The Emergence of Insect Odorant Receptor-Based Biosensors
Abstract
1. Introduction
2. Development of Bioelectronic Sensing
2.1. Bioelectronic VOC Sensing
2.2. Cell-Based Bioelectronic Nose
2.3. OR-Based Bioelectronic Nose
2.4. The Advantages of Insect ORs as Biorecognition Elements
3. OR-Based Biosensors
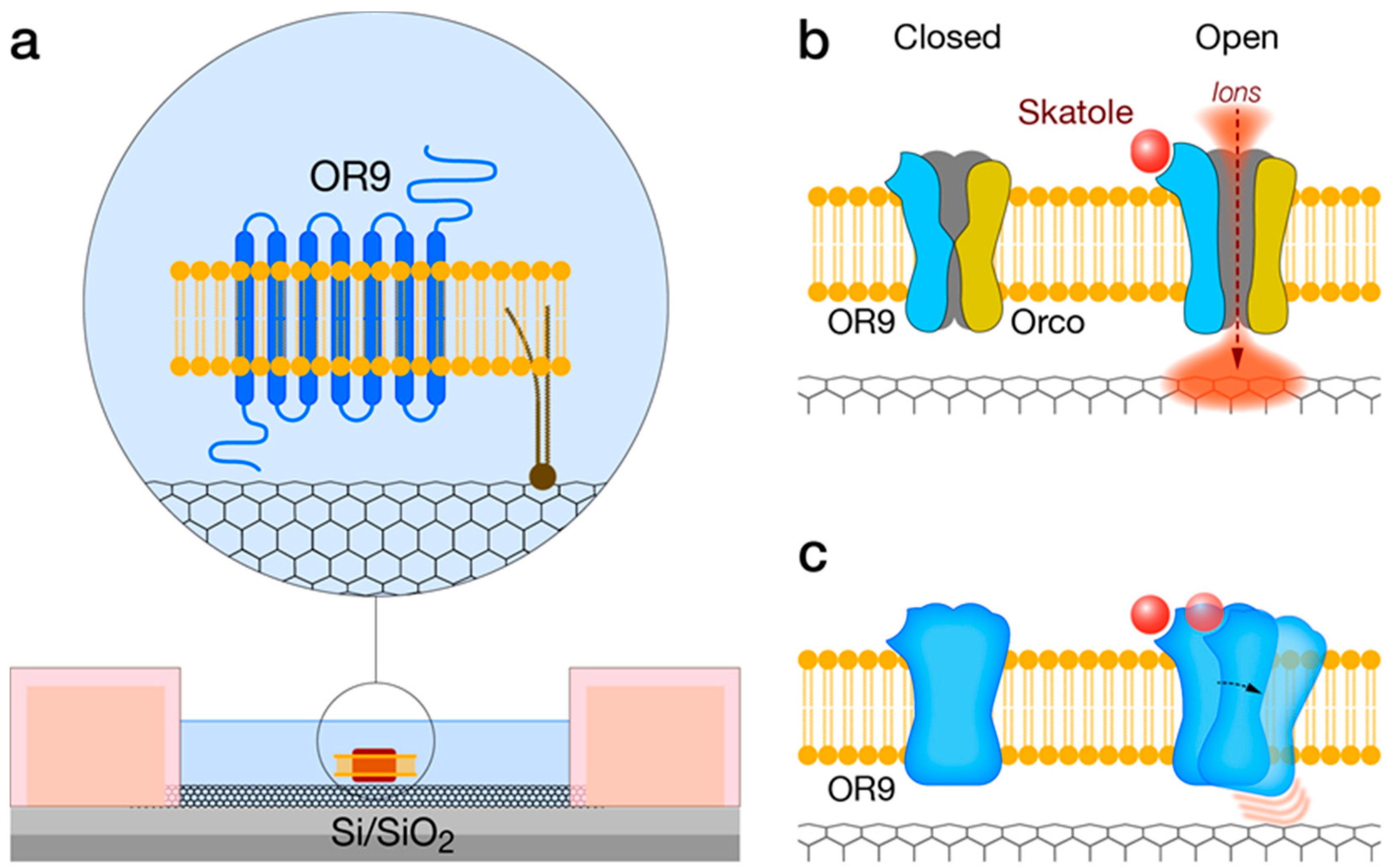
3.1. Solid-State—OR Interfacing
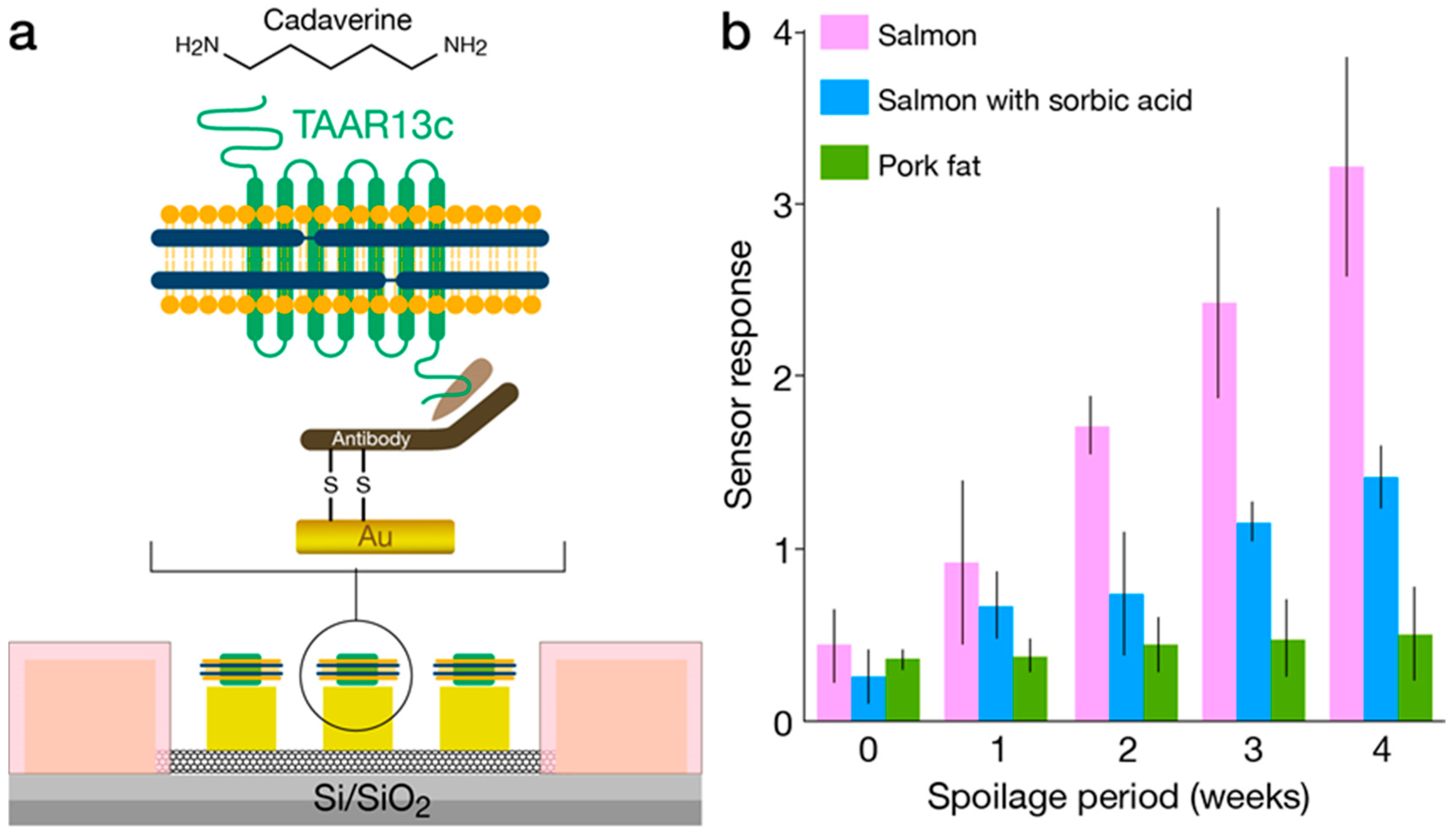
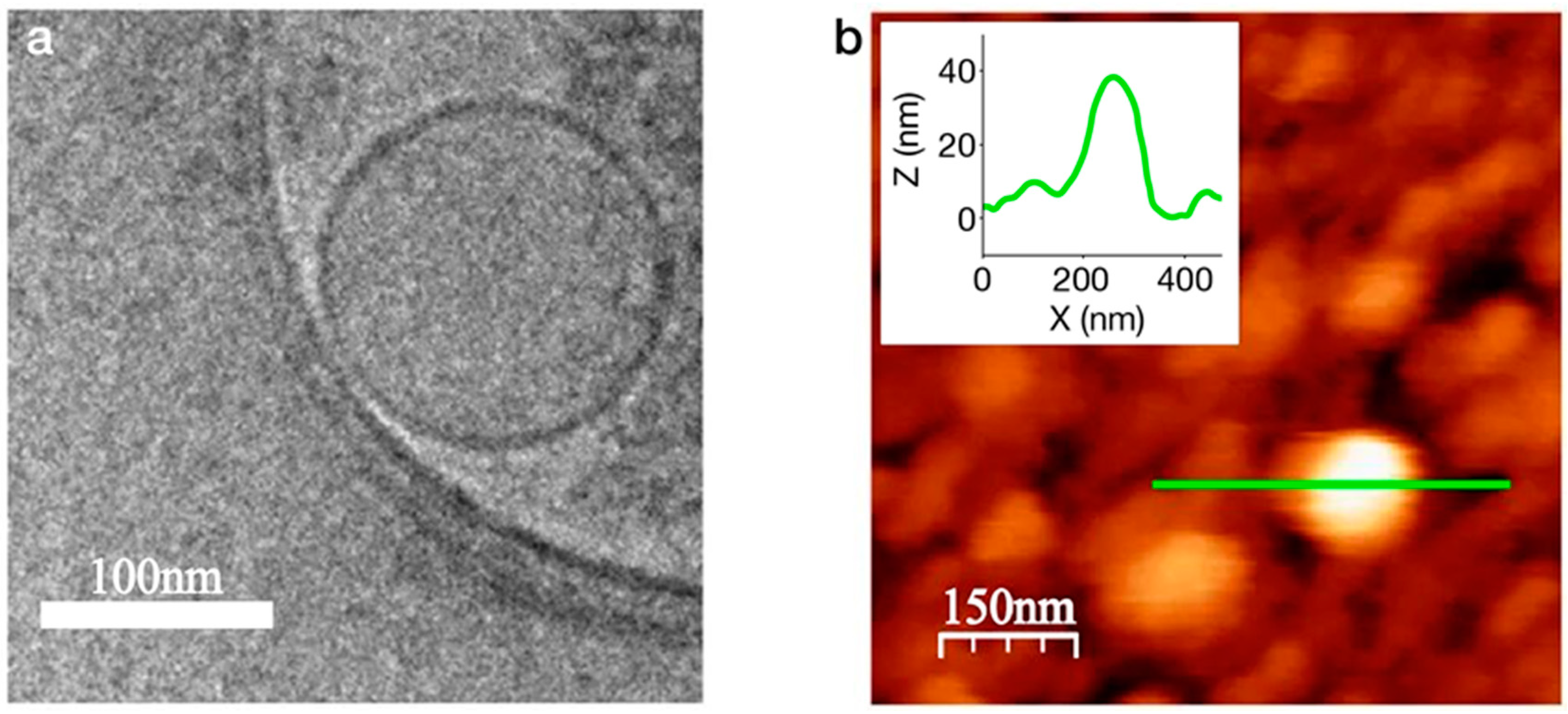
3.2. Nanodiscs
3.3. Nanovesicles
3.4. Suspended Lipid Bilayers
3.5. Immobilization Methods
3.6. Chemo-Electronic Signal Transduction
3.7. Phase Transfer in the Electrical Detection of VOCs
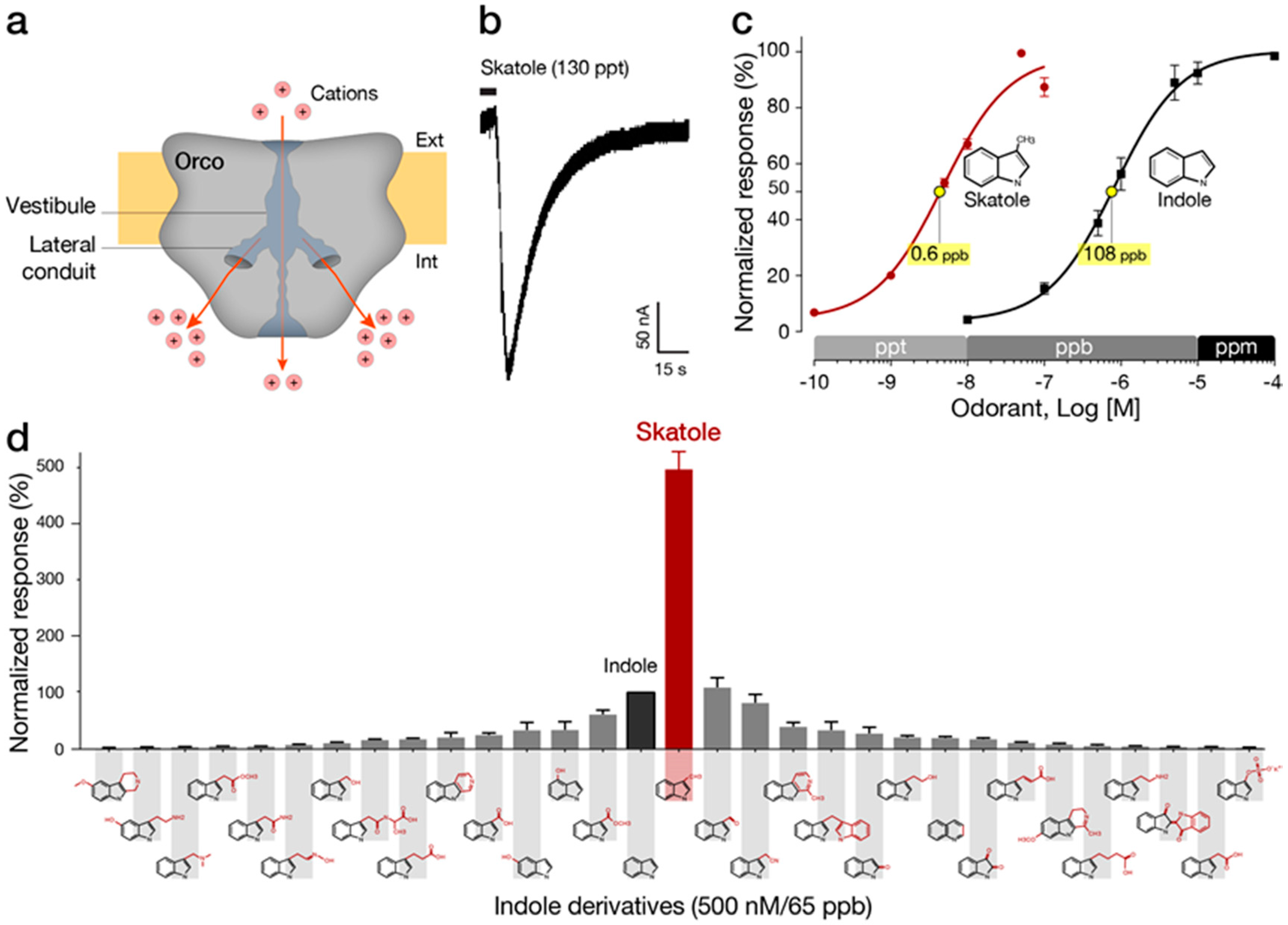
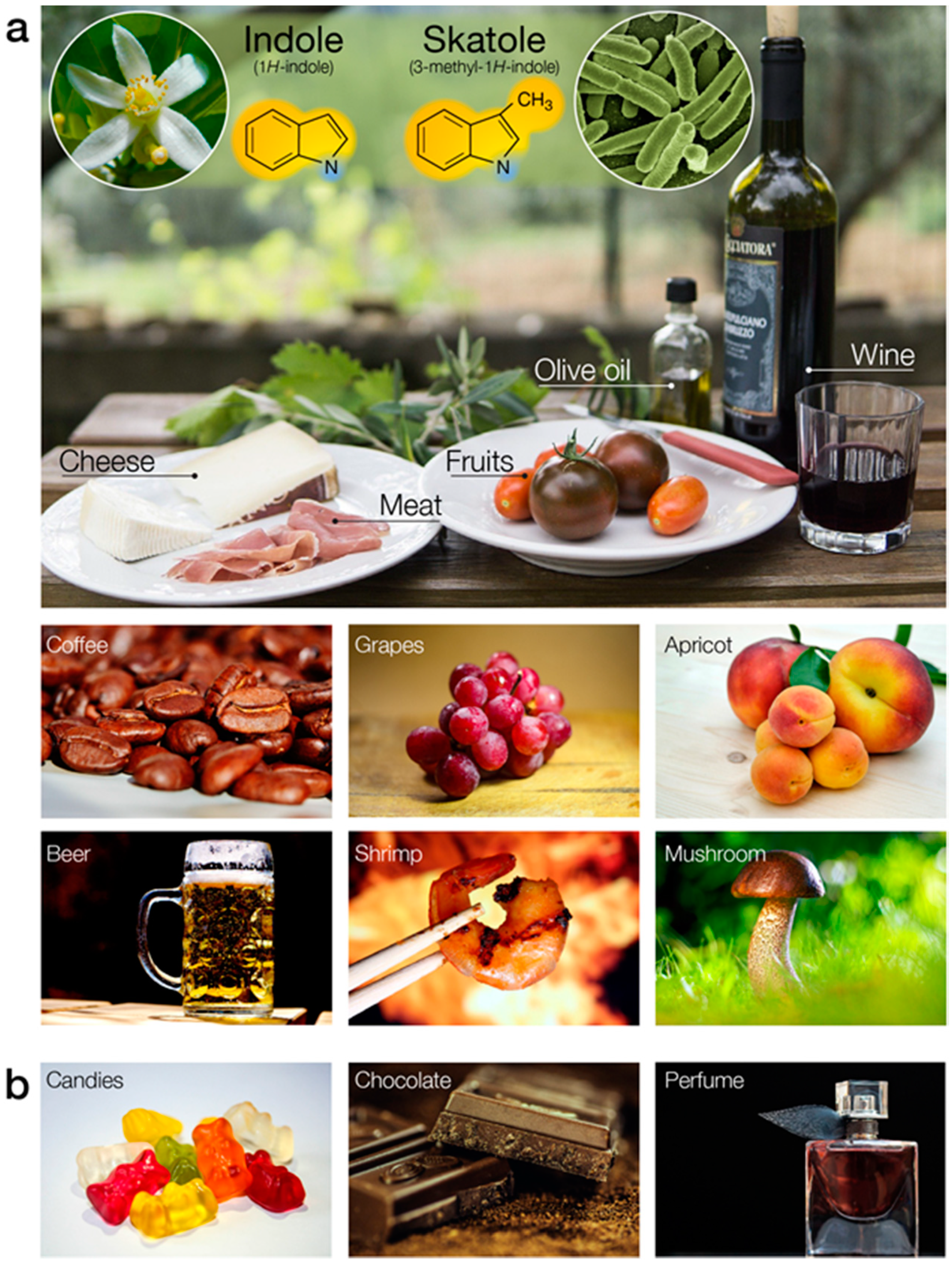
4. Indole-Sensitive ORs (“IndolORs”)
4.1. Chemical Description of Indole and Skatole
4.2. Diagnostic Significance of Indole and Skatole
4.3. Discovery and Pharmacological Properties of IndolORs
4.4. IndolOR-Based Biosensor
5. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chang, Y.; Tang, N.; Qu, H.M.; Liu, J.; Zhang, D.H.; Zhang, H.; Pang, W.; Duan, X.X. Detection of volatile organic compounds by self-assembled monolayer coated sensor array with concentration-independent fingerprints. Sci. Rep. 2016, 6, 23970. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Lee, S.C.; Chan, L.Y.; Li, W.M. Risk assessment of exposure to volatile organic compounds in different indoor environments. Environ. Res. 2004, 94, 57–66. [Google Scholar] [CrossRef]
- Barr, D.B.; Wang, R.Y.; Needham, L.L. Biologic monitoring of exposure to environmental chemicals throughout the life stages: Requirements and issues for consideration for the National Children’s Study. Environ. Health Perspect. 2005, 113, 1083–1091. [Google Scholar] [CrossRef] [PubMed]
- Mølhave, L.; Bach, B.; Pedersen, O.F. Human reactions to low concentrations of volatile organic compounds. Environ. Int. 1986, 12, 167–175. [Google Scholar] [CrossRef]
- Fleming-Jones, M.E.; Smith, R.E. Volatile organic compounds in foods: A five year study. J. Agric. Food Chem. 2003, 51, 8120–8127. [Google Scholar] [CrossRef]
- Hong-Geller, E.; Adikari, S. Volatile organic compound and metabolite signatures as pathogen identifiers and biomarkers of infectious disease. InTech 2018. [Google Scholar] [CrossRef]
- Bos, L.D.J.; Sterk, P.J.; Schultz, M.J. Volatile metabolites of pathogens: A systematic review. PLoS Pathog. 2013, 9, 5. [Google Scholar] [CrossRef]
- Zhu, J.J.; Hill, J.E. Detection of Escherichia coli via VOC profiling using secondary electrospray ionization-mass spectrometry (SESI-MS). Food Microbiol. 2013, 34, 412–417. [Google Scholar] [CrossRef]
- Perez-Hurtado, P.; Palmer, E.; Owen, T.; Aldcroft, C.; Allen, M.H.; Jones, J.; Creaser, C.S.; Lindley, M.R.; Turner, M.A.; Reynolds, J.C. Direct analysis of volatile organic compounds in foods by headspace extraction atmospheric pressure chemical ionisation mass spectrometry. Rapid Commun. Mass Spectrom. 2017, 31, 1947–1956. [Google Scholar] [CrossRef]
- Chen, J.; Tang, J.N.; Shi, H.; Tang, C.; Zhang, R. Characteristics of volatile organic compounds produced from five pathogenic bacteria by headspace-solid phase micro-extraction/gas chromatography-mass spectrometry. J. Basic Microb. 2017, 57, 228–237. [Google Scholar] [CrossRef]
- Mohsen Sohrabi, L.Z.K.Z.A.A.; Ming, Q.W. Volatile organic compounds as novel markers for the detection of bacterial infections. Clin. Microbiol. Open Access 2014, 3, 3. [Google Scholar]
- Fung, A.O.; Mykhaylova, N. Analysis of airborne biomarkers for point-of-care diagnostics. JALA J. Lab. Autom. 2014, 19, 225–247. [Google Scholar] [CrossRef] [PubMed]
- Knobloch, H.; Schroedl, W.; Turner, C.; Chambers, M.; Reinhold, P. Electronic nose responses and acute phase proteins correlate in blood using a bovine model of respiratory infection. Sens. Actuators B Chem. 2010, 144, 81–87. [Google Scholar] [CrossRef]
- Bhattacharyya, D.; Kumar, P.; Mohanty, S.K.; Smith, Y.R.; Misra, M. Detection of four distinct volatile indicators of colorectal cancer using functionalized titania nanotubular arrays. Sensors 2017, 17, 1795. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.L.; Xiao, P.; Fang, H.L.; Dai, H.F.; Qiao, L.; Zhang, Y.H. Single-walled carbon nanotube-based biosensors for the detection of volatile organic compounds of lung cancer. Phys. E Low Dimens. Syst. Nanostruct. 2011, 44, 367–372. [Google Scholar] [CrossRef]
