Three-Year Study of Markers of Oxidative Stress in Exhaled Breath Condensate in Workers Producing Nanocomposites, Extended by Plasma and Urine Analysis in Last Two Years
Abstract
1. Introduction
2. Materials and Methods
2.1. Operations Description
2.2. Subjects
Questionnaire
2.3. Workplace Aerosol Measurements
2.4. Environmental Contamination
2.5. Collection of Biological Samples and Analysis of Oxidative Stress Biomarkers
2.6. Statistics
3. Results
3.1. Subjects
3.1.1. Exposure Characteristics
3.1.2. Spirometry
3.2. Workplace Aerosol
3.2.1. Mass Concentrations and Mass Share of Analyzed Elements
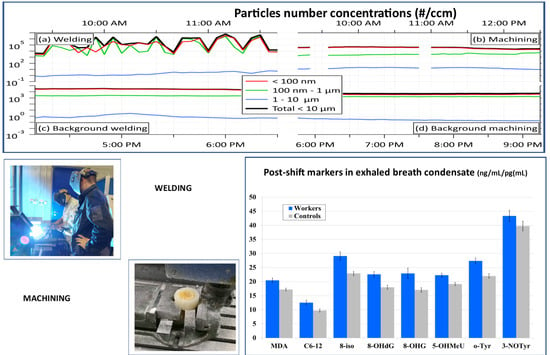
3.2.2. Number Concentrations
3.2.3. Elemental Composition
3.3. Oxidative Stress Markers
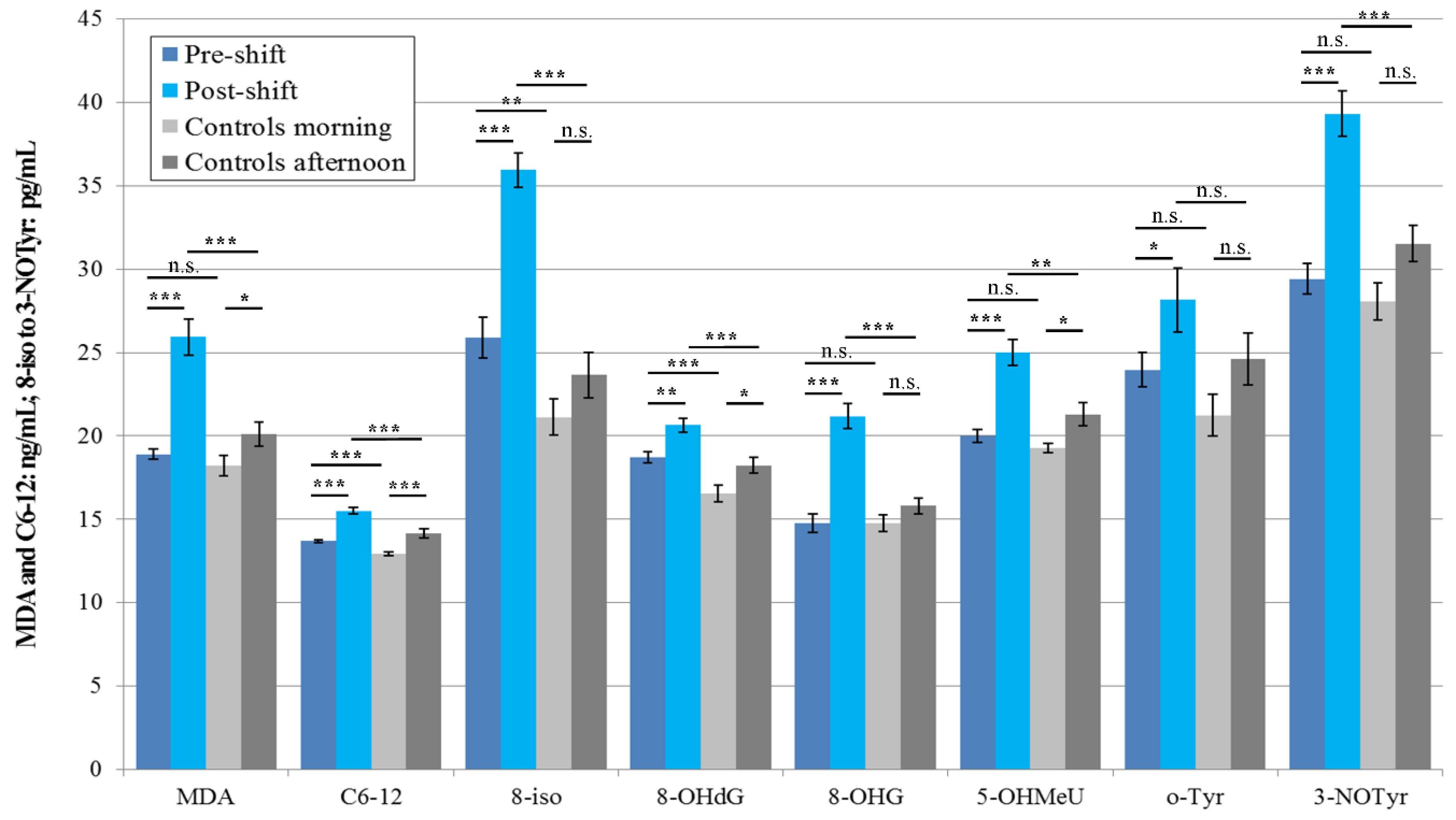
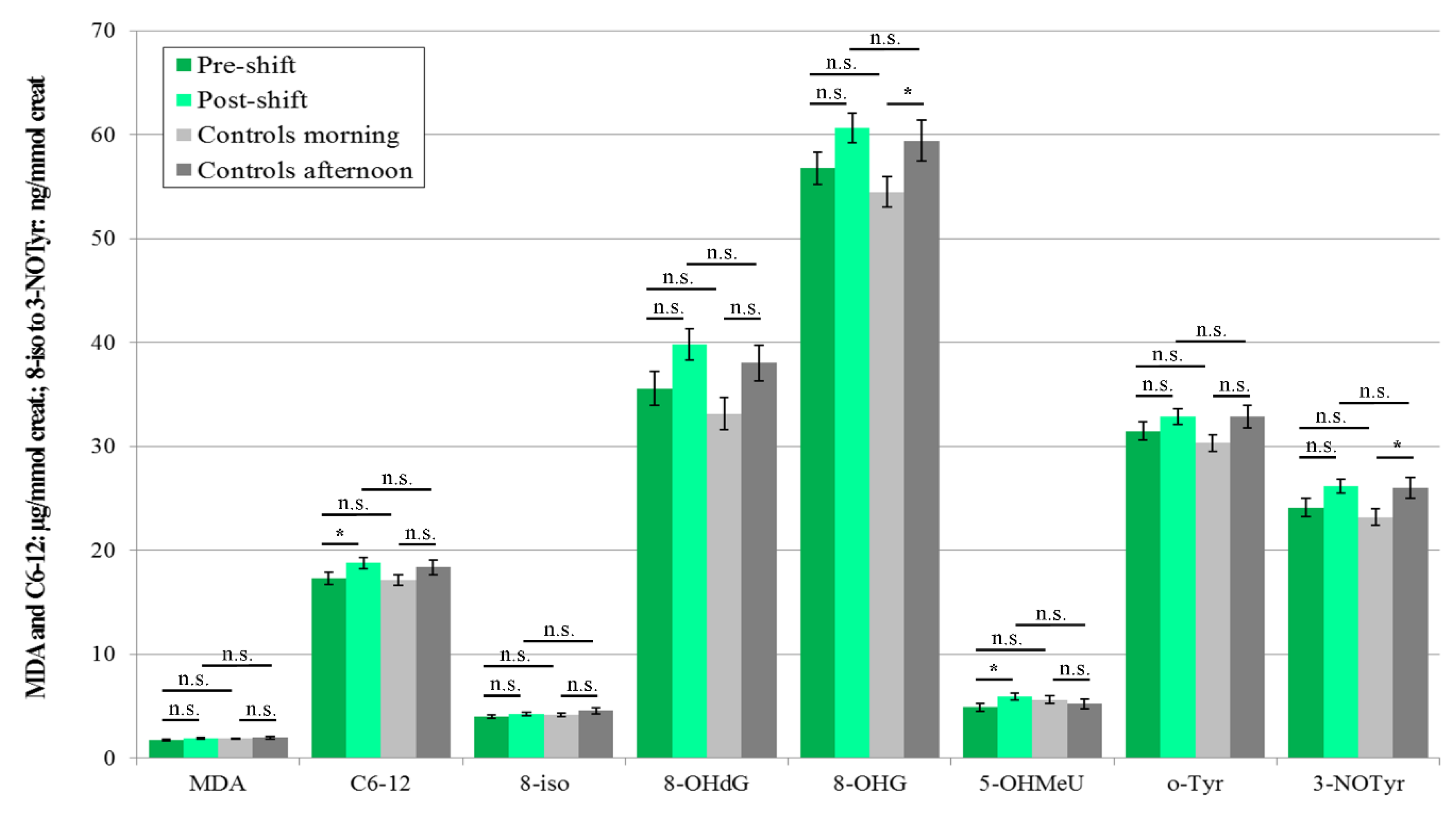
3.3.1. EBC
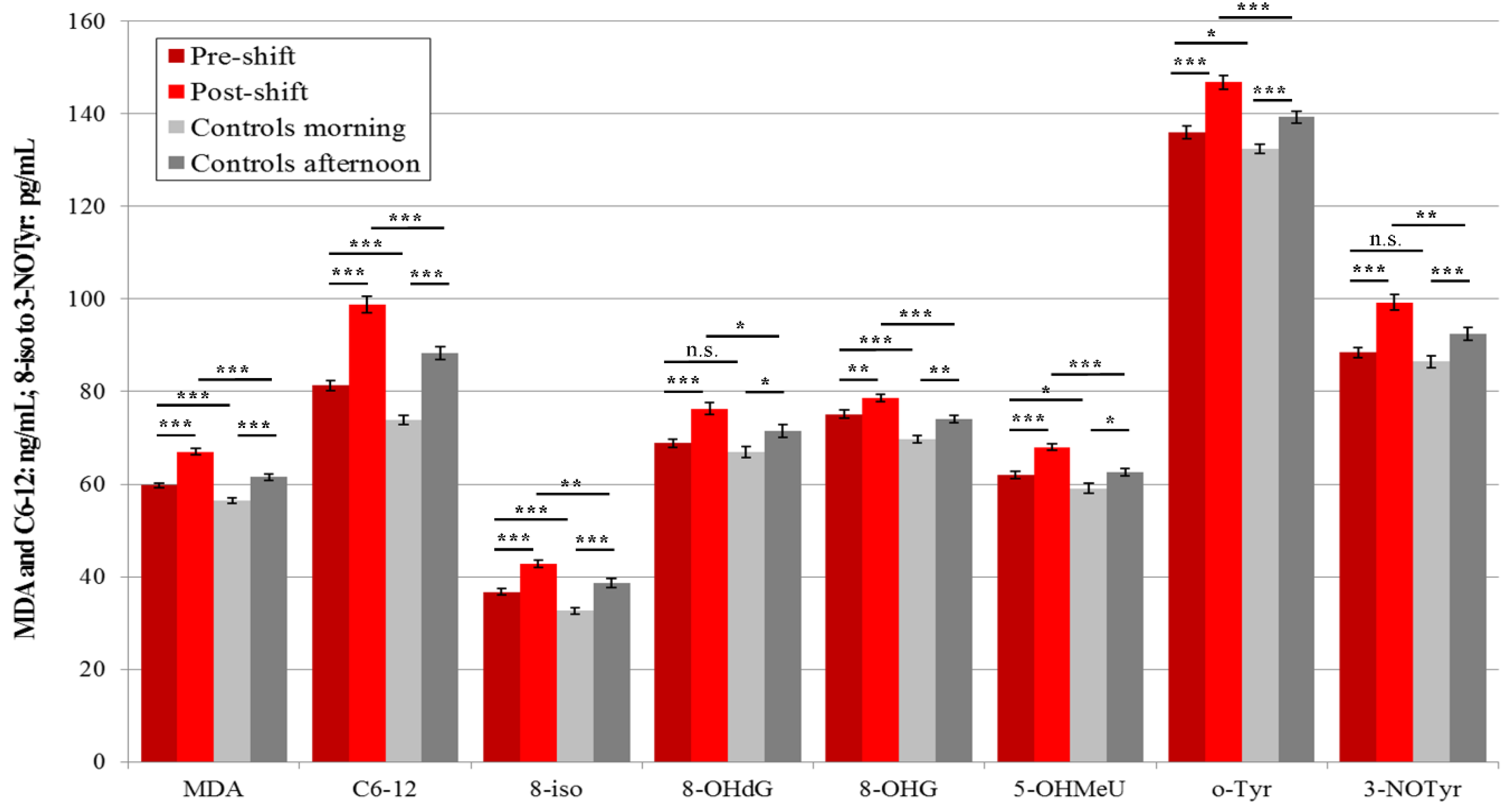
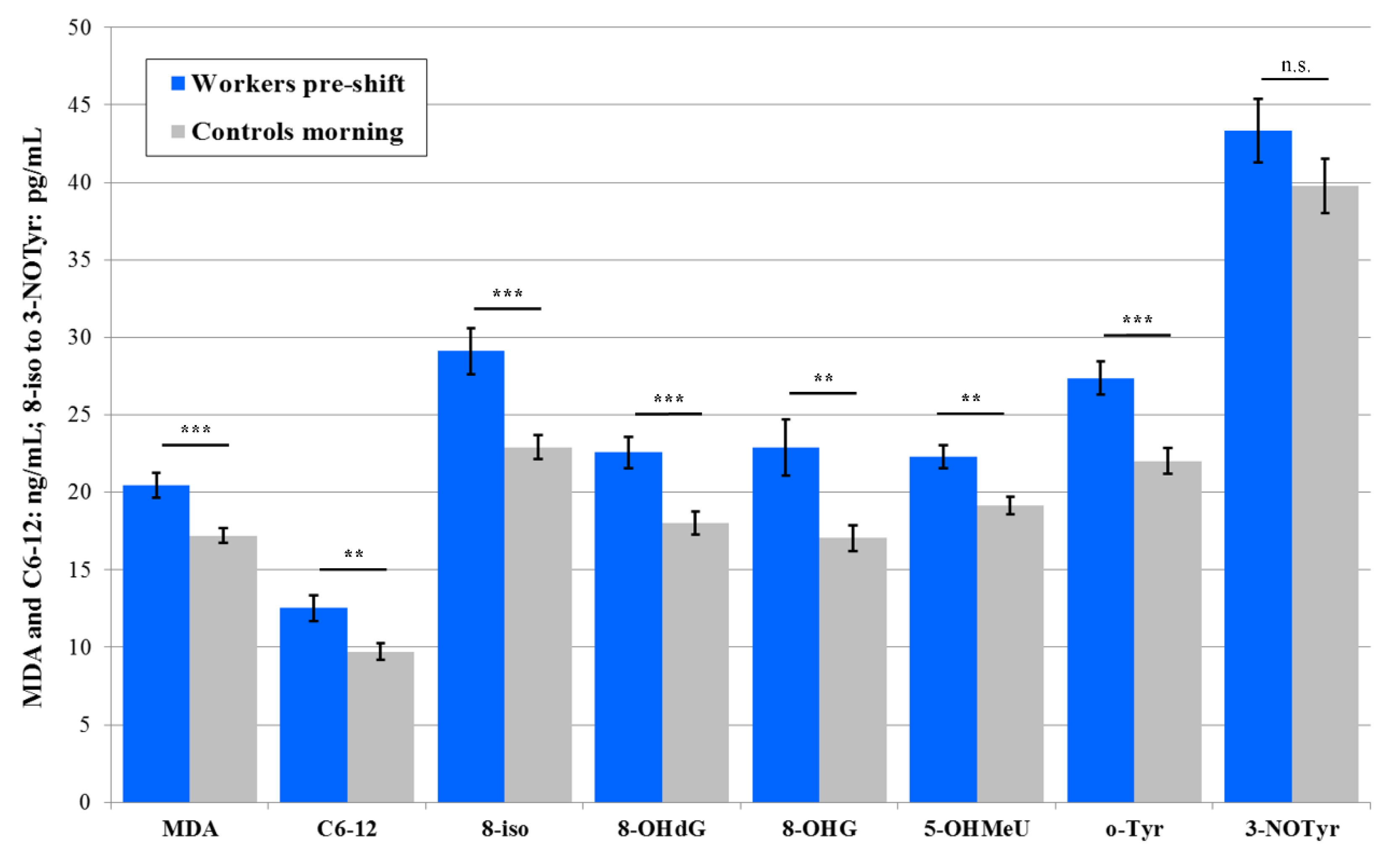
3.3.2. Plasma
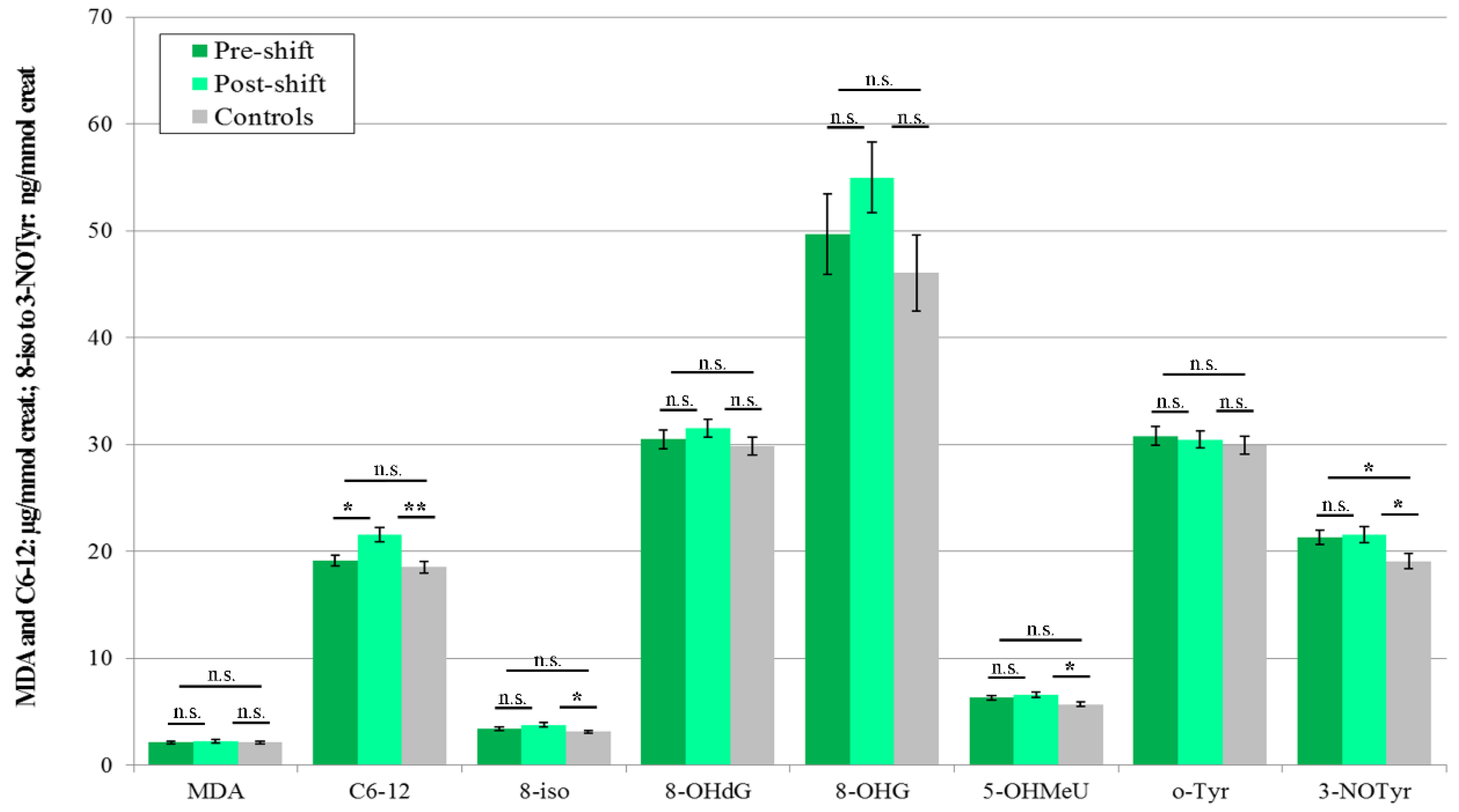
3.3.3. Urine
3.4. Acute Exposure Effect
3.5. Chronic Effect
3.5.1. Three Years’ Analysis of EBC Samples
3.5.2. Trends in 17 Repeated Examinations in 3 Biological Fluids
3.5.3. Occupational Factors
- Years of exposure
- Daily exposure length
- Latency since last exposure and pre-shift markers
3.6. Individual Characteristics
3.6.1. Workers Pre-Shift in 2017 and 2018
3.6.2. Workers Post-Shift in 2017 and 2018
3.6.3. Control Subjects in 2018
3.7. Correlation of One Marker in Different Fluid Samples
3.8. Environmental Contamination
4. Discussion
- Acute effect
- Chronic effect
- Duration of exposure and latency
- Workshops
- Methodology
- Age, gender, BMI
- Smoking, alcohol
- Food
- Physical activity
- Symptoms
- Diseases
- Circadian fluctuations
- Environmental contamination
5. Strengths and Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shoman, Y.; Wild, P.; Hemmendinger, M.; Graille, M.; Sauvain, J.J.; Hopf, N.B.; Canu, I.G. Reference Ranges of 8-Isoprostane Concentrations in Exhaled Breath Condensate (EBC): A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2020, 21, 3822. [Google Scholar] [CrossRef] [PubMed]
- Graille, M.; Wild, P.; Sauvain, J.J.; Hemmendinger, M.; Canu, I.G.; Hopf, N.B. Urinary 8-isoprostane as a biomarker for oxidative stress. A systematic review and meta-analysis. Toxicol. Lett. 2020, 328, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Graille, M.; Wild, P.; Sauvain, J.J.; Hemmendinger, M.; Canu, I.G.; Hopf, N.B. Urinary 8-OHdG as a Biomarker for Oxidative Stress: A Systematic Literature Review and Meta-Analysis. Int. J. Mol. Sci. 2020, 21, 3743. [Google Scholar] [CrossRef] [PubMed]
- Hemmendinger, M.; Wild, P.; Shoman, Y.; Graille, M.; Bergamaschi, E.; Hopf, N.; Canu, I.G. Reference ranges of oxidative stress biomarkers selected for non-invasive biological surveillance of nanotechnology workers: Study protocol and meta-analysis results for 8-OHdG in exhaled breath condensate. Toxicol. Lett. 2020, 327, 41–47. [Google Scholar] [CrossRef]
