Effect of Different Colored LED Lighting on the Growth and Pigment Content of Isochrysis zhanjiangensis under Laboratory Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Microalgae Culture Condition
2.2. Measurement of I. Zhanjiangensis Growth
2.3. Measurement of Photosynthetic Pigments, Protein, and Carbohydrate Content
2.4. Statistical Analysis
3. Results
3.1. Specific Growth Rate and Biomass Productivity
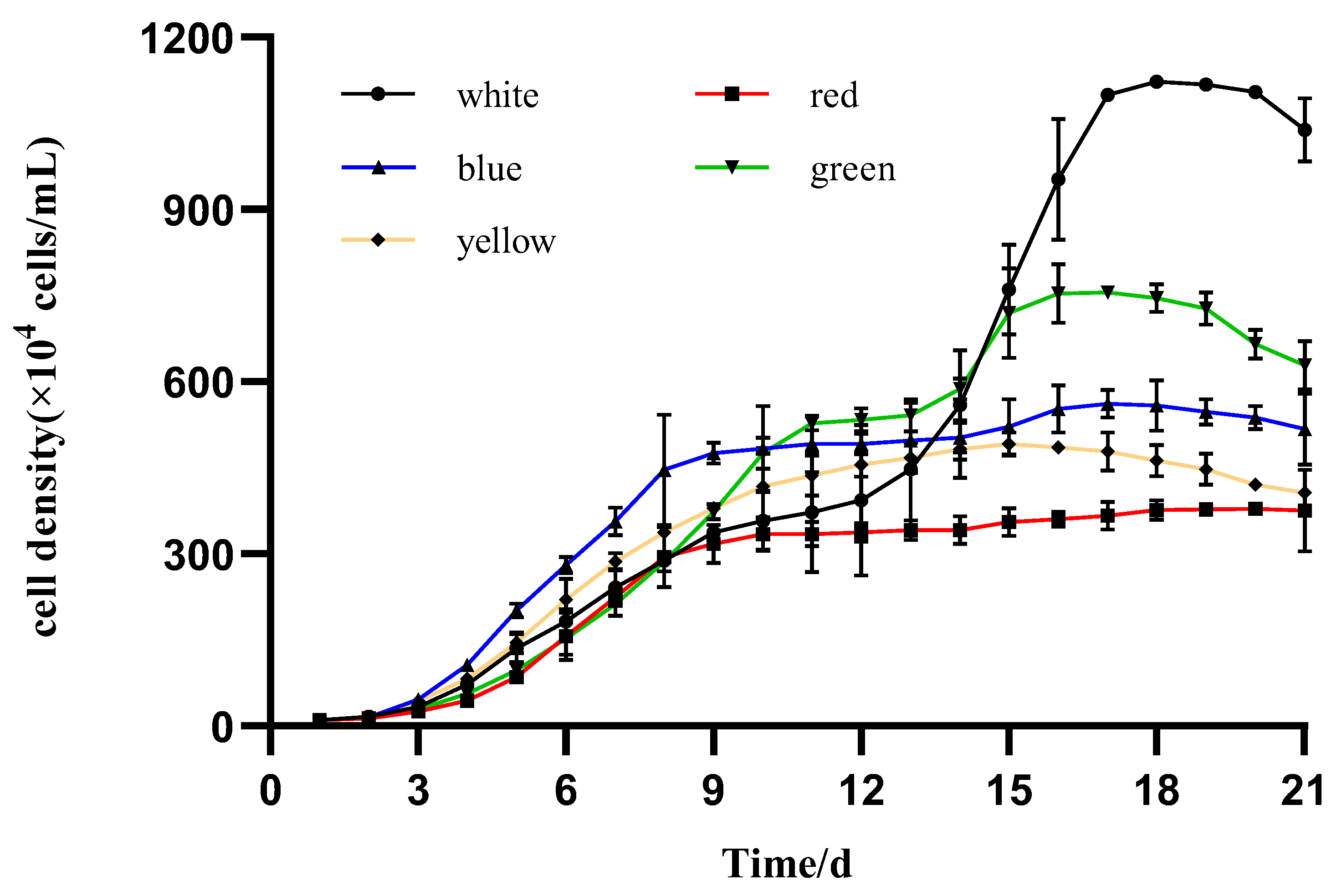
3.2. Algal Growth
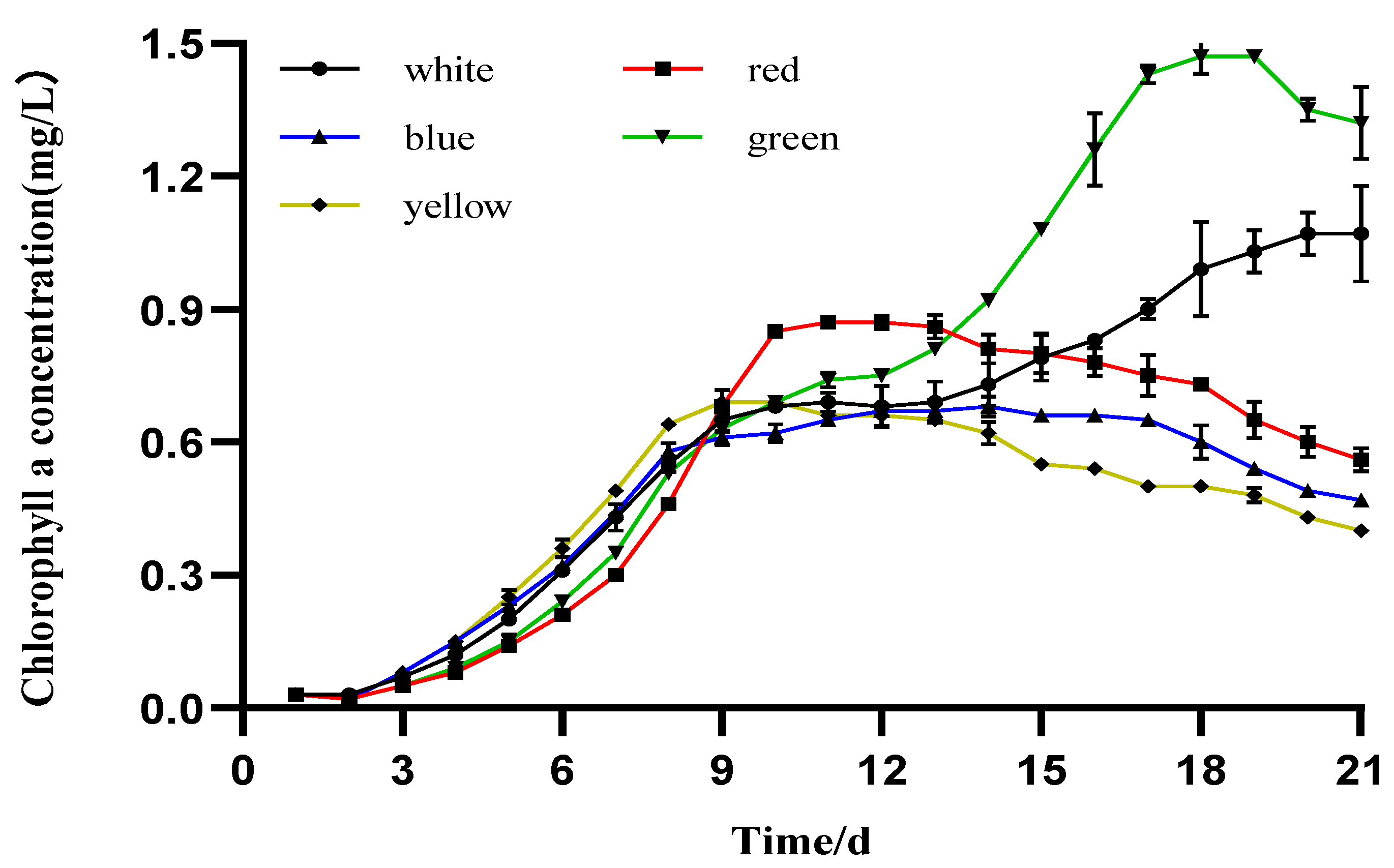
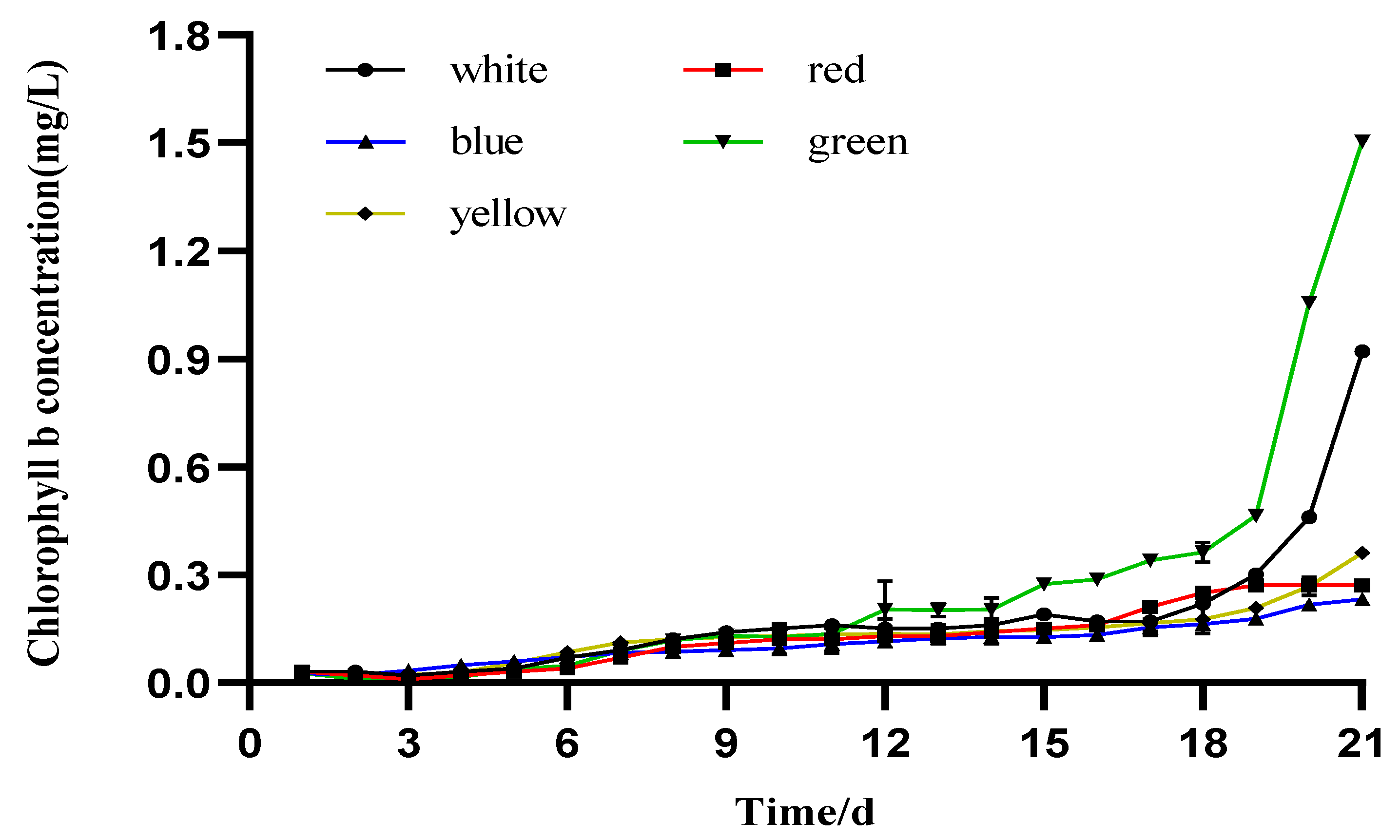
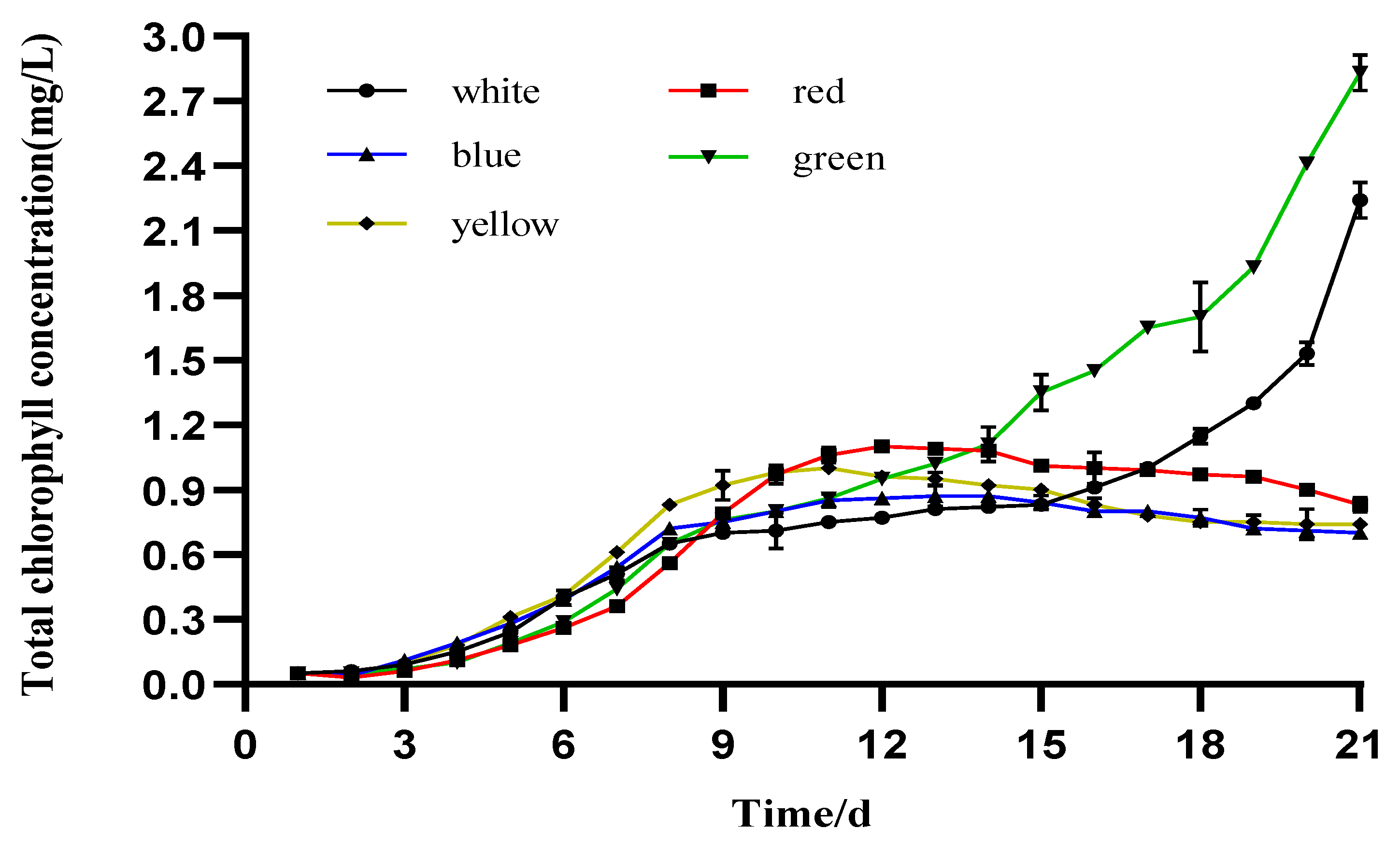
3.3. Photosynthetic Pigment Production
3.4. Protein and Soluble Carbohydrates’ Production
4. Discussion
4.1. Specific Growth Rate and Biomass Productivity of I. zhangjiangensis
4.2. The Effect of Different LED Colors on the Pigment Content of I. zhangjiangensis
4.3. The Effect of Different LED Colors on the Protein and Carbohydrate Content of I. Zhangjiangensis
5. Conclusions
6. Ethics Statement
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Hu, H.; Lu, S.; Liu, R. A New Species of Isochrysis (Isochrysidales)-I. Zhanjiangensis and Its Observation on the Fine Structure. Acta Oceanol. Sin. 2007, 29, 111–119. [Google Scholar]
- Phatarpekar, P.V.; Sreepada, R.A.; Pednekar, C.; Achuthankutty, C.T. A Comparative Study on Growth Performance and Biochemical Composition of Mixed Culture of Isochrysis galbana and Chaetoceros calcitrans with Monocultures. Aquaculture 2000, 181, 141–155. [Google Scholar] [CrossRef]
- Cao, J.-Y.; Kong, Z.-Y.; Ye, M.-W.; Ling, T.; Chen, K.; Xu, J.-L.; Zhou, C.-X.; Liao, K.; Zhang, L.; Yan, X.-J. Comprehensive Comparable Study of Metabolomic and Transcriptomic Profiling of Isochrysis galbana Exposed to High Temperature, an Important Diet Microalgal Species. Aquaculture 2020, 521, 735034. [Google Scholar] [CrossRef]
- Zhu, C.; Han, D.; Li, Y.; Zhai, X.; Chi, Z.; Zhao, Y.; Cai, H. Cultivation of Aquaculture Feed Isochrysis zhangjiangensis in Low-Cost Wave Driven Floating Photobioreactor without Aeration Device. Bioresour. Technol. 2019, 293, 122018. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Xu, J.; Zong, C.; Zhu, S.; Ye, M.; Zhou, C.; Chen, H.; Yan, X. Effect of High Temperature on the Lipid Composition of Isochrysis galbana Parke in Logarithmic Phase. Aquac. Int. 2017, 25, 327–339. [Google Scholar] [CrossRef]
- Yang, S.; Li, X.; Zang, Z.; Li, J.; Wang, A.; Shi, Y.; Zhang, X.; Gu, Z.; Zheng, X.; Vasquez, H.E. Effect of Fresh and Spray-Dried Microalgal Diets on the Growth, Digestive Enzymatic Activity, and Gut Microbiota of Juvenile Winged Pearl Oyster Pteria penguin. Aquac. Rep. 2022, 25, 101251. [Google Scholar] [CrossRef]
