In Situ Ultraviolet Polymerization Using Upconversion Nanoparticles: Nanocomposite Structures Patterned by Near Infrared Light
Abstract
:1. Introduction
2. Materials and Methods
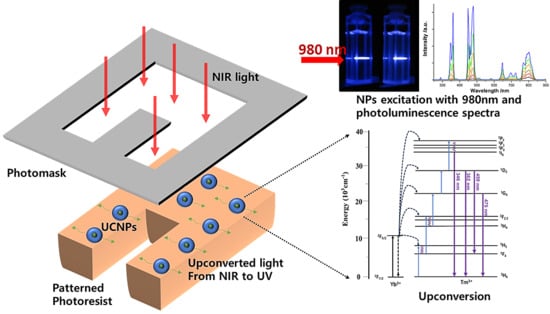
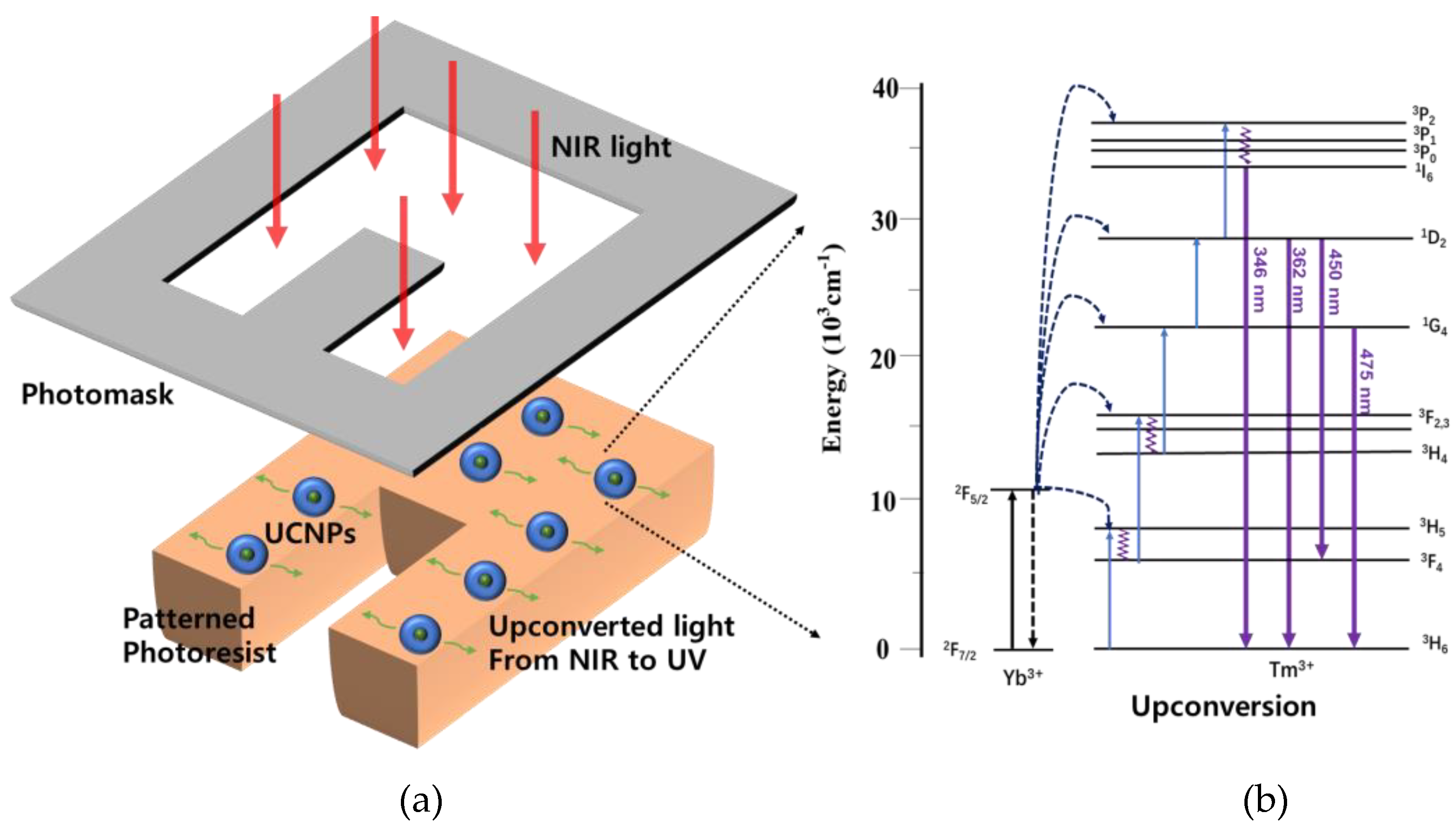
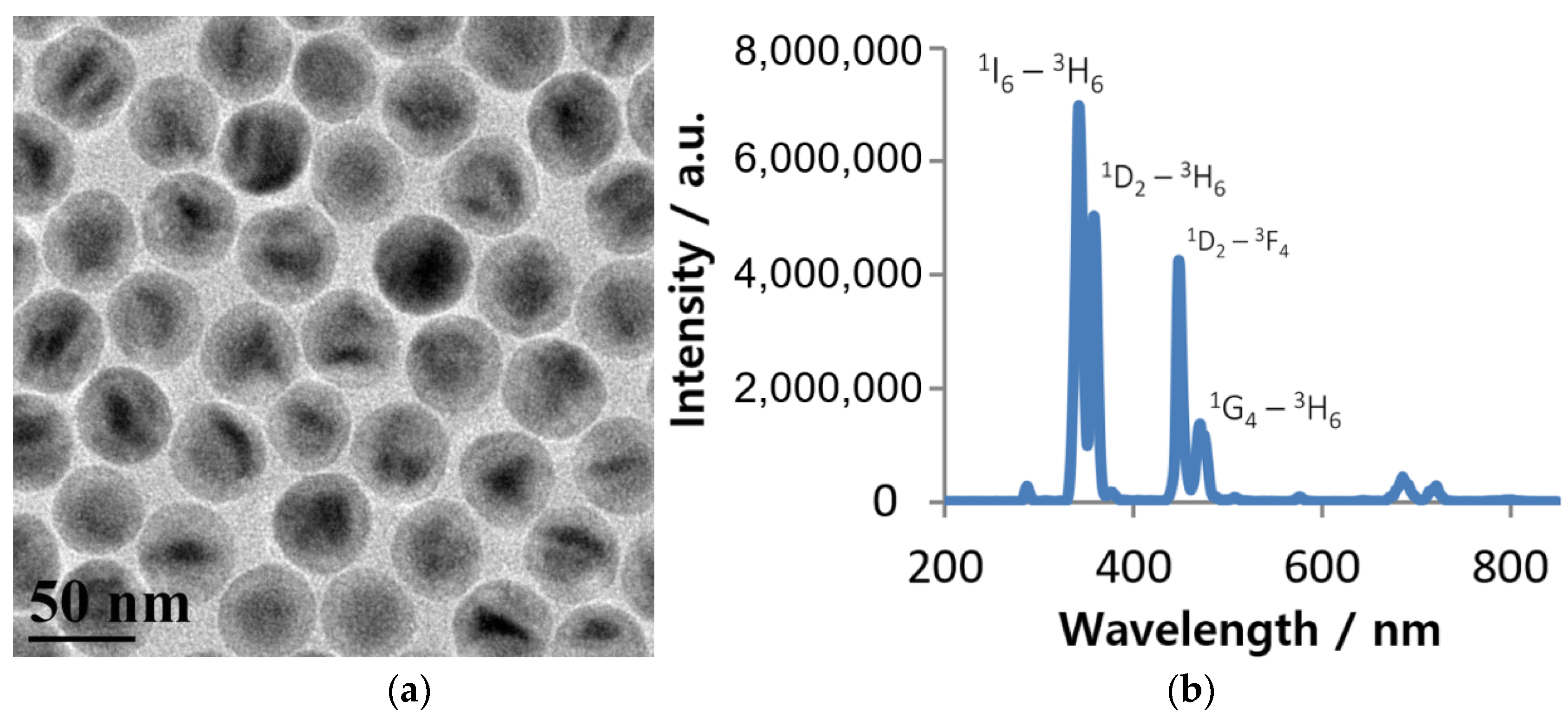
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Oster, G.; Yang, N. Photopolymerization of vinyl monomers. Chem. Rev. 1968, 68, 125–151. [Google Scholar] [CrossRef]
- Lee, H.; Hong, S.; Yang, K.; Choi, K. Fabrication of nano-sized resist patterns on flexible plastic film using thermal curing nano-imprint lithography. Microelectron. Eng. 2006, 83, 323–327. [Google Scholar] [CrossRef]
- Eklund, N.; Huang, J. Planographic Printing Plate. U.S. Patent 4,483,913, 20 November 1984. [Google Scholar]
- Miller, S.M.; Troian, S.M. Direct printing of polymer microstructures on flat and spherical surfaces using a letterpress technique. J. Vac. Sci. Technol. B 2002, 20, 2320–2327. [Google Scholar] [CrossRef] [Green Version]
- Yang, K.X.; Vervust, T.; Vanfleteren, J. Multifunctional and miniaturized flexible sensor patch: Design and application for in situ monitoring of epoxy polymerization. Sens. Actuators B Chem. 2018, 261, 144–152. [Google Scholar] [CrossRef]
- Ferracane, J.L. Developing a more complete understanding of stresses produced in dental composites during polymerization. Dent. Mater. J. 2005, 21, 36–41. [Google Scholar] [CrossRef]
- Madou, M.J. Fundamentals of microfabrication, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2002; pp. 4–70. [Google Scholar]
- Jančovičová, V.; Mikula, M.; Havlínová, B.; Jakubíková, Z. Influence of UV-curing conditions on polymerization kinetics and gloss of urethane acrylate coatings. Prog. Org. Coat. 2013, 76, 432–438. [Google Scholar] [CrossRef]
- Endruweit, A.; Johnson, M.S.; Long, A.C. Curing of composite components by ultraviolet radiation: A review. Polym. Compos. 2006, 27, 119–128. [Google Scholar] [CrossRef]
