A Novel Fluorescence and SPE Adsorption Nanomaterials of Molecularly Imprinted Polymers Based on Quantum Dot-Grafted Covalent Organic Frameworks for the High Selectivity and Sensitivity Detection of Ferulic Acid
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Instrumentation
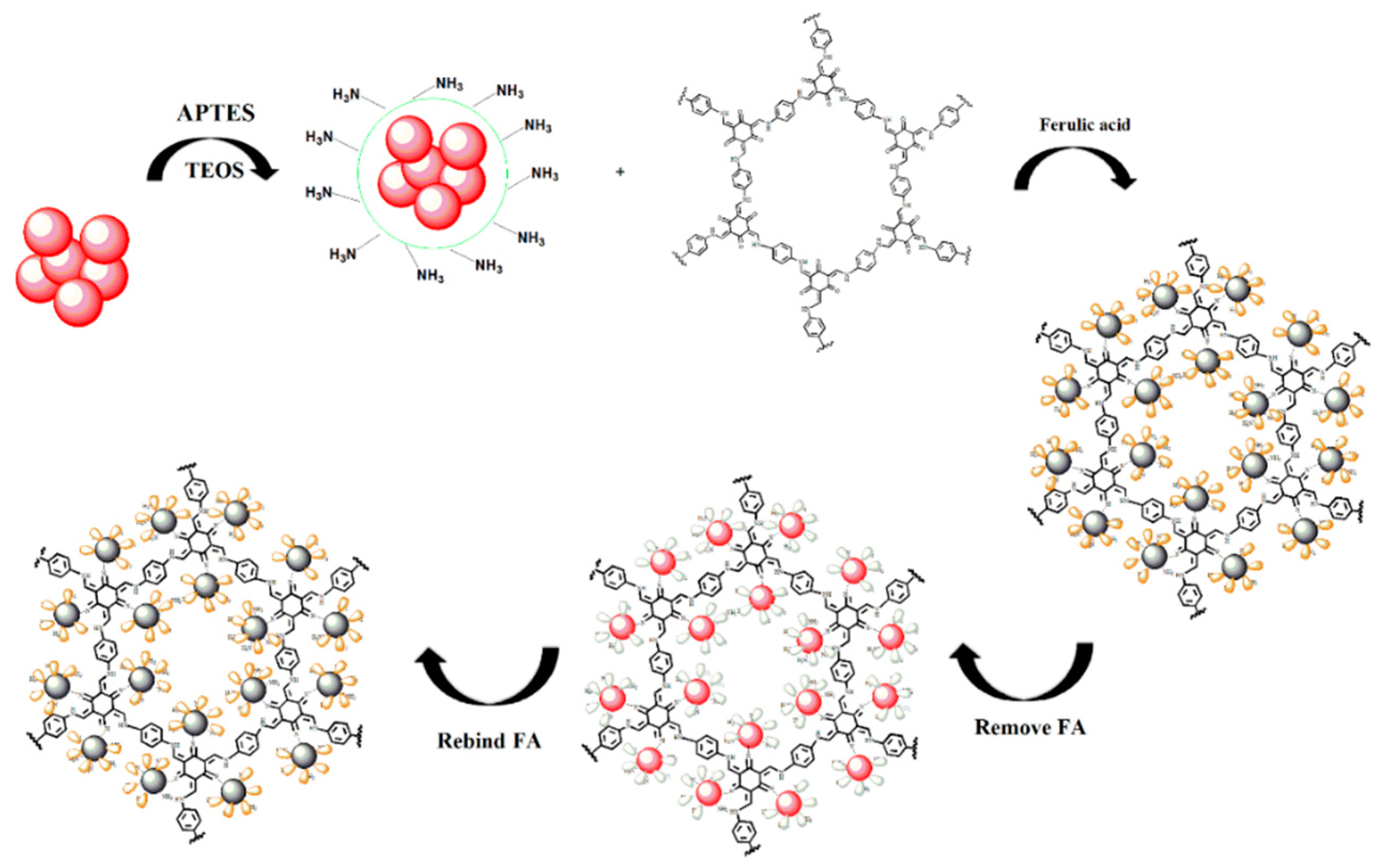
2.3. Preparation of MIPs Based on QD-Grafted COFs
2.4. Fluorescence Analysis of MIPs Based on QD-Grafted COFs
2.5. Procedures for SPE Enrichment Coupled with HPLC
3. Results
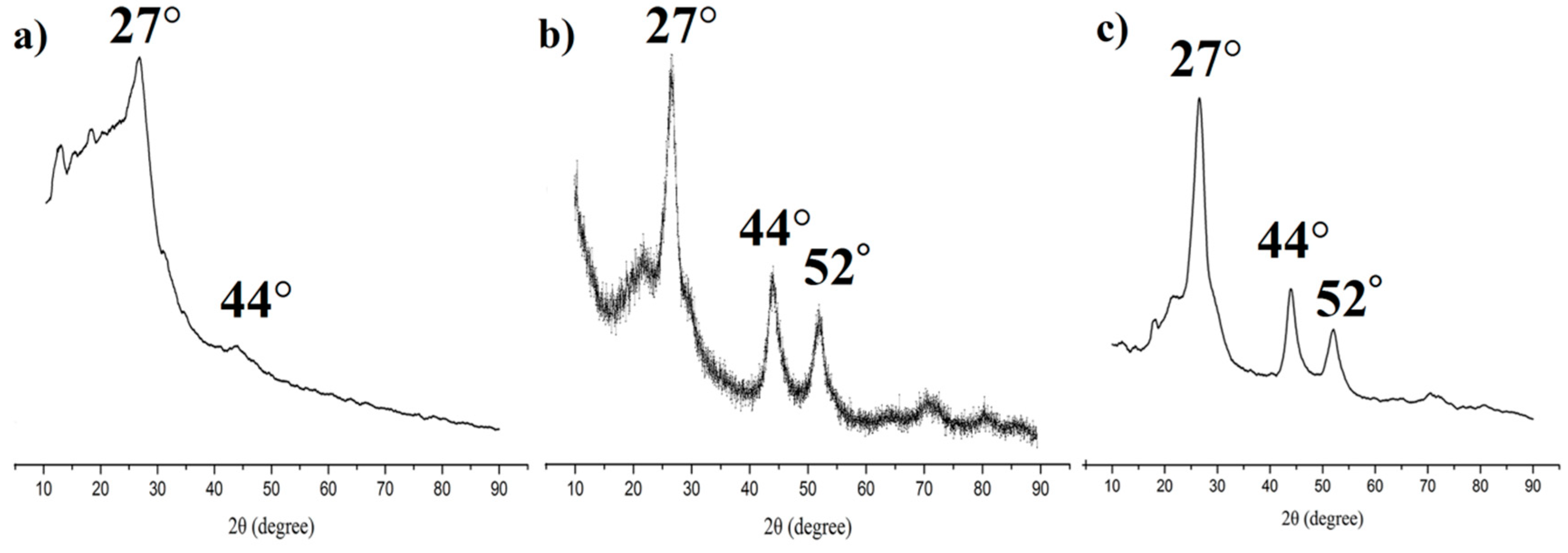
3.1. Synthesis and Characterization of the MIPs Based on QD-Grafted COFs
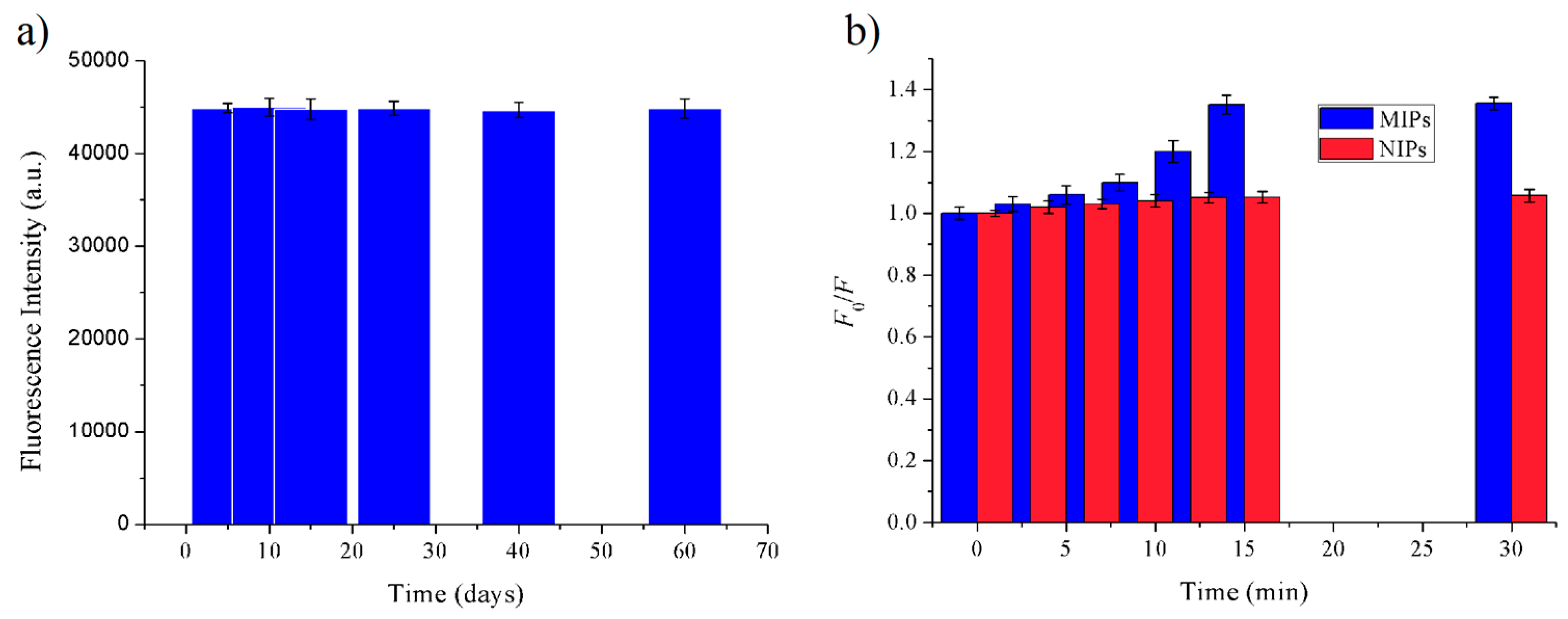
3.2. Fluorescent Properties of MIPs Based on QD-Grafted COFs
3.3. Adsorption Properties
3.3.1. Adsorption Time of MIPs Based on QD-Grafted COFs for FA Molecules
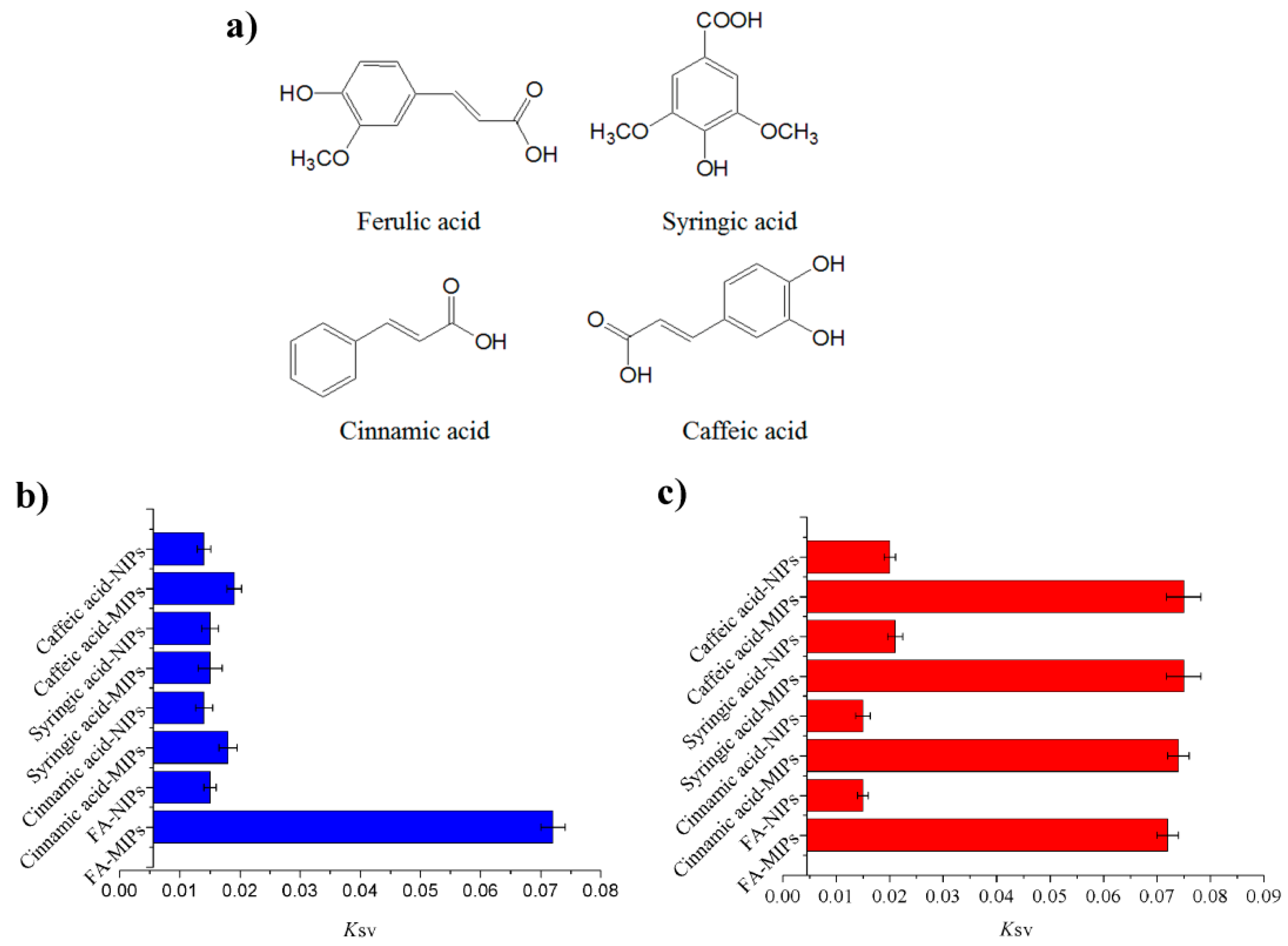
3.3.2. Selectivity of the MIPs Based on QD-Grafted COFs
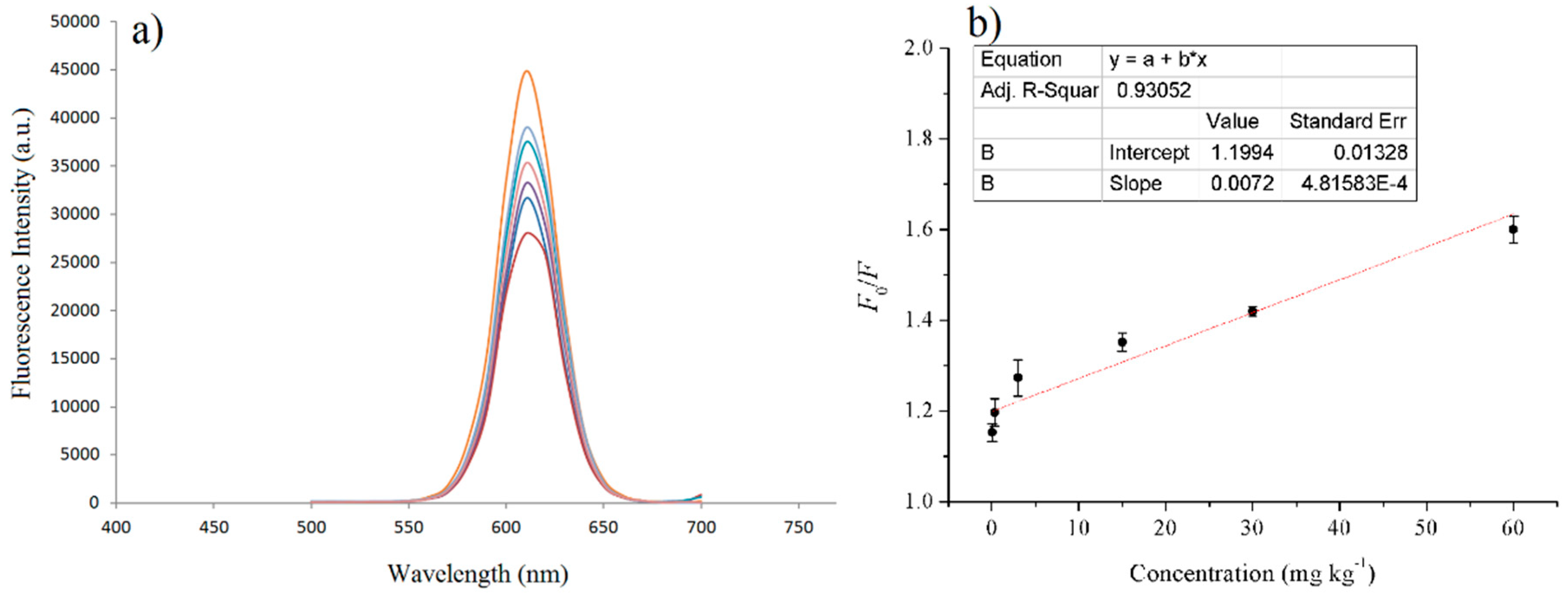
3.4. The Standard Curve and the Detection Limit of MIPs Based on QD-Grafted COFs
3.5. FA Detection in Real Samples
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Peng, X. An essay on synthetic chemistry of colloidal nanocrystals. Nano Res. 2009, 2, 425–447. [Google Scholar] [CrossRef]
