The Antifungal Mechanism of Amphotericin B Elucidated in Ergosterol and Cholesterol-Containing Membranes Using Neutron Reflectometry
Abstract
1. Introduction
2. Materials and Methods
3. Results
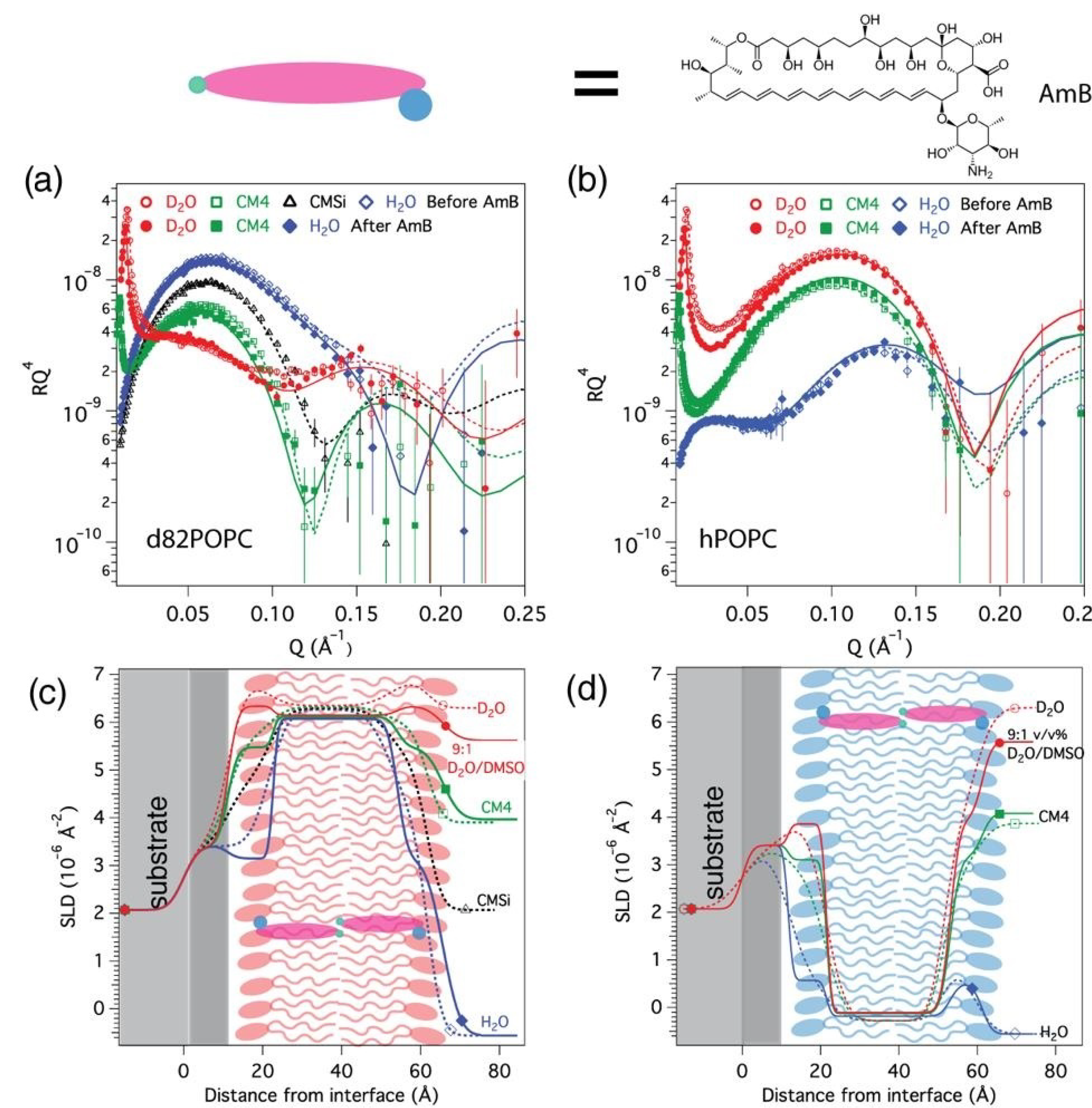
3.1. AmB Interaction with Sterol-Free Bilayers: hPOPC and d82POPC
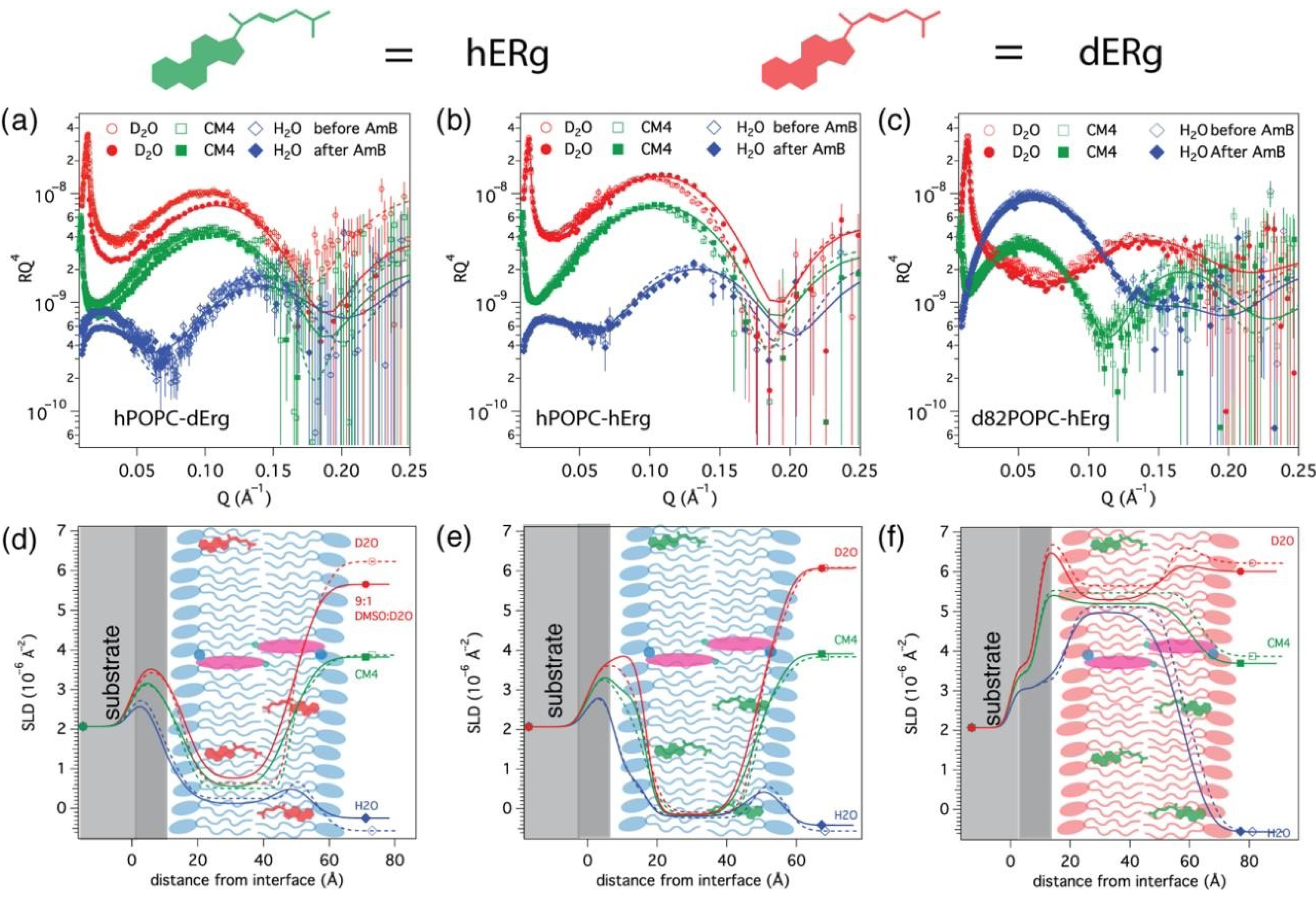
3.2. AmB Interaction with Ergosterol-Containing Bilayers
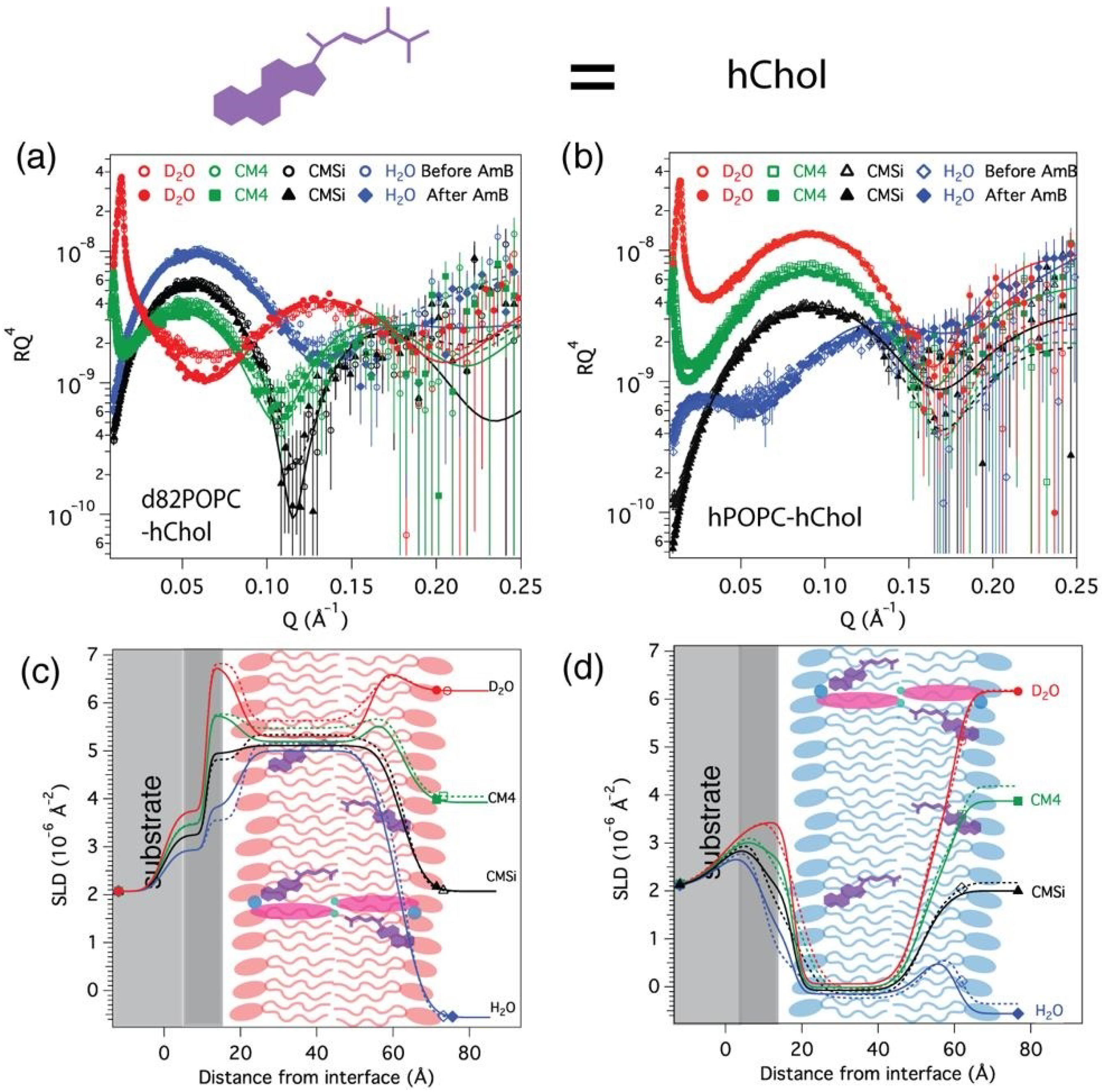
3.3. AmB Interaction with Cholesterol-Containing Bilayers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gallis, H.A.; Drew, R.H.; Pickard, W.W. Amphotericin B: 30 Years of Clinical Experience. Clin. Infect. Dis. 1990, 12, 308–329. [Google Scholar] [CrossRef] [PubMed]
- Hartsel, S.; Bolard, J. Amphotericin B: New life for an old drug. Trends Pharmacol. Sci. 1996, 17, 445–449. [Google Scholar] [CrossRef]
- Hamill, R.J. Amphotericin B formulations: A comparative review of efficacy and toxicity. Drugs 2013, 73, 919–934. [Google Scholar] [CrossRef] [PubMed]
- Matsumori, N.; Tahara, K.; Yamamoto, H.; Morooka, A.; Doi, M.; Oishi, T.; Murata, M. Direct interaction between amphotericin B and ergosterol in lipid bilayers as revealed by 2H NMR spectroscopy. J. Am. Chem. Soc. 2009, 131, 11855–11860. [Google Scholar] [CrossRef]
- Gray, K.C.; Palacios, D.S.; Dailey, I.; Endo, M.M.; Uno, B.E.; Wilcock, B.C.; Burke, M.D. Amphotericin primarily kills yeast by simply binding ergosterol. Proc. Natl. Acad. Sci. USA 2012, 109, 2234–2239. [Google Scholar] [CrossRef]
- Gabrielska, J.; Gagos, M.; Gubernator, J.; Gruszecki, W.I. Binding of antibiotic amphotericin B to lipid membranes: A 1H NMR study. FEBS Lett. 2006, 580, 2677–2685. [Google Scholar] [CrossRef]
- Ciesielski, F.; Griffin, D.C.; Loraine, J.; Rittig, M.; Delves-Broughton, J.; Bonev, B.B. Recognition of Membrane Sterols by Polyene Antifungals Amphotericin B and Natamycin, A 13C MAS NMR Study. Front. Cell Dev. Biol. 2016, 4, 212–253. [Google Scholar] [CrossRef]
- Huang, W.; Zhang, Z.; Han, X.; Tang, J.; Wang, J.; Dong, S.; Wang, E. Ion Channel Behavior of Amphotericin B in Sterol-Free and Cholesterol- or Ergosterol-Containing Supported Phosphatidylcholine Bilayer Model Membranes Investigated by Electrochemistry and Spectroscopy. Biophys. J. 2002, 83, 3245–3255. [Google Scholar] [CrossRef]
- Baginski, M.; Resat, H.; Borowski, E. Comparative molecular dynamics simulations of amphotericin B–cholesterol/ergosterol membrane channels. Biochim. Biophys. Acta (BBA) Biomembr. 2002, 1567, 63–78. [Google Scholar] [CrossRef]
