006, 6, 51. [Google Scholar] [CrossRef]
- Bostwick, D.G.; Woody, J.; Hunt, C.; Budd, W. Antimicrobial resistance genes and modelling of treatment failure in bacterial vaginosis: Clinical study of 289 symptomatic women. J. Med. Microbiol. 2016, 65, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Petrina, M.A.; Cosentino, L.A.; Rabe, L.K.; Hillier, S.L. Susceptibility of bacterial vaginosis (BV)-associated bacteria to secnidazole compared to metronidazole, tinidazole and clindamycin. Anaerobe 2017, 47, 115–119. [Google Scholar] [CrossRef]
- Beigi, R.H.; Austin, M.N.; Meyn, L.A.; Krohn, M.A.; Hillier, S.L. Antimicrobial resistance associated with the treatment of bacterial vaginosis. Am. J. Obstet. Gynecol. 2004, 191, 1124–1129. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.W.; Moore, J.E.; Rao, J.R. Antimicrobial resistance (AMR): Significance to food quality and safety. Food Qual. Saf. 2019, 3, 15–22. [Google Scholar] [CrossRef]
- Tally, F.; Malamy, M. Mechanism of antimicrobial resistance and resistance transfer in anaerobic bacteria. Scand. J. Infect. Dis. Suppl. 1982, 35, 37–44. [Google Scholar]
- Acar, J.; Rostel, B. Antimicrobial resistance: An overview. Revue Scientifique et Technique-Office International des Epizooties 2001, 20, 797–810. [Google Scholar] [CrossRef]
- Tenover, F.C. Mechanisms of antimicrobial resistance in bacteria. Am. J. Med. 2006, 119, S3–S10. [Google Scholar] [CrossRef]
- Sykes, R. The 2009 Garrod lecture: The evolution of antimicrobial resistance: A Darwinian perspective. J. Antimicrob. Chemother. 2010, 65, 1842–1852. [Google Scholar] [CrossRef]
- Holmes, A.H.; Moore, L.S.; Sundsfjord, A.; Steinbakk, M.; Regmi, S.; Karkey, A.; Guerin, P.J.; Piddock, L.J. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet 2016, 387, 176–187. [Google Scholar] [CrossRef]
- Ali, J.; Rafiq, Q.A.; Ratcliffe, E. Antimicrobial resistance mechanisms and potential synthetic treatments. Future Sci. OA 2018, 4, FSO290. [Google Scholar] [CrossRef]
- Abe, K.; Nomura, N.; Suzuki, S. Biofilms: Hot spots of horizontal gene transfer (HGT) in aquatic environments, with a focus on a new HGT mechanism. FEMS Microbiol. Ecol. 2020, 96. [Google Scholar] [CrossRef] [PubMed]
- Castro, J.; Rosca, A.S.; Muzny, C.A.; Cerca, N. Atopobium vaginae and Prevotella bivia are able to incorporate and influence gene expression in a pre-formed Gardnerella vaginalis biofilm. Pathogens 2021, 10, 247. [Google Scholar] [CrossRef] [PubMed]
- Shishpal, P.; Kasarpalkar, N.; Singh, D.; Bhor, V.M. Characterization of Gardnerella vaginalis membrane vesicles reveals a role in inducing cytotoxicity in vaginal epithelial cells. Anaerobe 2020, 61, 102090. [Google Scholar] [CrossRef] [PubMed]
- Hill, G.B. The microbiology of bacterial vaginosis. Am. J. Obstet. Gynecol. 1993, 169, 450–454. [Google Scholar] [CrossRef]
- Majeroni, B.A. Bacterial vaginosis: An update. Am. Fam. Physician 1998, 57, 1285–1289. [Google Scholar]
- Hainer, B.L.; Gibson, M.V. Vaginitis: Diagnosis and treatment. Am. Fam. Physician 2011, 83, 807–815. [Google Scholar]
- Amsel, R.; Totten, P.A.; Spiegel, C.A.; Chen, K.C.; Eschenbach, D.; Holmes, K.K. Nonspecific vaginitis: Diagnostic criteria and microbial and epidemiologic associations. Am. J. Med. 1983, 74, 14–22. [Google Scholar] [CrossRef]
- Spiegel, C.A.; Amsel, R.; Holmes, K. Diagnosis of bacterial vaginosis by direct gram stain of vaginal fluid. J. Clin. Microbiol. 1983, 18, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Money, D. The laboratory diagnosis of bacterial vaginosis. Can. J. Infect. Dis. Med. Microbiol. 2005, 16, 77–79. [Google Scholar] [CrossRef]
- Nugent, R.P.; Krohn, M.A.; Hillier, S.L. Reliability of diagnosing bacterial vaginosis is improved by a standardized method of gram stain interpretation. J. Clin. Microbiol. 1991, 29, 297–301. [Google Scholar] [CrossRef]
- Moussavi, Z.; Behrouzi, R. Diagnostic Amsel criteria compared standardized method of Gram stain for the diagnosis of bacterial vaginosis. Int. Congr. 2004, 1271, 392–395. [Google Scholar] [CrossRef]
- Mohammadzadeh, F.; Dolatian, M.; Jorjani, M.; Majd, H.A. Diagnostic value of Amsel’s clinical criteria for diagnosis of bacterial vaginosis. Glob. J. Health Sci. 2015, 7, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Bhat, G.; Kotigadde, S.; Shenoy, S. Comparison of the methods of diagnosis of bacterial vaginosis. J. Clin. Diagn. Res. 2011, 5, 498–501. [Google Scholar]
- Rafiq, S.; Nauman, N.; Tariq, A.; Jalali, S. Diagnosis of bacterial vaginosis in females with vaginal discharge using Amsel’s clinical criteria and Nugent scoring. J. Rawalpindi Med Coll. 2015, 19, 230–234. [Google Scholar]
- Gutman, R.E.; Peipert, J.F.; Weitzen, S.; Blume, J. Evaluation of clinical methods for diagnosing bacterial vaginosis. Obstet. Gynecol. 2005, 105, 551–556. [Google Scholar] [CrossRef]
- Mittal, V.; Jain, A.; Pradeep, Y. Development of modified diagnostic criteria for bacterial vaginosis at peripheral health centres in developing countries. J. Infect. Dev. Ctries. 2012, 6, 373–377. [Google Scholar] [CrossRef]
- Bansal, R.; Garg, P.; Garg, A. Comparison of Amsel’s criteria and Nugent’s criteria for diagnosis of bacterial vaginosis in tertiary care centre. Int. J. Reprod. Contracept. Obstet. Gynecol. 2019, 8, 637–640. [Google Scholar] [CrossRef]
- Ison, C.; Hay, P. Validation of a simplified grading of Gram stained vaginal smears for use in genitourinary medicine clinics. Sex. Transm. Infect. 2002, 78, 413–415. [Google Scholar] [CrossRef]
- Chawla, R.; Bhalla, P.; Chadha, S.; Grover, S.; Garg, S. Comparison of Hay’s criteria with Nugent’s scoring system for diagnosis of bacterial vaginosis. BioMed Res. Int. 2013, 2013, 365194. [Google Scholar] [CrossRef]
- Larsson, P.-G.; Carlsson, B.; Fåhraeus, L.; Jakobsson, T.; Forsum, U. Diagnosis of bacterial vaginosis: Need for validation of microscopic image area used for scoring bacterial morphotypes. Sex. Transm. Infect. 2004, 80, 63–67. [Google Scholar] [CrossRef]
- Antonucci, F.; Mir, W.; Fontana, C. Comparison between Nugent’s and Hay/Ison scoring criteria for the diagnosis of bacterial vaginosis in WASP prepared vaginal samples. Clin. Investig. 2017, 7, 89–93. [Google Scholar] [CrossRef]
- Shujatullah, F.; Khan, H.M.; Khatoon, R.; Rabbani, T.; Malik, A. An evaluation of OSOM BV blue test in the diagnosis of bacterial vaginosis. Asian Pac. J. Trop. Med. 2010, 3, 574–576. [Google Scholar] [CrossRef]
- Khatoon, R.; Ahmad, S.; Jahan, N. OSOM BV blue test: A new point-of-care test for diagnosing bacterial vaginosis and its comparison with Gram staining. Afr. J. Microbiol. Res. 2013, 7, 4103–4106. [Google Scholar] [CrossRef]
- Coleman, J.S.; Gaydos, C.A. Molecular diagnosis of bacterial vaginosis: An update. J. Clin. Microbiol. 2018, 56, e00342-18. [Google Scholar] [CrossRef] [PubMed]
