Growth Kinetics of the Selected Intermetallic Phases in Ni/Al/Ni System with Various Nickel Substrate Microstructure
Abstract
:1. Introduction
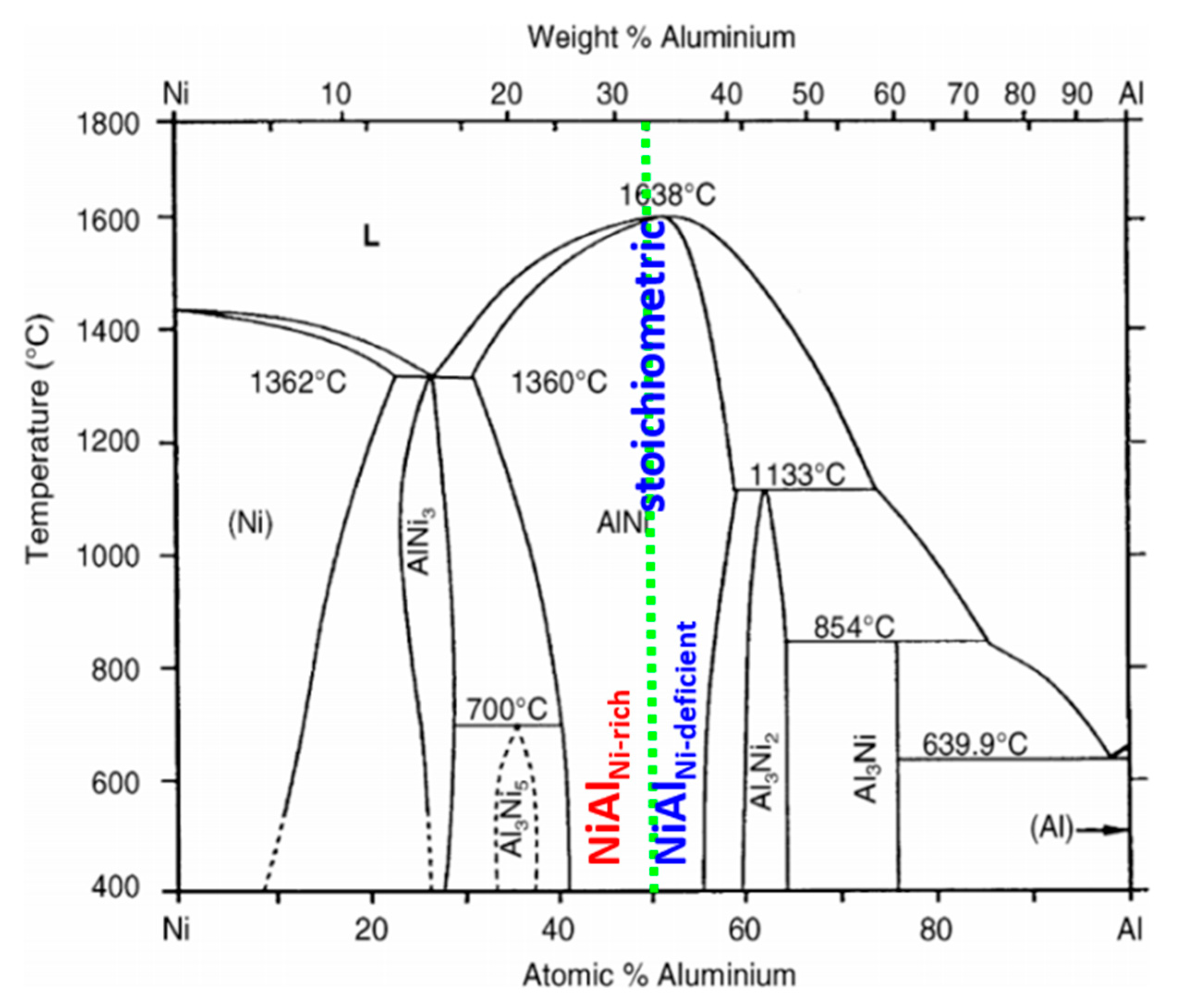
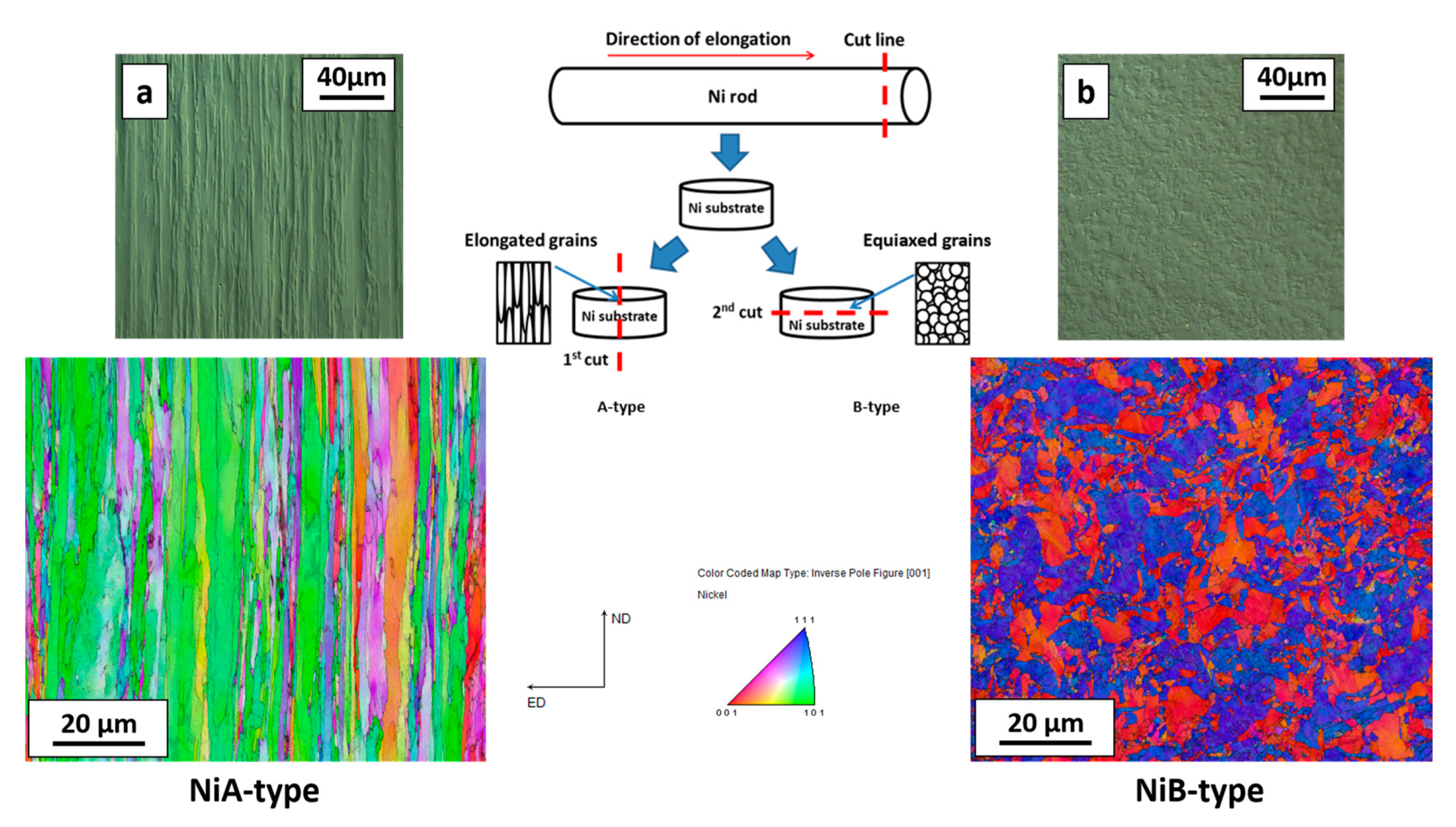
2. Materials and Methods
3. Results and Discussion
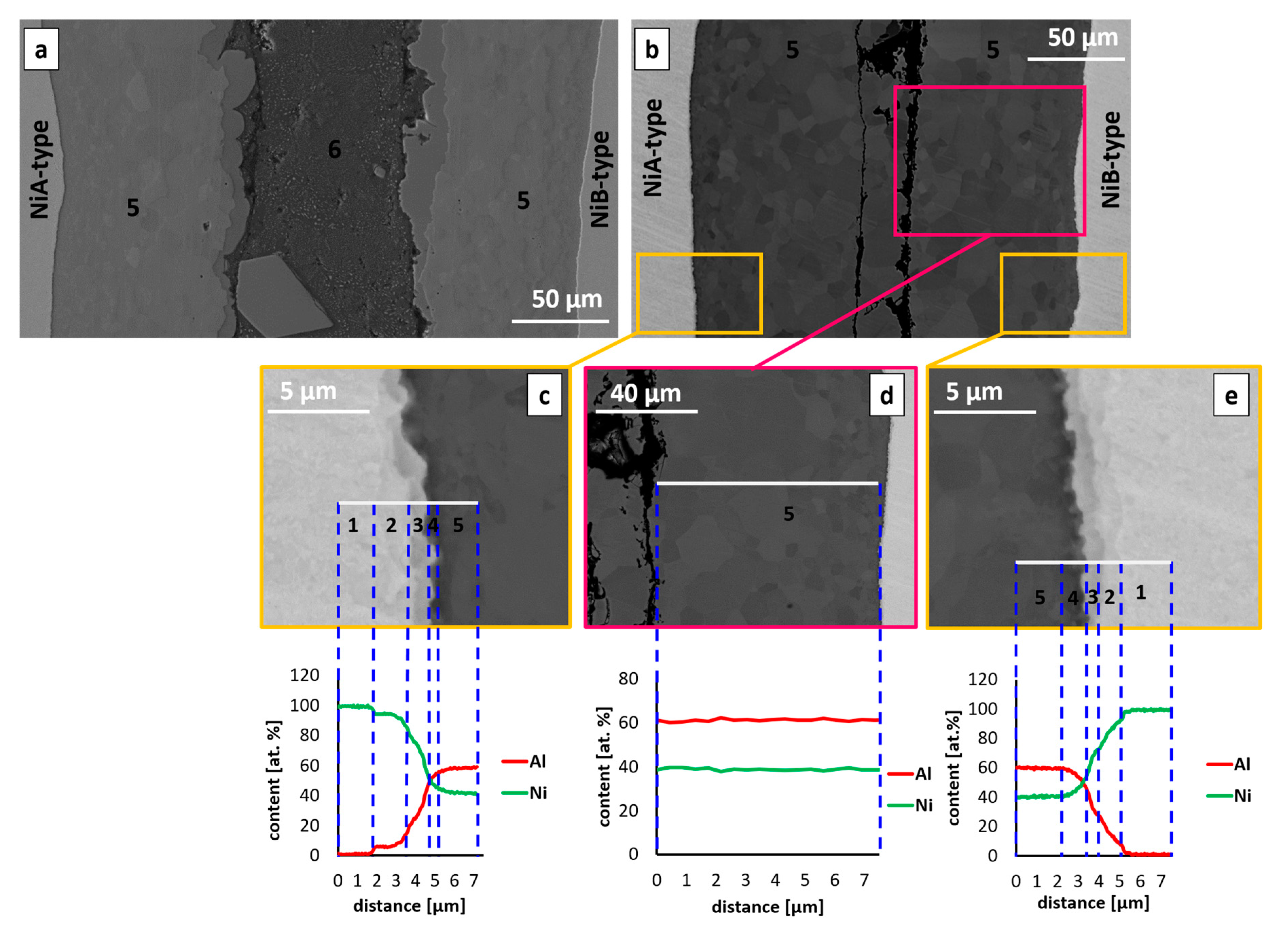
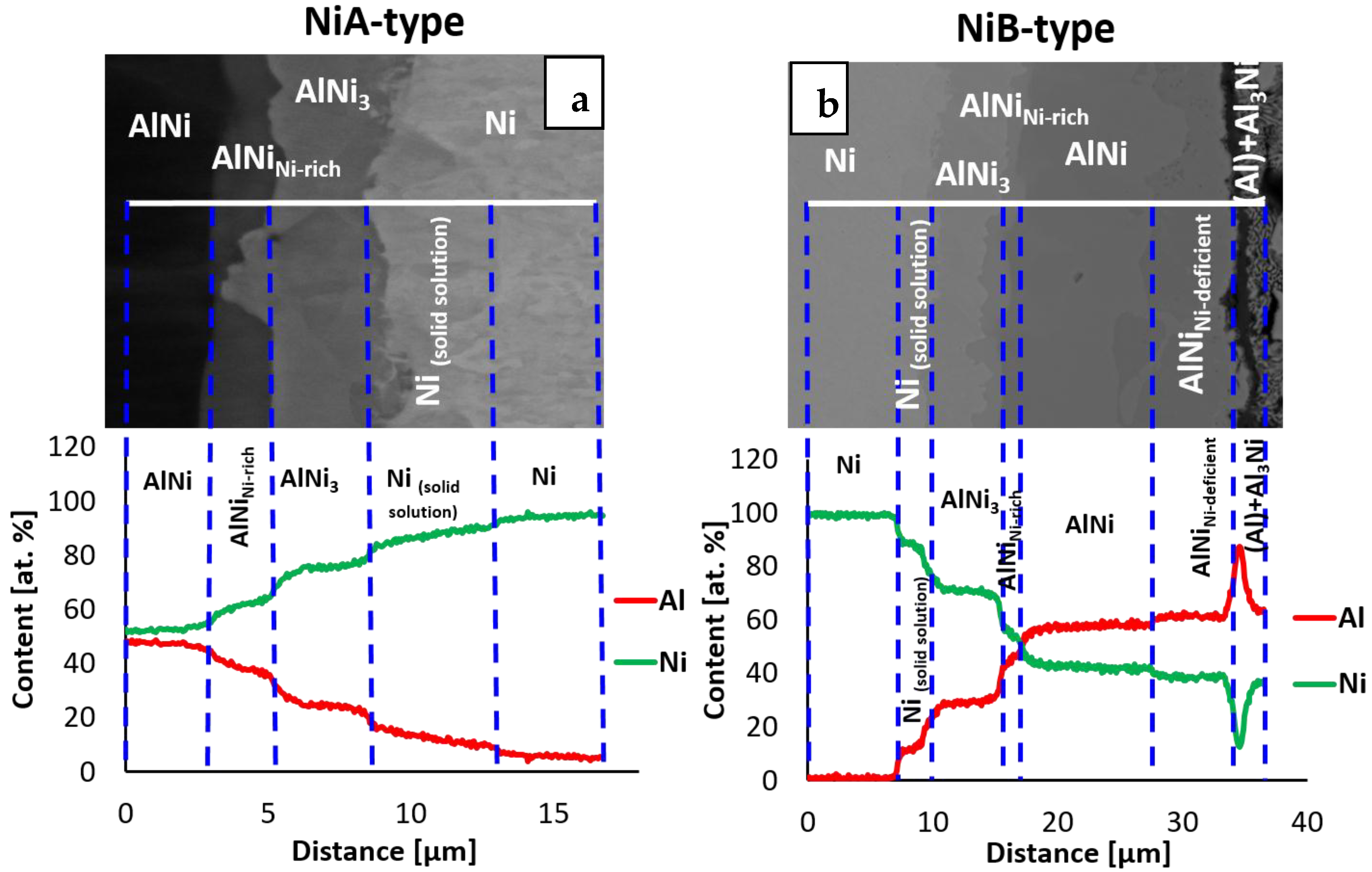
3.1. Sequence of Intermetallic Phases in Interconnections
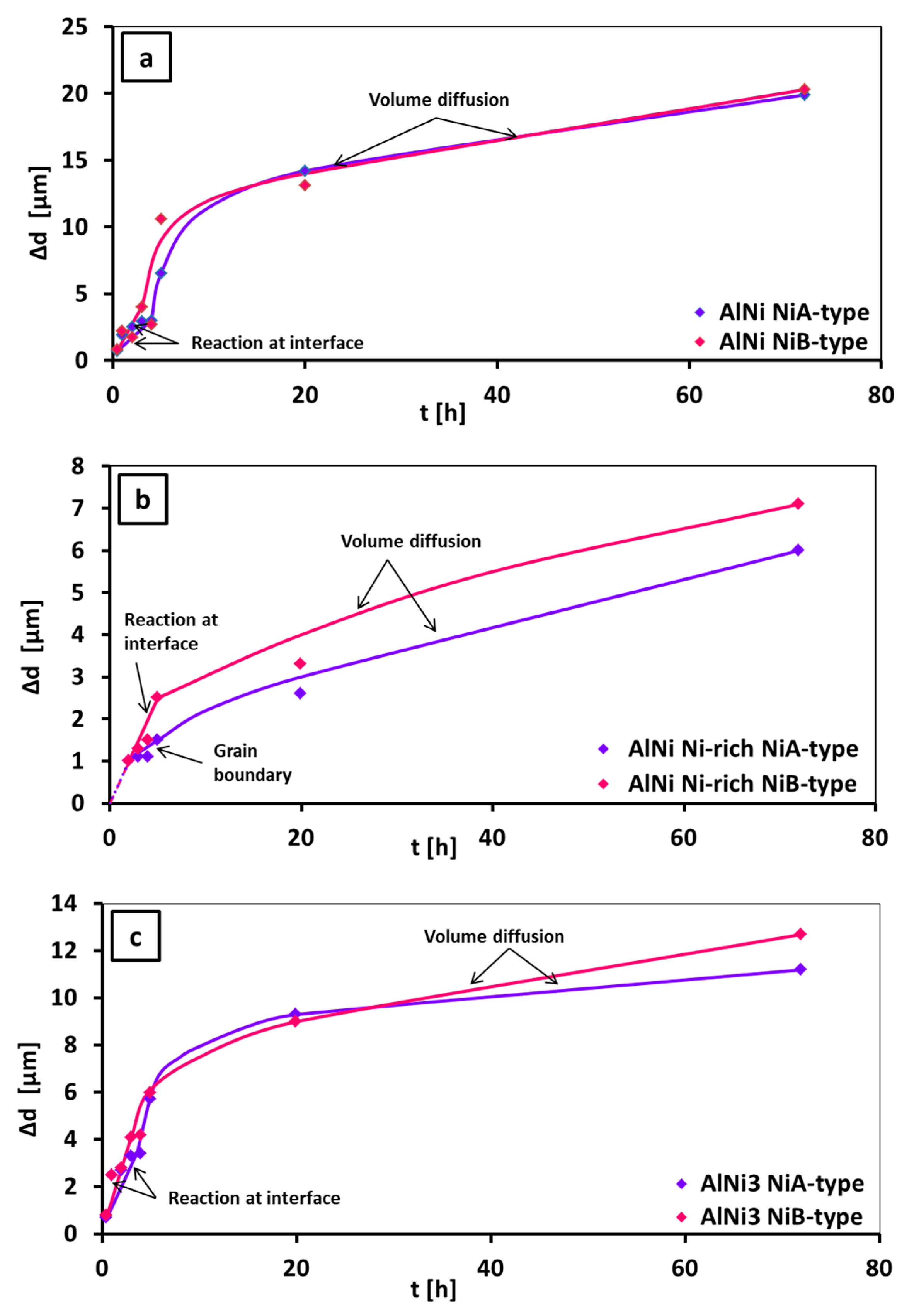
3.2. Growth Rate of the Intermetallic Phases
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Miracle, D.B. The physical and mechanical properties of NiAl. Acta Metal. Mater. 1993, 41, 649–684. [Google Scholar] [CrossRef]
- Liu, C.T.; Stiegler, J.O. Ductile ordered intermetallic alloys. Science 1984, 226, 636–642. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.B.; Minamino, Y.; Yamane, T.; Saji, S. Reaction diffusion and formation of Al3Ni and Al3Ni2 phases in the Al-Ni system. J. Mater. Sci. Lett. 1993, 12, 1684–1686. [Google Scholar] [CrossRef]
- Bradley, A.J.; Taylor, A. An X-ray of the nickel-aluminium system. Proc. R. Soc. Lond. 1937, 159, 56–72. [Google Scholar]
- Kodentsov, A. Interdiffusion studies in β- and γ’-intermetallic phases of the binary Ni-Al system. Diffus. Fund. 2017, 13, 56–97. [Google Scholar] [CrossRef]