- Takada, J.; Murase, N.; Abe, M.; Noda, M.; Suda, Y. Growth and Photosynthesis of Ulva prolifera under Different Light Quality from Light Emitting Diodes. Aquac. Sci. 2011, 59, 101–107. [Google Scholar]
- Kwan, P.P.; Banerjee, S.; Shariff, M.; Yusoff, F.M. Influence of Light on Biomass and Lipid Production in Microalgae Cultivation. Aquac. Res. 2021, 52, 1337–1347. [Google Scholar] [CrossRef]
- Li, Y.; Li, R.; Yi, X. Effects of Light Quality on Growth Rates and Pigments of Chaetoceros gracilis (Bacillariophyceae). J. Ocean. Limnol. 2020, 38, 795–801. [Google Scholar] [CrossRef]
- Ravelonandro, P.H.; Ratianarivo, D.H.; Joannis-Cassan, C.; Isambert, A.; Raherimandimby, M. Influence of Light Quality and Intensity in the Cultivation of Spirulina platensis from Toliara (Madagascar) in a Closed System. J. Chem. Technol. Biotechnol. 2008, 83, 842–848. [Google Scholar] [CrossRef]
- Shu, C.-H.; Tsai, C.-C.; Liao, W.-H.; Chen, K.-Y.; Huang, H.-C. Effects of Light Quality on the Accumulation of Oil in a Mixed Culture of Chlorella sp. and Saccharomyces cerevisiae. J. Chem. Technol. Biotechnol. 2012, 87, 601–607. [Google Scholar] [CrossRef]
- You, T.; Barnett, S.M. Effect of Light Quality on Production of Extracellular Polysaccharides and Growth Rate of Porphyridium cruentum. Biochem. Eng. J. 2004, 19, 251–258. [Google Scholar] [CrossRef]
- Metsoviti, M.N.; Papapolymerou, G.; Karapanagiotidis, I.T.; Katsoulas, N. Effect of Light Intensity and Quality on Growth Rate and Composition of Chlorella vulgaris. Plants 2019, 9, 31. [Google Scholar] [CrossRef]
- Pang, N.; Fu, X.; Fernandez, J.S.M.; Chen, S. Multilevel Heuristic LED Regime for Stimulating Lipid and Bioproducts Biosynthesis in Haematococcus pluvialis under Mixotrophic Conditions. Bioresour. Technol. 2019, 288, 121525. [Google Scholar] [CrossRef] [PubMed]
- Hallenbeck, P.C.; Grogger, M.; Mraz, M.; Veverka, D. The Use of Design of Experiments and Response Surface Methodology to Optimize Biomass and Lipid Production by the Oleaginous Marine Green Alga, Nannochloropsis gaditana in Response to Light Intensity, Inoculum Size and CO2. Bioresour. Technol. 2015, 184, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Solovchenko, A.E.; Khozin-Goldberg, I.; Didi-Cohen, S.; Cohen, Z.; Merzlyak, M.N. Effects of Light Intensity and Nitrogen Starvation on Growth, Total Fatty Acids and Arachidonic Acid in the Green Microalga Parietochloris incisa. J. Appl. Phycol. 2008, 20, 245–251. [Google Scholar] [CrossRef]
