Accuracy of Continuous Glucose Monitoring before, during, and after Aerobic and Anaerobic Exercise in Patients with Type 1 Diabetes Mellitus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Procedures
2.3. Data and Statistical Analysis
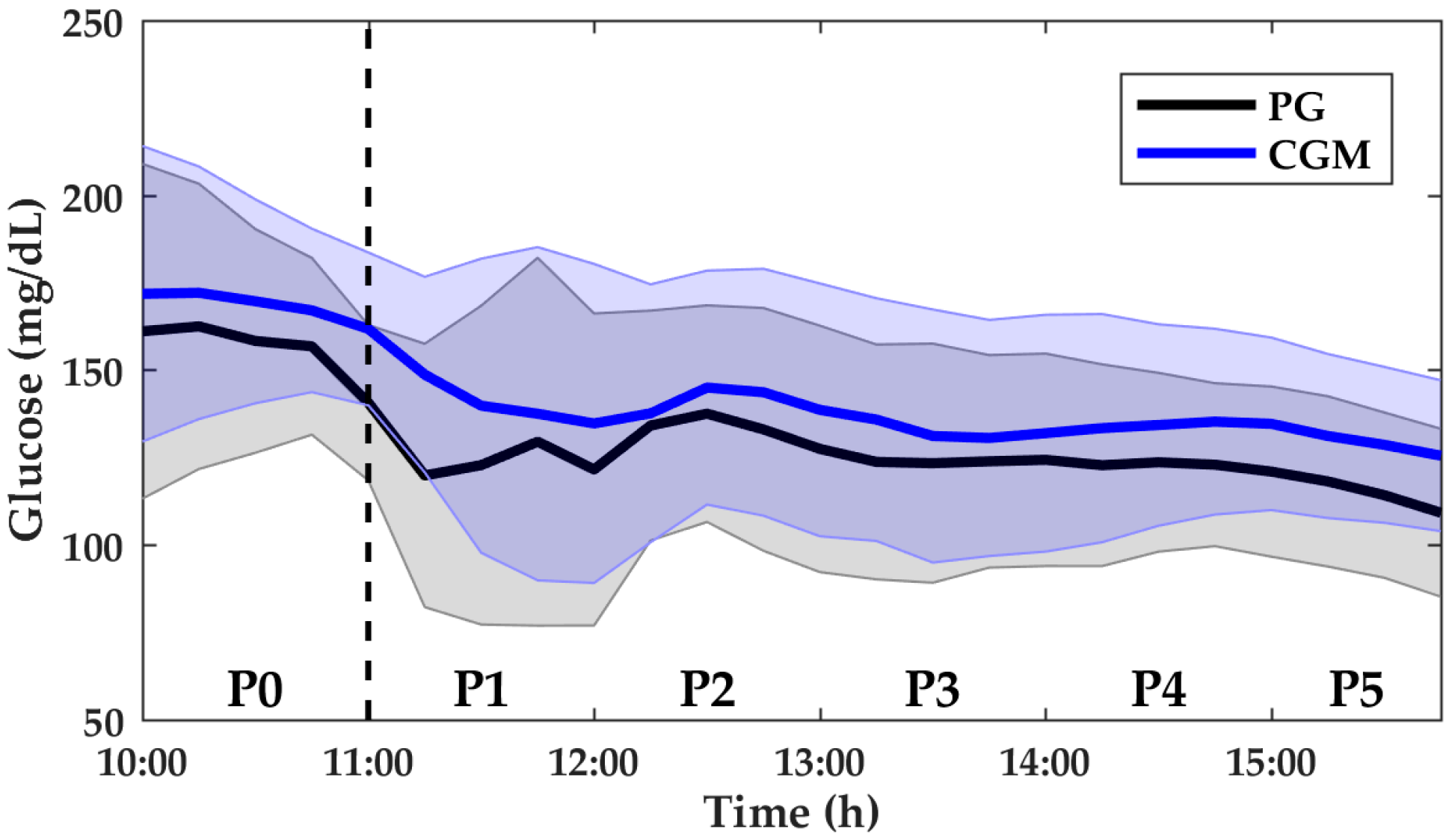
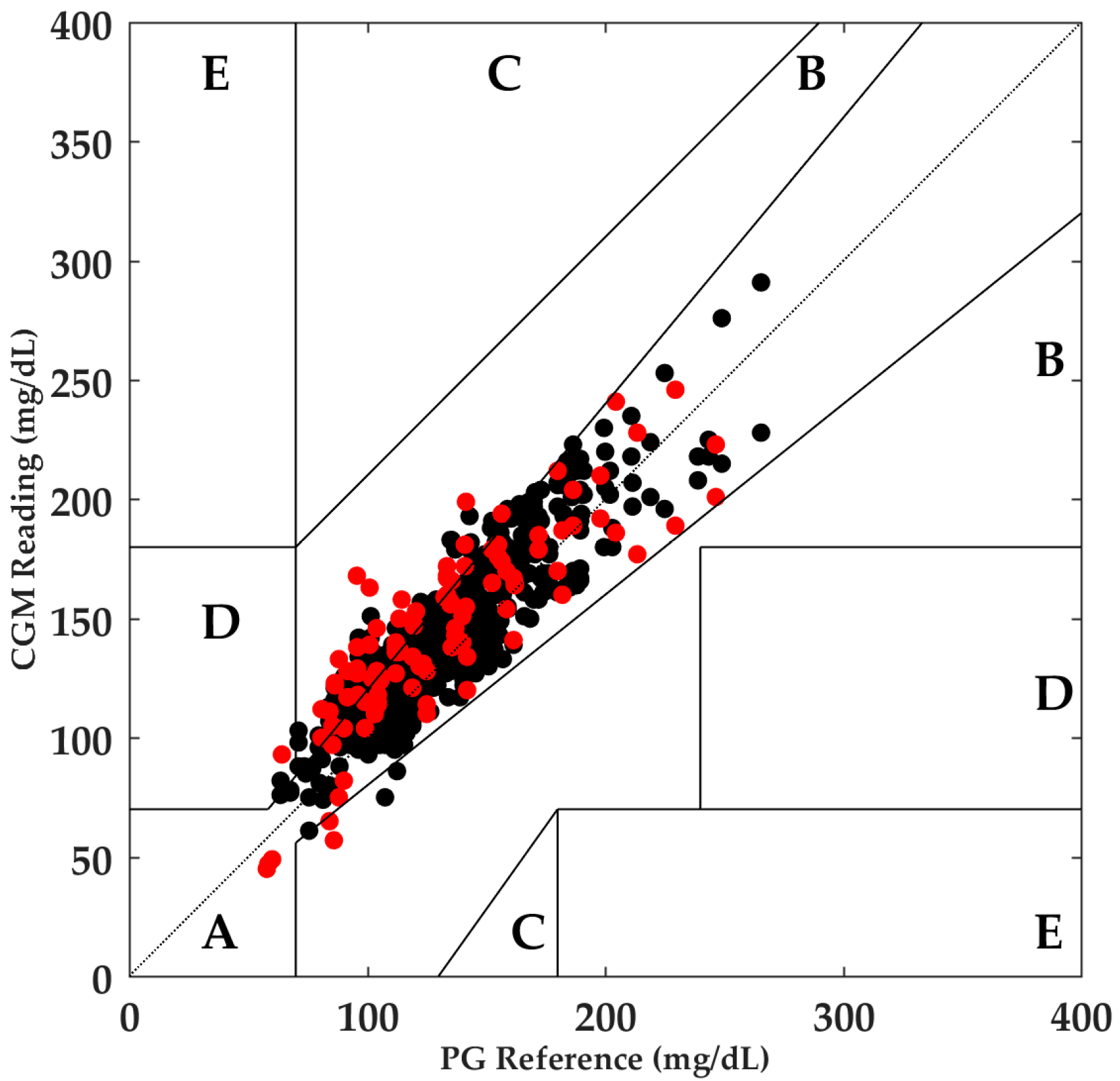
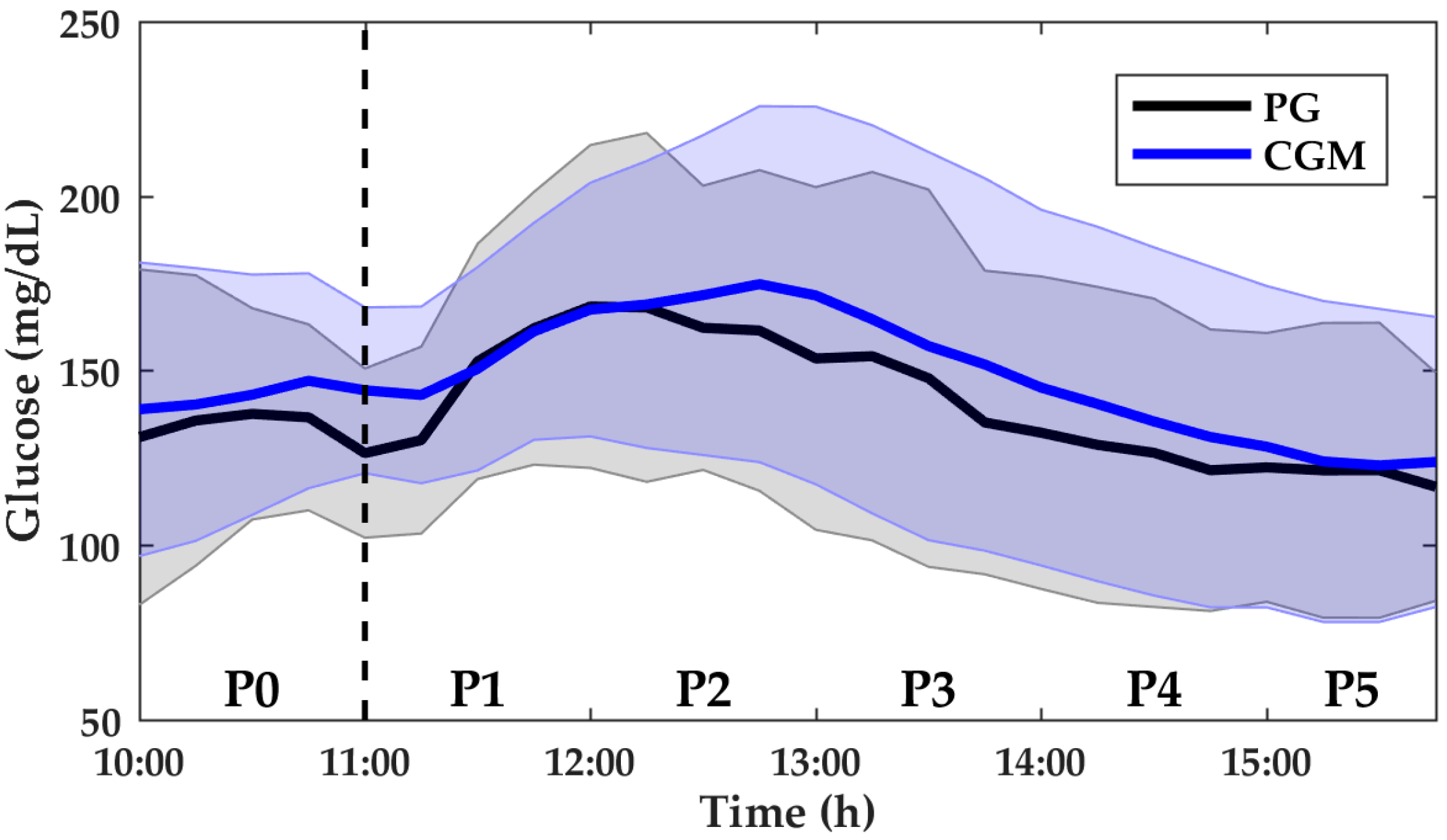
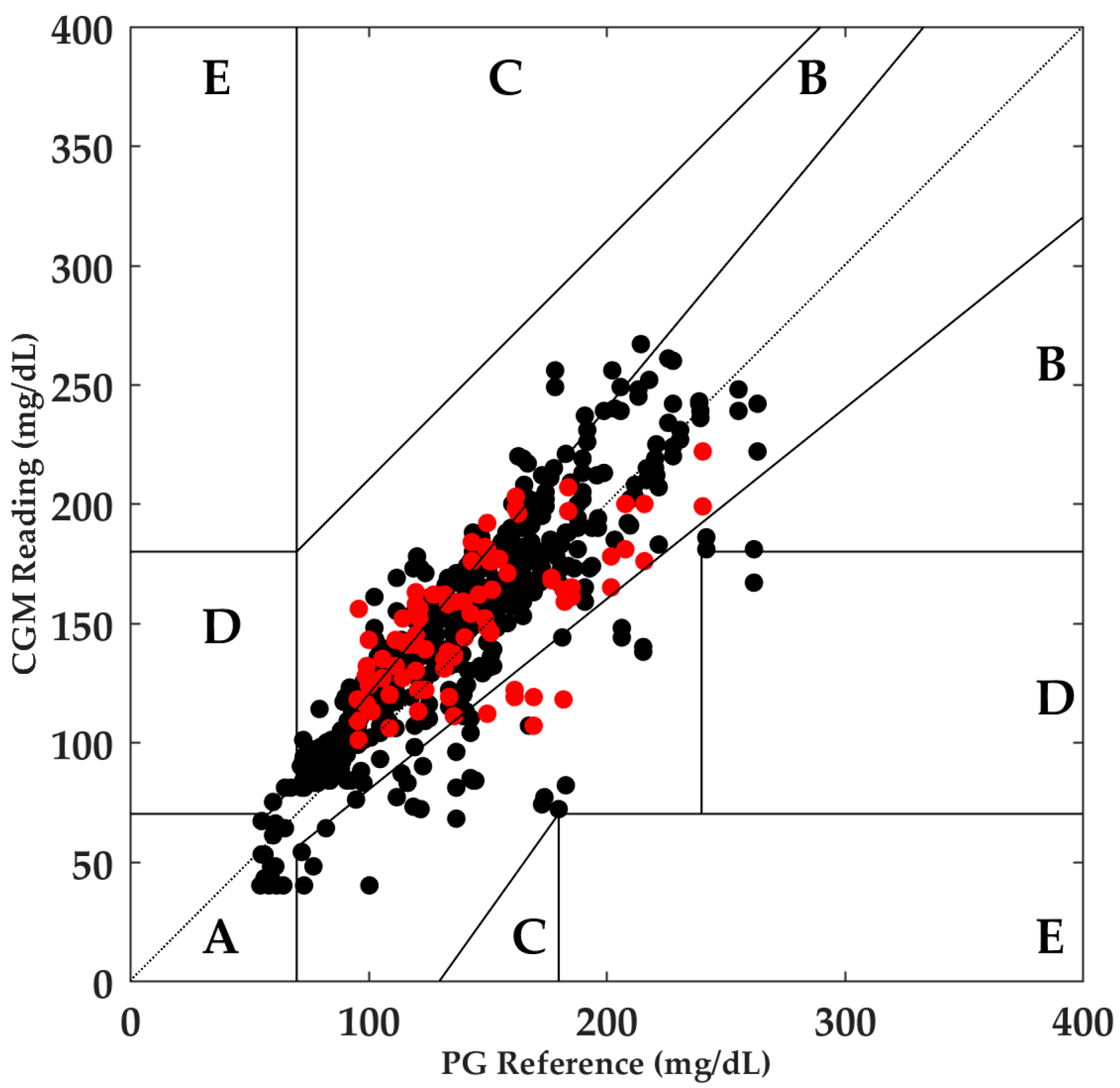
3. Results
3.1. Aerobic Exercise
3.2. Anaerobic Exercise
4. Discussion and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pickup, J.C.; Freeman, S.C.; Sutton, A.J. Glycaemic control in type 1 diabetes during real time continuous glucose monitoring compared with self monitoring of blood glucose: Meta-analysis of randomised controlled trials using individual patient data. BMJ 2011, 343, d3805. [Google Scholar] [CrossRef] [PubMed]
- Rodbard, D. Continuous Glucose Monitoring: A Review of Recent Studies Demonstrating Improved Glycemic Outcomes. Diabetes Technol. Ther. 2017, 19, S25–S37. [Google Scholar] [CrossRef] [PubMed]
- Beck, R.W.; Riddlesworth, T.; Ruedy, K.; Ahmann, A.; Bergenstal, R.; Haller, S.; Kollman, C.; Kruger, D.; McGill, J.; Polonsky, W.; et al. Effect of continuous glucose monitoring on glycemic control in adults with type 1 diabetes using insulin injections: The diamond randomized clinical