- Venegas, B.; González-Damián, J.; Celis, H.; Ortega-Blake, I. Amphotericin B Channels in the Bacterial Membrane: Role of Sterol and Temperature. Biophys. J. 2003, 85, 2323–2332. [Google Scholar] [CrossRef][Green Version]
- Matsuoka, S.; Murata, M. Membrane permeabilizing activity of amphotericin B is affected by chain length of phosphatidylcholine added as minor constituent. Biochim. Biophys. Acta (BBA) Biomembr. 2003, 1617, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, S.; Ikeuchi, H.; Matsumori, N.; Murata, M. Dominant formation of a single-length channel by amphotericin B in dimyristoylphosphatidylcholine membrane evidenced by 13C-31P rotational echo double resonance. Biochemistry 2005, 44, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Mouri, R.; Konoki, K.; Matsumori, N.; Oishi, T.; Murata, M. Complex formation of amphotericin B in sterol-containing membranes as evidenced by surface plasmon resonance. Biochemistry 2008, 47, 7807–7815. [Google Scholar] [CrossRef] [PubMed]
- Romero, E.A.; Valdivieso, E.; Cohen, B.E. Formation of Two Different Types of Ion Channels by Amphotericin B in Human Erythrocyte Membranes. J. Membr. Biol. 2009, 230, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Anderson, T.M.; Clay, M.C.; Cioffi, A.G.; Diaz, K.A.; Hisao, G.S.; Tuttle, M.D.; Nieuwkoop, A.J.; Comellas, G.; Maryum, N.; Wang, S.; et al. Amphotericin forms an extramembranous and fungicidal sterol sponge. Nat. Chem. Biol. 2014, 10, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Dufourc, E.J. Sterols and membrane dynamics. J. Chem. Biol. 2008, 1, 63–77. [Google Scholar] [CrossRef]
- Chudzik, B.; Koselski, M.; Czuryło, A.; Trębacz, K.; Gagoś, M. A new look at the antibiotic amphotericin B effect on Candida albicans plasma membrane permeability and cell viability functions. Eur. Biophys. J. 2015, 44, 77–90. [Google Scholar] [CrossRef]
- Foglia, F.; Drake, A.F.; Terry, A.E.; Rogers, S.E.; Lawrence, M.J.; Barlow, D.J. Small-angle neutron scattering studies of the effects of amphotericin B on phospholipid and phospholipid–sterol membrane structure. Biochim. Biophys. Acta (BBA) Biomembr. 2011, 1808, 1574–1580. [Google Scholar] [CrossRef]
- Foglia, F.; Lawrence, M.J.; Demeė, B.; Fragneto, G.; Barlow, D. Neutron diffraction studies of the interaction between amphotericin B and lipid-sterol model membranes. Sci. Rep. 2012, 2, 806–809. [Google Scholar] [CrossRef]
- Foglia, F.; Fragneto, G.; Clifton, L.A.; Lawrence, M.J.; Barlow, D.J. Interaction of Amphotericin B with Lipid Monolayers. Langmuir 2014, 30, 9147–9156. [Google Scholar] [CrossRef]
