Abstract
Benzo(a)pyrene (BaP) is a recognized reprotoxic compound and the most widely investigated polycyclic aromatic hydrocarbon in ambient air; it is widespread by the incomplete combustion of fossil fuels along with cerium dioxide nanomaterials (CeO2 NMs), which are used in nano-based diesel additives to decrease the emission of toxic compounds and to increase fuel economy. The toxicity of CeO2 NMs on reproductive organs and cells has also been shown. However, the effect of the combined interactions of BaP and CeO2 NMs on reproduction has not been investigated. Herein, human and rat gametes were exposed in vitro to combusted CeO2 NMs or BaP or CeO2 NMs and BaP in combination. CeO2 NMs were burned at 850 °C prior to mimicking their release after combustion in a diesel engine. We demonstrated significantly higher amounts of DNA damage after exposure to combusted CeO2 NMs (1 µg·L−1) or BaP (1.13 µmol·L−1) in all cell types considered compared to unexposed cells. Co-exposure to the CeO2 NMs-BaP mixture induced additive DNA damage in sperm and cumulus cells, whereas no additive effect was observed in rat oocytes. This result could be related to the structural protection of the oocyte by cumulus cells and to the oocyte’s efficient system to repair DNA damage compared to that of cumulus and sperm cells.
1. Introduction
Diesel engines are one of many sources of ambient particulate matter and gaseous air pollutants [1]. Diesel exhaust is a complex mixture of particles, commonly known as soot and gases and contains more than one hundred different organic and inorganic compounds, including many chemicals that have been designated as air pollutants [2]. In 2012, the International Agency for Research on Cancer (IARC), part of the World Health Organization (WHO), upgraded the carcinogenicity of diesel emissions from Group 2 A (probably carcinogenic) to Group 1 (carcinogenic with sufficient evidence) [3]. For instance, diesel engines are significant sources of polycyclic aromatic hydrocarbons (PAHs) in urban air [4]. Despite the hazards induced by PAHs to humans, there are no motor vehicle emission limits for these compounds in most countries. Sixteen PAHs compounds have been classified by the U.S. EPA as a priority pollutant because of various toxicological concerns [5] and significant health impacts [6]. Among them, benzoapyrene (BaP) is recognized as a powerful carcinogen, mutagen, and reprotoxic compound [7,8]. The exposure to PAHs is mostly through ingestion and air inhalation, the BaP “virtually safe dose” is depending on countries legislation and is between 0.7–1 ng/m3 [9]. BaP is associated with increased genotoxicity [10,11,12] and DNA fragmentation [13] towards sperm cells and oocytes. BaP exposure decreases sperm motility and morphology and increases DNA damage [14,15,16]. In vivo experimental studies have also shown that postnatal exposure to BaP destroys ovarian follicles due to the inhibition of follicle growth and then causes premature ovarian failure [17,18,19,20]. More recently, nanomaterials (NMs) have been increasingly used in Europe and elsewhere as fuel-borne catalysts in diesel engines [21,22,23] as CeO2 NMs [24,25]. These CeO2 NMs are used to decrease the emission of toxic compounds in exhaust [26], but they have also been shown to increase the emission of ultrafine particles and the amount of Ce released [26]. Compared to that of BaP, the potential effect of the released CeO2 NMs on health is still not fully understood [27,28], and up to now, there are still few studies regarding the exposure to CeO2 NMs, and no secure data are reported concerning the humans exposure limits. However, few in vivo and in vitro studies have demonstrated the potential toxicity of CeO2 NMs on reproductive cells [29,30,31,32,33,34], which likely occurs via the generation of reactive oxygen species (ROS), leading to oxidative stress and DNA damage [26,32,33]. Interestingly, the biological effects of NMs depend not only on their own structure and chemistry but also on their interactions (e.g., adsorption, complexation) with other pollutants, such as PAHs, metals, metalloids, etc. [35,36]. To date, most research on the effects of chemicals on biological systems is conducted on one chemical at a time, while in the real world (as with diesel exhaust), people are exposed to chemical mixtures whose effects are extremely complex and need further investigation [37]. Within mixtures, chemicals (organic, inorganic, dissolved, and nanoparticulate) could interact additively (which results in the sum of toxicity of each agent), synergistically (inducing toxic effects greater than the sum of the effects of the individual chemicals) or antagonistically (where the combined effect of two or more compounds is less toxic than the individual effects) [38]. This study aimed to investigate the combined biological effects of one commercialized CeO2 NM-based diesel additive (EnviroxTM from Energenics Europe Ltd., Begbroke, UK) and one PAH (BaP), both of which are likely released in the atmosphere after combustion in a diesel engine [4,25,39]. Prior to the in vitro exposure of germ cells, EnviroxTM was combusted at 850 °C to mimic its physico-chemical transformations in a diesel engine [40]. Then, the potential genotoxicity induced by the in vitro co-exposure of human and rat gametes to combusted CeO2 NMs along with BaP was investigated using the comet assay. Herein, we will study how the interactions between combusted CeO2 NMs and BaP molecules in diesel exhaust may additively, synergistically, or antagonistically impact the previously observed genotoxicities of the individual compounds on human and rat germ cells (sperm, follicular cells, and oocytes).
2. Materials and Methods
2.1. Solution and Suspension Preparation Prior to Exposure
Metabolic activation of benzo(a)pyrene (BaP). BaP was purchased from Sigma Aldrich (Saint-Quentin Fallavier, France). A BaP stock suspension was prepared in dimethyl sulfoxide (DMSO) (Sigma Aldrich) at 10 mM to obtain complete dissolution [41]. To activate BaP metabolism, we used an S9 mix [39,42,43] that consisted of the following cofactors: pooled S9 rat liver (Sigma Aldrich), 1 M KCl, 0.25 M MgCl2*6H2O, 0.2 M glucose-6-phosphate, and 0.04 M NADP [44]. The final concentration of BaP at 1.13 µmol·L−1 was then prepared in Ferticult® medium (JCD Laboratories, Lyon, France), with 1% S9 mix and 1% DMSO as previously described by Baumgartner et al. (2012) [45]. The working concentration was mainly chosen because of previously published toxicological data, but also due to the solubility limits in biological media [45].
Aging of the diesel fuel additive. CeO2 NMs were extracted from EnviroxTM, a fuel-borne catalyst scientifically and commercially proven CeO2 NM-based diesel additive supplied by Energenics Europe Ltd. The EnviroxTM was combusted and characterized following the protocol already published in ref [40,46]. Briefly, the EnviroxTM was by ultracentrifugated at 396,750x g and 20 °C for 1 h. The pellets containing CeO2 NMs were freeze-dried (Heto PowerDry LL3000, Thermo Fisher Scientific, Strasbourg, France) for 5 days and combusted at 850 °C [30,40]. A stock suspension of the combusted EnviroxTM (called aged CeO2 NMs) was prepared in Milli-Q water at 10.15 g·L−1 CeO2 and put under magnetic stirring to avoid the formation of large aggregates. The final concentration (1 µg·L−1) was prepared in Ferticult® medium. This concentration of CeO2 NMs was chosen because it was the lowest studied concentration responsible for significant DNA damage in human and rat sperm cells [30].
