Microcrystalline Cellulose Extracted from Native Plants as an Excipient for Solid Dosage Formulations in Drug Delivery
Abstract
1. Introduction
2. Materials and Methods
2.1. Cellulose Samples
2.2. BC Extraction
2.3. Preparation of BC as an Excipient
2.4. Characterization of Excipients
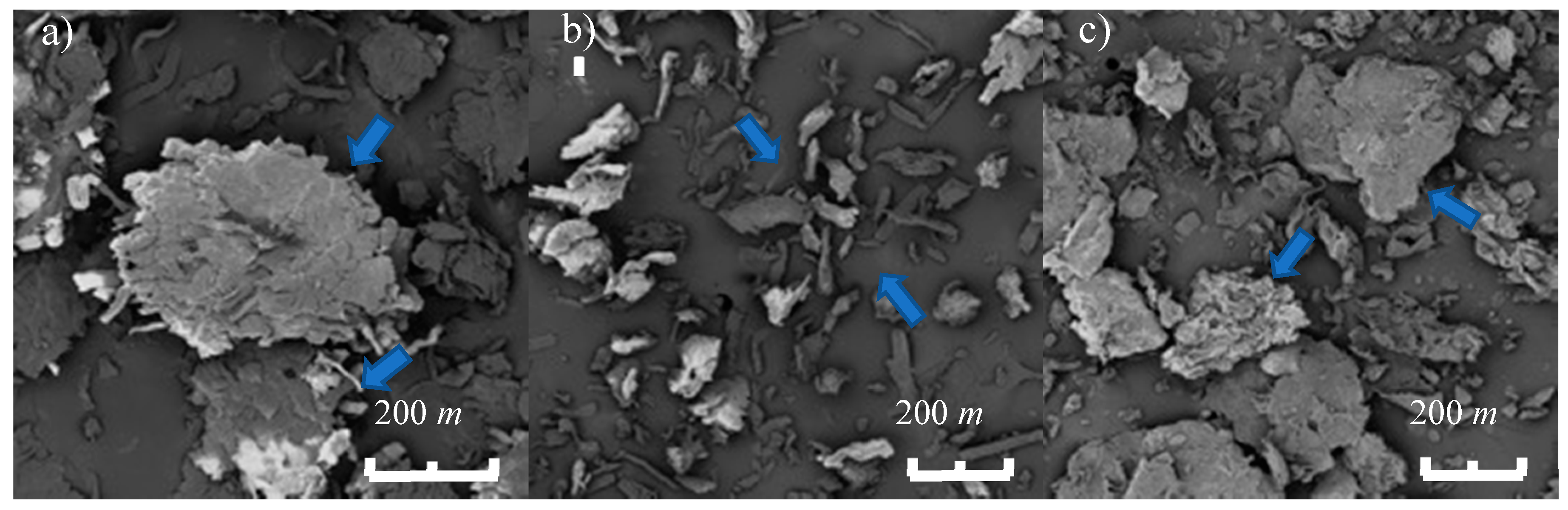
2.4.1. Scanning Electron Microscopy (SEM)
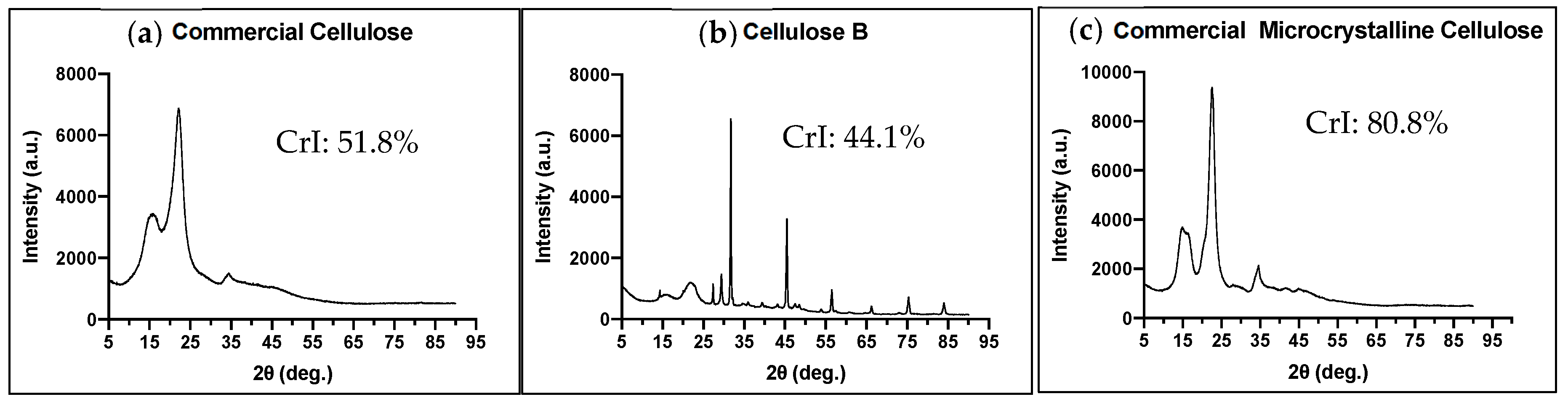
2.4.2. X-ray Diffraction (XRD)
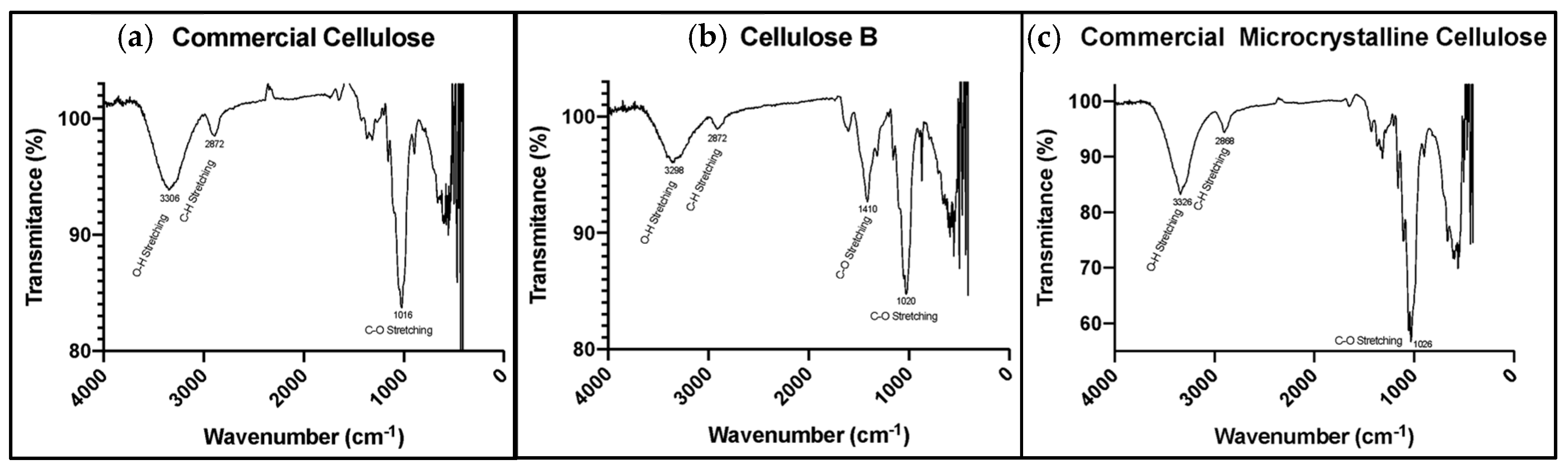
2.4.3. Fourier-Transform Infrared (FTIR) Spectroscopy
2.5. Rheological Properties of Excipients
2.5.1. Loss on Drying
2.5.2. Carr’s Index and Hausner’s Ratio
2.5.3. Angle of Repose
2.5.4. Statistical Analysis
2.6. Compression
2.7. Quality Control of Tablets
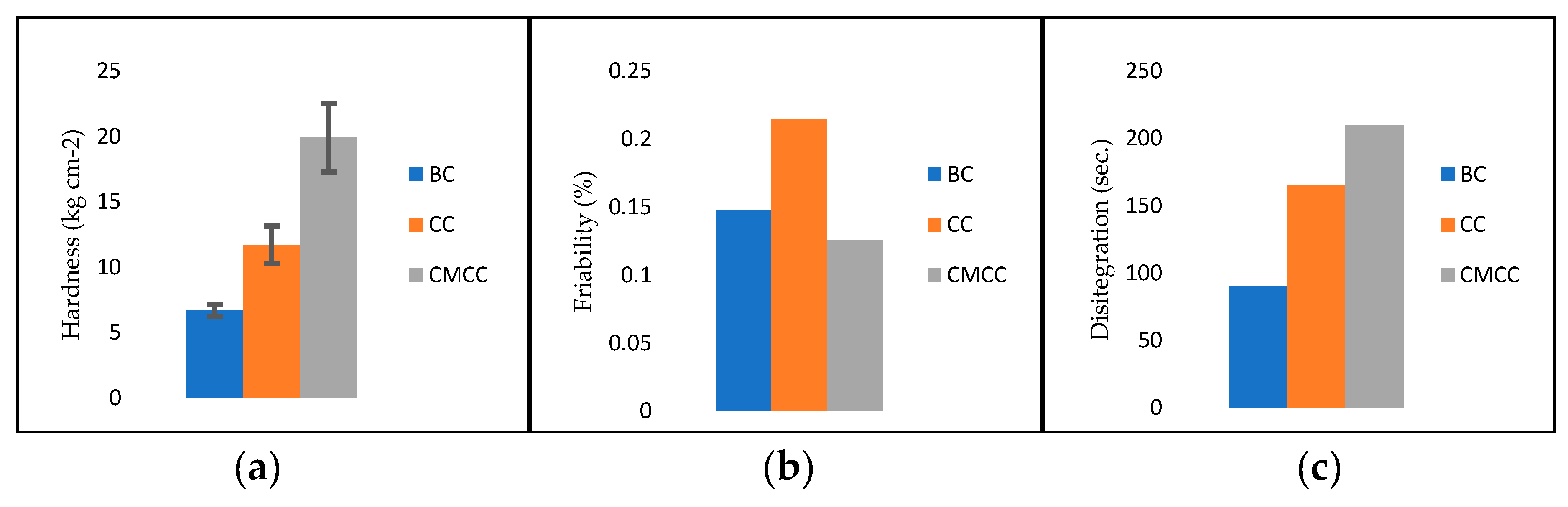
2.7.1. Hardness
2.7.2. Friability
2.7.3. Disintegration Time
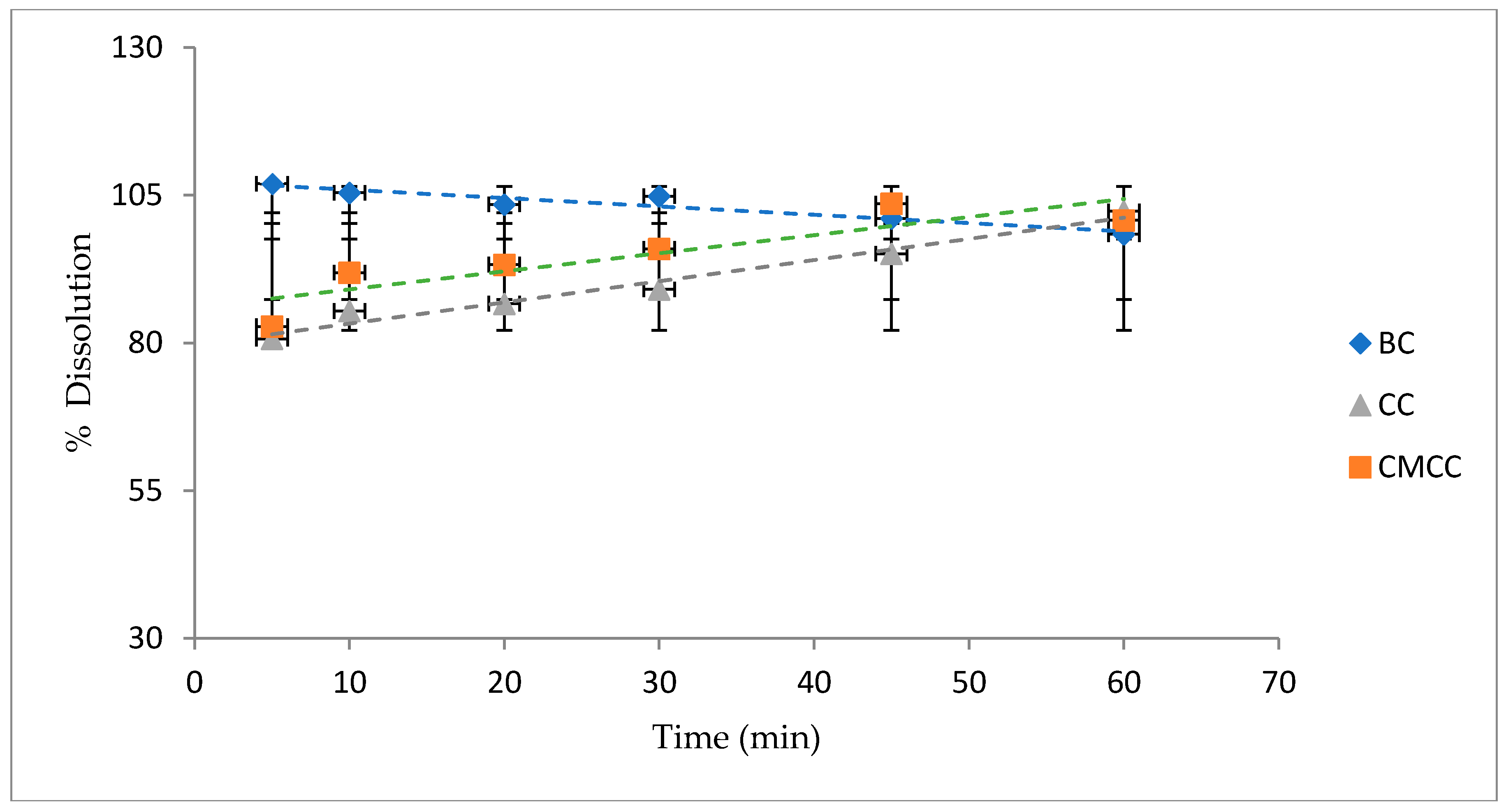
2.7.4. Dissolution Test
3. Results
3.1. Characterization of Excipients
3.1.1. Crystallinity of the Cellulose Excipients
3.1.2. Chemical Composition of the Cellulose Excipients
3.1.3. Morphology of the Cellulose Excipients
3.2. Loss on Drying of B Cellulose
3.3. Rheological Properties of Excipients
3.3.1. Carr’s Index and Hausner’s Ratio
3.3.2. Angle of Repose
3.4. Fabrication and Quality Control of Tablets
3.4.1. Hardness, Friability and Disintegration
3.4.2. Acetaminophen Dissolution Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pharmaceutical Outsourcing. Solid Dosage Processing and Packaging. Available online: https://www.pharmoutsourcing.com/Featured-Articles/350152-Solid-Dosage-Processing-and-Packaging/ (accessed on 7 September 2019).
