Versatile Production of Poly(Epsilon-Caprolactone) Fibers by Electrospinning Using Benign Solvents
Abstract
:1. Introduction
2. Results and Discussion
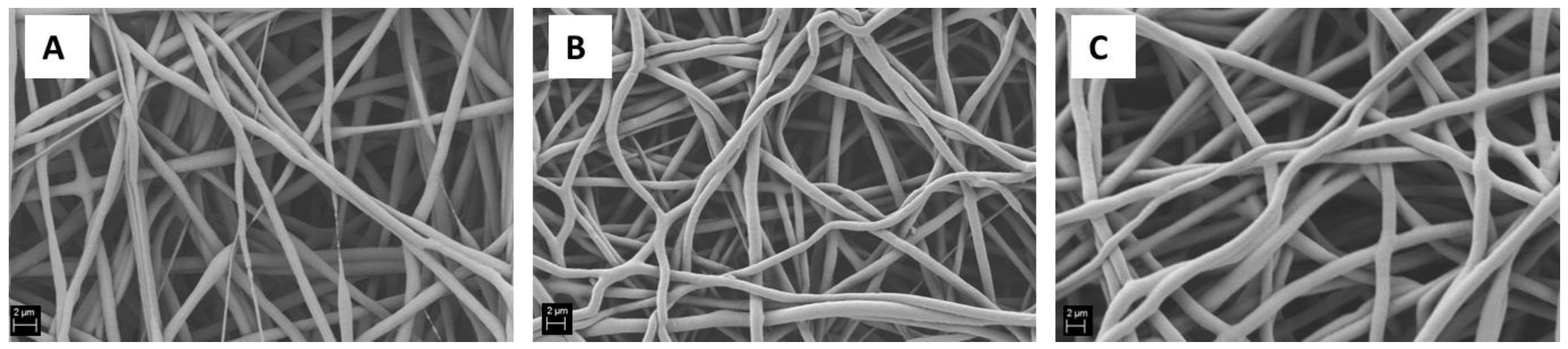
2.1. Influence of Solution Parameters
2.2. Influence of the Applied Voltage
2.3. Optimization of Solvent Mixture for Obtaining Nanofibers
2.4. Macroporosities in Electrospun Fiber Mats
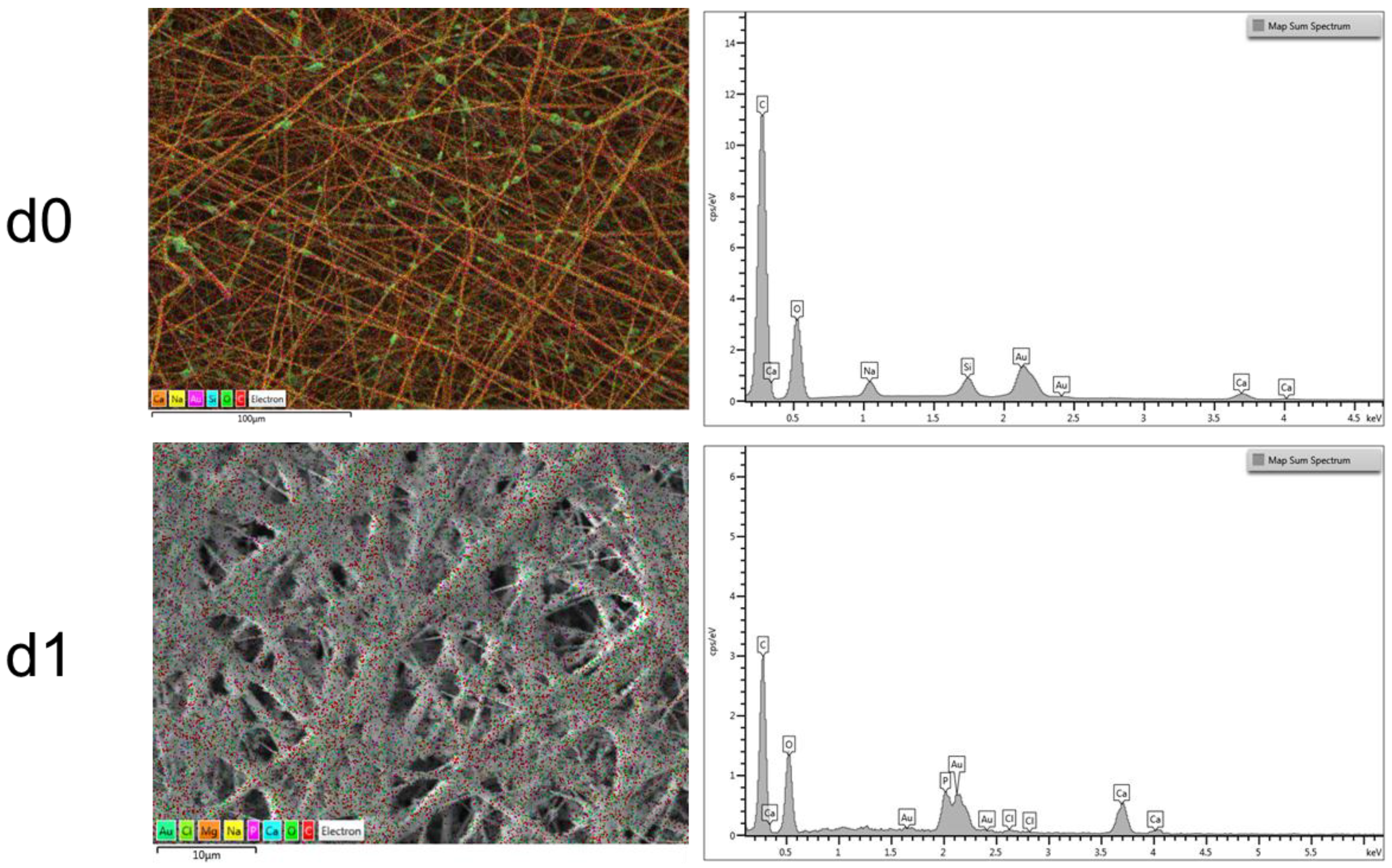
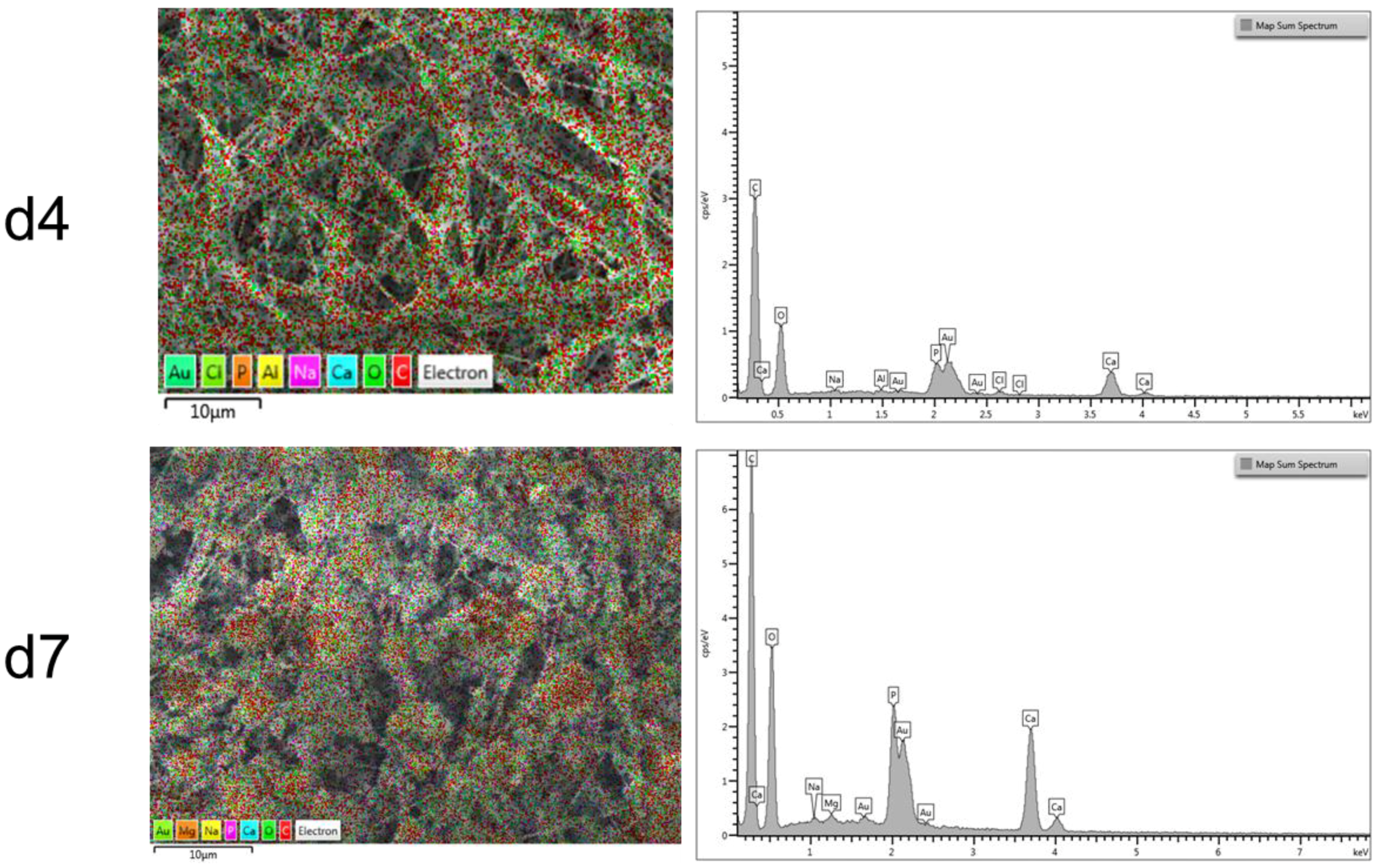
2.5. Composite Electrospun Fibers
2.6. ATR-FTIR Analysis
2.7. Mechanical Properties
3. Experimental Section
3.1. Synthesis
3.2. Electrospinning Process
3.3. Characterization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jiang, T.; Carbone, E.J.; Lo, K.W.-H.; Laurencin, C.T. Electrospinning of polymer nanofibers for tissue regeneration. Prog. Polym. Sci. 2015, 46, 1–24. [Google Scholar] [CrossRef]
- Sun, B.; Long, Y.Z.; Zhang, H.D.; Li, M.M.; Duvail, J.L.; Jiang, X.Y.; Yin, H.L. Advances in three-dimensional nanofibrous macrostructures via electrospinning. Prog. Polym. Sci. 2014, 39, 862–890. [Google Scholar] [CrossRef]
- Putti, M.; Simonet, M.; Solberg, R.; Peters, G.W.M. Electrospinning poly(ε-caprolactone) under controlled environmental conditions: Influence on fiber morphology and orientation. Polymer (Guildf) 2015, 63, 189–195. [Google Scholar] [CrossRef]
- Agarwal, S.; Greiner, A. On the way to clean and safe electrospinning-green electrospinning: Emulsion and suspension electrospinning. Polym. Adv. Technol. 2011, 22, 372–378. [Google Scholar] [CrossRef]
- Ferreira, J.L.; Gomes, S.; Henriques, C.; Borges, J.P.; Silva, J.C. Electrospinning polycaprolactone dissolved in glacial acetic acid: Fiber production, nonwoven characterization, and in vitro evaluation. J. Appl. Polym. Sci. 2014, 131. [Google Scholar] [CrossRef]
