Combined Treatments of Magnetic Intra-Lysosomal Hyperthermia with Doxorubicin Promotes Synergistic Anti-Tumoral Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemical Reagents
2.2. Cell Culture
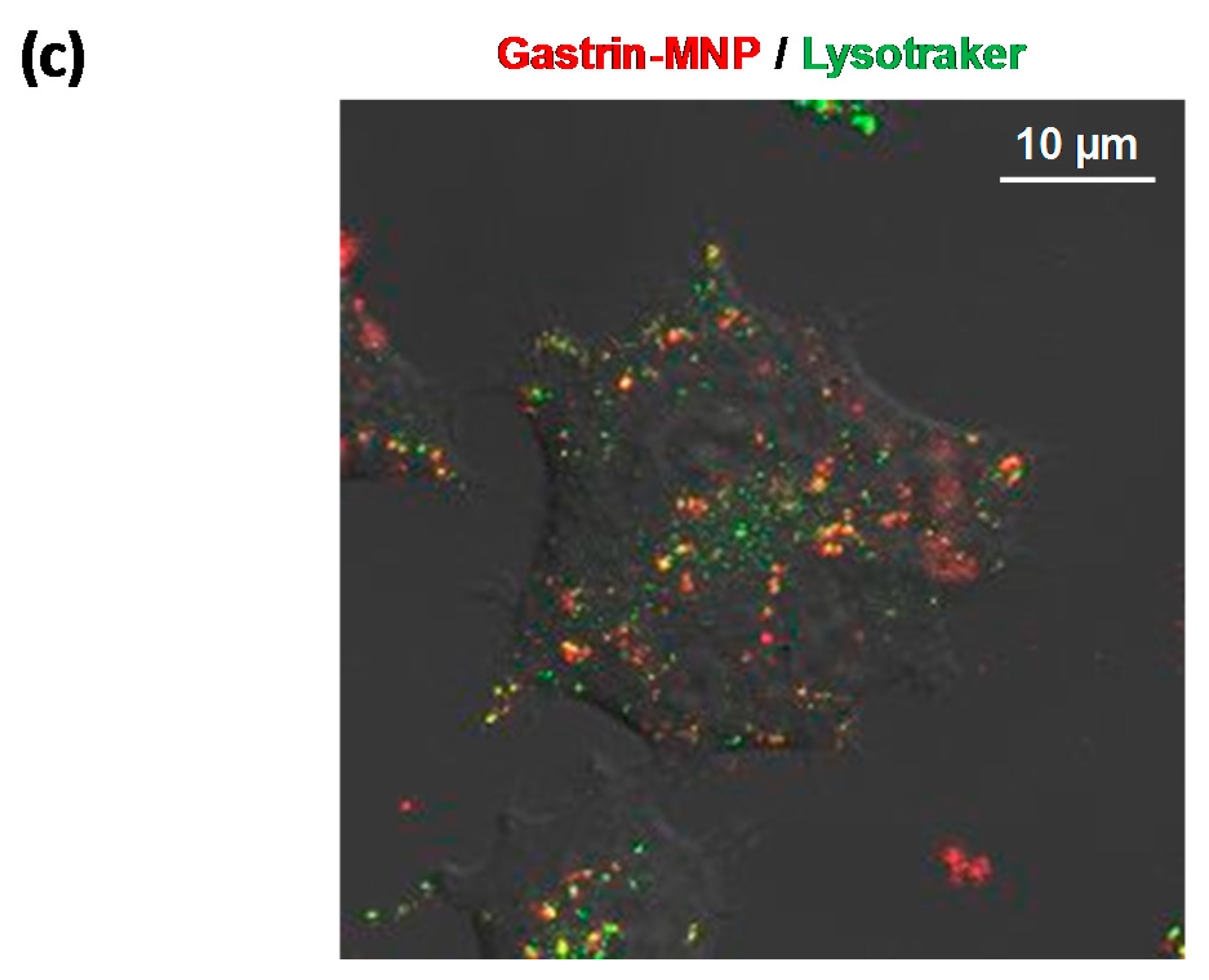
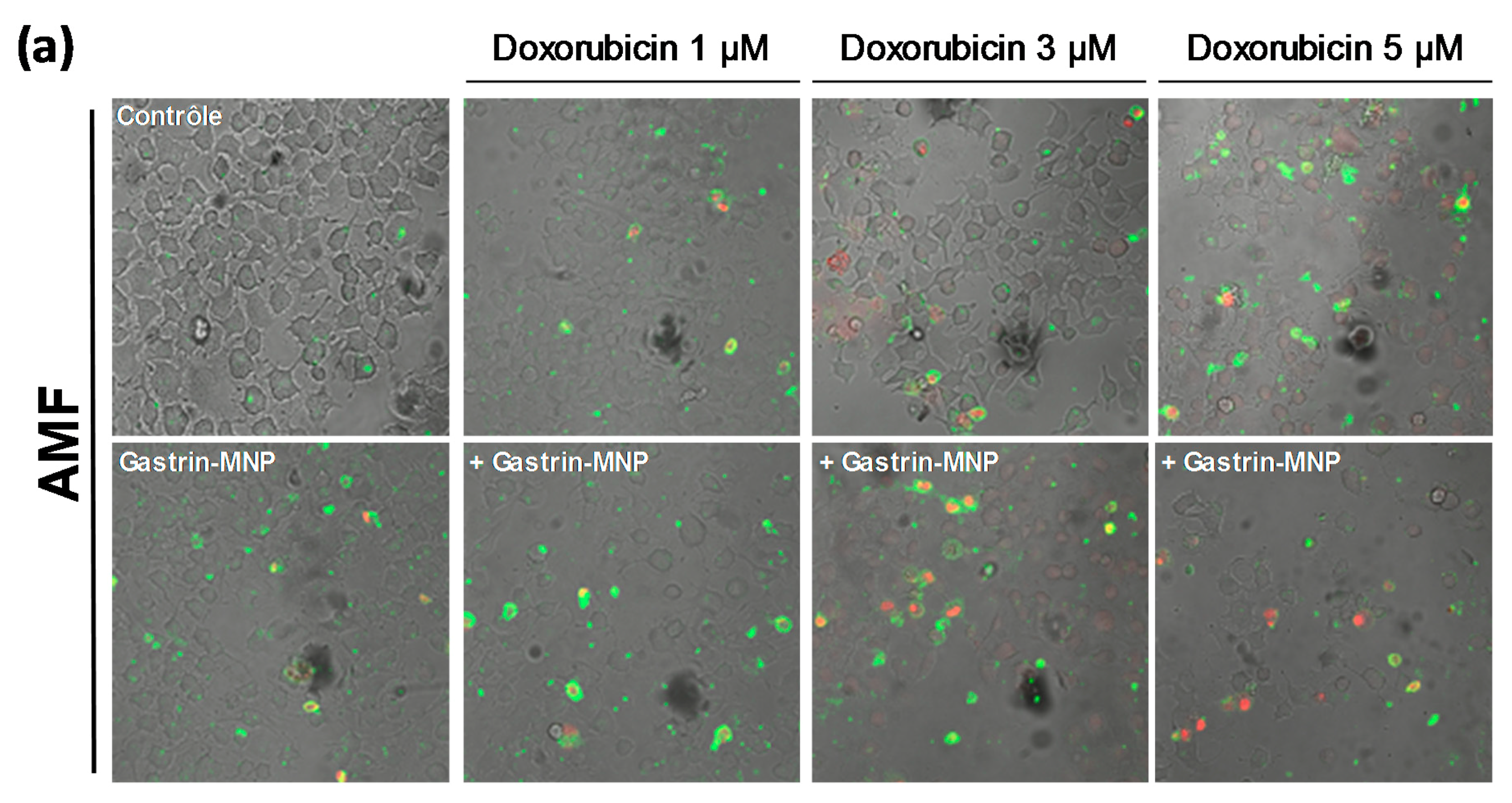
2.3. Gastrin–MNP Localization in Lysosomes by Confocal Microscopy
2.4. Cell Exposure to Alternating Magnetic Field
2.5. Cell Viability Analysis
2.6. Cell Death
2.7. Synergism/Additivity Analysis by the Chou–Talalay Method
2.8. Analysis of Caspase-1 and Caspase-3 Activation
2.9. Statistical Analysis
3. Results
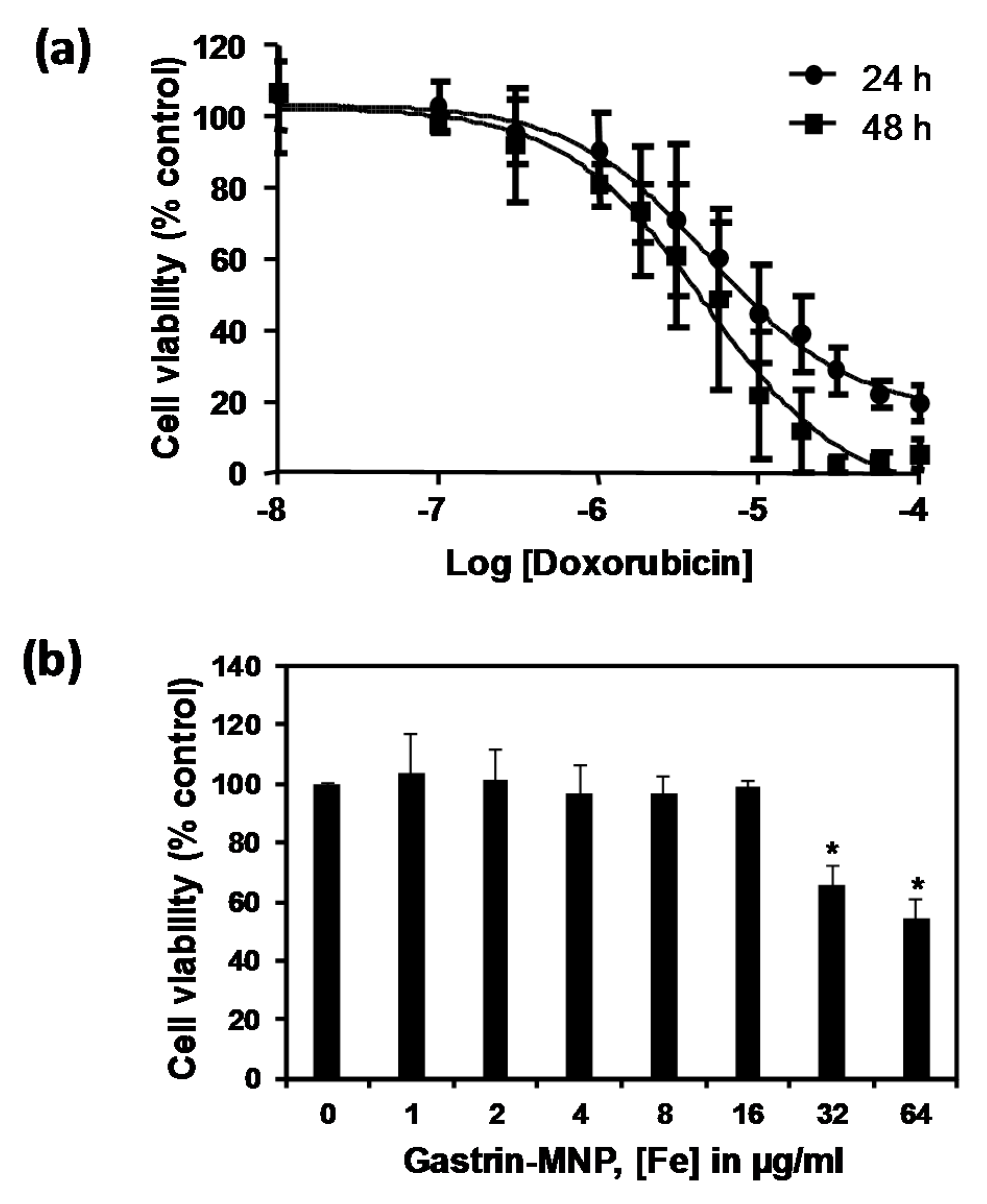
3.1. Doxorubicin and Gastrin–MNP Cytotoxicity
3.2. Effects of the Combination of Magnetic Intra-Lysosomal Hyperthermia and Doxorubicin Treatments
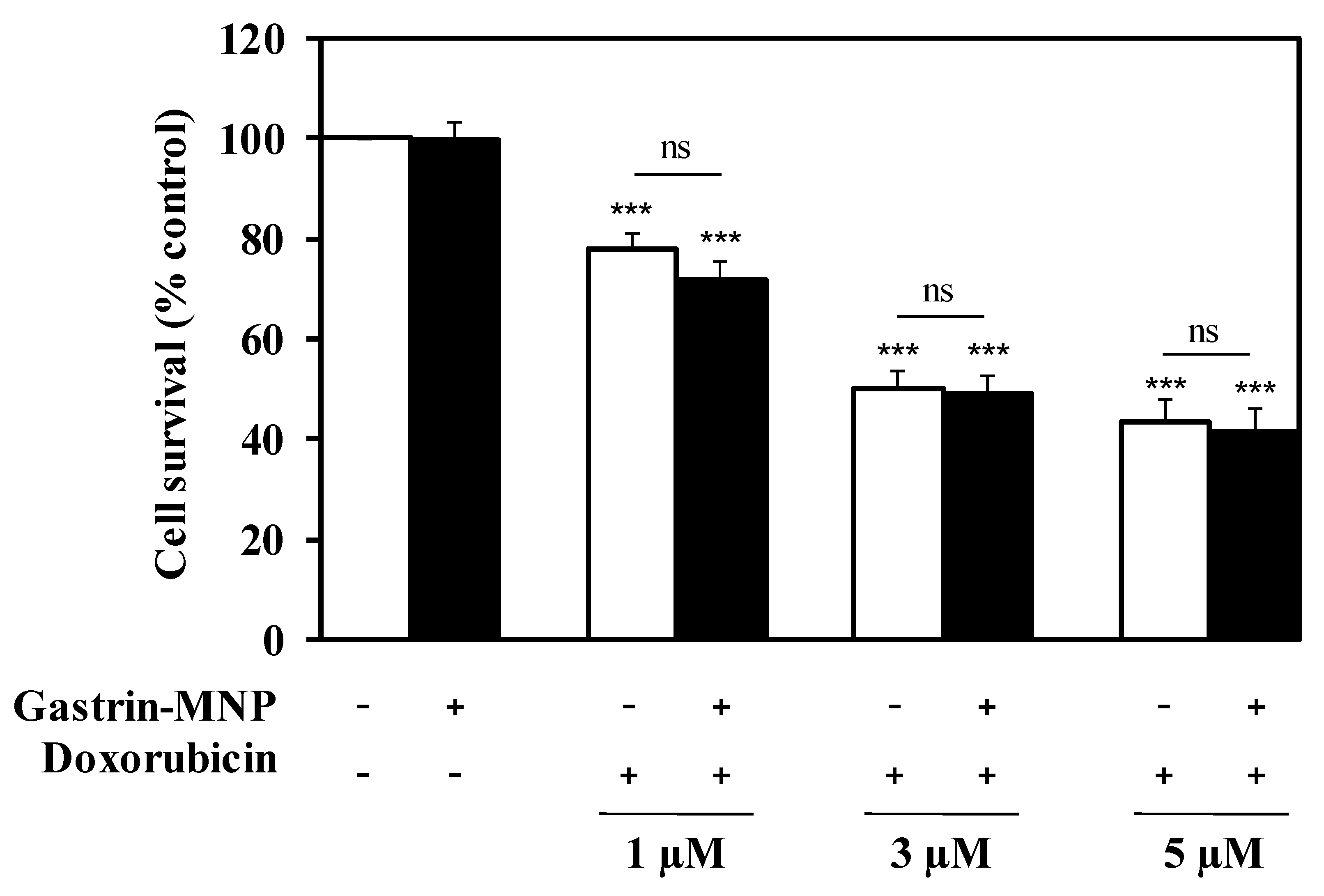
3.2.1. Cell Viability Analysis
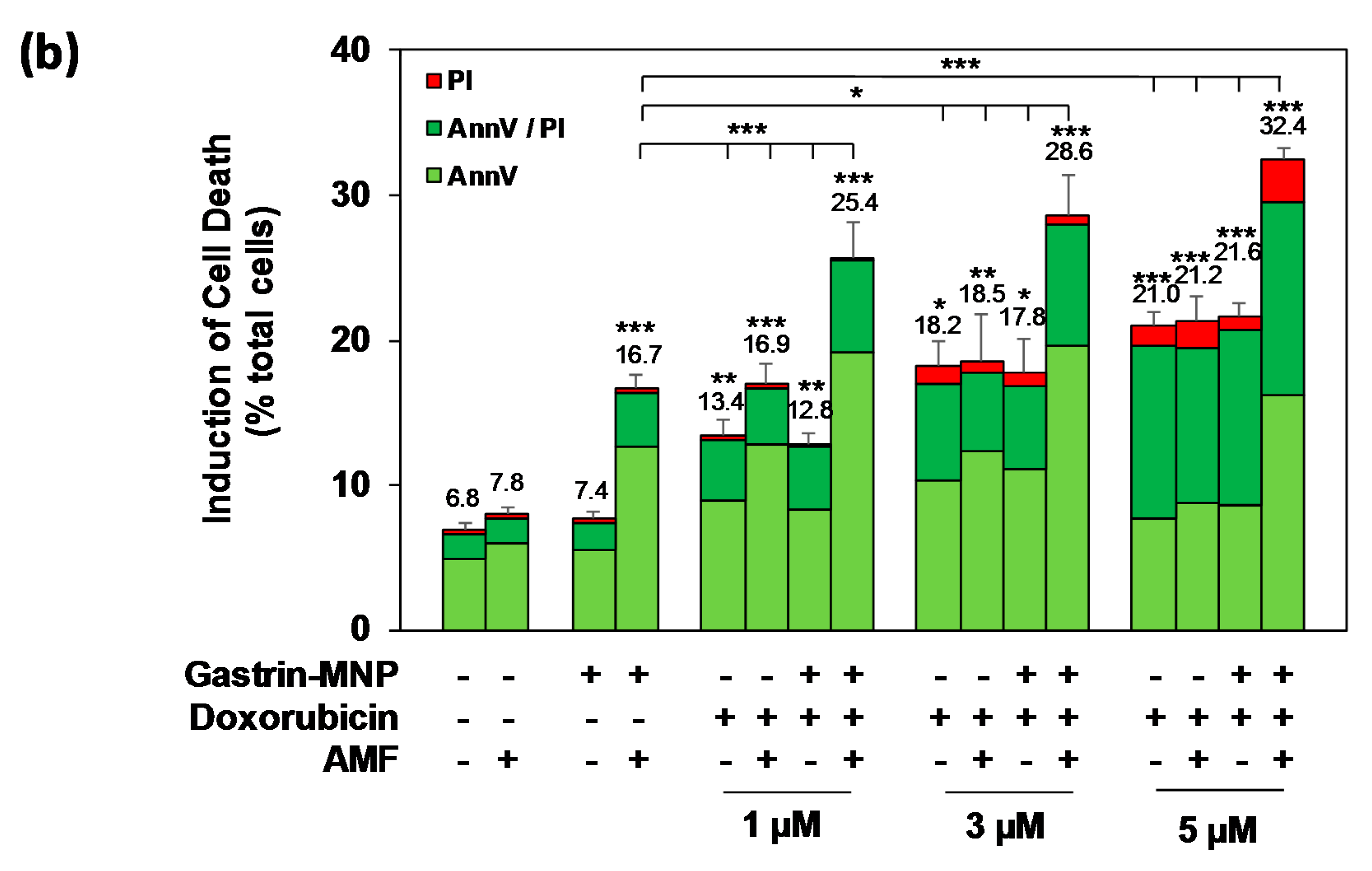
3.2.2. Cell Death Study
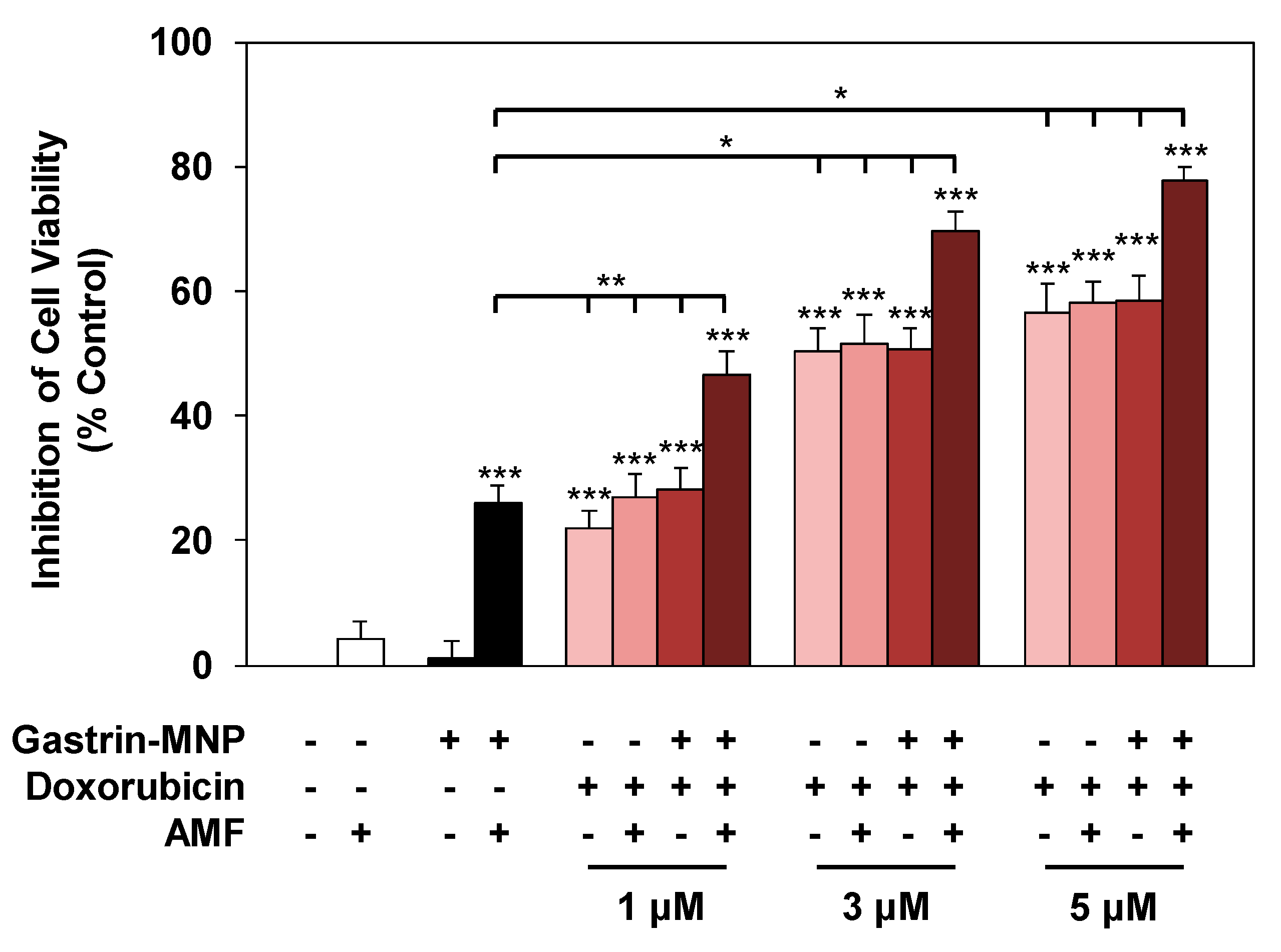
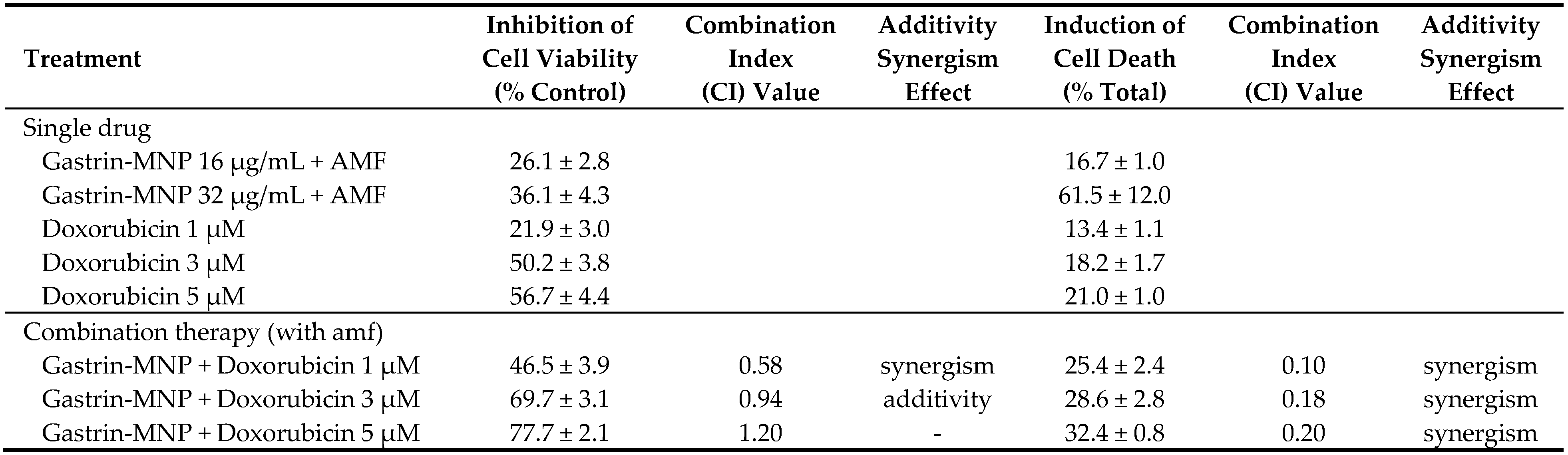
3.2.3. Synergism/Additivity Analysis
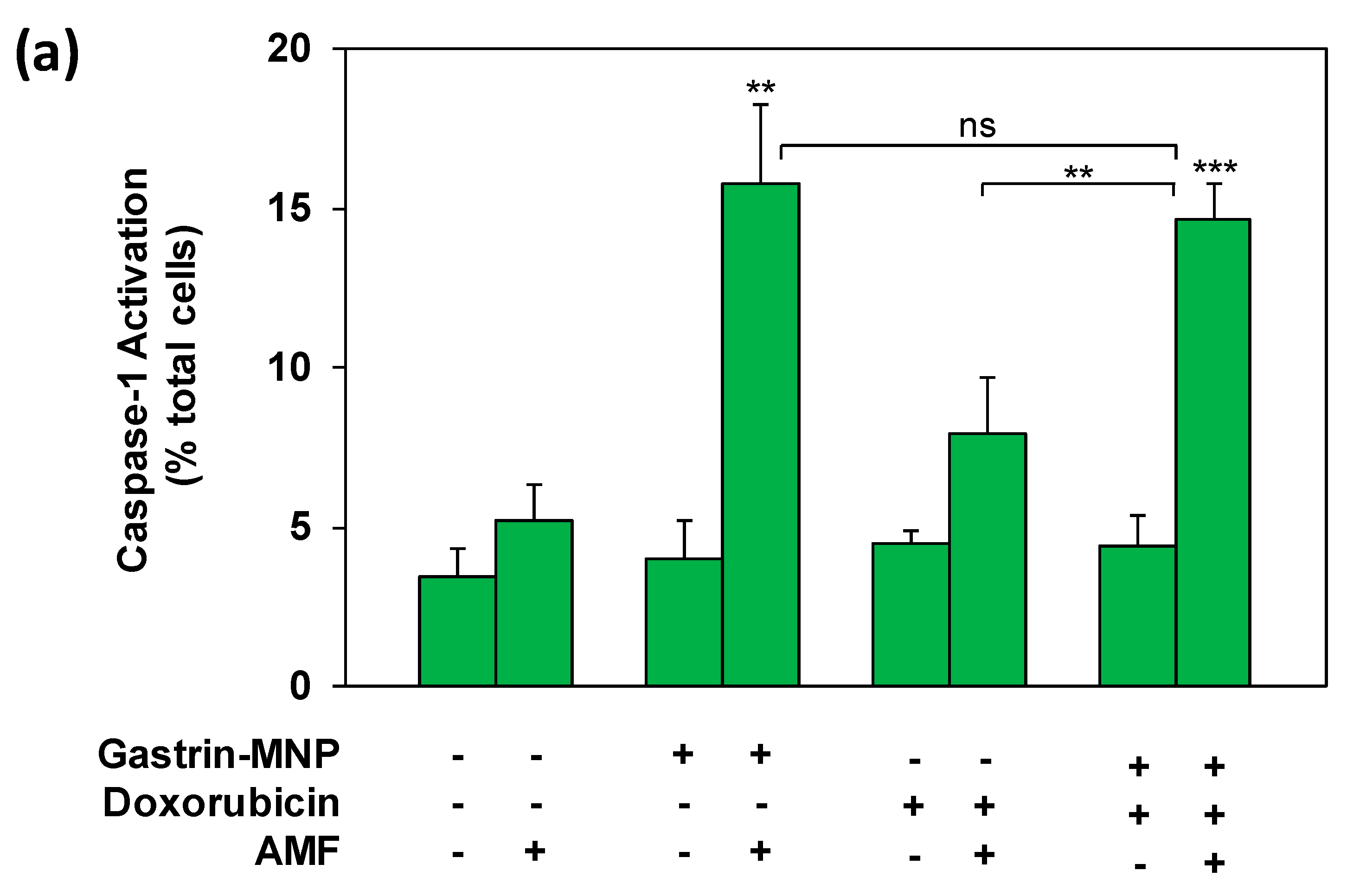
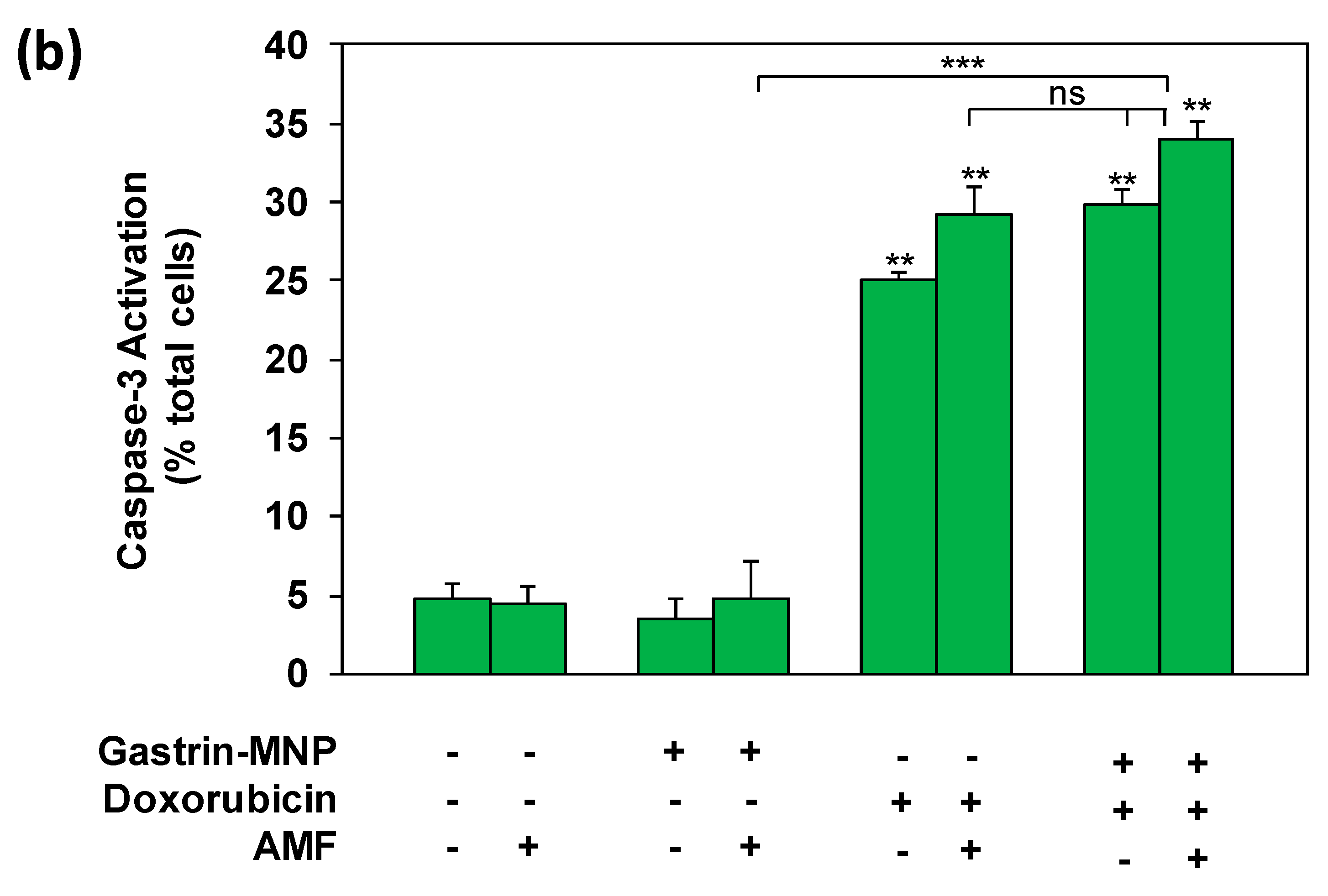
3.3. Mechanism of Cell Death: Analysis of Capsase-1 and Caspase-3 Activation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Scoazec, J.Y. Endocrine tumors: Biology and physiopathology. Ann. Pathol. 2005, 25, 447–461. [Google Scholar] [CrossRef]
- Tacar, O.; Sriamornsak, P.; Dass, C.R. Doxorubicin: An update on anticancer molecular action, toxicity and novel drug delivery systems. J. Pharm. Pharmacol. 