trial. JAMA 2017, 317, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Colberg, S.R.; Sigal, R.J.; Yardley, J.E.; Riddell, M.C.; Dunstan, D.W.; Dempsey, P.C.; Horton, E.S.; Castorino, K.; Tate, D.F. Physical Activity/Exercise and Diabetes: A Position Statement of the American Diabetes Association. Diabetes Care 2016, 39, 2065–2079. [Google Scholar] [CrossRef] [PubMed]
- Riddell, M.C.; Gallen, I.W.; Smart, C.E.; Taplin, C.E.; Adolfsson, P.; Lumb, A.N.; Kowalski, A.; Rabasa-Lhoret, R.; McCrimmon, R.J.; Hume, C.; et al. Exercise management in type 1 diabetes: A consensus statement. Lancet Diabetes Endocrinol. 2017, 5, 377–390. [Google Scholar] [CrossRef]
- Doyle, F.J.; Huyett, L.M.; Lee, J.B.; Zisser, H.C.; Dassau, E. Closed-Loop Artificial Pancreas Systems: Engineering the Algorithms. Diabetes Care 2014, 37, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Garg, S.K.; Weinzimer, S.A.; Tamborlane, W.V.; Buckingham, B.A.; Bode, B.W.; Bailey, T.S.; Brazg, R.L.; Ilany, J.; Slover, R.H.; Anderson, S.M.; et al. Glucose Outcomes with the In-Home Use of a Hybrid Closed-Loop Insulin Delivery System in Adolescents and Adults with Type 1 Diabetes. Diabetes Technol. Ther. 2017, 19, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Riddell, M.C.; Zaharieva, D.P.; Yavelberg, L.; Cinar, A.; Jamnik, V.K. Exercise and the Development of the Artificial Pancreas: One of the More Difficult Series of Hurdles. J. Diabetes Sci. Technol. 