- Zhang, H.; Cui, Z.; Wang, Y.; Zhang, K.; Ji, X.; Lü, C.; Yang, B.; Gao, M. From water-soluble CdTe nanocrystals to fluorescent nanocrystal–polymer transparent composites using polymerizable surfactants. Adv. Mater. 2003, 15, 777–780. [Google Scholar] [CrossRef]
- Dai, X.; Zhang, Z.; Jin, Y.; Niu, Y.; Cao, H.; Liang, X.; Chen, L.; Wang, J.; Peng, X. Solution-processed, high-performance light-emitting diodes based on quantum dots. Nature 2014, 515, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Susha, A.S.; Javier, A.M.; Parak, W.J.; Rogach, A.L. Luminescent CdTe nanocrystals as ion probes and pH sensors in aqueous solutions. Colloids Surf. A Physicochem. Eng. Asp. 2006, 281, 40–43. [Google Scholar] [CrossRef]
- Liu, J.; Yang, X.; He, X.; Wang, K.; Wang, Q.; Guo, Q.; Shi, H.; Huang, J.; Huo, X. Fluorescent nanoparticles for chemical and biological sensing. Sci. China Chem. 2011, 54, 1157. [Google Scholar] [CrossRef]
- Zhang, W.; He, X.-W.; Chen, Y.; Li, W.-Y.; Zhang, Y.-K. Composite of CdTe quantum dots and molecularly imprinted polymer as a sensing material for cytochrome c. Biosens. Bioelectron. 2011, 26, 2553–2558. [Google Scholar] [CrossRef] [PubMed]
- The Huy, B.; Seo, M.-H.; Zhang, X.; Lee, Y.-I. Selective optosensing of clenbuterol and melamine using molecularly imprinted polymer-capped CdTe quantum dots. Biosens. Bioelectron. 