- Harayama, T.; Riezman, H. Understanding the diversity of membrane lipid composition. Nat. Rev. Mol. Cell Biol. 2018, 19, 281. [Google Scholar] [CrossRef] [PubMed]
- Yepuri, N.R.; Darwish, T.A.; Krause-Heuer, A.M.; Leung, A.E.; Delhom, R.; Wacklin, H.P.; Holden, P.J. Synthesis of Perdeuterated 1-Palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine ([D82]POPC) and Characterisation of Its Lipid Bilayer Membrane Structure by Neutron Reflectometry. ChemPlusChem 2016, 81, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Fragneto, G.; Delhom, R.; Joly, L.; Scoppola, E. Neutrons and model membranes: Moving towards complexity. Curr. Opin. Colloid Interface Sci. 2018, 38, 108–121. [Google Scholar] [CrossRef]
- Gerelli, Y.; de Ghellinck, A.; Jouhet, J.; Laux, V.; Haertlein, M.; Fragneto, G. Multi-lamellar organization of fully deuterated lipid extracts of yeast membranes. Acta Crystallogr. Sect. D Biol. Crystallogr. 2014, 70, 3167–3176. [Google Scholar] [CrossRef]
- Luchini, A.; Delhom, R.; Cristiglio, V.; Knecht, W.; Wacklin-Knecht, H.; Fragneto, G. Effect of ergosterol on the interlamellar spacing of deuterated yeast phospholipid multilayers. Chem. Phys. Lipids 2020, 227, 104873. [Google Scholar] [CrossRef]
- Luchini, A.; Delhom, R.; Demé, B.; Laux, V.; Moulin, M.; Haertlein, M.; Pichler, H.; Strohmeier, G.A.; Wacklin, H.; Fragneto, G. The impact of deuteration on natural and synthetic lipids: A neutron diffraction study. Colloids Surf. B Biointerfaces 2018, 168, 126–133. [Google Scholar] [CrossRef]
- de Ghellinck, A.; Schaller, H.; Laux, V.; Haertlein, M.; Sferrazza, M.; Maréchal, E.; Wacklin, H.; Jouhet, J.; Fragneto, G. Production and Analysis of Perdeuterated Lipids from Pichia pastoris Cells. PLoS ONE 2014, 9, e92999. [Google Scholar] [CrossRef]
- de Ghellinck, A.; Fragneto, G.; Laux, V.; Haertlein, M.; Jouhet, J.; Sferrazza, M.; Wacklin, H. Lipid polyunsaturation determines the extent of membrane structural changes induced by Amphotericin B in Pichia pastoris yeast. Biochim. Biophys. Acta (BBA) Biomembr. 2015, 1848, 2317–2325. [Google Scholar] [CrossRef]
- Wacklin, H.P. Composition and asymmetry in supported membranes formed by vesicle fusion. Langmuir 2011, 27, 7698–7707. [Google Scholar] [CrossRef]
- Hayter, J.B.; Highfield, R.R.; Pullman, B.J.; Thomas, R.K.; McMullen, A.I.; Penfold, J. Critical reflection of neutrons. A new technique for investigating interfacial phenomena. J. Chem. Soc. Faraday Trans. 1 Phys. Chem. Condens. Phases 1981, 77, 1437–1448. [Google Scholar] [CrossRef]
