Comparative Molecular Immunological Activity of Physiological Metal Oxide Nanoparticle and its Anticancer Peptide and RNA Complexes
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cytokine Panel
2.3. Ex Vivo Splenocyte Assay
2.4. High Throughput Tumor Proteomics Analysis
2.5. Molecular Dynamics
2.6. IL-6 Assay
2.7. MTT Assay
3. Results and Discussion
3.1. In Vitro Immunogenicity
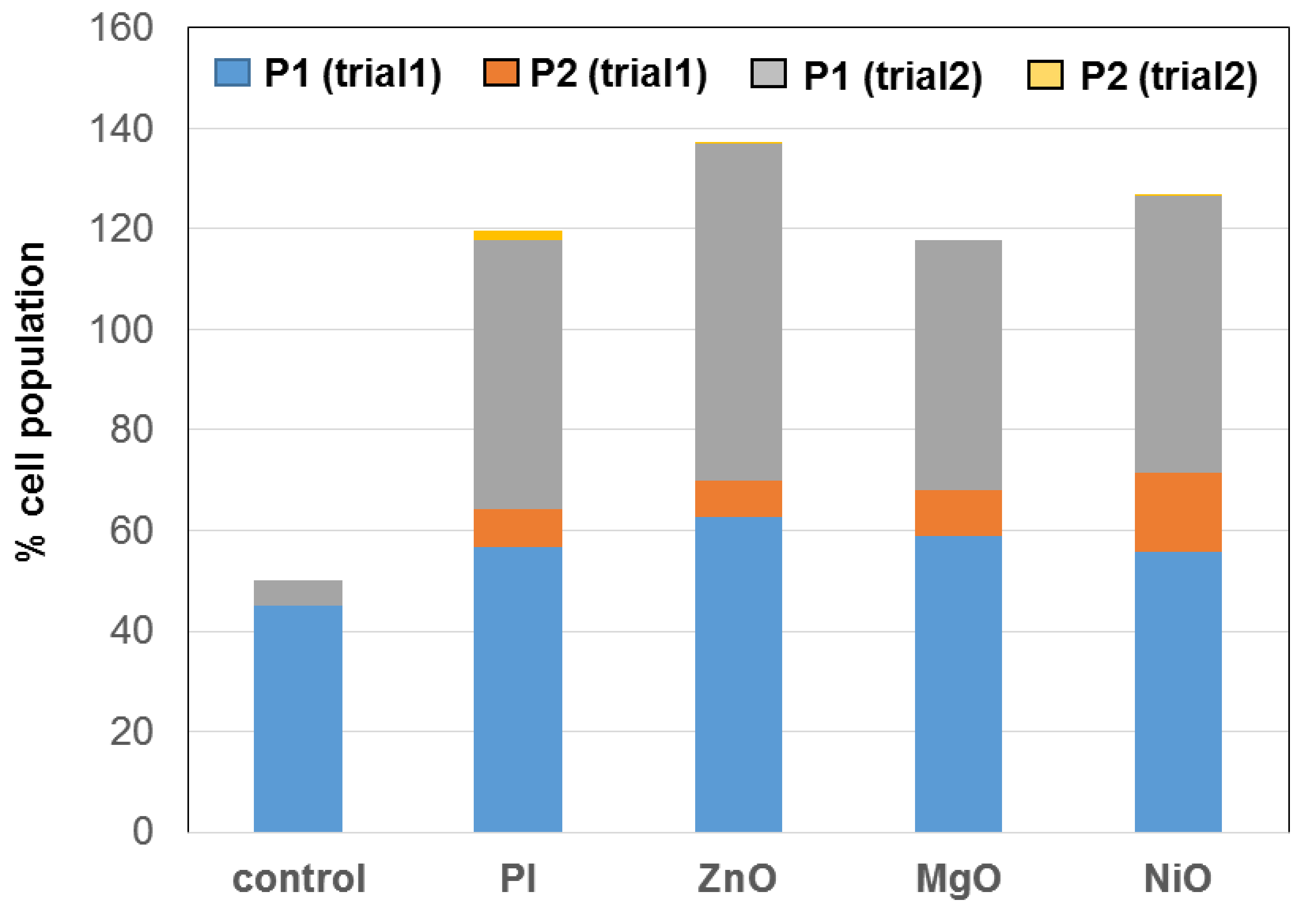
3.2. Ex Vivo Splenocyte Assay
3.3. In Vivo Direct Intratumoral Injection Model
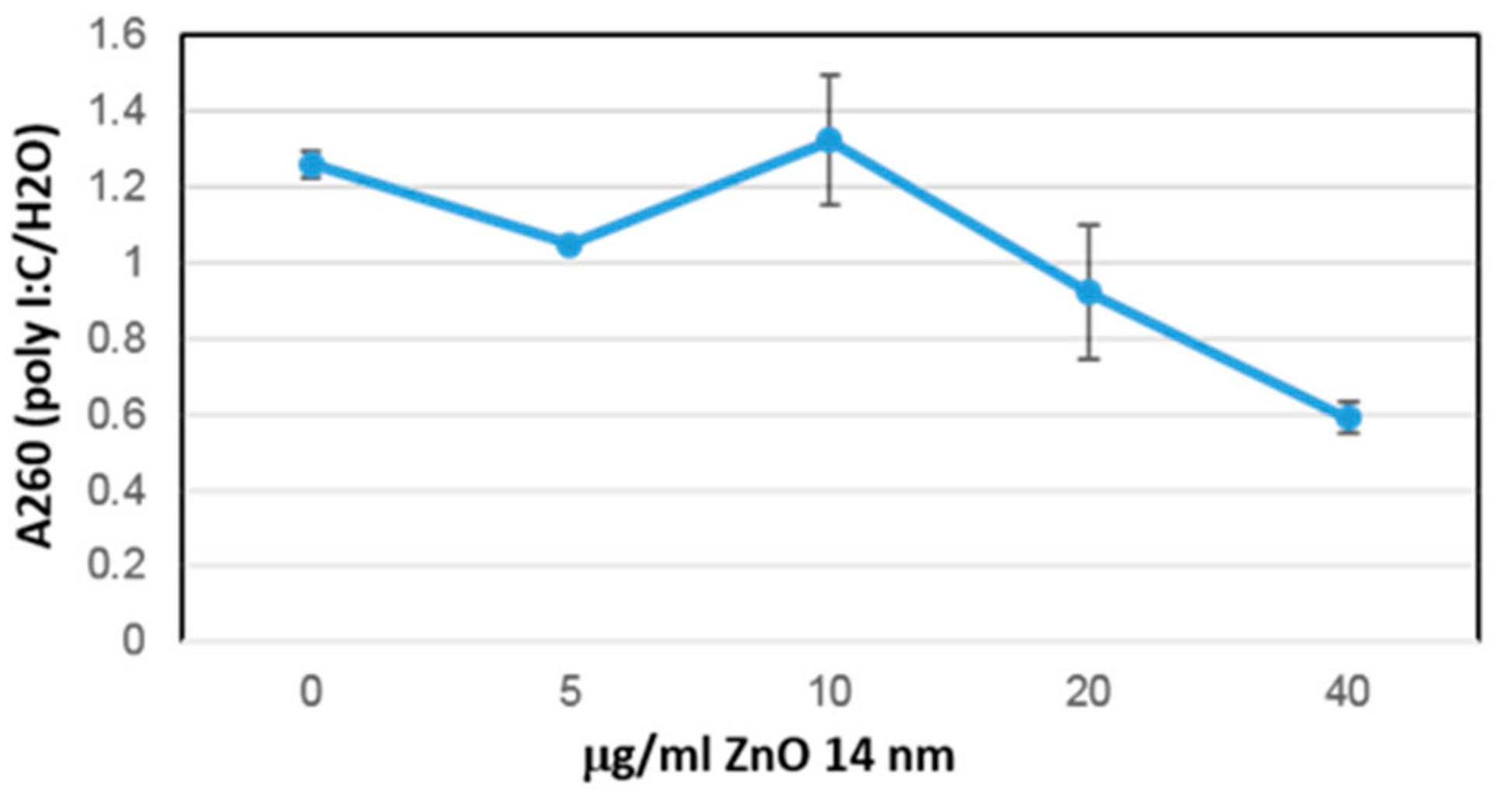
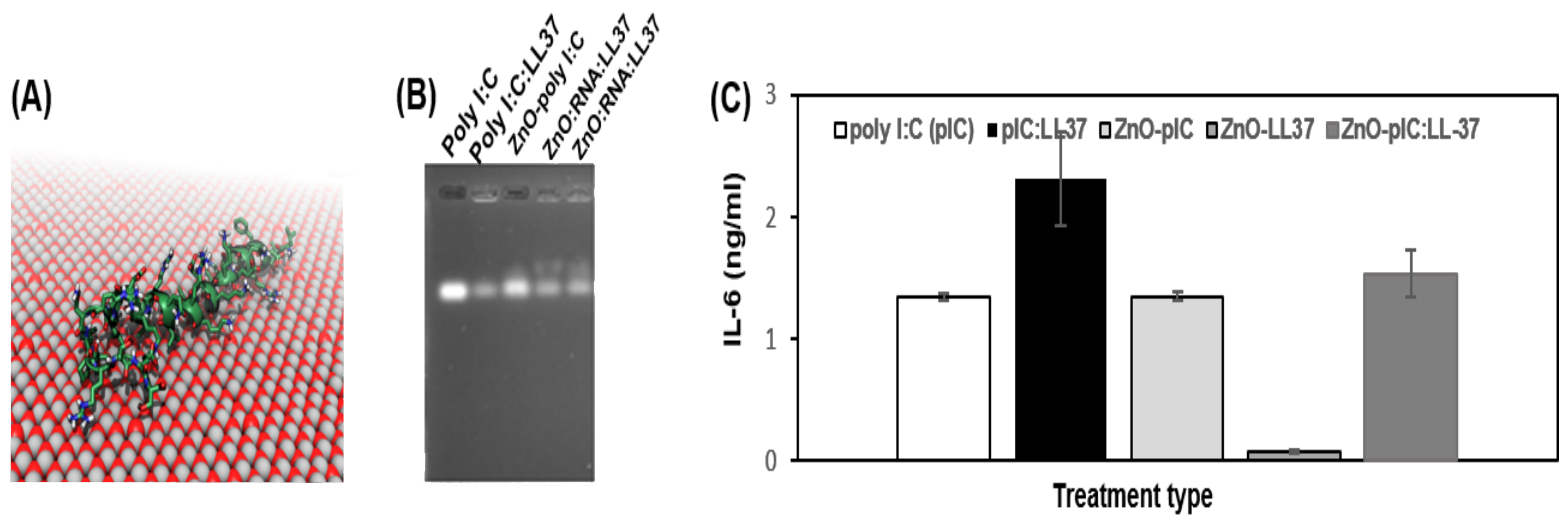
3.4. Immunological Activity of ZnO NP Antimicrobial Peptide Complexes
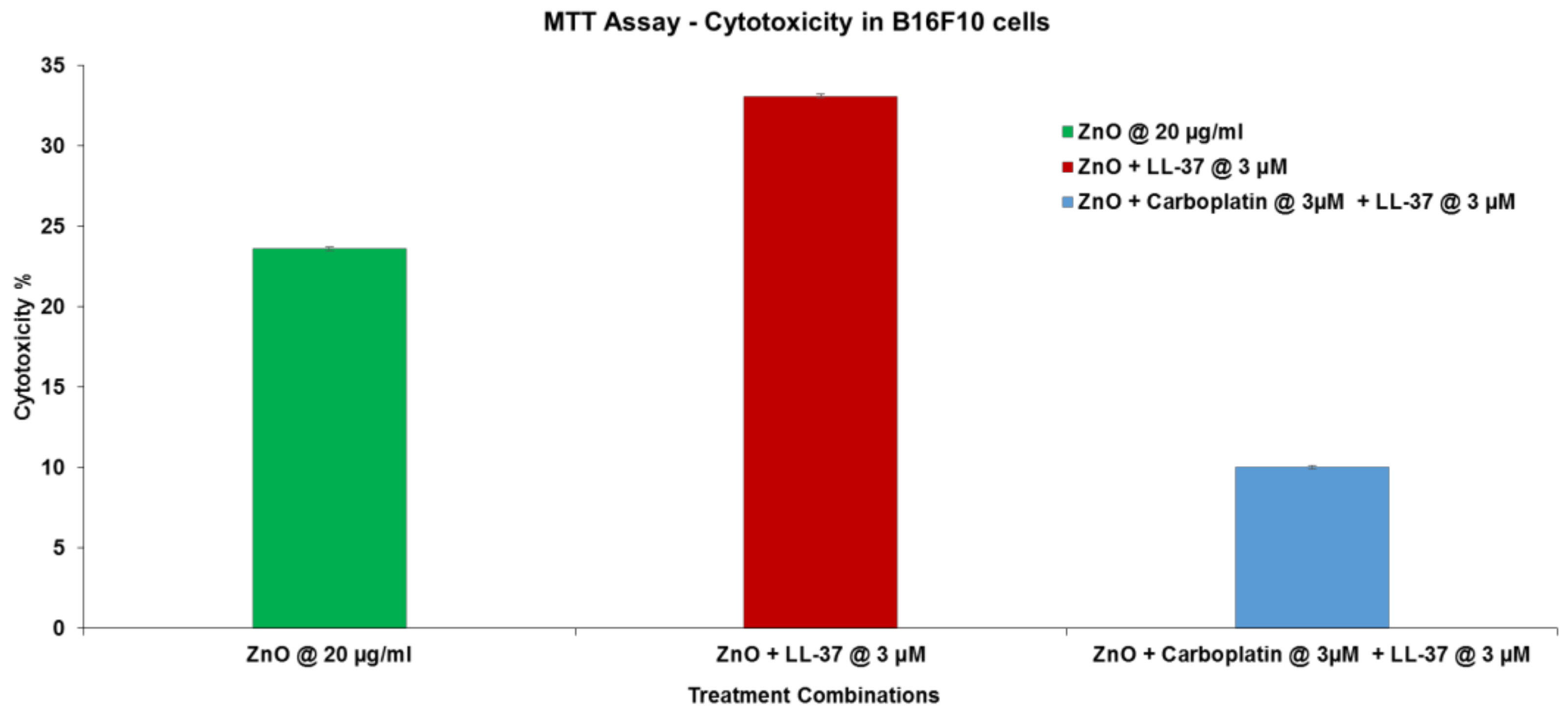
3.5. Combination Therapy Experiment
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kauffman, K.J.; Dorkin, J.R.; Yang, J.H.; Heartlein, M.W.; DeRosa, F.; Mir, F.F.; Fenton, O.S.; Anderson, D.G. Optimization of Lipid Nanoparticle Formulations for mRNA Delivery in Vivo with Fractional Factorial and Definitive Screening Designs. Nano Lett. 2015, 15, 7300–7306. [Google Scholar] [CrossRef]