- Madhivanan, P.; Krupp, K.; Li, T.; Ravi, K.; Selezneva, J.; Srinivas, V.; Arun, A.; Klausner, J.D. Performance of BVBlue rapid test in detecting bacterial vaginosis among women in Mysore, India. Infect. Dis. Obstet. Gynecol. 2014, 2014, 908313. [Google Scholar] [CrossRef] [PubMed]
- Myziuk, L.; Romanowski, B.; Johnson, S.C. BVBlue test for diagnosis of bacterial vaginosis. J. Clin. Microbiol. 2003, 41, 1925–1928. [Google Scholar] [CrossRef]
- West, B.; Morison, L.; Van Der Loeff, M.S.; Gooding, E.; Awasana, A.A.; Demba, E.; Mayaud, P. Evaluation of a new rapid diagnostic kit (FemExam) for bacterial vaginosis in patients with vaginal discharge syndrome in The Gambia. Sex. Transm. Dis. 2003, 30, 483–489. [Google Scholar] [CrossRef]
- Kairu, A.W. Rapid point of care testing for sexually transmitted diseases and bacterial vaginosis: Cost estimation and budget impact analysis. Master’s Thesis, University of Cape Town, Cape Town, South Africa, 2018. [Google Scholar]
- Miller, L. Can Fem Exam card use facilitate bacterial vaginosis diagnosis on day of abortion to prevent postabortion endometritis? Obstet. Gynecol. 2001, 97, S58–S59. [Google Scholar] [CrossRef]
- Theroux, R. Women’s self-diagnostic skills: Developing the science. Nurs. Womens Health 2010, 14, 399–404. [Google Scholar] [CrossRef]
- Lynch, T.; Peirano, G.; Lloyd, T.; Read, R.; Carter, J.; Chu, A.; Shaman, J.A.; Jarvis, J.P.; Diamond, E.; Ijaz, U.Z.; et al. Molecular diagnosis of vaginitis: Comparing quantitative PCR and microbiome profiling approaches to current microscopy scoring. J. Clin. Microbiol. 2019, 57, e00300-19. [Google Scholar] [CrossRef] [PubMed]
- Briselden, A.M.; Hillier, S.L. Evaluation of affirm VP Microbial Identification Test for Gardnerella vaginalis and Trichomonas vaginalis. J. Clin. Microbiol. 1994, 32, 148–152. [Google Scholar] [CrossRef]
- Brown, H.L.; Fuller, D.D.; Jasper, L.T.; Davis, T.E.; Wright, J.D. Clinical evaluation of Affirm VPIII in the detection and identification of Trichomonas vaginalis, Gardnerella vaginalis, and Candida species in vaginitis/vaginosis. Infect. Dis. Obstet. Gynecol. 2004, 12, 934876. [Google Scholar] [CrossRef] [PubMed]
- Byun, S.W.; Park, Y.J.; Hur, S.Y. Affirm VPIII microbial identification test can be used to detect Gardnerella vaginalis, Candida albicans and Trichomonas vaginalis microbial infections in Korean women. J. Obstet. Gynaecol. Res. 2016, 42, 422–426. [Google Scholar] [CrossRef]
- Pavlov, A.R.; Pavlova, N.V.; Kozyavkin, S.A.; Slesarev, A.I. Recent developments in the optimization of thermostable DNA polymerases for efficient applications. Trends Biotechnol. 2004, 22, 253–260. [Google Scholar] [CrossRef]
- Srinivasan, S.; Fredricks, D.N. The human vaginal bacterial biota and bacterial vaginosis. Interdiscip. Perspect. Infect. Dis. 2008, 2008, 750479. [Google Scholar] [CrossRef]
- Balashov, S.V.; Mordechai, E.; Adelson, M.E.; Sobel, J.D.; Gygax, S.E. Multiplex quantitative polymerase chain reaction assay for the identification and quantitation of major vaginal lactobacilli. Diagn. Microbiol. Infect. Dis. 2014, 78, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Hilbert, D.W.; Smith, W.L.; Chadwick, S.G.; Toner, G.; Mordechai, E.; Adelson, M.E.; Aguin, T.J.; Sobel, J.D.; Gygax, S.E. Development and validation of a highly accurate quantitative real-time PCR assay for diagnosis of bacterial vaginosis. J. Clin. Microbiol. 2016, 54, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Ramsuran, V.; Mitchev, N.; Niehaus, A.J.; Han, K.S.S.; Osman, F.; Ngcapu, S.; Karim, S.A.; Rompalo, A.; Garrett, N.; et al. Assessing a diagnosis tool for bacterial vaginosis. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 1481–1485. [Google Scholar] [CrossRef]
- Cartwright, C.P.; Lembke, B.D.; Ramachandran, K.; Body, B.A.; Nye, M.B.; Rivers, C.A.; Schwebke, J.R. Comparison of nucleic acid amplification assays with BD affirm VPIII for diagnosis of vaginitis in symptomatic women. J. Clin. Microbiol. 2013, 51, 3694–3699. [Google Scholar] [CrossRef] [PubMed]
- Schwebke, J.R.; Taylor, S.N.; Ackerman, R.; Schlaberg, R.; Quigley, N.B.; Gaydos, C.A.; Chavoustie, S.E.; Nyirjesy, P.; Remillard, C.V.; Estes, P.; et al. Clinical validation of the Aptima bacterial vaginosis and Aptima Candida/Trichomonas vaginitis assays: Results from a prospective multicenter clinical study. J. Clin. Microbiol. 2020, 58, e01643-19. [Google Scholar] [CrossRef] [PubMed]
- Gaydos, C.A.; Beqaj, S.; Schwebke, J.R.; Lebed, J.; Smith, B.; Davis, T.E.; Fife, K.H.; Nyirjesy, P.; Spurrell, T.; Furgerson, D.; et al. Clinical validation of a test for the diagnosis of vaginitis. Obstet. Gynecol. 2017, 130, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Sherrard, J. Evaluation of the BD MAX™ Vaginal Panel for the detection of vaginal infections in a sexual health service in the UK. Int. J. STD AIDS 2019, 30, 411–414. [Google Scholar] [CrossRef]
- Koenig, M.; Jejeebhoy, S.; Singh, S.; Sridhar, S. Investigating women’s s gynaecological morbidity in India: Not just another KAP survey. Reprod. Health Matters 1998, 6, 84–97. [Google Scholar] [CrossRef]
- Forney, L.J.; Gajer, P.; Williams, C.J.; Schneider, G.M.; Koenig, S.S.; McCulle, S.L.; Karlebach, S.; Brotman, R.M.; Davis, C.C.; Ault, K.; et al. Comparison of self-collected and physician-collected vaginal swabs for microbiome analysis. J. Clin. Microbiol. 2010, 48, 1741–1748. [Google Scholar] [CrossRef]
- Ogale, Y.; Yeh, P.T.; Kennedy, C.E.; Toskin, I.; Narasimhan, M. Self-collection of samples as an additional approach to deliver testing services for sexually transmitted infections: A systematic review and meta-analysis. BMJ Glob. Health 2019, 4, e001349. [Google Scholar] [CrossRef]
- Hammerschlag, M.; Brownell, A.; Shapiro, R. P021 Use of non-FDA cleared assays for detection of STIs in children being evaluated for suspected sexual abuse. Sex. Transm. Infect. 2019, 95, A90. [Google Scholar]
- Davies, J. Where have all the antibiotics gone? Can. J. Infect. Dis. Med. Microbiol. 2006, 17, 707296. [Google Scholar] [CrossRef] [PubMed]
- Organización Mundial de la Salud (Suiza); World Health Organization Staff; Światowa Organizacja Zdrowia; World Health Organization; UNAIDS. Guidelines for the Management of Sexually Transmitted Infections; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Clinical Effectiveness Group; British Association for Sexual Health and HIV. UK National Guideline for the Management of Bacterial Vaginosis 2012. Available online: https://www.guidelinecentral.com/summaries/uk-national-guideline-for-the-management-of-bacterial-vaginosis-2012/#section-society (accessed on 28 January 2021).
- Workowski, K.A. Centers for Disease Control and Prevention Sexually Transmitted Diseases Treatment Guidelines. Clin. Infect. Dis. 2015, 61, S759–S762. [Google Scholar] [CrossRef]
- Australia, S.H.A. Australian STI Management Guidelines for Use in Primary Care. Available online: http://sti.guidelines.org.au/ (accessed on 28 January 2021).