- Pinchuk, I.; Weber, D.; Kochlik, B.; Stuetz, W.; Toussaint, O.; Debacq-Chainiaux, F.; Dolle, M.E.T.; Jansen, E.H.J.M.; Gonos, E.S.; Sikora, E.; et al. Gender- and age-dependencies of oxidative stress, as detected based on the steady state concentrations of different biomarkers in the MARK-AGE study. Redox Biol. 2019, 24. [Google Scholar] [CrossRef]
- Lichtenberg, D.; Pinchuk, I.; Weber, D. Oxidative stress, as assayed by a single test, cannot be used as a diagnostic tool. BioFactors 2018, 44, 222–223. [Google Scholar] [CrossRef]
- Ghafari, J.; Moghadasi, N.; Shekaftik, S.O. Oxidative stress induced by occupational exposure to nanomaterials: A systematic review. Ind. Health 2020. [Google Scholar] [CrossRef]
- Miller, M.R.; Poland, C.A. Nanotoxicology: The Need for a Human Touch? Small 2020, 16. [Google Scholar] [CrossRef]
- Bradna, P.; Ondrackova, L.; Zdimal, V.; Navratil, T.; Pelclova, D. Detection of nanoparticles released at finishing of dental composite materials. Mon. Chem. 2017, 148, 531–537. [Google Scholar] [CrossRef]
- Larner, F.; Gulson, B.; McCall, M.; Oytam, Y.; Rehkamper, M. An inter-laboratory comparison of high precision stable isotope ratio measurements for nanoparticle tracing in biological samples. J. Anal. At. Spectrom. 2014, 29, 471–477. [Google Scholar] [CrossRef]
- Pelclova, D.; Navratil, T.; Fenclova, Z.; Vlckova, S. Markers of oxidative stress after three days of nanoTiO2 sunscreen use in humans: A pilot study. Cent. Eur. J. Public Health 2020, 28, S17–S21. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.L.; Ding, L.; Yao, C.J.; Li, C.C.; Xing, X.J.; Huang, Y.A.; Gu, T.J.; Wu, M.H. Toxic effects of metal oxide nanoparticles and their underlying mechanisms. Sci. China-Mater. 2017, 60, 93–108. [Google Scholar] [CrossRef]
- Pelclova, D.; Zdimal, V.; Schwarz, J.; Dvorackova, S.; Komarc, M.; Ondracek, J.; Kostejn, M.; Kacer, P.; Vlckova, S.; Fenclova, Z.; et al. Markers of Oxidative Stress in the Exhaled Breath Condensate of Workers Handling Nanocomposites. Nanomaterials 2018, 8, 611. [Google Scholar] [CrossRef] [PubMed]
- Pelclova, D.; Zdimal, V.; Komarc, M.; Vlckova, S.; Fenclova, Z.; Ondracek, J.; Schwarz, J.; Kostejn, M.; Kacer, P.; Dvorackova, S.; et al. Deep Airway Inflammation and Respiratory Disorders in Nanocomposite Workers. Nanomaterials 2018, 8, 731. [Google Scholar] [CrossRef] [PubMed]
- Horvath, I.; Barnes, P.J.; Loukides, S.; Sterk, P.J.; Hogman, M.; Olin, A.C.; Amann, A.; Antus, B.; Baraldi, E.; Bikov, A.; et al. A European Respiratory Society technical standard: Exhaled biomarkers in lung disease. Eur. Respir. J. 2017, 49. [Google Scholar] [CrossRef]
- Hung, T.D. New Generation of Geopolymer Composite for Fire-Resistance; IntechOpen: Rijeka, Croatia, 2011. [Google Scholar]
- Novotna, B.; Pelclova, D.; Rossnerova, A.; Zdimal, V.; Ondracek, J.; Lischkova, L.; Vlckova, S.; Fenclova, Z.; Klusackova, P.; Zavodna, T.; et al. The genotoxic effects in the leukocytes of workers handling nanocomposite materials. Mutagenesis 2020, 35, 331–340. [Google Scholar] [CrossRef]