- Ghosal, K.; Thomas, S.; Kalarikkal, N.; Gnanamanis, A. Collagen coated electrospun polycaprolactone (PCL) with titanium dioxide (TiO2) from an environmentally benign solvent: Preliminary physico-chemical studies for skin substitute. J. Polym. Res. 2014, 21, 2–6. [Google Scholar] [CrossRef]
- Dash, T.K.; Konkimalla, V.B. Poly-ε-caprolactone based formulations for drug delivery and tissue engineering: A review. J. Control. Release 2012, 158, 15–33. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xiong, C.; Deng, X. Biodegradable polyester blends for biomedical application. J. Appl. Polym. Sci. 1995, 56, 103–112. [Google Scholar] [CrossRef]
- Duncan, T.V. Applications of nanotechnology in food packaging and food safety: Barrier materials, antimicrobials and sensors. J. Colloid Interface Sci. 2011, 363, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Castro, N.J.; Cheng, X.; Keidar, M.; Zhang, L.G. Cold Atmospheric Plasma Modified Electrospun Scaffolds with Embedded Microspheres for Improved Cartilage Regeneration. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Tiaw, K.S.; Goh, S.W.; Hong, M.; Wang, Z.; Lan, B.; Teoh, S.H. Laser surface modification of poly(ε-caprolactone) (PCL) membrane for tissue engineering applications. Biomaterials 2005, 26, 763–769. [Google Scholar] [CrossRef] [PubMed]