- Vacklin, H.P.; Tiberg, F.; Fragneto, G.; Thomas, R.K. Composition of Supported Model Membranes Determined by Neutron Reflection. Langmuir 2005, 21, 2827–2837. [Google Scholar] [CrossRef] [PubMed]
- Wacklin, H.P.; Bremec, B.B.; Moulin, M.; Rojko, N.; Haertlein, M.; Forsyth, T.; Anderluh, G.; Norton, R.S. Neutron reflection study of the interaction of the eukaryotic pore-forming actinoporin equinatoxin II with lipid membranes reveals intermediate states in pore formation. Biochim. Biophys. Acta (BBA) Biomembr. 2016, 1858, 640–652. [Google Scholar] [CrossRef] [PubMed]
- James, M.; Nelson, A.; Holt, S.A.; Saerbeck, T.; Hamilton, W.A.; Klose, F. The multipurpose time-of-flight neutron reflectometer “Platypus” at Australia’s OPAL reactor. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrometers Detect. Assoc. Equip. 2011, 632, 112–123. [Google Scholar] [CrossRef]
- Nelson, A. Co-refinement of multiple-contrast neutron/X-ray reflectivity data using MOTOFIT. J. Appl. Crystallogr. 2006, 39, 273–276. [Google Scholar] [CrossRef]
- Born, M.; Wolf, E. Principles of Optics: Electromagnetic Theory of Propagation, Interference and Diffraction of Light, 7th ed.; Cambridge University Press: Cambridge, UK, 1999. [Google Scholar] [CrossRef]
- Armen, R.S.; Uitto, O.D.; Feller, S.E. Phospholipid component volumes: Determination and application to bilayer structure calculations. Biophys. J. 1998, 75, 734–744. [Google Scholar] [CrossRef]
- Nagle, J.F.; Tristram-Nagle, S. Structure of lipid bilayers. Biochim. Biophys. Acta (BBA) Rev. Biomembr. 2000, 1469, 159–195. [Google Scholar] [CrossRef]
- Greenwood, A.I.; Tristram-Nagle, S.; Nagle, J.F. Partial molecular volumes of lipids and cholesterol. Chem. Phys. Lipids 2006, 143, 1–10. [Google Scholar] [CrossRef]
- Johnson, S.J.; Bayerl, T.M.; McDermott, D.C.; Adam, G.W.; Rennie, A.R.; Thomas, R.K.; Sackmann, E. Structure of an Adsorbed Dimyristoylphosphatidylcholine Bilayer Measured with Specular Reflection of Neutrons. Biophys. J. 1991, 59, 289–294. [Google Scholar] [CrossRef]
- Cremer, P.S.; Boxer, S.G. Formation and spreading of lipid bilayers on planar glass supports. J. Phys. Chem. B 1999, 103, 2554–2559. [Google Scholar] [CrossRef]
- Zwang, T.J.; Fletcher, W.R.; Lane, T.J.; Johal, M.S. Quantification of the Layer of Hydration of a Supported Lipid Bilayer. Langmuir 2010, 26, 4598–4601. [Google Scholar] [CrossRef]
