Novel Pastes Containing Polymeric Nanoparticles for Dentin Hypersensitivity Treatment: An In Vitro Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Nanoparticle Production and Description
2.2. Experimental Toothpaste Preparation
2.3. Dentin Disks Preparation
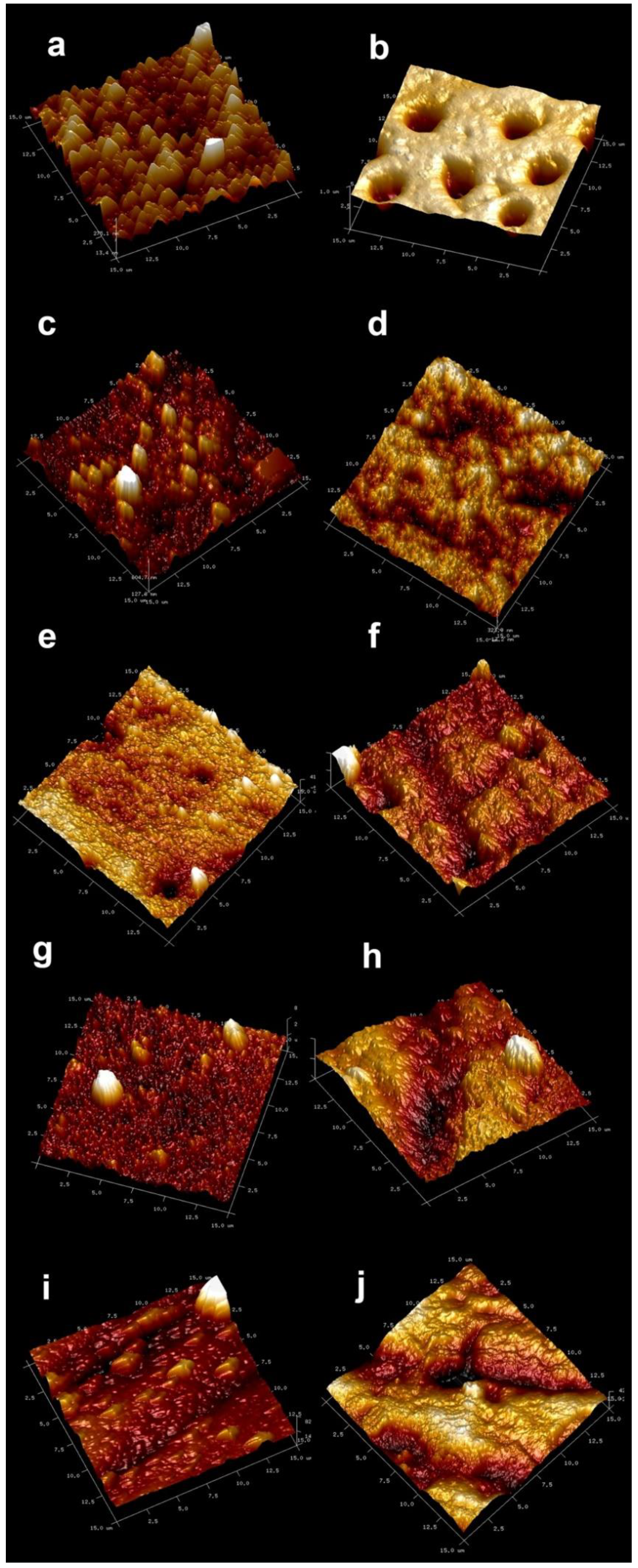
2.4. Atomic Force Microscopy (AFM) Imaging
2.5. Nanomechanical Properties Analysis
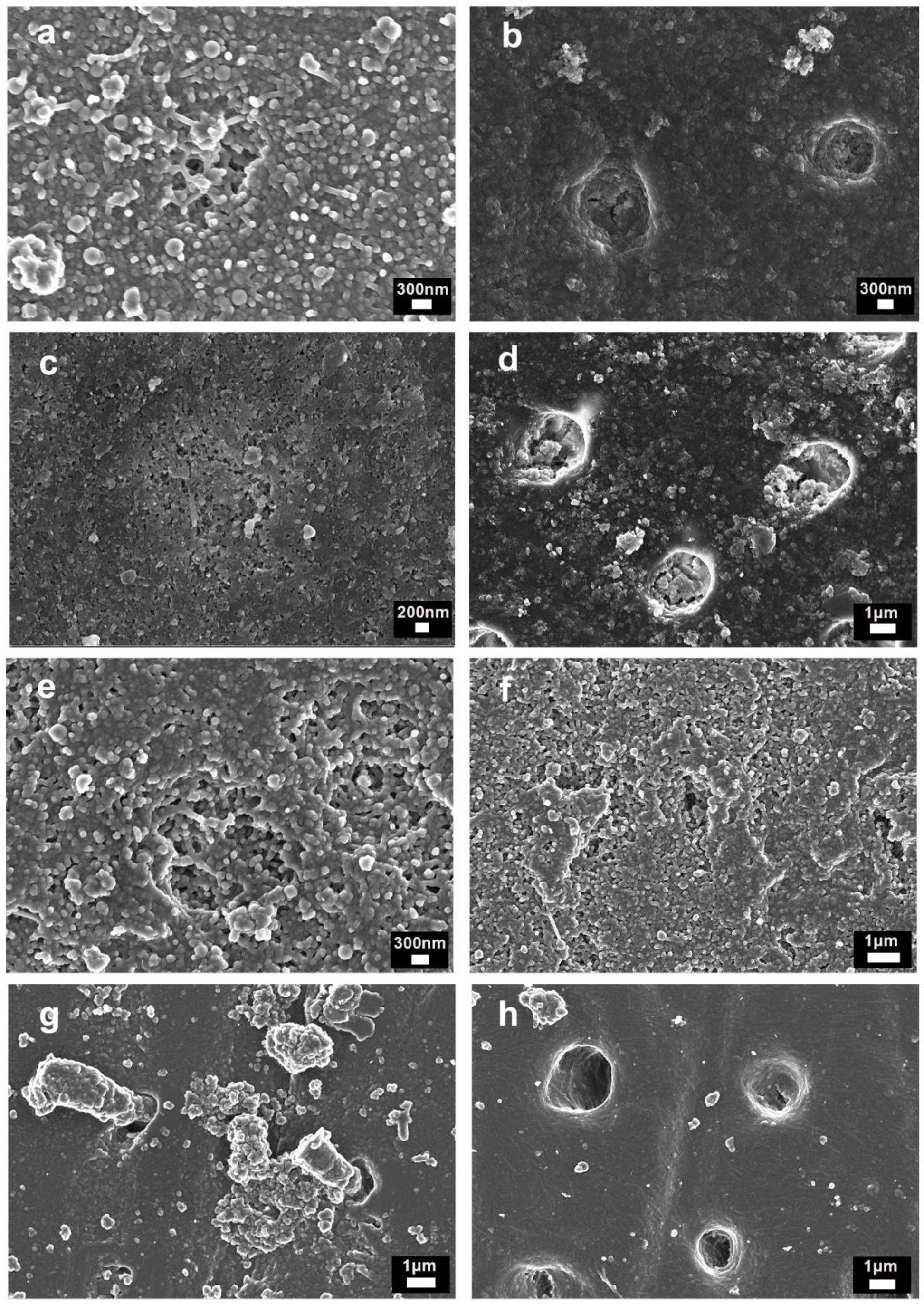
2.6. Field Emission Scanning Electron Microscopy (FESEM)
2.7. Statistical Analysis
3. Results
3.1. AFM Imaging
3.2. Nanomechanical Properties Analysis
3.3. Field Emission Scanning Electron Microscopy (FESEM)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Anthoney, D.; Zahid, S.; Khalid, H.; Khurshid, Z.; Shah, A.T.; Chaudhry, A.A.; Khan, A.S. Effectiveness of Thymoquinone and Fluoridated Bioactive Glass/Nano-Oxide Contained Dentifrices on Abrasion and Dentine Tubules Occlusion: An Ex Vivo Study. Eur. J. Dent. 2020, 14, 45–54. [Google Scholar] [CrossRef] [Green Version]
- West, N.X.; Sanz, M.; Lussi, A.; Bartlett, D.; Bouchard, P.; Bourgeois, D. Prevalence of Dentine Hypersensitivity and Study of Associated Factors: A European Population-Based Cross-Sectional Study. J. Dent. 2013, 41, 841–851. [Google Scholar] [CrossRef]
- Jena, A.; Shashirekha, G. Comparison of Efficacy of Three Different Desensitizing Agents for In-Office Relief of Dentin Hypersensitivity: A 4 Weeks Clinical Study. J. Conserv. Dent. 2015, 18, 389–393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- West, N.X.; Lussi, A.; Seong, J.; Hellwig, E. Dentin Hypersensitivity: Pain Mechanisms and Aetiology of Exposed Cervical Dentin. Clin Oral Investig 2013, 17 (Suppl. 1), S9–S19. [Google Scholar] [CrossRef] [Green Version]
- Närhi, M.; Jyväsjärvi, E.; Virtanen, A.; Huopaniemi, T.; Ngassapa, D.; Hirvonen, T. Role of Intradental A- and C-Type Nerve Fibres in Dental Pain Mechanisms. Proc. Finn. Dent. Soc. 1992, 88 (Suppl. 1), 507–516. [Google Scholar]