- Marletta, G.; Ciapetti, G.; Satriano, C.; Perut, F.; Salerno, M.; Baldini, N. Improved osteogenic differentiation of human marrow stromal cells cultured on ion-induced chemically structured poly-ε-caprolactone. Biomaterials 2007, 28, 1132–1140. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Teoh, S.H. Surface modification of ultra thin poly(epsilon-caprolactone) films using acrylic acid and collagen. Biomaterials 2004, 25, 1991–2001. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, G.R.; Lima, T.H.; Oréfice, R.L.; Fernandes-Cunha, G.M.; Silva-Cunha, A.; Zhao, M.; Behar-Cohen, F. In vitro and in vivo ocular biocompatibility of electrospun poly(ɛ-caprolactone) nanofibers. Eur. J. Pharm. Sci. 2015, 73, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xie, J.; Yuan, X.; Xia, Y. Coating electrospun poly(ε-caprolactone) fibers with gelatin and calcium phosphate and their use as biomimetic scaffolds for bone tissue engineering. Langmuir 2008, 24, 14145–14150. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.M.; Zhang, Y.Z.; Kotaki, M.; Ramakrishna, S. A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Compos. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Yoshimoto, H.; Shin, Y.M.; Terai, H.; Vacanti, J.P. A biodegradable nanofiber scaffold by electrospinning and its potential for bone tissue engineering. Biomaterials 2003, 24, 2077–2082. [Google Scholar] [CrossRef]
- Suggs, L.J.; Moore, S.A.; Mikos, A.G. Physical Properties of Polymers Handbook. In Physical Properties of Polymers Handbook; Mark, J.E., Ed.; Springer: Berlin, Germany, 2007; pp. 942–943. [Google Scholar]
- Pitt, C.G. Poly-ε-Caprolactone and Its Copolymers. In Biodegradable Polymers As Drug Delivery Systems; Chasin, M., Langer, R., Eds.; Marcel Dekker Inc.: New York, NY, USA, 1990; pp. 71–120. [Google Scholar]