- Sherrard, J.; Wilson, J.; Donders, G.; Mendling, W.; Jensen, J.S. 2018 European (IUSTI/WHO) International Union against sexually transmitted infections (IUSTI) World Health Organisation (WHO) guideline on the management of vaginal discharge. Int. J. STD AIDS 2018, 29, 1258–1272. [Google Scholar] [CrossRef]
- Wang, J. Bacterial vaginosis. Prim. Care Update OB/GYNS 2000, 7, 181–185. [Google Scholar] [CrossRef]
- Kroon, S.J.; Ravel, J.; Huston, W.M. Cervicovaginal microbiota, women’s health, and reproductive outcomes. Fertil. Steril. 2018, 110, 327–336. [Google Scholar] [CrossRef]
- Lossick, J.G. Treatment of sexually transmitted vaginosis/vaginitis. Rev. Infect. Dis. 1990, 12, S665–S681. [Google Scholar] [CrossRef] [PubMed]
- Livengood, C.H.I.; Soper, D.E.; Sheehan, K.L.; Fenner, D.E.; Martens, M.G.; Nelson, A.L.; Ismail, M.; Thorp, J.M.; Lappin, M.; Long, B.J.; et al. Comparison of once-daily and twice-daily dosing of 0.75% metronidazole gel in the treatment of bacterial vaginosis. Sex. Transm. Dis. 1999, 26, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Edwards, D.I. Mechanisms of selective toxicity of metronidazole and other nitroimidazole drugs. Sex. Transm. Infect. 1980, 56, 285–290. [Google Scholar] [CrossRef]
- Müller, M. Mode of action of metronidazole on anaerobic bacteria and protozoa. Surgery 1983, 93, 165–171. [Google Scholar]
- Leyva-Gómez, G.; Del Prado-Audelo, M.L.; Ortega-Peña, S.; Mendoza-Muñoz, N.; Urbán-Morlán, Z.; González-Torres, M.; González-Del Carmen, M.; Figueroa-González, G.; Reyes-Hernández, O.D.; Cortés, H. Modifications in vaginal microbiota and their influence on drug release: Challenges and opportunities. Pharmaceutics 2019, 11, 217. [Google Scholar] [CrossRef]
- Oduyebo, O.O.; Anorlu, R.I.; Ogunsola, F.T. The effects of antimicrobial therapy on bacterial vaginosis in non-pregnant women. Cochrane Database Syst. Rev. 2009, CD006055. [Google Scholar] [CrossRef] [PubMed]
- Kasten, M.J. Clindamycin, metronidazole, and chloramphenicol. Mayo Clin. Proc. 1999, 74, 825–833. [Google Scholar] [CrossRef] [PubMed]
- Spížek, J.; Řezanka, T. Lincomycin, clindamycin and their applications. Appl. Microbiol. Biotechnol. 2004, 64, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.I.; Eady, E.A.; Cove, J.H.; Jones, C.E.; Ratyal, A.H.; Miller, Y.W.; Vyakrnam, S.; Cunliffe, W.J. Clinical resistance to erythromycin and clindamycin in cutaneous propionibacteria isolated from acne patients is associated with mutations in 23S rRNA. Antimicrob. Agents Chemother. 1997, 41, 1162–1165. [Google Scholar] [CrossRef]
- Leclercq, R. Mechanisms of resistance to macrolides and lincosamides: Nature of the resistance elements and their clinical implications. Clin. Infect. Dis. 2002, 34, 482–492. [Google Scholar] [CrossRef] [PubMed]
- Malbruny, B.; Werno, A.M.; Murdoch, D.R.; Leclercq, R.; Cattoir, V. Cross-resistance to lincosamides, streptogramins A, and pleuromutilins due to the lsa (C) gene in Streptococcus agalactiae UCN70. Antimicrob. Agents Chemother. 2011, 55, 1470–1474. [Google Scholar] [CrossRef]
- Sun, L.; Klein, E.Y.; Laxminarayan, R. Seasonality and temporal correlation between community antibiotic use and resistance in the United States. Clin. Infect. Dis. 2012, 55, 687–694. [Google Scholar] [CrossRef]
- Mtsher, A.M.; Aziz, Z.S. Estimation of erythromycin and inducible clindamycin resistance in Saphylococcus aureus Isolated from Clinical Cases. Res. J.Pharm. Technol. 2020, 13, 2920–2924. [Google Scholar] [CrossRef]
- Austin, M.; Beigi, R.; Meyn, L.; Hillier, S. Microbiologic response to treatment of bacterial vaginosis with topical clindamycin or metronidazole. J. Clin. Microbiol. 2005, 43, 4492–4497. [Google Scholar] [CrossRef]
- Boris, J.; Påhlson, C.; Larsson, P.G. Six years observation after successful treatment of bacterial vaginosis. Infect. Dis. Obstet. Gynecol. 1997, 5, 297–302. [Google Scholar] [CrossRef]
- Mattila, J.; Männistö, P.; Mäntylä, R.; Nykänen, S.; Lamminsivu, U. Comparative pharmacokinetics of metronidazole and tinidazole as influenced by administration route. Antimicrob. Agents Chemother. 1983, 23, 721–725. [Google Scholar] [CrossRef]
- Paavonen, J.; Vesterinen, E.; Purola, E.; Jokipii, A.; Jokipii, L.; Holttinen, K.; Vartiainen, E. Single dose of tinidazole in the treatment of vaginal discharge. Scand. J. Urol. Nephrol. Suppl. 1984, 86, 237–240. [Google Scholar] [PubMed]
- Piot, P.; Van Dyck, E.; Godts, P.; Vanderheyden, J. A placebo-controlled, double-blind comparison of tinidazole and triple sulfonamide cream for the treatment of nonspecific vaginitis. Am. J. Obstet. Gynecol. 1983, 147, 85–89. [Google Scholar] [CrossRef]
- van der Meijden, W.I. Treatment of non-specific vaginitis with a single dose of tinidazole. Scand. J. Infect. Dis. Suppl. 1983, 40, 85–89. [Google Scholar] [PubMed]
- Ekgren, J.; Norling, B.K.; Degre, M.; Midtvedt, T. Comparison of tinidazole given as a single dose and on 2 consecutive days for the treatment of nonspecific bacterial vaginosis. Gynecol. Obstet. Invest. 1988, 26, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, N.R.; Wilson, J.D. Tinidazole in the treatment of bacterial vaginosis. Int. J. Women’s Health 2009, 1, 59–65. [Google Scholar] [CrossRef]
- Baylson, F.A.; Nyirjesy, P.; Weitz, M.V. Treatment of recurrent bacterial vaginosis with tinidazole. Obstet. Gynecol. 2004, 104, 931–932. [Google Scholar] [CrossRef]
- Raja, I.M.; Basavareddy, A.; Mukherjee, D.; Meher, B.R. Randomized, double-blind, comparative study of oral metronidazole and tinidazole in treatment of bacterial vaginosis. Indian J. Pharmacol. 2016, 48, 654–658. [Google Scholar] [CrossRef]
- Mendling, W.; Weissenbacher, E.R.; Gerber, S.; Prasauskas, V.; Grob, P. Use of locally delivered dequalinium chloride in the treatment of vaginal infections: A review. Arch. Gynecol. Obstet. 2016, 293, 469–484. [Google Scholar] [CrossRef]
- Weissenbacher, E.R.; Donders, G.; Unzeitig, V.; De Tejada, B.M.; Gerber, S.; Halaška, M.; Špaček, J.; Group, F.S. A comparison of dequalinium chloride vaginal tablets (Fluomizin®) and clindamycin vaginal cream in the treatment of bacterial vaginosis: A single-blind, randomized clinical trial of efficacy and safety. Gynecol. Obstet. Invest. 2012, 73, 8–15. [Google Scholar] [CrossRef]
- Allsworth, J.E.; Lewis, V.A.; Peipert, J.F. Viral sexually transmitted infections and bacterial vaginosis: 2001–2004 National Health and Nutrition Examination Survey data. Sex. Transm. Dis. 2008, 35, 791–796. [Google Scholar] [CrossRef]
- Valenti, P.; Rosa, L.; Capobianco, D.; Lepanto, M.S.; Schiavi, E.; Cutone, A.; Paesano, R.; Mastromarino, P. Role of lactobacilli and lactoferrin in the mucosal cervicovaginal defense. Front. Immunol. 2018, 9, 376. [Google Scholar] [CrossRef]
- Kechagia, M.; Basoulis, D.; Konstantopoulou, S.; Dimitriadi, D.; Gyftopoulou, K.; Skarmoutsou, N.; Fakiri, E.M. Health benefits of probiotics: A review. Int. Sch. Res. Nutri. 2013, 2013, 481651. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.H.; Balakrishnan, K.; Thiagarajah, K.; Ismail, N.I.M.; Yin, O.S. Beneficial properties of probiotics. Trop. Life Sci. Res. 2016, 27, 73–90. [Google Scholar] [CrossRef] [PubMed]
- McLean, N.W.; Rosenstein, I.J. Characterisation and selection of a Lactobacillus species to re-colonise the vagina of women with recurrent bacterial vaginosis. J. Med. Microbiol. 2000, 49, 543–552. [Google Scholar] [CrossRef]