2014, 57, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Liu, H.; Geng, W.; Wang, Y. A dual-function molecularly imprinted optopolymer based on quantum dots-grafted covalent-organic frameworks for the sensitive detection of tyramine in fermented meat products. Food Chem. 2019, 277, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Ni, T.; Mu, L.; Zhang, D.; Wang, J.; Wang, S.; Sun, B. Sensitive detection of pyrraline with a molecularly imprinted sensor based on metal-organic frameworks and quantum dots. Sens. Actuators B Chem. 2018, 256, 1038–1044. [Google Scholar] [CrossRef]
- Huang, C.-P.; Li, Y.-K.; Chen, T.-M. A highly sensitive system for urea detection by using CdSe/ZnS core-shell quantum dots. Biosens. Bioelectron. 2007, 22, 1835–1838. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Han, C. Sonochemical synthesis of cyclodextrin-coated quantum dots for optical detection of pollutant phenols in water. Chem. Mater. 2008, 20, 6053–6059. [Google Scholar] [CrossRef]
- Liu, H.; Zhou, K.; Wu, D.; Wang, J.; Sun, B. A novel quantum dots-labeled on the surface of molecularly imprinted polymer for turn-off optosensing of dicyandiamide in dairy products. Biosens. Bioelectron. 2016, 77, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Côté, A.P.; Benin, A.I.; Ockwig, N.W.; Keeffe, M.; Matzger, A.J.; Yaghi, O.M. Porous, crystalline, covalent organic frameworks. Science 2005, 310, 1166–1170. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, Y.; Xia, H.; Mu, Y.; Liu, X. A robust and luminescent covalent organic framework as a highly sensitive and selective sensor for the detection of Cu2+ ions. Chem. Commun. 2016, 52, 6613–6616. [Google Scholar] [CrossRef] [PubMed]
- Waller, P.J.; Gándara, F.; Yaghi, O.M. Chemistry of covalent organic frameworks. Acc. Chem. Res. 2015, 48, 3053–3063. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.-Y.; Wang, W. Covalent organic frameworks (COFs): From design to applications. Chem. Soc. Rev. 2013, 42, 548–568. [Google Scholar] [CrossRef] [PubMed]
- Kandambeth, S.; Biswal, B.P.; Chaudhari, H.D.; Rout, K.C.; Kunjattu, H.S.; Mitra, S.; Karak, S.; Das, A.; Mukherjee, R.; Kharul, U.K.; et al. Selective molecular sieving in self-standing porous covalent-organic-framework membranes. Adv. Mater. 2017, 29, 1603945. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Xu, F.; Sun, B.; Fu, R.; He, H.; Matyjaszewski, K. Design and preparation of porous polymers. Chem. Rev. 2012, 112, 3959–4015. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Bonde, S.J.; Kamra, T.; Bülow, L.; Leo, J.C.; Linke, D.; Ye, L. Bacterial imprinting at pickering emulsion interfaces. Angew. Chem. Int. Ed. 2014, 53, 10687–10690. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Gao, J.; Liu, X.; Gao, X.; Ma, T.; Lu, X.; Li, J. Ultrasensitive detection of clenbuterol by a covalent imprinted polymer as a biomimetic antibody. Food Chem. 2017, 228, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Lan, J.; Gao, X.; Liu, X.; Zhang, D.; Wei, L.; Gao, Z.; Li, J. Determination of clenbuterol in pork and potable water samples by molecularly imprinted polymer through the use of covalent imprinting method. Food Chem. 2016, 190, 952–959. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, M.; Sudheer, A.R.; Menon, V.P. Ferulic acid: Therapeutic potential through its antioxidant property. J. Clin. Biochem. Nutr. 2007, 40, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Urbaniak, A.; Szelag, M.; Molski, M. Theoretical investigation of stereochemistry and solvent influence on antioxidant activity of ferulic acid. Comput. Theor. Chem. 2013, 1012, 33–40. [Google Scholar] [CrossRef]
- Lima, E.; Flores, J.; Cruz, A.S.; Leyva-Gómez, G.; Krötzsch, E. Controlled release of ferulic acid from a hybrid hydrotalcite and its application as an antioxidant for human fibroblasts. Microporous Mesoporous Mater. 2013, 181, 1–7. [Google Scholar] [CrossRef]