- Soliman, S.; Sant, S.; Nichol, J.W.; Khabiry, M.; Traversa, E.; Khademhosseini, A. Controlling the porosity of fibrous scaffolds by modulating the fiber diameter and packing density. J. Biomed. Mater. Res. Part A 2011, 96A, 566–574. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Kim, H.Y.; Khil, M.S.; Ra, Y.M.; Lee, D.R. Characterization of nano-structured poly(ε-caprolactone) nonwoven mats via electrospinning. Polymer (Guildf) 2003, 44, 1287–1294. [Google Scholar] [CrossRef]
- Li, W.-J.; Danielson, K.G.; Alexander, P.G.; Tuan, R.S. Biological response of chondrocytes cultured in three-dimensional nanofibrous poly(epsilon-caprolactone) scaffolds. J. Biomed. Mater. Res. A 2003, 67, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Prabhakaran, M.P.; Venugopal, J.R.; Chyan, T.T.; Hai, L.B.; Chan, C.K.; Lim, A.Y.; Ramakrishna, S. Electrospun biocomposite nanofibrous scaffolds for neural tissue engineering. Tissue Eng. Part A 2008, 14, 1787–1797. [Google Scholar] [CrossRef] [PubMed]
- Van Der Schueren, L.; De Schoenmaker, B.; Kalaoglu, Ö.I.; De Clerck, K. An alternative solvent system for the steady state electrospinning of polycaprolactone. Eur. Polym. J. 2011, 47, 1256–1263. [Google Scholar] [CrossRef]
- Kanani, A.G.; Bahrami, S.H. Effect of Changing Solvents on Poly(ε-Caprolactone) Nanofibrous Webs Morphology. J. Nanomater. 2011, 2011, 1–10. [Google Scholar] [CrossRef]
- Katsogiannis, K.A.G.; Vladisavljević, G.T.; Georgiadou, S. Porous electrospun polycaprolactone (PCL) fibres by phase separation. Eur. Polym. J. 2015, 69, 284–295. [Google Scholar] [CrossRef]