- McMillan, A.; Dell, M.; Zellar, M.P.; Cribby, S.; Martz, S.; Hong, E.; Fu, J.; Abbas, A.; Dang, T.; Miller, W.; et al. Disruption of urogenital biofilms by lactobacilli. Colloids Surf. B Biointerfaces 2011, 86, 58–64. [Google Scholar] [CrossRef]
- Petricevic, L.; Witt, A. The role of Lactobacillus casei rhamnosus Lcr35 in restoring the normal vaginal flora after antibiotic treatment of bacterial vaginosis. BJOG 2008, 115, 1369–1374. [Google Scholar] [CrossRef]
- Marcone, V.; Calzolari, E.; Bertini, M. Effectiveness of vaginal administration of Lactobacillus rhamnosus following conventional metronidazole therapy: How to lower the rate of bacterial vaginosis recurrences. New Microbiol. 2008, 31, 429–433. [Google Scholar]
- Marcone, V.; Rocca, G.; Lichtner, M.; Calzolari, E. Long-term vaginal administration of Lactobacillus rhamnosus as a complementary approach to management of bacterial vaginosis. Int. J. Gynecol. Obstet. 2010, 110, 223–226. [Google Scholar] [CrossRef]
- Hemmerling, A.; Harrison, W.; Schroeder, A.; Park, J.; Korn, A.; Shiboski, S.; Foster-Rosales, A.; Cohen, C.R. Phase 2a study assessing colonization efficiency, safety, and acceptability of Lactobacillus crispatus CTV-05 in women with bacterial vaginosis. Sex. Transm. Dis. 2010, 37, 745–750. [Google Scholar] [CrossRef]
- Cohen, C.R.; Wierzbicki, M.R.; French, A.L.; Morris, S.; Newmann, S.; Reno, H.; Green, L.; Miller, S.; Powell, J.; Parks, T.; et al. Randomized trial of Lactin-V to prevent recurrence of bacterial vaginosis. N. Engl. J. Med. 2020, 382, 1906–1915. [Google Scholar] [CrossRef] [PubMed]
- Recine, N.; Palma, E.; Domenici, L.; Giorgini, M.; Imperiale, L.; Sassu, C.; Musella, A.; Marchetti, C.; Muzii, L.; Panici, P.B. Restoring vaginal microbiota: Biological control of bacterial vaginosis. A prospective case–control study using Lactobacillus rhamnosus BMX 54 as adjuvant treatment against bacterial vaginosis. Arch. Gynecol. Obstet. 2016, 293, 101–107. [Google Scholar] [CrossRef]
- Ling, Z.; Liu, X.; Chen, W.; Luo, Y.; Yuan, L.; Xia, Y.; Nelson, K.E.; Huang, S.; Zhang, S.; Wang, Y.; et al. The restoration of the vaginal microbiota after treatment for bacterial vaginosis with metronidazole or probiotics. Microb. Ecol. 2013, 65, 773–780. [Google Scholar] [CrossRef]
- Cianci, A.; Cicinelli, E.; De Leo, V.; Fruzzetti, F.; Massaro, M.G.; Bulfoni, A.; Parazzini, F.; Perino, A. Observational prospective study on Lactobacillus plantarum P 17630 in the prevention of vaginal infections, during and after systemic antibiotic therapy or in women with recurrent vaginal or genitourinary infections. J. Obstet. Gynaecol. 2018, 38, 693–696. [Google Scholar] [CrossRef]
- Sudha, M.R.; Yelikar, K.A.; Deshpande, S. Clinical Study of Bacilluscoagulans Unique IS-2 (ATCC PTA-11748) in the Treatment of Patients with Bacterial Vaginosis. Indian J. Microbiol. 2012, 52, 396–399. [Google Scholar] [CrossRef]
- Martinez, R.C.; Franceschini, S.A.; Patta, M.C.; Quintana, S.M.; Gomes, B.C.; De Martinis, E.C.; Reid, G. Improved cure of bacterial vaginosis with single dose of tinidazole (2 g), Lactobacillus rhamnosus GR-1, and Lactobacillus reuteri RC-14: A randomized, double-blind, placebo-controlled trial. Can. J. Microbiol. 2009, 55, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Heczko, P.B.; Tomusiak, A.; Adamski, P.; Jakimiuk, A.J.; Stefański, G.; Mikołajczyk-Cichońska, A.; Suda-Szczurek, M.; Strus, M. Supplementation of standard antibiotic therapy with oral probiotics for bacterial vaginosis and aerobic vaginitis: A randomised, double-blind, placebo-controlled trial. BMC Womens Health 2015, 15, 115. [Google Scholar] [CrossRef]
- Anukam, K.; Osazuwa, E.; Ahonkhai, I.; Ngwu, M.; Osemene, G.; Bruce, A.W.; Reid, G. Augmentation of antimicrobial metronidazole therapy of bacterial vaginosis with oral probiotic Lactobacillus rhamnosus GR-1 and Lactobacillus reuteri RC-14: Randomized, double-blind, placebo controlled trial. Microb. Infect. 2006, 8, 1450–1454. [Google Scholar] [CrossRef]
- Hummelen, R.; Changalucha, J.; Butamanya, N.L.; Cook, A.; Habbema, J.D.F.; Reid, G. Lactobacillus rhamnosus GR-1 and L. reuteri RC-14 to prevent or cure bacterial vaginosis among women with HIV. Int. J. Gynecol. Obstet 2010, 111, 245–248. [Google Scholar] [CrossRef]
- Larsson, P.-G.; Stray-Pedersen, B.; Ryttig, K.R.; Larsen, S. Human lactobacilli as supplementation of clindamycin to patients with bacterial vaginosis reduce the recurrence rate; a 6-month, double-blind, randomized, placebo-controlled study. BMC Womens Health 2008, 8, 3. [Google Scholar] [CrossRef]
- Eriksson, K.; Carlsson, B.; Forsum, U.; Larsson, P. A double-blind treatment study of bacterial vaginosis with normal vaginal lactobacilli after an open treatment with vaginal clindamycin ovules. Acta Derm. Venereol. 2005, 85, 42–46. [Google Scholar] [CrossRef]
- Laue, C.; Papazova, E.; Liesegang, A.; Pannenbeckers, A.; Arendarski, P.; Linnerth, B.; Domig, K.; Kneifel, W.; Petricevic, L.; Schrezenmeir, J. Effect of a yoghurt drink containing Lactobacillus strains on bacterial vaginosis in women–a double-blind, randomised, controlled clinical pilot trial. Benef. Microbes 2018, 9, 35–50. [Google Scholar] [CrossRef] [PubMed]
- van de Wijgert, J.H.H.M.; Verwijs, M.C.; Agaba, S.K.; Bronowski, C.; Mwambarangwe, L.; Uwineza, M.; Lievens, E.; Nivoliez, A.; Ravel, J.; Darby, A.C. Intermittent Lactobacilli-containing vaginal probiotic or metronidazole use to prevent bacterial vaginosis recurrence: A pilot study incorporating microscopy and sequencing. Sci. Rep. 2020, 10, 3884. [Google Scholar] [CrossRef] [PubMed]
- Marcotte, H.; Larsson, P.G.; Andersen, K.K.; Zuo, F.; Mikkelsen, L.S.; Brandsborg, E.; Gray, G.; Laher, F.; Otwombe, K. An exploratory pilot study evaluating the supplementation of standard antibiotic therapy with probiotic lactobacilli in south African women with bacterial vaginosis. BMC Infect. Dis. 2019, 19, 824. [Google Scholar] [CrossRef]
- Happel, A.-U.; Singh, R.; Mitchev, N.; Mlisana, K.; Jaspan, H.B.; Barnabas, S.L.; Passmore, J.-A.S. Testing the regulatory framework in South Africa–a single-blind randomized pilot trial of commercial probiotic supplementation to standard therapy in women with bacterial vaginosis. BMC Infect. Dis. 2020, 20, 491. [Google Scholar] [CrossRef]
- Ehrstrom, S.; Daroczy, K.; Rylander, E.; Samuelsson, C.; Johannesson, U.; Anzen, B.; Pahlson, C. Lactic acid bacteria colonization and clinical outcome after probiotic supplementation in conventionally treated bacterial vaginosis and vulvovaginal candidiasis. Microbes Infect 2010, 12, 691–699. [Google Scholar] [CrossRef] [PubMed]
- Hallen, A.; Jarstrand, C.; Pahlson, C. Treatment of bacterial vaginosis with lactobacilli. Sex. Transm. Dis. 1992, 19, 146–148. [Google Scholar] [CrossRef]
- Bomba, A.; Nemcová, R.r.; Mudroňová, D.; Guba, P. The possibilities of potentiating the efficacy of probiotics. Trends Food Sci. Technol. 2002, 13, 121–126. [Google Scholar] [CrossRef]
- Timmerman, H.; Koning, C.; Mulder, L.; Rombouts, F.; Beynen, A. Monostrain, multistrain and multispecies probiotics—A comparison of functionality and efficacy. Int. J. Food Microbiol. 2004, 96, 219–233. [Google Scholar] [CrossRef]
