Antimicrobials and Food-Related Stresses as Selective Factors for Antibiotic Resistance along the Farm to Fork Continuum
Abstract
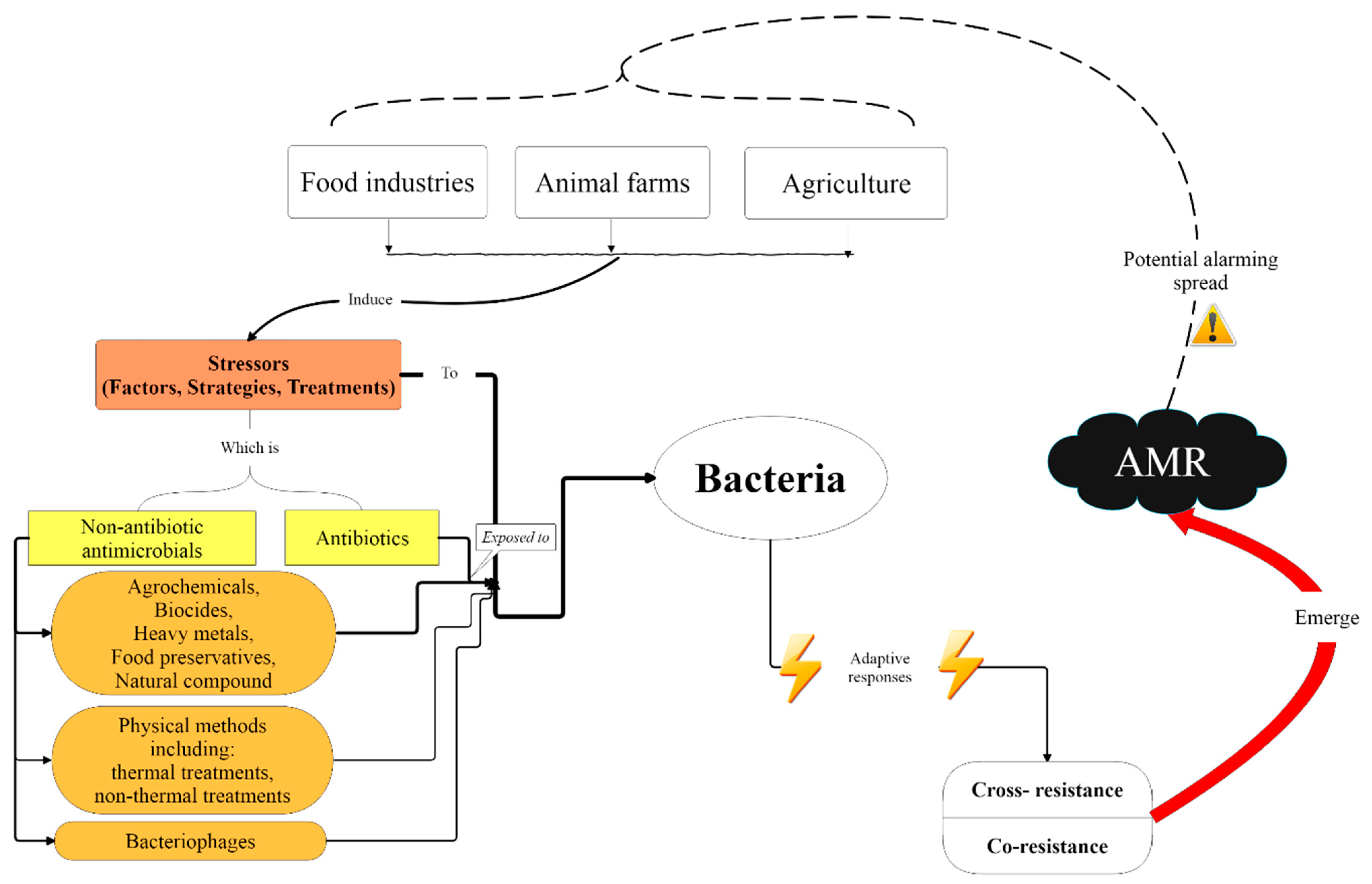
1. Introduction
2. Interaction of the Use of Non-Antibiotic Antimicrobials with A Potential Antibiotic Decreased Susceptibility
2.1. Agrochemicals
2.2. Biocides
2.3. Heavy Metals
2.4. Food Preservatives and Decontaminants
2.5. Natural Compounds
3. Physical Methods of Food Processing That May Influence Antibiotic Susceptibility
3.1. Thermal Treatments
3.2. Non-Thermal Treatments
3.2.1. Ultraviolet (UV) and UV-Based Advanced Oxidation Processes
3.2.2. Non-Thermal or Cold Atmospheric Plasma
4. Influence of Bacteriophage Application on Antibiotic Susceptibility
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Estimating the Burden of Foodborne Diseases. Available online: https://www.who.int/activities/estimating-the-burden-of-foodborne-diseases (accessed on 20 April 2021).
- Caniça, M.; Manageiro, V.; Abriouel, H.; Moran-Gilad, J.; Franz, C.M.A.P. Antibiotic resistance in foodborne bacteria. Trends Food Sci. Technol. 2019, 84, 41–44. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority); ECDC (European Centre for Disease Prevention and Control). The European Union Summary Report on Antimicrobial Resistance in zoonotic and indicator bacteria from humans, animals and food in 2018/2019. EFSA J. 2021, 19, 6490. [Google Scholar] [CrossRef]
- Capita, R.; Alonso-Calleja, C. Antibiotic-Resistant Bacteria: A Challenge for the Food Industry. Crit. Rev. Food Sci. Nutr. 2013, 53, 11–48. [Google Scholar] [CrossRef] [PubMed]
- Doyle, M.P.; Busta, F.; Cords, B.R.; Davidson, P.M.; Hawke, J.; Hurd, H.S.; Isaacson, R.E.; Matthews, K.; Maurer, J.; Meng, J.; et al. Antimicrobial resistance: Implications for the food system. Compr. Rev. Food Sci. Food Saf. 2006, 5, 71–137. [Google Scholar] [CrossRef]
- Begley, M.; Hill, C. Stress adaptation in foodborne pathogens. Annu. Rev. Food Sci. Technol. 2015, 6, 191–210. [Google Scholar] [CrossRef]
- Liao, X.; Ma, Y.; Daliri, E.B.M.; Koseki, S.; Wei, S.; Liu, D.; Ye, X.; Chen, S.; Ding, T. Interplay of antibiotic resistance and food-associated stress tolerance in foodborne pathogens. Trends Food Sci. Technol. 2020, 95, 97–106. [Google Scholar] [CrossRef]
- De Silva, N.I.; Brooks, S.; Lumyong, S.; Hyde, K.D. Use of endophytes as biocontrol agents. Fungal Biol. Rev. 2019, 33, 133–148. [Google Scholar] [CrossRef]
- Carvalho, F.P. Agriculture, pesticides, food security and food safety. Environ. Sci. Policy 2006, 9, 685–692. [Google Scholar] [CrossRef]
- Kleiner, D.K.; Katz, S.E.; Ward, P.M.L. Development of in vitro antimicrobial resistance in bacteria exposed to residue level exposures of antimicrobial drugs, pesticides and veterinary drugs. Chemotherapy 2007, 53, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Bordas, A.C.; Brady, M.S.; Siewierski, M.; Katz, S.E. In vitro enhancement of antibiotic resistance development—Interaction of residue levels of pesticides and antibiotics. J. Food Prot. 1997, 60, 531–536. [Google Scholar] [CrossRef]
- Kurenbach, B.; Marjoshi, D.; Amábile-Cuevas, C.F.; Ferguson, G.C.; Godsoe, W.; Gibson, P.; Heinemann, J.A. Sublethal exposure to commercial formulations of the herbicides dicamba, 2,4-dichlorophenoxyacetic acid, and glyphosate cause changes in antibiotic susceptibility in Escherichia coli and Salmonella enterica serovar Typhimurium. MBio 2015, 6, e00009-15. [Google Scholar] [CrossRef]
- Xing, Y.; Wu, S.; Men, Y. Exposure to environmental levels of pesticides stimulates and diversifies evolution in Escherichia coli toward higher antibiotic resistance. Environ. Sci. Technol. 2020, 54, 8770–8778. [Google Scholar] [CrossRef]
- Capita, R.; Riesco-Peláez, F.; Alonso-Hernando, A.; Alonso-Calleja, C. Exposure of Escherichia coli ATCC 12806 to sublethal concentrations of food-grade biocides influences its ability to form biofilm, resistance to antimicrobials, and ultrastructure. Appl. Environ. Microbiol. 2014, 80, 1268–1280. [Google Scholar] [CrossRef]
- Karatzas, K.A.G.; Webber, M.A.; Jorgensen, F.; Woodward, M.J.; Piddock, L.J.V.; Humphrey, T.J. Prolonged treatment of Salmonella enterica serovar Typhimurium with commercial disinfectants selects for multiple antibiotic resistance, increased efflux and reduced invasiveness. J. Antimicrob. Chemother. 2007, 60, 947–955. [Google Scholar] [CrossRef]
- Sonbol, F.I.; El-Banna, T.E.; Abd El-Aziz, A.A.; El-Ekhnawy, E. Impact of triclosan adaptation on membrane properties, efflux and antimicrobial resistance of Escherichia coli clinical isolates. J. Appl. Microbiol. 2019, 126, 730–739. [Google Scholar] [CrossRef]
- Noguchi, N.; Tamura, M.; Narui, K.; Wakasugi, K.; Sasatsu, M. Frequency and genetic characterization of multidrug-resistant mutants of Staphylococcus aureus after selection with individual antiseptics and fluoroquinolones. Biol. Pharm. Bull. 2002, 25, 1129–1132. [Google Scholar] [CrossRef][Green Version]
- Yu, T.; Jiang, X.; Zhang, Y.; Ji, S.; Gao, W.; Shi, L. Effect of benzalkonium chloride adaptation on sensitivity to antimicrobial agents and tolerance to environmental stresses in Listeria monocytogenes. Front. Microbiol. 2018, 9, 2906. [Google Scholar] [CrossRef]
- Mc Cay, P.H.; Ocampo-Sosa, A.A.; Fleming, G.T.A. Effect of subinhibitory concentrations of benzalkonium chloride on the competitiveness of Pseudomonas aeruginosa grown in continuous culture. Microbiology 2010, 156, 30–38. [Google Scholar] [CrossRef]