- Oh, S.H.; Park, I.K.; Kim, J.M.; Lee, J.H. In vitro and in vivo characteristics of PCL scaffolds with pore size gradient fabricated by a centrifugation method. Biomaterials 2007, 28, 1664–1671. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Wang, C.; Wei, Y. One-dimensional composite nanomaterials: Synthesis by electrospinning and their applications. Small 2009, 5, 2349–2370. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, J.R.; Low, S.; Choon, A.T.; Kumar, A.B.; Ramakrishna, S. Nanobioengineered electrospun composite nanofibers and osteoblasts for bone regeneration. Artif. Organs 2008, 32, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.M.; Moon, J.H.; Yi, G.R.; Heo, C.J.; Yang, S.M. Fabrication of one-dimensional colloidal assemblies from electrospun nanofibers. Langmuir 2006, 22, 3445–3449. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, A.; Güldal, N.S.; Boccaccini, A.R. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 2011, 32, 2757–2774. [Google Scholar] [CrossRef] [PubMed]
- Pham, Q.P.; Sharma, U.; Mikos, A.G. Electrospun Poly(ε-caprolactone) Microfiber and Multilayer Nanofiber/Microfiber Scaffolds: Characterization of Scaffolds and Measurement of Cellular Infiltration. Biomacromolecules 2006, 2796–2805. [Google Scholar] [CrossRef] [PubMed]
- Roosa, S.M.M.; Kemppainen, J.M.; Moffitt, E.N.; Krebsbach, P.H.; Hollister, S.J. The pore size of polycaprolactone scaffolds has limited influence on bone regeneration in an in vivo model. J. Biomed. Mater. Res. Part A 2010, 92, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Liverani, L.; Boccaccini, A.R. Electrospinning with benign solvents: Feasibility study and versatile use of poly(epsilon-caprolactone) fibers. In Proceedings of 10th World Biomaterials Congress, Montreal, QC, Canada, 17–22 May 2016. Front. Bioeng. Biotechnol.; Conference Abstract. [CrossRef]