- Lavra, L.; Catini, A.; Ulivieri, A.; Capuano, R.; Salehi, L.B.; Sciacchitano, S.; Bartolazzi, A.; Nardis, S.; Paolesse, R.; Martinelli, E.; et al. Investigation of VOCs associated with different characteristics of breast cancer cells. Sci. Rep. 2015, 5, 13246. [Google Scholar] [CrossRef]
- Wilson, A.D. Advances in Electronic-Nose Technologies for the detection of volatile biomarker metabolites in the human breath. Metabolites 2015, 5, 140–163. [Google Scholar] [CrossRef]
- Chen, G.; Paronyan, T.M.; Harutyunyan, A.R. Sub-ppt gas detection with pristine graphene. Appl. Phys. Lett. 2012, 101, 053119. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, Y.; Wang, Z.; Yang, L.; Wu, X.; Han, N.; Chen, Y. Synergetic p+n field-effect transistor circuits for ppb-level xylene detection. IEEE Sens. J. 2018, 18, 3875–3882. [Google Scholar] [CrossRef]
- Macias, M.M.; Agudo, J.E.; Manso, A.G.; Orellana, C.J.G.; Velasco, H.M.G.; Caballero, R.G. A compact and low cost electronic nose for aroma detection. Sensors 2013, 13, 5528–5541. [Google Scholar] [CrossRef]
- Turner, A.P.; Magan, N. Electronic noses and disease diagnostics. Nat. Rev. Microbiol. 2004, 2, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Misawa, N.; Osaki, T.; Takeuchi, S. Membrane protein-based biosensors. J. R. Soc. Interface 2018, 15, 20170952. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Wu, C.; Liu, Q.; Huang, L.; Wang, P. Recent advances in olfactory receptor-based biosensors. Biosens. Bioelectron. 2013, 42, 570–580. [Google Scholar] [CrossRef] [PubMed]
- Wasilewski, T.; Gębicki, J.; Kamysz, W. Advances in olfaction-inspired biomaterials applied to bioelectronic noses. Sens. Actuators B Chem. 2018, 257, 511–537. [Google Scholar] [CrossRef]
- Wu, T.Z. A piezoelectric biosensor as an olfactory receptor for odour detection: Electronic nose. Biosens. Bioelectron. 1999, 14, 9–18. [Google Scholar] [CrossRef]
- Ko, H.J.; Park, T.H. Piezoelectric olfactory biosensor: Ligand specificity and dose-dependence of an olfactory receptor expressed in a heterologous cell system. Biosens. Bioelectron. 2005, 20, 1327–1332. [Google Scholar] [CrossRef]
- Ko, H.J.; Park, T.H. Functional analysis of olfactory receptors expressed in a HEK-293 cell system by using cameleons. J. Microbiol. Biotechnol. 2007, 17, 928–933. [Google Scholar]
- Lee, S.H.; Jun, S.B.; Ko, H.J.; Kim, S.J.; Park, T.H. Cell-based olfactory biosensor using microfabricated planar electrode. Biosens. Bioelectron. 2009, 24, 2659–2664. [Google Scholar] [CrossRef]
- Saito, R.; Sato, K.; Araujo, P.T.; Mafra, D.L.; Dresselhaus, M.S. Gate modulated Raman spectroscopy of graphene and carbon nanotubes. Solid State Commun. 2013, 175, 18–34. [Google Scholar] [CrossRef]
- Marrakchi, M.; Vidic, J.; Jaffrezic-Renault, N.; Martelet, C.; Pajot-Augy, E. A new concept of olfactory biosensor based on interdigitated microelectrodes and immobilized yeasts expressing the human receptor OR17–40. Eur. Biophys. J. 2007, 36, 1015–1018. [Google Scholar] [CrossRef]
- Misawa, N.; Mitsuno, H.; Kanzaki, R.; Takeuchi, S. Highly sensitive and selective odorant sensor using living cells expressing insect olfactory receptors. Proc. Natl. Acad. Sci. USA 2010, 107, 15340–15344. [Google Scholar] [CrossRef] [PubMed]
- Terutsuki, D.; Mitsuno, H.; Sakurai, T.; Okamoto, Y.; Tixier-Mita, A.; Toshiyoshi, H.; Mita, Y.; Kanzaki, R. Increasing cell-device adherence using cultured insect cells for receptor-based biosensors. R. Soc. Open Sci. 2018, 5, 172366. [Google Scholar] [CrossRef] [PubMed]
- Termtanasombat, M.; Mitsuno, H.; Misawa, N.; Yamahira, S.; Sakurai, T.; Yamaguchi, S.; Nagamune, T.; Kanzaki, R. Cell-based odorant sensor array for odor discrimination based on insect odorant receptors. J. Chem. Ecol. 2016, 42, 716–724. [Google Scholar] [CrossRef] [PubMed]
- Vidic, J.M.; Grosclaude, J.; Persuy, M.A.; Aioun, J.; Salesse, R.; Pajot-Augy, E. Quantitative assessment of olfactory receptors activity in immobilized manosomes: A novel concept for bioelectronic nose. Lab Chip 2006, 6, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.J.; Lee, S.H.; Kim, T.H.; Park, J.; Song, H.S.; Park, T.H.; Hong, S. Nanovesicle-based bioelectronic nose platform mimicking human olfactory signal transduction. Biosens. Bioelectron. 2012, 35, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, B.R.; Mitala, J.J.; Josue, J.; Castro, A.; Lerner, M.B.; Bayburt, T.H.; Khamis, S.M.; Jones, R.A.; Brand, J.G.; Sligar, S.G.; et al. Biomimetic chemical sensors using nanoelectronic readout of olfactory receptor proteins. ACS Nano 2011, 5, 5408–5416. [Google Scholar] [CrossRef]