- Fraud, S.; Campigotto, A.J.; Chen, Z.; Poole, K. MexCD-OprJ multidrug efflux system of Pseudomonas aeruginosa: Involvement in chlorhexidine resistance and induction by membrane-damaging agents dependent upon the AlgU stress response sigma factor. Antimicrob. Agents Chemother. 2008, 52, 4478–4482. [Google Scholar] [CrossRef]
- Nasr, A.M.; Mostafa, M.S.; Arnaout, H.H.; Elshimy, A.A.A. The effect of exposure to sub-inhibitory concentrations of hypochlorite and quaternary ammonium compounds on antimicrobial susceptibility of Pseudomonas aeruginosa. Am. J. Infect. Control 2018, 46, e57–e63. [Google Scholar] [CrossRef]
- Wand, M.E.; Bock, L.J.; Bonney, L.C.; Sutton, J.M. Mechanisms of increased resistance to chlorhexidine and cross-resistance to colistin following exposure of Klebsiella pneumoniae clinical isolates to chlorhexidine. Antimicrob. Agents Chemother. 2017, 61, e01162-16. [Google Scholar] [CrossRef] [PubMed]
- Potenski, C.J.; Gandhi, M.; Matthews, K.R. Exposure of Salmonella Enteritidis to chlorine or food preservatives increases susceptibility to antibiotics. FEMS Microbiol. Lett. 2003, 220, 181–186. [Google Scholar] [CrossRef]
- Obe, T.; Nannapaneni, R.; Sharma, C.S.; Kiess, A. Homologous stress adaptation, antibiotic resistance, and biofilm forming ability of Salmonella enterica serovar Heidelberg ATCC8326 on different food-contact surfaces following exposure to sublethal chlorine concentrations. Poult. Sci. 2018, 97, 951–961. [Google Scholar] [CrossRef]
- Templeton, M.R.; Oddy, F.; Leung, W.K.; Rogers, M. Chlorine and UV disinfection of ampicillin-resistant and trimethoprim-resistant Escherichia coli. Can. J. Civ. Eng. 2009, 36, 889–894. [Google Scholar] [CrossRef]
- Huang, J.J.; Hu, H.Y.; Wu, Y.H.; Wei, B.; Lu, Y. Effect of chlorination and ultraviolet disinfection on tetA-mediated tetracycline resistance of Escherichia coli. Chemosphere 2013, 90, 2247–2253. [Google Scholar] [CrossRef]
- Pang, Y.; Huang, J.; Xi, J.; Hu, H.; Zhu, Y. Effect of ultraviolet irradiation and chlorination on ampicillin-resistant Escherichia coli and its ampicillin resistance gene. Front. Environ. Sci. Eng. 2016, 10, 522–530. [Google Scholar] [CrossRef]
- Venieri, D.; Gounaki, I.; Bikouvaraki, M.; Binas, V.; Zachopoulos, A.; Kiriakidis, G.; Mantzavinos, D. Solar photocatalysis as disinfection technique: Inactivation of Klebsiella pneumoniae in sewage and investigation of changes in antibiotic resistance profile. J. Environ. Manag. 2017, 195, 140–147. [Google Scholar] [CrossRef]
- Hou, A.-M.; Yang, D.; Miao, J.; Shi, D.-Y.; Yin, J.; Yang, Z.-W.; Shen, Z.-Q.; Wang, H.-R.; Qiu, Z.-G.; Liu, W.-L.; et al. Chlorine injury enhances antibiotic resistance in Pseudomonas aeruginosa through over expression of drug efflux pumps. Water Res. 2019, 156, 366–371. [Google Scholar] [CrossRef]
- Nguyen, C.C.; Hugie, C.N.; Kile, M.L.; Navab-Daneshmand, T. Association between heavy metals and antibiotic-resistant human pathogens in environmental reservoirs: A review. Front. Environ. Sci. Eng. 2019, 13, 46. [Google Scholar] [CrossRef]
- Al-Nabulsi, A.A.; Osaili, T.M.; Elabedeen, N.A.Z.; Jaradat, Z.W.; Shaker, R.R.; Kheirallah, K.A.; Tarazi, Y.H.; Holley, R.A. Impact of environmental stress desiccation, acidity, alkalinity, heat or cold on antibiotic susceptibility of Cronobacter sakazakii. Int. J. Food Microbiol. 2011, 146, 137–143. [Google Scholar] [CrossRef]
- McMahon, M.A.S.; Xu, J.; Moore, J.E.; Blair, I.S.; McDowell, D.A. Environmental stress and antibiotic resistance in food-related pathogens. Appl. Environ. Microbiol. 2007, 73, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Al-Nabulsi, A.A.; Osaili, T.M.; Shaker, R.R.; Olaimat, A.N.; Jaradat, Z.W.; Zain Elabedeen, N.A.; Holley, R.A. Effects of osmotic pressure, acid, or cold stresses on antibiotic susceptibility of Listeria monocytogenes. Food Microbiol. 2015, 46, 154–160. [Google Scholar] [CrossRef]
- Komora, N.; Bruschi, C.; Magalhães, R.; Ferreira, V.; Teixeira, P. Survival of Listeria monocytogenes with different antibiotic resistance patterns to food-associated stresses. Int. J. Food Microbiol. 2017, 245, 79–87. [Google Scholar] [CrossRef]
- Ebinesh, A.; Vijaykumar, G.S.; Kiran, T.S. Exposure to stress minimizes the zone of antimicrobial action: A phenotypic demonstration with six Acinetobacter baumannii strains. MicroMedicine 2018, 6, 16–35. [Google Scholar] [CrossRef]
- Bikels-Goshen, T.; Landau, E.; Saguy, S.; Shapira, R. Staphylococcal strains adapted to epigallocathechin gallate (EGCG) show reduced susceptibility to vancomycin, oxacillin and ampicillin, increased heat tolerance, and altered cell morphology. Int. J. Food Microbiol. 2010, 138, 26–31. [Google Scholar] [CrossRef]
- Levinger, O.; Bikels-Goshen, T.; Landau, E.; Fichman, M.; Shapira, R. Epigallocatechin gallate induces upregulation of the two-component VraSR system by evoking a cell wall stress response in Staphylococcus aureus. Appl. Environ. Microbiol. 2012, 78, 7954–7959. [Google Scholar] [CrossRef] [PubMed]
- McMahon, M.A.S.; Tunney, M.M.; Moore, J.E.; Blair, I.S.; Gilpin, D.F.; McDowell, D.A. Changes in antibiotic susceptibility in staphylococci habituated to sub-lethal concentrations of tea tree oil (Melaleuca alternifolia). Lett. Appl. Microbiol. 2008, 47, 263–268. [Google Scholar] [CrossRef]
- Becerril, R.; Nerín, C.; Gómez-Lus, R. Evaluation of bacterial resistance to essential oils and antibiotics after exposure to oregano and cinnamon essential oils. Foodborne Pathog. Dis. 2012, 9, 699–705. [Google Scholar] [CrossRef]
