Improved Photostability in Fluorinated 2D Perovskite Single Crystals
Abstract
1. Introduction
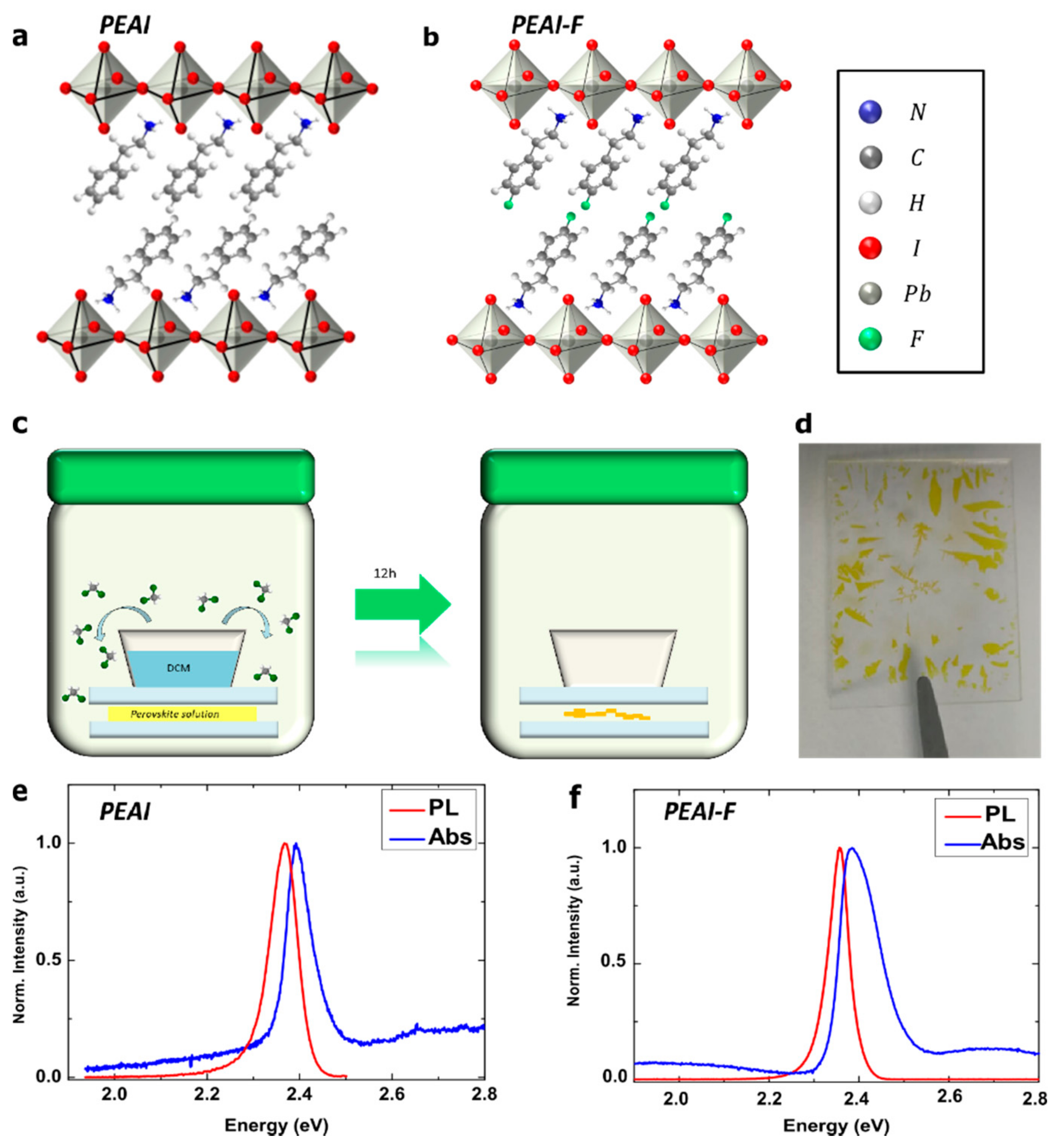
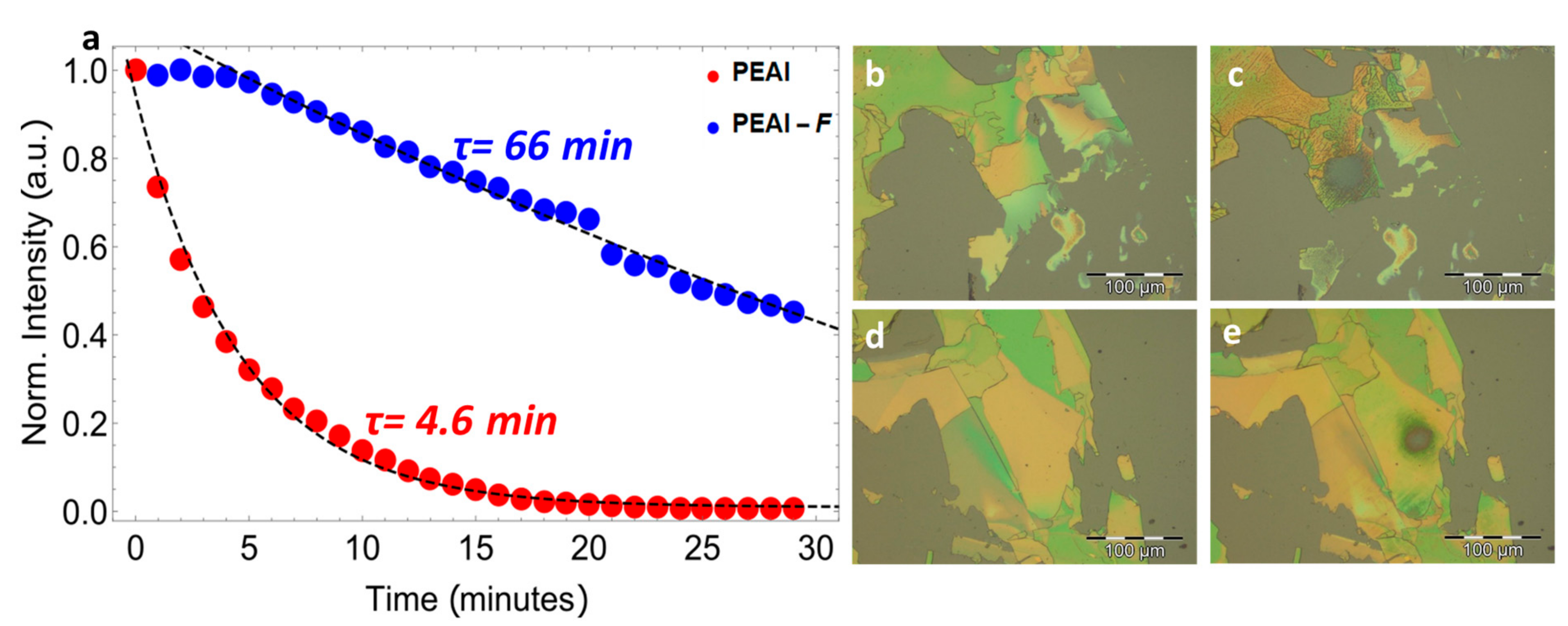
2. Results
3. Conclusions
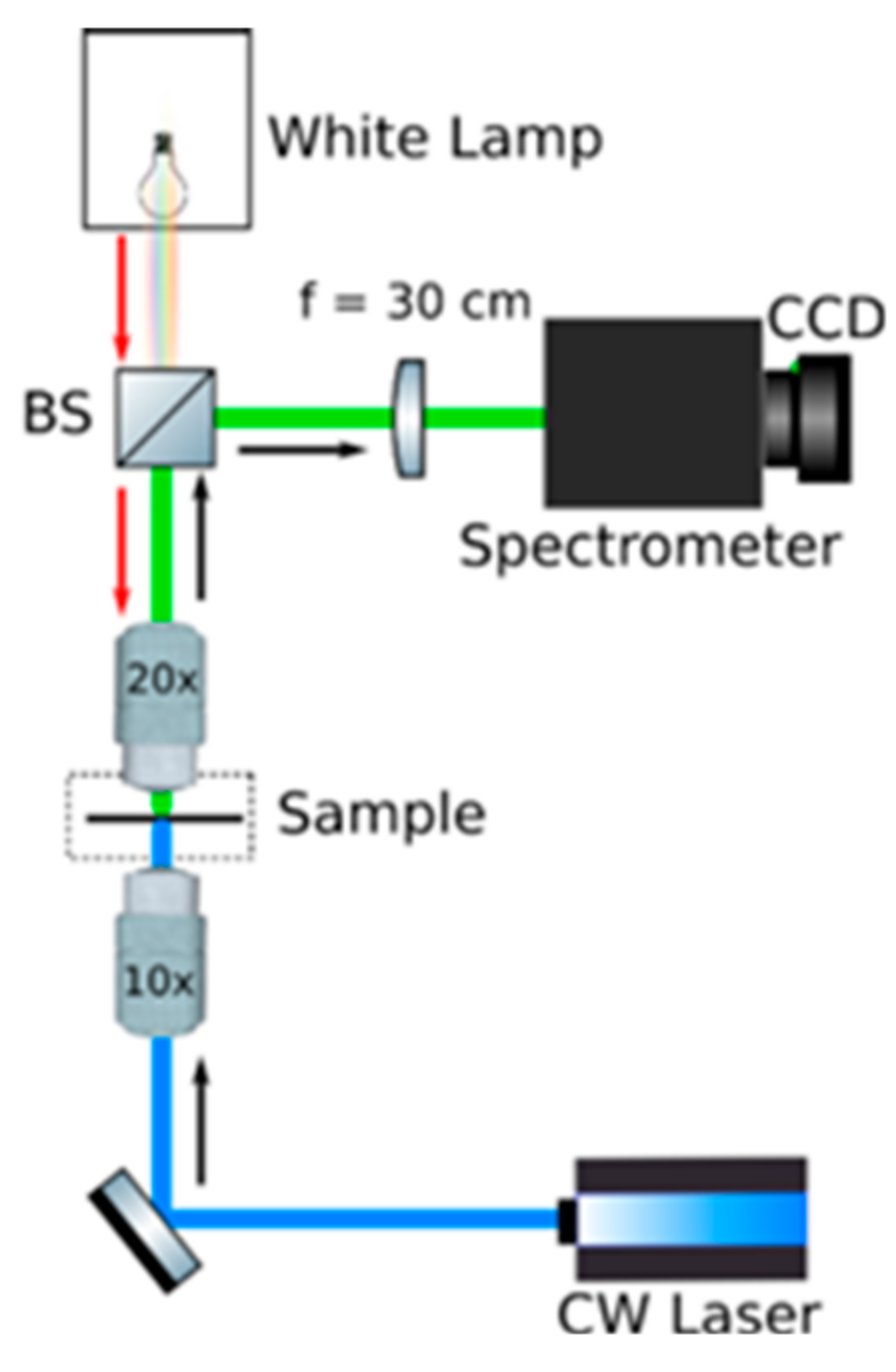
4. Experimental Details
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Green, M.A.; Dunlop, E.D.; Levi, D.H.; Hohl-Ebinger, J.; Yoshita, M.; Ho-Baillie, A.W. Solar cell efficiency tables (version 54). Prog. Photovolt. Res. Appl. 2019, 27, 565–575. [Google Scholar] [CrossRef]
- Sutherland, B.R.; Sargent, E.H. Perovskite photonic sources. Nat. Phot. 2016, 10, 295. [Google Scholar] [CrossRef]
- Pedesseau, L.; Sapori, D.; Traore, B.; Robles, R.; Fang, H.-H.; Loi, M.A.; Tsai, H.; Nie, W.; Blancon, J.-C.; Neukirch, A.; et al. Advances and promises of layered halide hybrid perovskite semiconductors. ACS Nano 2016, 10, 9776–9786. [Google Scholar] [CrossRef] [PubMed]
- Su, R.; Diederichs, C.; Wang, J.; Liew, T.C.; Zhao, J.; Liu, S.; Xu, W.; Chen, Z.; Xiong, Q. Room-temperature polariton lasing in all-inorganic perovskite nanoplatelets. Nano Lett. 2017, 17, 3982–3988. [Google Scholar] [CrossRef] [PubMed]
- Adjokatse, S.; Fang, H.-H.; Loi, M.A. Broadly tunable metal halide perovskites for solid-state light-emission applications. Mater. Today 2017, 20, 413–424. [Google Scholar] [CrossRef]
- Kanemitsu, Y. Luminescence spectroscopy of lead-halide perovskites: Materials properties and application as photovoltaic devices. J. Mater. Chem. C 2017, 5, 3427–3437. [Google Scholar] [CrossRef]
- Fieramosca, A.; De Marco, L.; Passoni, M.; Polimeno, L.; Rizzo, A.; Rosa, B.L.; Cruciani, G.; Dominici, L.; De Giorgi, M.; Gigli, G.; et al. Tunable out-of-plane excitons in 2D single-crystal perovskites. ACS Photonics 2018, 5, 4179–4185. [Google Scholar] [CrossRef]
