Wireless Direct Microampere Current in Wound Healing: Clinical and Immunohistological Data from Two Single Case Reports
Abstract
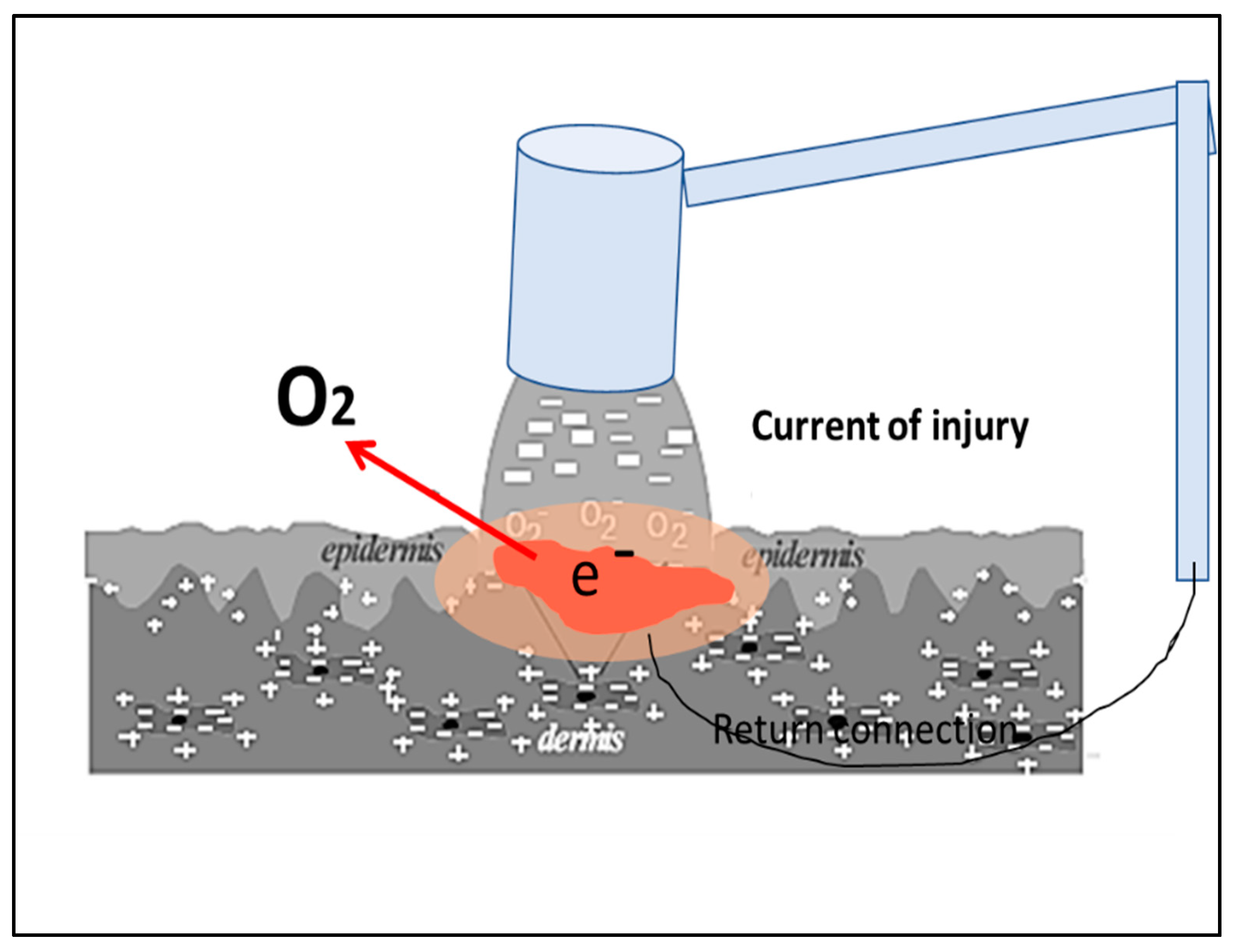
1. Introduction
2. Materials and Methods
2.1. Ethical Considerations
2.2. Study Participants—Cases Presentation
2.3. WMCS Device and Set-Up
2.4. Immunohistochemical Analysis of Biopsies
2.5. Hematoxylin and Eosin
2.6. Human CD117/c-Kit 17 Antigen
2.7. Masson’s Trichrome
2.8. Measurement of Wound Closure
3. Results
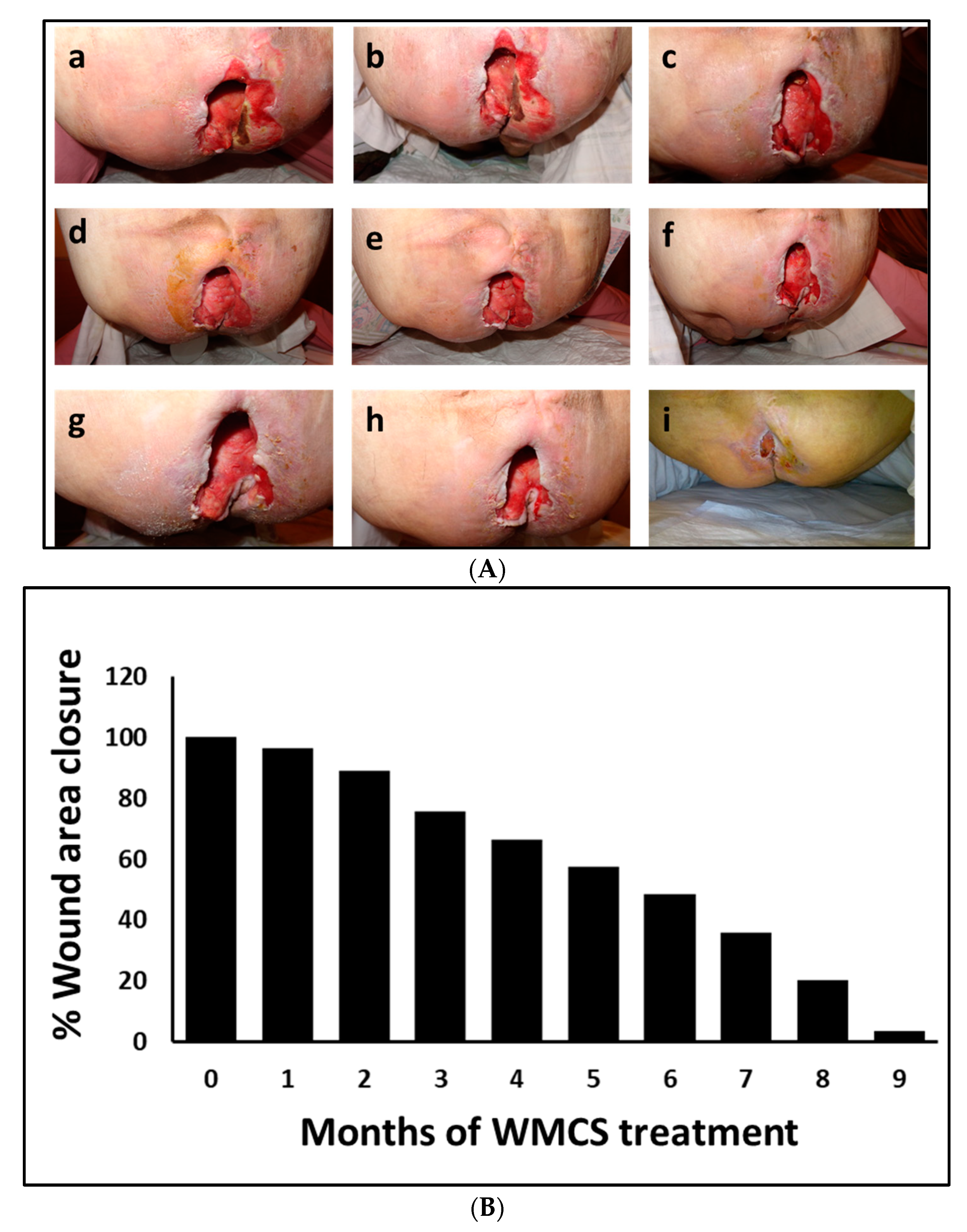
3.1. Clinical Outcome
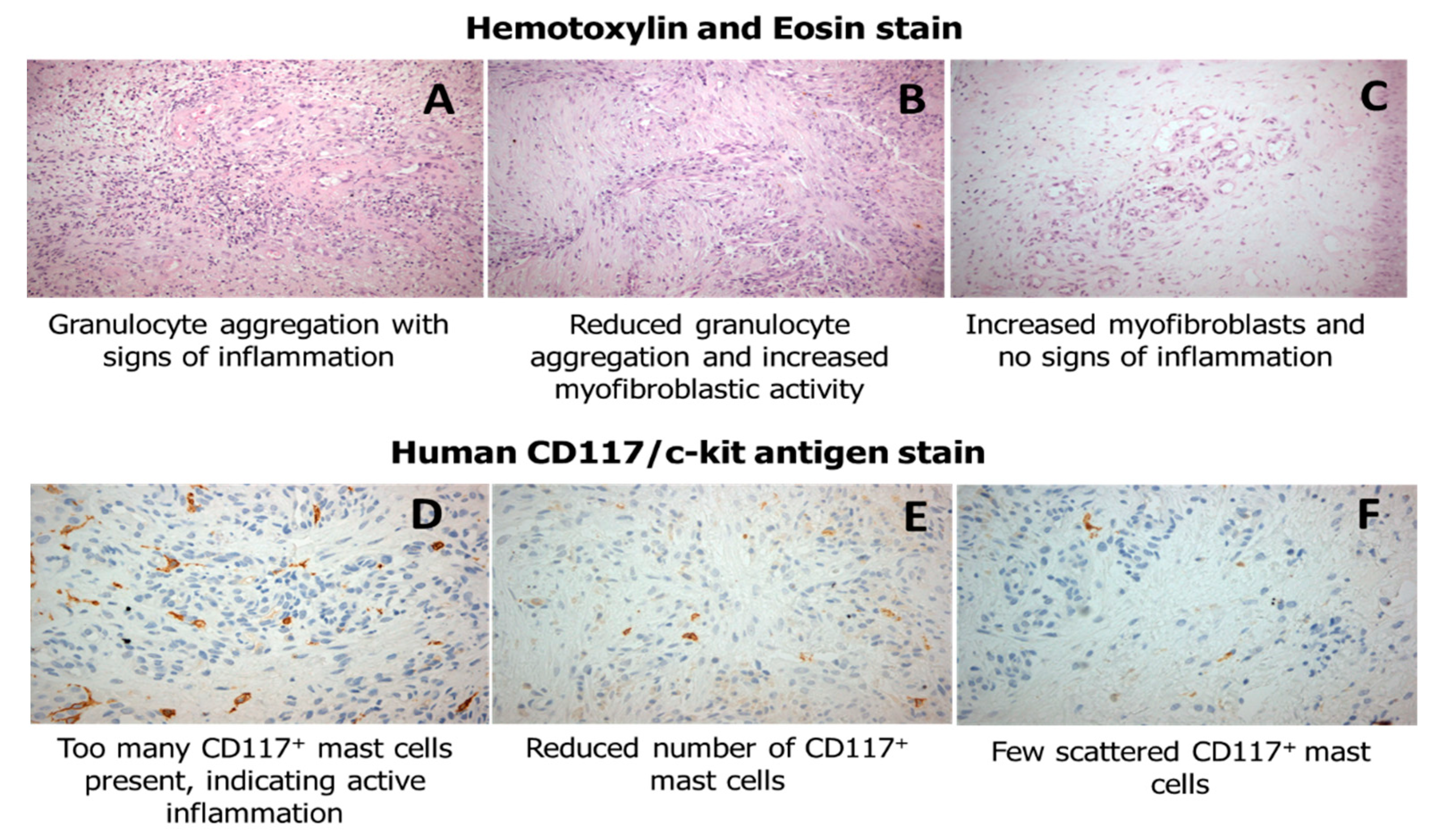
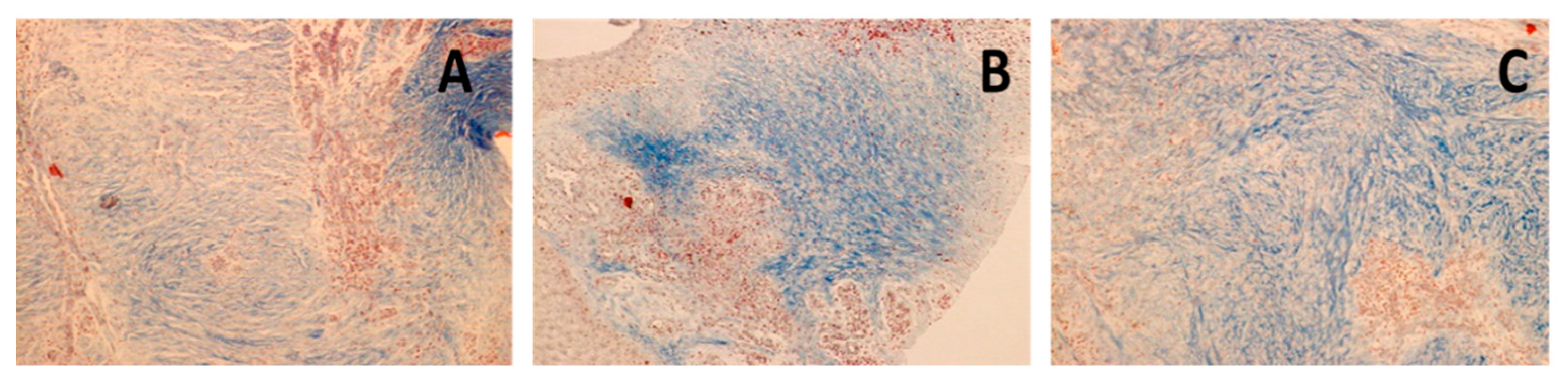
3.2. Immunohistochemical Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Avishai, E.; Yeghiazaryan, K.; Golubnitschaja, O. Impaired wound healing: Facts and hypotheses for multi-professional considerations in predictive, preventive and personalised medicine. EPMA J. 2017, 8, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; DiPietro, L.A. Factors affecting wound healing. J. Dent. Res. 2010, 89, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Augustin, M.; Brocatti, L.K.; Rustenbach, S.J.; Schäfer, I.; Herberger, K. Cost-of-illness