- Johny, A.K.; Frye, J.G.; Donoghue, A.; Donoghue, D.J.; Porwollik, S.; McClelland, M.; Venkitanarayanan, K. Gene expression response of Salmonella enterica Serotype Enteritidis phage type 8 to subinhibitory concentrations of the plant-derived compounds trans-cinnamaldehyde and eugenol. Front. Microbiol. 2017, 8, 1828. [Google Scholar] [CrossRef]
- Myers, J.P.; Antoniou, M.N.; Blumberg, B.; Carroll, L.; Colborn, T.; Everett, L.G.; Hansen, M.; Landrigan, P.J.; Lanphear, B.P.; Mesnage, R.; et al. Concerns over use of glyphosate-based herbicides and risks associated with exposures: A consensus statement. Environ. Health 2016, 15, 19. [Google Scholar] [CrossRef]
- Pöppe, J.; Bote, K.; Merle, R.; Makarova, O.; Roesler, U. Minimum inhibitory concentration of glyphosate and a glyphosate-containing herbicide in Salmonella enterica isolates originating from different time periods, hosts, and serovars. Eur. J. Microbiol. Immunol. 2019, 9, 35–41. [Google Scholar] [CrossRef]
- Pöppe, J.; Bote, K.; Ramesh, A.; Murugaiyan, J.; Kuropka, B.; Kühl, M.; Johnston, P.; Roesler, U.; Makarova, O. Selection for resistance to a glyphosate-containing herbicide in Salmonella enterica does not result in a sustained activation of the tolerance response or increased cross-tolerance and cross-resistance to clinically important antibiotics. Appl. Environ. Microbiol. 2020, 86, e01204-20. [Google Scholar] [CrossRef]
- Lu, W.; Li, L.; Chen, M.; Zhou, Z.; Zhang, W.; Ping, S.; Yan, Y.; Wang, J.; Lin, M. Genome-wide transcriptional responses of Escherichia coli to glyphosate, a potent inhibitor of the shikimate pathway enzyme 5-enolpyruvylshikimate-3- phosphate synthase. Mol. Biosyst. 2013, 9, 522–530. [Google Scholar] [CrossRef]
- Kurenbach, B.; Gibson, P.S.; Hill, A.M.; Bitzer, A.S.; Silby, M.W.; Godsoe, W.; Heinemann, J.A. Herbicide ingredients change Salmonella enterica sv. Typhimurium and Escherichia coli antibiotic responses. Microbiology 2017, 163, 1791–1801. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.R.; Mortimer, D.; Rose, M.; Smith, F.; Steel, Z.; Panton, S. Recently listed Stockholm convention POPs: Analytical methodology, occurrence in food and dietary exposure. Sci. Total Environ. 2019, 678, 793–800. [Google Scholar] [CrossRef]
- Zhou, Q.; Wu, W.; Lin, C.; Liang, H.; Long, C.; Lv, F.; Pan, J.; Liu, Z.; Wang, B.; Yang, X.; et al. Occurrence and dietary exposure assessment of pentachlorophenol in livestock, poultry, and aquatic foods marketed in Guangdong Province, China: Based on food monitoring data from 2015 to 2018. J. Food Sci. 2021, 86, 1132–1143. [Google Scholar] [CrossRef] [PubMed]
- Muller, J.F.; Stevens, A.M.; Craig, J.; Love, N.G. Transcriptome analysis reveals that multidrug efflux genes are upregulated to protect Pseudomonas aeruginosa from pentachlorophenol stress. Appl. Environ. Microbiol. 2007, 73, 4550–4558. [Google Scholar] [CrossRef]
- Paul, D.; Chakraborty, R.; Mandal, S.M. Biocides and health-care agents are more than just antibiotics: Inducing cross to co-resistance in microbes. Ecotoxicol. Environ. Saf. 2019, 174, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Davin-Regli, A.; Pages, J.M. Cross-resistance between biocides and antimicrobials: An emerging question. OIE Rev. Sci. Tech. 2012, 31, 89–104. [Google Scholar] [CrossRef]
- Lu, J.; Jin, M.; Nguyen, S.H.; Mao, L.; Li, J.; Coin, L.J.M.; Yuan, Z.; Guo, J. Non-antibiotic antimicrobial triclosan induces multiple antibiotic resistance through genetic mutation. Environ. Int. 2018, 118, 257–265. [Google Scholar] [CrossRef]
- Beier, R.C.; Byrd, J.A.; Andrews, K.; Caldwell, D.; Crippen, T.L.; Anderson, R.C.; Nisbet, D.J. Disinfectant and antimicrobial susceptibility studies of the foodborne pathogen Campylobacter jejuni isolated from the litter of broiler chicken houses. Poult. Sci. 2021, 100, 1024–1033. [Google Scholar] [CrossRef]
- Dann, A.B.; Hontela, A. Triclosan: Environmental exposure, toxicity and mechanisms of action. J. Appl. Toxicol. 2011, 31, 285–311. [Google Scholar] [CrossRef]
- Zhang, Y.; Gu, A.Z.; He, M.; Li, D.; Chen, J. Subinhibitory concentrations of disinfectants promote the horizontal transfer of multidrug resistance genes within and across genera. Environ. Sci. Technol. 2017, 51, 570–580. [Google Scholar] [CrossRef]
- Pagès, J.M.; James, C.E.; Winterhalter, M. The porin and the permeating antibiotic: A selective diffusion barrier in Gram-negative bacteria. Nat. Rev. Microbiol. 2008, 6, 893–903. [Google Scholar] [CrossRef]
- Thorrold, C.A.; Letsoalo, M.E.; Dusé, A.G.; Marais, E. Efflux pump activity in fluoroquinolone and tetracycline resistant Salmonella and E. coli implicated in reduced susceptibility to household antimicrobial cleaning agents. Int. J. Food Microbiol. 2007, 113, 315–320. [Google Scholar] [CrossRef]
- Tkachenko, O.; Shepard, J.; Aris, V.M.; Joy, A.; Bello, A.; Londono, I.; Marku, J.; Soteropoulos, P.; Peteroy-Kelly, M.A. A triclosan-ciprofloxacin cross-resistant mutant strain of Staphylococcus aureus displays an alteration in the expression of several cell membrane structural and functional genes. Res. Microbiol. 2007, 158, 651–658. [Google Scholar] [CrossRef]
- Zhang, Y.; Gu, A.Z.; Cen, T.; Li, X.; He, M.; Li, D.; Chen, J. Sub-inhibitory concentrations of heavy metals facilitate the horizontal transfer of plasmid-mediated antibiotic resistance genes in water environment. Environ. Pollut. 2018, 237, 74–82. [Google Scholar] [CrossRef]