- Cinquino, M.; Polimeno, L.; Lerario, G.; De Giorgi, M.; Moliterni, A.; Olieric, V.; Fieramosca, A.; Carallo, S.; Mastria, R.; Ardizzone, V.; et al. One-step synthesis at room temperature of low dimensional perovskite single crystals with high optical quality. J. Lumin. 2020, 221, 117079. [Google Scholar] [CrossRef]
- Stoumpos, C.C.; Cao, D.H.; Clark, D.J.; Young, J.; Rondinelli, J.M.; Jang, J.I.; Hupp, J.T.; Kanatzidis, M.G. Ruddlesden-Popper hybrid lead iodide perovskite 2D homologous semiconductors. Chem. Mater. 2016, 28, 2852–2867. [Google Scholar] [CrossRef]
- Fieramosca, A.; Polimeno, L.; Ardizzone, V.; De Marco, L.; Pugliese, M.; Maiorano, V.; De Giorgi, M.; Dominici, L.; Gigli, G.; Gerace, D.; et al. Two-dimensional hybrid perovskites sustaining strong polariton interactions at room temperature. Sci. Adv. 2019, 5, eaav9967. [Google Scholar] [CrossRef] [PubMed]
- Polimeno, L.; Fieramosca, A.; Lerario, G.; Cinquino, M.; De Giorgi, M.; Ballarini, D.; Todisco, F.; Dominici, L.; Ardizzone, V.; Pugliese, M.; et al. Observation of two thresholds leading to polariton condensation in 2D hybrid perovskites. Adv. Opt. Mater. 2020, 8, 2000176. [Google Scholar] [CrossRef]
- Ardizzone, V.; De Marco, L.; De Giorgi, M.; Dominici, L.; Ballarini, D.; Sanvitto, D. Emerging 2D materials for room-temperature polaritonics. Nanophotonics 2019, 8, 1547–1558. [Google Scholar] [CrossRef]
- Jemli, K.; Diab, H.; Lédée, F.; Trippé-Allard, G.; Garrot, D.; Geffroy, B.; Lauret, J.-S.; Audebert, P.; Deleporte, E. Using low temperature photoluminescence spectroscopy to investigate CH3NH3PbI3 hybrid perovskite degradation. Molecules 2016, 21, 885. [Google Scholar] [CrossRef]