of leg ulcers in the community. Int. Wound, J. 2014, 11, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Frykberg, R.G.; Banks, J. Challenges in the treatment of chronic wounds. Adv. Wound Care 2015, 4, 560–582. [Google Scholar] [CrossRef] [PubMed]
- Järbrink, K.; Ni, G.; Sönnergren, H.; Schmidtchen, A.; Pang, C.; Bajpai, R.; Car, J. The humanistic and economic burden of chronic wounds: A protocol for a systematic review. Syst. Rev. 2017, 6, 15. [Google Scholar] [CrossRef] [PubMed]
- Phillips, C.J.; Humphreys, I.; Fletcher, J.; Harding, K.; Chamberlain, G.; Macey, S. Estimating the costs associated with the management of patients with chronic wounds using linked routine data. Int. Wound J. 2016, 13, 1193–1197. [Google Scholar] [CrossRef] [PubMed]
- Posnett, J.; Franks, P.J. The burden of chronic wounds in the UK. Nurs. Times 2008, 104, 44–45. [Google Scholar] [PubMed]
- Agale, S.V. Chronic leg ulcers: Epidemiology, aetiopathogenesis, and management. Ulcers 2013, 2013, 1–9. [Google Scholar] [CrossRef]
- Gosain, A.; DiPietro, L.A. Aging and wound healing. World J. Surg. 2004, 28, 321–326. [Google Scholar] [CrossRef]
- Diegelmann, R.F.; Evans, M.C. Wound healing: An overview of acute, fibrotic and delayed healing. Front. Biosci. 2004, 9, 283–289. [Google Scholar] [CrossRef]
- Han, G.; Ceilley, R. Chronic wound healing: A review of current management and treatments. Adv. Ther. 2017, 34, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Korzendorfer, H.; Hettrick, H. Biophysical technologies for management of wound bioburden. Adv. Wound Care 2014, 3, 733. [Google Scholar] [CrossRef] [PubMed]
- Janssen, T.; Smit, C.; Hopman, M. Prevention and treatment of pressure ulcers using electrical stimulation. In Pressure Ulcer Research; Bader, D.L., Bouten, C., Colin, D., Oomens, C.W.J., Eds.; Springer-Verlag: Berlin/Heidelberg, Germany, 2005; pp. 89–107. ISBN 978-3-540-28804-6. [Google Scholar]
- Liu, L.Q.; Moody, J.; Traynor, M.; Dyson, S.; Gall, A. A systematic review of electrical stimulation for pressure ulcer prevention and treatment in people with spinal cord injuries. J. Spinal Cord Med. 2014, 37, 703–718. [Google Scholar] [CrossRef] [PubMed]
