Sorption of Tannin and Related Phenolic Compounds and Effects on Extraction of Soluble-N in Soil Amended with Several Carbon Sources
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field site and Sampling
| Amendment | pHwater (1:1) | CEC (cmolc·kg−1) | Total-C (%) | Total- N (%) | C:N |
|---|---|---|---|---|---|
| Cotton fiber | 5.23 (0.02) | 14.7 (0.2) | 1.65 (0.08) | 0.16 (0.01) | 10.4 (0.3) |
| Wheat residue | 5.22 (0.03) | 15.2 (0.3) | 1.65 (0.06) | 0.16 (0.01) | 10.4 (0.3) |
| No amendment | 5.21 (0.03) | 14.4 (0.6) | 1.71 (0.20) | 0.16 (0.02) | 10.7 (0.5) |
| Brassica crop | 5.22 (0.03) | 15.0 (0.3) | 1.88 (0.08) | 0.18 (0.01) | 10.3 (0.3) |
| Brassica residue | 5.19 (0.03) | 16.3 (0.3) | 1.90 (0.11) | 0.18 (0.01) | 10.4 (0.3) |
| Sucrose | 5.20 (0.03 | 14.6 (0.4) | 1.97 (0.20) | 0.16 (0.01) | 11.8 (0.7) |
| Grass | 5.22 (0.04) | 15.6 (0.2) | 2.01 (0.10) | 0.18 (0.01) | 10.9 (0.2) |
| Compost | 5.14 (0.05) | 15.9 (0.6) | 2.23 (0.27) | 0.18 (0.01) | 12.2 (1.2) |
| Alfalfa | 5.20 (0.04) | 15.6 (0.4) | 2.26 (0.13) | 0.22 (0.01) | 10.6 (0.5) |
| Manure | 5.21 (0.05) | 17.5 (0.4) | 2.50 (0.28) | 0.24 (0.02) | 10.4 (0.3) |
| Wood | 5.20 (0.05) | 15.2 (0.4) | 2.75 (0.17) | 0.18 (0.01) | 15.4 (1.1) |
| Biosolids | 5.18 (0.04) | 15.7 (0.6) | 2.90 (0.23) | 0.30 (0.02) | 9.6 (0.2) |
2.2. Sorption of Test Compounds
2.3. Test Compounds
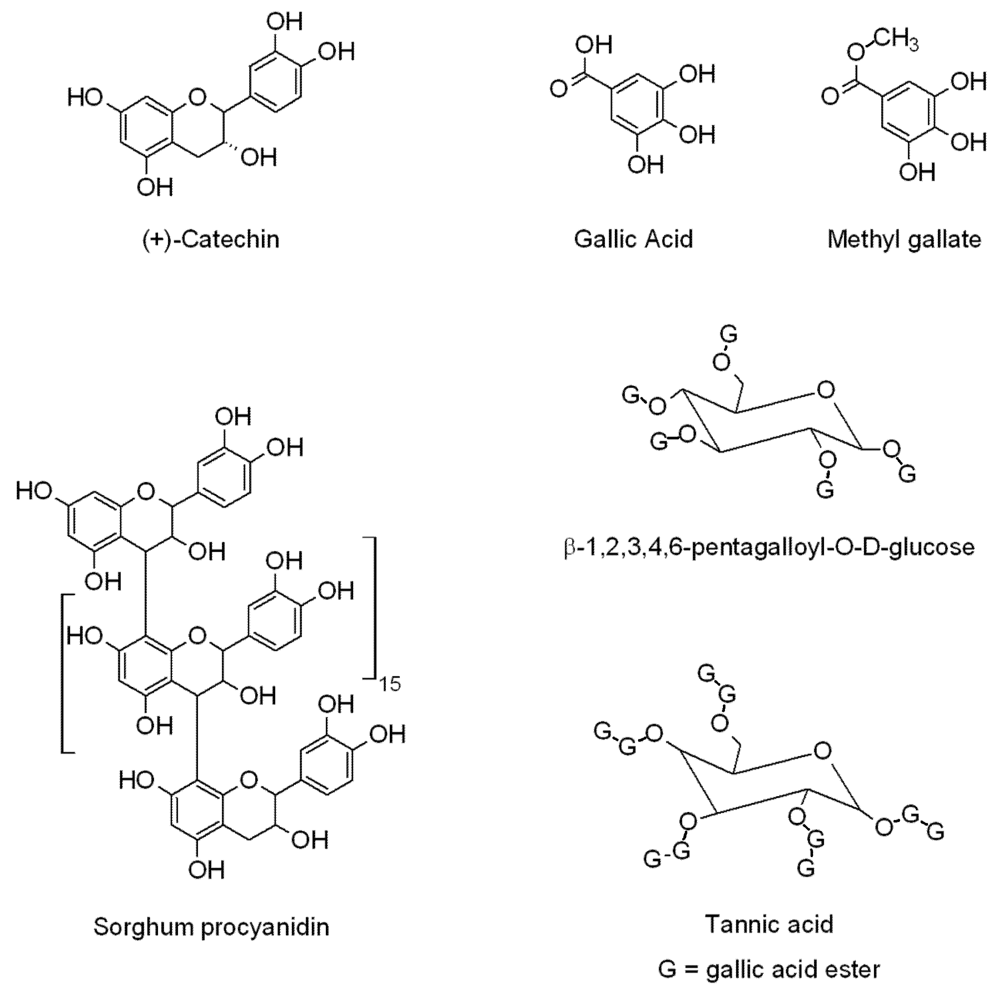
2.4. Procedure
| Compound/Treatment | Class | Source | Compound characteristics | Treatment¶ | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| MW† | C‡ | N‡ | C (g mol−1) | KOW§ | Soluble-C | Soluble-N | Phenolics# μmol GA | Solution pH | |||
| (%) | (%) | (mg/kg soil) | (mg/kg soil) | equiv/kg | 1mg/mL) | ||||||
| Methyl 3,4,5-trihydroxybenzoate, 98% | Phenolic organic ester | Indofine Chemical Co., Hillsborough, NJ | 184 | 51.7 | 0.084 | 96 | 6.3 | 5122 | 0.6 | 41.7 | 4.4 |
| (Methyl Gallate) (MG) | |||||||||||
| Gallic Acid, Certified (GA) | Phenolic organic acid | Fisher Scientific, Pittsburgh, PA | 170 | 47.7 | 0.106 | 85 | 0.3 | 4654 | 0.6 | 53.7 | 3.3 |
| Tannic Acid, Certified (TA) | Mixture of gallotannins | Fisher Scientific, Pittsburgh, PA | 902 | 49.4 | 0.142 | 474 | ND | 4669 | 2.0 | 30.1 | 3.5 |
| β-1,2,3,4,6 penta-O-galloyl-D-glucose (PGG) | Gallotannin | Purified from Tannic Acid (Fisher) | 941 | 49.7 | 0.099 | 492 | 129 | 4919 | 0.6 | 29.4 | 5.1 |
| (+)-Catechin hydrate, >98% | Flavonoid | Sigma, | 290 | 61.6 | 0 | 180 | 2.4 | 5810 | 0.7 | 32.6 | 5.6 |
| (CAT) | St Louis, MO | ||||||||||
| [(4β->8)-epicatechin}15-(4β->8)-catechin | Polymeric flavonoid | Sorghum grain | 4624 | 48.6 | 0.094 | 2880 | 0.002 | 4833 | 6.8 | 13.7 | 6.0 |
| (Sorghum Proanthocyanidin) (SOR) | [Sorghum bicolor (L.) Moench] | ||||||||||