- Waldie, S.; Lind, T.K.; Browning, K.; Moulin, M.; Haertlein, M.; Forsyth, V.T.; Luchini, A.; Strohmeier, G.A.; Pichler, H.; Maric, S.; et al. Localization of Cholesterol within Supported Lipid Bilayers Made of a Natural Extract of Tailor-Deuterated Phosphatidylcholine. Langmuir 2018, 34, 472–479. [Google Scholar] [CrossRef]
- Marquardt, D.; Heberle, F.A.; Greathouse, D.V.; Koeppe, R.E.; Standaert, R.F.; Van Oosten, B.J.; Harroun, T.A.; Kinnun, J.J.; Williams, J.A.; Wassall, S.R.; et al. Lipid bilayer thickness determines cholesterol’s location in model membranes. Soft Matter 2016, 12, 9417–9428. [Google Scholar] [CrossRef] [PubMed]
- Åkesson, A.; Lind, T.; Ehrlich, N.; Stamou, D.; Wacklin, H.; Cárdenas, M. Composition and structure of mixed phospholipid supported bilayers formed by POPC and DPPC. Soft Matter 2012, 8, 5658–5665. [Google Scholar] [CrossRef]
- Soranzo, T.; Martin, D.K.; Lenormand, J.-L.; Watkins, E.B. Coupling neutron reflectivity with cell-free protein synthesis to probe membrane protein structure in supported bilayers. Sci. Rep. 2017, 7, 3399. [Google Scholar] [CrossRef] [PubMed]
- Kučerka, N.; Nieh, M.-P.; Katsaras, J. Fluid phase lipid areas and bilayer thicknesses of commonly used phosphatidylcholines as a function of temperature. Biochim. Biophys. Acta (BBA) Biomembr. 2011, 1808, 2761–2771. [Google Scholar] [CrossRef]
- Dabkowska, A.P.; Collins, L.E.; Barlow, D.J.; Barker, R.; McLain, S.E.; Lawrence, M.J.; Lorenz, C.D. Modulation of Dipalmitoylphosphatidylcholine Monolayers by Dimethyl Sulfoxide. Langmuir 2014, 30, 8803–8811. [Google Scholar] [CrossRef]
- Hung, W.-C.; Lee, M.-T.; Chung, H.; Sun, Y.-T.; Chen, H.; Charron, N.E.; Huang, H.W. Comparative Study of the Condensing Effects of Ergosterol and Cholesterol. Biophys. J. 2016, 110, 2026–2033. [Google Scholar] [CrossRef][Green Version]
- Baran, M.; Borowski, E.; Mazerski, J. Molecular modeling of amphotericin B–ergosterol primary complex in water II. Biophys. Chem. 2009, 141, 162–168. [Google Scholar] [CrossRef]
- Urbina, J.A.; Pekerar, S.; Le, H.-B.; Patterson, J.; Montez, B.; Oldfield, E. Molecular order and dynamics of phosphatidylcholine bilayer membranes in the presence of cholesterol, ergosterol and lanosterol: A comparative study using 2H-, 13C- and 31P-NMR spectroscopy. Biochim. Biophys. Acta (BBA) Biomembr. 1995, 1238, 163–176. [Google Scholar] [CrossRef]
- Readio, J.D.; Bittman, R. Equilibrium binding of amphotericin B and its methyl ester and borate complex to sterols. Biochim. Biophys. Acta (BBA) Biomembr. 1982, 685, 219–224. [Google Scholar] [CrossRef]