- Hamman, J.H.; Tarirai, C. Functional excipients. Chem. Today 2006, 24, 57–62. [Google Scholar]
- IPEC. The Joint IPEC-PQG Good Manufacturing Practices Audit Guideline for Pharmaceutical Excipients; World Health: Brussels, Belgium, 2008. [Google Scholar]
- Zatz, J.; Kushla, G. Pharmaceutical Dosage Forms: Disperse Systems; Banker, G.S., Rieger, M.M., Lieberman, H.A., Eds.; CRC Press: New York, NY, USA, 1996; pp. 171–203. [Google Scholar]
- Pal, R.S.; Pal, Y.; Wal, A.; Wal, P. Current Review on Plant based Pharmaceutical Excipients. Open Med. J. 2019, 6, 1–5. [Google Scholar] [CrossRef]
- Balandrin, M.; Klocke, J.; Wurtele, E.; Bollinger, W. Natural plant chemicals: Sources of industrial and medicinal materials. Science 1985, 228, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Mahmood, T.; Shameem, A.; Bagga, P.; Ahmad, N. A review on Herbal Excipients and their pharmaceutical applications. Sch. Acad. J. Pharm. 2016, 5, 53–57. [Google Scholar]
- Shokri, J.; Adibki, K. Application of Cellulose and Cellulose Derivatives in Pharmaceutical Industries. In Cellulose—Medical, Pharmaceutical and Electronic Applications; Van De Ven, T.G.M., Ed.; InTech: London, UK, 2013; ISBN 9789535111917. [Google Scholar]
- Lee, C.K.; Diesendruck, C.E.; Lu, E.; Pickett, A.N.; May, P.A.; Moore, J.S.; Braun, P.V. Solvent swelling activation of a mechanophore in a polymer network. Macromolecules 2014, 47, 2690–2694. [Google Scholar] [CrossRef]
- Sun, B.; Zhang, M.; Shen, J.; He, Z.; Fatehi, P.; Ni, Y. Applications of Cellulose-based Materials in Sustained Drug Delivery Systems. Curr. Med. Chem. 2019, 26, 2485–2501. [Google Scholar] [CrossRef] [PubMed]