- Jutkina, J.; Marathe, N.P.; Flach, C.F.; Larsson, D.G.J. Antibiotics and common antibacterial biocides stimulate horizontal transfer of resistance at low concentrations. Sci. Total Environ. 2018, 616–617, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Karatzas, K.A.G.; Randall, L.P.; Webber, M.; Piddock, L.J.V.; Humphrey, T.J.; Woodward, M.J.; Coldham, N.G. Phenotypic and proteomic characterization of multiply antibiotic-resistant variants of Salmonella enterica serovar Typhimurium selected following exposure to disinfectants. Appl. Environ. Microbiol. 2008, 74, 1508–1516. [Google Scholar] [CrossRef] [PubMed]
- Chuanchuen, R.; Beinlich, K.; Hoang, T.T.; Becher, A.; Karkhoff-Schweizer, R.R.; Schweizer, H.P. Cross-resistance between triclosan and antibiotics in Pseudomonas aeruginosa is mediated by multidrug efflux pumps: Exposure of a susceptible mutant strain to triclosan selects nfxB mutants overexpressing MexCD-OprJ. Antimicrob. Agents Chemother. 2001, 45, 428–432. [Google Scholar] [CrossRef]
- Pumbwe, L.; Randall, L.P.; Woodward, M.J.; Piddock, L.J.V. Evidence for multiple-antibiotic resistance in Campylobacter jejuni not mediated by CmeB or CmeF. Antimicrob. Agents Chemother. 2005, 49, 1289–1293. [Google Scholar] [CrossRef] [PubMed]
- Hernández, A.; Ruiz, F.M.; Romero, A.; Martínez, J.L. The binding of triclosan to SmeT, the repressor of the multidrug efflux pump SmeDEF, induces antibiotic resistance in Stenotrophomonas maltophilia. PLoS Pathog. 2011, 7, e1002103. [Google Scholar] [CrossRef]
- Dodd, M.C. Potential impacts of disinfection processes on elimination and deactivation of antibiotic resistance genes during water and wastewater treatment. J. Environ. Monit. 2012, 14, 1754–1771. [Google Scholar] [CrossRef]
- Wang, H.; Wang, J.; Li, S.; Ding, G.; Wang, K.; Zhuang, T.; Huang, X.; Wang, X. Synergistic effect of UV/chlorine in bacterial inactivation, resistance gene removal, and gene conjugative transfer blocking. Water Res. 2020, 185, 116290. [Google Scholar] [CrossRef]
- Fiorentino, A.; Ferro, G.; Alferez, M.C.; Polo-López, M.I.; Fernández-Ibañez, P.; Rizzo, L. Inactivation and regrowth of multidrug resistant bacteria in urban wastewater after disinfection by solar-driven and chlorination processes. J. Photochem. Photobiol. B Biol. 2015, 148, 43–50. [Google Scholar] [CrossRef]
- Jin, M.; Liu, L.; Wang, D.-N.; Yang, D.; Liu, W.-L.; Yin, J.; Yang, Z.-W.; Wang, H.-R.; Qiu, Z.-G.; Shen, Z.-Q.; et al. Chlorine disinfection promotes the exchange of antibiotic resistance genes across bacterial genera by natural transformation. ISME J. 2020, 14, 1847–1856. [Google Scholar] [CrossRef]
- Squadrone, S. Water environments: Metal-tolerant and antibiotic-resistant bacteria. Environ. Monit. Assess. 2020, 192, 238. [Google Scholar] [CrossRef]
- Moore, B. A new screen test and selective medium for the rapid detection of epidemic strains of Staph. aureus. Lancet 1960, 276, 453–458. [Google Scholar] [CrossRef]
- Imran, M.; Das, K.R.; Naik, M.M. Co-selection of multi-antibiotic resistance in bacterial pathogens in metal and microplastic contaminated environments: An emerging health threat. Chemosphere 2019, 215, 846–857. [Google Scholar] [CrossRef]
- Bacon, R.T.; Sofos, J.N.; Kendall, P.A.; Belk, K.E.; Smith, G.C. Comparative analysis of acid resistance between susceptible and multi-antimicrobial-resistant Salmonella strains cultured under stationary-phase acid tolerance-inducing and noninducing conditions. J. Food Prot. 2003, 66, 732–740. [Google Scholar] [CrossRef]
- Hughes, M.K.; Yanamala, S.; Francisco, M.S.; Loneragan, G.H.; Miller, M.F.; Brashears, M.M. Reduction of multidrug-resistant and drug-susceptible Salmonella in ground beef and freshly harvested beef briskets after exposure to commonly used industry antimicrobial interventions. J. Food Prot. 2010, 73, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Faezi-Ghasemi, M.; Kazemi, S. Effect of sub-lethal environmental stresses on the cell survival and antibacterial susceptibility of Listeria monocytogenes PTCC1297. Zahedan J. Res. Med. Sci. 2014, 17, e1915. [Google Scholar]
- EFSA Panel on Food Contact Materials; Enzymes and Processing Aids (CEP); Silano, V.; Baviera, B.; Manuel, J.; Bolognesi, C.; Brüschweiler, B.J.; Chesson, A.; Cocconcelli, P.S.; Crebelli, R.; et al. Evaluation of the safety and efficacy of the organic acids lactic and acetic acids to reduce microbiological surface contamination on pork carcasses and pork cuts. EFSA J. 2018, 16, 5482. [Google Scholar] [CrossRef]
- Skandamis, P.N.; Stopforth, J.D.; Kendall, P.A.; Belk, K.E.; Scanga, J.A.; Smith, G.C.; Sofos, J.N. Modeling the effect of inoculum size and acid adaptation on growth/no growth interface of Escherichia coli O157:H7. Int. J. Food Microbiol. 2007, 120, 237–249. [Google Scholar] [CrossRef]
- Samelis, J.; Sofos, J.N.; Kendall, P.A.; Smith, G.C. Effect of acid adaptation on survival of Escherichia coli O157:H7 in meat decontamination washing fluids and potential effects of organic acid interventions on the microbial ecology of the meat plant environment. J. Food Prot. 2002, 65, 33–40. [Google Scholar] [CrossRef]
- Mani-López, E.; García, H.S.; López-Malo, A. Organic acids as antimicrobials to control Salmonella in meat and poultry products. Food Res. Int. 2012, 45, 713–721. [Google Scholar] [CrossRef]