- Hirano, T.; Kitagawa, S.; Ohtani, H. Methacrylate-ester-based Reversed Phase Monolithic Columns for High Speed Separation Prepared by Low Temperature UV Photo-polymerization. Anal. Sci. 2009, 25, 1107–1113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Decker, C.; Viet, T.N.T. Photocrosslinking of functionalized rubbers IX. Thiol-ene polymerization of styrene-butadiene-block-copolymers. Polymers 2000, 41, 3905–3912. [Google Scholar]
- Singha, A.S.; Thakur, V.K. Chemical Resistance, Mechanical and Physical Properties of Biofibers-Based Polymer Composites. Polym-Plast. Technol. 2009, 44, 736–744. [Google Scholar] [CrossRef]
- Tasic, S.; Bozic, B.; Dunjic, B. Synthesis of new hyperbranched urethane-acrylates and their evaluation in UV-curable coatings. Prog. Org. Coat. 2004, 51, 320–327. [Google Scholar] [CrossRef]
- Dill, F.H.; Hornberger, W.P.; Hauge, P.S.; Shaw, J.M. Characterization of positive photoresist. IEEE Trans. Electron Devices 1975, 22, 445–452. [Google Scholar] [CrossRef]
- Rehab, A. New photosensitive polymers as negative photoresist materials. Eur. Polym. 1998, 34, 1845–1855. [Google Scholar] [CrossRef]
- Leach, S.J.; Scheraga, H.A. Effect of Light Scattering on Ultraviolet Difference Spectra. JACS 1960, 82, 4790–4792. [Google Scholar] [CrossRef]
- Bertsch, A.; Lorenz, H.; Renaud, P. 3D microfabrication by combining microstereolithography and thick resist UV lithography. Sens. Actuators A Phys. 1999, 73, 14–23. [Google Scholar] [CrossRef]
- Alsharif, S.O.; Akil, H.B.M.; El-Aziz, N.A.A.; Ahmad, Z.A.B. Effect of alumina particles loading on the mechanical properties of light-cured dental resin composites. Mater. Des. 2014, 54, 430–435. [Google Scholar] [CrossRef]
- Teh, P.L.; Mariatti, J.; Akil, H.M.; Yeoh, C.K.; Seetharamu, K.N.; Wagiman, A.N.R.; Beh, K.S. The properties of epoxy resin coated silica fillers composites. Mater. Lett. 2007, 61, 2156–2158. [Google Scholar] [CrossRef]
- Sanchez, C.; Julián, B.; Belleville, P.; Popall, M. Applications of hybrid organic–inorganic nanocomposites. J. Mater. Chem. 2005, 15, 3559–3592. [Google Scholar] [CrossRef]
- Sangerano, M.; Pallaro, E.; Roppolo, I.; Rizza, G. UV-cured epoxy coating reinforced with sepiolite as inorganic filler. J. Mater. Sci. 2009, 44, 3165–3171. [Google Scholar] [CrossRef]