- Oberli, M.A.; Reichmuth, A.M.; Dorkin, J.R.; Mitchell, M.J.; Fenton, O.S.; Jaklenec, A.; Anderson, D.G.; Langer, R.; Blankschtein, D. Lipid Nanoparticle Assisted mRNA Delivery for Potent Cancer Immunotherapy. Nano Lett. 2017, 17, 1326–1335. [Google Scholar] [CrossRef]
- DeLong, R.K.; Curtis, C.B. Toward RNA nanoparticle vaccines: Synergizing RNA and inorganic nanoparticles to achieve immunopotentiation. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, e1415. [Google Scholar] [CrossRef]
- Gondan, A.I.B.; Ruiz-de-Angulo, A.; Zabaleta, A.; Blanco, N.G.; Cobaleda-Siles, B.M.; García-Granda, M.J.; Padro, D.; Llop, J.; Arnaiz, B.; Gato, M.; et al. Effective cancer immunotherapy in mice by polyIC-imiquimod complexes and engineered magnetic nanoparticles. Biomaterials 2018, 170, 95–115. [Google Scholar] [CrossRef]
- Cobaleda-Siles, M.; Henriksen-Lacey, M.; Ruiz de Angulo, A.; Bernecker, A.; Gómez Vallejo, V.; Szczupak, B.; Llop, J.; Pastor, G.; Plaza-Garcia, S.; Jauregui-Osoro, M.; et al. An iron oxide nanocarrier for dsRNA to target lymph nodes and strongly activate cells of the immune system. Small 2014, 10, 5054–5067. [Google Scholar] [CrossRef]