- Rossnerova, A.; Honkova, K.; Pelclova, D.; Zdimal, V.; Hubacek, J.A.; Chvojkova, I.; Vrbova, K.; Rossner, P.; Topinka, J.; Vlckova, S.; et al. DNA Methylation Profiles in a Group of Workers Occupationally Exposed to Nanoparticles. Int. J. Mol. Sci. 2020, 21, 2420. [Google Scholar] [CrossRef]
- Stefancova, L.; Schwarz, J.; Makela, T.; Hillamo, R.; Smolik, J. Comprehensive Characterization of Original 10-Stage and 7-Stage Modified Berner Type Impactors. Aerosol Sci. Technol. 2011, 45, 88–100. [Google Scholar] [CrossRef]
- Syslova, K.; Kacer, P.; Kuzma, M.; Klusackova, P.; Fenclova, Z.; Lebedova, J.; Pelclova, D. Determination of 8-iso-prostaglandin F(2alpha) in exhaled breath condensate using combination of immunoseparation and LC-ESI-MS/MS. J. Chromatogr. B 2008, 867, 8–14. [Google Scholar] [CrossRef]
- Syslova, K.; Kacer, P.; Kuzma, M.; Pankracova, A.; Fenclova, Z.; Vlckova, S.; Lebedova, J.; Pelclova, D. LC-ESI-MS/MS method for oxidative stress multimarker screening in the exhaled breath condensate of asbestosis/silicosis patients. J. Breath Res. 2010, 4, 17104. [Google Scholar] [CrossRef]
- Syslova, K.; Bohmova, A.; Mikoska, M.; Kuzma, M.; Pelclova, D.; Kacer, P. Multimarker screening of oxidative stress in aging. Oxidative Med. Cell. Longev. 2014, 2014, 562860. [Google Scholar] [CrossRef] [PubMed]
- Neprasova, M.; Maixnerova, D.; Novak, J.; Reily, C.; Julian, B.A.; Boron, J.; Novotny, P.; Suchanek, M.; Tesar, V.; Kacer, P. Toward Noninvasive Diagnosis of IgA Nephropathy: A Pilot Urinary Metabolomic and Proteomic Study. Dis. Markers 2016. [Google Scholar] [CrossRef] [PubMed]
- Delanghe, J.R.; Speeckaert, M.M. Creatinine determination according to Jaffe-what does it stand for? NDT Plus 2011, 4, 83–86. [Google Scholar] [CrossRef] [PubMed]
- Khanna, P.; Ong, C.; Bay, B.H.; Baeg, G.H. Nanotoxicity: An Interplay of Oxidative Stress, Inflammation and Cell Death. Nanomaterials 2015, 5, 1163–1180. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.W.; Cambre, M.; Lee, H.J. The Toxicity of Nanoparticles Depends on Multiple Molecular and Physicochemical Mechanisms. Int. J. Mol. Sci. 2017, 18, 2702. [Google Scholar] [CrossRef]
- Runa, S.; Hussey, M.; Payne, C.K. Nanoparticle-Cell Interactions: Relevance for Public Health. J. Phys. Chem. B 2018, 122, 1009–1016. [Google Scholar] [CrossRef]
- Riccelli, M.G.; Goldoni, M.; Andreoli, R.; Mozzoni, P.; Pinelli, S.; Alinovi, R.; Selis, L.; Mutti, A.; Corradi, M. Biomarkers of exposure to stainless steel tungsten inert gas welding fumes and the effect of exposure on exhaled breath condensate. Toxicol. Lett. 2018, 292, 108–114. [Google Scholar] [CrossRef]
- Wu, C.M.; Adetona, A.; Song, C.; Naeher, L.; Adetona, O. Measuring acute pulmonary responses to occupational wildland fire smoke exposure using exhaled breath condensate. Arch. Environ. Occup. Health 2020, 75, 65–69. [Google Scholar] [CrossRef]