- Neumann, A.; Baginski, M.; Czub, J. How Do Sterols Determine the Antifungal Activity of Amphotericin B? Free Energy of Binding between the Drug and Its Membrane Targets. J. Am. Chem. Soc. 2010, 132, 18266–18272. [Google Scholar] [CrossRef] [PubMed]
- Tancréde, P.; Barwicz, J.; Jutras, S.; Gruda, I. The effect of surfactants on the aggregation state of amphotericin B. Biochim. Biophys. Acta (BBA) Biomembr. 1990, 1030, 289–295. [Google Scholar] [CrossRef]
- Borman, A.M.; Muller, J.; Walsh-Quantick, J.; Szekely, A.; Patterson, Z.; Palmer, M.D.; Fraser, M.; Johnson, E.M. MIC distributions for amphotericin B, fluconazole, itraconazole, voriconazole, flucytosine and anidulafungin and 35 uncommon pathogenic yeast species from the UK determined using the CLSI broth microdilution method. J. Antimicrob. Chemother. 2020, 75, 1194–1205. [Google Scholar] [CrossRef] [PubMed]
- Barwicz, J.; Tancrède, P. The effect of aggregation state of amphotericin-B on its interactions with cholesterol- or ergosterol-containing phosphatidylcholine monolayers. Chem. Phys. Lipids 1997, 85, 145–155. [Google Scholar] [CrossRef]
- Kamiński, D.M.; Czernel, G.; Murphy, B.; Runge, B.; Magnussen, O.M.; Gagoś, M. Effect of cholesterol and ergosterol on the antibiotic amphotericin B interactions with dipalmitoylphosphatidylcholine monolayers: X-ray reflectivity study. Biochim. Biophys. Acta (BBA) Biomembr. 2014, 1838, 2947–2953. [Google Scholar] [CrossRef]
- Van Hoogevest, P.; De Kruijff, B. Effect of amphotericin B on cholesterol-containing liposomes of egg phosphatidylcholine and didocosenoyl phosphatidylcholine. A refinement of the model for the formation of pores by amphotericin B in membranes. Biochim. Biophys. Acta (BBA) Biomembr. 1978, 511, 397–407. [Google Scholar] [CrossRef]
- Walker, L.; Sood, P.; Lenardon, M.D.; Milne, G.; Olson, J.; Jensen, G.; Wolf, J.; Casadevall, A.; Adler-Moore, J.; Gow, N.A.R. The Viscoelastic Properties of the Fungal Cell Wall Allow Traffic of AmBisome as Intact Liposome Vesicles. mBio 2018, 9, e02383-17. [Google Scholar] [CrossRef]
- Gerelli, Y.; Porcar, L.; Lombardi, L.; Fragneto, G. Lipid Exchange and Flip-Flop in Solid Supported Bilayers. Langmuir 2013, 29, 12762–12769. [Google Scholar] [CrossRef]
- Garg, S.; Porcar, L.; Woodka, A.C.; Butler, P.D.; Perez-Salas, U. Noninvasive Neutron Scattering Measurements Reveal Slower Cholesterol Transport in Model Lipid Membranes. Biophys. J. 2011, 101, 370–377. [Google Scholar] [CrossRef]
| POPC | Ergosterol | Cholesterol | AmB | |
|---|---|---|---|---|