2.5. Measurements of Final CEC and Total Soil -C
2.6. Statistical Analysis
3. Results and Discussion
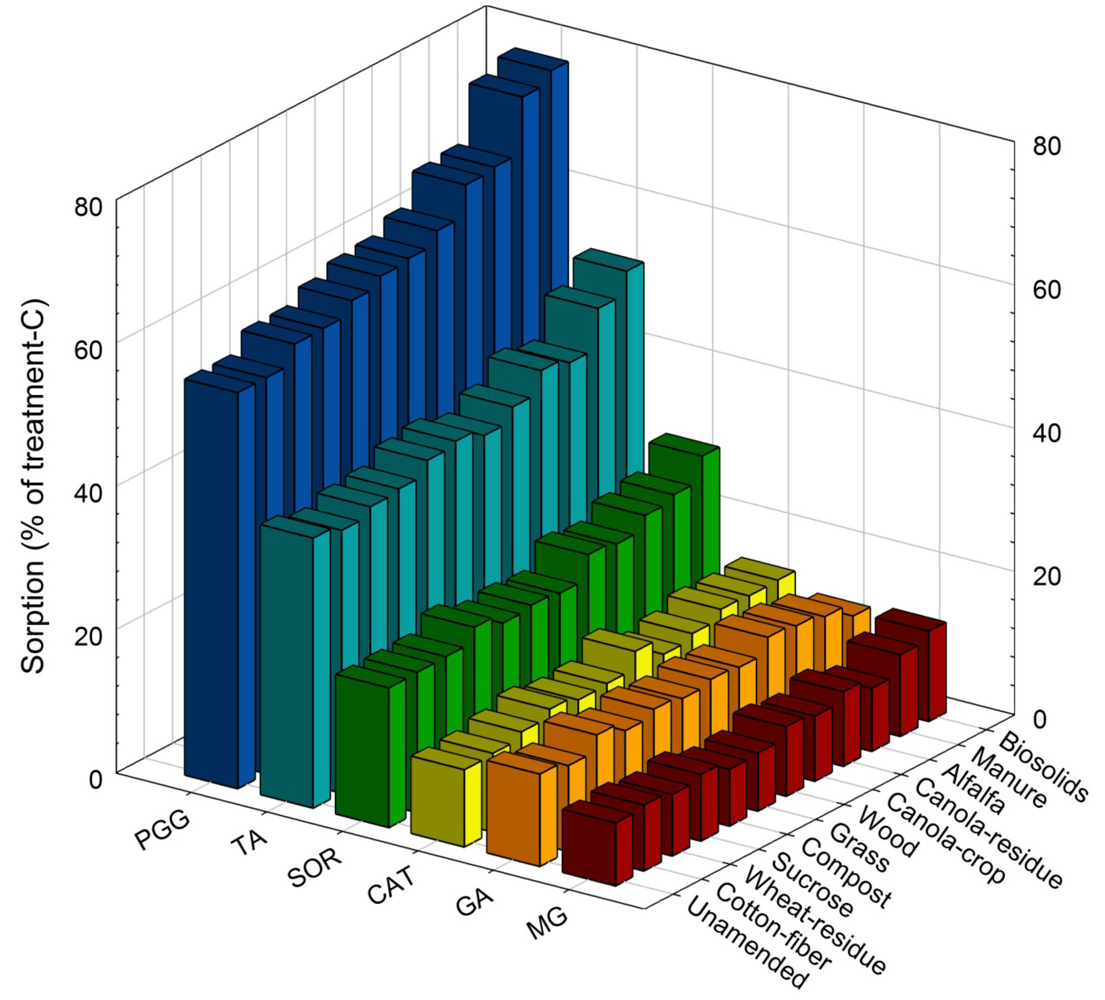
3.1. Sorption of Treatment-C
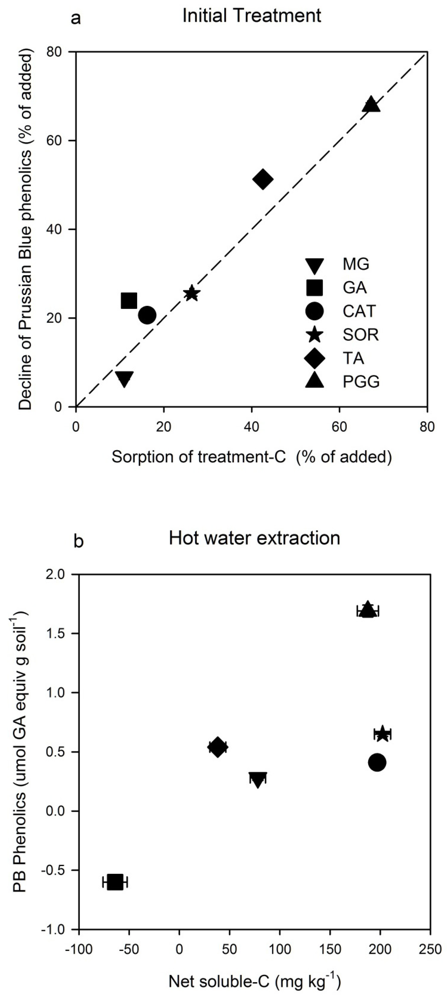
| Amendment | Sorption of treatment-C | Net Soluble-N | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (% of amount added) ‡ | (% difference from Control) ‡ | |||||||||||||||
| Control Soluble-C | MG | GA | CAT | SOR | TA | PGG | Average | Control Soluble-N | MG | GA | CAT | SOR | TA | PGG | Average | |
| (mg/kg) | (n = 48) | (mg/kg) | (n = 48) | |||||||||||||
| No amendment | 314 (34) | 10.3ab4 | 12.4a4 | 13.9a4 | 23.2c3 | 39.0c2 | 60.0cd1 | 26.5E | 29 (2) | 21.5a1 | −13.8a3 | 7.8a2 | 1.0a2 | −3.4a23 | 2.4a2 | 2.6 |
| Cotton fiber | 325 (27) | 10.8ab4 | 11.6a4 | 13.9a4 | 22.8c3 | 37.9c2 | 59.3d1 | 26.1E | 19 (1) | 20.3ab1 | −27.9a4 | 1.4a2 | −11.8a3 | −19.3a34 | −10.6a3 | −8.0 |
| Compost | 330 (22) | 8.8a5 | 10.6a5 | 15.6a4 | 23.8bc3 | 39.8c2 | 64.3bcd | 27.2DE | 42 (4) | 15.4ab1 | −16.4a4 | 7.0a12 | 1.4a23 | −9.6a34 | −1.4a23 | −0.6 |
| Sucrose | 346 (36) | 10.6ab5 | 11.8a5 | 16.1a4 | 25.2bc3 | 39.3c2 | 61.4cd1 | 27.4CDE | 32 (5) | 17.2ab1 | −20.4a4 | −1.2a2 | −7.9a23 | −13.6a34 | −6.5a23 | −5.4 |
| Wheat residue | 332 (30) | 10.4ab5 | 12.9a45 | 15.3a4 | 24.3bc3 | 38.9c2 | 62.9cd1 | 27.4CDE | 32 (3) | 18.7ab1 | −18.1a4 | 6.4a2 | −2.3a23 | −10.7a34 | −4.4a23 | −1.7 |
| Grass | 373 (17) | 10.0ab5 | 11.4a5 | 15.6a4 | 24.8bc3 | 41.8bc2 | 65.3bcd1 | 28.1CDE | 27 (2) | 20.4ab1 | −22.0a4 | 4.7a2 | −7.2a3 | −14.8a34 | −8.9a3 | −4.6 |
| Wood | 425 (26) | 11.4ab5 | 12.7a5 | 18.4a4 | 25.4bc3 | 39.9c2 | 64.5bcd1 | 28.7BCDE | 23 (4) | 19.7ab1 | −25.9a4 | −2.1a2 | −11.9a23 | −18.9a34 | −9.6a23 | −8.1 |
| Brassica crop | 345 (22) | 10.7ab5 | 12.8a45 | 15.8a4 | 27.5abc3 | 42.8bc2 | 67.8bc1 | 29.6BCDE | 64 (18) | 13.7ab1 | −18.4a4 | 3.0a12 | −1.6a23 | −11.8a34 | −4.9a23 | −3.3 |
| Brassica residue | 343 (24) | 11.6ab5 | 12.4a45 | 16.3a4 | 26.6bc3 | 44.6bc2 | 71.9ab1 | 30.6BCD | 55 (9) | 12.6ab1 | −19.0a4 | 4.1a12 | −4.8a23 | −12.5a34 | −7.2a234 | −4.5 |
| Alfalfa | 394 (20) | 10.8ab5 | 12.9a5 | 18.7a4 | 29.7ab3 | 44.2bc2 | 72.5ab1 | 31.5ABC | 119 (31) | 10.0ab1 | −17.7a4 | 3.5a12 | −1.6a123 | −12.6a34 | −4.1a23 | −3.8 |
| Manure | 428 (11) | 12.6ab5 | 10.6a5 | 17.8a4 | 29.7ab3 | 49.0ab2 | 77.9a1 | 33.0AB | 106 (23) | 11.9ab1 | −16.2a4 | 5.3a12 | −2.8a23 | −14.2a34 | −6.0a234 | −3.7 |
| Biosolids | 368 (15) | 14.1a45 | 12.7a5 | 17.4a4 | 33.7a3 | 53.5a2 | 78.4a1 | 35.0A | 136 (28) | 4.5b1 | −15.2a3 | 0.3a1 | −1.8a12 | −13.1a23 | −6.3a123 | −5.3 |
| Average | 11.0F | 12.1E | 16.2D | 26.4C | 42.6B | 67.2A | 15.5A | −19.2E | 3.4B | −4.3C | −12.9D | −5.6C | ||||