- Messerli, M.A.; Graham, D.M. Extracellular electrical fields direct wound healing and regeneration. Biol. Bull. 2011, 221, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Tai, G.; Tai, M.; Zhao, M. Electrically stimulated cell migration and its contribution to wound healing. Burn. Trauma 2018, 6, 20. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.; Zhao, M. Regulation of tissue repair and regeneration by electric fields. Chin. J. Traumatol. 2010, 13, 55–61. [Google Scholar] [PubMed]
- Kambouris, M.; Zagoriti, Z.; Lagoumintzis, G.; Poulas, K. From therapeutic electrotherapy to electroceuticals: Formats, applications and prospects of electrostimulation. Annu. Res. Rev. Biol. 2014, 4, 3054–3070. [Google Scholar] [CrossRef]
- Adunsky, A.; Ohry, A.; DDCT Group. Decubitus direct current treatment (DDCT) of pressure ulcers: Results of a randomized double-blinded placebo controlled study. Arch. Gerontol. Geriatr. 2005, 41, 261–269. [Google Scholar] [CrossRef]
- Guerriero, F.; Botarelli, E.; Mele, G.; Polo, L.; Zoncu, D.; Renati, P.; Sgarlata, C.; Rollone, M.; Ricevuti, G.; Maurizi, N.; et al. Effectiveness of an innovative pulsed