- Graczyk, H.; Lewinski, N.; Zhao, J.Y.; Sauvain, J.J.; Suarez, G.; Wild, P.; Danuser, B.; Riediker, M. Increase in oxidative stress levels following welding fume inhalation: A controlled human exposure study. Part. Fibre Toxicol. 2016, 13. [Google Scholar] [CrossRef]
- Pelclova, D.; Zdimal, V.; Kacer, P.; Fenclova, Z.; Vlckova, S.; Syslova, K.; Navratil, T.; Schwarz, J.; Zikova, N.; Barosova, H.; et al. Oxidative stress markers are elevated in exhaled breath condensate of workers exposed to nanoparticles during iron oxide pigment production. J. Breath Res. 2016, 10, 16004. [Google Scholar] [CrossRef]
- Pelclova, D.; Zdimal, V.; Kacer, P.; Zikova, N.; Komarc, M.; Fenclova, Z.; Vlckova, S.; Schwarz, J.; Makes, O.; Syslova, K.; et al. Markers of lipid oxidative damage in the exhaled breath condensate of nano TiO2 production workers. Nanotoxicology 2017, 11, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Pelclova, D.; Zdimal, V.; Fenclova, Z.; Vlckova, S.; Turci, F.; Corazzari, I.; Kacer, P.; Schwarz, J.; Zikova, N.; Makes, O.; et al. Markers of oxidative damage of nucleic acids and proteins among workers exposed to TiO2 (nano) particles. Occup. Environ. Med. 2016, 73, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Pelclova, D.; Zdimal, V.; Kacer, P.; Vlckova, S.; Fenclova, Z.; Navratil, T.; Komarc, M.; Schwarz, J.; Zikova, N.; Makes, O.; et al. Markers of nucleic acids and proteins oxidation among office workers exposed to air pollutants including (nano) TiO2 particles. Neuro Endocrinol. Lett. 2016, 37, 13–16. [Google Scholar] [PubMed]
- Pelclova, D.; Zdimal, V.; Kacer, P.; Komarc, M.; Fenclova, Z.; Vlckova, S.; Zikova, N.; Schwarz, J.; Makes, O.; Navratil, T.; et al. Markers of lipid oxidative damage among office workers exposed intermittently to air pollutants including nanoTiO2 particles. Rev. Environ. Health 2017, 32, 193–200. [Google Scholar] [CrossRef]
- Pelclova, D.; Fenclova, Z.; Syslova, K.; Vlckova, S.; Lebedova, J.; Pecha, O.; Belacek, J.; Navratil, T.; Kuzma, M.; Kacer, P. Oxidative Stress Markers in Exhaled Breath Condensate in Lung Fibroses Are Not Significantly Affected by Systemic Diseases. Ind. Health 2011, 49, 746–754. [Google Scholar] [CrossRef]
- Pelclova, D.; Navratil, T.; Kacerova, T.; Zamostna, B.; Fenclova, Z.; Vlckova, S.; Kacer, P. NanoTiO2 Sunscreen Does Not Prevent Systemic Oxidative Stress Caused by UV Radiation and a Minor Amount of NanoTiO2 is Absorbed in Humans. Nanomaterials 2019, 9, 888. [Google Scholar] [CrossRef]
- Rossnerova, A.; Pelclova, D.; Zdimal, V.; Rossner, P.; Elzeinova, F.; Vrbova, K.; Topinka, J.; Schwarz, J.; Ondracek, J.; Kostejn, M.; et al. The repeated cytogenetic analysis of subjects occupationally exposed to nanoparticles: A pilot study. Mutagenesis 2019, 34, 253–263. [Google Scholar] [CrossRef]
- Rossnerova, A.; Pokorna, M.; Svecova, V.; Sram, R.J.; Topinka, J.; Zolzer, F.; Rossner, P. Adaptation of the human population to the environment: Current knowledge, clues from Czech cytogenetic and “omics” biomonitoring studies and possible mechanisms. Mutat. Res. Rev. Mutat. Res. 2017, 773, 188–203. [Google Scholar] [CrossRef]