| (n = 96) | ||||||||||||||||
| Amendment | Control Soluble-C | Treatment-C removed by hot water (% change from Control) ‡ | Average | Control Soluble-N | Net Soluble-N (% change from Control) ‡ | Average | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (mg/kg) | MG | GA | CAT | SOR | TA | PGG | (n = 48) | (mg/kg) | MG | GA | CAT | SOR | TA | PGG | (n = 48) | |
| No ammendment | 603 (102) | 16.1a3 | 0.0ab4 | 35.1a12 | 32.2a2 | 14.9a3 | 48.0a1 | 24.4 | 46 (9) | −12.9a2 | −21.8ab23 | 4.2a1 | −1.2ab1 | −24.4a3 | −21.2a23 | −12.9 A |
| Cotton fiber | 598 (63) | 16.6a23 | −1.0ab4 | 31.6a1 | 28.8a12 | 12.7a3 | 38.3ab1 | 21.2 | 46 (5) | −13.4a2 | −26.0abc3 | 1.8a1 | −3.7b1 | −28.2a3 | −25.3a3 | −15.8 ABC |
| Compost | 822 (123) | 9.7a2 | −10.2ab3 | 29.2a1 | 26.9a1 | 2.3a2 | 37.5ab1 | 15.9 | 68 (11) | −18.1a2 | −32.1bc3 | −2.8a1 | −2.5ab1 | −31.4a3 | −25.6a23 | −18.8 ABC |
| Sucrose | 751 (142) | 14.0a23 | −1.7ab4 | 29.7a1 | 25.8a12 | 11.4a3 | 31.4abcd1 | 18.4 | 52 (7) | −14.7a2 | −24.7abc3 | 2.1a1 | −3.9b1 | −27.2a3 | −26.4a3 | −15.8 ABC |
| Wheat residue | 694 (73) | 14.0a2 | −6.6ab3 | 31.2a1 | 34.0a1 | 9.5a2 | 38.6ab1 | 20.1 | 53 (5) | −15.4a2 | −28.7bc3 | 0.7a1 | −0.9ab1 | −28.5a3 | −27.0a3 | −16.7 ABC |
| Grass | 791 (37) | 12.6a23 | −9.2ab4 | 23.7a12 | 26.2a1 | 6.3a3 | 26.6abcd1 | 14.4 | 65 (3) | −16.0a2 | −32.0bc3 | −4.6a1 | −4.8b1 | −31.0a3 | −29.2a3 | −19.6 ABC |
| Wood | 890 (92) | 12.8a23 | −2.6ab4 | 26.5a1 | 31.1a1 | 4.8a34 | 19.4bcd12 | 15.3 | 56 (7) | −14.3a2 | −25.8abc3 | −0.5a1 | 0.7ab1 | −29.9a3 | −27.1a3 | −16.1 ABC |
| Brassica crop | 707 (67) | 14.1a2 | −0.4ab3 | 31.0a1 | 29.7a1 | 12.2a2 | 35.6abc1 | 20.3 | 56 (5) | −14.2a2 | −23.8abc3 | 1.5a1 | −0.1ab1 | −27.4a3 | −27.1a3 | −15.2 ABC |
| Brassica residue | 815 (103) | 10.0a2 | −13.7b3 | 27.4a1 | 26.4a1 | 4.0a2 | 29.8abcd1 | 14.0 | 69 (9) | −19.9a2 | −35.7c3 | −4.6a1 | −9.2b1 | −34.7a3 | −33.0a3 | −22.9 C |
| Alfalfa | 952 (76) | 11.9a2 | −7.7ab3 | 27.5a1 | 24.8a1 | 5.4a2 | 26.5abcd1 | 15.0 | 86 (9) | −15.3a2 | −27.7abc3 | −2.6a1 | −2.9ab1 | −29.5a3 | −27.4a3 | −17.5 ABC |
| Manure | 1055 (120) | 9.0a23 | −13.8ab4 | 22.5a1 | 20.8a12 | 2.0a3 | 12.9cd123 | 8.9 | 102 (11) | −17.5a2 | −31.1bc3 | −5.8a1 | −5.1b1 | −31.4a3 | −33.5a3 | −20.7 BC |
| Biosolids | 810 (82) | 10.2a23 | 4.7a3 | 22.9a12 | 30.3a1 | 10.7a23 | 9.6d3 | 14.7 | 85 (8) | −15.4a3 | −15.7a3 | −5.8a2 | 9.9a1 | −27.3a4 | −31.0a4 | −14.2 AB |
| Average | 12.6 B | −5.2 D | 28.2 A | 28.2 A | 8.0 C | 29.5 A | −15.6 B | −27.1 C | −1.4 A | −2.0 A | −29.1 D | −27.8 CD | ||||
| (n = 96) | ||||||||||||||||
3.2. Effects on Soluble-N

3.3. Total Soil-C and CEC
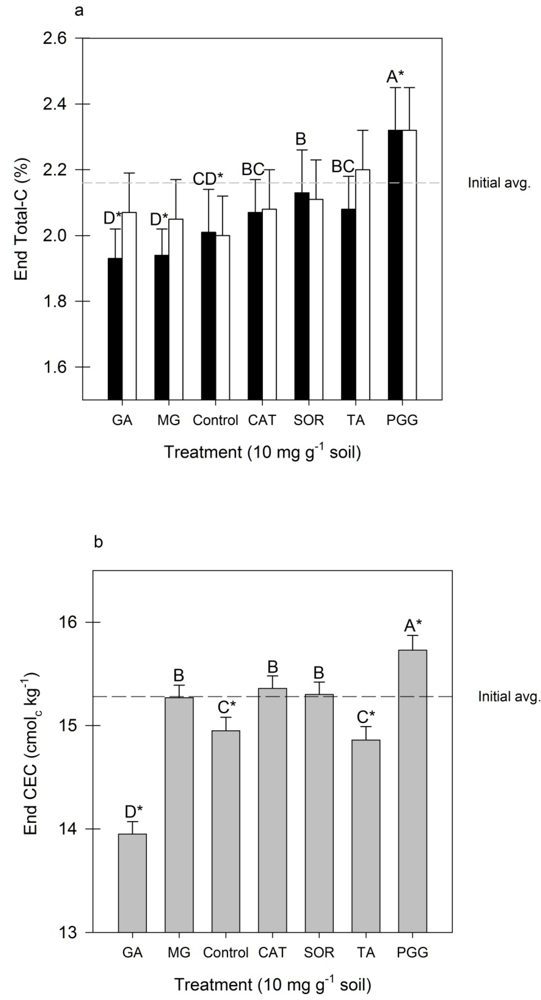
4. Conclusions
Disclaimer