2013, 65, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Muller, I.; Niethammer, D.; Bruchelt, G. Anthracycline-derived chemotherapeutics in apoptosis and free radical cytotoxicity (review). Int. J. Mol. Med. 1998, 1, 491–494. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, C.; Santos, R.X.; Cardoso, S.; Correia, S.; Oliveira, P.J.; Santos, M.S.; Moreira, P.I. Doxorubicin: The good, the bad and the ugly effect. Curr. Med. Chem. 2009, 16, 3267–3285. [Google Scholar] [CrossRef] [PubMed]
- Singal, P.K.; Iliskovic, N. Doxorubicin-induced cardiomyopathy. N. Engl. J. Med. 1998, 339, 900–905. [Google Scholar] [CrossRef] [PubMed]
- Vejpongsa, P.; Yeh, E.T. Prevention of anthracycline-induced cardiotoxicity: Challenges and opportunities. J. Am. Coll. Cardiol. 2014, 64, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Roviello, F.; Caruso, S.; Marrelli, D.; Pedrazzani, C.; Neri, A.; De Stefano, A.; Pinto, E. Treatment of peritoneal carcinomatosis with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy: State of the art and future developments. Surg. Oncol. 2011, 20, e38–e54. [Google Scholar] [CrossRef] [PubMed]
- Huilgol, N.G.; Gupta, S.; Sridhar, C.R. Hyperthermia with radiation in the treatment of locally advanced head and neck cancer: A report of randomized trial. J. Cancer Res. Ther. 2010, 6, 492–496. [Google Scholar] [CrossRef] [PubMed]
- Harima, Y.; Nagata, K.; Harima, K.; Ostapenko, V.V.; Tanaka, Y.; Sawada, S. A randomized clinical trial of radiation therapy versus thermoradiotherapy in stage IIIB cervical carcinoma. Int. J. Hyperther. 