- Mitsuno, H.; Sakurai, T.; Namiki, S.; Mitsuhashi, H.; Kanzaki, R. Novel cell-based odorant sensor elements based on insect odorant receptors. Biosens. Bioelectron. 2015, 65, 287–294. [Google Scholar] [CrossRef]
- Terutsuki, D.; Mitsuno, H.; Okamoto, Y.; Sakurai, T.; Tixier-Mita, A.; Toshiyoshi, H.; Mita, Y.; Kanzaki, R. Odor-sensitive field effect transistor (OSFET) based on insect cells expressing insect odorant receptors. In Proceedings of the 2017 IEEE 30th International Conference on Micro Electro Mechanical Systems (MEMS), Las Vegas, NV, USA, 22–26 January 2017; pp. 394–397. [Google Scholar]
- Huotari, M.; Lantto, V. Measurements of odours based on response analysis of insect olfactory receptor neurons. Sens. Actuators B Chem. 2007, 127, 284–287. [Google Scholar] [CrossRef]
- Pelosi, P.; Zhu, J.; Knoll, W. From gas sensors to biomimetic artificial noses. Chemosensors 2018, 6, 3. [Google Scholar] [CrossRef]
- Pelosi, P. The role of perireceptor events in vertebrate olfaction. Cell. Mol. Life Sci. 2001, 58, 503–509. [Google Scholar] [CrossRef]
- Tegoni, M.; Campanacci, V.; Cambillau, C. Structural aspects of sexual attraction and chemical communication in insects. Trends Biochem. Sci. 2004, 29, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Brandazza, A.; Pelosi, P. Binding of polycyclic aromatic hydrocarbons to mutants of odorant-binding protein: A first step towards biosensors for environmental monitoring. Biochim. Biophys. Acta 2008, 1784, 666–671. [Google Scholar] [CrossRef]
- Pelosi, P.; Mastrogiacomo, R.; Iovinella, I.; Tuccori, E.; Persaud, K.C. Structure and biotechnological applications of odorant-binding proteins. Appl. Microbiol. Biotechnol. 2014, 98, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, A.J.M.; Oliveira, A.R.; Roque, A.C.A. Protein- and peptide-based biosensors in artificial olfaction. Trends Biotechnol. 2018, 36, 1244–1258. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Kim, D.; Kim, J.; Moon, D.; Song, H.S.; Lee, M.; Hong, S.; Park, T.H. Nanodisc-based bioelectronic nose using olfactory receptor produced in Escherichia coli for the assessment of the death-associated odor cadaverine. ACS Nano 2017, 11, 11847–11855. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Yang, H.; Kim, D.; Yang, M.; Park, T.H.; Hong, S. Human-like smelling of a rose scent using an olfactory receptor nanodisc-based bioelectronic nose. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef]
- Murugathas, T.; Zheng, H.Y.; Colbert, D.; Kralicek, A.V.; Carraher, C.; Plank, N.O.V. Biosensing with insect odorant receptor nanodiscs and carbon nanotube field-effect transistors. ACS Appl. Mater. Interfaces 2019, 11, 9530–9538. [Google Scholar] [CrossRef]
- Khadka, R.; Aydemir, N.; Carraher, C.; Hamiaux, C.; Colbert, D.; Cheema, J.; Malmstrom, J.; Kralicek, A.; Travas-Sejdic, J. An ultrasensitive electrochemical impedance-based biosensor using insect odorant receptors to detect odorants. Biosens. Bioelectron. 2019, 126, 207–213. [Google Scholar] [CrossRef]
- Sung, J.H.; Ko, H.J.; Park, T.H. Piezoelectric biosensor using olfactory receptor protein expressed in Escherichia coli. Biosens. Bioelectron. 2006, 21, 1981–1986. [Google Scholar] [CrossRef]
- Lim, J.H.; Park, J.; Ahn, J.H.; Jin, H.J.; Hong, S.; Park, T.H. A peptide receptor-based bioelectronic nose for the real-time determination of seafood quality. Biosens. Bioelectron. 2013, 39, 244–249. [Google Scholar] [CrossRef]
- Oh, E.H.; Lee, S.H.; Ko, H.J.; Park, T.H. Odorant detection using liposome containing olfactory receptor in the SPR system. Sens. Actuators B Chem. 2014, 198, 188–193. [Google Scholar] [CrossRef]