- Shankar, S.; Seigle, L.L. Interdiffusion and intrinsic diffusion in the NiAl (δ) phase of the AL-Ni system. Metal. Trans. A 1978, 9A, 1467–1476. [Google Scholar] [CrossRef]
- Yang, T.Y.; Wu, S.K.; Shiue, R.K. Interfacial reaction of infrared brazed NiAl/Al/NiAl and Ni3Al/Al/Ni3Al joints. Intermetallics 2001, 9, 341–347. [Google Scholar] [CrossRef]
- Tsao, C.L.; Chen, S.W. Interfacial reaction in the liquid diffusion couples of Mg/Ni, Al/Ni, Al/(Ni)-Al2O3 systems. J. Mater. Sci. 1995, 30, 5215–5222. [Google Scholar] [CrossRef]
- Castleman, L.S.; Froot, H.A. Nucleation of Intermetallic Phases in Aluminium-Nickel Diffusion couples. Trans. ASM 1963, 56, 205–208. [Google Scholar]
- Lieblich, M.; Gonzalez-Crrasco, J.L.; Caruna, G. Thermal stability of an Al/Ni3 composite processed by powder metallurgy. Intermetallics 1997, 5, 515–524. [Google Scholar] [CrossRef]
- Gong, X.; Ma, Y.; Guo, H.; Gong, S. Effect of thermal cycling on microstructure evolution and elements diffusion behavior near the interface of Ni/NiAl diffusion couple. J. Alloy. Compd. 2015, 642, 117–123. [Google Scholar] [CrossRef]
- Fujiwara, K.; Horita, Z. Measurement of intrinsic diffusion coefficients of Al and Ni in Ni3Al using Ni/NiAl diffusion couples. Acta Mater. 2002, 50, 1571–1579. [Google Scholar] [CrossRef]