Acknowledgements
References
- Kraus, T.E.C.; Dahlgren, R.A.; Zasoski, R.J. Tannins in nutrient dynamics of forest ecosystems—A review. Plant Soil 2003, 256, 41–66. [Google Scholar] [CrossRef]
- Hattenschwiler, S.; Vitousek, P.M. The role of polyphenols in terrestrial ecosystem nutrient cycling. Trends Ecol. Evol. 2000, 15, 238–243. [Google Scholar] [CrossRef]
- Norris, C.; Preston, C.; Hogg, K.; Titus, B. The influence of condensed tannin structure on rate of microbial mineralization and reactivity to chemical assays. J. Chem. Ecol. 2011, 37, 311–319. [Google Scholar] [CrossRef]
- Horner, J.D.; Gosz, J.R.; Cates, R.G. The role of carbon-based plant secondary metabolites in decomposition in terrestrial ecosystems. Am. Nat. 1988, 132, 869–883. [Google Scholar]
- Adamczyk, B.; Adamczyk, S.; Smolander, A.; Kitunen, V. Tannic acid and norway spruce condensed tannins can precipitate various organic nitrogen compounds. Soil Biol. Biochem. 2011, 43, 628–637. [Google Scholar] [CrossRef]
- Rillig, M.C.; Caldwell, B.A.; Wösten, H.A.B.; Sollins, P. Role of proteins in soil carbon and nitrogen storage: Controls on persistence. Biogeochemistry 2007, 85, 25–44. [Google Scholar] [CrossRef]
- Kaal, J.; Nierop, K.G.J.; Verstraten, J.M. Retention of tannic acid and condensed tannin by Fe-oxide-coated quartz sand. J. Colloid Interface Sci. 2005, 287, 72–79. [Google Scholar] [CrossRef]
- Halvorson, J.J.; Gonzalez, J.M.; Hagerman, A.E.; Smith, J.L. Sorption of tannin and related phenolic compounds and effects on soluble-N in soil. Soil Biol. Biochem. 2009, 41, 2002–2010. [Google Scholar] [CrossRef]
- Halvorson, J.J.; Gonzalez, J.M.; Hagerman, A.E. Repeated applications of tannins and related phenolic compounds are retained by soil and affect cation exchange capacity. Soil Biol. Biochem. 2011, 43, 1139–1147. [Google Scholar] [CrossRef]
- Zibilske, L.M.; Bradford, J.M. Oxygen effects on carbon, polyphenols, and nitrogen mineralization potential in soil. Soil Sci. Soc. Am. J. 2007, 71, 133–139. [Google Scholar]
- Joanisse, G.D.; Bradley, R.L.; Preston, C.M.; Munson, A.D. Soil enzyme inhibition by condensed litter tannins may drive ecosystem structure and processes: The case of kalmia angustifolia. New Phytol. 2007, 175, 535–546. [Google Scholar] [CrossRef]
- Halvorson, J.J.; Gonzalez, J.M. Tannic acid reduces recovery of water-soluble carbon and nitrogen from soil and affects the composition of Bradford-reactive soil protein. Soil Biol. Biochem. 2008, 40, 186–197. [Google Scholar] [CrossRef]
- Appel, H.M. Phenolics in ecological interactions: The importance of oxidation. J. Chem. Ecol. 1993, 19, 1521–1552. [Google Scholar] [CrossRef]