- Alonso-Calleja, C.; Guerrero-Ramos, E.; Alonso-Hernando, A.; Capita, R. Adaptation and cross-adaptation of Escherichia coli ATCC 12806 to several food-grade biocides. Food Control 2015, 56, 86–94. [Google Scholar] [CrossRef]
- Lerma, L.L.; Benomar, N.; Valenzuela, A.S.; Muñoz, M.d.C.C.; Gálvez, A.; Abriouel, H. Role of EfrAB efflux pump in biocide tolerance and antibiotic resistance of Enterococcus faecalis and Enterococcus faecium isolated from traditional fermented foods and the effect of EDTA as EfrAB inhibitor. Food Microbiol. 2014, 44, 249–257. [Google Scholar] [CrossRef]
- Romero, J.L.; Grande Burgos, M.J.; Pérez-Pulido, R.; Gálvez, A.; Lucas, R. Resistance to antibiotics, biocides, preservatives and metals in bacteria isolated from seafoods: Co-selection of strains resistant or tolerant to different classes of compounds. Front. Microbiol. 2017, 8, 1650. [Google Scholar] [CrossRef] [PubMed]
- Gadea, R.; Glibota, N.; Pérez Pulido, R.; Gálvez, A.; Ortega, E. Effects of exposure to biocides on susceptibility to essential oils and chemical preservatives in bacteria from organic foods. Food Control 2017, 80, 176–182. [Google Scholar] [CrossRef]
- Ma, Y.; Lan, G.; Li, C.; Cambaza, E.M.; Liu, D.; Ye, X.; Chen, S.; Ding, T. Stress tolerance of Staphylococcus aureus with different antibiotic resistance profiles. Microb. Pathog. 2019, 133, 103549. [Google Scholar] [CrossRef]
- Jalal Uddin, M.; Jeon, G.; Ahn, J. Variability in the adaptive response of antibiotic-resistant Salmonella Typhimurium to environmental stresses. Microb. Drug Resist. 2019, 25, 182–192. [Google Scholar] [CrossRef]
- Capita, R. Variation in Salmonella resistance to poultry chemical decontaminants, based on serotype, phage type, and antibiotic resistance patterns. J. Food Prot. 2007, 70, 1835–1843. [Google Scholar] [CrossRef]
- McMahon, M.A.S.; Blair, I.S.; Moore, J.E.; Mc Dowell, D.A. The rate of horizontal transmission of antibiotic resistance plasmids is increased in food preservation-stressed bacteria. J. Appl. Microbiol. 2007, 103, 1883–1888. [Google Scholar] [CrossRef] [PubMed]
- Combarros-Fuertes, P.; Fresno, J.M.; Estevinho, M.M.; Sousa-Pimenta, M.; Tornadijo, M.E.; Estevinho, L.M. Honey: Another alternative in the fight against antibiotic-resistant bacteria? Antibiotics 2020, 9, 774. [Google Scholar] [CrossRef] [PubMed]
- Durán-Lara, E.F.; Valderrama, A.; Marican, A. Natural organic compounds for application in organic farming. Agriculture 2020, 10, 41. [Google Scholar] [CrossRef]
- Dubois-Brissonnet, F.; Naïtali, M.; Mafu, A.A.; Briandet, R. Induction of fatty acid composition modifications and tolerance to biocides in Salmonella enterica serovar Typhimurium by plant-derived terpenes. Appl. Environ. Microbiol. 2011, 77, 906–910. [Google Scholar] [CrossRef]
- Liu, X.; Shen, B.; Du, P.; Wang, N.; Wang, J.; Li, J.; Sun, A. Transcriptomic analysis of the response of Pseudomonas fluorescens to epigallocatechin gallate by RNA-seq. PLoS ONE 2017, 12, e0177938. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.F.; Rileya, T.V. Effects of Melaleuca alternifolia (tea tree) essential oil and the major monoterpene component terpinen-4-ol on the development of single- and multistep antibiotic resistance and antimicrobial susceptibility. Antimicrob. Agents Chemother. 2012, 56, 909–915. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.F.; Riley, T.V.; Nielsen, J.B. A review of the toxicity of Melaleuca alternifolia (tea tree) oil. Food Chem. Toxicol. 2006, 44, 616–625. [Google Scholar] [CrossRef]
- De Sá Silva, C.; de Figueiredo, H.M.; Stamford, T.L.M.; Silva, L.H.M. Inhibition of Listeria monocytogenes by Melaleuca alternifolia (tea tree) essential oil in ground beef. Int. J. Food Microbiol. 2019, 293, 79–86. [Google Scholar] [CrossRef]
- McMahon, M.A.S.; Blair, I.S.; Moore, J.E.; McDowell, D.A. Habituation to sub-lethal concentrations of tea tree oil (Melaleuca alternifolia) is associated with reduced susceptibility to antibiotics in human pathogens. J. Antimicrob. Chemother. 2006, 59, 125–127. [Google Scholar] [CrossRef] [PubMed]
- Fadli, M.; Chevalier, J.; Hassani, L.; Mezrioui, N.E.; Pagès, J.M. Natural extracts stimulate membrane-associated mechanisms of resistance in Gram-negative bacteria. Lett. Appl. Microbiol. 2014, 58, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Moken, M.C.; McMurry, L.M.; Levy, S.B. Selection of multiple-antibiotic-resistant (mar) mutants of Escherichia coli by using the disinfectant pine oil: Roles of the mar and acrAB loci. Antimicrob. Agents Chemother. 1997, 41, 2770–2772. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.; Seng, Z.J.; Kohli, G.S.; Yang, L.; Yuk, H.-G. Stress resistance development and genome-wide transcriptional response of Escherichia coli O157:H7 adapted to sublethal thymol, carvacrol, and trans-cinnamaldehyde. Appl. Environ. Microbiol. 2018, 84, e01616-18. [Google Scholar] [CrossRef]
- Brul, S.; Coote, P. Preservative agents in foods: Mode of action and microbial resistance mechanisms. Int. J. Food Microbiol. 1999, 50, 1–17. [Google Scholar] [CrossRef]
- Ibarra-Sánchez, L.A.; El-Haddad, N.; Mahmoud, D.; Miller, M.J.; Karam, L. Advances in nisin use for preservation of dairy products. J. Dairy Sci. 2020, 103, 2041–2052. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food & Drug Administration. Microorganisms & Microbial-Derived Ingredients Used in Food (Partial List). Available online: https://www.fda.gov/food/generally-recognized-safe-gras/microorganisms-microbial-derived-ingredients-used-food-partial-list (accessed on 10 April 2021).