- Park, S.J.; Kwon, O.S.; Lee, S.H.; Song, H.S.; Park, T.H.; Jang, J. Ultrasensitive flexible graphene based field-effect transistor (FET)-type bioelectronic nose. Nano Lett. 2012, 12, 5082–5090. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.H.; Lim, J.H.; Park, J.; Oh, E.H.; Son, M.; Hong, S.; Park, T.H. Screening of target-specific olfactory receptor and development of olfactory biosensor for the assessment of fungal contamination in grain Screening of target-specific olfactory receptor and development of olfactory biosensor for the assessment of fungal contamination in grain. Sens. Actuators B Chem. 2015, 210, 9–16. [Google Scholar]
- Ruel, D.M.; Yakir, E.; Bohbot, J.D. Supersensitive odorant receptor underscores pleiotropic roles of indoles in mosquito ecology. Front. Cell. Neurosci. 2019, 12. [Google Scholar] [CrossRef]
- Fleischer, J.; Pregitzer, P.; Breer, H.; Krieger, J. Access to the odor world: Olfactory receptors and their role for signal transduction in insects. Cell. Mol. Life Sci. 2018, 75, 485–508. [Google Scholar] [CrossRef]
- Bohbot, J.D.; Dickens, J.C. Odorant receptor modulation: Ternary paradigm for mode of action of insect repellents. Neuropharmacology 2012, 62, 2086–2095. [Google Scholar] [CrossRef]
- Bohbot, J.D.; Dickens, J.C. Insect repellents: Modulators of mosquito odorant receptor activity. PLoS ONE 2010, 5, e12138. [Google Scholar] [CrossRef]
- Tsitoura, P.; Koussis, K.; Iatrou, K. Inhibition of Anopheles gambiae odorant receptor function by mosquito repellents. J. Biol. Chem. 2015, 290, 7961–7972. [Google Scholar] [CrossRef]
- Butterwick, J.A.; Del Marmol, J.; Kim, K.H.; Kahlson, M.A.; Rogow, J.A.; Walz, T.; Ruta, V. Cryo-EM structure of the insect olfactory receptor Orco. Nature 2018, 560, 447–452. [Google Scholar] [CrossRef]
- Nath, A.; Atkins, W.M.; Sligar, S.G. Applications of phospholipid bilayer nanodiscs in the study of membranes and membrane proteins. Biochemistry 2007, 46, 2059–2069. [Google Scholar] [CrossRef]
- Bayburt, T.H.; Sligar, S.G. Membrane protein assembly into Nanodiscs. FEBS Lett. 2010, 584, 1721–1727. [Google Scholar] [CrossRef]
- Ritchie, T.K.; Grinkova, Y.V.; Bayburt, T.H.; Denisov, I.G.; Zolnerciks, J.K.; Atkins, W.M.; Sligar, S.G. Chapter eleven—Reconstitution of membrane proteins in phospholipid bilayer nanodiscs. Methods Enzymol. 2009, 464, 211–231. [Google Scholar] [PubMed]
- Inagaki, S.; Ghirlando, R.; Grisshammer, R. Biophysical characterization of membrane proteins in nanodiscs. Methods 2013, 59, 287–300. [Google Scholar] [CrossRef] [PubMed]
- Denisov, I.G.; Sligar, S.G. Nanodiscs for structural and functional studies of membrane proteins. Nat. Struct. Mol. Biol. 2016, 23, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Sackmann, E. Supported membranes: Scientific and practical applications. Science 1996, 271, 43–48. [Google Scholar] [CrossRef]
- Helfrich, W. Stiffening of fluid membranes and entropy loss of membrane closure: Two effects of thermal undulations. Eur. Phys. J. B 1998, 1, 481–489. [Google Scholar] [CrossRef]
- West, A.; Ma, K.; Chung, J.L.; Kindt, J.T. Simulation studies of structure and edge tension of lipid bilayer edges: Effects of tail structure and Force-field. J. Phys. Chem. A 2013, 117, 7114–7123. [Google Scholar] [CrossRef]
- Yoon, J.; Jo, W.; Jeong, D.; Kim, J.; Jeong, H.; Park, J. Generation of nanovesicles with sliced cellular membrane fragments for exogenous material delivery. Biomaterials 2015, 59, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Moran-Mirabal, J.M.; Edel, J.B.; Meyer, G.D.; Throckmorton, D.; Singh, A.K.; Craighead, H.G. Micrometer-sized supported lipid bilayer arrays for bacterial toxin binding studies through total internal reflection fluorescence microscopy. Biophys. J. 2005, 89, 296–305. [Google Scholar] [CrossRef]
- Sanmartí-Espinal, M.; Iavicoli, P.; Calò, A.; Taulés, M.; Galve, R.; Marco, M.P.; Samitier, J. Quantification of interacting cognate odorants with olfactory receptors in nanovesicles. Sci. Rep. 2017, 7, 17483. [Google Scholar] [CrossRef]
- Rajapaksha, S.P.; Wang, X.; Lu, H.P. Suspended lipid bilayer for optical and electrical measurements of single ion channel proteins. Anal. Chem. 2013, 85, 8951–8955. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.-S.; Jain, D.; Burke, P.J. Biomembrane-gated carbon nanotube transistor as a sensing platform. In Proceedings of the 15th International Conference on Miniaturized Systems for Chemistry and Life Sciences, Seatle, WA, USA, 2–6 October 2011. [Google Scholar]
- Gangupomu, V.K.; Capaldi, F.M. Interactions of carbon nanotube with lipid bilayer membranes. J. Nanomater. 2011, 31. [Google Scholar] [CrossRef]
