Lead-Free Metal Halide Perovskites for Hydrogen Evolution from Aqueous Solutions
Abstract
:1. Introduction
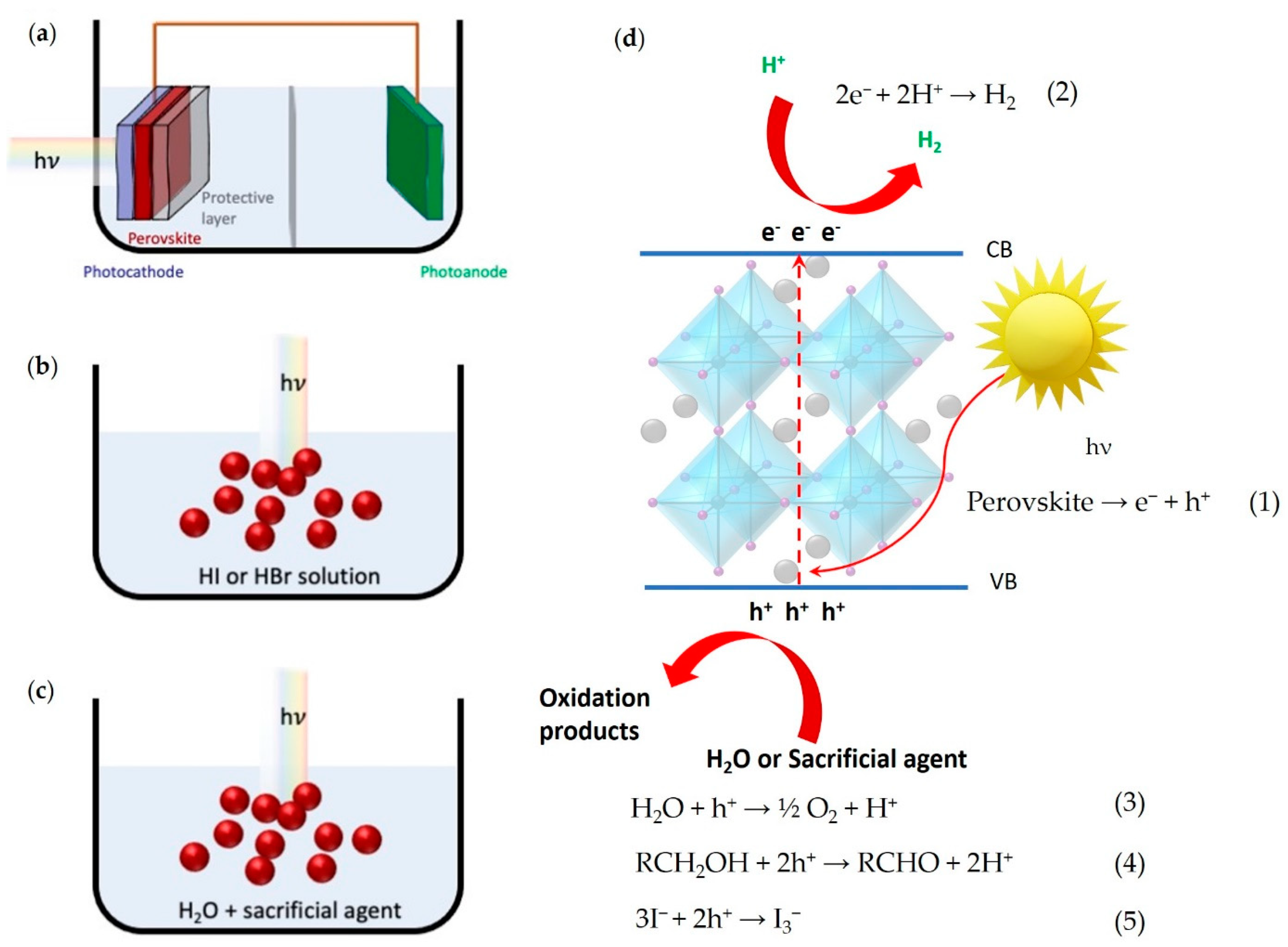
2. General Mechanisms and Figures of Merit
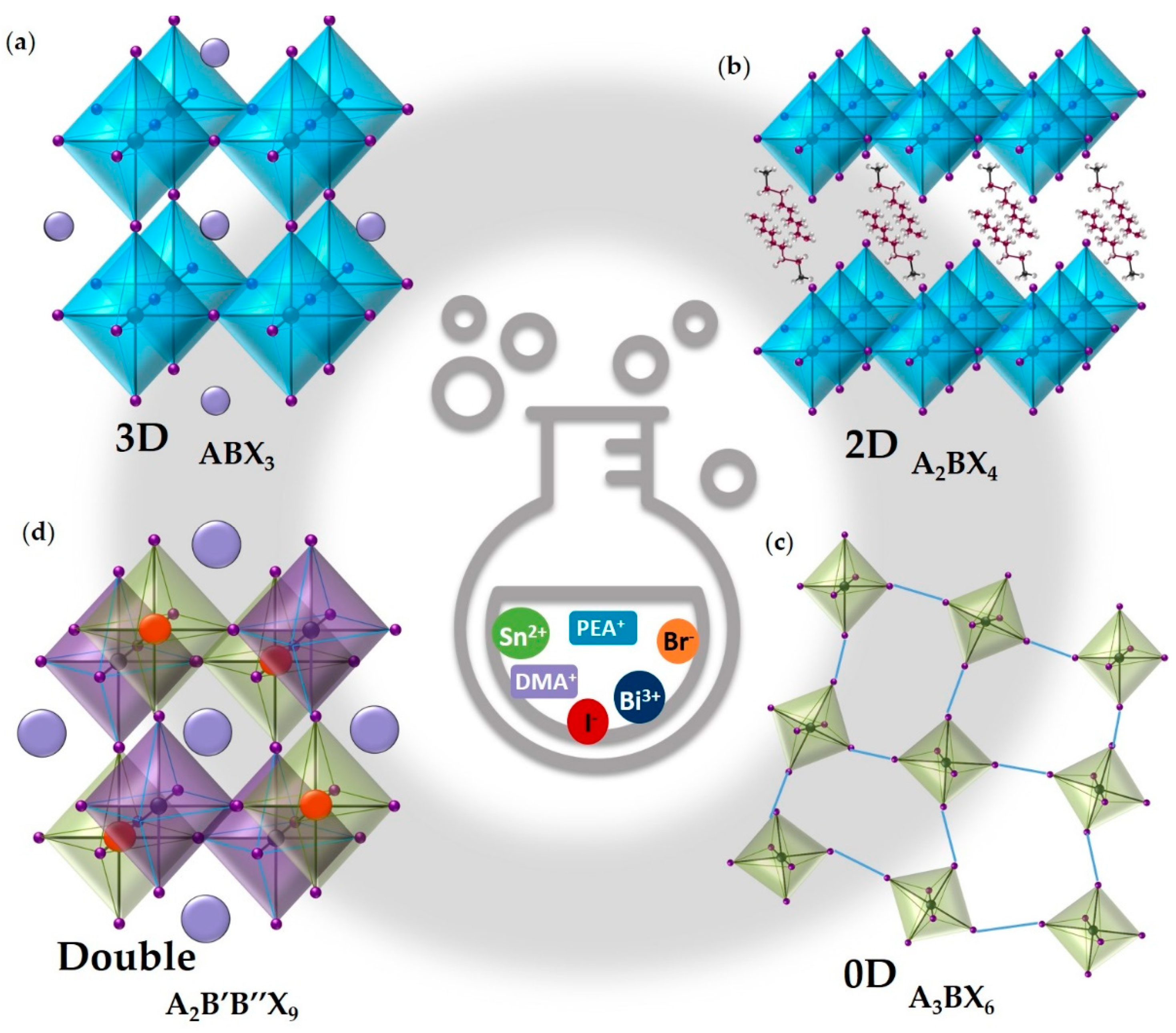
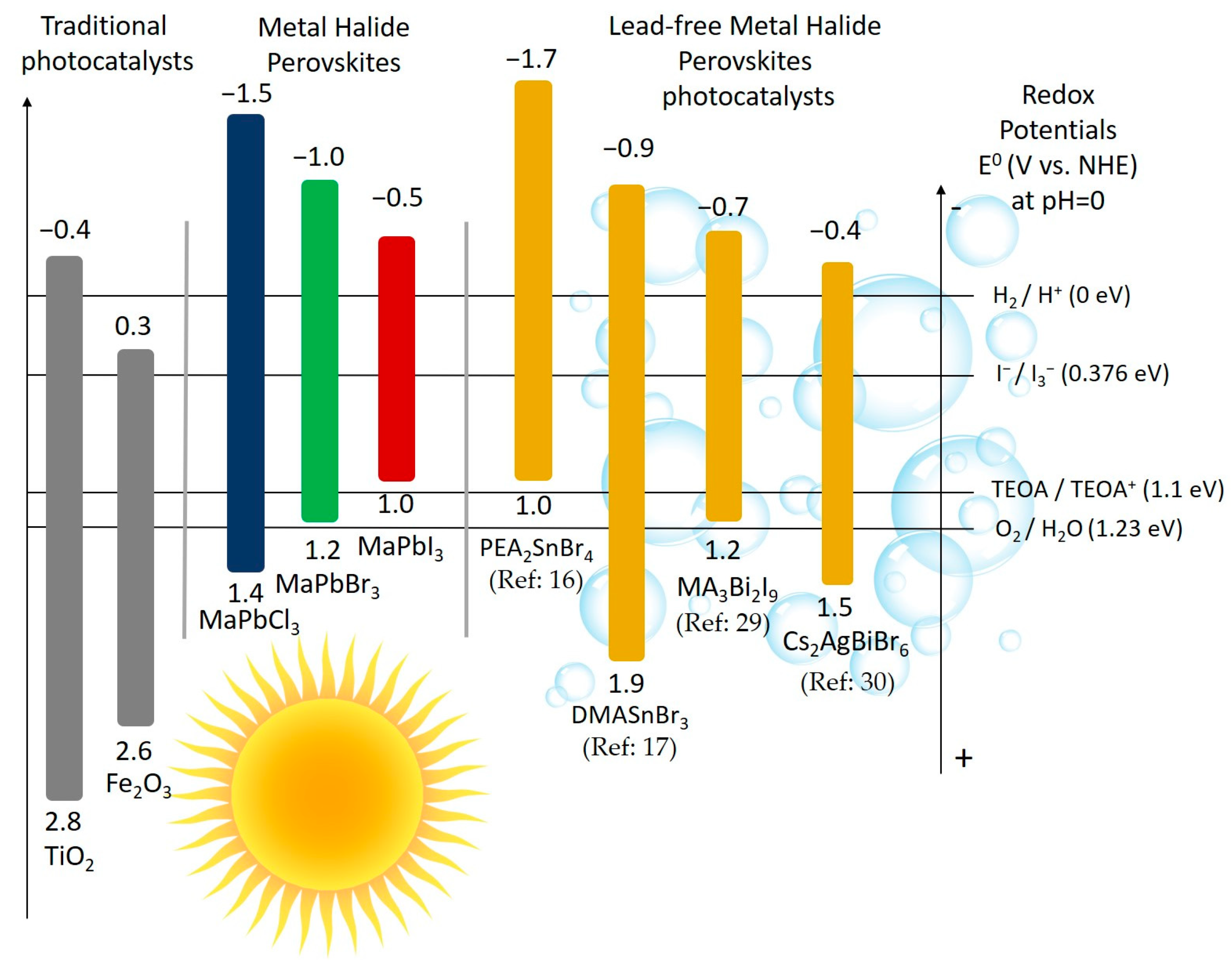
3. Pb-Free Perovskites for H2 Evolution
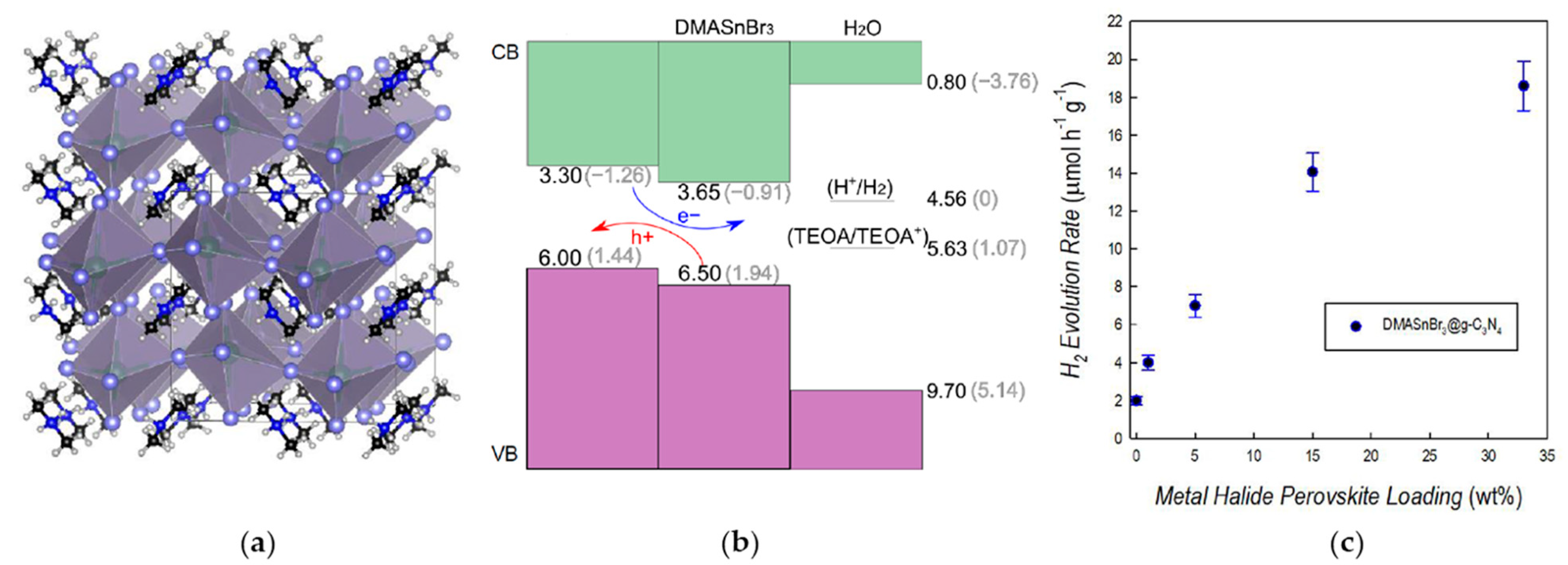
3.1. 3D Sn-Based MHPs
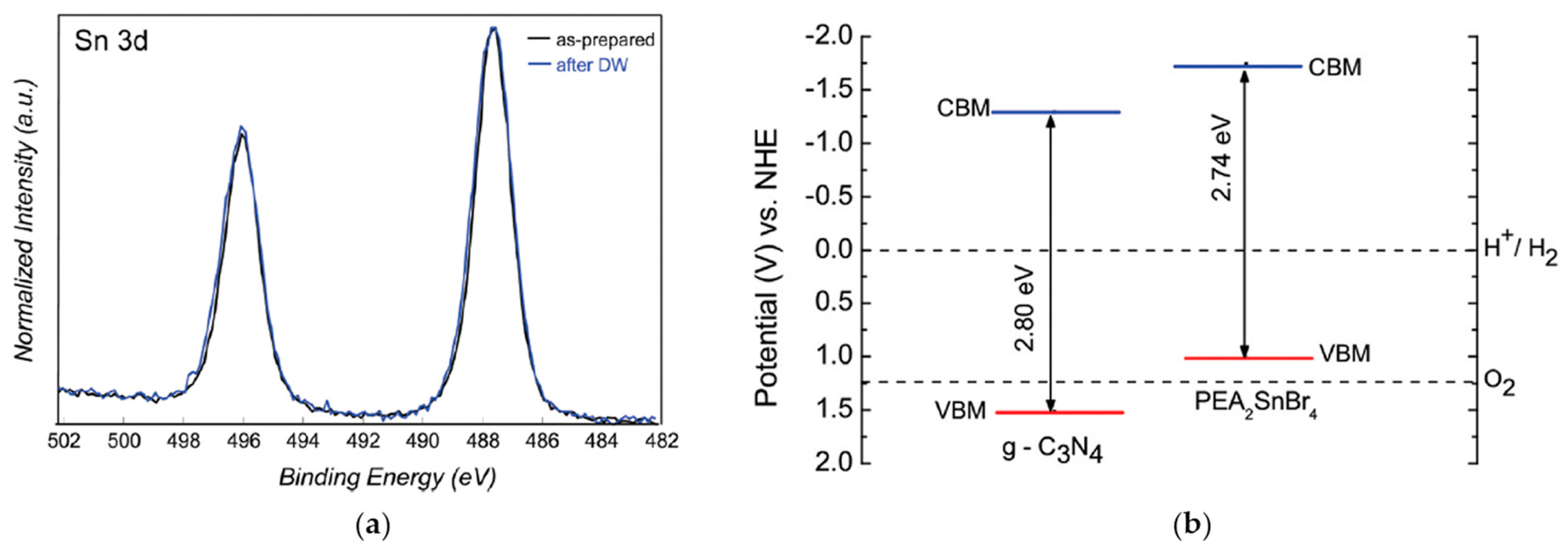
3.2. 2D Sn-Based MHPs
3.3. 0D Bi-Based and Sb-Based MHPs
3.4. Double MHPs
| Photocatalyst | Aqueous Media | Measurement Conditions | Activity (μmol g−1 h−1) | Ref. |
|---|---|---|---|---|
| DMASnBr3/Pt 10 wt% | DI water + 10% v/v TEOA | simulated solar light 500 Wm−2 for 5 h UV filter | 111 | [34] |
| g-C3N4/DMASnBr3 33 wt% | DI water + 10% v/v TEOA | simulated solar light 500 Wm−2 for 6 h UV filter | 19 | [17] |
| g-C3N4/DMASnBr3 33 wt%/Pt 3 wt% | DI water + 10% v/v TEOA | simulated solar light 500 Wm−2 for 6 h UV filter | 1730 | [17] |
| g-C3N4/DMASnBr3 33 wt%/Pt 3 wt% | DI water + glucose 0.1M | simulated solar light 500 Wm−2 for 6 h UV filter | 300 | [17] |
| DMASnI3 | DI water | 300 W Xe lamp for 5 h a 10 °C | 3 | [15] |
| PEA2SnBr4/Pt 3 wt% | DI water + 10% v/v TEOA | simulated solar light 500 Wm−2 for 6 h UV filter | 4 | [16] |
| g-C3N4/PEA2SnBr4 5 wt%/Pt 3 wt% | DI water + 10% v/v TEOA | simulated solar light 500 W m−2 for 6 h | 1613 | [16] |
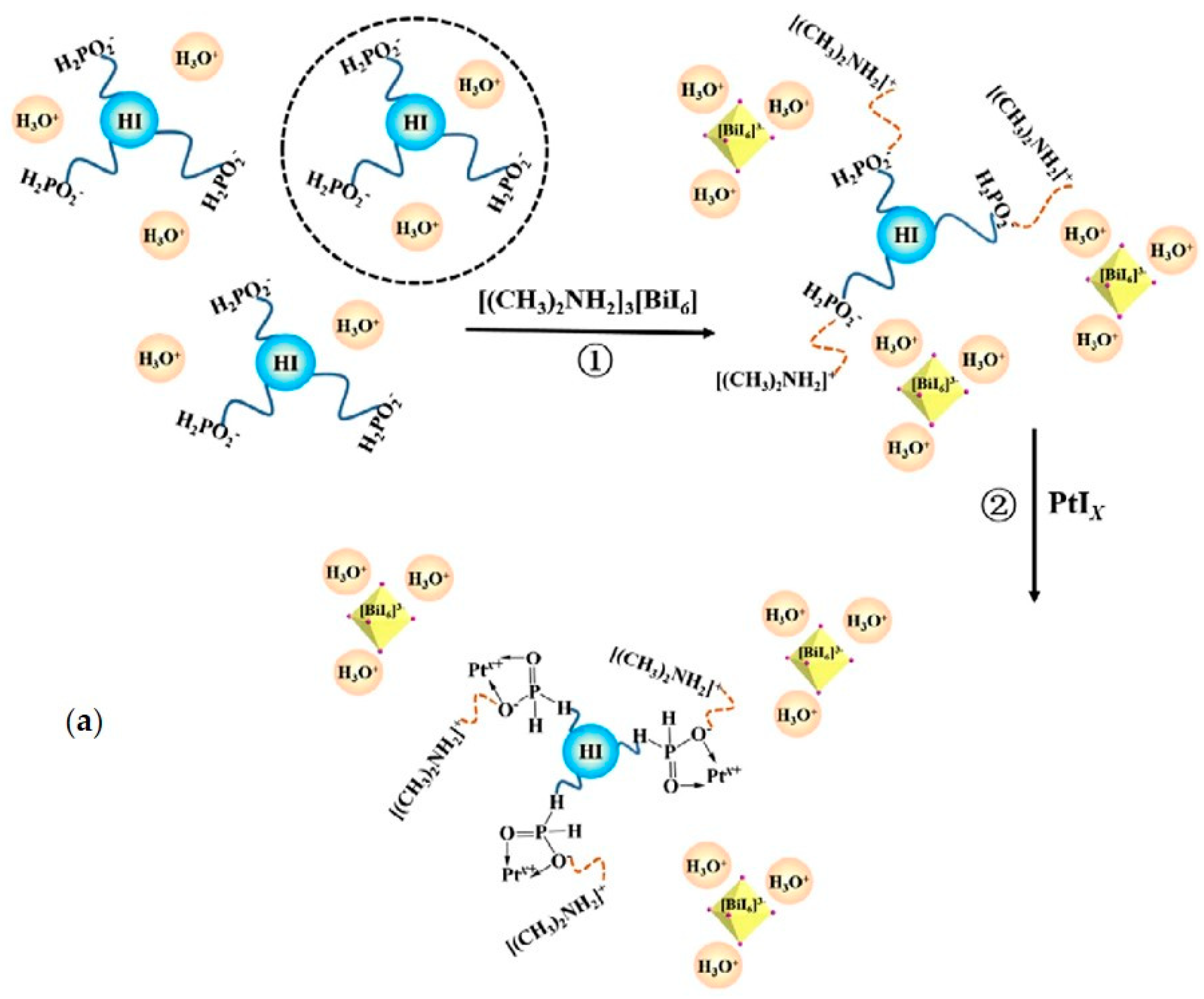
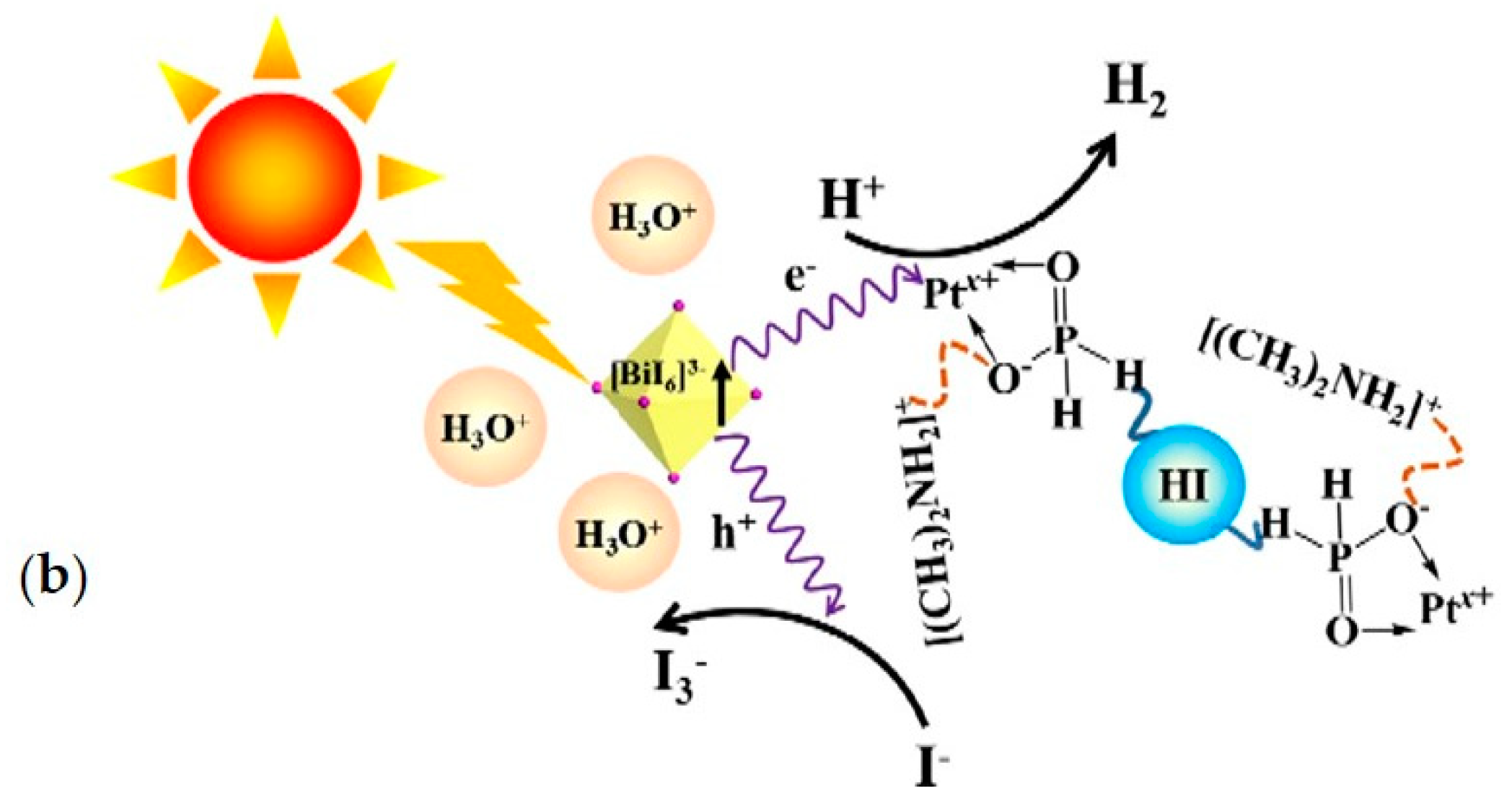
| DMA3BiI6 | 20 mg/mL in aqueous HI:H3PO2 1:4 | commercial LED for 6 h 425 nm, light intensity 8 mW | 3.5 | [31] |
| DMA3BiI6/Pb 1wt% | 20 mg/mL in aqueous HI:H3PO2 1:4 | commercial LED for 6 h 425 nm, light intensity 8 mW | 46 | [31] |
| DMA3BiI6 | aqueous saturated HI solution at 15 °C | 300 W Xe lamp (λ ≥ 420 nm) for 10 h | 25 | [78] |
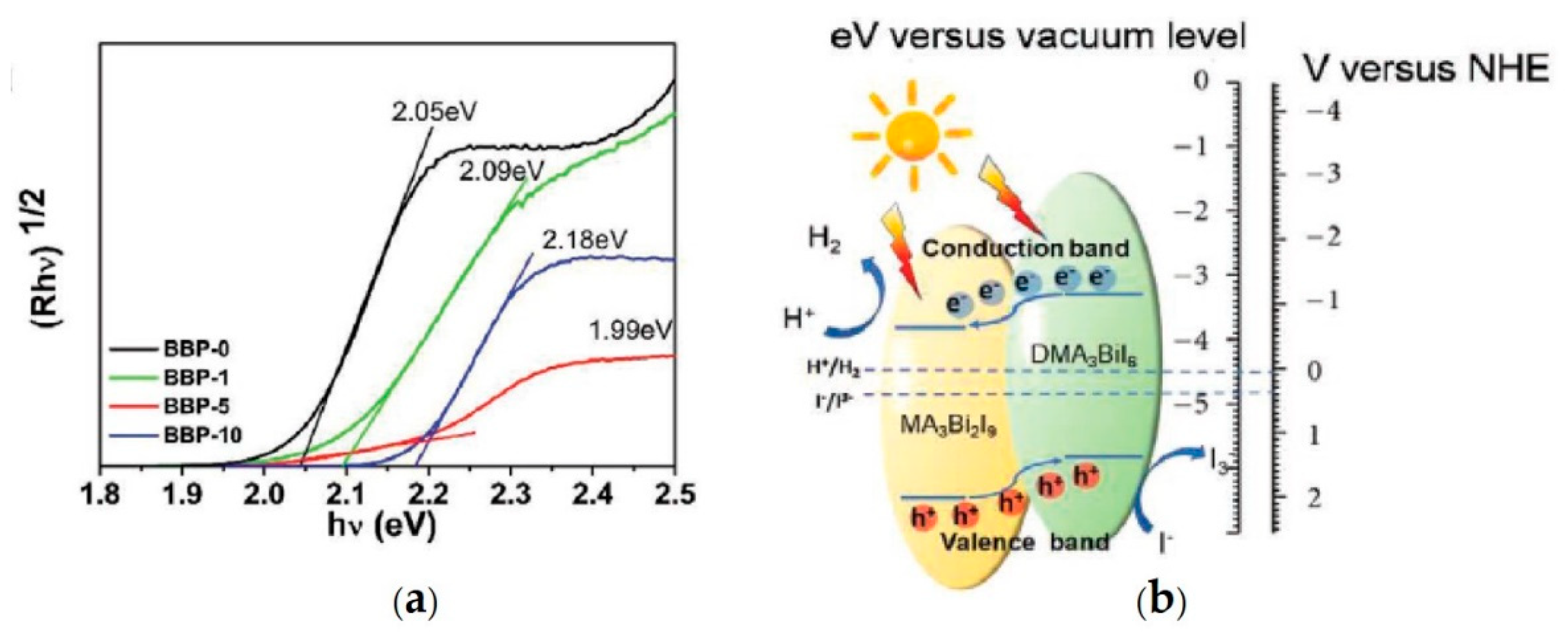
| DMA3BiI6/MA3Bi2I9 | aqueous saturated HI solution at 15 °C | 300 W Xe lamp (λ ≥ 420 nm) for 10h | 198 | [78] |
| MA3Bi2I9 | aqueous saturated HI solution + H3PO2 | 300 W Xe lamp (λ ≥ 420nm) for 10 h | 13 | [29] |
| MA3Bi2I9/Pt | aqueous saturated HI solution + H3PO2 | 300 W Xe lamp (λ ≥ 420nm) for 10 h | 169 | [29] |
| Cs3Bi2I9 | aqueous saturated HI + H3PO2 9:1 | 300 W Xe lamp for 10 h at 15 °C AM 1.5 cut-off filter | 2 | [42] |
| Cs3Bi2I9 | aqueous saturated HI + H3PO2 9:1 + Cs2CO3) | 300 W Xe lamp for 10 h at 15 °C AM 1.5 cut-off filter | 22 | [42] |
| Cs3Sb2I9 | aqueous saturated HI + H3PO2 9:1 + Cs2CO3 | 300 W Xe lamp for 10 h at 15 °C AM 1.5 cut-off filter | 60 | [42] |
| Cs3Bi0.6Sb1.4I9 | aqueous saturated HI + H3PO2 9:1 + Cs2CO3 | 300 W Xe lamp for 10h at 15 °C AM 1.5 cut-off filter | 157 | [42] |
| Cs3Bi0.6Sb1.4I9/Pt | aqueous saturated HI + H3PO2 9:1 + Cs2CO3 | 300W Xe lamp for 10 h at 15°C AM 1.5 cut-off filter | 926 | [42] |
| Cs2AgBiBr6 | aqueous saturated HBr solution | 300 W Xe lamp for 3 h (λ ≥ 420 nm) at 15 °C | 0.7 | [30] |
| Cs2AgBiBr6 | aqueous saturated HBr:H3PO2 5:1 solution | 300 W Xe lamp for 3 h (λ ≥ 420 nm) at 15 °C | 18 | [30] |
| Cs2AgBiBr6/Pt | aqueous saturated HBr:H3PO2 5:1 solution | 300 W Xe lamp for 3 h (λ ≥ 420 nm) at 15 °C | 22 | [30] |