- Osorio, R.; Toledano-Osorio, M.; Osorio, E.; Aguilera, F.S.; Padilla-Mondéjar, S.; Toledano, M. Zinc and Silica Are Active Components to Efficiently Treat in Vitro Simulated Eroded Dentin. Clin. Oral Investig. 2018, 22, 2859–2870. [Google Scholar] [CrossRef] [PubMed]
- Gandolfi, M.G.; Iacono, F.; Pirani, C.; Prati, C. The Use of Calcium-Silicate Cements to Reduce Dentine Permeability. Arch. Oral Biol. 2012, 57, 1054–1061. [Google Scholar] [CrossRef]
- Wang, Z.; Jiang, T.; Sauro, S.; Pashley, D.H.; Toledano, M.; Osorio, R.; Liang, S.; Xing, W.; Sa, Y.; Wang, Y. The Dentine Remineralization Activity of a Desensitizing Bioactive Glass-Containing Toothpaste: An in Vitro Study. Aust. Dent. J. 2011, 56, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Sa, Y.; Sauro, S.; Chen, H.; Xing, W.; Ma, X.; Jiang, T.; Wang, Y. Effect of Desensitising Toothpastes on Dentinal Tubule Occlusion: A Dentine Permeability Measurement and SEM in Vitro Study. J. Dent. 2010, 38, 400–410. [Google Scholar] [CrossRef] [PubMed]
- Lynch, E.; Brauer, D.S.; Karpukhina, N.; Gillam, D.G.; Hill, R.G. Multi-Component Bioactive Glasses of Varying Fluoride Content for Treating Dentin Hypersensitivity. Dent. Mater. 2012, 28, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Arnold, W.H.; Gröger, C.; Bizhang, M.; Naumova, E.A. Dentin Abrasivity of Various Desensitizing Toothpastes. Head Face Med. 2016, 12, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pashley, D.H. Dentin Permeability, Dentin Sensitivity, and Treatment through Tubule Occlusion. J. Endod. 1986, 12, 465–474. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Taddei, P.; Siboni, F.; Modena, E.; De Stefano, E.D.; Prati, C. Biomimetic Remineralization of Human Dentin Using Promising Innovative Calcium-Silicate Hybrid “Smart” Materials. Dent. Mater. 2011, 27, 1055–1069. [Google Scholar] [CrossRef] [PubMed]
- Osorio, R.; Alfonso-Rodríguez, C.A.; Medina-Castillo, A.L.; Alaminos, M.; Toledano, M. Bioactive Polymeric Nanoparticles for Periodontal Therapy. PLoS ONE 2016, 11, e0166217. [Google Scholar] [CrossRef]