- Kinraide, T.B.; Hagerman, A.E. Interactive intoxicating and ameliorating effects of tannic acid, aluminum (Al3+), copper (Cu2+), and selenate (SeO42−) in wheat roots: A descriptive and mathematical assessment. Physiol. Plant. 2010, 139, 68–79. [Google Scholar] [CrossRef]
- Nina, W.; Ronald, L.H. Plant litter chemistry and mycorrhizal roots promote a nitrogen feedback in a temperate forest. J. Ecol. 2009, 97, 528–536. [Google Scholar] [CrossRef]
- Talbot, J.; Finzi, A. Differential effects of sugar maple, red oak, and hemlock tannins on carbon and nitrogen cycling in temperate forest soils. Oecologia 2008, 155, 583–592. [Google Scholar] [CrossRef]
- Mutabaruka, R.; Hairiah, K.; Cadisch, G. Microbial degradation of hydrolysable and condensed tannin polyphenol-protein complexes in soils from different land-use histories. Soil Biol. Biochem. 2007, 39, 1479–1492. [Google Scholar] [CrossRef]
- Souto, X.C.; Chiapusio, G.; Pellissier, F. Relationships between phenolics and soil microorganisms in spruce forests: Significance for natural regeneration. J. Chem. Ecol. 2000, 26, 2025–2034. [Google Scholar] [CrossRef]
- Bhat, T.K.; Singh, B.; Sharma, O.P. Microbial degradation of tannins—A current perspective. Biodegradation 1998, 9, 343–357. [Google Scholar] [CrossRef]
- Fierer, N.; Schimel, J.P.; Cates, R.G.; Zou, J. Influence of balsam poplar tannin fractions on carbon and nitrogen dynamics in alaskan taiga floodplain soils. Soil Biol. Biochem. 2001, 33, 1827–1839. [Google Scholar] [CrossRef]
- Schimel, J.P.; Cates, R.G.; Ruess, R. The role of balsam poplar secondary chemicals in controlling soil nutrient dynamics through succession in the alaskan taiga. Biogeochemistry 1998, 42, 221–234. [Google Scholar] [CrossRef]
- Kraus, T.E.C.; Zasoski, R.J.; Dahlgren, R.A.; Horwath, W.R.; Preston, C.M. Carbon and nitrogen dynamics in a forest soil amended with purified tannins from different plant species. Soil Biol. Biochem. 2004, 36, 309–321. [Google Scholar] [CrossRef]
- Kraus, T.E.C.; Yu, Z.; Preston, C.M.; Dahlgren, R.A.; Zasoski, R.J. Linking chemical reactivity and protein precipitation to structural characteristics of foliar tannins. J. Chem. Ecol. 2003, 29, 703–730. [Google Scholar] [CrossRef]
- Madritch, M.D.; Jordan, L.M.; Lindroth, R.L. Interactive effects of condensed tannin and cellulose additions on soil respiration. Can. J. For. Res. 2007, 37, 2063–2067. [Google Scholar] [CrossRef]
- Schweitzer, J.A.; Bailey, J.K.; Hart, S.C.; Wimp, G.M.; Chapman, S.K.; Whitham, T.G. The interaction of plant genotype and herbivory decelerate leaf litter decomposition and alter nutrient dynamics. Oikos 2005, 110, 133–145. [Google Scholar] [CrossRef]