| Cs2AgBiBr6/Pt | aqueous saturated HBr:H3PO2 5:1 solution | 300 W Xe lamp for 3 h (λ ≥ 420 nm) at 15 °C | 55 | [30] |
| Cs2AgBiBr6 | aqueous saturated HBr + H3PO2 solution 5:1 | 300 W Xe lamp for 10 h (λ ≥ 420 nm) at 5 °C | 0.6 | [32] |
| Cs2AgBiBr6/Pt | aqueous saturated HBr + H3PO2 solution 5:1 | 300 W Xe lamp for 10 h (λ ≥ 420 nm) at 5 °C | 1 | [32] |
| Cs2AgBiBr6/2.5 wt% RGO | aqueous saturated HBr + H3PO2 solution 5:1 | 300 W Xe lamp for 10 h (λ ≥ 420nm) at 5 °C | 49 | [32] |
4. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 0D | Zero-dimensional |
| 1D | One-dimensional |
| 2D | Two-dimensional |
| 3D | Three-dimensional |
| AM 1.5 G | Air Mass 1.5 Global |
| AQE | Apparent Quantum Efficiency |
| Bi | Bismuth |
| CB | Conduction Band |
| CBM | Conduction Band Minimum |
| DI | Deionized |
| DMA | Dimethylammonium |
| DMF | Dimethylformamide |
| Eg | Energy of Band Gap |
| EIS | Electrochemical Impedance Spectroscopy |
| FA | Formamidinium |
| g-C3N4 | Graphitic Carbon Nitride |
| GC-TCD | Thermal Conductivity Detection |
| H2 | Hydrogen |
| IPA | Isopropanol |
| Jsc | Photocurrent Density |
| MA | Methylammonium |
| MHP | Metal Halide Perovskite |
| NHE | Normal Hydrogen Electrode |
| O2 | Oxygen |
| Pb | Lead |
| PEC | Photoelectrochemical |
| PL | Photoluminescence |
| PV | Photovoltaic |
| RGO | Reduced Graphene Oxide |
| Sb | Antimony |
| Sn | Tin |
| TBAPF6 | Tetrabutylammonium Hexafluorophosphate |
| TEOA | Triethanolamine |
| UPS | Ultraviolet Photoelectron Spectroscopy |
| VB | Valence Band |
| VBM | Valence Band Minimum |
| XPS | X-ray Photoelectron Spectroscopy |
| XRD | X-ray Diffraction |
| ηSTH | Conversion Efficiency |
References
- Listorti, A.; Durrant, J.; Barber, J. Artificial photosynthesis: Solar to fuel. Nat. Mater. 2009, 8, 929–930. [Google Scholar] [CrossRef] [PubMed]
- IEA. Key World Energy Statistics 2020; International Energy Agency: Paris, France, 2020; Volume 33, p. 4649. [Google Scholar]
- Abdin, Z.; Zafaranloo, A.; Rafiee, A.; Mérida, W.; Lipiński, W.; Khalilpour, K.R. Hydrogen as an energy vector. Renew. Sustain. Energy Rev. 2020, 120. [Google Scholar] [CrossRef]
- Armaroli, N.; Balzani, V. The hydrogen issue. ChemSusChem 2011, 4, 21–36. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Qi, Z.; Zhang, S.; Su, J.; Somorjai, G.A. Catalytic hydrogen production from methane: A review on recent progress and prospect. Catalysts 2020, 10, 858. [Google Scholar] [CrossRef]
- Zhai, P.; Haussener, S.; Ager, J.; Sathre, R.; Walczak, K.; Greenblatt, J.; McKone, T. Net primary energy balance of a solar-driven photoelectrochemical water-splitting device. Energy Environ. Sci. 2013, 6, 2380–2389. [Google Scholar] [CrossRef] [Green Version]
- Bosso, P.; Milella, A.; Barucca, G.; Mengucci, P.; Armenise, V.; Fanelli, F.; Giannuzzi, R.; Maiorano, V.; Fracassi, F. Plasma-assisted deposition of iron oxide thin films for photoelectrochemical water splitting. Plasma Process. Polym. 2020, 1–12. [Google Scholar] [CrossRef]