- Nobles, D.R.; Romanovicz, D.K.; Brown, R.M. Cellulose in cyanobacteria. Origin of vascular plant cellulose synthase? Plant Physiol. 2001, 127, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.M. Algae as tools in studying the biosynthesis of cellulose, nature’s most abundant macromolecule. In Cell Walls and Surfaces, Reproduction, Photosynthesis; Wiessner, W., Robinson, D.G., Starr, R.C., Eds.; Springer: Berlin/Heidelberg, Germany, 1990; Volume 1, pp. 20–39. ISBN 9783642486548. [Google Scholar]
- Kimura, S.; Itoh, T. A new cellulosic structure, the tunic cord in the ascidian Polyandrocarpa misakiensis. Protoplasma 1998, 204, 94–102. [Google Scholar] [CrossRef]
- Trache, D.; Hussin, M.H.; Hui Chuin, C.T.; Sabar, S.; Fazita, M.R.N.; Taiwo, O.F.A.; Hassan, T.M.; Haafiz, M.K.M. Microcrystalline cellulose: Isolation, characterization and bio-composites application—A review. Int. J. Biol. Macromol. 2016, 93, 789–804. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, G.T.; Gowthamarajan, K.; Dhobe, R.R.; Yohanan, F.; Suresh, B. Development of controlled release spheriods using natural polysaccharide as release modifier. Drug Deliv. 2005, 12, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Morán, J.I.; Alvarez, V.A.; Cyras, V.P.; Vázquez, A. Extraction of cellulose and preparation of nanocellulose from sisal fibers. Cellulose 2008, 15, 149–159. [Google Scholar] [CrossRef]
- Robles, L.V. Los excipientes y su funcionalidad en productos farmacéuticos sólidos. Revista Mexicana de Ciencias Farmacéuticas 2011, 42, 18–36. [Google Scholar]