- Castells, E. Indirect Effects of Phenolics on Plant Performance by Altering Nitrogen Cycling: Another Mechanism of Plant–Plant Negative Interactions. In Allelopathy in Sustainable Agriculture and Forestry; Zeng, R.S., Mallik, A.U., Luo, S.M., Eds.; Springer: New York, NY, USA, 2008; pp. 137–156. [Google Scholar]
- Wurzburger, N.; Hendrick, R.L. Plant litter chemistry and mycorrhizal roots promote a nitrogen feedback in a temperate forest. J. Ecol. 2009, 97, 528–536. [Google Scholar] [CrossRef]
- Bending, G.D.; Read, D.J. Effects of the soluble polyphenol tannic acid on the activities of ericoid and ectomycorrhizal fungi. Soil Biol. Biochem. 1996, 28, 1595–1602. [Google Scholar] [CrossRef]
- Bending, G.D.; Read, D.J. Nitrogen mobilization from protein-polyphenol complex by ericoid and ectomycorrhizal fungi. Soil Biol. Biochem. 1996, 28, 1603–1612. [Google Scholar] [CrossRef]
- Halvorson, J.J.; Gonzalez, J.M. Bradford reactive soil protein in appalachian soils: Distribution and response to incubation, extraction reagent and tannins. Plant Soil 2006, 286, 339–356. [Google Scholar] [CrossRef]
- Halvorson, J.J.; Gonzalez, J.M.; Hagerman, A.E. Changes in soluble-N in forest and pasture soil after repeated applications of tannins and related phenolic compounds. Int. J. Agron. Spec. Ed. N Miner. Prod. Agric. 2012, in press. [Google Scholar]
- Rasmussen, P.E.; Parton, W.J. Long-term effects of residue management in wheat-fallow: I. Inputs, yield, and soil organic matter. Soil Sci. Soc. Am. J. 1994, 58, 523–530. [Google Scholar]
- Zuzel, J.F.; Allmaras, R.R.; Greenwalt, R.N. Temporal distribution of runoff and soil erosion at a site in northeastern Oregon. J. Soil Water Conserv. 1993, 48, 373–378. [Google Scholar]
- Nelson, D.W.; Sommers, L.E. Total Carbon, Organic Carbon and Organic Matter. In Methods of Soil Analysis Part 3: Chemical Methods. No 5. In the Soil Science Society of America Books Series; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnson, C.T., Sumner, M.E., Eds.; Soil Science Society of America, Inc.: Madison, WI, USA, 1996; pp. 961–1010. [Google Scholar]
- Ciesielski, H.; Sterckeman, T. Determination of cation exchange capacity and exchangeable cations in soils by means of cobalt hexamine trichloride. Effects of experimental conditions. Agronomie 1997, 17, 1–7. [Google Scholar] [CrossRef]
- Ciesielski, H.; Sterckeman, T. A comparison between three methods for the determination of cation exchange capacity and exchangeable cations in soils. Agronomie 1997, 17, 9–16. [Google Scholar] [CrossRef]
- ISO 23470:2007, Soil quality- determination of effective cation exchange capacity (cec) and exchangeable cations using a hexamminecobalt trichloride solution. ISO/TC 190, Soil quality Subcommitte SC3, Chemical methods and soil characteristics. 2007.