- Takeshita, T.; Ota, S.; Yamazaki, T.; Hirata, A.; Zachleder, V.; Kawano, S. Starch and Lipid Accumulation in Eight Strains of Six Chlorella Species under Comparatively High Light Intensity and Aeration Culture Conditions. Bioresour. Technol. 2014, 158, 127–134. [Google Scholar] [CrossRef]
- Yeesang, C.; Cheirsilp, B. Effect of Nitrogen, Salt, and Iron Content in the Growth Medium and Light Intensity on Lipid Production by Microalgae Isolated from Freshwater Sources in Thailand. Bioresour. Technol. 2011, 102, 3034–3040. [Google Scholar] [CrossRef]
- Mandotra, S.K.; Kumar, P.; Suseela, M.R.; Nayaka, S.; Ramteke, P.W. Evaluation of Fatty Acid Profile and Biodiesel Properties of Microalga Scenedesmus abundans under the Influence of Phosphorus, PH and Light Intensities. Bioresour. Technol. 2016, 201, 222–229. [Google Scholar] [CrossRef]
- Sirisuk, P.; Ra, C.-H.; Jeong, G.-T.; Kim, S.-K. Effects of Wavelength Mixing Ratio and Photoperiod on Microalgal Biomass and Lipid Production in a Two-Phase Culture System Using LED Illumination. Bioresour. Technol. 2018, 253, 175–181. [Google Scholar] [CrossRef]
- Babuskin, S.; Radhakrishnan, K.; Babu, P.A.S.; Sivarajan, M.; Sukumar, M. Effect of Photoperiod, Light Intensity and Carbon Sources on Biomass and Lipid Productivities of Isochrysis galbana. Biotechnol. Lett. 2014, 36, 1653–1660. [Google Scholar] [CrossRef] [PubMed]
- Krzemińska, I.; Pawlik-Skowrońska, B.; Trzcińska, M.; Tys, J. Influence of Photoperiods on the Growth Rate and Biomass Productivity of Green Microalgae. Bioprocess Biosyst. Eng. 2014, 37, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Ding, W.; Zhou, X.; Jin, W.; Han, W.; Chi, K.; Chen, Y.; Zhao, Z.; He, Z.; Jiang, G. Sub-Pilot Scale Cultivation of Tetradesmus dimorphus in Wastewater for Biomass Production and Nutrients Removal: Effects of Photoperiod, CO2 Concentration and Aeration Intensity. J. Water Process Eng. 2022, 49, 103003. [Google Scholar] [CrossRef]
- De Mooij, T.; de Vries, G.; Latsos, C.; Wijffels, R.H.; Janssen, M. Impact of Light Color on Photobioreactor Productivity. Algal Res. 2016, 15, 32–42. [Google Scholar] [CrossRef]
- Rugnini, L.; Rossi, C.; Antonaroli, S.; Rakaj, A.; Bruno, L. The Influence of Light and Nutrient Starvation on Morphology, Biomass and Lipid Content in Seven Strains of Green Microalgae as a Source of Biodiesel. Microorganisms 2020, 8, 1254. [Google Scholar] [CrossRef]
- Jeon, Y.-C.; Cho, C.-W.; Yun, Y.-S. Measurement of Microalgal Photosynthetic Activity Depending on Light Intensity and Quality. Biochem. Eng. J. 2005, 27, 127–131. [Google Scholar] [CrossRef]
- Elisabeth, B.; Rayen, F.; Behnam, T. Microalgae Culture Quality Indicators: A Review. Crit. Rev. Biotechnol. 2021, 41, 457–473. [Google Scholar] [CrossRef]