Mixture of aged CeO2 NMs and BaP. One µg·L−1 of aged CeO2 NMs was incubated with 1.13 µmol·L−1 BaP in abiotic Ferticult® supplemented with 1% S9 mix and 1% DMSO for 1 h at room temperature (RT) prior to exposure to the cells. To estimate the stability of BaP in supplemented Ferticult®, pure suspensions of BaP at 50 µmol·L−1 were also incubated without NMs in supplemented Ferticult®, centrifuged (1 h at 4000× g), or settled (1 h), and their supernatant was measured by UV-vis spectrometry (mySPEC Twin UV-vis spectrometer, VWR, Val-de-Marne, France). Standard curves obtained at two wavelengths corresponding to the BaP signal (300 and 384 nm) are provided in Supporting Information. We estimated that 30 ± 6% of the BaP was removed from the solution just by 1h settling and 57 ± 11% by 1h centrifugation. This could highlight the incomplete dissolution but also to the chemical instability of BaP in these abiotic conditions related to its high affinity for serum components (i.e., as albumin in Ferticult®) [47,48,49,50]. UV-vis spectrometry was used to estimate the affinity of BaP for the surface of the aged CeO2 NMs in abiotic conditions. To be in the detection range of the apparatus (see standard curves in Supplementary Materials, Figure S1), 10 µg·L−1 aged CeO2 NMs were mixed with 11.3 µmol·L−1 BaP (similar [CeO2]/[BaP] ratio of concentration to those used with the cells) in Ferticult® medium supplemented with 1% S9 mix and 1% DMSO for 1 h under mechanical stirring at RT. After 1 h, the samples were centrifuged (1 h at 4000× g), and the supernatant was recovered. No washing step was performed in order to access both the weak and strong surface affinity of BaP for NMs. The absorbance corresponding to BaP was measured in the supernatant by UV-vis at two wavelengths (300 and 384 nm). The percentage of BaP adsorbed at the surface of NMs was estimated taking into account the BaP instability in abiotic Ferticult® (with NMs) following centrifugation.
2.2. Gamete Collection
Rat cumulus–oocytes complex (COC) collection. Female superovulation was induced in prepubescent rats by an intraperitoneal injection of pregnant mare serum gonadotropin (20 U.I. PMSG) on day one and human chorionic gonadotropin (40 U.I. HCG) on day three. Twelve hours later, we collected oviducts containing oocytes surrounded by follicle cells after cervical dislocation euthanasia [51]. Once the cells from each oviduct were recovered, we left them equilibrate in Ferticult® medium at 37 °C and CO2 5% for 1 h [46].
Rat sperm cell collection. Male rats were previously anesthetized (Sevoflurane, vol % 8) and then euthanized with a 10 mL injection of Dolethal. After sacrifice, we collected and cut the epididymis to allow the exit of sperm into HTF-BSA culture medium (Human Tubal Fluid, Millipore, St-Quentin-en-Yvelines, France, with 0.4% BSA: Bovine Serum Albumin, Sigma-Aldrich, St. Quentin-Fallavier, France) for 1 h at 37 °C and CO2 5% under mineral oil (Sigma-Aldrich®, France) [30].
Human sperm collection. We used frozen human sperm from healthy fertile donors. After thawing, we aliquoted the preparation and centrifuged it for 10 minutes at 420× g. The supernatants were discarded, and the pellets were exposed to various exposure conditions [30].
2.3. Ethical Authorization
Ethical authorization for animal sampling of gametes was obtained from the National Ethics Committee on Animal Experimentation (2018061110211950-V2 #15447). We used Sprague-Dawley rats, Oncins France Strain A (623OFA), which were purchased from Charles River Laboratories (Lyon, France). Sexually mature 60-day-old male rats and prepubescent 26-day-old female rats were housed with free access to food and water until sacrifice.
Human sperm cells were purchased from GERMETHEQUE Biobank (BB-0033-00081 Marseille, France); informed consent was obtained from each donor for the inclusion of samples in the biobank and for their use in research experiments regarding human fertility in accordance with the 1975 Helsinki Declaration on human experimentation. The Scientific Committee approved the present study design (number 20130102).
2.4. Gamete Exposure and DNA Damage Evaluation by the Comet Assay
We exposed human sperm, rat sperm, and COCs to three experimental conditions: (i) aged CeO2 NMs at 1 µg·L−1 (called NMs); (ii) BaP at 1.13 µmol·L−1 (called BaP); (iii) aged CeO2 NMs at 1 µg·L−1 previously incubated with 1.13 µmol·L−1 BaP (called NMs+BaP). FertiCult® medium alone and Ferticult® medium containing 1% S9 mix and 1% DMSO were used as the negative control and internal control (IC), respectively. As a protocol verification, we also exposed rat sperm cells to Ferticult® medium 1% S9 mix, 1% DMSO, and CeO2 NMs (1 µg·L−1) (see Supplementary Materials, Figure S2). H2O2 (110 µmol·L−1) in Ferticult® medium was used as a positive control, and the H2O2 concentration was chosen based on previous studies [11,31,32]. At least three different experiments were performed for each condition. After exposure, we recovered all motile sperm cells by swim-up [8], and we measured sperm viability by eosin-nigrosine staining according to the WHO (WHO, 1999, Appendix IV.2) technique (100 cells were evaluated per condition). We then performed the alkaline comet assay according to the procedure described by Singh et al. (1988) [52] and adapted by Baumgartner et al. (2009) [53], which has already been described in ref [30,31]. DNA damage was quantified by the percentage of DNA in the tail of 100 randomly selected sperm cells from each triplicate slide per condition (at least 300 raw values analyzed per experiment, at least 900 in total per condition). Regarding the COC, we performed a comet assay according to the protocol described by Berthelot-Ricou et al. (2011) [54] and adapted by Préaubert et al. (2015) [32]. DNA damage was quantified by Olive Tail Moment (OTM) [55] in 2 replicated slides of each condition per experiment (at least 100 cumulus cells per experiment, 300 in total per condition, and at least 30 oocytes per experiment, 90 in total per condition).
The data are presented as the medians of % tail DNA or olive tail moment (OTM) values with 1st and 3rd quartiles. We performed a linear mixed model analysis with “condition” (exposure condition) as a fixed effect and “cells” (sperm cells, follicle cells, or oocytes) within the replicate slide as a random effect using the linear mixed effects regression (LMER) function of R software, version 3.6.0 (R Foundation for Statistical Computing, Vienna, Austria), to compare DNA damage among the various conditions. Pairwise differences of least-square means for all conditions were post hoc assessed. Statistical significance was set at p < 0.05.
3. Results and Discussion
3.1. DNA Damage in Sperm Cells Induced by Aged CeO2 NMs and/or BaP
In human and rat sperm cells, a significant increase in DNA damage was observed after 1 h of in vitro exposure to NMs+BaP versus that in the negative control and NMs and BaP alone groups (p < 0.001) (Figure 1a,b, Table 1). It is noteworthy that all the viability rates were over the normality threshold as stated by the WHO criteria [56]. The results are presented as the distribution of median values of the % tail DNA with 1st and 3rd quartiles obtained from three independent experiments. These values could inform about additive, synergistic, or antagonistic effects within the mixture [57]. In both human and rat sperm cells, a significantly higher genotoxicity was detected after exposure to the NM+BaP mixture compared to the toxicity of single contaminants, highlighting the additive effects of NMs and BaP when sperm cells are simultaneously exposed.
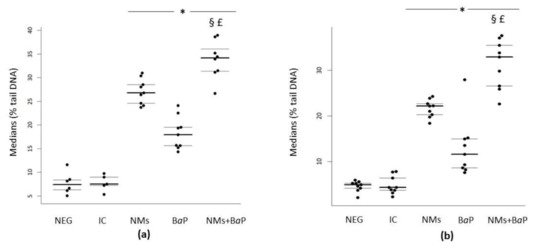
Figure 1.