2001, 17, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Colombo, R.; Da Pozzo, L.F.; Salonia, A.; Rigatti, P.; Leib, Z.; Baniel, J.; Caldarera, E.; Pavone-Macaluso, M. Multicentric study comparing intravesical chemotherapy alone and with local microwave hyperthermia for prophylaxis of recurrence of superficial transitional cell carcinoma. J. Clin. Oncol. 2003, 21, 4270–4276. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, T.; Kokura, S.; Sakamoto, N.; Ando, T.; Imamoto, E.; Hattori, T.; Oyamada, H.; Yoshinami, N.; Sakamoto, M.; Kitagawa, K.; et al. Phase II trial of combined regional hyperthermia and gemcitabine for locally advanced or metastatic pancreatic cancer. Int. J. Hyperther. 2012, 28, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Stauffer, P.R.; Cetas, T.C.; Fletcher, A.M.; DeYoung, D.W.; Dewhirst, M.W.; Oleson, J.R.; Roemer, R.B. Observations on the use of ferromagnetic implants for inducing hyperthermia. IEEE Trans. Bio-Med. Eng. 1984, 31, 76–90. [Google Scholar] [CrossRef] [PubMed]
- Banobre-Lopez, M.; Teijeiro, A.; Rivas, J. Magnetic nanoparticle-based hyperthermia for cancer treatment. Rep. Pract. Oncol. Radiother. 2013, 18, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Ito, A.; Tanaka, K.; Honda, H.; Abe, S.; Yamaguchi, H.; Kobayashi, T. Complete regression of mouse mammary carcinoma with a size greater than 15 mm by frequent repeated hyperthermia using magnetite nanoparticles. J. Biosci. Bioeng. 2003, 96, 364–369. [Google Scholar] [CrossRef]
- Silva, A.C.; Oliveira, T.R.; Mamani, J.B.; Malheiros, S.M.; Malavolta, L.; Pavon, L.F.; Sibov, T.T.; Amaro, E., Jr.; Tannus, A.; Vidoto, E.L.; et al. Application of hyperthermia induced by superparamagnetic iron oxide nanoparticles in glioma treatment. Int. J. Nanomed. 2011, 6, 591–603. [Google Scholar]