- Rostok, M.; Hütt, P.; Rööp, T.; Smidt, I.; Štšepetova, J.; Salumets, A.; Mändar, R. Potential vaginal probiotics: Safety, tolerability and preliminary effectiveness. Benef. Microbes 2019, 10, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Vujic, G.; Knez, A.J.; Stefanovic, V.D.; Vrbanovic, V.K. Efficacy of orally applied probiotic capsules for bacterial vaginosis and other vaginal infections: A double-blind, randomized, placebo-controlled study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 168, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Hantoushzadeh, S.; Golshahi, F.; Javadian, P.; Khazardoost, S.; Aram, S.; Hashemi, S.; Mirarmandehi, B.; Borna, S. Comparative efficacy of probiotic yoghurt and clindamycin in treatment of bacterial vaginosis in pregnant women: A randomized clinical trial. J. Matern. Fetal Neonatal Med. 2012, 25, 1021–1024. [Google Scholar] [CrossRef]
- Mastromarino, P.; Macchia, S.; Meggiorini, L.; Trinchieri, V.; Mosca, L.; Perluigi, M.; Midulla, C. Effectiveness of Lactobacillus-containing vaginal tablets in the treatment of symptomatic bacterial vaginosis. Clin. Microbiol. Infect. 2009, 15, 67–74. [Google Scholar] [CrossRef]
- Hemalatha, R.; Mastromarino, P.; Ramalaxmi, B.; Balakrishna, N.; Sesikeran, B. Effectiveness of vaginal tablets containing lactobacilli versus pH tablets on vaginal health and inflammatory cytokines: A randomized, double-blind study. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 3097–3105. [Google Scholar] [CrossRef] [PubMed]
- Ya, W.; Reifer, C.; Miller, L.E. Efficacy of vaginal probiotic capsules for recurrent bacterial vaginosis: A double-blind, randomized, placebo-controlled study. Am. J. Obstet. Gynecol. 2010, 203, 120.e1–120.e6. [Google Scholar] [CrossRef]
- Vicariotto, F.; Mogna, L.; Del Piano, M. Effectiveness of the two microorganisms Lactobacillus fermentum LF15 and Lactobacillus plantarum LP01, formulated in slow-release vaginal tablets, in women affected by bacterial vaginosis: A pilot study. J. Clin. Gastroenterol. 2014, 48, S106–S112. [Google Scholar] [CrossRef] [PubMed]
- Neri, A.; Sabah, G.; Samra, Z. Bacterial vaginosis in pregnancy treated with yoghurt. Acta Obstet. Gynecol. Scand. 1993, 72, 17–19. [Google Scholar] [CrossRef] [PubMed]
- Pineiro, M.; Asp, N.-G.; Reid, G.; Macfarlane, S.; Morelli, L.; Brunser, O.; Tuohy, K. FAO Technical meeting on prebiotics. J. Clin. Gastroenterol. 2008, 42, S156–S159. [Google Scholar] [CrossRef]
- Markowiak, P.; Śliżewska, K. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Özmen, Ş.; Turhan, N.Ö.; Seçkin, N.C. Garnerella-associated vaginitis: Comparison of three treatment modalities. Turk. J. Med. Sci. 1998, 28, 171–174. [Google Scholar]
- Coste, I.; Judlin, P.; Lepargneur, J.-P.; Bou-Antoun, S. Safety and efficacy of an intravaginal prebiotic gel in the prevention of recurrent bacterial vaginosis: A randomized double-blind study. Obstet. Gynecol. Int. 2012, 2012, 147867. [Google Scholar] [CrossRef] [PubMed]
- Hakimi, S.; Farhan, F.; Farshbaf-Khalili, A.; Dehghan, P.; Javadzadeh, Y.; Abbasalizadeh, S.; Khalvati, B. The effect of prebiotic vaginal gel with adjuvant oral metronidazole tablets on treatment and recurrence of bacterial vaginosis: A triple-blind randomized controlled study. Arch. Gynecol. Obstet. 2018, 297, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Chavez, S.A.; Arevalo-Gallegos, S.; Rascon-Cruz, Q. Lactoferrin: Structure, function and applications. Int. J. Antimicrob. Agents 2009, 33, 301.e1–301.e8. [Google Scholar] [CrossRef]
- Adlerova, L.; Bartoskova, A.; Faldyna, M. Lactoferrin: A review. Vet. Med. 2008, 53, 457–468. [Google Scholar] [CrossRef]
- Ward, P.P.; Paz, E.; Conneely, O.M. Multifunctional roles of lactoferrin: A critical overview. Cell. Mol. Life Sci. 2005, 62, 2540–2548. [Google Scholar] [CrossRef]
- Superti, F.; De Seta, F. Warding off recurrent yeast and bacterial vaginal infections: Lactoferrin and Lactobacilli. Microorganisms 2020, 8, 130. [Google Scholar] [CrossRef]
- De Alberti, D.; Russo, R.; Terruzzi, F.; Nobile, V.; Ouwehand, A.C. Lactobacilli vaginal colonisation after oral consumption of Respecta® complex: A randomised controlled pilot study. Arch. Gynecol. Obstet. 2015, 292, 861–867. [Google Scholar] [CrossRef]
- Jang, S.E.; Jeong, J.J.; Choi, S.Y.; Kim, H.; Han, M.J.; Kim, D.H. Lactobacillus rhamnosus HN001 and Lactobacillus acidophilus La-14 attenuate Gardnerella vaginalis-infected bacterial vaginosis in mice. Nutrients 2017, 9, 531. [Google Scholar] [CrossRef]
- Russo, R.; Karadja, E.; De Seta, F. Evidence-based mixture containing Lactobacillus strains and lactoferrin to prevent recurrent bacterial vaginosis: A double blind, placebo controlled, randomised clinical trial. Benef. Microbes 2019, 10, 19–26. [Google Scholar] [CrossRef]
- Liu, S.-X.; Li, Y.-H.; Dai, W.-K.; Li, X.-S.; Qiu, C.-Z.; Ruan, M.-L.; Zou, B.; Dong, C.; Liu, Y.-H.; He, J.-Y.; et al. Fecal microbiota transplantation induces remission of infantile allergic colitis through gut microbiota re-establishment. World J. Gastroenterol. 2017, 23, 8570–8581. [Google Scholar] [CrossRef]
- Aron-Wisnewsky, J.; Clément, K.; Nieuwdorp, M. Fecal microbiota transplantation: A future therapeutic option for obesity/diabetes? Curr. Diab. Rep. 2019, 19, 51. [Google Scholar] [CrossRef]
- Vaughn, B.P.; Rank, K.M.; Khoruts, A. Fecal microbiota transplantation: Current status in treatment of GI and liver disease. Clin. Gastroenterol. Hepatol. 2019, 17, 353–361. [Google Scholar] [CrossRef]
- Benech, N.; Sokol, H. Fecal microbiota transplantation in gastrointestinal disorders: Time for precision medicine. Genome Med. 2020, 12, 58. [Google Scholar] [CrossRef]
- Shasthry, S.M. Fecal microbiota transplantation in alcohol related liver diseases. Clin. Mol. Hepatol. 2020, 26, 294–301. [Google Scholar] [CrossRef]
- Kaakoush, N.O. Fecal transplants as a microbiome-based therapeutic. Curr. Opin. Microbiol. 2020, 56, 16–23. [Google Scholar] [CrossRef]
- Kaźmierczak-Siedlecka, K.; Daca, A.; Fic, M.; van de Wetering, T.; Folwarski, M.; Makarewicz, W. Therapeutic methods of gut microbiota modification in colorectal cancer management–fecal microbiota transplantation, prebiotics, probiotics, and synbiotics. Gut Microbes 2020, 11, 1518–1530. [Google Scholar] [CrossRef]
- DeLong, K.; Bensouda, S.; Zulfiqar, F.; Zierden, H.C.; Hoang, T.M.; Abraham, A.G.; Coleman, J.S.; Cone, R.A.; Gravitt, P.E.; Hendrix, C.W.; et al. Conceptual design of a universal donor screening approach for vaginal microbiota transplant. Front. Cell. Infect. Microbiol. 2019, 9, 306. [Google Scholar] [CrossRef]
- Lev-Sagie, A.; Goldman-Wohl, D.; Cohen, Y.; Dori-Bachash, M.; Leshem, A.; Mor, U.; Strahilevitz, J.; Moses, A.E.; Shapiro, H.; Yagel, S.; et al. Vaginal microbiome transplantation in women with intractable bacterial vaginosis. Nat. Med. 2019, 25, 1500–1504. [Google Scholar] [CrossRef]
- US Food Drug Administration. Fecal Microbiota for Transplantation: Safety Communication-Risk of Serious Adverse Reactions Due to Transmission of Multi-Drug Resistant Organisms. Available online: https://www.fda.gov/safety/medical-product-safety-information/fecal-microbiota-transplantation-safety-communication-risk-serious-adverse-reactions-due (accessed on 13 January 2021).