electromagnetic fields stimulation in healing of untreatable skin ulcers in the frail elderly: Two case reports. Case Rep. Dermatol. Med. 2015, 2015, 1–6. [Google Scholar] [CrossRef]
- Lundeberg, T.C.; Eriksson, S.V.; Malm, M. Electrical nerve stimulation improves healing of diabetic ulcers. Ann. Plast. Surg. 1992, 29, 328–331. [Google Scholar] [CrossRef]
- Castana, O.; Dimitrouli, A.; Argyrakos, T.; Theodorakopoulou, E.; Stampolidis, N.; Papadopoulos, E.; Pallantzas, A.; Stasinopoulos, I.; Poulas, K. Wireless electrical stimulation. Int. J. Low. Extrem. Wounds 2013, 12, 18–21. [Google Scholar] [CrossRef] [PubMed]
- Ramadhinara, A.; Poulas, K. Use of wireless microcurrent stimulation for the treatment of diabetes-related wounds. Adv. Skin Wound Care 2013, 26, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Bansal, C.; Scott, R.; Stewart, D.; Cockerell, C.J. Decubitus ulcers: A review of the literature. Int. J. Dermatol. 2005, 44, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Prevention and Treatment of Pressure Ulcers: Quick Reference Guide; Cambridge Media: Cambridge, UK, 2009; ISBN 978-0-9579343-6-8.