- Lopez, G.A.; Sommadossi, S.; Zieba, P.; Gust, W.; Mittemeijer, E.J. Kinetic behavior of diffusion-soldered Ni/Al/Ni interconnections. Mater. Chem. Phys. 2002, 78, 459–463. [Google Scholar] [CrossRef]
- Tumminello, S.; Sommadossi, S. Growth Kinetics of Intermetallic Phases in Transient Liquid Phase Bonding Process (TLPB) in Al/Ni System. Defect. Diffus. Froum. 2012, 323–325, 465–470. [Google Scholar] [CrossRef]
- Urrutia, A.; Tumminello, S.; Lames, D.G.; Sommadossi, S. X-Ray Characterization of intermetallic phases in Al/Ni multilayer system. Proc. Mat. Sci. 2015, 8, 1150–1159. [Google Scholar] [CrossRef]
- Maj, L.; Morgiel, J. In-situ transmission electron microscopy observations of nucleation and growth of intermetallic phases during reaction of Ni(V)/Al multilayers. Thin Solid Films 2017, 621, 165–170. [Google Scholar] [CrossRef]
- Maj, L.; Morgiel, J.; Mars, K.; Grzegorek, J.; Faryna, M.; Godlewska, E. Microstructure and hardness of Ti6Al4V/NiAl/Ti6Al4V joints obtained through resistive heating. J. Mater. Process. Tech. 2018, 255, 689–695. [Google Scholar] [CrossRef]
- Simoes, S.; Viana, F.; Kocak, M.; Ramos, A.S.; Viera, M.T.; Viera, M.F. Diffusion bonding of TiAl using reactive Ni/Al nanolayers and Ti and Ni foils. Mater. Chem. Phys. 2011, 128, 202–207. [Google Scholar] [CrossRef]