- US Food Drug Administration. Fecal Microbiota for Transplantation: Safety Alert-Risk of Serious Adverse Events Likely due to Transmission of Pathogenic Organisms. Available online: https://www.fda.gov/safety/medical-product-safety-information/fecal-microbiota-transplantation-safety-alert-risk-serious-adverse-events-likely-due-transmission. (accessed on 13 January 2021).
- Mehta, S.D. Systematic review of randomized trials of treatment of male sexual partners for improved bacteria vaginosis outcomes in women. Sex. Transm. Dis. 2012, 39, 822–830. [Google Scholar] [CrossRef]
- Amaya-Guio, J.; Viveros-Carreño, D.A.; Sierra-Barrios, E.M.; Martinez-Velasquez, M.Y.; Grillo-Ardila, C.F.; Group, C.S. Antibiotic treatment for the sexual partners of women with bacterial vaginosis. Cochrane Database Syst. Rev. 2016, CD011701. [Google Scholar] [CrossRef]
- Reid, G.; Charbonneau, D.; Erb, J.; Kochanowski, B.; Beuerman, D.; Poehner, R.; Bruce, A.W. Oral use of Lactobacillus rhamnosus GR-1 and L. fermentum RC-14 significantly alters vaginal flora: Randomized, placebo-controlled trial in 64 healthy women. FEMS Immunol. Med. Microbiol. 2003, 35, 131–134. [Google Scholar] [CrossRef]
- Senok, A.C.; Verstraelen, H.; Temmerman, M.; Botta, G.A. Probiotics for the treatment of bacterial vaginosis. Cochrane Database Syst. Rev. 2009, CD006289. [Google Scholar] [CrossRef]
- Bisanz, J.E.; Seney, S.; McMillan, A.; Vongsa, R.; Koenig, D.; Wong, L.; Dvoracek, B.; Gloor, G.B.; Sumarah, M.; Ford, B.; et al. A systems biology approach investigating the effect of probiotics on the vaginal microbiome and host responses in a double blind, placebo-controlled clinical trial of post-menopausal women. PLoS ONE 2014, 9, e104511. [Google Scholar] [CrossRef] [PubMed]
- Gille, C.; Boer, B.; Marschal, M.; Urschitz, M.S.; Heinecke, V.; Hund, V.; Speidel, S.; Tarnow, I.; Mylonas, I.; Franz, A.; et al. Effect of probiotics on vaginal health in pregnancy. EFFPRO, a randomized controlled trial. Am. J. Obstet. Gynecol. 2016, 215, 608.e1–608.e7. [Google Scholar] [CrossRef]
- Strus, M.; Chmielarczyk, A.; Kochan, P.; Adamski, P.; Chełmicki, Z.; Chełmicki, A.; Pałucha, A.; Heczko, P.B. Studies on the effects of probiotic Lactobacillus mixture given orally on vaginal and rectal colonization and on parameters of vaginal health in women with intermediate vaginal flora. Eur. J. Obstet. Gynecol. Reprod. Biol. 2012, 163, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Ser, H.-L.; Letchumanan, V.; Goh, B.-H.; Wong, S.H.; Lee, L.-H. The use of fecal microbiome transplant in treating human diseases: Too early for poop? Front. Microbiol. 2021, 12, 1005. [Google Scholar] [CrossRef] [PubMed]
- Akram, A.; Maley, M.; Gosbell, I.; Nguyen, T.; Chavada, R. Utility of 16S rRNA PCR performed on clinical specimens in patient management. Int. J. Infect. Dis. 2017, 57, 144–149. [Google Scholar] [CrossRef]
- Sobel, J.D.; Kaur, N.; Woznicki, N.A.; Boikov, D.; Aguin, T.; Gill, G.; Akins, R.A. Prognostic indicators of recurrence of bacterial vaginosis. J. Clin. Microbiol. 2019, 57, e00227-19. [Google Scholar] [CrossRef]
- Aslan, M.M.; Uslu Yuvacı, H.; Köse, O.; Toptan, H.; Akdemir, N.; Köroğlu, M.; Cevrioğlu, A.S.; Özden, S. SARS-CoV-2 is not present in the vaginal fluid of pregnant women with COVID-19. J. Matern. Fetal Neonatal Med. 2020, 1–3. [Google Scholar] [CrossRef]
- Li, D.; Jin, M.; Bao, P.; Zhao, W.; Zhang, S. Clinical characteristics and results of semen tests among men with coronavirus disease 2019. JAMA Network Open 2020, 3, e208292. [Google Scholar] [CrossRef]
- Qiu, L.; Liu, X.; Xiao, M.; Xie, J.; Cao, W.; Liu, Z.; Morse, A.; Xie, Y.; Li, T.; Zhu, L. SARS-CoV-2 is not detectable in the vaginal fluid of women with severe COVID-19 infection. Clin. Infect. Dis. 2020, 71, 813–817. [Google Scholar] [CrossRef]
- Paoli, D.; Pallotti, F.; Turriziani, O.; Mazzuti, L.; Antonelli, G.; Lenzi, A.; Lombardo, F. SARS-CoV-2 presence in seminal fluid: Myth or reality. Andrology 2021, 9, 23–26. [Google Scholar] [CrossRef]
- Schwartz, A.; Yogev, Y.; Zilberman, A.; Alpern, S.; Many, A.; Yousovich, R.; Gamzu, R. Detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in vaginal swabs of women with acute SARS-CoV-2 infection: A prospective study. BJOG 2021, 128, 97–100. [Google Scholar] [CrossRef]
- Sharun, K.; Tiwari, R.; Dhama, K. SARS-CoV-2 in semen: Potential for sexual transmission in COVID-19. Int. J. Surg. 2020, 84, 156–158. [Google Scholar] [CrossRef] [PubMed]
- Tur-Kaspa, T.; Hildebrand, G.; Cohen, D.; Tur-Kaspa, I. Is COVID-19 a sexually transmitted disease? A systematic review. Fertil. Steril. 2020, 114, e522–e528. [Google Scholar] [CrossRef]
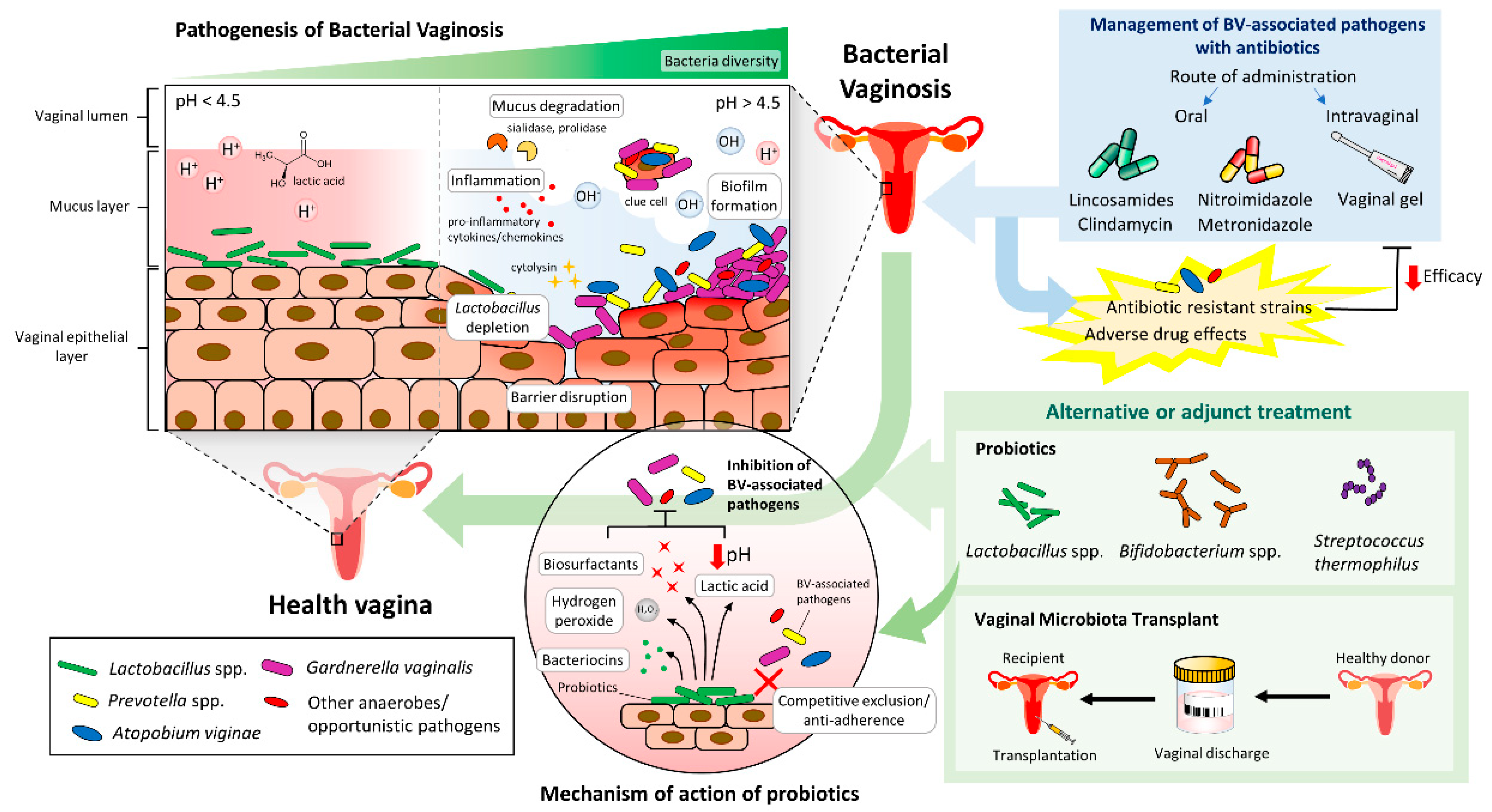
| Study | Country | Study Period | Age Range (Years Old) | Diagnostic Process | Population Characteristics | BV Prevalence | ||
|---|---|---|---|---|---|---|---|---|
| Overall (95% CI or Proportion) | Based on Age (95% CI or Proportion) | Based on Race/Ethnicity | ||||||