- Ghani, A.; Dexter, M.; Perrott, K.W. Hot-water extractable carbon in soils: A sensitive measurement for determining impacts of fertilisation, grazing and cultivation. Soil Biol. Biochem. 2003, 35, 1231–1243. [Google Scholar] [CrossRef]
- Curtin, D.; Wright, C.E.; Beare, M.H.; McCallum, F.M. Hot water-extractable nitrogen as an indicator of soil nitrogen availability. Soil Sci. Soc. Am. J. 2006, 70, 1512–1521. [Google Scholar]
- Gallet, C.; Lebreton, P. Evolution of phenolic patterns in plants and associated litters and humus of a mountain forest ecosystem. Soil Biol. Biochem. 1995, 27, 157–165. [Google Scholar] [CrossRef]
- Hagerman, A.E. Tannin Handbook; Miami University: Oxford, OH, USA, 2002. [Google Scholar]
- Hagerman, A.E.; Butler, L.G. Condensed tannin purification and characterization of tannin-associated proteins. J. Agric. Food Chem. 1980, 28, 947–952. [Google Scholar] [CrossRef]
- Graham, H.D. Stabilization of the Prussian blue color in the determination of polyphenols. J. Agric. Food Chem. 1992, 40, 801–805. [Google Scholar] [CrossRef]
- Schofield, J.A.; Hagerman, A.E.; Harold, A. Loss of tannins and other phenolics from willow leaf litter. J. Chem. Ecol. 1998, 24, 1409–1421. [Google Scholar] [CrossRef]
- Hagerman, A.E.; Rice, M.E.; Ritchard, N.T. Mechanisms of protein precipitation for two tannins, pentagalloyl glucose and epicatechin16 (4→8) catechin (procyanidin). J. Agric. Food Chem. 1998, 46, 2590–2595. [Google Scholar] [CrossRef]
- Lu, Z.; Nie, G.; Belton, P.S.; Tang, H.; Zhao, B. Structure-activity relationship analysis of antioxidant ability and neuroprotective effect of gallic acid derivatives. Neurochem. Int. 2006, 48, 263–274. [Google Scholar] [CrossRef]
- Littell, R.C.; Milliken, G.A.; Stroup, W.W.; Wolfinger, R.D. SAS System for Mixed Models; SAS Institute Inc.: Cary, NC, USA, 1996. [Google Scholar]
- SAS OnlineDoc®, Version 8; SAS Inst., Inc.: Cary, NC, USA, 1999.