- A decade of perovskite photovoltaics. Nat. Energy 2019, 4, 20181221. [CrossRef] [Green Version]
- Huang, H.; Pradhan, B.; Hofkens, J.; Roeffaers, M.B.J.; Steele, J.A. Solar-Driven Metal Halide Perovskite Photocatalysis: Design, Stability, and Performance. ACS Energy Lett. 2020, 5, 1107–1123. [Google Scholar] [CrossRef]
- Crespo-Quesada, M.; Pazos-Outón, L.M.; Warnan, J.; Kuehnel, M.F.; Friend, R.H.; Reisner, E. Metal-encapsulated organolead halide perovskite photocathode for solar-driven hydrogen evolution in water. Nat. Commun. 2016, 7, 6–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.; Chang, W.J.; Lee, C.W.; Park, S.; Ahn, H.Y.; Nam, K.T. Photocatalytic hydrogen generation from hydriodic acid using methylammonium lead iodide in dynamic equilibrium with aqueous solution. Nat. Energy 2017, 2, 1–8. [Google Scholar] [CrossRef]
- Han, C.; Zhu, X.; Martin, J.S.; Lin, Y.; Spears, S.; Yan, Y. Recent Progress in Engineering Metal Halide Perovskites for Efficient Visible-Light-Driven Photocatalysis. ChemSusChem 2020, 13, 4005–4025. [Google Scholar] [CrossRef]
- Huynh, K.A.; Nguyen, D.L.T.; Nguyen, V.H.; Vo, D.V.N.; Trinh, Q.T.; Nguyen, T.P.; Kim, S.Y.; Le, Q. Van Halide perovskite photocatalysis: Progress and perspectives. J. Chem. Technol. Biotechnol. 2020, 95, 2579–2596. [Google Scholar] [CrossRef]
- Romani, L.; Malavasi, L. Solar-Driven Hydrogen Generation by Metal Halide Perovskites: Materials, Approaches, and Mechanistic View. ACS Omega 2020, 5, 25511–25519. [Google Scholar] [CrossRef] [PubMed]
- Ju, D.; Zheng, X.; Liu, J.; Chen, Y.; Zhang, J.; Cao, B.; Xiao, H.; Mohammed, O.F.; Bakr, O.M.; Tao, X. Reversible Band Gap Narrowing of Sn-Based Hybrid Perovskite Single Crystal with Excellent Phase Stability. Angew. Chem. Int. Ed. 2018, 57, 14868–14872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romani, L.; Bala, A.; Kumar, V.; Speltini, A.; Milella, A.; Fracassi, F.; Listorti, A.; Profumo, A.; Malavasi, L. PEA2SnBr4: A water-stable lead-free two-dimensional perovskite and demonstration of its use as a co-catalyst in hydrogen photogeneration and organic-dye degradation. J. Mater. Chem. C 2020, 8, 9189–9194. [Google Scholar] [CrossRef]
- Romani, L.; Speltini, A.; Ambrosio, F.; Mosconi, E.; Profumo, A.; Marelli, M.; Margadonna, S.; Milella, A.; Fracassi, F.; Listorti, A.; et al. Water-Stable DMASnBr3 Lead-Free Perovskite for Effective Solar-Driven Photocatalysis. Angew. Chem. Int. Ed. 2020, 2–10. [Google Scholar] [CrossRef]
- Yin, W.J.; Shi, T.; Yan, Y. Unique properties of halide perovskites as possible origins of the superior solar cell performance. Adv. Mater. 2014, 26, 4653–4658. [Google Scholar] [CrossRef]
- Eperon, G.E.; Paternò, G.M.; Sutton, R.J.; Zampetti, A.; Haghighirad, A.A.; Cacialli, F.; Snaith, H.J. Inorganic caesium lead iodide perovskite solar cells. J. Mater. Chem. A 2015, 3, 19688–19695. [Google Scholar] [CrossRef]
- Premkumar, M.P.; Thiruvengadaravi, K.V.; Senthil Kumar, P.; Nandagopal, J.; Sivanesan, S. Eco-Friendly Treatment Strategies for Wastewater Containing Dyes and Heavy Metals; Springer: Berlin/Heidelberg, Germany, 2018; ISBN 9789811073328. [Google Scholar]