2015, 9, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Kumareswaran, K.; Elleri, D.; Allen, J.M.; Caldwell, K.; Nodale, M.; Wilinska, M.E.; Amiel, S.A.; Hovorka, R.; Murphy, H.R. Accuracy of Continuous Glucose Monitoring During Exercise in Type 1 Diabetes Pregnancy. Diabetes Technol. Ther. 2013, 15, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Biagi, L.; Bertachi, A.H.; Conget, I.; Quirós, C.; Giménez, M.; Ampudia-Blasco, F.J.; Rossetti, P.; Bondia, J.; Vehí, J. Extensive Assessment of Blood Glucose Monitoring during Postprandial Period and Its Impact on Closed-Loop Performance. J. Diabetes Sci. Technol. 2017, 11, 1089–1095. [Google Scholar] [CrossRef] [PubMed]
- Taleb, N.; Emami, A.; Suppere, C.; Messier, V.; Legault, L.; Chiasson, J.-L.; Rabasa-Lhoret, R.; Haidar, A. Comparison of Two Continuous Glucose Monitoring Systems, Dexcom G4 Platinum and Medtronic Paradigm Veo Enlite System, at Rest and During Exercise. Diabetes Technol. Ther. 2016, 18, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Bally, L.; Zueger, T.; Pasi, N.; Carlos, C.; Paganini, D.; Stettler, C. Accuracy of continuous glucose monitoring during differing exercise conditions. Diabetes Res. Clin. Pract. 