- Wutticharoenmongkol, P.; Sanchavanakit, N.; Pavasant, P.; Supaphol, P. Preparation and characterization of novel bone scaffolds based on electrospun polycaprolactone fibers filled with nanoparticles. Macromol. Biosci. 2006, 6, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Gönen, S.Ö.; Taygun, M.E.; Küçükbayrak, S. Fabrication of Bioactive Glass Containing Nanocomposite Fiber Mats For Bone Tissue Engineering Applications. Compos. Struct. 2016, 138, 96–106. [Google Scholar] [CrossRef]
- Kouhi, M.; Morshed, M.; Varshosaz, J.; Fathi, M.H. Poly(epsilon-caprolactone) incorporated bioactive glass nanoparticles and simvastatin nanocomposite nanofibers: Preparation, characterization and in vitro drug release for bone regeneration applications. Chem. Eng. J. 2013, 228, 1057–1065. [Google Scholar] [CrossRef]
- Catledge, S.A.; Clem, W.C.; Shrikishen, N.; Chowdhury, S.; Stanishevsky, A.V.; Koopman, M.; Vohra, Y.K. An electrospun triphasic nanofibrous scaffold for bone tissue engineering. Biomed. Mater. 2007, 2, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Morshed, M.; Nasr-Esfahani, M.-H.; Ramakrishna, S. Electrospun poly(ε-caprolactone)/gelatin nanofibrous scaffolds for nerve tissue engineering. Biomaterials 2008, 29, 4532–4539. [Google Scholar] [CrossRef] [PubMed]
- Gaharwar, A.K.; Nikkhah, M.; Sant, S.; Khademhosseini, A. Anisotropic poly(glycerol sebacate)-poly(ε-caprolactone) electrospun fibers promote endothelial cell guidance. Biofabrication 2014, 7. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, H.; Serra, J.; González, P.; León, B. Structural study of sol–gel silicate glasses by IR and Raman spectroscopies. J. Non. Cryst. Solids 2009, 355, 475–480. [Google Scholar] [CrossRef]