- Kumari, N.; Dwarakanath, B.S.; Das, A.; Bhatt, A.N. Role of interleukin-6 in cancer progression and therapeutic resistance. Tumor Biol. 2016, 7, 11553–11572. [Google Scholar] [CrossRef]
- Shao, K.; Singha, S.; Clemente-Casares, X.; Tsai, S.; Yang, Y.; Santamaria, P. Nanoparticle-based immunotherapy for cancer. ACS Nano. 2015, 9, 16–30. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.; Das, M.; Dwivedi, P.D. Toxicological mode of action of ZnO nanoparticles: Impact on immune cells. Mol. Immunol. 2015, 63, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Delong, R.K.; Risor, A.; Kanomata, M.; Laymon, A.; Jones, B.; Zimmerman, S.D.; Williams, J.; Witkowski, C.; Warner, M.; Ruff, M.; et al. Characterization of biomolecular nanoconjugates by high-throughput delivery and spectroscopic difference. Nanomedicine 2012, 7, 1851–1862. [Google Scholar] [CrossRef] [PubMed]
- Medberry, P.; Dennis, S.; Van Hecke, T.; DeLong, R.K. pDNA bioparticles: Comparative heterogeneity, surface, binding, and activity analyses. Biochem. Biophys. Res. Commun. 2004, 319, 426–432. [Google Scholar] [CrossRef] [PubMed]
- DeLong, R.K.; Akhtar, U.; Sallee, M.; Parker, B.; Barber, S.; Zhang, J.; Craig, M.; Garrad, R.; Hickey, A.J.; Engstrom, E. Characterization and performance of nucleic acid nanoparticles combined with protamine and gold. Biomaterials 2009, 30, 6451–6459. [Google Scholar] [CrossRef] [PubMed]
- Knowle, R.; Werner, A.; DeLong, R.K. R4 peptide-pDNA nanoparticle coated HepB vaccine microparticles: Sedimentation, partitioning, and spray freeze dry bioprocesses. J. Nanosci. Nanotechnol. 2006, 6, 2783–2789. [Google Scholar] [CrossRef] [PubMed]
- Ramani, M.; Mudge, M.C.; Morris, R.T.; Zhang, Y.; Warcholek, S.A.; Hurst, M.N.; Riviere, J.E.; DeLong, R.K. Zinc Oxide Nanoparticle-Poly I:C RNA Complexes: Implication as Therapeutics against Experimental Melanoma. Mol. Pharm. 2017, 14, 614–625. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.R.; Kisin, E.; Inman, A.; Young, S.H.; Muhammed, M.; Burks, T.; Uheida, A.; Tkach, A.; Waltz, M.; Castranova, V.; et al. Oxidative stress and dermal toxicity of iron oxide nanoparticles in vitro. Cell Biochem. Biophys. 2013, 67, 461–476. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. A power primer. Psychol. Bull. 1992, 112, 155. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates Publishers: New York, NY, USA, 1988. [Google Scholar]
- Raymand, D.; van Duin, A.C.; Baudin, M.; Hermansson, K. A reactive force field (ReaxFF) for zinc oxide. Surf. Sci. 2008, 602, 1020–1031. [Google Scholar] [CrossRef]
- Plimpton, S. Fast parallel algorithms for short-range molecular dynamics. J. Comput. Phys. 1995, 117, 1–19. [Google Scholar] [CrossRef]
- Huang, J.; Rauscher, S.; Nawrocki, G.; Ran, T.; Feig, M.; de Groot, B.; Grubmüller, H.; MacKerell, A., Jr. CHARMM36m: An improved force field for folded and intrinsically disordered proteins. Nat. Meth. 2017, 14, 71–73. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, R.; Riviere, J.E.; Comer, J. Extraction of aromatic organochlorines from water by ZnO nanoparticles. submitted.