- Hubicki, Z.; Koodynsk, D. Selective Removal of Heavy Metal Ions from Waters and Waste Waters Using Ion Exchange Methods. Ion Exch. Technol. 2012. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Duan, J.; Yang, X.; Duan, Y.; Yang, P.; Tang, Q. Review on recent progress of lead-free halide perovskites in optoelectronic applications. Nano Energy 2021, 80, 105526. [Google Scholar] [CrossRef]
- Li, J.; Cao, H.L.; Jiao, W.B.; Wang, Q.; Wei, M.; Cantone, I.; Lü, J.; Abate, A. Biological impact of lead from halide perovskites reveals the risk of introducing a safe threshold. Nat. Commun. 2020, 11, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Wen, J.; Xie, J.; Chen, X.; Li, X. A review on g-C 3 N 4-based photocatalysts. Appl. Surf. Sci. 2017, 391, 72–123. [Google Scholar] [CrossRef]
- Kondarides, D.I.; Daskalaki, V.M.; Patsoura, A.; Verykios, X.E. Hydrogen production by photo-induced reforming of biomass components and derivatives at ambient conditions. Catal. Lett. 2008, 122, 26–32. [Google Scholar] [CrossRef]
- Roger, I.; Shipman, M.A.; Symes, M.D. Earth-abundant catalysts for electrochemical and photoelectrochemical water splitting. Nat. Rev. Chem. 2017, 1. [Google Scholar] [CrossRef]
- Colón, G. Towards the hydrogen production by photocatalysis. Appl. Catal. A Gen. 2016, 518, 48–59. [Google Scholar] [CrossRef]
- Hisatomi, T.; Kubota, J.; Domen, K. Recent advances in semiconductors for photocatalytic and photoelectrochemical water splitting. Chem. Soc. Rev. 2014, 43, 7520–7535. [Google Scholar] [CrossRef]
- Guo, Y.; Liu, G.; Li, Z.; Lou, Y.; Chen, J.; Zhao, Y. Stable Lead-Free (CH3NH3)3Bi2I9 Perovskite for Photocatalytic Hydrogen Generation. ACS Sustain. Chem. Eng. 2019, 7, 15080–15085. [Google Scholar] [CrossRef]
- Shi, M.; Li, G.; Tian, W.; Jin, S.; Tao, X.; Jiang, Y.; Pidko, E.A.; Li, R.; Li, C. Understanding the Effect of Crystalline Structural Transformation for Lead-Free Inorganic Halide Perovskites. Adv. Mater. 2020, 32, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Li, Y.; Zhang, B.; Xu, T.; Wang, C. PtIx/[(CH3)2NH2]3[BiI6] as a well-dispersed photocatalyst for hydrogen production in hydroiodic acid. Nano Energy 2018, 50, 665–674. [Google Scholar] [CrossRef]
- Wang, T.; Yue, D.; Li, X.; Zhao, Y. Lead-free double perovskite Cs2AgBiBr6/RGO composite for efficient visible light photocatalytic H2 evolution. Appl. Catal. B Environ. 2020, 268, 118399. [Google Scholar] [CrossRef]
- Guan, Z.; Wu, Y.; Wang, P.; Zhang, Q.; Wang, Z.; Zheng, Z.; Liu, Y.; Dai, Y.; Whangbo, M.H.; Huang, B. Perovskite photocatalyst CsPbBr3-xIx with a bandgap funnel structure for H2 evolution under visible light. Appl. Catal. B Environ. 2019, 245, 522–527. [Google Scholar] [CrossRef]
- Pisanu, A.; Speltini, A.; Quadrelli, P.; Drera, G.; Sangaletti, L.; Malavasi, L. Enhanced air-stability of Sn-based hybrid perovskites induced by dimethylammonium (DMA): Synthesis, characterization, aging and hydrogen photogeneration of the MA1-xDMAxSnBr3 system. J. Mater. Chem. C 2019, 7, 7020–7026. [Google Scholar] [CrossRef]