- Toledano-Osorio, M.; Osorio, E.; Aguilera, F.S.; Luis Medina-Castillo, A.; Toledano, M.; Osorio, R. Improved Reactive Nanoparticles to Treat Dentin Hypersensitivity. Acta Biomater. 2018, 72, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Toledano, M.; Osorio, R.; Osorio, E.; Medina-Castillo, A.L.; Toledano-Osorio, M.; Aguilera, F.S. Ions-Modified Nanoparticles Affect Functional Remineralization and Energy Dissipation through the Resin-Dentin Interface. J. Mech. Behav. Biomed. Mater. 2017, 68, 62–79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medina-Castillo, A.L.; Fernandez-Sanchez, J.F.; Segura-Carretero, A.; Fernandez-Gutierrez, A. Micrometer and Submicrometer Particles Prepared by Precipitation Polymerization: Thermodynamic Model and Experimental Evidence of the Relation between Flory’s Parameter and Particle Size. Macromolecules 2010, 43, 5804–5813. [Google Scholar] [CrossRef]
- Medina-Castillo, A.L. Thermodynamic Principles of Precipitation Polymerization and Role of Fractal Nanostructures in the Particle Size Control. Macromolecules 2020, 53, 5687–5700. [Google Scholar] [CrossRef]
- Ryou, H.; Romberg, E.; Pashley, D.H.; Tay, F.R.; Arola, D. Nanoscopic Dynamic Mechanical Properties of Intertubular and Peritubular Dentin. J. Mech. Behav. Biomed. Mater. 2012, 7, 3–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pashley, D.H.; Tay, F.R.; Carvalho, R.M.; Rueggeberg, F.A.; Agee, K.A.; Carrilho, M.; Donnelly, A.; García-Godoy, F. From Dry Bonding to Water-Wet Bonding to Ethanol-Wet Bonding. A Review of the Interactions between Dentin Matrix and Solvated Resins Using a Macromodel of the Hybrid Layer. Am. J. Dent. 2007, 20, 7–20. [Google Scholar] [PubMed]
- Han, L.; Grodzinsky, A.J.; Ortiz, C. Nanomechanics of the Cartilage Extracellular Matrix. Annu. Rev. Mater. Res. 2011, 41, 133–168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farooq, I.; Moheet, I.A.; AlShwaimi, E. In Vitro Dentin Tubule Occlusion and Remineralization Competence of Various Toothpastes. Arch. Oral Biol. 2015, 60, 1246–1253. [Google Scholar] [CrossRef]