- Weber, D.; Stuetz, W.; Toussaint, O.; Debacq-Chainiaux, F.; Dolle, M.E.T.; Jansen, E.; Gonos, E.S.; Franceschi, C.; Sikora, E.; Hervonen, A.; et al. Associations between Specific Redox Biomarkers and Age in a Large European Cohort: The MARK-AGE Project. Oxidative Med. Cell. Longev. 2017, 2017. [Google Scholar] [CrossRef]
- Hofer, T.; Fontana, L.; Anton, S.D.; Weiss, E.P.; Villareal, D.; Malayappan, B.; Leeuwenburgh, C. Long-term effects of caloric restriction or exercise on DNA and RNA oxidation levels in white blood cells and urine in humans. Rejuvenation Res. 2008, 11, 793–799. [Google Scholar] [CrossRef]
- Brieger, K.; Schiavone, S.; Miller, F.J.; Krause, K.H. Reactive oxygen species: From health to disease. Swiss Med. Wkly. 2012, 142. [Google Scholar] [CrossRef] [PubMed]
- Kurti, S.P.; Emerson, S.R.; Rosenkranz, S.K.; Teeman, C.S.; Emerson, E.M.; Cull, B.J.; Smith, J.R.; Harms, C.A. Post-prandial systemic 8-isoprostane increases after consumption of moderate and high-fat meals in insufficiently active males. Nutr. Res. 2017, 39, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Kullmann, T.; Barta, I.; Antus, B.; Horvath, I. Drinking influences exhaled breath condensate acidity. Lung 2008, 186, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Kanabrocki, E.L.; Murray, D.; Hermida, R.C.; Scott, G.S.; Bremner, W.F.; Ryan, M.D.; Ayala, D.E.; Third, J.L.H.C.; Shirazi, P.; Nemchausky, B.A.; et al. Circadian variation in oxidative stress markers in healthy and type II diabetic men. Chronobiol. Int. 2002, 19, 423–439. [Google Scholar] [CrossRef] [PubMed]
- Morissette, M.C.; Murray, N.; Turmel, J.; Milot, J.; Boulet, L.P.; Bougault, V. Increased exhaled breath condensate 8-isoprostane after a swimming session in competitive swimmers. Eur. J. Sport Sci. 2016, 16, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Kurti, S.P.; Emerson, S.R.; Smith, J.R.; Rosenkranz, S.K.; Alexander, S.A.; Lovoy, G.M.; Harms, C.A. Older women exhibit greater airway 8-isoprostane responses to strenuous exercise compared with older men and younger controls. Appl. Physiol. Nutr. Metab. 2018, 43, 497–503. [Google Scholar] [CrossRef]
- Finkler, M.; Lichtenberg, D.; Hochman, A.; Pinchuk, I. The relationship between oxidative stress and exercise. Free Radic. Biol. Med. 2012, 53, S202–S203. [Google Scholar] [CrossRef]
- Kuban, P.; Foret, F. Exhaled breath condensate: Determination of non-volatile compounds and their potential for clinical diagnosis and monitoring. A review. Anal. Chim. Acta 2013, 805, 1–18. [Google Scholar] [CrossRef]
- Pelclova, D.; Fenclova, Z.; Vlckova, S.; Klusackova, P.; Lebedova, J.; Syslova, K.; Belacek, J.; Kuzma, M.; Navratil, T.; Zakharov, S.; et al. Occupational asthma follow-up—Which markers are elevated in exhaled breath condensate and plasma? Int. J. Occup. Med. Environ. Health 2014, 27, 206–215. [Google Scholar] [CrossRef]