- Han, Y.; Taylor, A.; Knowles, K.M. Scratch resistance and adherence of novel organic–inorganic hybrid coatings on metallic and non-metallic substrates. Surf. Coat. Technol. 2009, 203, 2871–2877. [Google Scholar] [CrossRef]
- Chen, C.C.; Chung, M.; Hseih, T.; Liu, M.O.; Lin, J.; Chu, W.; Tang, R.; Tsai, Y.; Juang, F. Synthesis, thermal characterization, and gas barrier properties of UV curable organic/inorganic hybrid nanocomposites with metal alloys and their application for encapsulation of organic solar cells. Compos. Sci. Technol. 2008, 68, 3041–3046. [Google Scholar] [CrossRef]
- Liu, B.; Tang, S.; Yu, Y.; Lin, S. High-refractive-index polymer/inorganic hybrid films containing high TiO2 contents. Colloids Surf. A 2011, 377, 138–143. [Google Scholar] [CrossRef]
- Wouters, M.E.L.; Wolfs, D.P.; Linde, M.C.; Hovens, J.H.P.; Tinnemans, A.H.A. Transparent UV curable antistatic hybrid coatings on polycarbonate prepared by the sol–gel method. Prog. Org. Coat. 2004, 51, 312–319. [Google Scholar] [CrossRef]
- Wang, Y.; Su, J.; Li, T.; Ma, P.; Bai, H.; Xie, Y.; Chen, M.; Dong, W. A Novel UV-Shielding and Transparent Polymer Film: When Bioinspired Dopamine–Melanin Hollow Nanoparticles Join Polymers. Appl. Mater. Interfaces 2017, 9, 36281–36289. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zeng, X.; King, X.; Bian, S.; Chen, J. A simple two-step method to fabricate highly transparent ITO/polymer nanocomposite films. Appl. Surf. Sci. 2012, 258, 8564–8569. [Google Scholar] [CrossRef]
- Wellman, E. UV Radiation in Photomorphogenesis; Springer: Berlin, Germany, 1983; pp. 745–756. [Google Scholar]
- Hollósy, F. Effects of ultraviolet radiation on plant cells. Micron 2002, 33, 179–197. [Google Scholar] [CrossRef]
- Nylander-French, L.A.; Fischer, T.; Hultengren, M.; Lewne, M.; Rosen, G. Assessment of Worker Exposure in the Processing of Ultraviolet Radiation-Cured Acrylate Lacquer-Coated Wood Products. Appl. Occup. Environ. Hyg. 2011, 9, 962–976. [Google Scholar] [CrossRef]
- Kocaarslan, A.; Tabanli, S.; Eryurek, G.; Yagci, Y. Near-Infrared Free-Radical and Free-Radical-Promoted Cationic Photopolymerizations by In-Source Lighting Using Upconverting Glass. Angew. Chem. Int. 2017, 56, 14507–14510. [Google Scholar] [CrossRef]