Evaluation of DNA damage using the comet assay following in vitro exposure of human (a) and rat sperm (b) to NMs+BaP. Tested concentrations: Negative control = Figure 1. S9 mix, 1% DMSO), NMs: aged CeO2 NMs at 1 µg·L−1; BaP: BaP at 1.13 µmol·L−1; NMs+BaP: aged CeO2 NMs at 1 µg·L−1 previously incubated with 1.13 µmol·L−1 BaP. p < 0.05, for differences compared versus *: negative control (NEG); §: vs. NMs, £: vs. BaP.

Table 1.
Median values of the % tail DNA of each condition of three experiments, with 1st and 3rd quartiles, in rat and human sperm.
NMs and BaP are known to individually induce DNA damage on sperm cells, resulting in adverse effects on the fertilization rate [32] and sperm nucleus [8]. Our previous in vitro studies showed a significant increase in DNA damage in human sperm after exposure to 10 µg·L−1 of pristine CeO2 NMs. The mechanisms of the genotoxicity were indirectly attributed to oxidative stress via the adjunction of an antioxidant (L-ergothioneine) in the exposure medium [31]. We also observed a significant increase in intracellular ROS production after in vitro exposure to 1 µg·L−1 of aged CeO2 NMs in human sperm cells. This enhanced oxidative stress was attributed to a potential reductive dissolution of Ce(IV) in the vicinity of the plasma membrane of the cells into Ce(III) with pro-oxidant abilities [30]. It is noteworthy that CeO2 NM internalization within sperm cells was never observed under any exposure condition [30,31].
Conversely, it is well known that BaP directly penetrates sperm cells. Its metabolism involves the activation of the aryl hydrocarbon receptor, which increases the expression of cytochrome P450 1A1 and 1B1, followed by the generation of reactive metabolites (4,5-diol, 7,8-diol, and 9,10-diol). After the reactive bay region, diol epoxide may covalently bind to DNA and other cellular macromolecules, which initiate its toxicity, mutagenesis, and carcinogenesis [58]. BaP exposure in human is associated with BPDE-DNA adducts and ROS production in sperm [8,20,59,60,61,62]. Moreover, Zhang et al. (2019) recently demonstrated that in vivo exposure to BaP can also significantly change the DNA methylation of rat sperm, mainly through hypomethylation [63]. These changes are associated with alterations in embryonic and reproductive system development and with many genetic diseases, but it is still not understood whether these epigenetic changes are transgenerational and can then be transmitted to offspring [63].
Few recent toxicological studies have started considering the co-exposure to NMs and other contaminants [57]. For instance, Asweto et al. (2017) showed for the first time a synergistic interaction between Si-based NMs and BaP involved in enhancing their individual toxicity after in vitro co-exposure of endothelial cells. It causes excessive oxidative stress, leading to DNA damage, cell cycle arrest, and apoptosis [35]. Herein, we assessed whether the physicochemical interactions between BaP and aged CeO2 NMs might modify the behavior of BaP under abiotic conditions using UV-vis spectrometry. For [CeO2] over a [BaP] ratio of concentrations similar to that used with the cells (11.3 µmol·L−1 BaP for 10 µg·L−1 CeO2 NMs), we estimated that 44 ± 9% of BaP interacted with the surface of the NMs. This affinity is in agreement with previous studies showing the effect of ultrafine, airborne, carrier (nano)particles on the deposition, retention, and biological fate of PAHs [64,65,66,67]. Herein, we demonstrated that co-exposure to aged CeO2 NMs and BaP additively impact sperm cells. Consequently, the potential affinity of the BaP for the CeO2 NMs surface observed in abiotic media did not impact the toxicity. This could be either attributed to the BaP desorption from the CeO2 NMs surface related to reductive dissolution of nanocrystalline Ce(IV)O2 into Ce(III) at the vicinity of the cell membrane [30], but also to the limited number of BaP binding sites at the surface of the sperm cells and to the limited capacity of the constitutive CYP1A (cytochrome P4501A) enzymatic activity in sperm [47].
3.2. DNA Damage in COCs Induced by Aged CeO2 NMs and/or BaP
Interactions and close communication between cumulus cells and oocytes in COCs are critically important for oocyte maturation and quality. Cumulus cells are particularly sensitive to exogenous contaminants [68] and provide oocyte protection against short-lived perturbations in the surrounding environment [69,70,71].
In rat cumulus cells, significantly higher DNA damage was observed after 1 h of in vitro exposure to the NMs+BaP mixture compared to the negative control, NMs alone, and BaP alone (p < 0.001) (Figure 2a and Table 2). The results are presented as the distribution of median values of OTM with 1st and 3rd quartiles obtained from three independent experiments. The significantly different toxicities observed after exposure to the NM+BaP mixture highlight the additive effect of NMs and BaP upon co-exposure to CCs.
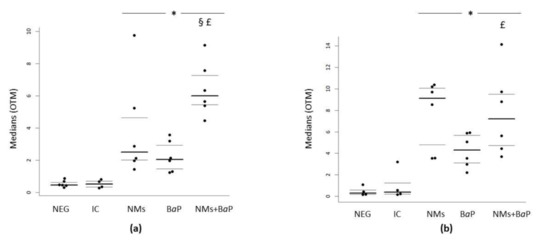
Figure 2.
Evaluation of DNA damage using the comet assay following in vitro exposure of rat cumulus cells (a) and oocytes (b) to NMs+BaP. Tested concentrations: negative control = Ferticult® medium, IC = intern control (Ferticult® 1% S9 mix, 1% DMSO), NMs: aged CeO2 NMs at 1 µg·L−1; BaP: BaP at 1.13 µmol·L−1; NMs+BaP: aged CeO2 NMs at 1 µg·L−1 previously incubated with 1.13 µmol·L−1 BaP. p < 0.05, for differences compared versus *: negative control (NEG); §: vs. NMs, £: vs. BaP.

Table 2.
Median values of % olive tail moment (OTM) of each condition of three experiments, with 1° and 3° quartiles, in cumulus–oocytes complexes (COCs).
It is well known that BaP metabolites impair follicle growth in vitro and increase primordial follicle atresia through the induction of apoptosis [72,73,74,75]. Siddique et al. (2013) demonstrated that in vitro exposure to BaP [1.5–45 µg·L−1] for 13 days induces oxidative stress in cumulus cells, highlighted by a significant increase in 8-OH-dG, which is a general biomarker of cellular oxidative stress and DNA oxidative damage [76]. Einaudi et al. (2014) showed a significant increase in DNA damage and BPDE-DNA adducts in cumulus cells after in vivo exposure to a single dose of BaP [13 mg/kg body weight] [11]. They observed BaP-induced genotoxicity [11], which was related to the different follicle maturation stages [77,78,79]. Conversely, there is still a large gap in the literature regarding the potential effect induced by NMs exposure on cumulus cells. A few previous studies reported a significant dose-dependent genotoxicity in cumulus cells exposed in vitro to 2.103 to 1·105 µg·L−1 pristine NMs, likely related to oxidative stress [33]. Moreover, during in vitro exposure of COCs to NMs, Courbiere et al. (2013) showed the ability of cumulus cells to internalize pristine CeO2 NMs (~8 nm) by endosomal trapping after in vitro exposure to 10·104 µg·L−1 CeO2 NMs [32,33]. Based on this internalization and contrary to the case of sperm cells, a so-called “Trojan horse effect” could have occurred in cumulus cells. Indeed, metal oxide NMs have already been shown to enhance the toxicity of contaminants adsorbed on their surface via modification of their bioavailability [57]. However, Figure 2a shows that despite the affinity of BaP for the surface of aged CeO2 NMs, co-exposure to NMs and BaP resulted in additive genotoxicity of the two single contaminants towards cumulus. Consequently, no Trojan horse effect modifying the toxicity of BaP or aged CeO2 NMs has been observed under our experimental conditions.