- Geng, J.; Kim, K.; Zhang, J.F.; Escalada, A.; Tunuguntla, R.; Comolli, L.R.; Allen, F.I.; Shnyrova, A.V.; Cho, K.R.; Munoz, D.; et al. Stochastic transport through carbon nanotubes in lipid bilayers and live cell membranes. Nature 2014, 514, 612–615. [Google Scholar] [CrossRef] [PubMed]
- Bradley, K.; Davis, A.; Gabriel, J.C.P.; Gruner, G. Integration of cell membranes and nanotube transistors. Nano Lett. 2005, 5, 841–845. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.J.; Moran-Mirabal, J.M.; Craighead, H.G.; McEuen, P.L. Supported lipid bilayer/carbon nanotube hybrids. Nat. Nanotechnol. 2007, 2, 185–190. [Google Scholar] [CrossRef]
- Fujii, S.; Nobukawa, A.; Osaki, T.; Morimoto, Y.; Kamiya, K.; Misawa, N.; Takeuchi, S. Pesticide vapor sensing using an aptamer, nanopore, and agarose gel on a chip. Lab Chip 2017, 17, 2421–2425. [Google Scholar] [CrossRef]
- Osaki, T.; Takeuchi, S. Artificial cell membrane systems for biosensing applications. Anal. Chem. 2017, 89, 216–231. [Google Scholar] [CrossRef]
- Nikoleli, G.P.; Siontorou, C.G.; Nikolelis, M.T.; Bratakou, S.; Bendos, D.K. Recent lipid membrane-based biosensing platforms. Appl. Sci. 2019, 9, 1745. [Google Scholar] [CrossRef]
- Choi, Y.K.; Moody, I.S.; Sims, P.C.; Hunt, S.R.; Corso, B.L.; Perez, I.; Weiss, G.A.; Collins, P.G. Single-molecule lysozyme dynamics monitored by an electronic circuit. Science 2012, 335, 319–324. [Google Scholar] [CrossRef]
- Chen, R.J.; Zhang, Y.; Wang, D.; Dai, H. Noncovalent sidewall functionalization of single-walled carbon nanotubes for protein immobilization. J. Am. Chem. Soc. 2001, 123, 3838–3839. [Google Scholar] [CrossRef]
- Sorgenfrei, S.; Chiu, C.-Y.; Johnston, M.; Nuckolls, C.; Shepard, K.L. Debye screening in single-molecule carbon nanotube field-effect sensors. Nano Lett. 2011, 11, 3739–3743. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Park, T.H. Recent advances in the development of bioelectronic nose. Biotechnol. Bioprocess Eng. 2010, 15, 22–29. [Google Scholar] [CrossRef]
- Du, L.; Wu, C.; Peng, H.; Zou, L.; Zhao, L.; Huang, L.; Wang, P. Piezoelectric olfactory receptor biosensor prepared by aptamer-assisted immobilization. Sens. Actuators B Chem. 2013, 187, 481–487. [Google Scholar] [CrossRef]
- Chen, Q.; Xiao, L.; Liu, Q.; Ling, S.; Yin, Y.; Dong, Q.; Wang, P. An olfactory bulb slice-based biosensor for multi-site extracellular recording of neural networks. Biosens. Bioelectron. 2011, 26, 3313–3319. [Google Scholar] [CrossRef]
- Huotari, M.J. Biosensing by insect olfactory receptor neurons. Sens. Actuators B Chem. 2000, 71, 212–222. [Google Scholar] [CrossRef]
- Alfinito, E.; Pennetta, C.; Reggiani, L. Olfactory receptor-based smell nanobiosensors: An overview of theoretical and experimental results. Sens. Actuators B Chem. 2010, 146, 554–558. [Google Scholar] [CrossRef]
- Alfinito, E.; Millithaler, J.F.; Pennetta, C.; Reggiani, L. A single protein based nanobiosensor for odorant recognition. Microelectron. J. 2010, 41, 718–722. [Google Scholar] [CrossRef]
- Alfinito, E.; Millithaler, J.F.; Reggiani, L. Olfactory receptors for a smell sensor: A comparative study of the electrical responses of rat I7 and human 17–40. Measure. Sci. Technol. 2011, 22, 124004. [Google Scholar] [CrossRef]
- Yoon, H.; Lee, S.H.; Kwon, O.S.; Song, H.S.; Oh, E.H.; Park, T.H.; Jang, J. Polypyrrole nanotubes conjugated with human olfactory receptors: High-performance transducers for FET-type bioelectronic noses. Angew. Chem. Int. Ed. Engl. 2009, 48, 2755–2758. [Google Scholar] [CrossRef]
- Kwon, O.S.; Song, H.S.; Park, S.J.; Lee, S.H.; An, J.H.; Park, J.W.; Yang, H.; Yoon, H.; Bae, J.; Park, T.H.; et al. An ultrasensitive, selective, multiplexed superbioelectronic nose that mimics the human sense of smell. Nano Lett. 2015, 15, 6559–6567. [Google Scholar] [CrossRef]
- Tans, S.J.; Verschueren, A.R.M.; Dekker, C. Room-temperature transistor based on a single carbon nanotube. Nature 1998, 393, 49–52. [Google Scholar] [CrossRef]
- Javey, A.; Guo, J.; Wang, Q.; Lundstrom, M.; Dai, H.J. Ballistic carbon nanotube field-effect transistors. Nature 2003, 424, 654–657. [Google Scholar] [CrossRef] [PubMed]
- Allen, B.L.; Kichambare, P.D.; Star, A. Carbon nanotube field-effect-transistor-based biosensors. Adv. Mater. 2007, 19, 1439–1451. [Google Scholar] [CrossRef]