- Gravesen, A.; Jydegaard Axelsen, A.M.; Mendes da Silva, J.; Hansen, T.B.; Knøchel, S. Frequency of bacteriocin resistance development and associated fitness costs in Listeria monocytogenes. Appl. Environ. Microbiol. 2002, 68, 756–764. [Google Scholar] [CrossRef]
- Kumar, S.; Devi, S.; Sood, S.K.; Kapila, S.; Narayan, K.S.; Shandilya, S. Antibiotic resistance and virulence genes in nisin-resistant Enterococcus faecalis isolated from raw buffalo milk modulate the innate functions of rat macrophages. J. Appl. Microbiol. 2019, 127, 897–910. [Google Scholar] [CrossRef] [PubMed]
- Martínez, B.; Rodríguez, A. Antimicrobial susceptibility of nisin resistant Listeria monocytogenes of dairy origin. FEMS Microbiol. Lett. 2005, 252, 67–72. [Google Scholar] [CrossRef][Green Version]
- Mantovani, H.C.; Russell, J.B. Nisin resistance of Streptococcus bovis. Appl. Environ. Microbiol. 2001, 67, 808–813. [Google Scholar] [CrossRef]
- Apolónio, J.; Faleiro, M.L.; Miguel, M.G.; Neto, L. No induction of antimicrobial resistance in Staphylococcus aureus and Listeria monocytogenes during continuous exposure to eugenol and citral. FEMS Microbiol. Lett. 2014, 354, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.R.; Domingues, F.C.; Ferreira, S. The influence of resveratrol adaptation on resistance to antibiotics, benzalkonium chloride, heat and acid stresses of Staphylococcus aureus and Listeria monocytogenes. Food Control 2017, 73, 1420–1425. [Google Scholar] [CrossRef]
- Chen, Y.; Wen, Q.; Chen, S.; Guo, D.; Xu, Y.; Liang, S.; Xia, X.; Yang, B.; Shi, C. Effect of thymoquinone on the resistance of Cronobacter sakazakii to environmental stresses and antibiotics. Food Control 2020, 109, 106944. [Google Scholar] [CrossRef]
- Monte, D.F.M.; Tavares, A.G.; Albuquerque, A.R.; Sampaio, F.C.; Oliveira, T.C.R.M.; Franco, O.L.; Souza, E.L.; Magnani, M. Tolerance response of multidrug-resistant Salmonella enterica strains to habituation to Origanum vulgare L. essential oil. Front. Microbiol. 2014, 5, 721. [Google Scholar] [CrossRef]
- Rodríguez-Verdugo, A.; Gaut, B.S.; Tenaillon, O. Evolution of Escherichia coli rifampicin resistance in an antibiotic-free environment during thermal stress. BMC Evol. Biol. 2013, 13, 50. [Google Scholar] [CrossRef]
- Taher, E.M.; Hemmatzadeh, F.; Aly, S.A.; Elesswy, H.A.; Petrovski, K.R. Molecular characterization of antimicrobial resistance genes on farms and in commercial milk with emphasis on the effect of currently practiced heat treatments on viable but nonculturable formation. J. Dairy Sci. 2020, 103, 9936–9945. [Google Scholar] [CrossRef]
- Delorme, M.M.; Guimarães, J.T.; Coutinho, N.M.; Balthazar, C.F.; Rocha, R.S.; Silva, R.; Margalho, L.P.; Pimentel, T.C.; Silva, M.C.; Freitas, M.Q.; et al. Ultraviolet radiation: An interesting technology to preserve quality and safety of milk and dairy foods. Trends Food Sci. Technol. 2020, 102, 146–154. [Google Scholar] [CrossRef]
- Guo, M.; Huang, J.; Hu, H.; Liu, W.; Yang, J. UV inactivation and characteristics after photoreactivation of Escherichia coli with plasmid: Health safety concern about UV disinfection. Water Res. 2012, 46, 4031–4036. [Google Scholar] [CrossRef]
- Zhang, G.; Li, W.; Chen, S.; Zhou, W.; Chen, J. Problems of conventional disinfection and new sterilization methods for antibiotic resistance control. Chemosphere 2020, 254, 126831. [Google Scholar] [CrossRef]
- Umar, M.; Roddick, F.; Fan, L. Moving from the traditional paradigm of pathogen inactivation to controlling antibiotic resistance in water—Role of ultraviolet irradiation. Sci. Total Environ. 2019, 662, 923–939. [Google Scholar] [CrossRef]
- Guo, M.T.; Yuan, Q.-B.; Yang, J. Distinguishing effects of ultraviolet exposure and chlorination on the horizontal transfer of antibiotic resistance genes in municipal wastewater. Environ. Sci. Technol. 2015, 49, 5771–5778. [Google Scholar] [CrossRef]
- Álvarez-Molina, A.; de Toro, M.; Ruiz, L.; López, M.; Prieto, M.; Alvarez-Ordóñez, A. Selection for antimicrobial resistance in foodborne pathogens through exposure to UV light and nonthermal atmospheric plasma decontamination techniques. Appl. Environ. Microbiol. 2020, 86. [Google Scholar] [CrossRef]
- Rizzo, L.; Fiorentino, A.; Anselmo, A. Advanced treatment of urban wastewater by UV radiation: Effect on antibiotics and antibiotic-resistant E. coli strains. Chemosphere 2013, 92, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.M.; Xu, L.M.; Wang, X.C.; Zhuang, K.; Liu, Q.Q. Effects of ultraviolet disinfection on antibiotic-resistant Escherichia coli from wastewater: Inactivation, antibiotic resistance profiles and antibiotic resistance genes. J. Appl. Microbiol. 2017, 123, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Hou, A.; Chen, T.; Yang, D.; Chen, Z.; Shen, Z.; Qiu, Z.; Yin, J.; Yang, Z.; Shi, D.; et al. Decreased antibiotic susceptibility in Pseudomonas aeruginosa surviving UV irradiation. Front. Microbiol. 2021, 12, 93. [Google Scholar] [CrossRef]
- Guo, M.T.; Yuan, Q.-B.; Yang, J. Ultraviolet reduction of erythromycin and tetracycline resistant heterotrophic bacteria and their resistance genes in municipal wastewater. Chemosphere 2013, 93, 2864–2868. [Google Scholar] [CrossRef] [PubMed]
- Sousa, J.M.; Macedo, G.; Pedrosa, M.; Becerra-Castro, C.; Castro-Silva, S.; Pereira, M.F.R.; Silva, A.M.T.; Nunes, O.C.; Manaia, C.M. Ozonation and UV254nm radiation for the removal of microorganisms and antibiotic resistance genes from urban wastewater. J. Hazard. Mater. 2017, 323, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, B.A.; Vance, C.C.; Gentry, T.J.; Karthikeyan, R. Effects of chlorination and ultraviolet light on environmental tetracycline-resistant bacteria and tet(W) in water. J. Environ. Chem. Eng. 2017, 5, 777–784. [Google Scholar] [CrossRef]
- Sanganyado, E.; Gwenzi, W. Antibiotic resistance in drinking water systems: Occurrence, removal, and human health risks. Sci. Total Environ. 2019, 669, 785–797. [Google Scholar] [CrossRef] [PubMed]
- Chu, L.; Chen, D.; Wang, J.; Yang, Z.; Yang, Q.; Shen, Y. Degradation of antibiotics and inactivation of antibiotic resistance genes (ARGs) in Cephalosporin C fermentation residues using ionizing radiation, ozonation and thermal treatment. J. Hazard. Mater. 2020, 382, 121058. [Google Scholar] [CrossRef]