In rat oocytes, we detected a significant increase in DNA damage after in vitro exposure to NMs+BaP compared to the negative control and BaP alone (p < 0.001). In contrast to sperm and cumulus cells, we did not observe any significant difference in NMs+BaP exposure versus NMs alone (p > 0.05) (Figure 2b, Table 2). This result did not highlight any additive effect when oocytes were co-exposed to NMs and BaP. The results are presented as the distribution of median values of OTM with 1st and 3rd quartiles obtained from three independent experiments.
Few studies have explored the effect of CeO2 NMs on oocytes. In mouse the genotoxicity induced at 10 µg·L−1 of pristine CeO2 NM was attributed to oxidative stress [32]. Despite the protection of the zona pellucida, TEM analysis showed pristine CeO2 NMs in the perivitelline space (between the plasma membrane and the zona pellucida) after in vitro exposure to 10·104 µg·L−1 [32], highlighting the incomplete protection of cumulus cells against contaminants. Regarding BaP toxicity towards oocytes, it has been shown that the ovary possesses the ability to metabolically process BaP and obtain more reactive intermediates [80,81]. The generation of these metabolites is of importance, as they are capable of inducing cellular toxicity through the production of ROS and oxidative DNA damage [82], which has been linked to BaP-induced subfertility [83]. Rekhadevi et al. (2014) demonstrated that in vitro exposure of human ovarian subcellular fractions to 1 and 3 µmol·L−1 BaP induces metabolite accumulation, which contributes to premature ovarian failure [80]. An in vivo study showed that BaP oral exposure induced oxidative stress with an increased level of ROS and apoptosis in cumulus-denuded oocytes in mice after administration of BaP (10, 20, or 40 mg/kg body weight per day for 10 d), highlighting that oxidative stress is one of the mechanisms responsible for BaP metabolite-induced toxicity [84]. Additionally, Einaudi et al. (2014) also detected a significant increase in DNA damage in mouse oocytes after oral in vivo exposure to a single dose of BaP (13 mg/kg body weight) depending on the maturation stages [11]. The lower sensitivity of mature oocytes (exposed in antral follicles) to BaP-induced DNA damage could be due to oocytes that have reached the nuclear maturity required to repair DNA damage [77,78,79]. It has been shown that even the zona pellucida protects the oocytes, excluding some contaminants [85]; most biologically active molecules can pass through independently of the developmental stage [86].
Herein, we demonstrated that NMs+BaP exposure of oocytes did not induce any additive effect compared to NMs exposure alone, contrary to what we observed with sperm and cumulus cells. This result could be explained by the particular architecture and biology of COCs. First, there is structural protection around the oocyte due to the multiple layers of zona pellucida and cumulus cells [87,88]. These protective layers are gatekeepers for the oocyte [88,89] and act as a barrier between the oocyte and the extrafollicular environment [74,90,91], with cumulus cells able to select and process the metabolites that oocyte will receive [89]. This protection limits the contact between CeO2 NMs and the oocyte plasma membrane compared to CeO2 NMs interactions with sperm and cumulus cells. Second, in contrast to sperm and cumulus cells, oocytes have an efficient system to repair a variety of DNA lesions [92]. DNA repair activity in the zygote and during early development is, by definition, of maternal origin [93]. It is particularly important for germ cells to correct damage to their DNA, to avoid apoptosis, and prevent the transmission of genetic mutations to offspring [90,94]. Instead, sperm cells generally lack cytosolic antioxidants and fully functional DNA repair machinery, as they only possess the first enzyme in the base-excision repair pathway, OGG1, which removes the oxidized base, leaving a vulnerable abasic site [91]. Consequently, following the co-exposure of oocytes, efficient system repair protected the cells from NM-BaP-induced oxidative DNA damage, therefore resulting in a non-additive effect of the mixture of contaminants.
4. Conclusions
Drawing upon previous studies, we investigated the potential interaction between aged CeO2 NMs and BaP and the consequential impact on reproductive cells. We demonstrated additive toxic effects of NM+BaP exposure on sperm and cumulus cells compared to those generated by the individual pollutants. However, we did not show any additive effect in rat oocytes. This was attributed to the protection of the oocyte by the cumulus cells and to the oocyte’s efficient system to repair DNA damage compared to that of cumulus and sperm cells. The exposure of COCs and the subsequent genotoxic analysis by the comet assay of both cell types separately allowed us to analyze the impact of cumulus cells on the DNA damage of oocytes, which complies with the real exposure conditions. To further understand the impact of co-exposure on reproduction, in vivo studies are required. In vivo, the behavior, the time exposure, and fate of the two pollutants are expected to be different, which should affect their bioavailability, bioaccumulation, and toxicity.
5. Limitations and Strengths
Our study considers for the first time a co-contamination scenario of germ cells that is close to the real conditions in which humans are likely exposed to different emissions of ultrafine particles and many other pollutants. We considered the potential exposure of human and rat gametes to the combination of aged CeO2 NMs and BaP, which are released in the atmosphere after combustion in a diesel engine. The CeO2 NMs used in this study are representative of nano-based diesel fuel additives likely released by combustion in a diesel engine and to which people are potentially exposed [30]. Even though this study reflects realistic exposure conditions because of co-exposure to low concentrations of aged CeO2 NMs and BaP, it is limited by its in vitro nature.
Supplementary Materials
The following are available online at https://www.mdpi.com/2079-4991/11/2/478/s1, Figure S1. Supplementary controls used during the assessment of DNA damage by the comet assay. Figure S2. Standard curves for BaP measurements by UV-Vis spectrometry.
Author Contributions
Conceptualization, M.A., J.R., and J.P.; data curation, M.C.; formal analysis, M.C. and N.R.; funding acquisition, M.A., J.R., and J.P.; investigation, M.C.; methodology, M.C. and V.T.; project administration, J.R. and J.P.; supervision, M.A., J.R., and J.P.; validation, M.A., J.R., and J.P.; visualization, J.P.; writing—original draft, M.C.; writing—review and editing, M.C., M.A., J.R., and J.P. All authors have read and agreed to the published version of the manuscript.
Funding
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement no. 713750. Additionally, it was performed with the financial support of the Regional Council of Provence- Alpes-Côte d’Azur and A*MIDEX (n° ANR- 11-IDEX-0001-02), funded by the Investissements d’Avenir project funded by the French Government and managed by the French National Research Agency (ANR). The project also received funding from the Fédération de Recherche ECCOREV n° 3098.
Data Availability Statement
Data is contained within the article or supplementary material.
Acknowledgments
This work is a contribution to the Labex Serenade (No. ANR-11-LABX-0064) funded by the “Investissements d’Avenir” French Government programme of the French National Research Agency (ANR) through the A*MIDEX project (no. ANR-11-IDEX-0001-02). This work is also a contribution to the OSU-Institut Pythéas. The authors acknowledge the CNRS for the funding of the IRP iNOVE.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Institute, H.E. Diesel Exhaust: Critical Analysis of Emissions, Exposure, and Health Effects. Available online: https://www.healtheffects.org/publication/diesel-exhaust-critical-analysis-emissions-exposure-and-health-effects (accessed on 2 January 2021).