- Shi, D.; Adinolfi, V.; Comin, R.; Yuan, M.; Alarousu, E.; Buin, A.; Chen, Y.; Hoogland, S.; Rothenberger, A.; Katsiev, K.; et al. Low trap-state density and long carrier diffusion in organolead trihalide perovskite single crystals. Science 2015, 347, 519–522. [Google Scholar] [CrossRef]
- Ding, J.; Lian, Z.; Li, Y.; Wang, S.; Yan, Q. The role of surface defects in photoluminescence and decay dynamics of high-quality perovskite MAPbI3 single crystals. J. Phys. Chem. Lett. 2018, 9, 4221–4226. [Google Scholar] [CrossRef]
- Luo, C.; Yan, C.; Li, W.; Chun, F.; Xie, M.; Zhu, Z.; Gao, Y.; Guo, B.; Yang, W. Ultrafast Thermodynamic Control for Stable and Efficient Mixed Halide Perovskite Nanocrystals. Adv. Funct. Mater. 2020, 30, 2000026. [Google Scholar] [CrossRef]
- Aristidou, N.; Sanchez-Molina, I.; Chotchuangchutchaval, T.; Brown, M.; Martinez, L.; Rath, T.; Haque, S.A. The role of oxygen in the degradation of methylammonium lead trihalide perovskite photoactive layers. Angew. Chem. Int. Ed. 2015, 54, 8208–8212. [Google Scholar] [CrossRef] [PubMed]
- Fang, H.H.; Yang, J.; Tao, S.; Adjokatse, S.; Kamminga, M.E.; Ye, J.; Blake, G.R.; Even, J.; Loi, M.A. Unravelling Light-Induced Degradation of Layered Perovskite Crystals and Design of Efficient Encapsulation for Improved Photostability. Adv. Funct. Mater. 2018, 28, 1800305. [Google Scholar] [CrossRef]
- Seitz, M.; Gant, P.; Castellanos-Gomez, A.; Prins, F. Long-Term Stabilization of Two-Dimensional Perovskites by Encapsulation with Hexagonal Boron Nitride. Nanomaterials 2019, 9, 1120. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Audebert, P.; Galmiche, L.; Lauret, J.-S.; Deleporte, E. Photostability of 2D organic-inorganic hybrid perovskites. Materials 2014, 7, 4789. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Oswald, I.W.; Stuard, S.J.; Nahid, M.M.; Zhou, N.; Williams, O.F.; Guo, Z.; Yan, L.; Hu, H.; Chen, Z.; et al. Synthetic control over orientational degeneracy of spacer cations enhances solar cell efficiency in two-dimensional perovskites. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef]