- Mallard, A.R.; Hollekim-Strand, S.M.; Ingul, C.B.; Coombes, J.S. High day-to-day and diurnal variability of oxidative stress and inflammation biomarkers in people with type 2 diabetes mellitus and healthy individuals. Redox Rep. 2020, 25, 64–69. [Google Scholar] [CrossRef]
- Wilking, M.; Ndiaye, M.; Mukhtar, H.; Ahmad, N. Circadian Rhythm Connections to Oxidative Stress: Implications for Human Health. Antioxid. Redox Signal. 2013, 19, 192–208. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Moral, M.P.; Kannan, K. How stable is oxidative stress level? An observational study of intra- and inter-individual variability in urinary oxidative stress biomarkers of DNA, proteins, and lipids in healthy individuals. Environ. Int. 2019, 123, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Barregard, L.; Moller, P.; Henriksen, T.; Mistry, V.; Koppen, G.; Rossner, P.; Sram, R.J.; Weimann, A.; Poulsen, H.E.; Nataf, R.; et al. Human and Methodological Sources of Variability in the Measurement of Urinary 8-Oxo-7,8-dihydro-2′-deoxyguanosine. Antioxid. Redox Signal. 2013, 18, 2377–2391. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.C.; Zhu, T.; Kipen, H.; Wang, G.F.; Hu, M.; Ohman-Strickland, P.; Lu, S.E.; Zhang, L.; Wang, Y.D.; Zhu, P.; et al. Malondialdehyde in exhaled breath condensate and urine as a biomarker of air pollution induced oxidative stress. J. Expo. Sci. Environ. Epidemiol. 2013, 23, 322–327. [Google Scholar] [CrossRef]
- Calderon-Garciduenas, L.; Herrera-Soto, A.; Jury, N.; Maher, B.A.; Gonzalez-Maciel, A.; Reynoso-Robles, R.; Ruiz-Rudolph, P.; van Zundert, B.; Varela-Nallar, L. Reduced repressive epigenetic marks, increased DNA damage and Alzheimer’s disease hallmarks in the brain of humans and mice exposed to particulate urban air pollution. Environ. Res. 2020, 183. [Google Scholar] [CrossRef]
- Iavicoli, I.; Leso, V.; Schulte, P.A. Biomarkers of susceptibility: State of the art and implications for occupational exposure to engineered nanomaterials. Toxicol. Appl. Pharmacol. 2016, 299, 112–124. [Google Scholar] [CrossRef]
- Canu, I.G.; Schulte, P.A.; Riediker, M.; Fatkhutdinova, L.; Bergamaschi, E. Methodological, political and legal issues in the assessment of the effects of nanotechnology on human health. J. Epidemiol. Community Health 2018, 72, 148–153. [Google Scholar] [CrossRef]
- Mendoza, R.P.; Brown, J.M. Engineered nanomaterials and oxidative stress: Current understanding and future challenges. Curr. Opin. Toxicol. 2019, 13, 74–80. [Google Scholar] [CrossRef]
- Liou, S.H.; Tsai, C.S.J.; Pelclova, D.; Schubauer-Berigan, M.K.; Schulte, P.A. Assessing the first wave of epidemiological studies of nanomaterial workers. J. Nanoparticle Res. 2015, 17. [Google Scholar] [CrossRef]
- Schulte, P.A.; Iavicoli, I.; Rantanen, J.H.; Dahmann, D.; Iavicoli, S.; Pipke, R.; Canu, I.G.; Boccuni, F.; Ricci, M.; Polci, M.L.; et al. Assessing the protection of the nanomaterial workforce. Nanotoxicology 2016, 10, 1013–1019. [Google Scholar] [CrossRef]