- Glanz, K. Using behavioral theories to guide decisions of what to measure, and why. Int. J. Hydrogen Energy 2010, 27, 991–1022. [Google Scholar]
- Walter, M.G.; Warren, E.L.; McKone, J.R.; Boettcher, S.W.; Mi, Q.; Santori, E.A.; Lewis, N.S. Solar water splitting cells. Chem. Rev. 2010, 110, 6446–6473. [Google Scholar] [CrossRef] [PubMed]
- Döscher, H.; Geisz, J.F.; Deutsch, T.G.; Turner, J.A. Sunlight absorption in water-efficiency and design implications for photoelectrochemical devices. Energy Environ. Sci. 2014, 7, 2951–2956. [Google Scholar] [CrossRef]
- Chen, Z.; Jaramillo, T.F.; Deutsch, T.G.; Kleiman-Shwarsctein, A.; Forman, A.J.; Gaillard, N.; Garland, R.; Takanabe, K.; Heske, C.; Sunkara, M.; et al. Accelerating materials development for photoelectrochemical hydrogen production: Standards for methods, definitions, and reporting protocols. J. Mater. Res. 2010, 25, 3–16. [Google Scholar] [CrossRef]
- Varghese, O.K.; Grimes, C.A. Appropriate strategies for determining the photoconversion efficiency of water photoelectrolysis cells: A review with examples using titania nanotube array photoanodes. Sol. Energy Mater. Sol. Cells 2008, 92, 374–384. [Google Scholar] [CrossRef]
- Grimes, C.A.; Varghese, O.K.; Ranjan, S. From Hydrocarbons to Hydrogen: Towards a Sustainable Future; Springer: Berlin/Heidelberg, Germany, 2007; ISBN 9780387331980. [Google Scholar]
- Dotan, H.; Mathews, N.; Hisatomi, T.; Grätzel, M.; Rothschild, A. On the solar to hydrogen conversion efficiency of photoelectrodes for water splitting. J. Phys. Chem. Lett. 2014, 5, 3330–3334. [Google Scholar] [CrossRef]
- Chen, G.; Wang, P.; Wu, Y.; Zhang, Q.; Wu, Q.; Wang, Z.; Zheng, Z.; Liu, Y.; Dai, Y.; Huang, B. Lead-Free Halide Perovskite Cs3Bi2xSb2–2xI9 (x ≈ 0.3) Possessing the Photocatalytic Activity for Hydrogen Evolution Comparable to that of (CH3NH3)PbI3. Adv. Mater. 2020, 32, 1–7. [Google Scholar] [CrossRef]
- Stranks, S.D.; Snaith, H.J. Metal-halide perovskites for photovoltaic and light-emitting devices. Nat. Nanotechnol. 2015, 10, 391–402. [Google Scholar] [CrossRef]
- Saparov, B.; Mitzi, D.B. Organic-Inorganic Perovskites: Structural Versatility for Functional Materials Design. Chem. Rev. 2016, 116, 4558–4596. [Google Scholar] [CrossRef]
- Mitzi, D.B. Templating and structural engineering in organic-inorganic perovskites. J. Chem. Soc. Dalt. Trans. 2001, 1–12. [Google Scholar] [CrossRef]
- Bresolin, B.M.; Park, Y.; Bahnemann, D.W. Recent progresses on metal halide perovskite-based material as potential photocatalyst. Catalysts 2020, 10, 709. [Google Scholar] [CrossRef]
- Chen, Q.; De Marco, N.; Yang, Y.; Song, T.B.; Chen, C.C.; Zhao, H.; Hong, Z.; Zhou, H.; Yang, Y. Under the spotlight: The organic-inorganic hybrid halide perovskite for optoelectronic applications. Nano Today 2015, 10, 355–396. [Google Scholar] [CrossRef] [Green Version]
- Bernasconi, A.; Rizzo, A.; Listorti, A.; Mahata, A.; Mosconi, E.; De Angelis, F.; Malavasi, L. Synthesis, Properties, and Modeling of Cs1-xRbxSnBr3 Solid Solution: A New Mixed-Cation Lead-Free All-Inorganic Perovskite System. Chem. Mater. 2019, 31, 3527–3533. [Google Scholar] [CrossRef]