- Markou, G. Effect of Various Colors of Light-Emitting Diodes (LEDs) on the Biomass Composition of Arthrospira platensis Cultivated in Semi-Continuous Mode. Appl. Biochem. Biotechnol. 2014, 172, 2758–2768. [Google Scholar] [CrossRef]
- Atta, M.; Idris, A.; Bukhari, A.; Wahidin, S. Intensity of Blue LED Light: A Potential Stimulus for Biomass and Lipid Content in Fresh Water Microalgae Chlorella vulgaris. Bioresour. Technol. 2013, 148, 373–378. [Google Scholar] [CrossRef]
- Hultberg, M.; Jönsson, H.L.; Bergstrand, K.-J.; Carlsson, A.S. Impact of Light Quality on Biomass Production and Fatty Acid Content in the Microalga Chlorella vulgaris. Bioresour. Technol. 2014, 159, 465–467. [Google Scholar] [CrossRef]
- Teo, C.L.; Atta, M.; Bukhari, A.; Taisir, M.; Yusuf, A.M.; Idris, A. Enhancing Growth and Lipid Production of Marine Microalgae for Biodiesel Production via the Use of Different LED Wavelengths. Bioresour. Technol. 2014, 162, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Ra, C.-H.; Kang, C.-H.; Jung, J.-H.; Jeong, G.-T.; Kim, S.-K. Effects of Light-Emitting Diodes (LEDs) on the Accumulation of Lipid Content Using a Two-Phase Culture Process with Three Microalgae. Bioresour. Technol. 2016, 212, 254–261. [Google Scholar] [CrossRef] [PubMed]
- McGee, D.; Archer, L.; Fleming, G.T.A.; Gillespie, E.; Touzet, N. Influence of Spectral Intensity and Quality of Led Lighting on Photoacclimation, Carbon Allocation and High-Value Pigments in Microalgae. Photosynth. Res. 2020, 143, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.-H.; Chan, M.-C.; Liu, C.-C.; Chen, C.-Y.; Lee, W.-L.; Lee, D.-J.; Chang, J.-S. Enhancing Lutein Productivity of an Indigenous Microalga Scenedesmus obliquus Fsp-3 Using Light-Related Strategies. Bioresour. Technol. 2014, 152, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Bialevich, V.; Zachleder, V.; Bišová, K. The Effect of Variable Light Source and Light Intensity on the Growth of Three Algal Species. Cells 2022, 11, 1293. [Google Scholar] [CrossRef] [PubMed]
- Li, S.F.; Fanesi, A.; Martin, T.; Lopes, F. Biomass Production and Physiology of Chlorella vulgaris during the Early Stages of Immobilized State Are Affected by Light Intensity and Inoculum Cell Density. Algal Res. 2021, 59, 102453. [Google Scholar] [CrossRef]
- He, Q.; Yang, H.; Wu, L.; Hu, C. Effect of Light Intensity on Physiological Changes, Carbon Allocation and Neutral Lipid Accumulation in Oleaginous Microalgae. Bioresour. Technol. 2015, 191, 219–228. [Google Scholar] [CrossRef]
- Beale, S.I.; Appleman, D. Chlorophyll Synthesis in Chlorella: Regulation by Degree of Light Limitation of Growth. Plant Physiol. 1971, 47, 230–235. [Google Scholar] [CrossRef]