- Arslan-Alaton, I.; Karatas, A.; Pehlivan, Ö.; Ucun, O.K.; Ölmez-Hancı, T. Effect of UV-A-assisted iron-based and UV-C-driven oxidation processes on organic matter and antibiotic resistance removal in tertiary treated urban wastewater. Catal. Today 2021, 361, 152–158. [Google Scholar] [CrossRef]
- Guo, L.; Xu, R.; Zhao, Y.; Liu, D.; Liu, Z.; Wang, X.; Chen, H.; Kong, M.G. Gas plasma pre-treatment increases antibiotic sensitivity and persister eradication in methicillin-resistant Staphylococcus aureus. Front. Microbiol. 2018, 9, 537. [Google Scholar] [CrossRef] [PubMed]
- Zuo, P.; Yu, P.; Alvarez, P.J.J. Beta-lactam-induced outer membrane alteration confers E. coli a fortuitous competitive advantage through cross-resistance to bacteriophages. Environ. Sci. Technol. Lett. 2020, 7, 428–433. [Google Scholar] [CrossRef]
- Breitbart, M.; Bonnain, C.; Malki, K.; Sawaya, N.A. Phage puppet masters of the marine microbial realm. Nat. Microbiol. 2018, 3, 754–766. [Google Scholar] [CrossRef]
- Clokie, M.R.J.; Millard, A.D.; Letarov, A.V.; Heaphy, S. Phages in nature. Bacteriophage 2011, 1, 31–45. [Google Scholar] [CrossRef]
- Lin, D.M.; Koskella, B.; Lin, H.C. Phage therapy: An alternative to antibiotics in the age of multi-drug resistance. World J. Gastrointest. Pharmacol. Ther. 2017, 8, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Fernández, L.; Gutiérrez, D.; Rodríguez, A.; García, P. Application of bacteriophages in the agro-food sector: A long way toward approval. Front. Cell. Infect. Microbiol. 2018, 8, 296. [Google Scholar] [CrossRef]
- Burmeister, A.R.; Fortier, A.; Roush, C.; Lessing, A.J.; Bender, R.G.; Barahman, R.; Grant, R.; Chan, B.K.; Turner, P.E. Pleiotropy complicates a trade-off between phage resistance and antibiotic resistance. Proc. Natl. Acad. Sci. USA 2020, 117, 11207–11216. [Google Scholar] [CrossRef]
- Chan, B.K.; Sistrom, M.; Wertz, J.E.; Kortright, K.E.; Narayan, D.; Turner, P.E. Phage selection restores antibiotic sensitivity in MDR Pseudomonas aeruginosa. Sci. Rep. 2016, 6, 26717. [Google Scholar] [CrossRef]
- Kim, M.; Jo, Y.; Hwang, Y.J.; Hong, H.W.; Hong, S.S.; Park, K.; Myung, H. Phage-antibiotic synergy via delayed lysis. Appl. Environ. Microbiol. 2018, 84, e02085-18. [Google Scholar] [CrossRef]
- Allen, R.C.; Pfrunder-Cardozo, K.R.; Meinel, D.; Egli, A.; Hall, A.R. Associations among antibiotic and phage resistance phenotypes in natural and clinical Escherichia coli isolates. MBio 2017, 8, e01341-17. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.J.; Kim, B.; Dawan, J.; Ding, T.; Kim, J.C.; Ahn, J. Assessment of antibiotic resistance in bacteriophage-insensitive Klebsiella pneumoniae. Microb. Pathog. 2019, 135, 103625. [Google Scholar] [CrossRef] [PubMed]
- Moulton-Brown, C.E.; Friman, V.-P. Rapid evolution of generalized resistance mechanisms can constrain the efficacy of phage-antibiotic treatments. Evol. Appl. 2018, 11, 1630–1641. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Gómez, C.; Blanco-Picazo, P.; Brown-Jaque, M.; Quirós, P.; Rodríguez-Rubio, L.; Cerdà-Cuellar, M.; Muniesa, M. Infectious phage particles packaging antibiotic resistance genes found in meat products and chicken feces. Sci. Rep. 2019, 9, 13281. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Kim, Y.; Ma, Q.; Hong, S.H.; Pokusaeva, K.; Sturino, J.M.; Wood, T.K. Cryptic prophages help bacteria cope with adverse environments. Nat. Commun. 2010, 1, 147. [Google Scholar] [CrossRef]
- Bertozzi Silva, J.; Storms, Z.; Sauvageau, D. Host receptors for bacteriophage adsorption. FEMS Microbiol. Lett. 2016, 363, fnw002. [Google Scholar] [CrossRef] [PubMed]
- German, G.J.; Misra, R. The TolC protein of Escherichia coli serves as a cell-surface receptor for the newly characterized TLS bacteriophage. J. Mol. Biol. 2001, 308, 579–585. [Google Scholar] [CrossRef]
- Parent, K.N.; Erb, M.L.; Cardone, G.; Nguyen, K.; Gilcrease, E.B.; Porcek, N.B.; Pogliano, J.; Baker, T.S.; Casjens, S.R. OmpA and OmpC are critical host factors for bacteriophage Sf6 entry in Shigella. Mol. Microbiol. 2014, 92, 47–60. [Google Scholar] [CrossRef]
- Gurney, J.; Pradier, L.; Griffin, J.S.; Gougat-Barbera, C.; Chan, B.K.; Turner, P.E.; Kaltz, O.; Hochberg, M.E. Phage steering of antibiotic-resistance evolution in the bacterial pathogen, Pseudomonas aeruginosa. Evol. Med. Public Health 2020, 2020, 148–157. [Google Scholar] [CrossRef]
- Hao, G.; Chen, A.I.; Liu, M.; Zhou, H.; Egan, M.; Yang, X.; Kan, B.; Wang, H.; Goulian, M.; Zhu, J. Colistin resistance-mediated bacterial surface modification sensitizes phage infection. Antimicrob. Agents Chemother. 2019, 63, e01609-19. [Google Scholar] [CrossRef] [PubMed]
- Broeker, N.K.; Barbirz, S. Not a barrier but a key: How bacteriophages exploit host’s O-antigen as an essential receptor to initiate infection. Mol. Microbiol. 2017, 105, 353–357. [Google Scholar] [CrossRef] [PubMed]
| Non-Antibiotic Antimicrobials | Bacterial Species | Antibiotics to which Susceptibility was Decreased | Bacterial Adaptation/Mechanism of Resistance | Reference |
|---|---|---|---|---|
| Agrochemicals | ||||
| Combination of three pesticides: captan, carbaryl, and malathion | S. aureus | Sulfamethazine | Not reported | [11] |
| Dicamba | E. coli/ S. Typhimurium | Chloramphenicol, ciprofloxacin, tetracycline/ Ampicillin, chloramphenicol, ciprofloxacin, tetracycline | [12] | |
| 2,4-dichlorophenoxyacetic acid | Ampicillin, ciprofloxacin/ Ampicillin, chloramphenicol, ciprofloxacin, tetracycline | Efflux pumps and induction of the soxRS regulon account for the change in susceptibility in E. coli. Dicamba plus chloramphenicol and Roundup plus kanamycin | ||
| Glyphosate (RoudUp) | Ciprofloxacin/ Ciprofloxacin, kanamycin | |||
| Mixture of 23 pesticides | E. coli | Streptomycin | Mutations associated with the antibiotic target | [13] |
| Biocides | ||||
| Sodium hypochlorite | E. coli | Spectinomycin, nalidixic acid, ampicillin-sulbactam | Increase in cell surface hydrophobicity and biofilm formation, changes in cell morphology and ultrastructure | [14] |
| Quaternary ammonium disinfectant or triclosan | S. Typhimurium | Chloramphenicol, ciprofloxacin, tetracycline, ampicillin | Overexpression of AcrAB efflux pump and reduction in outer membrane porins | [15] |