- Días, C.; Camacho, A. Elaboración de Productos Farmaceúticos y Parafarmaceúticos, 1st ed.; McGraw-Hill: Madrid, Spain, 2005; ISBN 978-0-00-940150-3. [Google Scholar]
- Montanari, D.; Agostini, A.; Bonini, M.; Corti, G.; Del Ventisette, C. The use of empirical methods for testing granular materials in analogue modelling. Materials 2017, 10, 635. [Google Scholar] [CrossRef] [PubMed]
- Pharmapproach. Quality Control Tests for Tablets. Available online: https://www.pharmapproach.com/quality-control-tests-for-tablets/2 (accessed on 15 December 2019).
- Al-Gousous, J.; Langguth, P. Oral Solid Dosage Form Disintegration Testing—The Forgotten Test. J. Pharm. Sci. 2015, 104, 2664–2675. [Google Scholar] [CrossRef] [PubMed]
- The United States Pharmacopeia (USP). Monograph: Dissolution; USP 32 NF; The United States Pharmacopeial Convention: Rockville, MD, USA, 2016. [Google Scholar]
- Pèrez López, E. “In vitro” solution test of acetaminophen tablets, hplc quantifying with electrochemical detector. Intersedes 2014, 16, 27–37. [Google Scholar]
- Klemm, D.; Heublein, B.; Fink, H.P.; Bohn, A. Cellulose: Fascinating biopolymer and sustainable raw material. Angew. Chem. Int. Ed. 2005, 44, 3358–3393. [Google Scholar] [CrossRef] [PubMed]
- Markl, D.; Zeitler, J.A. A Review of Disintegration Mechanisms and Measurement Techniques. Pharm. Res. 2017, 34, 890–917. [Google Scholar] [CrossRef] [PubMed]
- Chandran, I.S.; Prasanna, P.M. Drug-excipient interaction studies of loperamide loaded in polsorbate 80 liposomes. Orient. J. Chem. 2015, 31, 2201–2206. [Google Scholar] [CrossRef]
- Bravo, I.; Figueroa, F.; Swasy, M.I.; Attia, M.F.; Ateia, M.; Encalada, D.; Vizuete, K.; Galeas, S.; Guerrero, V.H.; Debut, A.; et al. Cellulose Particles Capture Pollutants. RSC Adv. 2020, 10, 7967–7975. [Google Scholar] [CrossRef]