- Da Silva Ferreira, V.; Sant’Anna, C. Impact of Culture Conditions on the Chlorophyll Content of Microalgae for Biotechnological Applications. World J. Microbiol. Biotechnol. 2017, 33, 20. [Google Scholar] [CrossRef]
- Yoshioka, M.; Yago, T.; Yoshie-Stark, Y.; Arakawa, H.; Morinaga, T. Effect of High Frequency of Intermittent Light on the Growth and Fatty Acid Profile of Isochrysis galbana. Aquaculture 2012, 338–341, 111–117. [Google Scholar] [CrossRef]
- Che, C.A.; Kim, S.H.; Hong, H.J.; Kityo, M.K.; Sunwoo, I.Y.; Jeong, G.-T.; Kim, S.-K. Optimization of Light Intensity and Photoperiod for Isochrysis galbana Culture to Improve the Biomass and Lipid Production Using 14-L Photobioreactors with Mixed Light Emitting Diodes (LEDs) Wavelength under Two-Phase Culture System. Bioresour. Technol. 2019, 285, 121323. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, J.; Bougaran, G.; Jauffrais, T.; Lefebvre, S.; Rouxel, C.; Saint-Jean, B.; Lukomska, E.; Robert, R.; Cadoret, J.P. Effects of Blue Light on the Biochemical Composition and Photosynthetic Activity of Isochrysis sp. (T-Iso). J. Appl. Phycol. 2013, 25, 109–119. [Google Scholar] [CrossRef]
- Fernandes, B.D.; Dragone, G.M.; Teixeira, J.A.; Vicente, A.A. Light Regime Characterization in an Airlift Photobioreactor for Production of Microalgae with High Starch Content. Appl. Biochem. Biotechnol. 2010, 161, 218–226. [Google Scholar] [CrossRef]
- Sukumaran, P.; Nulit, R.; Halimoon, N.; Simoh, S.; Omar, H.; Ismail, A. Formulation of Cost-Effective Medium Using Urea as a Nitrogen Source for Arthrospira platensis Cultivation under Real Environment. ARRB 2018, 22, 1–12. [Google Scholar] [CrossRef]
- Jeffrey, S.W.; Humphrey, G.F. New Spectrophotometric Equations for Determining Chlorophylls a, b, C1 and C2 in Higher Plants, Algae and Natural Phytoplankton. Biochem. Physiol. Pflanz. 1975, 167, 191–194. [Google Scholar] [CrossRef]
- Mohsenpour, S.F.; Willoughby, N. Luminescent Photobioreactor Design for Improved Algal Growth and Photosynthetic Pigment Production through Spectral Conversion of Light. Bioresour. Technol. 2013, 142, 147–153. [Google Scholar] [CrossRef]
- Lowry, O.H. Protein Measurement with the Folin Phenol Reagent. J. biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Somani, B.L.; Khanade, J.; Sinha, R. A Modified Anthrone-Sulfuric Acid Method for the Determination of Fructose in the Presence of Certain Proteins. Anal. Biochem. 1987, 167, 327–330. [Google Scholar] [CrossRef]