- Zhang, S.; Tang, L.; Wang, X.; Liu, D.; Zhou, Q. Studies on the near infrared laser induced photopolymerization employing a cyanine dye–borate complex as the photoinitiator. Polymer 2001, 42, 7575–7582. [Google Scholar] [CrossRef]
- Beyazit, S.; Ambrosini, S.; Marchyk, N.; Palo, E.; Kale, V.; Soukka, T.; Bui, B.T.S.; Haupt, K. Versatile Synthetic Strategy for Coating Upconverting Nanoparticles with Polymer Shells through Localized Photopolymerization by Using the Particles as Internal Light Sources. Angew. Chem. Int. 2014, 53, 8919–8923. [Google Scholar] [CrossRef]
- Demina, P.; Arkharova, N.; Asharchuk, I.; Khaydukov, K.; Karimov, D.; Rocheva, V.; Nechaev, A.; Grigoriev, Y.; Generalova, A.; Khaydukov, E. Polymerization Assisted by Upconversion Nanoparticles under NIR Light. Molecules 2019, 24, 2476. [Google Scholar] [CrossRef] [Green Version]
- Chen, Z.; Wang, X.; Li, S.; Liu, S.; Miao, H.; Wu, S. Near-Infrared Light Driven Photopolymerization Based On Photon Upconversion. ChemPhotoChem 2019, 3, 1077–1083. [Google Scholar] [CrossRef]
- Shen, J.; Chen, G.; Ohulchanskyy, T.Y.; Kesseli, S.J.; Buchholz, S.; Li, Z.; Prasad, P.N.; Han, G. Tunable to ultraviolet upconversion luminescence enhancement in (α-NaYF4:Yb,Tm3+)/CaF2 core/shell nanoparticles for in situ real-time recorded biocompatible photoactivation. Small 2013, 9, 3213–3217. [Google Scholar] [CrossRef] [PubMed]
- Pliss, A.; Ohulchanskyy, T.Y.; Chen, G.; Damasco, J.; Bass, C.E.; Prasad, P.N. Subcellular Optogenetics Enacted by Targeted Nanotransformers of Near-Infrared Light. ACS Photonics 2017, 4, 806–814. [Google Scholar] [CrossRef]
- Shukla, S.; Kim, K.; Baev, A.; Yoon, Y.K.; Litchinitser, N.M.; Prasad, P.N. Fabrication and Characterization of Gold−Polymer Nanocomposite Plasmonic Nanoarrays in a Porous Alumina Template. ACS Nano 2010, 4, 2249–2255. [Google Scholar] [CrossRef]
- Oh, H.S.; Jee, H.; Baev, A.; Swihart, M.T.; Prasad, P.N. Dramatic Structural Enhancement of Chirality in Photopatternable Nanocomposites of Chiral Poly(fluorene-alt-benzothiadiazole) (PFBT) in Achiral SU-8 Photoresist. Adv. Funct. Mater. 2012, 22, 5074–5080. [Google Scholar] [CrossRef]