- Murgia, N.; Barregard, L.; Sallsten, G.; Almstrand, A.C.; Montuschi, P.; Ciabattoni, G.; Olin, A.C. 8-isoprostane in exhaled breath condensate after experimental exposure to wood smoke in humans. J. Biol. Regul. Homeost. Agents 2016, 30, 263–270. [Google Scholar] [PubMed]


| 2016 | 2017 | 2018 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Workers | Controls | pa | Workers | Controls | pa | Workers | Controls | pa | |
| N (male/female) | 20 (15/5) | 21 (15/6) | 0.796 | 20 (13/7) | 20 (13/7) | 1 | 21 (16/5) | 18 (12/6) | 1 |
| Age (years) | 42 ± 11 | 38.7 ± 9.1 | 0.334 | 39 ± 11 | 39.9 ± 7.3 | 0.647 | 40 ± 12 | 45 ± 13 | 0.253 |
| Mean ± SD (range) | (29–63) | (20–55) | (23–64) | (27–55) | (24–65) | (21–72) | |||
| BMI (kg/m2) | 28.0 ± 6.2 | 25.0 ± 4.7 | 0.1 | 24.6 ± 4.6 | 26.1 ± 4.6 | 0.294 | 27.0 ± 5.3 | 26.5 ± 5.4 | 0.789 |
| Mean ± SD (range) | (18–42) | (18–37) | (18–34) | (20–37) | (19–37) | (20–39) | |||
| Smoking | 1 | 4 | 0.169 | 3 | 3 | 1 | 3 | 3 | 0.91 |
| Alcohol (occasionally) | 18 | 16 | 0.24 | 16 | 18 | 0.389 | 18 | 16 | 0.842 |
| Exposure Time | 2016 | 2017 | 2018 |
|---|---|---|---|
| Mean ± SD (Range) | Mean ± SD (Range) | Mean ± SD (Range) | |
| Total (years) | 18 ± 10 (5–40) | 12.2 ± 9.3 (2–31) | 13.9 ± 9.4 (1–32) |
| Usually per day (min) | 101 ± 60 (30–240) | 128 ± 87 (30–360) | 123 ± 16 (60–270) |
| On monitoring day (min) | 156 ± 62 (60–330) | 204 ± 55 (150–360) ** | 156 ± 7.4 (120–240) |
| Latency since last exposure (hour) | 266 ± 458 (14–1440) | 81 ± 125 (15–504) | 160 ± 190 (14–720) |
| Operation: Machining | Size Range (nm) | Sum of Elements Analyzed by SEM/EDS (µg/m3) |
|---|---|---|
| 2016 | 25–100 | 0.143 |
| 2017 | 25–100 | 0.550 |
| 2018 | 25–100 | 0.801 |
| Medians of Total Number Concentrations of PM Fractions per cm3 | |||||
|---|---|---|---|---|---|
| Operations and Backgrounds | <100 nm | 100 nm–1 µm | 1–10 µm | Total <10 µm | |
| 2016 | MAG welding—workshop 1 | 4.88 × 104 | 7.29 × 104 | 3.33 × 101 | 1.22 × 105 |
| Smelting—workshop 1 | 4.60 × 104 | 2.60 × 103 | 3.26 | 4.86 × 104 | |
| Background—workshop 1 | 2.01 × 104 | 6.72 × 102 | 1.05 | 2.08 × 104 | |
| Machining—workshop 2 | 3.22 × 105 | 2.04 × 105 | 2.96 × 101 | 5.26 × 105 | |
| Background—workshop 2 | 1.16 × 105 | 1.68 × 105 | 1.86 × 101 | 2.84 × 105 | |
| 2017 | Machining—workshop 2 | 8.99 × 104 | 3.95 × 103 | 4.75 × 101 | 9.39 × 104 |
| Background—workshop 2 | 4.68 × 103 | 4.10 × 103 | 6.04 × 10−1 | 8.77 × 103 | |
| 2018 | MAG welding—workshop 1 | 5.52 × 103 | 3.79 × 103 | 1.31 | 9.31 × 103 |
| Background—workshop 1 | 6.83 × 103 | 2.37 × 103 | 4.98 × 10−1 | 9.21 × 103 | |
| Machining—workshop 2 | 2.61 × 104 | 3.04 × 103 | 1.85 × 101 | 2.91 × 104 | |
| Background—workshop 2 | 3.64 × 103 | 1.68 × 103 | 2.38 × 10−1 | 5.32 × 103 | |
| Year | Processes | Proportion of PM Fractions (number %) | ||
|---|---|---|---|---|
| <100 nm | 100 nm–1 µm | 1 μm–10 µm | ||
| 2016 | MAG Welding (workshop 1) | 40.13 | 59.85 | 0.02 |