- Bolton, L. Evidence corner: Evidence-based care for malignant wounds. Wounds Compend. Clin. Res. Pract. 2016, 28, 214–216. [Google Scholar]
- Moore, Z.E.; Webster, J.; Samuriwo, R. Wound-care teams for preventing and treating pressure ulcers. Cochrane Database Syst. Rev. 2015, 16, CD011011. [Google Scholar] [CrossRef] [PubMed]
- Reddy, M.; Gill, S.S.; Rochon, P.A. Preventing pressure ulcers: A systematic review. JAMA 2006, 296, 974. [Google Scholar] [CrossRef] [PubMed]
- Burdette-Taylor, S.R.; Kass, J. Heel ulcers in critical care units: A major pressure problem. Crit. Care Nurs. Q. 2002, 25, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Demarré, L.; Van Lancker, A.; Van Hecke, A.; Verhaeghe, S.; Grypdonck, M.; Lemey, J.; Annemans, L.; Beeckman, D. The cost of prevention and treatment of pressure ulcers: A systematic review. Int. J. Nurs. Stud. 2015, 52, 1754–1774. [Google Scholar] [CrossRef]
- Pattanshetty, R.B.; Prasade, P.M.; Aradhana, K.M. Risk assessment of decubitus ulcers using four scales among patients admitted in medical and surgical intensive care units in a tertiary care set up: A cross-sectional study. Int. J. Physiother. Res. 2015, 3, 971–977. [Google Scholar] [CrossRef]
- Reddy, M.; Gill, S.S.; Kalkar, S.R.; Wu, W.; Anderson, P.J.; Rochon, P.A. Treatment of pressure ulcers. JAMA 2008, 300, 2647. [Google Scholar] [CrossRef]
- Enoch, S.; Price, P.E. Cellular, molecular and biochemical differences in the pathophysiology of healing between acute wounds, chronic wounds and wounds in the aged. World Wide Wounds 2004, 2005, 12. [Google Scholar]
- Snyder, S.; DeJulius, C.; Willits, R.K. Electrical stimulation increases random migration of human dermal fibroblasts. Ann. Biomed. Eng. 2017, 45, 2049–2060. [Google Scholar] [CrossRef] [PubMed]
- Piccolino, M. Luigi Galvani’s path to animal electricity. C. R. Biol. 2006, 329, 303–318. [Google Scholar] [CrossRef] [PubMed]
- Finger, S.; Piccolino, M.; Stahnisch, F.W. Alexander von Humboldt: Galvanism, animal electricity, and self-experimentation part 2: The electric eel, animal electricity, and later years. J. Hist. Neurosci. 2013, 22, 327–352. [Google Scholar] [CrossRef]
- Foulds, I.S.; Barker, A.T. Human skin battery potentials and their possible role in wound healing. Br. J. Dermatol. 1983, 109, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Lagoumintzis, G.; Sideris, S.; Kampouris, M.; Koutosjannis, C.; Rennekampff, H.-O.; Poulas, K. Wireless Micro Current Stimulation Technology Improves Firework Burn Healing Clinical Applications of Wmcs Technology. In Transforming Healthcare Through Innovations in Mobile and Wireless Technologies, Proceedings of the 4th International Conference on Wireless Mobile Communication and Healthcare, Athens, Greece, 3–5 November 2014; ICST: South Portland, MM, USA, 2014. [Google Scholar]
- Wirsing, P.G.; Konstantakaki, M.; Poulas, K.A. Martorell’s ulcer successfully treated by wireless microcurrent stimulation technology. Adv. Skin Wound Care 2019, 32, 81–84. [Google Scholar] [CrossRef] [PubMed]