- Chung, S.Y.; Champagne, E.T. Ferulic acid enhances IgE binding to peanut allergens in Western blots. Food Chem. 2011, 124, 1639–1642. [Google Scholar] [CrossRef]
- Sgarbossa, A.; Monti, S.; Lenci, F.; Bramanti, E.; Bizzarri, R.; Barone, V. The effects of ferulic acid on β-amyloid fibrillar structures investigated through experimental and computational techniques. Biochim. Biophys. Acta 2013, 1830, 2924–2937. [Google Scholar] [CrossRef] [PubMed]
- Ni, T.; Zhang, D.; Wang, J.; Wang, S.; Liu, H.; Sun, B. Grafting of quantum dots on covalent organic frameworks via a reverse microemulsion for highly selective and sensitive protein optosensing. Sens. Actuators B Chem. 2018, 269, 340–345. [Google Scholar] [CrossRef]
- Liu, H.; Fang, G.; Wang, S. Molecularly imprinted optosensing material based on hydrophobic CdSe quantum dots via a reverse microemulsion for specific recognition of ractopamine. Biosens. Bioelectron. 2014, 55, 127–132. [Google Scholar] [CrossRef] [PubMed]

| The samples | Fluorescence method | SPE-HPLC/MS method | ||
|---|---|---|---|---|
| Content (mg kg−1) | RSD (%) | Content (mg kg−1) | RSD (%) | |
| Highland barley bran | 3.10 ± 0.12 | 3.87% | 3.11 ± 0.09 | 2.89% |
| Wheat bran | 1.64 ± 0.04 | 2.44% | 1.63 ± 0.03 | 1.84% |
| Corn silk | 2.33 ± 0.06 | 2.58% | 2.33 ± 0.04 | 1.72% |