- Kagawa, J. Health Effects of Diesel Exhaust Emissions--a Mixture of Air Pollutants of Worldwide Concern. Toxicology 2002, 181–182, 349–353. [Google Scholar] [CrossRef]
- Humans, I.W.G. on the E. of C.R. to Diesel and Gasoline Engine Exhausts and Some Nitroarenes. International Agency for Research on Cancer: Lion, France, 2014; ISBN 978-92-832-1328-4. [Google Scholar]
- Marr, L.C.; Kirchstetter, T.W.; Harley, R.A.; Miguel, A.H.; Hering, S.V.; Hammond, S.K. Characterization of Polycyclic Aromatic Hydrocarbons in Motor Vehicle Fuels and Exhaust Emissions. Environ. Sci. Technol. 1999, 33, 3091–3099. [Google Scholar] [CrossRef]
- Keith, L.H. The Source of U.S. EPA’s Sixteen PAH Priority Pollutants. Polycycl. Aromat. Compd. 2015, 35, 147–160. [Google Scholar] [CrossRef]
- Zheng, X.; Wu, Y.; Zhang, S.; Hu, J.; Zhang, K.M.; Li, Z.; He, L.; Hao, J. Characterizing Particulate Polycyclic Aromatic Hydrocarbon Emissions from Diesel Vehicles Using a Portable Emissions Measurement System. Sci. Rep. 2017, 7, 10058. [Google Scholar] [CrossRef] [PubMed]
- European Commission Proposal for a DIRECTIVE OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL Relating to Restrictions on the Marketing and Use of Certain Polycyclic Aromatic Hydrocarbons in Extender Oils and Tyres (Twenty-Seventh Amendment of Council Directive 76/769/EEC) EUR-Lex—52004PC0098—EN. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:52004PC0098&from=HU (accessed on 2 January 2021).
- Perrin, J.; Tassistro, V.; Mandon, M.; Grillo, J.-M.; Botta, A.; Sari-Minodier, I. Tobacco Consumption and Benzo(a)Pyrene-Diol-Epoxide-DNA Adducts in Spermatozoa: In Smokers, Swim-up Procedure Selects Spermatozoa with Decreased DNA Damage. Fertil. Steril. 2011, 95, 2013–2017. [Google Scholar] [CrossRef] [PubMed]
- European Commission W.G.O.P.A.H. Ambient Air Pollution by Polycyclic Aromatic Hydrocarbons (PAH)—Cerca Con Google. Available online: https://ec.europa.eu/environment/air/pdf/annex_pah.pdf (accessed on 1 February 2021).
- Watanabe, S.; Kamiguchi, Y. Chromosome Analysis of Human Spermatozoa Following in Vitro Exposure to Cyclophosphamide, Benzo(a)Pyrene and N-Nitrosodimethylamine in the Presence of Rat Liver S9. Mutat. Res. 2001, 491, 57–63. [Google Scholar] [CrossRef]
- Einaudi, L.; Courbiere, B.; Tassistro, V.; Prevot, C.; Sari-Minodier, I.; Orsiere, T.; Perrin, J. In Vivo Exposure to Benzo(a)Pyrene Induces Significant DNA Damage in Mouse Oocytes and Cumulus Cells. Hum. Reprod. Oxf. Engl. 2014, 29, 548–554. [Google Scholar] [CrossRef]
- Sipinen, V.; Laubenthal, J.; Baumgartner, A.; Cemeli, E.; Linschooten, J.O.; Godschalk, R.W.L.; Van Schooten, F.J.; Anderson, D.; Brunborg, G. In Vitro Evaluation of Baseline and Induced DNA Damage in Human Sperm Exposed to Benzo[a]Pyrene or Its Metabolite Benzo[a]Pyrene-7,8-Diol-9,10-Epoxide, Using the Comet Assay. Mutagenesis 2010, 25, 417–425. [Google Scholar] [CrossRef]
- Alamo, A.; Condorelli, R.A.; Mongioì, L.M.; Cannarella, R.; Giacone, F.; Calabrese, V.; La Vignera, S.; Calogero, A.E. Environment and Male Fertility: Effects of Benzo-α-Pyrene and Resveratrol on Human Sperm Function In Vitro. J. Clin. Med. 2019, 8, 561. [Google Scholar] [CrossRef]
- Ginsberg, G.L.; Atherholt, T.B. Transport of DNA-Adducting Metabolites in Mouse Serum Following Benzo[a]Pyrene Administration. Carcinogenesis 1989, 10, 673–679. [Google Scholar] [CrossRef]
- Mattison, D.R.; Singh, H.; Takizawa, K.; Thomford, P.J. Ovarian Toxicity of Benzo(a)Pyrene and Metabolites in Mice. Reprod. Toxicol. 1989, 3, 115–125. [Google Scholar] [CrossRef]
- Neal, M.S.; Zhu, J.; Holloway, A.C.; Foster, W.G. Follicle Growth Is Inhibited by Benzo-[a]-Pyrene, at Concentrations Representative of Human Exposure, in an Isolated Rat Follicle Culture Assay. Hum. Reprod. Oxf. Engl. 2007, 22, 961–967. [Google Scholar] [CrossRef][Green Version]
- Mattison, D.R.; White, N.B.; Nightingale, M.R. The Effect of Benzo(a)Pyrene on Fertility, Primordial Oocyte Number, and Ovarian Response to Pregnant Mare’s Serum Gonadotropin. Pediatr. Pharmacol. 1980, 1, 143–151. [Google Scholar]
- Sheng, F.; Ji, Y.; Ma, Y.; Ding, H.; Zhang, Q.; Li, W. Polycyclic Aromatic Hydrocarbons Cause Follicle Atresia and Apoptosis in Mouse Ovarian Follicles Cultured in Vitro That Can Be Reduced with the Activator of PI3K/Akt Pathway, 740Y-P. Crit. Care Obstet. Gynecol. 2018, 4. [Google Scholar] [CrossRef]
- Neal, M.S.; Mulligan Tuttle, A.M.; Casper, R.F.; Lagunov, A.; Foster, W.G. Aryl Hydrocarbon Receptor Antagonists Attenuate the Deleterious Effects of Benzo[a]Pyrene on Isolated Rat Follicle Development. Reprod. Biomed. Online 2010, 21, 100–108. [Google Scholar] [CrossRef]
- Revel, A.; Raanani, H.; Younglai, E.; Xu, J.; Han, R.; Savouret, J.F.; Casper, R.F. Resveratrol, a Natural Aryl Hydrocarbon Receptor Antagonist, Protects Sperm from DNA Damage and Apoptosis Caused by Benzo(a)Pyrene. Reprod. Toxicol. Elmsford N 2001, 15, 479–486. [Google Scholar] [CrossRef]
- Shi, J.P.; Evans, D.E.; Khan, A.A.; Harrison, R.M. Sources and Concentration of Nanoparticles (<10 nm Diameter) in the Urban Atmosphere. Atmos. Environ. 2001, 35, 1193–1202. [Google Scholar] [CrossRef]
- Slezakova, K.; Morais, S.; do Carmo Pereira, M. Atmospheric Nanoparticles and Their Impacts on Public Health. Curr. Top. Public Health 2013. [Google Scholar] [CrossRef]
- Donaldson, K.; Stone, V.; Tran, C.L.; Kreyling, W.; Borm, P.J.A. Nanotoxicology. Occup. Environ. Med. 2004, 61, 727–728. [Google Scholar] [CrossRef]
- Gaiser, B.K.; Fernandes, T.F.; Jepson, M.; Lead, J.R.; Tyler, C.R.; Stone, V. Assessing Exposure, Uptake and Toxicity of Silver and Cerium Dioxide Nanoparticles from Contaminated Environments. Environ. Health Glob. Access Sci. Source 2009, 8 Suppl. 1, S2. [Google Scholar] [CrossRef] [PubMed]