- Koc, C.; Anderson, G.A.; Kommareddy, A. Use of Red and Blue Light-Emitting Diodes (LED) and Fluorescent Lamps to Grow Microalgae in a Photobioreactor. Isr. J. Aquac.-Bamidgeh 2013, 65, 20661. [Google Scholar] [CrossRef]
- Oldenhof, H.; Zachleder, V.; Ende, H. Blue Light Delays Commitment to Cell Division in Chlamydomonas Reinhardtii. Plant Biology. 2004, 6, 689–695. [Google Scholar] [CrossRef]
- Izadpanah, M.; Gheshlaghi, R.; Mahdavi, M.A.; Elkamel, A. Effect of Light Spectrum on Isolation of Microalgae from Urban Wastewater and Growth Characteristics of Subsequent Cultivation of the Isolated Species. Algal Res. 2018, 29, 154–158. [Google Scholar] [CrossRef]
- Gómez-Loredo, A.; Benavides, J.; Rito-Palomares, M. Growth Kinetics and Fucoxanthin Production of Phaeodactylum tricornutum and Isochrysis galbana Cultures at Different Light and Agitation Conditions. J. Appl. Phycol. 2016, 28, 849–860. [Google Scholar] [CrossRef]
- Li, Y.; Liu, J. Analysis of Light Absorption and Photosynthetic Activity by Isochrysis galbana under Different Light Qualities. Aquac. Res. 2020, 51, 2893–2902. [Google Scholar] [CrossRef]
- Vadiveloo, A.; Moheimani, N.R.; Cosgrove, J.J.; Bahri, P.A.; Parlevliet, D. Effect of Different Light Spectra on the Growth and Productivity of Acclimated Nannochloropsis sp. (Eustigmatophyceae). Algal Res. 2015, 8, 121–127. [Google Scholar] [CrossRef]
- Carvalho, A.P.; Silva, S.O.; Baptista, J.M.; Malcata, F.X. Light Requirements in Microalgal Photobioreactors: An Overview of Biophotonic Aspects. Appl. Microbiol. Biotechnol. 2011, 89, 1275–1288. [Google Scholar] [CrossRef]
- Kommareddy, A.; Anderson, G. Study of Light as a Parameter in the Growth of Algae in a Photo-Bio Reactor (PBR). In Proceedings of the 2003 ASAE Annual Meeting, Las Vegas, NV, USA, 27–30 July 2003. [Google Scholar] [CrossRef]
- Ueno, Y.; Aikawa, S.; Kondo, A.; Akimoto, S. Adaptation of Light-Harvesting Functions of Unicellular Green Algae to Different Light Qualities. Photosynth. Res. 2019, 139, 145–154. [Google Scholar] [CrossRef]
- Masojídek, J.; Torzillo, G.; Koblížek, M. Handbook of Microalgal Culture: Applied Phycology and Biotechnolog, 2nd ed.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2004; ISBN 9781118567166. [Google Scholar]
- Wang, C.-Y.; Fu, C.-C.; Liu, Y.-C. Effects of Using Light-Emitting Diodes on the Cultivation of Spirulina platensis. Biochem. Eng. J. 2007, 37, 21–25. [Google Scholar] [CrossRef]
- Das, P.; Lei, W.; Aziz, S.S.; Obbard, J.P. Enhanced Algae Growth in Both Phototrophic and Mixotrophic Culture under Blue Light. Bioresour. Technol. 2011, 102, 3883–3887. [Google Scholar] [CrossRef] [PubMed]