| Triclosan | E. coli | Ampicillin, ampicillin-sulbactam, cefazoline, cefaclor, cefotaxime, cefepime, erythromycin, azithromycin, gentamicin, chloramphenicol, tetracycline, ciprofloxacin, lomefloxacin, imipenem | Changes in bacterial membrane properties and enhancing the efflux system | [16] |
| Quaternary ammonium disinfectant | S. aureus | Fluoroquinolones | Increased expression of norA | [17] |
| Quaternary ammonium disinfectant (benzalkonium chloride) | L. monocytogenes | Cefotaxime, cephalothin, ciprofloxacin | Increased expression of MdrL efflux pump | [18] |
| Benzalkonium chloride or chlorhexidine | P. aeruginosa | Ciprofloxacin, novobiocin | Decrease in the expression of the repressor gene mexR and increase the activity of MexAB-OprM and MexCD-OprJ efflux pumps | [19,20] |
| Didecyldimonium chloride | P. aeruginosa | Colistin, ceftazidime, amikacin, meropenem, gentamicin, piperacillin-tazobactam, ciprofloxacin | Not reported | [21] |
| Sodium hypochlorite | P. aeruginosa | Amikacin, gentamicin, meropenem, ciprofloxacin | Not reported | [21] |
| Chlorhexidine | K. pneumoniae | Colistin | Mutations in Tet repressor gene (smvR) and up-regulation of the smvA gene, both involved in MFS efflux pump system; modification of LPS | [22] |
| Chlorine | Salmonella Enteritidis | Tetracycline, nalidixic acid, chloramphenicol | MarRAB operon and increased expression of efflux pumps | [23] |
| Chlorine | S. enterica serovar Heidelberg | Gentamicin, streptomycin, ampicillin, ciprofloxacin (adapted rugose); sulphamethoxazole/trimethoprim and streptomycin (adapted smoothly) | Not reported | [24] |
| Chlorine | E. coli | Trimethoprim | Not reported | [25] |
| Chlorine (>1.0 mg Cl2/L) | E. coli | Tetracycline | Not reported | [26] |
| Chlorine (2 mg/L) | E. coli | Ampicillin | Not reported | [27] |
| Chlorine (1 and 5 mg/L) | K. pneumoniae | Ampicillin | Not reported | [28] |
| Chlorine (4 and 8 mg/L) | P. aeruginosa | Ceftazidime, chloramphenicol, ampicillin | Not reported | [29] |
| Chlorine | P. aeruginosa | Amikacin, gentamicin | Not reported | [21] |
| Heavy Metals | ||||
| Cr Pb Cd Zn Cu | E. coli | Fluoroquinolone Vancomycin Quinolone Fluoroquinolone, ampicillin, cephalothin, and trimethoprim/sulfamethoxazole, vancomycin Ampicillin, cephalothin, trimethoprim/sulfamethoxazole | Not reported | [30] |
| Hg | Enterobacteriaceae | Various antibiotics (not specified) | Not reported | |
| Co, Cr, Cu, Hg, Ni, Zn Pb | Salmonella spp. | Penicillin Ampicillin, chloramphenicol, tetracycline | ||
| Ag, Cd, Cu, Ni, Pb, Zn Zn, Cu | P. aeruginosa | Aminoglycoside, amphenicol, macrolide, nitrofuran, penicillin, Quinolone, sulfonamide, tetracycline, trimethoprim/sulfamethoxazole; imipenem | Outer membrane proteins Co-regulation | |
| Cd | A. baumannii, Klebsiella spp., P. aeruginosa, Providencia spp, Proteus spp. | Penicillin, ampicillin | Not reported | |
| Hg | E. coli, Klebsiella spp., Shigella spp. | Tetracycline, Sulfamethoxazole/trimethoprim | Not reported | |
| Cu, Ni, Zn | Klebsiella spp., P. aeruginosa, Proteus spp. | Ampicillin, amoxicillin, tetracycline | Not reported | |
| Food preservatives and decontaminants | ||||
| Lactic acid | Cronobacter sakazakii | Neomycin, tetracycline, tilmicosin, florfenicol, Amoxicillin, ampicillin, vancomycin, ciprofloxacin, enrofloxacin | Not reported | [31] |
| Acidification with HCl | E. coli S. Typhimurium S. aureus | Amikacin, ceftriaxone, nalidixic acid Amikacin, ceftriaxone, trimethoprim Gentamicin, erythromycin | Not reported | [32] |
| Acetic acid, sodium benzoate, sodium nitrite | S. Enteritidis | Tetracycline | mar mutation | [23] |
| Trisodium phosphate | E. coli | Ampicillin | Not reported | [14] |
| Sodium nitrite | Spectinomycin, amikacin, kanamycin, streptomycin, cefazolin, cephalothin, cefotaxime, ceftazidime, cefepime, aztreonam, nalidixic acid, enrofloxacin, phosphomycin, nitrofurantoin | Increase in cell surface hydrophobicity and biofilm formation | ||
| Lactic acid (pH 6, 5.5, 5) | L. monocytogenes | Streptomycin, gentamicin, ampicillin, penicillin, ciprofloxacin, enrofloxacin | Not reported | [33] |
| Lactic acid (1%, pH 3.5) | L. monocytogenes | Ciprofloxacin, nitrofurantoin, erythromycin | Not reported | [34] |
| Sulphuric acid (pH 3, 5, 6) | Acinetobacter baumannii | Amikacin, piperacillin, tazobactam, imipenem, meropenem | Not reported | [35] |
| Natural Compounds | ||||
| Epigallocathechin gallate | S. epidermis, S. aureus | Vancomycin, oxacillin, ampicillin | Increased cell wall thickness, with a role of the two-component VraSR system | [36,37] |
| Melaleuca alternifolia oil | E. coli | Gentamicin, erythromycin, vancomycin, chloramphenicol, tetracycline, trimethoprim, mupirocin | Not reported | [38] |
| S. Enteritidis, S. Typhimurium | Gentamicin, chloramphenicol, tetracycline, streptomycin, trimethoprim, mupirocin | |||
| S. aureus | Gentamicin, vancomycin, chloramphenicol, trimethoprim, ampicillin, fusidic acid, mupirocin | |||
| Thymus marroccanus essential oil | E. coli | Chloramphenicol, nalidixic acid, tetracycline, erythromycin | Overexpression of AcrAB-tolC and decrease of the expression of outer membrane proteins | [39] |
| Pine oil | E. coli | Tetracycline, ampicillin, chloramphenicol | Overexpression of marA gene. | [40] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giacometti, F.; Shirzad-Aski, H.; Ferreira, S. Antimicrobials and Food-Related Stresses as Selective Factors for Antibiotic Resistance along the Farm to Fork Continuum. Antibiotics 2021, 10, 671. https://doi.org/10.3390/antibiotics10060671
Giacometti F, Shirzad-Aski H, Ferreira S. Antimicrobials and Food-Related Stresses as Selective Factors for Antibiotic Resistance along the Farm to Fork Continuum. Antibiotics. 2021; 10(6):671. https://doi.org/10.3390/antibiotics10060671
Chicago/Turabian StyleGiacometti, Federica, Hesamaddin Shirzad-Aski, and Susana Ferreira. 2021. "Antimicrobials and Food-Related Stresses as Selective Factors for Antibiotic Resistance along the Farm to Fork Continuum" Antibiotics 10, no. 6: 671. https://doi.org/10.3390/antibiotics10060671
APA StyleGiacometti, F., Shirzad-Aski, H., & Ferreira, S. (2021). Antimicrobials and Food-Related Stresses as Selective Factors for Antibiotic Resistance along the Farm to Fork Continuum. Antibiotics, 10(6), 671. https://doi.org/10.3390/antibiotics10060671