- Hirsiger, C.; Schmidlin, P.R.; Michaelis, M.; Hirsch, C.; Attin, T.; Heumann, C.; Doméjean, S.; Gernhardt, C.R. Efficacy of 8% Arginine on Dentin Hypersensitivity: A Multicenter Clinical Trial in 273 Patients over 24 Weeks. J. Dent. 2019, 83, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martins, C.C.; Firmino, R.T.; Riva, J.J.; Ge, L.; Carrasco-Labra, A.; Brignardello-Petersen, R.; Colunga-Lozano, L.E.; Granville-Garcia, A.F.; Costa, F.O.; Yepes-Nuñez, J.J.; et al. Desensitizing Toothpastes for Dentin Hypersensitivity: A Network Meta-Analysis. J. Dent. Res. 2020, 99, 514–522. [Google Scholar] [CrossRef]
- Hill, R.; Gillam, D. Future strategies for the development of desensitising products. In Dentine Hypersensitivity: Advances in Diagnosis, Management and Treatment; Springer: Basel, Switzerland, 2015; pp. 157–180. [Google Scholar]
- Pashley, D.H.; Michelich, V.; Kehl, T. Dentin Permeability: Effects of Smear Layer Removal. J. Prosthet. Dent. 1981, 46, 531–537. [Google Scholar] [CrossRef]
- O’Toole, S.; Bartlett, D. The Relationship between Dentine Hypersensitivity, Dietary Acid Intake and Erosive Tooth Wear. J. Dent. 2017, 67, 84–87. [Google Scholar] [CrossRef] [Green Version]
- Arnold, W.H.; Prange, M.; Naumova, E.A. Effectiveness of Various Toothpastes on Dentine Tubule Occlusion. J. Dent. 2015, 43, 440–449. [Google Scholar] [CrossRef] [Green Version]
- Degli Esposti, L.; Ionescu, A.C.; Brambilla, E.; Tampieri, A.; Iafisco, M. Characterization of a Toothpaste Containing Bioactive Hydroxyapatites and In Vitro Evaluation of Its Efficacy to Remineralize Enamel and to Occlude Dentinal Tubules. Materials 2020, 13, 2928. [Google Scholar] [CrossRef]
- Takatsuka, T.; Tanaka, K.; Iijima, Y. Inhibition of Dentine Demineralization by Zinc Oxide: In Vitro and in Situ Studies. Dent. Mater. 2005, 21, 1170–1177. [Google Scholar] [CrossRef]