- Park, B.; Donaldson, K.; Duffin, R.; Tran, L.; Kelly, F.; Mudway, I.; Morin, J.-P.; Guest, R.; Jenkinson, P.; Samaras, Z.; et al. Hazard and Risk Assessment of a Nanoparticulate Cerium Oxide-Based Diesel Fuel Additive—A Case Study. Inhal. Toxicol. 2008, 20, 547–566. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Nazarenko, Y.; Zhang, L.; Calderon, L.; Lee, K.-B.; Garfunkel, E.; Schwander, S.; Tetley, T.D.; Chung, K.F.; Porter, A.E.; et al. Impacts of a Nanosized Ceria Additive on Diesel Engine Emissions of Particulate and Gaseous Pollutants. Environ. Sci. Technol. 2013, 47, 13077–13085. [Google Scholar] [CrossRef]
- Benameur, L.; Auffan, M.; Cassien, M.; Liu, W.; Culcasi, M.; Rahmouni, H.; Stocker, P.; Tassistro, V.; Bottero, J.-Y.; Rose, J.; et al. DNA Damage and Oxidative Stress Induced by CeO2 Nanoparticles in Human Dermal Fibroblasts: Evidence of a Clastogenic Effect as a Mechanism of Genotoxicity. Nanotoxicology 2015, 9, 696–705. [Google Scholar] [CrossRef]
- Minarchick, V.C.; Stapleton, P.A.; Sabolsky, E.M.; Nurkiewicz, T.R. Cerium Dioxide Nanoparticle Exposure Improves Microvascular Dysfunction and Reduces Oxidative Stress in Spontaneously Hypertensive Rats. Front. Physiol. 2015, 6. [Google Scholar] [CrossRef]
- Adebayo, O.A.; Akinloye, O.; Adaramoye, O.A. Cerium Oxide Nanoparticle Elicits Oxidative Stress, Endocrine Imbalance and Lowers Sperm Characteristics in Testes of Balb/c Mice. Andrologia 2018, 50. [Google Scholar] [CrossRef]
- Cotena, M.; Auffan, M.; Robert, S.; Tassistro, V.; Resseguier, N.; Rose, J.; Perrin, J. CeO2 Nanomaterials from Diesel Engine Exhaust Induce DNA Damage and Oxidative Stress in Human and Rat Sperm In Vitro. Nanomaterials 2020, 10, 2327. [Google Scholar] [CrossRef] [PubMed]
- Préaubert, L.; Tassistro, V.; Auffan, M.; Sari-Minodier, I.; Rose, J.; Courbiere, B.; Perrin, J. Very Low Concentration of Cerium Dioxide Nanoparticles Induce DNA Damage, but No Loss of Vitality, in Human Spermatozoa. Toxicol. Vitro Int. J. Publ. Assoc. BIBRA 2018, 50, 236–241. [Google Scholar] [CrossRef]
- Preaubert, L.; Courbiere, B.; Achard, V.; Tassistro, V.; Greco, F.; Orsiere, T.; Bottero, J.-Y.; Rose, J.; Auffan, M.; Perrin, J. Cerium Dioxide Nanoparticles Affect in Vitro Fertilization in Mice. Nanotoxicology 2016, 10, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Courbiere, B.; Auffan, M.; Rollais, R.; Tassistro, V.; Bonnefoy, A.; Botta, A.; Rose, J.; Orsière, T.; Perrin, J. Ultrastructural Interactions and Genotoxicity Assay of Cerium Dioxide Nanoparticles on Mouse Oocytes. Int. J. Mol. Sci. 2013, 14, 21613–21628. [Google Scholar] [CrossRef]
- Qin, F.; Shen, T.; Li, J.; Qian, J.; Zhang, J.; Zhou, G.; Tong, J. SF-1 Mediates Reproductive Toxicity Induced by Cerium Oxide Nanoparticles in Male Mice. J. Nanobiotechnology 2019, 17, 41. [Google Scholar] [CrossRef]
- Asweto, C.O.; Wu, J.; Hu, H.; Feng, L.; Yang, X.; Duan, J.; Sun, Z. Combined Effect of Silica Nanoparticles and Benzo[a]Pyrene on Cell Cycle Arrest Induction and Apoptosis in Human Umbilical Vein Endothelial Cells. Int. J. Environ. Res. Public. Health 2017, 14, 289. [Google Scholar] [CrossRef] [PubMed]
- Auffan, M.; Rose, J.; Proux, O.; Masion, A.; Liu, W.; Benameur, L.; Ziarelli, F.; Botta, A.; Chaneac, C.; Bottero, J.-Y. Is There a Trojan-Horse Effect during Magnetic Nanoparticles and Metalloid Cocontamination of Human Dermal Fibroblasts? Environ. Sci. Technol. 2012, 46, 10789–10796. [Google Scholar] [CrossRef] [PubMed]
- Silins, I.; Högberg, J. Combined Toxic Exposures and Human Health: Biomarkers of Exposure and Effect. Int. J. Environ. Res. Public. Health 2011, 8, 629–647. [Google Scholar] [CrossRef]
- Roell, K.R.; Reif, D.M.; Motsinger-Reif, A.A. An Introduction to Terminology and Methodology of Chemical Synergy-Perspectives from Across Disciplines. Front. Pharmacol. 2017, 8, 158. [Google Scholar] [CrossRef] [PubMed]
- Phillipson, C.E.; Ioannides, C. Metabolic Action of Polycyclic Aromatic Hydrocarbons to Mutagens in the Ames Test by Various Animal Species Including Man. Mutat. Res. Mol. Mech. Mutagen. 1989, 211, 147–151. [Google Scholar] [CrossRef]
- Auffan, M.; Tella, M.; Liu, W.; Pariat, A.; Cabié, M.; Borschneck, D.; Angeletti, B.; Landrot, G.; Mouneyrac, C.; Giamberini, L.; et al. Structural and Physical–Chemical Behavior of a CeO2 Nanoparticle Based Diesel Additive during Combustion and Environmental Release. Environ. Sci. 2017, 4, 1974–1980. [Google Scholar] [CrossRef]
- Audebert, M.; Riu, A.; Jacques, C.; Hillenweck, A.; Jamin, E.L.; Zalko, D.; Cravedi, J.-P. Use of the ΓH2AX Assay for Assessing the Genotoxicity of Polycyclic Aromatic Hydrocarbons in Human Cell Lines. Toxicol. Lett. 2010, 199, 182–192. [Google Scholar] [CrossRef]
- Alvares, A.P.; Kappas, A. Heterogeneity of Cytochrome P-450s Induced by Polychlorinated Biphenyls. J. Biol. Chem. 1977, 252, 6373–6378. [Google Scholar] [CrossRef]
- Thomas, P.E.; Reik, L.M.; Ryan, D.E.; Levin, W. Induction of Two Immunochemically Related Rat Liver Cytochrome P-450 Isozymes, Cytochromes P-450c and P-450d, by Structurally Diverse Xenobiotics. J. Biol. Chem. 1983, 258, 4590–4598. [Google Scholar] [CrossRef]
- van Leeuwen, D.M.; Gottschalk, R.W.H.; van Herwijnen, M.H.; Moonen, E.J.; Kleinjans, J.C.S.; van Delft, J.H.M. Differential Gene Expression in Human Peripheral Blood Mononuclear Cells Induced by Cigarette Smoke and Its Constituents. Toxicol. Sci. Off. J. Soc. Toxicol. 2005, 86, 200–210. [Google Scholar] [CrossRef]
- Baumgartner, A.; Kurzawa-Zegota, M.; Laubenthal, J.; Cemeli, E.; Anderson, D. Comet-Assay Parameters as Rapid Biomarkers of Exposure to Dietary/Environmental Compounds—An in Vitro Feasibility Study on Spermatozoa and Lymphocytes. Mutat. Res. 2012, 743, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Greco, F.; Perrin, J.; Auffan, M.; Tassistro, V.; Orsière, T.; Courbiere, B. A New Approach for the Oocyte Genotoxicity Assay: Adaptation of Comet Assay on Mouse Cumulus–Oocyte Complexes. Lab. Anim. 2015. [Google Scholar] [CrossRef] [PubMed]
- Madureira, D.J.; Weiss, F.T.; Midwoud, P.V.; Helbling, D.E.; Sturla, S.J.; Schirmer, K. Systems Toxicology Approach to Understand the Kinetics of Benzo(a)Pyrene Uptake, Biotransformation, and DNA Adduct Formation in a Liver Cell Model. Available online: https://pubs.acs.org/doi/pdf/10.1021/tx400446q (accessed on 10 January 2021).