- Kossatz, S.; Ludwig, R.; Dahring, H.; Ettelt, V.; Rimkus, G.; Marciello, M.; Salas, G.; Patel, V.; Teran, F.J.; Hilger, I. High therapeutic efficiency of magnetic hyperthermia in xenograft models achieved with moderate temperature dosages in the tumor area. Pharm. Res. 2014, 31, 3274–3288. [Google Scholar] [CrossRef] [PubMed]
- Balivada, S.; Rachakatla, R.S.; Wang, H.; Samarakoon, T.N.; Dani, R.K.; Pyle, M.; Kroh, F.O.; Walker, B.; Leaym, X.; Koper, O.B.; et al. A/c magnetic hyperthermia of melanoma mediated by iron(0)/iron oxide core/shell magnetic nanoparticles: A mouse study. BMC Cancer 2010, 10, 119. [Google Scholar] [CrossRef] [PubMed]
- Johannsen, M.; Thiesen, B.; Jordan, A.; Taymoorian, K.; Gneveckow, U.; Waldofner, N.; Scholz, R.; Koch, M.; Lein, M.; Jung, K.; et al. Magnetic fluid hyperthermia (mfh)reduces prostate cancer growth in the orthotopic dunning r3327 rat model. Prostate 2005, 64, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Fourmy, D.; Carrey, J.; Gigoux, V. Targeted nanoscale magnetic hyperthermia: Challenges and potentials of peptide-based targeting. Nanomedicine (Lond.) 2015, 10, 893–896. [Google Scholar] [CrossRef] [PubMed]
- Creixell, M.; Bohorquez, A.C.; Torres-Lugo, M.; Rinaldi, C. Egfr-targeted magnetic nanoparticle heaters kill cancer cells without a perceptible temperature rise. ACS Nano 2011, 5, 7124–7129. [Google Scholar] [CrossRef] [PubMed]