2016, 112, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Moser, O.; Mader, J.; Tschakert, G.; Mueller, A.; Groeschl, W.; Pieber, T.; Koehler, G.; Messerschmidt, J.; Hofmann, P. Accuracy of Continuous Glucose Monitoring (CGM) during Continuous and High-Intensity Interval Exercise in Patients with Type 1 Diabetes Mellitus. Nutrients 2016, 8, 489. [Google Scholar] [CrossRef] [PubMed]
- Kropff, J.; Bruttomesso, D.; Doll, W.; Farret, A.; Galasso, S.; Luijf, Y.M.; Mader, J.K.; Place, J.; Boscari, F.; Pieber, T.R.; et al. Accuracy of two continuous glucose monitoring systems: A head-to-head comparison under clinical research centre and daily life conditions. Diabetes Obes. Metab. 2015, 17, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Clarke, W.L.; Cox, D.; Gonder-Frederick, L.A.; Carter, W.; Pohl, S.L. Evaluating Clinical Accuracy of Systems for Self-Monitoring of Blood Glucose. Diabetes Care 1987, 10, 622–628. [Google Scholar] [CrossRef] [PubMed]
| Number of Patients (Females) | 6 (1) |
|---|---|
| Age (years) * | 36.7 ± 8.9 |
| HbA1C (%) * | 7.9 ± 0.5 |
| Body mass index (kg/m2) * | 24.6 ± 1.0 |
| Time with T1D (years) * | 25.2 ± 12.7 |
| Time with pump (years) * | 4.8 ± 1.7 |
| P0 | P1 | P2 | P3 | P4 | P5 | |
|---|---|---|---|---|---|---|