- Osorio, R.; Cabello, I.; Medina-Castillo, A.L.; Osorio, E.; Toledano, M. Zinc-Modified Nanopolymers Improve the Quality of Resin-Dentin Bonded Interfaces. Clin. Oral Investig. 2016, 20, 2411–2420. [Google Scholar] [CrossRef] [Green Version]
- Toledano-Osorio, M.; Babu, J.P.; Osorio, R.; Medina-Castillo, A.L.; García-Godoy, F.; Toledano, M. Modified Polymeric Nanoparticles Exert In Vitro Antimicrobial Activity Against Oral Bacteria. Materials 2018, 11, 1013. [Google Scholar] [CrossRef] [Green Version]
- Osorio, R.; Osorio, E.; Medina-Castillo, A.L.; Toledano, M. Polymer Nanocarriers for Dentin Adhesion. J. Dent. Res. 2014, 93, 1258–1263. [Google Scholar] [CrossRef] [Green Version]
- Sánchez, M.C.; Toledano-Osorio, M.; Bueno, J.; Figuero, E.; Toledano, M.; Medina-Castillo, A.L.; Osorio, R.; Herrera, D.; Sanz, M. Antibacterial Effects of Polymeric PolymP-n Active Nanoparticles. An in Vitro Biofilm Study. Dent. Mater. 2019, 35, 156–168. [Google Scholar] [CrossRef] [PubMed]
- Toledano-Osorio, M.; Osorio, R.; Aguilera, F.S.; Medina-Castillo, A.L.; Toledano, M.; Osorio, E.; Acosta, S.; Chen, R.; Aparicio, C. Polymeric Nanoparticles Protect the Resin-Dentin Bonded Interface from Cariogenic Biofilm Degradation. Acta Biomater. 2020, 111, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Carrouel, F.; Viennot, S.; Ottolenghi, L.; Gaillard, C.; Bourgeois, D. Nanoparticles as Anti-Microbial, Anti-Inflammatory, and Remineralizing Agents in Oral Care Cosmetics: A Review of the Current Situation. Nanomaterials 2020, 10, 140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
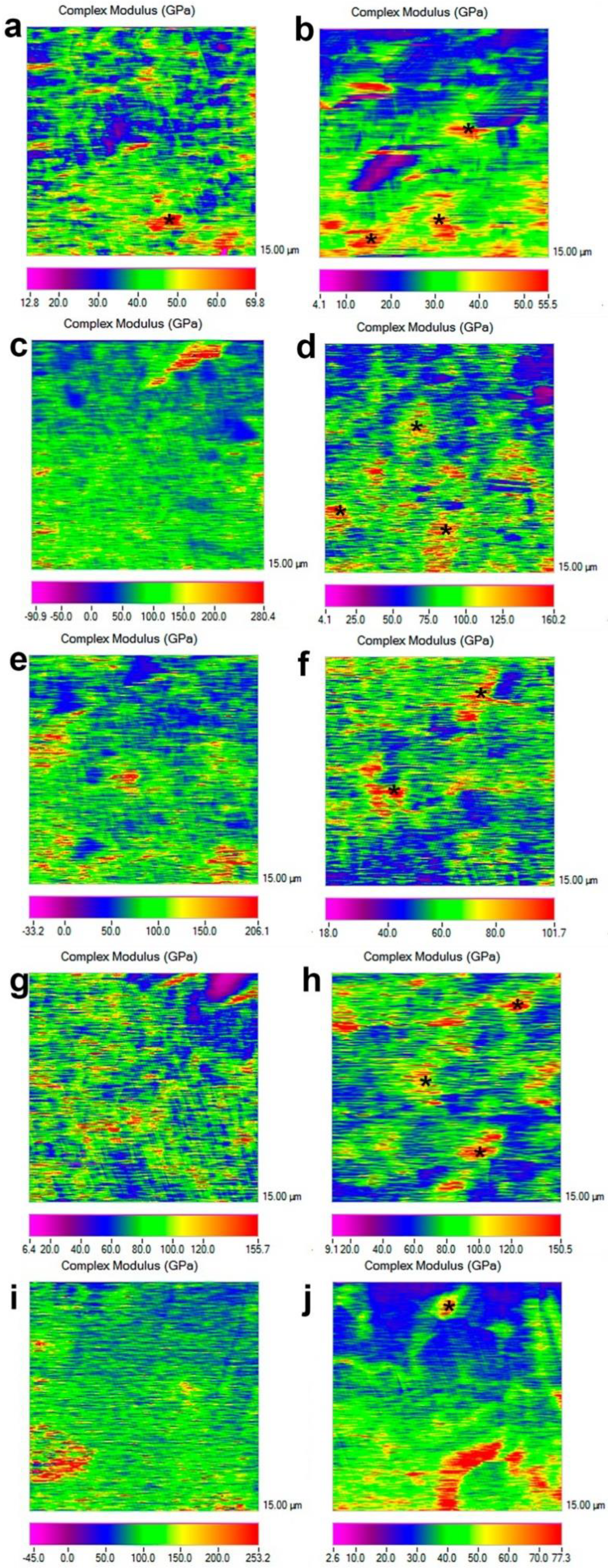

| Dentin Treatment | Complex Modulus (GPa) | |||
|---|---|---|---|---|
| Intratubular Dentin | Intertubular Dentin | |||
| Mean | SD | Mean | SD | |
| Distilled water (DW) | 57.37 b | 6.82 | 45.55 B | 6.70 |
| Zn-NP paste P1% | 147.87 f | 19.94 | 87.82 D | 10.50 |
| Zn-NP paste P5% | 133.99 def | 14.15 | 73.15 C | 8.04 |
| Zn-NP paste P10% | 110.64 d | 6.97 | 79.31 CD | 3.39 |
| Sensodyne paste ® | 140.93 ef | 39.12 | 89.26 D | 12.86 |