- Domenech, M.; Marrero-Berrios, I.; Torres-Lugo, M.; Rinaldi, C. Lysosomal membrane permeabilization by targeted magnetic nanoparticles in alternating magnetic fields. ACS Nano 2013, 7, 5091–5101. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, C.; El Hajj Diab, D.; Connord, V.; Clerc, P.; Meunier, E.; Pipy, B.; Payre, B.; Tan, R.P.; Gougeon, M.; Carrey, J.; et al. Targeting a g-protein-coupled receptor overexpressed in endocrine tumors by magnetic nanoparticles to induce cell death. ACS Nano 2014, 8, 1350–1363. [Google Scholar] [CrossRef] [PubMed]
- Clerc, P.; Jeanjean, P.; Hallalli, N.; Gougeon, M.; Pipy, B.; Carrey, J.; Fourmy, D.; Gigoux, V. Targeted magnetic intra-lysosomal hyperthermia produces lysosomal reactive oxygen species and causes caspase-1 dependent cell death. J. Control. Release 2017, 270, 120–134. [Google Scholar] [CrossRef] [PubMed]
- Connord, V.; Clerc, P.; Hallali, N.; El Hajj Diab, D.; Fourmy, D.; Gigoux, V.; Carrey, J. Real-time analysis of magnetic hyperthermia experiments on living cells under a confocal microscope. Small 2015, 11, 2437–2445. [Google Scholar] [CrossRef] [PubMed]
- Leung-Theung-Long, S.; Roulet, E.; Clerc, P.; Escrieut, C.; Marchal-Victorion, S.; Ritz-Laser, B.; Philippe, J.; Pradayrol, L.; Seva, C.; Fourmy, D.; et al. Essential interaction of egr-1 at an islet-specific response element for basal and gastrin-dependent glucagon gene transactivation in pancreatic alpha-cells. J. Biol. Chem. 2005, 280, 7976–7984. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.C.; Talalay, P. Quantitative-analysis of dose-effect relationships—The combined effects of multiple-drugs or enzyme-inhibitors. Adv. Enzym. Regul. 1984, 22, 27–55. [Google Scholar] [CrossRef]