- Konieczny, M. Mechanical properties and deformation behavior of laminated Ni-(Ni2Al3+NiAl3) and Ni-(Ni3Al+NiAl) composites. Mater. Sci. Eng. 2013, 586, 11–18. [Google Scholar] [CrossRef]
- Noro, J.; Ramos, A.S.; Viera, M.T. Intermettallic phase formation in nanometric Ni/Al multilayer thin films. Intermetallics 2008, 16, 1061–1065. [Google Scholar] [CrossRef]
- Brunelli, K.; Peruzzo, L.; Dabala, M. The effect of prolonged heat treatments on the microstructuralevolution of Al/Ni intermetallic compounds in multi layered composites. Mater. Chem. Phys. 2014, 149–150, 350–358. [Google Scholar]
- Wojewoda, J.; Lopez, G.A.; Zieba, P.; Mittemeijer, E.J. Diffusion processes in diffusion-soldered interconnections. Arch. Metall. Mater. 2004, 49, 277–291. [Google Scholar]
- Humpston, G.; Jacobson, D.M.; Sangha, S.P.S. Diffusion soldering for electronics manufacturing. Endeavour 1994, 18, 55–60. [Google Scholar] [CrossRef]
- Zhou, Y. Introduction to diffusion soldering/brazing. In Microjoining and Nanojoining; Zhou, Y., Ed.; Woodhead Publishing Limited: Cambridge, UK, 2008; Volume 1, pp. 269–298. [Google Scholar]
- Nash, P.; Singleton, M.F.; Murray, J.L. Phase Diagrams of Binary Nickel Alloys; Nash, P., Ed.; ASM International: Materials Park, OH, USA, 1991; pp. 3–11. [Google Scholar]
- Bouche, K.; Barbier, F.; Coulet, A. Phase Formation During Dissolution of Nickel in Liquid Aluminium. Z. Metallkd. 1997, 88, 446–451. [Google Scholar]