- Wang, G. Structures of human host defense cathelicidin LL-37 and its smallest antimicrobial peptide KR-12 in lipid micelles. J. Biol. Chem. 2008, 283, 32637–32643. [Google Scholar] [CrossRef]
- Phillips, J.C.; Braun, R.; Wang, W.; Gumbart, J.; Tajkhorshid, E.; Villa, E.; Chipot, C.; Skeel, R.D.; Kale, L.; Schulten, K. Scalable molecular dynamics with NAMD. J. Comput. Chem. 2005, 26, 1781–1802. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, C.W.; Le Grand, S.; Walker, R.C.; Roitberg, A.E. Long-time-step molecular dynamics through hydrogen mass repartitioning. J. Chem. Theory Comput. 2015, 11, 1864–1874. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, S.; Kollman, P.A. SETTLE: An analytical version of the SHAKE and RATTLE algorithm for rigid water models. J. Comput. Chem. 1992, 13, 952–962. [Google Scholar] [CrossRef]
- Feller, S.E.; Zhang, Y.H.; Pastor, R.W.; Brooks, B.R. Constant pressure molecular dynamics simulations—The Langevin piston method. J. Chem. Phys. 1995, 103, 4613–4621. [Google Scholar] [CrossRef]
- Singh, D.; Qi, R.; Jordan, J.L.; San Mateo, L.; Kao, C.C. The human antimicrobial peptide LL-37, but not the mouse ortholog, mCRAMP, can stimulate signaling by poly(I:C) through a FPRL1-dependent pathway. J. Biol. Chem. 2013, 288, 8258–8268. [Google Scholar] [CrossRef]
- Thomas, S.E.; Comer, J.; Kim, M.J.; Marroquin, S.; Murthy, V.; Ramani, M.; Hopke, T.G.; McCall, J.; Choi, S.O.; DeLong, R.K. Comparative functional dynamics studies on the enzyme nano-bio interface. Int. J. Nanomed. 2018, 13, 4523–4536. [Google Scholar] [CrossRef]
- Zhang, H.; Ji, Z.; Xia, T.; Meng, H.; Low-Kam, C.; Liu, R.; Pokhrel, S.; Lin, S.; Wang, X.; Liao, Y.P.; et al. Use of metal oxide nanoparticle band gap to develop a predictive paradigm for oxidative stress and acute pulmonary inflammation. ACS Nano. 2012, 6, 4349–4368. [Google Scholar] [CrossRef]
- Filon, F.L.; Crosera, M.; Timeus, E.; Adami, G.; Bovenzi, M.; Ponti, J.; Maina, G. Human skin penetration of cobalt nanoparticles through intact and damaged skin. Toxicol. In Vitro 2013, 27, 121–127. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Dash, S.K.; Ghosh, T.; Das, S.; Tripathy, S.; Mandal, D.; Das, D.; Pramanik, P.; Roy, S. Anticancer and immunostimulatory role of encapsulated tumor antigen containing cobalt oxide nanoparticles. J. Biol. Inorg. Chem. 2013, 18, 957–973. [Google Scholar] [CrossRef]
- Park, E.J.; Oh, S.Y.; Kim, Y.; Yoon, C.; Lee, B.S.; Kim, S.D.; Kim, J.S. Distribution and immunotoxicity by intravenous injection of iron nanoparticles in a murine model. J. Appl. Toxicol. 2016, 36, 414–423. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, J.; Liu, J.; Ling, D.; Xia, J. Functional assembly of protein fragments induced by spatial confinement. PLoS ONE 2015, 10, e0122101. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, A.; Wu, X.; Wang, J.; Brodeur, A.; Thomas, R.; Thakkar, R.; Hadi, H.; Glaspell, G.P.; Duszynski, M.; Wanekaya, A.; et al. Two-Dimensional Fluorescence Difference Spectroscopy of ZnO and Mg Composites in the Detection of Physiological Protein and RNA Interactions. Materials 2017, 10, 1430. [Google Scholar] [CrossRef] [PubMed]
- Ramani, M.; Nguyen, T.D.T.; Aryal, S.; Ghosh, K.C.; DeLong, R.K. Elucidating the RNA Nano-bio Interface: Mechanisms of Anti-Cancer Poly I: C RNA and Zinc Oxide Nanoparticle Interaction. J. Phys. Chem. C 2017, 121, 15702–15710. [Google Scholar] [CrossRef]
- Intratumoral Injections of LL37 for Melanoma. Available online: https://clinicaltrials.gov/ct2/show/NCT02225366 (accessed on 22 August 2019).