- Travis, W.; Glover, E.N.K.; Bronstein, H.; Scanlon, D.O.; Palgrave, R.G. On the application of the tolerance factor to inorganic and hybrid halide perovskites: A revised system. Chem. Sci. 2016, 7, 4548–4556. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartel, C.J.; Sutton, C.; Goldsmith, B.R.; Ouyang, R.; Musgrave, C.B.; Ghiringhelli, L.M.; Scheffler, M. New tolerance factor to predict the stability of perovskite oxides and halides. Sci. Adv. 2019, 5, eaav0693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klejna, S.; Mazur, T.; Wlaźlak, E.; Zawal, P.; Soo, H.S.; Szaciłowski, K. Halogen-containing semiconductors: From artificial photosynthesis to unconventional computing. Coord. Chem. Rev. 2020, 415. [Google Scholar] [CrossRef]
- Radicchi, E.; Ambrosio, F.; Mosconi, E.; Alasmari, A.A.; Alasmary, F.A.S.; De Angelis, F. Combined Computational and Experimental Investigation on the Nature of Hydrated Iodoplumbate Complexes: Insights into the Dual Role of Water in Perovskite Precursor Solutions. J. Phys. Chem. B 2020. [Google Scholar] [CrossRef] [PubMed]
- Grancini, G.; Nazeeruddin, M.K. Dimensional tailoring of hybrid perovskites for photovoltaics. Nat. Rev. Mater. 2019, 4, 4–22. [Google Scholar] [CrossRef]
- Wang, A.; Guo, Y.; Zhou, Z.; Niu, X.; Wang, Y.; Muhammad, F.; Li, H.; Zhang, T.; Wang, J.; Nie, S.; et al. Aqueous acid-based synthesis of lead-free tin halide perovskites with near-unity photoluminescence quantum efficiency. Chem. Sci. 2019, 10, 4573–4579. [Google Scholar] [CrossRef] [Green Version]
- Almutlaq, J.; Yin, J.; Mohammed, O.F.; Bakr, O.M. The Benefit and Challenges of Zero-Dimensional Perovskites. J. Phys. Chem. Lett. 2018, 9, 4131–4138. [Google Scholar] [CrossRef]
- Akkerman, Q.A.; Abdelhady, A.L.; Manna, L. Zero-Dimensional Cesium Lead Halides: History, Properties, and Challenges. J. Phys. Chem. Lett. 2018, 9, 2326–2337. [Google Scholar] [CrossRef] [Green Version]
- Nie, R.; Sumukam, R.R.; Reddy, S.H.; Banavoth, M.; Seok, S. Il Lead-free perovskite solar cells enabled by hetero-valent substitutes. Energy Environ. Sci. 2020, 13, 2363–2385. [Google Scholar] [CrossRef]
- Hu, H.; Dong, B.; Zhang, W. Low-toxic metal halide perovskites: Opportunities and future challenges. J. Mater. Chem. A 2017, 5, 11436–11449. [Google Scholar] [CrossRef]
- Ke, W.; Kanatzidis, M.G. Prospects for low-toxicity lead-free perovskite solar cells. Nat. Commun. 2019, 10, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Igbari, F.; Wang, Z.K.; Liao, L.S. Progress of Lead-Free Halide Double Perovskites. Adv. Energy Mater. 2019, 9, 1–32. [Google Scholar] [CrossRef]
- Meyer, E.; Mutukwa, D.; Zingwe, N.; Taziwa, R. Lead-free halide double perovskites: A review of the structural, optical, and stability properties as well as their viability to replace lead halide perovskites. Metals (Basel) 2018, 8, 667. [Google Scholar] [CrossRef] [Green Version]
- Dave, K.; Fang, M.H.; Bao, Z.; Fu, H.T.; Liu, R.S. Recent Developments in Lead-Free Double Perovskites: Structure, Doping, and Applications. Chem. Asian J. 2020, 15, 242–252. [Google Scholar] [CrossRef]