- Clerch, A. Aportación al Diseño de un Nuevo Excipiente Tipo “Coprocessed Product” Para Compresión Directa. Doctoral Thesis, Universidad de Barcelona, Barcelona, Spain, 2008. [Google Scholar]
- Cornejo, L.; Cordero, M. Evaluación de las Propiedades Farmacotécnicas en el Diseño y Formulación de Tabletas de Clorfenamina por Compresión Directa. Bachelor’s Thesis, Universidad Nacional Mayor de San Marcos, Lima, Peru, 2007. [Google Scholar]
- Bordallo, J.S. Determinación por Hplc de 4-Epianhidro-Tetraciclina Como Producto de Degradación en Formas Orales de Tetraciclina Condicionada por Nuevos Diseños Galenicos. Bachelor’s Thesis, Universidad Complutense de Madrid, Madrid, Spain, 1994. [Google Scholar]
- Council of Europe. European Pharmacopoeia European Pharmacopoeia 7.0, 7th ed.; Council of Europe: Strasbourg, France, 2010; pp. 1634–1637. [Google Scholar]
- Gad, S.C. (Ed.) Pharmaceutical Manufacturing Handbook: Production and Processes; Jonh Willey & Sons: Hoboken, NJ, USA, 2008; ISBN 9780470259580. [Google Scholar]
- Dyatlov, N.A. Pharmaceutical formulation and investigations of sustained-release paracetamol matrix tablets and their potential antipyretic effect in children. Sci. Pract. J. Obstet. Gynecol. 2018, 112–117. [Google Scholar] [CrossRef]
- Vila, J.L. Tecnología Farmacéutica: Formas Farmaceúticas; Síntesis: Madrid, Spain, 2001; Volume 2, ISBN 9788477385387. [Google Scholar]
| Component | Function | Amount Used (%) | Mass Used (g) |
|---|---|---|---|
| Acetaminophen | Active Principle | 10 | 2 |