| DW + Citric Acid (CA) | 24.18 a | 4.21 | 31.93 A | 6.15 |
| Zn-NP paste P1% + CA | 116.75 de | 6.52 | 74.43 C | 6.59 |
| Zn-NP paste P5% + CA | 83.28 c | 11.15 | 69.72 C | 8.26 |
| Zn-NP paste P10% + CA | 127.38 def | 13.21 | 77.54 CD | 7.73 |
| Sensodyne® paste + CA | 51.10 b | 13.84 | 48.41 B | 8.83 |
| Dentin Treatment | Tubular Occlusion | ||
|---|---|---|---|
|
Open Tubules |
Filled/Partially Filled Tubules | %Total Open Tubules | |
| EDTA | 273 | 0 | 100 |
| EDTA+Zn-NP paste P1% | 0 | 0 | 0 |
| EDTA+Zn-NP paste P5% | 0 | 3 | 0 |
| EDTA+Zn-NP paste P10% | 0 | 9 | 0 |
| EDTA+Sensodyne® paste | 0 | 25 | 0 |
| EDTA+Citric Acid (CA) | 274 | 0 | 100 |
| EDTA+Zn-NP paste P1% + CA | 0 | 12 | 0 |
| EDTA+Zn-NP paste P5% + CA | 2 | 189 | 0.007 |
| EDTA+Zn-NP paste P10% + CA | 3 | 9 | 0.01 |
| EDTA+Sensodyne® paste +CA | 86 | 30 | 31.38 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toledano-Osorio, M.; Osorio, R.; Osorio, E.; Medina-Castillo, A.L.; Toledano, M. Novel Pastes Containing Polymeric Nanoparticles for Dentin Hypersensitivity Treatment: An In Vitro Study. Nanomaterials 2021, 11, 3150. https://doi.org/10.3390/nano11113150
Toledano-Osorio M, Osorio R, Osorio E, Medina-Castillo AL, Toledano M. Novel Pastes Containing Polymeric Nanoparticles for Dentin Hypersensitivity Treatment: An In Vitro Study. Nanomaterials. 2021; 11(11):3150. https://doi.org/10.3390/nano11113150
Chicago/Turabian StyleToledano-Osorio, Manuel, Raquel Osorio, Estrella Osorio, Antonio L. Medina-Castillo, and Manuel Toledano. 2021. "Novel Pastes Containing Polymeric Nanoparticles for Dentin Hypersensitivity Treatment: An In Vitro Study" Nanomaterials 11, no. 11: 3150. https://doi.org/10.3390/nano11113150
APA StyleToledano-Osorio, M., Osorio, R., Osorio, E., Medina-Castillo, A. L., & Toledano, M. (2021). Novel Pastes Containing Polymeric Nanoparticles for Dentin Hypersensitivity Treatment: An In Vitro Study. Nanomaterials, 11(11), 3150. https://doi.org/10.3390/nano11113150