- Zheng, K.; Solodovnyk, A.; Li, W.; Goudouri, O.-M.; Stähli, C.; Nazhat, S.N.; Boccaccini, A.R. Aging Time and Temperature Effects on the Structure and Bioactivity of Gel-Derived 45S5 Glass-Ceramics. J. Am. Ceram. Soc. 2015, 98, 30–38. [Google Scholar] [CrossRef]
- Lin, H.M.; Lin, Y.H.; Hsu, F.Y. Preparation and characterization of mesoporous bioactive glass/polycaprolactone nanofibrous matrix for bone tissues engineering. J. Mater. Sci. Mater. Med. 2012, 23, 2619–2630. [Google Scholar] [CrossRef] [PubMed]
- Jo, J.H.; Lee, E.J.; Shin, D.S.; Kim, H.E.; Kim, H.W.; Koh, Y.H.; Jang, J.H. In vitro/in vivo biocompatibility and mechanical properties of bioactive glass nanofiber and poly(ε-caprolactone) composite materials. J. Biomed. Mater. Res. Part B 2009, 91, 213–220. [Google Scholar] [CrossRef] [PubMed]

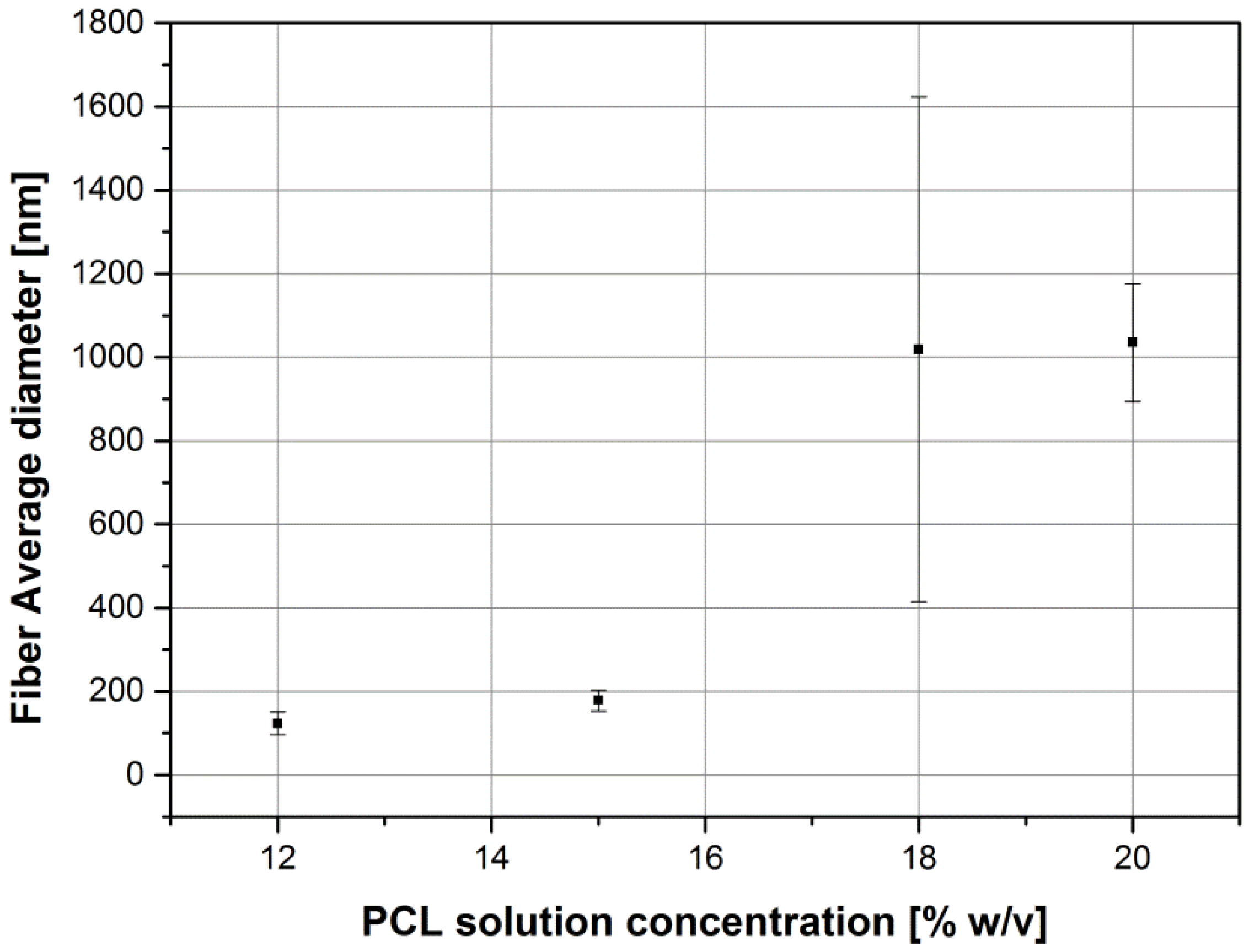




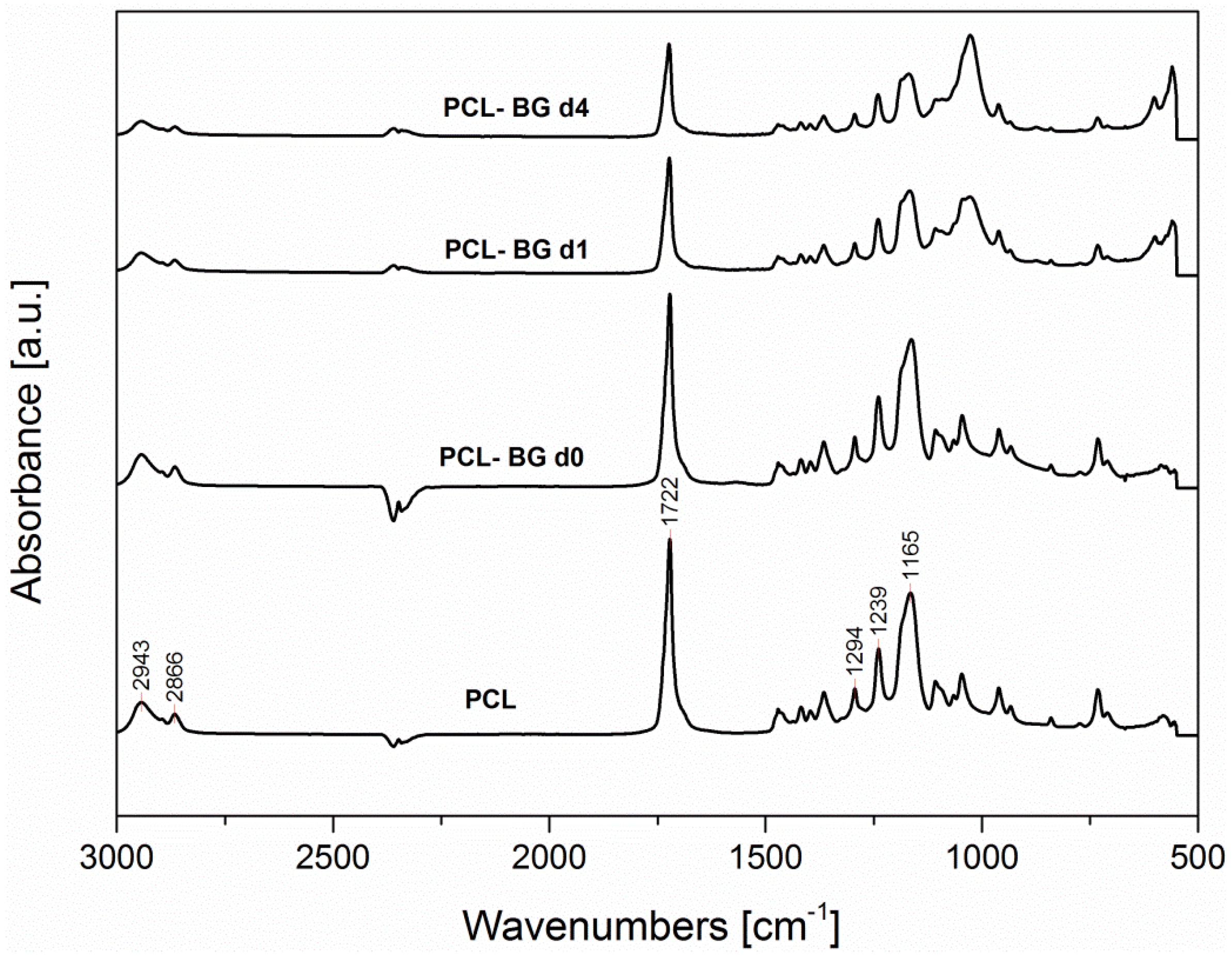
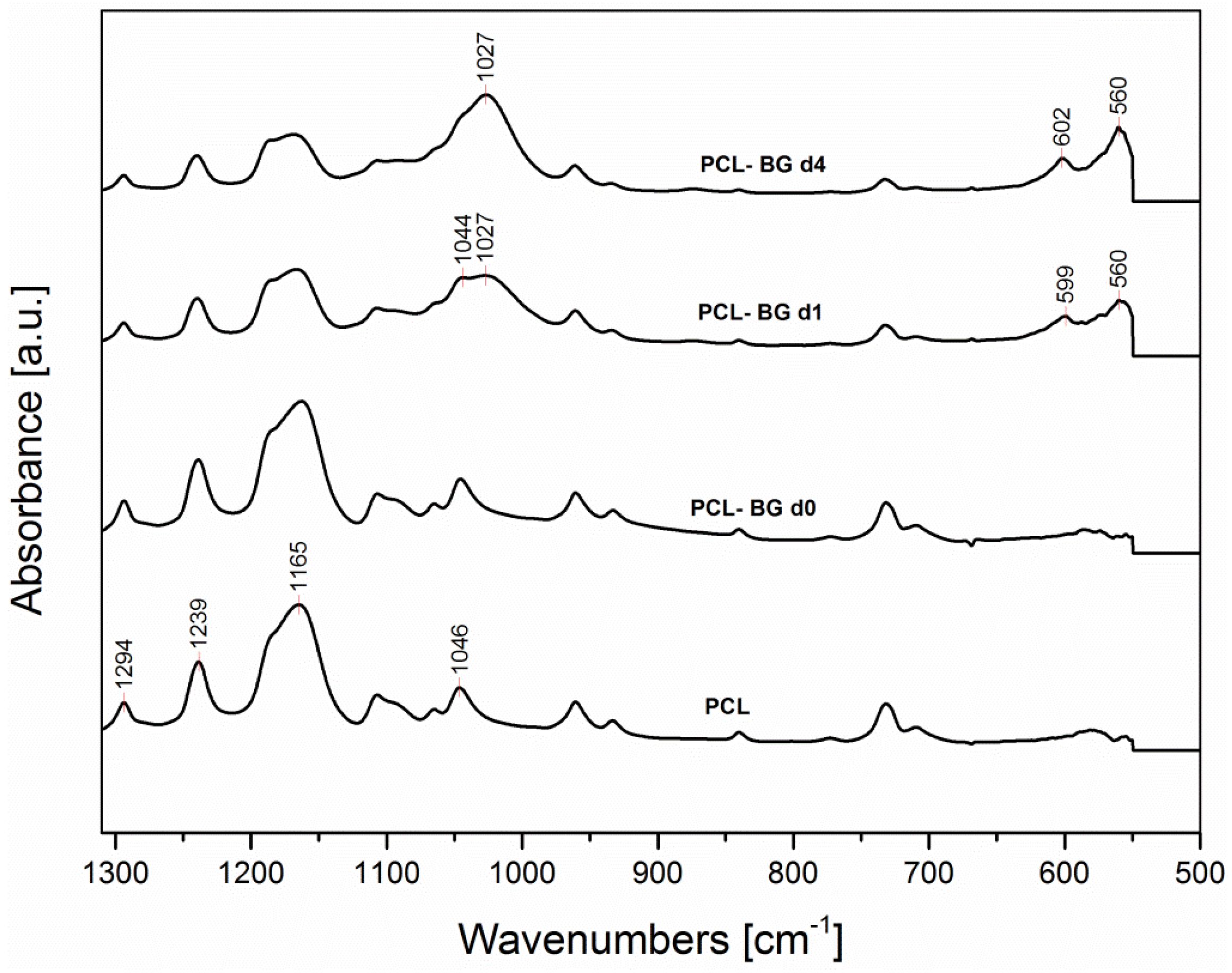

| Sample | Fiber Average Diameter | Minimum Fiber Diameter | Maximum Fiber Diameter |
|---|---|---|---|
| PCL12_AA_15 * | 0.12 ± 0.03 μm | 0.09 μm | 0.2 μm |
| PCL15_AA_15 * | 0.18 ± 0.02 μm | 0.1 μm | 0.2 μm |
| PCL18_AA_15 | 1.0 ± 0.6 μm | 0.3 μm | 3.6 μm |
| PCL20_AA_15 | 1.0 ± 0.1 μm | 0.7 μm | 1.2 μm |
| Sample Name | Average Fiber Diameter (μm) | Young’s Modulus (Mpa) | UTS (Mpa) | Tensile Strain (%) |
|---|---|---|---|---|
| PCL20_AA_15 | 1.0 ± 0.1 | 12 ± 5 | 1.2 ± 0.3 | 83 ± 10 |
| PCL15_AAFA | 0.20 ± 0.04 | 11.0 ± 0.8 | 6.2 ± 0.9 | 115 ± 2 |
| PCL-BG | 0.5 ± 0.2 | 4.2 ± 0.9 | 1.2 ± 0.3 | 90 ± 18 |