- Wolczynski, W.; Guzik, E.; Janczak-Rusch, J.; Kopycinski, D.; Golczewski, J.; Mo Lee, H.; Kloch, J. Morphological characteristics of multi-layer/substrate systems. Mater. Charact. 2006, 56, 274–280. [Google Scholar] [CrossRef]
- Wolczynski, W.; Okane, T.; Senderowski, C.; Kania, B.; Zasada, D.; Janczak-Rusch, J. Meta-Stable Conditions of Diffusion Brazing. Arch. Metall. Mater. 2011, 56, 311–323. [Google Scholar] [CrossRef]
- Ding, Z.; Hu, Q.; Lu, W.; Sun, S.; Xia, M.; Li, J. In situ observation on the formation of intermetallics compounds at the interface of liquid Al/ solid Ni. Scripta Mater. 2017, 130, 214–218. [Google Scholar] [CrossRef]
- Tseng, Y.C.; Lee, H.; Tsai, S.C.; Yen, Y.W.; Chen, C.M. Suppression effect of Ni grain size on the Ni3Sn4 growth at the Sn/Ni interface. Mater. Charact. 2017, 128, 232–273. [Google Scholar] [CrossRef]
- Ainutdinov, F.A.; Khairidinov, S.K.; Vakhobov, A.V. Doklady Akademii Nauk Tadzhiskoj SSR 3; Akademija Nauk SSR: Moscow, Russia, 1987; Volume 30, pp. 169–172. [Google Scholar]
- Pieraggi, B. Calculations of Parabolic Reaction Rate Constants. Oxid. Met. 1987, 27, 177–185. [Google Scholar] [CrossRef]







| Conditions: | Temperature 720 °C, Vacuum, Slight Mechanical Pressure, Heating 18 °C/min Cooling with Furnace | ||
|---|---|---|---|