- Hasan, M.; Ruksznis, C.; Wang, Y.; Leifer, C. Antimicrobial peptides inhibit polyinosinic-polycytidylic acid-induced immune responses. J. Immunol. 2011, 187, 5653–5659. [Google Scholar] [CrossRef] [PubMed]
- Quagliariello, V.; Masarone, M.; Armenia, E.; Giudice, A.; Barbarisi, M.; Caraglia, M.; Barbarisi, A.; Persico, M. Chitosan-coated liposomes loaded with butyric acid demonstrate anticancer and anti-inflammatory activity in human hepatoma HepG2 cells. Oncol. Rep. 2019, 41, 1476–1486. [Google Scholar] [CrossRef] [PubMed]
- Serri, C.; Quagliariello, V.; Iaffaioli, R.V.; Fusco, S.; Botti, G.; Mayol, L.; Biondi, M. Combination therapy for the treatment of pancreatic cancer through hyaluronic acid-decorated nanoparticles loaded with quercetin and gemcitabine: A preliminary in vitro study. J. Cell Physiol. 2019, 234, 4959–4969. [Google Scholar] [CrossRef]
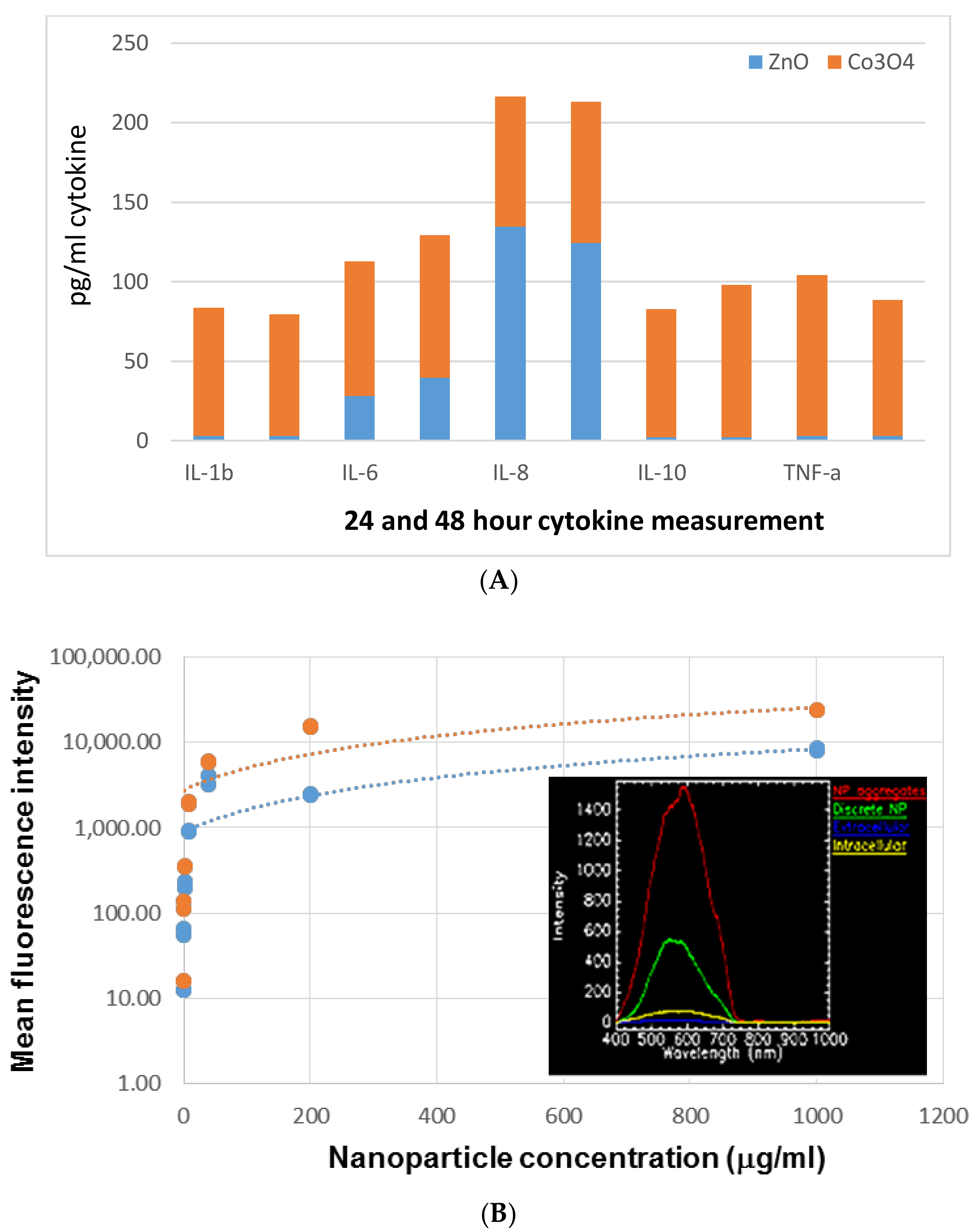
| Cytokine | Putative Role in Metastatic Tumor Niche | Effect of ZnO-Poly I:C (Fold Over Control) | Effect Size Analysis (Cohen’s d Measure) |
|---|---|---|---|
| TNF-α | Chemotaxis, leukocyte recruitment, extracellular killing | 148.3+/−33.7 | −0.59 |
| IFN-γ | Secreted by cancer, T, natural killer (NK) cells and macrophages (Macs), MHCI/II, involved in Th1 and Th2 | 6.0+/−0.92 | −2.81 |