| Smelting (workshop 1) | 94.64 | 5.35 | 0.01 | |
| Background (workshop 1) | 96.76 | 3.23 | 0.00 | |
| Machining (workshop 2) | 61.23 | 38.76 | 0.01 | |
| Background (workshop 2) | 40.89 | 59.10 | 0.01 | |
| 2017 | Machining (workshop 2) | 95.74 | 4.20 | 0.05 |
| Background (workshop 2) | 53.30 | 46.69 | 0.01 | |
| 2018 | MAG Welding (workshop 1) | 59.29 | 40.70 | 0.01 |
| Background (workshop 1) | 74.24 | 25.76 | 0.00 | |
| Machining (workshop 2) | 89.50 | 10.43 | 0.07 | |
| Background (workshop 2) | 68.42 | 31.58 | 0.00 | |
| Year | Fe | Mn | Al | Si | S | F | Na | Cl | K | Ca | Cr | Total | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2016 | Welding | 84.70 | 8.46 | 0.00 | 5.35 | 0.61 | 0.23 | 0.44 | 0.20 | 0.00 | 0.00 | 0.00 | 100.00 |
| Smelting | 80.13 | 7.48 | 0.36 | 4.40 | 1.24 | 0.07 | 2.85 | 1.90 | 1.58 | 0.00 | 0.00 | 100.00 | |
| Machining | 45.86 | 0.00 | 5.30 | 11.91 | 12.11 | 4.11 | 0.00 | 20.70 | 0.00 | 0.00 | 0.00 | 100.00 | |
| 2017 | Machining | 0.00 | 0.00 | 21.09 | 40.92 | 15.94 | 0.00 | 16.70 | 5.35 | 0.00 | 0.00 | 0.00 | 100.00 |
| 2018 | Machining | 68.00 | 11.86 | 1.04 | 3.64 | 2.65 | 0.00 | 0.87 | 0.00 | 0.00 | 0.00 | 11.94 | 100.00 |
| Welding | 40.74 | 3.99 | 0.55 | 2.95 | 1.59 | 28.73 | 4.39 | 0.67 | 8.89 | 7.49 | 0.00 | 100.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pelclova, D.; Zdimal, V.; Komarc, M.; Schwarz, J.; Ondracek, J.; Ondrackova, L.; Kostejn, M.; Vlckova, S.; Fenclova, Z.; Dvorackova, S.; et al. Three-Year Study of Markers of Oxidative Stress in Exhaled Breath Condensate in Workers Producing Nanocomposites, Extended by Plasma and Urine Analysis in Last Two Years. Nanomaterials 2020, 10, 2440. https://doi.org/10.3390/nano10122440
Pelclova D, Zdimal V, Komarc M, Schwarz J, Ondracek J, Ondrackova L, Kostejn M, Vlckova S, Fenclova Z, Dvorackova S, et al. Three-Year Study of Markers of Oxidative Stress in Exhaled Breath Condensate in Workers Producing Nanocomposites, Extended by Plasma and Urine Analysis in Last Two Years. Nanomaterials. 2020; 10(12):2440. https://doi.org/10.3390/nano10122440
Chicago/Turabian StylePelclova, Daniela, Vladimir Zdimal, Martin Komarc, Jaroslav Schwarz, Jakub Ondracek, Lucie Ondrackova, Martin Kostejn, Stepanka Vlckova, Zdenka Fenclova, Stepanka Dvorackova, and et al. 2020. "Three-Year Study of Markers of Oxidative Stress in Exhaled Breath Condensate in Workers Producing Nanocomposites, Extended by Plasma and Urine Analysis in Last Two Years" Nanomaterials 10, no. 12: 2440. https://doi.org/10.3390/nano10122440
APA StylePelclova, D., Zdimal, V., Komarc, M., Schwarz, J., Ondracek, J., Ondrackova, L., Kostejn, M., Vlckova, S., Fenclova, Z., Dvorackova, S., Lischkova, L., Klusackova, P., Kolesnikova, V., Rossnerova, A., & Navratil, T. (2020). Three-Year Study of Markers of Oxidative Stress in Exhaled Breath Condensate in Workers Producing Nanocomposites, Extended by Plasma and Urine Analysis in Last Two Years. Nanomaterials, 10(12), 2440. https://doi.org/10.3390/nano10122440