| No. | Sample Type | Time of Annealing [h] | |
| 1. | NiA/Al/NiB | 0.25 | |
| 2. | NiA/Al/NiB | 0.50 | |
| 3. | NiA/Al/NiA | NiB/Al/NiB | 1 |
| 4. | NiA/Al/NiB | 2 | |
| 5. | NiA/Al/NiA | NiB/Al/NiB | 3 |
| 6. | NiA/Al/NiB | 4 | |
| 7. | NiA/Al/NiA | NiB/Al/NiB | 5 |
| 8. | NiA/Al/NiB | 20 | |
| 9. | NiA/Al/NiB | 72 | |
| Phase | Present Work | Tumminello et al. [14] |
|---|---|---|
| Thickness [μm] | ||
| Whole interconnection zone | 280 | 140 |
| Eutectics Al + Al3Ni | 86 | 56 |
| Al3Ni | 15 | 9 |
| Al3Ni2 | 82 | 33 |
| Phase | The Range of Aluminum Content (at. %) in Interconnection | Average Content of Aluminum in the Interconnection (at. %) |
|---|---|---|
| Ni solid | 5.5–12.1 ± 0.2–0.5 | 8.9 ± 0.2 |
| AlNi3 | 21.6–30.5 ± 0.4–0.6 | 26.1 ± 0.5 |
| AlNiNi-rich | 33.4–42.0 ± 0.7–0.8 | 39.1 ± 0.8 |
| AlNi | 47.1–53.5 ± 0.9–1.1 | 50.6 ± 1.0 |
| AlNiNi-deficient | 53.7–57.8 ± 1.1–1.2 | 55.9 ± 1.1 |
| Al3Ni2 | 58.2–60.7 ± 1.2 | 59.6 ± 1.2 |
| Al3Ni | 75.5–76.3 ± 1.5 | 76.0 ± 1.5 |
| No. | Method | SEM | TEM | ||
|---|---|---|---|---|---|
| Phase | Content at. % | ||||
| Ni | Al | Ni | Al | ||
| 1. | Ni solid solution | 94.4 ± 1.9 | 5.6 ± 1.9 | 91.9 ± 1.8 | 8.1 ± 1.8 |
| 2. | AlNi3 | 78.4 ± 1.6 | 21.6 ± 1.6 | 78.4 ± 1.6 | 21.6 ± 1.6 |
| 3. | AlNiNi-rich | 66.6 ± 1.3 | 33.4 ± 1.3 | 60.8 ± 1.2 | 39.2 ± 1.2 |