| [76] | United States | 2001–2002 2003–2004 | 14–49 | Self-collected vaginal swabs and Nugent’s criteria | Female participants selected from the National Health and Nutrition Examination Survey study | 29.2 (27.2–31.3) | 14–19 years old: 23.3 (20.1–26.9) 20–29 years old: 31.2 (27.0–35.6) 30–39 years old: 28.8 (25.4–31.1) 40–49 years old: 31.3 (27.3–35.6) | White/non-Hispanic: 23.2 (20.8–25.8) Black/non-Hispanic: 51.6 (47.9–55.2) Mexican American: 32.1 (27.7–36.9) Other race/Hispanic: 36.6 (27.6–45.9) |
| [71] | 2001–2004 | 14–49 | Self-collected vaginal swabs and Nugent’s criteria | Female participants selected from the National Health and Nutrition Examination Survey study (80.5% of total interviewed or 83.6% of those examined) | 29.2 (27.2–31.3) | 14–19 years old: 23.3 (20.1–26.9) 20–29 years old: 31.1 (27.1–35.7) 30–39 years old: 28.1 (25.4–31.1) 40–49 years old: 31.3 (27.4–35.7) | White/non-Hispanic: 23.2 (20.8–25.9) Black/non-Hispanic: 51.4 (47.9–55.2) Mexican American: 31.9 (27.6–36.9) Other race/Hispanic: 25.9 (19.3–34.8) | |
| [77] | Mexico | January–July 1994 | Not available | Nugent’s criteria | Patients attended to the City Hospital for a regular gynecological consultation | 16.5 (67/405) | N/S | N/S |
| [78] | Grenada | January 2009–December 2011 | 15–49 | Pap smears | Women participated in the national centralized data bank at the General Hospital in St George’s, Grenada | 19.5 | 20–29 years old: 43.6% | N/S |
| [79] | Peru | December 1997–June 1998 | 18–67 | Vaginal swab collection at clinic, Amsel’s criteria, and Nugent’s criteria | Women registered through community-based organization in the ReproSalud Project (promote the improvement of reproductive health) | Amsel’s criteria: 33.6 (251/748) Nugent’s criteria: 40.8 (305/748) Either criteria: 43.7 (327/748) Both criteria: 30.6 (229/748) | >25 years old: 45.5 (275/604) ≤25 years old: 36.1 (52/144) | N/S |
| [80] | April–August 2001 | 18–30 | Self-collected vaginal swabs, sialidase, and PCR tests | Women recruited in a cross-sectional, population-based study to investigate the prevalence and the risk factors for HIV and other STDs | 26.6 (207/779) | N/S | N/S | |
| [81] | United Kingdom | January 1996–September 1998 | <45 | Amsel’s criteria | Women patients presenting for diagnosis at a genitourinary medicine clinic in Sheffield | 12.9 (1161/8989) | N/S | N/S |
| [82] | N/S | 15–49 | Spiegel’s criteria | Pregnant women (gestational ages of 15 and 24 weeks) attended the antenatal clinic at Barnsley District General Hospital, Barnsley | 3.54 (38/1073) | 11–20 years old: 3.23 (6/186) 21–30 years old: 4.32 (30/695) 31–35 years old: 1.48 (2/135) 36–40 years old: 0 (0/44) >40 years old: 0 (0/13) | N/S | |
| [83] | India | March–June 2002 | 18–40 | Nugent’s criteria | Women living in slum areas of Chennai recruited for HIV intervention study | 25 (120/487) | N/S | N/S |
| [84] | September–December 2002 | 15–49 | Vaginal swab at health centers and Nugent’s criteria | Community-based stud | 32.8 (26.38–39.22) | 15–24 years old: 34.2 (13/38) 25–49 years old: 32.5 (57/175) | N/S | |
| [85] | June–December 2004 | 18–40 | Nugent’s criteria | Women sex workers in Chennai recruited for HIV intervention study | 45 (40.6–48.7) | N/S | N/S | |
| [86] | December 2016–November 2018 | 42–64 | Vaginal swab at health centers and Nugent’s criteria | Outpatient female attended obstetrical and gynecological at Department of Obstetrics and Gynecology of Veer Surendra Sai Institute of Medical Sciences and Research, Burla, Odisha, India | 9.1 (19/209) | ≤55 years old: 43.1 (22/120) ≥55 years old: 56.9 (29/89) | N/S | |
| [87] | Nepal | November 2014–May 2015 | 10–60 | Nugent’s criteria | Women attended KIST Medical College Teaching Hospital | 24.4 (39/160) | 10–20 years old: 1.3 (2/7) 20–40 years old: 8.1 (13/48) 30–40 years old: 8.8 (14/75) 40–50 years old: 5 (8/22) 50–60 years old: 1.3 (2/8) | N/S |
| [88] | Ethiopia | November 2011–April 2012 | 18–40 | Vaginal swab collection at clinic and Nugent’s criteria | Outpatient female visited the obstetrical and gynecological clinical for antenatal care services of a tertiary level hospital in Addis Ababa | 19.4 (49/183) | ≤20 years old: 17.6 (3/17) 21–29 years old: 21.2 (33/156) ≥30 years old: 16.5 (13/79) | N/S |
| [89] | September 2015–July 2016 | 15–64 | Vaginal swab collection at clinic and Nugent’s criteria | Women attended gynecology and antenatal clinics | 48.6 (102/210) | 15–24 years old: 41.5 (22/53) 25–44 years old: 47.8 (56/117) 45–64 years old: 60 (24/40) | N/S | |
| [90] | Nigeria | 7 December 2010–15 July 2011 | 15–42 | Vaginal swab collection at clinic and Amsel’s criteria | Pregnant women presenting to the antenatal clinics of the University of Maiduguri Teaching Hospital | 17.3 (69/400) | 15–19 years old: 23.6 (25/106) 20–24 years old: 17.5 (32/183) 25–29 years old: 16.1 (10/62) 30–34 years old: 15 (6/40) 35–39 years old: 50 (3/6) 40–44 years old: (1/3) | |
| [91] | November 2011–March 2012 | 19–45 | Vaginal swab collection at clinic and Nugent’s criteria | Non-pregnant asymptomatic women attended the gynecological clinic | 40.1 (85/212) | <20 years old: 100 (1/1) 20–24 years old: 43.8 (7/16) 25–29 years old: 35.5 (22/62) 30–34 years old: 41.7 (28/67) 35–39 years old: 40.5 (15/37) ≥40 years old: 37.5 (12/32) | N/S | |
| [92] | Iran | 2006 | 18–40 | N/S | Pregnant women (before 20th week of gestation) registered in health centers | 17.5 (136/777) | <30 years old: 32.7 (124/379) >30 years old: 32 (39/122) | N/S |