- Zaporotskova, I.V.; Kozhitov, L.V.; Boroznina, N.P. Sensor activity with respect to alkali metals of a carbon nanotube edge-modified with amino group. Russ. J. Inorg. Chem. 2017, 62, 1458–1463. [Google Scholar] [CrossRef]
- Leyden, M.R.; Schuman, C.; Sharf, T.; Kevek, J.; Remcho, V.T.; Minot, E.D. Fabrication and characterization of carbon nanotube field-effect transistor biosensors. Org. Semicond. Sens. Bioelectron. III 2010, 7779, 16. [Google Scholar]
- Sorgenfrei, S.; Chiu, C.Y.; Gonzalez, R.L.; Yu, Y.J.; Kim, P.; Nuckolls, C.; Shepard, K.L. Label-free single-molecule detection of DNA-hybridization kinetics with a carbon nanotube field-effect transistor. Nat. Nanotechnol. 2011, 6, 125–131. [Google Scholar] [CrossRef]
- Tang, X.; Bansaruntip, S.; Nakayama, N.; Yenilmez, E.; Chang, Y.-L.; Wang, Q. Carbon nanotube DNA sensor and sensing mechanism. Nano Lett. 2006, 6, 1632–1636. [Google Scholar] [CrossRef]
- Vernick, S.; Trocchia, S.M.; Warren, S.B.; Young, E.F.; Bouilly, D.; Gonzalez, R.L.; Nuckolls, C.; Shepard, K.L. Electrostatic melting in a single-molecule field-effect transistor with applications in genomic identification. Nat. Commun. 2017, 8, 15450. [Google Scholar] [CrossRef]
- Bouilly, D.; Hon, J.; Daly, N.S.; Trocchia, S.; Vernick, S.; Yu, J.; Warren, S.; Wu, Y.; Gonzalez, R.L., Jr.; Shepard, K.L.; et al. Single-molecule reaction chemistry in patterned nanowells. Nano Lett. 2016, 16, 4679–4685. [Google Scholar] [CrossRef]
- Lerner, M.B.; Dailey, J.; Goldsmith, B.R.; Brisson, D.; Johnson, A.T.C. Detecting Lyme disease using antibody-functionalized single-walled carbon nanotube transistors. Biosens. Bioelectron. 2013, 45, 163–167. [Google Scholar] [CrossRef]
- Kim, T.H.; Lee, S.H.; Lee, J.; Song, H.S.; Oh, E.H.; Park, T.H.; Hong, S. Single-carbon-atomic-resolution detection of odorant molecules using a human olfactory receptor-based bioelectronic nose. Adv. Mater. 2009, 21, 91–94. [Google Scholar] [CrossRef]
- Son, M.; Kim, D.; Kang, J.; Lim, J.H.; Lee, S.H.; Ko, H.J.; Hong, S.; Park, T.H. Bioelectronic nose using odorant binding protein-derived peptide and carbon nanotube field-effect transistor for the assessment of salmonella contamination in food. Anal. Chem. 2016, 88, 11283–11287. [Google Scholar] [CrossRef] [PubMed]
- Mahmoodlu, M.G.; Pontedeiro, E.M.; Guerrero, J.S.P.; Raoof, A.; Hassanizadeh, S.M.; Van Genuchten, M.T. Dissolution kinetics of volatile organic compound vapors in water: An integrated experimental and computational study. J. Contam. Hydrol. 2017, 196, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Orakdogen, N.; Okay, O. Reentrant conformation transition in poly(N,N-dimethylacrylamide) hydrogels in water–organic solvent mixtures. Polymer 2006, 47, 561–568. [Google Scholar] [CrossRef]
- Kaushik, N.K.; Kaushik, N.; Attri, P.; Kumar, N.; Kim, C.H.; Verma, A.K.; Choi, E.H. Biomedical importance of indoles. Molecules 2013, 18, 6620–6662. [Google Scholar] [CrossRef] [PubMed]
- Ten, G.N.; Yakovleva, A.A.; Baranov, V.I. Theoretical study of hydrophobicity and hydrophilicity of indole, skatole, and ethanole. J. Struct. Chem. 2013, 54, 1018–1028. [Google Scholar] [CrossRef]
- Lee, J.H.; Wood, T.K.; Lee, J. Roles of indole as an interspecies and interkingdom signaling molecule. Trends Microbiol. 2015, 23, 707–718. [Google Scholar] [CrossRef] [PubMed]
- Oehlenschläger, J.; Luten, J.B. Review: Indole as a quality indicator in shrimps and prawns. Arch. Lebensmittelhyg. 2005, 56, 52–57. [Google Scholar]
- Quaranta, H.O.; Curzio, O.A. Free fatty acids in irradiated hake fillets. Int. J. Appl. Radiat. Isot. 1984, 35, 912–933. [Google Scholar] [CrossRef]
- Beacham, L.M. A study of decomposition in canned oysters and chuns. J. Assoc. Off. Agric. Chem. 1946, 29, 89. [Google Scholar]
- Duggan, R.E. Report on decomposition in shellfish—Indole in shrimp, oysters, and crabmeat. J. Assoc. Off. Agric. Chem. 1948, 31, 507. [Google Scholar] [CrossRef]
- King, W.H.; Flynn, F.F. Experimental studies on decomposition of oysters used for canning. J. Assoc. Off. Agric. Chem. 1945, 28, 385. [Google Scholar] [CrossRef]
- Pensabene, J.W.; Fiddler, W. Indole and skatole in fresh pork as possible markers of fecal contamination. J. Food Protect. 1996, 59, 663–665. [Google Scholar] [CrossRef] [PubMed]