| PG (mg/dL) | 155.0 (135.3–174.5) | 120.5 (99.5–149.9) | 124.0 (106.8–155.8) | 114.5 (101.3–146.3) | 118.3 (104.5–143.8) | 111.5 (101.0–129.8) |
| MARD (%) | 9.5 (4.7–13.9) | 16.5 (7.6–23.5) | 9.3 (5.4–16.3) | 11.6 (6.5–17.5) | 11.3 (6.2–16.0) | 12.9 (4.7–18.8) |
| n | 112 | 108 | 108 | 108 | 108 | 108 |
| P0 | P1 | P2 | P3 | P4 | P5 | |
|---|---|---|---|---|---|---|
| P0 | – | <0.001 | 0.986 | 0.177 | 0.281 | 0.060 |
| P1 | <0.001 | – | <0.001 | <0.01 | <0.001 | <0.05 |
| P2 | 0.986 | <0.001 | – | 0.189 | 0.346 | 0.075 |
| P3 | 0.177 | <0.01 | 0.189 | – | 0.683 | 0.452 |
| P4 | 0.281 | <0.001 | 0.346 | 0.683 | – | 0.241 |
| P5 | 0.060 | <0.05 | 0.075 | 0.452 | 0.241 | – |
| P0 | P1 | P2 | P3 | P4 | P5 | |
|---|---|---|---|---|---|---|
| PG (mg/dL) | 124.5 (110.8–155.8) | 138.0 (118.3–160.0) | 157.5 (139.5–195.0) | 149.3 (108.5–179.8) | 138.5 (84.5–159.0) | 129.8 (88.3–150.8) |
| MARD (%) | 15.5 (6.5–26.4) | 16.8 (7.9–24.5) | 12.7 (4.9–20.3) | 14.3 (4.8–26.5) | 14.3 (7.9–19.7) | 12.3 (5.7–18.8) |
| n | n = 76 | n = 86 | n = 88 | n = 88 | n = 89 | n = 88 |
| P0 | P1 | P2 | P3 | P4 | P5 | |
|---|---|---|---|---|---|---|
| P0 | – | 0.993 | 0.095 | 0.709 | 0.287 | <0.05 |
| P1 | 0.993 | – | 0.063 | 0.798 | 0.171 | <0.05 |
| P2 | 0.095 | 0.063 | – | 0.188 | 0.444 | 0.691 |
| P3 | 0.709 | 0.798 | 0.188 | – | 0.458 | 0.076 |
| P4 | 0.287 | 0.171 | 0.444 | 0.458 | – | 0.223 |