- Mueller-Harvey, I. Unravelling the conundrum of tannins in animal nutrition and health. J. Sci. Food Agric. 2006, 86, 2010–2037. [Google Scholar] [CrossRef]
- Mueller-Harvey, I.; Mlambo, V.; Sikosana, J.L.N.; Smith, T.; Owen, E.; Brown, R.H. Octanol-water partition coefficients for predicting the effects of tannins in ruminant nutrition. J. Agric. Food Chem. 2007, 55, 5436–5444. [Google Scholar]
- Jardine, P.M.; McCarthy, J.F.; Weber, N.L. Mechanisms of dissolved organic carbon adsorption on soil. Soil Sci. Soc. Am. J. 1989, 53, 1378–1385. [Google Scholar]
- Piccolo, A.; Mbagwu, J.S.C. Role of hydrophobic components of soil organic matter in soil aggregate stability. Soil Sci. Soc. Am. J. 1999, 63, 1801–1810. [Google Scholar] [CrossRef]
- Lützow, M.V.; Kögel-Knabner, I.; Ekschmitt, K.; Matzner, E.; Guggenberger, G.; Marschner, B.; Flessa, H. Stabilization of organic matter in temperate soils: Mechanisms and their relevance under different soil conditions—A review. Eur. J. Soil Sci. 2006, 57, 426–445. [Google Scholar]
- Spaccini, R.; Piccolo, A.; Conte, P.; Haberhauer, G.; Gerzabek, M.H. Increased soil organic carbon sequestration through hydrophobic protection by humic substances. Soil Biol. Biochem. 2002, 34, 1839–1851. [Google Scholar] [CrossRef]
- Kaiser, K.; Zech, W. Competitive sorption of dissolved organic matter fractions to soils and related mineral phases. Soil Sci. Soc. Am. J. 1997, 61, 64–69. [Google Scholar] [CrossRef]
- Maryganova, V.; Szajdak, L.W.; Tychinskaya, L. Hydrophobic and hydrophilic properties of humic acids from soils under shelterbelts of different ages. Chem. Ecol. 2010, 26, 25–33. [Google Scholar] [CrossRef]
- Hernández-Apaolaza, L.; Gascó, J.M.; Guerrero, F. Initial organic matter transformation of soil amended with composted sewage sludge. Biol. Fertil. Soils 2000, 32, 421–426. [Google Scholar] [CrossRef]
- Sánchez–Monedero, M.A.; Roig, A.; Cegarra, J.; Bernal, M.P. Relationships between water-soluble carbohydrate and phenol fractions and the humification indices of different organic wastes during composting. Bioresour. Technol. 1999, 70, 193–201. [Google Scholar] [CrossRef]
- Jindo, K.; Hernández, T.; García, C.; Sánchez-Monedero, M.A. Influence of stability and origin of organic amendments on humification in semiarid soils. Soil Sci. Soc. Am. J. 2011, 75, 2178–2187. [Google Scholar] [CrossRef]
- Stubbs, T.L.; Kennedy, A.C.; Reisenauer, P.E.; Burns, J.W. Chemical composition of residue from cereal crops and cultivars in dryland ecosystems. Agron. J. 2009, 101, 538–545. [Google Scholar] [CrossRef]
- Meinert, M.C.; Delmer, D.P. Changes in biochemical composition of the cell wall of the cotton fiber during development. Plant Phys. 1977, 59, 1088–1097. [Google Scholar] [CrossRef]
- Tang, H.R.; Covington, A.D.; Hancock, R.A. Structure–activity relationships in the hydrophobic interactions of polyphenols with cellulose and collagen. Biopolymers 2003, 70, 403–413. [Google Scholar] [CrossRef]
- Yu, X.; Chu, S.; Hagerman, A.E.; Lorigan, G.A. Probing the interaction of polyphenols with lipid bilayers by solid-state NMR spectroscopy. J. Agric. Food Chem. 2011, 59, 6783–6789. [Google Scholar]
- Šimon, T.; Javůrek, M.; Mikanová, O.; Vach, M. The influence of tillage systems on soil organic matter and soil hydrophobicity. Soil Tillage Res. 2009, 105, 44–48. [Google Scholar] [CrossRef]
- Monreal, C.M.; Schnitzer, M.; Schulten, H.R.; Campbell, C.A.; Anderson, D.W. Soil organic structures in macro and microaggregates of a cultivated brown chernozem. Soil Biol. Biochem. 1995, 27, 845–853. [Google Scholar] [CrossRef]
- Milanovsky, E.Yu.; Shein, E.V.; Tuygai, Z.N.; Vasil’eva, N.A. Distribution of hydrophobic and hydrophilic components of soil organic matter over granulometric fractions of chernozem. Geophys. Res. Abstr. 2005, 7, 01184:1–01184:2. [Google Scholar]
- Tschapek, M. Criteria for determining the hydrophilicity-hydrophobicity of soils. Z. Pflanzenernähr. Bodenkd. 1984, 147, 137–149. [Google Scholar]
- Leinweber, P.; Schulten, H.R.; Körschens, M. Hot water extracted organic matter: Chemical composition and temporal variations in a long-term field experiment. Biol. Fertil. Soils 1995, 20, 17–23. [Google Scholar] [CrossRef]
- Majcher, E.H.; Chorover, J.; Bollag, J.-M.; Huang, P.M. Evolution of CO2 during birnessite-induced oxidation of 14C-labeled catechol. Soil Sci. Soc. Am. J. 2000, 64, 157–163. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Halvorson, J.J.; Gollany, H.T.; Kennedy, A.C.; Hagerman, A.E.; Gonzalez, J.M.; Wuest, S.B. Sorption of Tannin and Related Phenolic Compounds and Effects on Extraction of Soluble-N in Soil Amended with Several Carbon Sources. Agriculture 2012, 2, 52-72. https://doi.org/10.3390/agriculture2010052
Halvorson JJ, Gollany HT, Kennedy AC, Hagerman AE, Gonzalez JM, Wuest SB. Sorption of Tannin and Related Phenolic Compounds and Effects on Extraction of Soluble-N in Soil Amended with Several Carbon Sources. Agriculture. 2012; 2(1):52-72. https://doi.org/10.3390/agriculture2010052
Chicago/Turabian StyleHalvorson, Jonathan J., Hero T. Gollany, Ann C. Kennedy, Ann E. Hagerman, Javier M. Gonzalez, and Stewart B. Wuest. 2012. "Sorption of Tannin and Related Phenolic Compounds and Effects on Extraction of Soluble-N in Soil Amended with Several Carbon Sources" Agriculture 2, no. 1: 52-72. https://doi.org/10.3390/agriculture2010052
APA StyleHalvorson, J. J., Gollany, H. T., Kennedy, A. C., Hagerman, A. E., Gonzalez, J. M., & Wuest, S. B. (2012). Sorption of Tannin and Related Phenolic Compounds and Effects on Extraction of Soluble-N in Soil Amended with Several Carbon Sources. Agriculture, 2(1), 52-72. https://doi.org/10.3390/agriculture2010052