| [93] | Albania | 1 January 2010–31 December 2010 | N/S | Vaginal swab at clinic and Amsel’s criteria | Pregnant women attended to the Regional Hospital of Fier | 23.3 (35/150) | N/S | N/S |
| [94] | Italy | April 1998–April 2001 | 40–79 | Vaginal swab at clinic and Nugent’s criteria | Non-pregnant women visited three clinics in northern Italy for routine gynecologic examinations | 7.6 (113/1486) | N/S | N/S |
| Amsel’s Criteria | Nugent’s Criteria * | Hay/Ison Criteria | |
|---|---|---|---|
| Type | Clinical and laboratory diagnosis | Laboratory diagnosis | Laboratory diagnosis |
| Diagnosis duration | Fast | Long | Long |
| Expertise requirement | Clinicians | Experienced laboratory technicians and pathologist | Experienced laboratory technicians |
| Laboratory requirement | Low | High | Moderate |
| Grading system | Diagnosis is confirmed when three out of four criteria are fulfilled.
| Score 0–4: Normal flora Score 4–6: Intermediate Score ≥ 7: BV | Group 1: Normal flora (Lactobacillus only) Group 2: Intermediate (Lactobacillus = Gardnerella) Group 3: BV (Lactobacillus < Gardnerella) |
| Guidelines | Regime Type | Drug (Dose), Frequency and Duration |
|---|---|---|
| Guidelines for the Management of Sexually Transmitted Infections [214] | Standard | Oral metronidazole (400 or 500 mg), twice daily for 7 days |
| Alternative | Oral metronidazole (2 g), single dose | |
| Intravaginal clindamycin cream (2%, 5 g), once daily for 7 days | ||
| Intravaginal metronidazole gel (0.75%, 5 g), twice daily for 5 days | ||
| Oral clindamycin (300 mg), twice daily for 7 days | ||
| UK National Guideline for the management of Bacterial Vaginosis [215] | Standard | Oral metronidazole (400 mg), twice daily for 5–7 days |
| Oral metronidazole (2 g), single dose | ||
| Alternative | Intravaginal metronidazole gel (0.75%), once daily for 5 days, OR | |
| Intravaginal clindamycin cream (2%), once daily for 7 days | ||
| Oral tinidazole (2 g), single dose | ||
| Sexually Transmitted Diseases Treatment Guidelines [216] | Standard | Oral metronidazole (500 mg), twice daily for 7 days |
| Intravaginal metronidazole gel (0.75%, 5 g), once daily for 5 days | ||
| Alternative | Intravaginal clindamycin cream (2%, 5 g), once daily for 7 days | |
| Oral tinidazole (2 g), once daily for 2 days | ||
| Oral tinidazole (1 g), once daily for 5 days | ||
| Oral clindamycin (300 mg), twice daily for 7 days | ||
| Intravaginal clindamycin ovules (100 mg), once daily for 3 days | ||
| Australian STI Management guidelines for use in primary care [217] | Standard | Oral metronidazole (400 mg), twice daily for 7 days |
| Intravaginal metronidazole gel (0.75%, 5g), once daily for 5 days | ||
| Alternative | Intravaginal clindamycin cream (2%), 5 g once daily for 7 days | |
| Oral clindamycin (300 mg), twice daily for 7 days | ||
| Oral metronidazole (2 g), single dose | ||
| Oral tinidazole (2 g), single dose | ||
| European International Union against sexually transmitted infections (IUSTI) World Health Organisation (WHO) guideline on the management of vaginal discharge [208,218] | Standard | Oral metronidazole (400–500 mg), twice daily for 5–7 days |
| Intravaginal metronidazole gel (0.75%), intravaginally, once daily for 5 days | ||
| Intravaginal clindamycin cream (2%), once daily for 7 days | ||
| Alternative | Oral metronidazole (2 g), single dose | |
| Oral tinidazole (2 g), single dose | ||
| Oral tinidazole (1 g) orally, once daily for 5 days | ||
| Oral metronidazole (300 mg), twice daily, for 7 days | ||
| Intravaginal dequalinium chloride tablet (10 mg), once daily for 6 days |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joseph, R.J.; Ser, H.-L.; Kuai, Y.-H.; Tan, L.T.-H.; Arasoo, V.J.T.; Letchumanan, V.; Wang, L.; Pusparajah, P.; Goh, B.-H.; Ab Mutalib, N.-S.; et al. Finding a Balance in the Vaginal Microbiome: How Do We Treat and Prevent the Occurrence of Bacterial Vaginosis? Antibiotics 2021, 10, 719. https://doi.org/10.3390/antibiotics10060719
Joseph RJ, Ser H-L, Kuai Y-H, Tan LT-H, Arasoo VJT, Letchumanan V, Wang L, Pusparajah P, Goh B-H, Ab Mutalib N-S, et al. Finding a Balance in the Vaginal Microbiome: How Do We Treat and Prevent the Occurrence of Bacterial Vaginosis? Antibiotics. 2021; 10(6):719. https://doi.org/10.3390/antibiotics10060719
Chicago/Turabian StyleJoseph, Rebecca Jane, Hooi-Leng Ser, Yi-He Kuai, Loh Teng-Hern Tan, Valliammai Jayanthi Thirunavuk Arasoo, Vengadesh Letchumanan, Lijing Wang, Priyia Pusparajah, Bey-Hing Goh, Nurul-Syakima Ab Mutalib, and et al. 2021. "Finding a Balance in the Vaginal Microbiome: How Do We Treat and Prevent the Occurrence of Bacterial Vaginosis?" Antibiotics 10, no. 6: 719. https://doi.org/10.3390/antibiotics10060719
APA StyleJoseph, R. J., Ser, H.-L., Kuai, Y.-H., Tan, L. T.-H., Arasoo, V. J. T., Letchumanan, V., Wang, L., Pusparajah, P., Goh, B.-H., Ab Mutalib, N.-S., Chan, K.-G., & Lee, L.-H. (2021). Finding a Balance in the Vaginal Microbiome: How Do We Treat and Prevent the Occurrence of Bacterial Vaginosis? Antibiotics, 10(6), 719. https://doi.org/10.3390/antibiotics10060719