- Tolod, K.R.; Hernández, S.; Russo, N. Recent advances in the BiVO4 photocatalyst for sun-driven water oxidation: Top-performing photoanodes and scale-up challenges. Catalysts 2017, 7, 13. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Hu, H.; Kang, W.; Qiu, G.; Liang, R.; Deng, L.; Yuan, H. Enhancing hydrogen evolution by photoelectrocatalysis of water splitting over a CdS flowers-loaded TiO2 nanotube array film on the Ti foil substrate. Ceram. Int. 2020, 46, 17606–17613. [Google Scholar] [CrossRef]
- Zhang, S.; Chen, S.; Liu, D.; Zhang, J.; Peng, T. Layered WS2/WO3 Z-scheme photocatalyst constructed via an in situ sulfurization of hydrous WO3 nanoplates for efficient H2 generation. Appl. Surf. Sci. 2020, 529, 147013. [Google Scholar] [CrossRef]
- Fominski, V.; Romanov, R.; Fominski, D.; Soloviev, A.; Rubinkovskaya, O.; Demin, M.; Maksimova, K.; Shvets, P.; Goikhman, A. Performance and mechanism of photoelectrocatalytic activity of mosx/wo3 heterostructures obtained by reactive pulsed laser deposition for water splitting. Nanomaterials 2020, 10, 871. [Google Scholar] [CrossRef]
- Nørskov, J.K.; Bligaard, T.; Logadottir, A.; Kitchin, J.R.; Chen, J.G.; Pandelov, S.; Stimming, U. Trends in the Exchange Current for Hydrogen Evolution. J. Electrochem. Soc. 2005, 152, J23. [Google Scholar] [CrossRef] [Green Version]
- Cao, S.; Piao, L.; Chen, X. Emerging Photocatalysts for Hydrogen Evolution. Trends Chem. 2020, 2, 57–70. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, C.; Sun, Y.; Chen, Q.; He, L.; Zhang, K.; Zhang, J.; Liu, B.; Chen, L.F. Design of metal-organic framework-based photocatalysts for hydrogen generation. Coord. Chem. Rev. 2020, 413, 213266. [Google Scholar] [CrossRef]
- Ye, S.; Wang, R.; Wu, M.Z.; Yuan, Y.P. A review on g-C 3 N 4 for photocatalytic water splitting and CO 2 reduction. Appl. Surf. Sci. 2015, 358, 15–27. [Google Scholar] [CrossRef]
- Bonomi, S.; Armenise, V.; Accorsi, G.; Colella, S.; Rizzo, A.; Fracassi, F.; Malavasi, L.; Listorti, A. The Effect of Extended Ball-Milling upon Three-Dimensional and Two-Dimensional Perovskite Crystals Properties. Appl. Sci. 2020, 10, 4775. [Google Scholar] [CrossRef]
- Slavney, A.H.; Leppert, L.; Bartesaghi, D.; Gold-Parker, A.; Toney, M.F.; Savenije, T.J.; Neaton, J.B.; Karunadasa, H.I. Defect-Induced Band-Edge Reconstruction of a Bismuth-Halide Double Perovskite for Visible-Light Absorption. J. Am. Chem. Soc. 2017, 139, 5015–5018. [Google Scholar] [CrossRef] [Green Version]
- Mitzi, D.B.; Feild, C.A.; Harrison, W.T.A.; Guloy, A.M. Conducting tin halides with a layered organic-based perovskite structure. Nature 1994, 369, 467–469. [Google Scholar] [CrossRef]
- Mao, L.; Stoumpos, C.C.; Kanatzidis, M.G. Two-Dimensional Hybrid Halide Perovskites: Principles and Promises. J. Am. Chem. Soc. 2019, 141, 1171–1190. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2photocatalysis: Mechanisms and materials. Chem. Rev. 2014, 114, 9919–9986. [Google Scholar] [CrossRef] [PubMed]