- Peters, T. Serum Albumin. Adv. Protein Chem. 1985, 37, 161–245. [Google Scholar] [CrossRef]
- Fasano, M.; Curry, S.; Terreno, E.; Galliano, M.; Fanali, G.; Narciso, P.; Notari, S.; Ascenzi, P. The Extraordinary Ligand Binding Properties of Human Serum Albumin. IUBMB Life 2005, 57, 787–796. [Google Scholar] [CrossRef]
- Motwani, H.V.; Westberg, E.; Törnqvist, M. Interaction of Benzo[a]Pyrene Diol Epoxide Isomers with Human Serum Albumin: Site Specific Characterisation of Adducts and Associated Kinetics. Sci. Rep. 2016, 6, 36243. [Google Scholar] [CrossRef]
- Roustan, A.; Perrin, J.; Berthelot-Ricou, A.; Lopez, E.; Botta, A.; Courbiere, B. Evaluating Methods of Mouse Euthanasia on the Oocyte Quality: Cervical Dislocation versus Isoflurane Inhalation. Lab. Anim. 2012, 46, 167–169. [Google Scholar] [CrossRef]
- Singh, N.P.; McCoy, M.T.; Tice, R.R.; Schneider, E.L. A Simple Technique for Quantitation of Low Levels of DNA Damage in Individual Cells. Exp. Cell Res. 1988, 175, 184–191. [Google Scholar] [CrossRef]
- Baumgartner, A.; Cemeli, E.; Anderson, D. The Comet Assay in Male Reproductive Toxicology. Cell Biol. Toxicol. 2009, 25, 81–98. [Google Scholar] [CrossRef] [PubMed]
- Berthelot-Ricou, A.; Perrin, J.; Di Giorgio, C.; De Meo, M.; Botta, A.; Courbiere, B. Comet Assay on Mouse Oocytes: An Improved Technique to Evaluate Genotoxic Risk on Female Germ Cells. Fertil. Steril. 2011, 95, 1452–1457. [Google Scholar] [CrossRef]
- Tice, R.R.; Agurell, E.; Anderson, D.; Burlinson, B.; Hartmann, A.; Kobayashi, H.; Miyamae, Y.; Rojas, E.; Ryu, J.C.; Sasaki, Y.F. Single Cell Gel/Comet Assay: Guidelines for in Vitro and in Vivo Genetic Toxicology Testing. Environ. Mol. Mutagen. 2000, 35, 206–221. [Google Scholar] [CrossRef]
- World Health Organization WHO Laboratory Manual for the Examination and Processing of Human Semen. Available online: https://www.who.int/publications-detail-redirect/9789241547789 (accessed on 3 January 2021).
- Deng, R.; Lin, D.; Zhu, L.; Majumdar, S.; White, J.C.; Gardea-Torresdey, J.L.; Xing, B. Nanoparticle Interactions with Co-Existing Contaminants: Joint Toxicity, Bioaccumulation and Risk. Nanotoxicology 2017, 11, 591–612. [Google Scholar] [CrossRef] [PubMed]
- Mumtaz, M.M.; George, J.D.; Gold, K.W.; Cibulas, W.; DeRosa, C.T. ATSDR Evaluation of Health Effects of Chemicals. IV. Polycyclic Aromatic Hydrocarbons (PAHs): Understanding a Complex Problem. Toxicol. Ind. Health 1996, 12, 742–971. [Google Scholar] [CrossRef] [PubMed]
- Oliveri Conti, G.; Calogero, A.E.; Giacone, F.; Fiore, M.; Barchitta, M.; Agodi, A.; Ferrante, M. B(a)P Adduct Levels and Fertility: A Cross-sectional Study in a Sicilian Population. Mol. Med. Rep. 2017, 15, 3398–3404. [Google Scholar] [CrossRef] [PubMed]
- Senft, A.P.; Dalton, T.P.; Nebert, D.W.; Genter, M.B.; Puga, A.; Hutchinson, R.J.; Kerzee, J.K.; Uno, S.; Shertzer, H.G. Mitochondrial Reactive Oxygen Production Is Dependent on the Aromatic Hydrocarbon Receptor. Free Radic. Biol. Med. 2002, 33, 1268–1278. [Google Scholar] [CrossRef]
- Zenzes, M.T.; Puy, L.A.; Bielecki, R.; Reed, T.E. Detection of Benzo[a]Pyrene Diol Epoxide-DNA Adducts in Embryos from Smoking Couples: Evidence for Transmission by Spermatozoa. Mol. Hum. Reprod. 1999, 5, 125–131. [Google Scholar] [CrossRef]
- Xia, Y.; Zhu, P.; Han, Y.; Lu, C.; Wang, S.; Gu, A.; Fu, G.; Zhao, R.; Song, L.; Wang, X. Urinary Metabolites of Polycyclic Aromatic Hydrocarbons in Relation to Idiopathic Male Infertility. Hum. Reprod. Oxf. Engl. 2009, 24, 1067–1074. [Google Scholar] [CrossRef]
- Zhang, C.M.; Sun, Z.X.; Wang, Z.L.; Chen, J.S.; Chang, Z.; Wang, Z.; Zhu, L.; Ma, Z.H.; Peng, Y.J.; Xu, Z.A.; et al. Abnormal Methylation of Spermatozoa Induced by Benzo(a)Pyrene in Rats. Hum. Exp. Toxicol. 2019, 38, 846–856. [Google Scholar] [CrossRef]
- Mahgoub, H.A. Nanoparticles Used for Extraction of Polycyclic Aromatic Hydrocarbons. Available online: https://www.hindawi.com/journals/jchem/2019/4816849/ (accessed on 3 January 2021).