| Sample Name | Solution Concentration (% w/v) | Solvent(s) | kV | Distance Tip-Target (cm) | Needle Diameter (G) | Flow Rate (mL/h) | T (°C) | Relative Humidity (RH) (%) | SEM Micrograph |
|---|---|---|---|---|---|---|---|---|---|
| PCL12 | 12 | AA | 15 | 11 | 23 | 0.4 | 23.6 | 42 | Figure 1A |
| PCL15_AA | 15 | AA | 15 | 11 | 23 | 0.4 | 23.5 | 43 | Figure 1B and Figure 4A |
| PCL15_AAFA | 15 | AA/FA | 20 | 11 | 23 | 1.3 | 23.6 | 43 | Figure 4B |
| PCL18 | 18 | AA | 15 | 15 | 23 | 0.4 | 23.5 | 49 | Figure 1C |
| PCL20_10 | 20 | AA | 10 | 11 | 23 | 0.4 | 29.0 | 45 | Figure 3A |
| PCL20_15 | 20 | AA | 15 | 11 | 23 | 0.4 | 28.0 | 48 | Figure 1D and Figure 3B |
| PCL20_20 | 20 | AA | 20 | 11 | 23 | 0.4 | 23.5 | 28 | Figure 3C |
| PCL-BG | 20 | AA | 15 | 11 | 21 | 0.8 | 23.6 | 49 | Figure 7A,B,C |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liverani, L.; Boccaccini, A.R. Versatile Production of Poly(Epsilon-Caprolactone) Fibers by Electrospinning Using Benign Solvents. Nanomaterials 2016, 6, 75. https://doi.org/10.3390/nano6040075
Liverani L, Boccaccini AR. Versatile Production of Poly(Epsilon-Caprolactone) Fibers by Electrospinning Using Benign Solvents. Nanomaterials. 2016; 6(4):75. https://doi.org/10.3390/nano6040075
Chicago/Turabian StyleLiverani, Liliana, and Aldo R. Boccaccini. 2016. "Versatile Production of Poly(Epsilon-Caprolactone) Fibers by Electrospinning Using Benign Solvents" Nanomaterials 6, no. 4: 75. https://doi.org/10.3390/nano6040075
APA StyleLiverani, L., & Boccaccini, A. R. (2016). Versatile Production of Poly(Epsilon-Caprolactone) Fibers by Electrospinning Using Benign Solvents. Nanomaterials, 6(4), 75. https://doi.org/10.3390/nano6040075