- Shi, J.; Gao, Y.; Gao, X.; Zhang, Y.; Zhang, J.; Jing, X.; Shao, M. Fluorinated Low-Dimensional Ruddlesden-Popper Perovskite Solar Cells with over 17% Power Conversion Efficiency and Improved Stability. Adv. Mater. 2019, 31, 1901673. [Google Scholar] [CrossRef] [PubMed]
- Fieramosca, A.; Polimeno, L.; Lerario, G.; De Marco, L.; De Giorgi, M.; Ballarini, D.; Dominici, L.; Ardizzone, V.; Pugliese, M.; Maiorano, V.; et al. Chromodynamics of photons in an artificial non-Abelian magnetic Yang-Mills field. arXiv 2019, arXiv:1912.09684. [Google Scholar]
- Polimeno, L.; De Giorgi, M.; Lerario, G.; De Marco, L.; Dominici, L.; Ardizzone, V.; Pugliese, M.; Prontera, C.T.; Maiorano, V.; Moliterni, A.; et al. Tuning the Berry curvature in 2D Perovskite. arXiv 2020, arXiv:2007.14945. [Google Scholar]
- Chong, W.K.; Thirumal, K.; Giovanni, D.; Goh, T.W.; Liu, X.; Mathews, N.; Mhaisalkar, S.; Sum, T.C. Dominant factors limiting the optical gain in layered two-dimensional halide perovskite thin films. Phys. Chem. Chem. Phys. 2016, 18, 14701–14708. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Su, R.; Xing, J.; Bao, D.; Diederichs, C.; Liu, S.; Liew, T.C.; Chen, Z.; Xiong, Q. Room temperature coherently coupled exciton-polaritons in two-dimensional organic-inorganic perovskite. ACS Nano 2018, 12, 8382–8389. [Google Scholar] [CrossRef] [PubMed]
- Lédée, F.; Trippé-Allard, G.; Diab, H.; Audebert, P.; Garrot, D.; Lauret, J.-S.; Deleporte, E. Fast growth of monocrystalline thin films of 2D layered hybrid perovskite. CrystEngComm 2017, 19, 2598–2602. [Google Scholar] [CrossRef]