| 4. | AlNi | 50.1 ± 1.0 | 49.9 ± 1.0 | 51.9 ± 1.0 | 48.1 ± 1.0 |
| 5. | AlNiNi-deficient | 45.6 ± 1.0 | 54.4 ± 1.0 | 45.8 ± 0.9 | 54.2 ± 0.9 |
| Time [h] | Ni-Type | Layer Thickness, Δd [μm] | |||||
|---|---|---|---|---|---|---|---|
| Al3Ni | Al3Ni2 | AlNiNi-deficient | AlNi | AlNiNirich | AlNi3 | ||
| 0.25 | A | 16.9 | 87.8 | - | - | - | - |
| B | 12.9 | 76.5 | - | - | - | - | |
| 0.50 | A | - | 94.2 | - | 0.7 | - | 0.7 |
| B | - | 96.3 | - | 0.8 | - | 0.8 | |
| 1 | A | - | 40.2 | - | 1.9 | - | 2.5 |
| B | - | 50.4 | - | 2.2 | - | 2.5 | |
| 2 | A | - | 108.7 | - | 2.5 | 1.0 | 2.7 |
| B | - | 103.7 | - | 1.7 | 1.0 | 2.8 | |
| 3 | A | - | - | 48.1 | 2.9 | 1.1 | 3.3 |
| B | - | - | 38.1 | 4 | 1.3 | 4.1 | |
| 4 | A | - | - | 77.6 | 3 | 1.1 | 3.4 |
| B | - | - | 45.5 | 2.7 | 1.5 | 4.2 | |
| 5 | A | - | - | - | 6.5 | 1.5 | 5.7 |
| B | - | - | 7.2 | 10.6 | 2.5 | 6 | |
| 20 | A | - | - | 7.5 | 14.2 | 2.6 | 9.3 |
| B | - | - | 10.5 | 13.1 | 3.3 | 9 | |
| 72 | A | - | - | - | 19.9 | 6.0 | 11.2 |
| B | - | - | - | 20.3 | 7.1 | 12.7 | |
| Ni A-Type | n | k | Mechanism | Ni B-Type | n | k | Mechanism |
|---|---|---|---|---|---|---|---|
| AlNi | 0.67 | mixed: volume diffusion and chemical reaction at interface | AlNi | 0.65 | mixed: volume diffusion and chemical reaction at interface | ||
| AlNiNi-rich | 0.51 | volume diffusion | AlNiNi-rich | 0.52 | volume diffusion | ||