| IL-2 | CD8 and NK activation, activates antigen presentation and B cell response (BCR) | 5.8+/−3.2 | 1.24 |
| IL-12 | Growth factor, increases NK action, stimulates antibody production | 9.9+/−4.3 | 18.58 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
DeLong, R.K.; Comer, J.; Mathew, E.N.; Jaberi-Douraki, M. Comparative Molecular Immunological Activity of Physiological Metal Oxide Nanoparticle and its Anticancer Peptide and RNA Complexes. Nanomaterials 2019, 9, 1670. https://doi.org/10.3390/nano9121670
DeLong RK, Comer J, Mathew EN, Jaberi-Douraki M. Comparative Molecular Immunological Activity of Physiological Metal Oxide Nanoparticle and its Anticancer Peptide and RNA Complexes. Nanomaterials. 2019; 9(12):1670. https://doi.org/10.3390/nano9121670
Chicago/Turabian StyleDeLong, Robert K., Jeffrey Comer, Elza Neelima Mathew, and Majid Jaberi-Douraki. 2019. "Comparative Molecular Immunological Activity of Physiological Metal Oxide Nanoparticle and its Anticancer Peptide and RNA Complexes" Nanomaterials 9, no. 12: 1670. https://doi.org/10.3390/nano9121670
APA StyleDeLong, R. K., Comer, J., Mathew, E. N., & Jaberi-Douraki, M. (2019). Comparative Molecular Immunological Activity of Physiological Metal Oxide Nanoparticle and its Anticancer Peptide and RNA Complexes. Nanomaterials, 9(12), 1670. https://doi.org/10.3390/nano9121670