| Vinasse | 1.67 ± 0.08 | 4.79% | 1.66 ± 0.03 | 1.81% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Wang, Y.; Liu, H. A Novel Fluorescence and SPE Adsorption Nanomaterials of Molecularly Imprinted Polymers Based on Quantum Dot-Grafted Covalent Organic Frameworks for the High Selectivity and Sensitivity Detection of Ferulic Acid. Nanomaterials 2019, 9, 305. https://doi.org/10.3390/nano9020305
Wang Y, Wang Y, Liu H. A Novel Fluorescence and SPE Adsorption Nanomaterials of Molecularly Imprinted Polymers Based on Quantum Dot-Grafted Covalent Organic Frameworks for the High Selectivity and Sensitivity Detection of Ferulic Acid. Nanomaterials. 2019; 9(2):305. https://doi.org/10.3390/nano9020305
Chicago/Turabian StyleWang, Yu, Yuzhen Wang, and Huilin Liu. 2019. "A Novel Fluorescence and SPE Adsorption Nanomaterials of Molecularly Imprinted Polymers Based on Quantum Dot-Grafted Covalent Organic Frameworks for the High Selectivity and Sensitivity Detection of Ferulic Acid" Nanomaterials 9, no. 2: 305. https://doi.org/10.3390/nano9020305
APA StyleWang, Y., Wang, Y., & Liu, H. (2019). A Novel Fluorescence and SPE Adsorption Nanomaterials of Molecularly Imprinted Polymers Based on Quantum Dot-Grafted Covalent Organic Frameworks for the High Selectivity and Sensitivity Detection of Ferulic Acid. Nanomaterials, 9(2), 305. https://doi.org/10.3390/nano9020305