- Bonneau, M.; Kempster, A.J.; Claus, R.; Claudi-Magnussen, C.; Diestre, A.; Tornberg, E.; Walstra, P.; Chevillon, P.; Weiler, U.; Cook, G.L. An international study on the importance of androstenone and skatole for boar taint: I. Presentation of the programme and measurement of boar taint compounds with different analytical procedures. Meat Sci. 2000, 54, 251–259. [Google Scholar] [CrossRef]
- Aluwe, M.; Tuyttens, F.A.M.; Bekaert, K.M.; De Smet, S.; De Brabander, D.L.; Millet, S. Evaluation of various boar taint detection methods. Animal 2012, 6, 1868–1877. [Google Scholar] [CrossRef][Green Version]
- Morlein, D.; Trautmann, J.; Gertheiss, J.; Meier-Dinkel, L.; Fischer, J.; Eynck, H.J.; Heres, L.; Looft, C.; Tholen, E. Interaction of skatole and androstenone in the olfactory perception of boar taint. J. Agric. Food Chem. 2016, 64, 4556–4565. [Google Scholar] [CrossRef]
- Thu, K.; Donham, K.; Ziegenhorn, R.; Reynolds, S.; Thorne, P.S.; Subramanian, P.; Whitten, P.; Stookesberry, J. A control study of the physical and mental health of residents living near a large-scale swine operation. J. Agric. Saf. Health 1997, 3, 13–26. [Google Scholar] [CrossRef]
- Carlson, J.R.; Yokoyama, M.T.; Dickinson, E.O. Induction of pulmonary edema and emphysema in cattle and goats with 3-methylindole. Science 1972, 176, 298–299. [Google Scholar] [CrossRef]
- Ruangyuttikarn, W.; Appleton, M.L.; Yost, G.S. Metabolism of 3-methylindole in human tissues. Drug Metab. Dispos. 1991, 19, 977–984. [Google Scholar]
- Peele, D.B.; Allison, S.D.; Bolon, B.; Prah, J.D.; Jensen, K.F.; Morgan, K.T. Functional deficits produced by 3-methylindole-induced olfactory mucosal damage revealed by a simple olfactory learning task. Toxicol. Appl. Pharmacol. 1991, 107, 191–202. [Google Scholar] [CrossRef]
- Zhou, Y.B.; Hallis, S.A.; Vitko, T.; Suffet, I.H. Identification, quantification and treatment of fecal odors released into the air at two wastewater treatment plants. J. Environ. Manag. 2016, 180, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Chappell, C.L.; Darkoh, C.; Shimmin, L.; Farhana, N.; Kim, D.K.; Okhuysen, P.C.; Hixson, J. Fecal indole as a biomarker of susceptibility to cryptosporidium infection. Infect. Immun. 2016, 84, 2299–2306. [Google Scholar] [CrossRef]
- Codipilly, D.; Kleinberg, I. Generation of indole/skatole during malodor formation in the salivary sediment model system and initial examination of the oral bacteria involved. J. Breath Res. 2008, 2, 017017. [Google Scholar] [CrossRef] [PubMed]
- Bollen, C.M.; Beikler, T. Halitosis: The multidisciplinary approach. Int. J. Oral Sci. 2012, 4, 55–63. [Google Scholar] [CrossRef]
- Van den Velde, S.; Nevens, F.; Van Hee, P.; Van Steenberghe, D.; Quirynen, M. GC-MS analysis of breath odor compounds in liver patients. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2008, 875, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Karlin, D.A.; Mastromarino, A.J.; Jones, R.D.; Stroehlein, J.R.; Lorentz, O. Fecal skatole and indole and breath methane and hydrogen in patients with large bowel polyps or cancer. J. Cancer Res. Clin. Oncol. 1985, 109, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Clyne, P.J.; Warr, C.G.; Freeman, M.R.; Lessing, D.; Kim, J.; Carlson, J.R. A novel family of divergent seven-transmembrane proteins: Candidate odorant receptors in Drosophila. Neuron 1999, 22, 327–338. [Google Scholar] [CrossRef]
- Gao, Q.; Chess, A. Identification of candidate Drosophila olfactory receptors from genomic DNA sequence. Genomics 1999, 60, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Vosshall, L.B.; Amrein, H.; Morozov, P.S.; Rzhetsky, A.; Axel, R. A spatial map of olfactory receptor expression in the Drosophila antenna. Cell 1999, 96, 725–736. [Google Scholar] [CrossRef]
- Hill, C.A.; Fox, A.N.; Pitts, R.J.; Kent, L.B.; Tan, P.L.; Chrystal, M.A.; Cravchik, A.; Collins, F.H.; Robertson, H.M.; Zwiebel, L.J. G protein-coupled receptors in Anopheles gambiae. Science 2002, 298, 176–178. [Google Scholar] [CrossRef]
- Bohbot, J.; Pitts, R.J.; Kwon, H.W.; Rutzler, M.; Robertson, H.M.; Zwiebel, L.J. Molecular characterization of the Aedes aegypti odorant receptor gene family. Insect Mol. Biol. 2007, 16, 525–537. [Google Scholar] [CrossRef] [PubMed]