| P5 | <0.05 | <0.05 | 0.691 | 0.076 | 0.223 | – |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Biagi, L.; Bertachi, A.; Quirós, C.; Giménez, M.; Conget, I.; Bondia, J.; Vehí, J. Accuracy of Continuous Glucose Monitoring before, during, and after Aerobic and Anaerobic Exercise in Patients with Type 1 Diabetes Mellitus. Biosensors 2018, 8, 22. https://doi.org/10.3390/bios8010022
Biagi L, Bertachi A, Quirós C, Giménez M, Conget I, Bondia J, Vehí J. Accuracy of Continuous Glucose Monitoring before, during, and after Aerobic and Anaerobic Exercise in Patients with Type 1 Diabetes Mellitus. Biosensors. 2018; 8(1):22. https://doi.org/10.3390/bios8010022
Chicago/Turabian StyleBiagi, Lyvia, Arthur Bertachi, Carmen Quirós, Marga Giménez, Ignacio Conget, Jorge Bondia, and Josep Vehí. 2018. "Accuracy of Continuous Glucose Monitoring before, during, and after Aerobic and Anaerobic Exercise in Patients with Type 1 Diabetes Mellitus" Biosensors 8, no. 1: 22. https://doi.org/10.3390/bios8010022
APA StyleBiagi, L., Bertachi, A., Quirós, C., Giménez, M., Conget, I., Bondia, J., & Vehí, J. (2018). Accuracy of Continuous Glucose Monitoring before, during, and after Aerobic and Anaerobic Exercise in Patients with Type 1 Diabetes Mellitus. Biosensors, 8(1), 22. https://doi.org/10.3390/bios8010022