- Holzinger, A.; Lütz, C. Algae and UV Irradiation: Effects on Ultrastructure and Related Metabolic Functions. Micron 2006, 37, 190–207. [Google Scholar] [CrossRef]
- Kuwano, K.; Abe, N.; Nishi, Y.; Seno, H.; Nishihara, G.N.; Iima, M.; Zachleder, V. Growth and Cell Cycle of Ulva Compressa (Ulvophyceae) under LED Illumination. J. Phycol. 2014, 50, 744–752. [Google Scholar] [CrossRef]
- Tsekos, I.; Niell, F.X.; Aguilera, J.; López-Figueroa, F.; Delivopoulos, S.G. Ultrastructure of the Vegetative Gametophytic Cells of Porphyra Leucosticta (Rhodophyta) Grown in Red, Blue and Green Light. Phycol. Res. 2002, 50, 251–264. [Google Scholar] [CrossRef]
- Gorai, T.; Katayama, T.; Obata, M.; Murata, A.; Taguchi, S. Low Blue Light Enhances Growth Rate, Light Absorption, and Photosynthetic Characteristics of Four Marine Phytoplankton Species. J. Exp. Mar. Biol. Ecol. 2014, 459, 87–95. [Google Scholar] [CrossRef]
- Danesi, E.D.G.; Rangel-Yagui, C.O.; Carvalho, J.C.M.; Sato, S. Effect of Reducing the Light Intensity on the Growth and Production of Chlorophyll by Spirulina platensis. Biomass Bioenergy 2004, 26, 329–335. [Google Scholar] [CrossRef]
- George, B.; Pancha, I.; Desai, C.; Chokshi, K.; Paliwal, C.; Ghosh, T.; Mishra, S. Effects of Different Media Composition, Light Intensity and Photoperiod on Morphology and Physiology of Freshwater Microalgae Ankistrodesmus falcatus-A Potential Strain for Bio-Fuel Production. Bioresour. Technol. 2014, 171, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Guðmundsson, Ó.; Paglia, G.; Herjólfsson, G.; Andrésson, Ó.S.; Palsson, B.Ø.; Brynjólfsson, S. Enhancement of Carotenoid Biosynthesis in the Green Microalga Dunaliella salina with Light-Emitting Diodes and Adaptive Laboratory Evolution. Appl. Microbiol. Biotechnol. 2013, 97, 2395–2403. [Google Scholar] [CrossRef]
- Gatamaneni Loganathan, B.; Orsat, V.; Lefsrud, M.; Wu, B.S. A Comprehensive Study on the Effect of Light Quality Imparted by Light-Emitting Diodes (LEDs) on the Physiological and Biochemical Properties of the Microalgal Consortia of Chlorella variabilis and Scenedesmus obliquus Cultivated in Dairy Wastewater. Bioprocess Biosyst. Eng. 2020, 43, 1445–1455. [Google Scholar] [CrossRef]
- Del Pilar Sánchez-Saavedra, M.; Maeda-Martínez, A.N.; Acosta-Galindo, S. Effect of Different Light Spectra on the Growth and Biochemical Composition of Tisochrysis lutea. J. Appl. Phycol. 2016, 28, 839–847. [Google Scholar] [CrossRef]
- Rivkin, R. Influence of Irradiance and Spectral Quality on the Carbon Metabolism of Phytoplankton I. Photosynthesis, Chemical Composition and Growth. Mar. Ecol. Prog. Ser. 1989, 55, 291–304. [Google Scholar] [CrossRef]