- Lala, D.; Spaulding, S.J.; Burke, S.M.; Houghton, P.E. Electrical stimulation therapy for the treatment of pressure ulcers in individuals with spinal cord injury: A systematic review and meta-analysis. Int. Wound J. 2016, 13, 1214–1226. [Google Scholar] [CrossRef] [PubMed]
- Smit, C.A.J.; de Groot, S.; Stolwijk-Swuste, J.M.; Janssen, T.W.J. Effects of electrical stimulation on risk factors for developing pressure ulcers in people with a spinal cord injury. Am. J. Phys. Med. Rehabil. 2016, 95, 535–552. [Google Scholar] [CrossRef]
- Khouri, C.; Kotzki, S.; Roustit, M.; Blaise, S.; Gueyffier, F.; Cracowski, J.-L. Hierarchical evaluation of electrical stimulation protocols for chronic wound healing: An effect size meta-analysis. Wound Repair Regen. 2017, 25, 883–891. [Google Scholar] [CrossRef]
- Thakral, G.; LaFontaine, J.; Najafi, B.; Talal, T.K.; Kim, P.; Lavery, L.A. Electrical stimulation to accelerate wound healing. Diabet. Foot Ankle 2013, 4, 22081. [Google Scholar] [CrossRef]
- Ud-Din, S.; Bayat, A. Electrical stimulation and cutaneous wound healing: A review of clinical evidence. Healthcare 2014, 2, 445–467. [Google Scholar] [CrossRef] [PubMed]
- Ashrafi, M.; Alonso-Rasgado, T.; Baguneid, M.; Bayat, A. The efficacy of electrical stimulation in lower extremity cutaneous wound healing: A systematic review. Exp. Dermatol. 2017, 26, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Kloth, L.C. Electrical stimulation technologies for wound healing. Adv. Wound Care 2014, 3, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Hunckler, J.; de Mel, A. A current affair: Electrotherapy in wound healing. J. Multidiscip. Healthc. 2017, 10, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Song, B.; Pu, J.; Wada, T.; Reid, B.; Tai, G.; Wang, F.; Guo, A.; Walczysko, P.; Gu, Y.; et al. Electrical signals control wound healing through phosphatidylinositol-3-OH kinase-γ and PTEN. Nature 2006, 442, 457–460. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lagoumintzis, G.; Zagoriti, Z.; Jensen, M.S.; Argyrakos, T.; Koutsojannis, C.; Poulas, K. Wireless Direct Microampere Current in Wound Healing: Clinical and Immunohistological Data from Two Single Case Reports. Biosensors 2019, 9, 107. https://doi.org/10.3390/bios9030107
Lagoumintzis G, Zagoriti Z, Jensen MS, Argyrakos T, Koutsojannis C, Poulas K. Wireless Direct Microampere Current in Wound Healing: Clinical and Immunohistological Data from Two Single Case Reports. Biosensors. 2019; 9(3):107. https://doi.org/10.3390/bios9030107
Chicago/Turabian StyleLagoumintzis, George, Zoi Zagoriti, Mogens S. Jensen, Theodoros Argyrakos, Constantinos Koutsojannis, and Konstantinos Poulas. 2019. "Wireless Direct Microampere Current in Wound Healing: Clinical and Immunohistological Data from Two Single Case Reports" Biosensors 9, no. 3: 107. https://doi.org/10.3390/bios9030107
APA StyleLagoumintzis, G., Zagoriti, Z., Jensen, M. S., Argyrakos, T., Koutsojannis, C., & Poulas, K. (2019). Wireless Direct Microampere Current in Wound Healing: Clinical and Immunohistological Data from Two Single Case Reports. Biosensors, 9(3), 107. https://doi.org/10.3390/bios9030107