- Chou, T.C. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev. 2006, 58, 621–681. [Google Scholar] [CrossRef] [PubMed]
- Tseng, W.A.; Thein, T.; Kinnunen, K.; Lashkari, K.; Gregory, M.S.; D’Amore, P.A.; Ksander, B.R. Nlrp3 inflammasome activation in retinal pigment epithelial cells by lysosomal destabilization: Implications for age-related macular degeneration. Investig. Ophthalmol. Visual Sci. 2013, 54, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Derangere, V.; Chevriaux, A.; Courtaut, F.; Bruchard, M.; Berger, H.; Chalmin, F.; Causse, S.Z.; Limagne, E.; Vegran, F.; Ladoire, S.; et al. Liver X receptor beta activation induces pyroptosis of human and murine colon cancer cells. Cell Death Differ. 2014, 21, 1914–1924. [Google Scholar] [CrossRef] [PubMed]
- Klostergaard, J.; Leroux, M.E.; Auzenne, E.; Khodadadian, M.; Spohn, W.; Wu, J.Y.; Donato, N.J. Hyperthermia engages the intrinsic apoptotic pathway by enhancing upstream caspase activation to overcome apoptotic resistance in mcf-7 breast adenocarcinoma cells. J. Cell. Biochem. 2006, 98, 356–369. [Google Scholar] [CrossRef] [PubMed]
- Wallner, K.E.; Banda, M.; Li, G.C. Hyperthermic enhancement of cell killing by mitomycin c in mitomycin c-resistant chinese hamster ovary cells. Cancer Res. 1987, 47, 1308–1312. [Google Scholar] [PubMed]
- Wust, P.; Hildebrandt, B.; Sreenivasa, G.; Rau, B.; Gellermann, J.; Riess, H.; Felix, R.; Schlag, P.M. Hyperthermia in combined treatment of cancer. Lancet Oncol. 2002, 3, 487–497. [Google Scholar] [CrossRef]
- Dewey, W.C. Arrhenius relationships from the molecule and cell to the clinic. Int. J. Hyperther. 1994, 10, 457–483. [Google Scholar] [CrossRef]