- Brinkmann, G.; Senger, H. The Development of Structure and Function in Chloroplasts of Greening Mutants of Scenedesmus IV. Blue Light-Dependent Carbohydrate and Protein Metabolism. Plant Cell Physiol. 1978, 19, 1427–1437. [Google Scholar] [CrossRef]
- Hammer, A.; Schumann, R.; Schubert, H. Light and Temperature Acclimation of Rhodomonas salina (Cryptophyceae): Photosynthetic Performance. Aquat. Microb. Ecol. 2002, 29, 287–296. [Google Scholar] [CrossRef]
| Light Condition | Dry Weight (g/L) | Specific Growth Rate (µ/day) | Productivity (g/L/day) |
|---|---|---|---|
| White | 1.1761± 0.0212 a | 0.2948 ± 0.0011 c | 0.0728 ± 0.0013 a |
| Red | 0.5683 ± 0.0284 c | 0.4431 ± 0.0055 a | 0.0620 ± 0.0032 b |
| Blue | 0.6278 ± 0.0184 c | 0.4089 ± 0.0029 b | 0.0617 ± 0.0018 b |
| Green | 0.8928 ± 0.0572 b | 0.2466 ± 0.0035 d | 0.0490 ± 0.0032 c |
| Yellow | 0.7902 ± 0.0555 b | 0.2158 ± 0.0034 e | 0.0390 ± 0.0028 d |
| Light Color | Maximum Total Protein Content (mg/L) | Maximum Carbohydrate Content (mg/L) |
|---|---|---|
| White | 524.1935 ± 6.5846 a | 8.5859 ± 0.0206 d |
| Red | 403.2258 ± 6.5846 c | 9.1667 ± 0.0206 c |
| Blue | 440.0325 ± 3.3723 b | 24.4697 ± 0.0206 a |
| Green | 406.6346 ± 3.3548 c | 9.3687 ± 0.0206 b |
| Yellow | 454.9462 ± 3.4375 b | 7.8455 ±0.1247 e |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lv, B.; Liu, Z.; Chen, Y.; Lan, S.; Mao, J.; Gu, Z.; Wang, A.; Yu, F.; Zheng, X.; Vasquez, H.E. Effect of Different Colored LED Lighting on the Growth and Pigment Content of Isochrysis zhanjiangensis under Laboratory Conditions. J. Mar. Sci. Eng. 2022, 10, 1752. https://doi.org/10.3390/jmse10111752
Lv B, Liu Z, Chen Y, Lan S, Mao J, Gu Z, Wang A, Yu F, Zheng X, Vasquez HE. Effect of Different Colored LED Lighting on the Growth and Pigment Content of Isochrysis zhanjiangensis under Laboratory Conditions. Journal of Marine Science and Engineering. 2022; 10(11):1752. https://doi.org/10.3390/jmse10111752
Chicago/Turabian StyleLv, Bu, Ziling Liu, Yu Chen, Shuaiqin Lan, Jing Mao, Zhifeng Gu, Aimin Wang, Feng Yu, Xing Zheng, and Hebert Ely Vasquez. 2022. "Effect of Different Colored LED Lighting on the Growth and Pigment Content of Isochrysis zhanjiangensis under Laboratory Conditions" Journal of Marine Science and Engineering 10, no. 11: 1752. https://doi.org/10.3390/jmse10111752
APA StyleLv, B., Liu, Z., Chen, Y., Lan, S., Mao, J., Gu, Z., Wang, A., Yu, F., Zheng, X., & Vasquez, H. E. (2022). Effect of Different Colored LED Lighting on the Growth and Pigment Content of Isochrysis zhanjiangensis under Laboratory Conditions. Journal of Marine Science and Engineering, 10(11), 1752. https://doi.org/10.3390/jmse10111752