- Cao, Y.; Gan, Z.; Jia, B.; Evans, R.A.; Gu, M. High-photosensitive resin for super-resolution direct-laser-writing based on photoinhibited polymerization. Opt. Express 2011, 19, 19486–19494. [Google Scholar] [CrossRef] [PubMed]
- Stepuk, A.; Mohn, D.; Grass, R.N.; Zehnder, M.; Kramer, K.W.; Pelle, F.; Ferrier, A.; Stark, W.J. Use of NIR light and upconversion phosphors in light-curable polymers. Dent. Mater. 2012, 28, 304–311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yakovliev, A.; Ohulchanskyy, T.Y.; Ziniuk, R.; Dias, T.; Wang, X.; Xu, H.; Chen, G.; Qu, J.; Gomes, A.S.L. Noninvasive Temperature Measurement in Dental Materials Using Nd3+, Yb3+ Doped Nanoparticles Emitting in the Near Infrared Region. Part. Part. Syst. Charact. 2020, 37, 1900445. [Google Scholar] [CrossRef]
- Mohsen, N.M.; Craig, R.G. Effect of silanization of fillers on their dispersability by monomer systems. J. Oral Rehabil. 1995, 22, 183–189. [Google Scholar] [CrossRef]
- Chen, M. Update on Dental Nanocomposites. J. Dent. Res. 2010, 89, 549–560. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jee, H.; Chen, G.; Prasad, P.N.; Ohulchanskyy, T.Y.; Lee, J. In Situ Ultraviolet Polymerization Using Upconversion Nanoparticles: Nanocomposite Structures Patterned by Near Infrared Light. Nanomaterials 2020, 10, 2054. https://doi.org/10.3390/nano10102054
Jee H, Chen G, Prasad PN, Ohulchanskyy TY, Lee J. In Situ Ultraviolet Polymerization Using Upconversion Nanoparticles: Nanocomposite Structures Patterned by Near Infrared Light. Nanomaterials. 2020; 10(10):2054. https://doi.org/10.3390/nano10102054
Chicago/Turabian StyleJee, Hongsub, Guanying Chen, Paras N. Prasad, Tymish Y. Ohulchanskyy, and Jaehyeong Lee. 2020. "In Situ Ultraviolet Polymerization Using Upconversion Nanoparticles: Nanocomposite Structures Patterned by Near Infrared Light" Nanomaterials 10, no. 10: 2054. https://doi.org/10.3390/nano10102054
APA StyleJee, H., Chen, G., Prasad, P. N., Ohulchanskyy, T. Y., & Lee, J. (2020). In Situ Ultraviolet Polymerization Using Upconversion Nanoparticles: Nanocomposite Structures Patterned by Near Infrared Light. Nanomaterials, 10(10), 2054. https://doi.org/10.3390/nano10102054