- Lyu, M.; Yun, J.H.; Cai, M.; Jiao, Y.; Bernhardt, P.V.; Zhang, M.; Wang, Q.; Du, A.; Wang, H.; Liu, G.; et al. Organic–inorganic bismuth (III)-based material: A lead-free, air-stable and solution-processable light-absorber beyond organolead perovskites. Nano Res. 2016, 9, 692–702. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Chen, Y.; Su, J.; Shi, J.; Wang, X.; Guo, L. Photocatalytic hydrogen production using twinned nanocrystals and an unanchored NiSxco-catalyst. Nat. Energy 2016, 1, 1–8. [Google Scholar] [CrossRef]
- Tang, Y.; Mak, C.H.; Liu, R.; Wang, Z.; Ji, L.; Song, H.; Tan, C.; Barrière, F.; Hsu, H.Y. In Situ Formation of Bismuth-Based Perovskite Heterostructures for High-Performance Cocatalyst-Free Photocatalytic Hydrogen Evolution. Adv. Funct. Mater. 2020, 30, 2006919. [Google Scholar] [CrossRef]
- Umari, P.; Mosconi, E.; De Angelis, F. Relativistic GW calculations on CH3 NH3 PbI 3 and CH3 NH3 SnI3 Perovskites for Solar Cell Applications. Sci. Rep. 2014, 4, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Bratsch, S.G. Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K. J. Phys. Chem. Ref. Data 1989, 18, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Steele, J.A.; Puech, P.; Keshavarz, M.; Yang, R.; Banerjee, S.; Debroye, E.; Kim, C.W.; Yuan, H.; Heo, N.H.; Vanacken, J.; et al. Giant electron-phonon coupling and deep conduction band resonance in metal halide double perovskite. ACS Nano 2018, 12, 8081–8090. [Google Scholar] [CrossRef]
- Hoye, R.L.Z.; Eyre, L.; Wei, F.; Brivio, F.; Sadhanala, A.; Sun, S.; Li, W.; Zhang, K.H.L.; MacManus-Driscoll, J.L.; Bristowe, P.D.; et al. Fundamental Carrier Lifetime Exceeding 1 µs in Cs2AgBiBr6 Double Perovskite. Adv. Mater. Interfaces 2018, 5, 2–9. [Google Scholar] [CrossRef] [Green Version]
- Ou, M.; Tu, W.; Yin, S.; Xing, W.; Wu, S.; Wang, H.; Wan, S.; Zhong, Q.; Xu, R. Amino-Assisted Anchoring of CsPbBr 3 Perovskite Quantum Dots on Porous g-C 3 N 4 for Enhanced Photocatalytic CO 2 Reduction. Angew. Chem. 2018, 130, 13758–13762. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, P.; Zhu, X.; Zhang, Q.; Wang, Z.; Liu, Y.; Zou, G.; Dai, Y.; Whangbo, M.H.; Huang, B. Composite of CH3NH3PbI3 with Reduced Graphene Oxide as a Highly Efficient and Stable Visible-Light Photocatalyst for Hydrogen Evolution in Aqueous HI Solution. Adv. Mater. 2018, 30, 2–7. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Armenise, V.; Colella, S.; Fracassi, F.; Listorti, A. Lead-Free Metal Halide Perovskites for Hydrogen Evolution from Aqueous Solutions. Nanomaterials 2021, 11, 433. https://doi.org/10.3390/nano11020433
Armenise V, Colella S, Fracassi F, Listorti A. Lead-Free Metal Halide Perovskites for Hydrogen Evolution from Aqueous Solutions. Nanomaterials. 2021; 11(2):433. https://doi.org/10.3390/nano11020433
Chicago/Turabian StyleArmenise, Vincenza, Silvia Colella, Francesco Fracassi, and Andrea Listorti. 2021. "Lead-Free Metal Halide Perovskites for Hydrogen Evolution from Aqueous Solutions" Nanomaterials 11, no. 2: 433. https://doi.org/10.3390/nano11020433