| AlNi3 | 0.51 | volume diffusion | AlNi3 | 0.50 | volume diffusion |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwiecien, I.; Bobrowski, P.; Wierzbicka-Miernik, A.; Litynska-Dobrzynska, L.; Wojewoda-Budka, J. Growth Kinetics of the Selected Intermetallic Phases in Ni/Al/Ni System with Various Nickel Substrate Microstructure. Nanomaterials 2019, 9, 134. https://doi.org/10.3390/nano9020134
Kwiecien I, Bobrowski P, Wierzbicka-Miernik A, Litynska-Dobrzynska L, Wojewoda-Budka J. Growth Kinetics of the Selected Intermetallic Phases in Ni/Al/Ni System with Various Nickel Substrate Microstructure. Nanomaterials. 2019; 9(2):134. https://doi.org/10.3390/nano9020134
Chicago/Turabian StyleKwiecien, Izabella, Piotr Bobrowski, Anna Wierzbicka-Miernik, Lidia Litynska-Dobrzynska, and Joanna Wojewoda-Budka. 2019. "Growth Kinetics of the Selected Intermetallic Phases in Ni/Al/Ni System with Various Nickel Substrate Microstructure" Nanomaterials 9, no. 2: 134. https://doi.org/10.3390/nano9020134
APA StyleKwiecien, I., Bobrowski, P., Wierzbicka-Miernik, A., Litynska-Dobrzynska, L., & Wojewoda-Budka, J. (2019). Growth Kinetics of the Selected Intermetallic Phases in Ni/Al/Ni System with Various Nickel Substrate Microstructure. Nanomaterials, 9(2), 134. https://doi.org/10.3390/nano9020134