| Cellulose | Diluent/disintegrant | 35 | 7 |
| Lactose | Diluent/binder | 53.75 | 10.75 |
| Aerosil | Glidant | 0.25 | 0.05 |
| PEG 600 | Lubricant | 1 | 0.2 |
| B Cellulose | Commercial Cellulose | Commercial MCC | |
|---|---|---|---|
| Carr’s index | 16.65 | 12.37 | 18.15 |
| Hausner’s ratio | 1.19 | 1.14 | 1.22 |
| BC | CC | CMCC |
|---|---|---|
| 25.38 | 31.78 | 41 |
| 25.27 | 29.05 | 41 |
| 24.26 | 30.6 | 41 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Viera-Herrera, C.; Santamaría-Aguirre, J.; Vizuete, K.; Debut, A.; Whitehead, D.C.; Alexis, F. Microcrystalline Cellulose Extracted from Native Plants as an Excipient for Solid Dosage Formulations in Drug Delivery. Nanomaterials 2020, 10, 975. https://doi.org/10.3390/nano10050975
Viera-Herrera C, Santamaría-Aguirre J, Vizuete K, Debut A, Whitehead DC, Alexis F. Microcrystalline Cellulose Extracted from Native Plants as an Excipient for Solid Dosage Formulations in Drug Delivery. Nanomaterials. 2020; 10(5):975. https://doi.org/10.3390/nano10050975
Chicago/Turabian StyleViera-Herrera, Camila, Javier Santamaría-Aguirre, Karla Vizuete, Alexis Debut, Daniel C. Whitehead, and Frank Alexis. 2020. "Microcrystalline Cellulose Extracted from Native Plants as an Excipient for Solid Dosage Formulations in Drug Delivery" Nanomaterials 10, no. 5: 975. https://doi.org/10.3390/nano10050975
APA StyleViera-Herrera, C., Santamaría-Aguirre, J., Vizuete, K., Debut, A., Whitehead, D. C., & Alexis, F. (2020). Microcrystalline Cellulose Extracted from Native Plants as an Excipient for Solid Dosage Formulations in Drug Delivery. Nanomaterials, 10(5), 975. https://doi.org/10.3390/nano10050975