- Kikuchi, K.; Takeoka, Y.; Rikukawa, M.; Sanui, K. Structure and optical properties of lead iodide based two-dimensional perovskite compounds containing fluorophenethylamines. Curr. Appl. Phys. 2004, 4, 599–602. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, B.; Liu, Y.; Deng, Y.; Bai, Y.; Dong, Q.; Huang, J. Scaling behavior of moisture-induced grain degradation in polycrystalline hybrid perovskite thin films. Energy Environ. Sci. 2017, 10, 516–522. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coriolano, A.; Polimeno, L.; De Giorgi, M.; Todisco, F.; Mastria, R.; Ardizzone, V.; Dominici, L.; Ballarini, D.; Rizzo, A.; Gigli, G.; et al. Improved Photostability in Fluorinated 2D Perovskite Single Crystals. Nanomaterials 2021, 11, 465. https://doi.org/10.3390/nano11020465
Coriolano A, Polimeno L, De Giorgi M, Todisco F, Mastria R, Ardizzone V, Dominici L, Ballarini D, Rizzo A, Gigli G, et al. Improved Photostability in Fluorinated 2D Perovskite Single Crystals. Nanomaterials. 2021; 11(2):465. https://doi.org/10.3390/nano11020465
Chicago/Turabian StyleCoriolano, Annalisa, Laura Polimeno, Milena De Giorgi, Francesco Todisco, Rosanna Mastria, Vincenzo Ardizzone, Lorenzo Dominici, Dario Ballarini, Aurora Rizzo, Giuseppe Gigli, and et al. 2021. "Improved Photostability in Fluorinated 2D Perovskite Single Crystals" Nanomaterials 11, no. 2: 465. https://doi.org/10.3390/nano11020465
APA StyleCoriolano, A., Polimeno, L., De Giorgi, M., Todisco, F., Mastria, R., Ardizzone, V., Dominici, L., Ballarini, D., Rizzo, A., Gigli, G., Sanvitto, D., & De Marco, L. (2021). Improved Photostability in Fluorinated 2D Perovskite Single Crystals. Nanomaterials, 11(2), 465. https://doi.org/10.3390/nano11020465