- Sun, J.D.; Wolff, R.K.; Kanapilly, G.M. Deposition, Retention, and Biological Fate of Inhaled Benzo(a)Pyrene Adsorbed onto Ultrafine Particles and as a Pure Aerosol. Toxicol. Appl. Pharmacol. 1982, 65, 231–244. [Google Scholar] [CrossRef]
- Yang, K.; Zhu, L.; Xing, B. Adsorption of Polycyclic Aromatic Hydrocarbons by Carbon Nanomaterials. Environ. Sci. Technol. 2006, 40, 1855–1861. [Google Scholar] [CrossRef]
- Sahle-Demessie, E.; Han, C.; Zhao, A.; Hahn, B.; Grecsek, H. Interaction of Engineered Nanomaterials with Hydrophobic Organic Pollutants. Nanotechnology 2016, 27, 284003. [Google Scholar] [CrossRef]
- Campen, K.A.; McNatty, K.P.; Pitman, J.L. A Protective Role of Cumulus Cells after Short-Term Exposure of Rat Cumulus Cell-Oocyte Complexes to Lifestyle or Environmental Contaminants. Reprod. Toxicol. Elmsford N 2017, 69, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Tatemoto, H.; Sakurai, N.; Muto, N. Protection of Porcine Oocytes Against Apoptotic Cell Death Caused by Oxidative Stress During In Vitro Maturation: Role of Cumulus Cells1. Biol. Reprod. 2000, 63, 805–810. [Google Scholar] [CrossRef]
- Tanaka, H.; Takeo, S.; Monji, Y.; Kuwayama, T.; Iwata, H. Maternal Liver Damage Delays Meiotic Resumption in Bovine Oocytes through Impairment of Signalling Cascades Originated from Low P38MAPK Activity in Cumulus Cells. Reprod. Domest. Anim. Zuchthyg. 2014, 49, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Shaeib, F.; Khan, S.N.; Ali, I.; Thakur, M.; Saed, M.G.; Dai, J.; Awonuga, A.O.; Banerjee, J.; Abu-Soud, H.M. The Defensive Role of Cumulus Cells Against Reactive Oxygen Species Insult in Metaphase II Mouse Oocytes. Reprod. Sci. Thousand Oaks Calif. 2016, 23, 498–507. [Google Scholar] [CrossRef]
- Mattison, D.R. The Mechanisms of Action of Reproductive Toxins. Am. J. Ind. Med. 1983, 4, 65–79. [Google Scholar] [CrossRef]
- Sadeu, J.C.; Foster, W.G. Cigarette Smoke Condensate Exposure Delays Follicular Development and Function in a Stage-Dependent Manner. Fertil. Steril. 2011, 95, 2410–2417. [Google Scholar] [CrossRef] [PubMed]
- Sadeu, J.C.; Foster, W.G. Effect of in Vitro Exposure to Benzo[a]Pyrene, a Component of Cigarette Smoke, on Folliculogenesis, Steroidogenesis and Oocyte Nuclear Maturation. Reprod. Toxicol. 2011, 31, 402–408. [Google Scholar] [CrossRef]
- Esengen, S.; Seçkin, U.; Borman, P.; Bodur, H.; Kutsal, Y.G.; Yücel, M. Drug Consumption in a Group of Elderly Residents of a Nursing Home: Relationship to Cognitive Impairment and Disability. J. Am. Med. Dir. Assoc. 2000, 1, 197–201. [Google Scholar]
- Siddique, S.; Sadeu, J.C.; Foster, W.G.; Feng, Y.-L.; Zhu, J. In Vitro Exposure to Cigarette Smoke Induces Oxidative Stress in Follicular Cells of F₁ Hybrid Mice. J. Appl. Toxicol. JAT 2014, 34, 224–226. [Google Scholar] [CrossRef]
- Murdoch, W.J.; Van Kirk, E.A. Estrogenic Upregulation of DNA Polymerase Beta in Oocytes of Preovulatory Ovine Follicles. Mol. Reprod. Dev. 2001, 58, 417–423. [Google Scholar] [CrossRef]
- Maman, E.; Prokopis, K.; Levron, J.; Carmely, A.; Dor, J.; Meirow, D. Does Controlled Ovarian Stimulation Prior to Chemotherapy Increase Primordial Follicle Loss and Diminish Ovarian Reserve? An Animal Study. Hum. Reprod. Oxf. Engl. 2009, 24, 206–210. [Google Scholar] [CrossRef]
- Meirow, D.; Epstein, M.; Lewis, H.; Nugent, D.; Gosden, R.G. Administration of Cyclophosphamide at Different Stages of Follicular Maturation in Mice: Effects on Reproductive Performance and Fetal Malformations. Hum. Reprod. Oxf. Engl. 2001, 16, 632–637. [Google Scholar] [CrossRef] [PubMed]
- Rekhadevi, P.; Diggs, D.; Huderson, A.; Harris, K.; Archibong, A.; Ramesh, A. Metabolism of the Environmental Toxicant Benzo(a)Pyrene by Subcellular Fractions of Human Ovary. Hum. Exp. Toxicol. 2014, 33, 196–202. [Google Scholar] [CrossRef]
- Mattison, D.R.; Shiromizu, K.; Nightingale, M.S. Oocyte Destruction by Polycyclic Aromatic Hydrocarbons. Am. J. Ind. Med. 1983, 4, 191–202. [Google Scholar] [CrossRef]
- Bolton, J.L.; Trush, M.A.; Penning, T.M.; Dryhurst, G.; Monks, T.J. Role of Quinones in Toxicology. Chem. Res. Toxicol. 2000, 13, 135–160. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, A.; Archibong, A.E.; Niaz, M.S. Ovarian Susceptibility to Benzo[a]Pyrene: Tissue Burden of Metabolites and DNA Adducts in F-344 Rats. J. Toxicol. Environ. Health A 2010, 73, 1611–1625. [Google Scholar] [CrossRef]
- Zhang, M.; Miao, Y.; Chen, Q.; Cai, M.; Dong, W.; Dai, X.; Lu, Y.; Zhou, C.; Cui, Z.; Xiong, B. BaP Exposure Causes Oocyte Meiotic Arrest and Fertilization Failure to Weaken Female Fertility. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2018, 32, 342–352. [Google Scholar] [CrossRef]
- Eaglesome, M.D.; Hare, W.C.; Singh, E.L. Embryo Transfer: A Discussion on Its Potential for Infectious Disease Control Based on a Review of Studies on Infection of Gametes and Early Embryos by Various Agents. Can. Vet. J. Rev. Veterinaire Can. 1980, 21, 106–112. [Google Scholar]
- Turner, K.; Horobin, R.W. Permeability of the Mouse Zona Pellucida: A Structure-Staining-Correlation Model Using Coloured Probes. J. Reprod. Fertil. 1997, 111, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, T. Determination of Follicle Growth Rate in the Ovary of the Immature Mouse. J. Reprod. Fertil. 1970, 21, 81–93. [Google Scholar] [CrossRef]
- Hou, C.-C.; Zhu, J.-Q. Nanoparticles and Female Reproductive System: How Do Nanoparticles Affect Oogenesis and Embryonic Development. Oncotarget 2017, 8, 109799–109817. [Google Scholar] [CrossRef] [PubMed]
- von Mengden, L.; Klamt, F.; Smitz, J. Redox Biology of Human Cumulus Cells: Basic Concepts, Impact on Oocyte Quality, and Potential Clinical Use. Antioxid. Redox Signal. 2020, 32, 522–535. [Google Scholar] [CrossRef] [PubMed]
- Derijck, A.; van der Heijden, G.; Giele, M.; Philippens, M.; de Boer, P. DNA Double-Strand Break Repair in Parental Chromatin of Mouse Zygotes, the First Cell Cycle as an Origin of de Novo Mutation. Hum. Mol. Genet. 2008, 17, 1922–1937. [Google Scholar] [CrossRef] [PubMed]
- Drevet, J.R.; Aitken, R.J. Oxidative Damage to Sperm DNA: Attack and Defense. Adv. Exp. Med. Biol. 2019, 1166, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Stringer, J.M.; Winship, A.; Liew, S.H.; Hutt, K. The Capacity of Oocytes for DNA Repair. Cell. Mol. Life Sci. CMLS 2018, 75, 2777–2792. [Google Scholar] [CrossRef] [PubMed]
- Ménézo, Y.; Dale, B.; Cohen, M. DNA Damage and Repair in Human Oocytes and Embryos: A Review. Zygote Camb. Engl. 2010, 18, 357–365. [Google Scholar] [CrossRef]
- van den Berg, M.M.J.; van Maarle, M.C.; van Wely, M.; Goddijn, M. Genetics of Early Miscarriage. Biochim. Biophys. Acta Mol. Basis Dis. 2012, 1822, 1951–1959. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).