- Xu, H.; Ju, D.; Jarois, T.; Xie, Y. Diminished feedback regulation of proteasome expression and resistance to proteasome inhibitors in breast cancer cells. Breast Cancer Res. Treat. 2008, 107, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Habash, R.W.; Bansal, R.; Krewski, D.; Alhafid, H.T. Thermal therapy, part 2: Hyperthermia techniques. Crit. Rev. Biomed. Eng. 2006, 34, 491–542. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, R.; Katsenovich, Y.; Lagos, L.; McIintosh, M.; Zhang, X.; Li, C.Z. Nanomedicine: Magnetic nanoparticles and their biomedical applications. Curr. Med. Chem. 2010, 17, 3120–3141. [Google Scholar] [CrossRef] [PubMed]
- Latorre, M.; Rinaldi, C. Applications of magnetic nanoparticles in medicine: Magnetic fluid hyperthermia. Puerto Rico Health Sci. J. 2009, 28, 227–238. [Google Scholar]
- Sato, I.; Umemura, M.; Mitsudo, K.; Kioi, M.; Nakashima, H.; Iwai, T.; Feng, X.; Oda, K.; Miyajima, A.; Makino, A.; et al. Hyperthermia generated with ferucarbotran (resovist(r)) in an alternating magnetic field enhances cisplatin-induced apoptosis of cultured human oral cancer cells. J. Physiol. Sci. JPS 2014, 64, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Berrios, M.P.; Castillo, A.; Rinaldi, C.; Torres-Lugo, M. Magnetic fluid hyperthermia enhances cytotoxicity of bortezomib in sensitive and resistant cancer cell lines. Int. J. Nanomed. 2014, 9, 145–153. [Google Scholar]
- Lee, J.S.; Rodriguez-Luccioni, H.L.; Mendez, J.; Sood, A.K.; Lpez-Berestein, G.; Rinaldi, C.; Torres-Lugo, M. Hyperthermia induced by magnetic nanoparticles improves the effectiveness of the anticancer drug cis-diamminedichloroplatinum. J. Nanosci. Nanotechnol. 2011, 11, 4153–4157. [Google Scholar] [CrossRef] [PubMed]