| V (Å3) | Heads: 322 a | 630 c | 630 c | 983 c |
| Chains: 934 b | ||||
| ρ (10−6 Å−2) d | Heads: 1.86 (h) 7.34 (d) | 0.43 (h) 7.70 (d) | 0.20(h) | 2.88 (D2O) 2.41 (CM4) 2.01 (CMSi) 1.50 (H2O) |
| ρ (10−6 Å−2) | Chains: −0.28 (h) 6.34 (d) |
| Before AmB Addition | After AmB Addition | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| τ (Å) | ρ (10−6 Å−2) | φ (%) | σ (Å) | A (Å2) | τ (Å) | ρ (10−6 Å−2) | φ (%) | σ (Å) | A (Å2) | |
| d82POPC | 5.5 ± 0.9 mol% AmB (heads and chains) | |||||||||
| Inner heads | 11 ± 1 | 7.35 ± 0.2 | 50 ± 5 | 4 ± 1 | 62 ± 10 | 11 ± 1 | 7.08 ± 0.1 a | 51 ± 5 | 2 ± 1 | 63 ± 12 |
| Chains | 31 ± 1 | 6.35 ± 0.05 | 1 ± 1 | 4 ± 1 | 61 ± 2 | 32 ± 1 | 6.15 ± 0.04 b | 0 ± 1 | 1 ± 1 | 63 ± 3 |
| Outer heads | 9 ± 1 | 7.35 ± 0.2 | 43 ± 5 | 4 ± 1 | 63 ± 10 | 11 ± 1 | 7.08 ± 0.1 a | 51 ± 5 | 2 ± 1 | 63 ± 12 |
| hPOPC | 5.5 ± 0.9 mol% AmB (chains) | |||||||||
| Inner heads | 10 ± 1 | 1.86 ± 0.2 | 46 ± 5 | 4 ± 1 | 63 ± 15 | 10 ± 1 | 1.86 ± 0.2 d | 53 ± 5 | 3 ± 1 | 74 ± 15 |
| Chains | 30 ± 1 | −0.28 ± 0.05 | 0 ± 1 | 4 ± 1 | 62 ± 3 | 32 ± 1 | −0.106 ± 0.06 c | 0 ± 1 | 1 ± 1 | 63 ± 3 |
| Outer heads | 10 ± 1 | 1.86 ± 0.2 | 46 ± 5 | 3 ± 1 | 63± 15 | 8± 1 | 1.86 ± 0.2 d | 57 ± 5 | 3 ± 1 | 99 ± 15 |
| Before AmB Addition | After AmB Addition | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| τ (Å) | ρ (10−6 Å−2) | φ (%) | σ (Å) | A (Å2) | τ (Å) | ρ (10−6 Å−2) | φ (%) | σ (Å) | A (Å2) | |
| hPOPC–dErg | 10 ± 1 mol% dErg | 3.0 ± 2.8 mol% AmB & 7.4 ± 1.5 mol% Erg | ||||||||
| Inner heads | 8 ± 1 | 1.86 ± 0.20 | 36 ± 8 | 3 ± 2 | 67 ± 13 | 8 ± 1 | 1.86 ± 0.20 | 47 ± 7 | 3 ± 2 | 76± 12 |
| Chains | 32 ± 1 | 0.3 ± 0.07 | 6 ± 2 | 3 ± 2 | 67± 4 | 30 ± 1 | 0.20 ± 0.06 a | 10 ± 2 | 5 ± 2 | 76 ± 5 |
| Outer heads | 8 ± 1 | 1.86 ± 0.20 | 36 ± 8 | 4 ± 2 | 67 ± 13 | 8 ± 1 | 1.86 ± 0.20 | 44 ± 8 | 5 ± 2 | 76 ± 12 |
| hPOPC–hErg | 14 ± 6 mol% hErg | 4.2 ± 3.8 mol% AmB & 7.7 ± 5.8 mol% Erg | ||||||||
| Inner heads | 9 ± 1 | 1.86 ± 0.20 | 42 ± 9 | 3 ± 2 | 65 ± 12 | 9 ± 1 | 1.86 ± 0.20 | 49 ± 8 | 3 ± 2 | 70 ± 12 |
| Chains | 32 ± 1 | −0.22 ± 0.04 | 2 ± 2 | 4 ± 2 | 64 ± 4 | 30 ± 1 | −0.14 ± 0.06 b | 2 ± 2 | 3 ± 2 | 68 ± 5 |
| Outer heads | 9 ± 1 | 1.86 ± 0.20 | 42 ± 9 | 3 ± 2 | 65 ± 12 | 9 ± 1 | 1.86 ± 0.20 | 48 ± 8 | 5 ± 2 | 72 ± 12 |
| d82POPC–hErg | 19 ± 2 mol% hErg | 21.7 ± 6.8 mol% AmB & 8.4 ± 4.7 mol% Erg | ||||||||
| Inner heads | 10 ± 1 | 7.35 ± 0.20 | 52 ± 8 | 2 ± 2 | 67 ± 11 | 10 ± 1 | 7.35 ± 0.20 | 52 ± 8 | 2 ± 2 | 75 ± 13 |
| Chains | 34 ± 1 | 5.60 ± 0.07 | 8 ± 2 | 3 ± 2 | 67 ± 4 | 32 ± 1 | 5.17 ± 0.09 c | 0 ± 2 | 4 ± 2 | 75 ± 3 |