- Hallem, E.A.; Ho, M.G.; Carlson, J.R. The molecular basis of odor coding in the Drosophila antenna. Cell 2004, 117, 965–979. [Google Scholar] [CrossRef]
- Carey, A.F.; Wang, G.; Su, C.Y.; Zwiebel, L.J.; Carlson, J.R. Odorant reception in the malaria mosquito Anopheles gambiae. Nature 2010, 464, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Carey, A.F.; Carlson, J.R.; Zwiebel, L.J. Molecular basis of odor coding in the malaria vector mosquito Anopheles gambiae. Proc. Natl. Acad. Sci. USA 2010, 107, 4418–4423. [Google Scholar] [CrossRef] [PubMed]
- Hughes, D.T.; Pelletier, J.; Luetje, C.W.; Leal, W.S. Odorant receptor from the southern house mosquito narrowly tuned to the oviposition attractant skatole. J. Chem. Ecol. 2010, 36, 797–800. [Google Scholar] [CrossRef]
- Dekel, A.; Yakir, E.; Bohbot, J.D. The evolutionarily conserved indolergic receptors of the non-hematophagous elephant mosquito Toxorhynchites amboinensis. Insect Biochem. Mol. Biol. 2019, 110, 45–51. [Google Scholar] [CrossRef]
- Bohbot, J.D.; Pitts, R.J. The narrowing olfactory landscape of insect odorant receptors. Front. Ecol. Evolut. 2015, 3, 39. [Google Scholar] [CrossRef]
- Khadka, R.; Carraher, C.; Hamiaux, C.; Travas-Sejdic, J.; Kralicek, A. Synergistic improvement in the performance of insect odorant receptor based biosensors in the presence of Orco. Biosens. Bioelectron. 2020, 153, 112040. [Google Scholar] [CrossRef]
- Warren, S.B.; Vernick, S.; Romano, E.; Shepard, K.L. Complementary metal-oxide-semiconductor integrated carbon nanotube arrays: Toward wide-bandwidth single-molecule sensing systems. Nano Lett. 2016, 16, 2674–2679. [Google Scholar] [CrossRef]
- Bohbot, J.D.; Dickens, J.C. Characterization of an enantioselective odorant receptor in the yellow fever mosquito Aedes aegypti. PLoS ONE 2009, 4, e7032. [Google Scholar] [CrossRef]
- Stensmyr, M.C.; Dweck, H.K.; Farhan, A.; Ibba, I.; Strutz, A.; Mukunda, L.; Linz, J.; Grabe, V.; Steck, K.; Lavista-Llanos, S.; et al. A conserved dedicated olfactory circuit for detecting harmful microbes in Drosophila. Cell 2012, 151, 1345–1357. [Google Scholar] [CrossRef] [PubMed]
| OR | VOC | Application | Dynamic Range or MDL | Device | References | |
|---|---|---|---|---|---|
| ODR-10 | Diacetyl | Food & beverages | 10−12–10−5 M | Quartz crystal microbalance | [50] |
| mOR174-9 | Eugenol | Fragrance development | 2 ppm | Nanodisc packaged OR-CNT-FETs | [36] |
| mOR256-17 | Cyclohexanone | Process manufacturing | 2250 ppm | ||
| hOR 2AG1 | Amylbutyrate | Food screening & medical diagnostics | 10−12–10−3 M | Nanovesicle-based OR-CNT-FETs | [35] |
| hOR 2AG1 | Amylbutyrate | Disease diagnostics food safety & environmental monitoring | 10−15–10−12 M | Graphene-based FET | [53] |
| OR-derived peptide | Trimethylamine | Food screening | 10−15–10−4 M | Nanovesicle-based OR-CNT-FETs | [51] |
| hOR3A1 | Helional | Process manufacturing | 10−7–10−3 M | Liposome-based OR-SPR | [52] |
| hOR8H2 | 1-octen-3-ol | Food screening | 10−15–10−9 M | Nanovesicle-based OR-CNT-FETs | [54] |
| TAAR13c | Cadaverine | food safety | 10−12–10−6 M | Nanodisc packaged OR-CNT-FETs | [46] |
| hOR1A2 | Geraniol | Fragrance development | 10−15–10−3 M | Nanodisc packaged OR-CNT-FETs | [47] |
| DmelOR10a | Methyl salicylate | Food screening | 10−15–10−4 M | OR/liposome gold sensor | [49] |
| DmelOR22a | Methyl hexanoate | 10−15–10−4 M | |||
| DmelOR71a | 4-Ethylguaiacol | 10−16–10−4 M | |||
| DmelOR10a | Methyl salicylate | Food screening | 10−15–10−12 M | Nanodisc packaged OR-CNT-FETs | [48] |
| DmelOR22a | Methyl hexanoate | ||||
| DmelOR35a | trans-2-Hexen1-al | ||||
| DmelOR71a | 4-Ethylguaiacol | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bohbot, J.D.; Vernick, S. The Emergence of Insect Odorant Receptor-Based Biosensors. Biosensors 2020, 10, 26. https://doi.org/10.3390/bios10030026
Bohbot JD, Vernick S. The Emergence of Insect Odorant Receptor-Based Biosensors. Biosensors. 2020; 10(3):26. https://doi.org/10.3390/bios10030026
Chicago/Turabian StyleBohbot, Jonathan D., and Sefi Vernick. 2020. "The Emergence of Insect Odorant Receptor-Based Biosensors" Biosensors 10, no. 3: 26. https://doi.org/10.3390/bios10030026
APA StyleBohbot, J. D., & Vernick, S. (2020). The Emergence of Insect Odorant Receptor-Based Biosensors. Biosensors, 10(3), 26. https://doi.org/10.3390/bios10030026