- Bull, J.M. An update on the anticancer effects of a combination of chemotherapy and hyperthermia. Cancer Res. 1984, 44, 4853s–4856s. [Google Scholar] [PubMed]
- Dahl, O. Interaction of hyperthermia and chemotherapy. In Application of Hyperthermia in the Treatment of Cancer; Springer: Berlin/Heidelberg, Germany, 1988; Volume 107, pp. 157–169. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El Hajj Diab, D.; Clerc, P.; Serhan, N.; Fourmy, D.; Gigoux, V. Combined Treatments of Magnetic Intra-Lysosomal Hyperthermia with Doxorubicin Promotes Synergistic Anti-Tumoral Activity. Nanomaterials 2018, 8, 468. https://doi.org/10.3390/nano8070468
El Hajj Diab D, Clerc P, Serhan N, Fourmy D, Gigoux V. Combined Treatments of Magnetic Intra-Lysosomal Hyperthermia with Doxorubicin Promotes Synergistic Anti-Tumoral Activity. Nanomaterials. 2018; 8(7):468. https://doi.org/10.3390/nano8070468
Chicago/Turabian StyleEl Hajj Diab, Darine, Pascal Clerc, Nizar Serhan, Daniel Fourmy, and Véronique Gigoux. 2018. "Combined Treatments of Magnetic Intra-Lysosomal Hyperthermia with Doxorubicin Promotes Synergistic Anti-Tumoral Activity" Nanomaterials 8, no. 7: 468. https://doi.org/10.3390/nano8070468
APA StyleEl Hajj Diab, D., Clerc, P., Serhan, N., Fourmy, D., & Gigoux, V. (2018). Combined Treatments of Magnetic Intra-Lysosomal Hyperthermia with Doxorubicin Promotes Synergistic Anti-Tumoral Activity. Nanomaterials, 8(7), 468. https://doi.org/10.3390/nano8070468