| Outer heads | 9 ± 1 | 7.35 ± 0.20 | 47 ± 8 | 3 ± 2 | 67 ± 11 | 9 ± 1 | 6.81 d ± 0.20 | 49 ± 9 | 6 ± 2 | 75 ± 16 |
| Before AmB Addition | After AmB Addition | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| τ (Å) | ρ (10−6 Å−2) | φ (%) | σ (Å) | A (Å2) | τ (Å) | ρ (10−6 Å−2) | φ (%) | σ (Å) | A (Å2) | |
| d82POPC–hChol | 18.3 ± 3 mol% hChol | 11.1 ± 3.3 mol% AmB & 18.4 ± 3.4 mol% hChol | ||||||||
| Inner heads | 10 ± 1 | 7.35 ± 0.20 | 48 ± 8 | 1 ± 2 | 66 ± 9 | 8 ± 1 | 7.15 ± 0.20 a | 43 ± 8 | 1 ± 2 | 68 ± 11 |
| Chains | 34 ± 1 | 5.58 ± 0.05 | 7 ± 3 | 2 ± 2 | 66 ± 4 | 35 ± 1 | 5.27 ± 0.20 b | 2 ± 2 | 2 ± 2 | 68 ± 3 |
| Outer heads | 9 ± 1 | 7.35 ± 0.20 | 44 ± 8 | 5± 2 | 66 ± 9 | 9 ± 1 | 7.15 ± 0.20 a | 37 ± 9 | 3 ± 2 | 68 ± 12 |
| hPOPC–hChol | 14.5 ± 8 mol% hChol | 5.7 ± 2.6 mol% AmB & 14.5 ± 3.7 mol% hChol | ||||||||
| Inner heads | 9 ± 1 | 1.86 ± 0.20 | 45 ± 7 | 3 ± 2 | 65 ± 7 | 8 ± 1 | 1.8 ± 0.20 | 38 ± 7 | 4 ± 2 | 65 ± 10 |
| Chains | 33 ± 1 | −0.23 ± 0.03 | 3 ± 2 | 5 ± 2 | 65 ± 3 | 34 ± 1 | −0.07 ± 0.06 c | 2 ± 2 | 2 ± 2 | 65 ± 4 |
| Outer heads | 10 ± 1 | 1.8 ± 0.20 | 51 ± 7 | 5 ± 2 | 65 ± 7 | 9 ± 1 | 1.8 ± 0.20 | 45 ± 7 | 5 ± 2 | 69 ± 10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delhom, R.; Nelson, A.; Laux, V.; Haertlein, M.; Knecht, W.; Fragneto, G.; Wacklin-Knecht, H.P. The Antifungal Mechanism of Amphotericin B Elucidated in Ergosterol and Cholesterol-Containing Membranes Using Neutron Reflectometry. Nanomaterials 2020, 10, 2439. https://doi.org/10.3390/nano10122439
Delhom R, Nelson A, Laux V, Haertlein M, Knecht W, Fragneto G, Wacklin-Knecht HP. The Antifungal Mechanism of Amphotericin B Elucidated in Ergosterol and Cholesterol-Containing Membranes Using Neutron Reflectometry. Nanomaterials. 2020; 10(12):2439. https://doi.org/10.3390/nano10122439
Chicago/Turabian StyleDelhom, Robin, Andrew Nelson, Valerie Laux, Michael Haertlein, Wolfgang Knecht, Giovanna Fragneto, and Hanna P. Wacklin-Knecht. 2020. "The Antifungal Mechanism of Amphotericin B Elucidated in Ergosterol and Cholesterol-Containing Membranes Using Neutron Reflectometry" Nanomaterials 10, no. 12: 2439. https://doi.org/10.3390/nano10122439
APA StyleDelhom, R., Nelson, A., Laux, V., Haertlein, M., Knecht, W., Fragneto, G., & Wacklin-Knecht, H. P. (2020). The Antifungal Mechanism of Amphotericin B Elucidated in Ergosterol and Cholesterol-Containing Membranes Using Neutron Reflectometry. Nanomaterials, 10(12), 2439. https://doi.org/10.3390/nano10122439





