Health—Promoting Properties of Highbush Blueberries Depending on Type of Fertilization
Abstract
1. Introduction
2. Materials and Methods
2.1. Location and Layout of the Study
- -
- ‘Treatment T1’—consisted of standard fertilization and foliar sprinkling without the use of bioactive components (control treatment);
- -
- ‘Treatment T2’—included foliar fertilization and typical sprinkling in addition to a solution containing phytohormone precursors and biostimulants;
- -
- ‘Treatment T3’—comprised traditional foliar fertilizing and sprinkling, with a new method of biostimulation based on extracts from various plants and sea algae, with bioactive qualities intended to improve the physiological processes in crops;
- -
- ‘Treatment T4’—biostimulant-containing formulations were used to fertilize the soil and the leaves.
2.2. Research Methods
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kruczek, A.; Krupa-Małkiewicz, M.; Lachowicz, S.; Oszmiański, J.; Ochmian, I. Health-Promoting Capacities of In Vitro and Cultivated Goji (Lycium chinense Mill.) Fruit and Leaves; Polyphenols, Antimicrobial Activity, Macro- and Microelements and Heavy Metals. Molecules 2020, 25, 5314. [Google Scholar] [CrossRef]
- Bieniasz, M.; Dziedzic, E.; Kaczmarczyk, E. The effect of storage and processing on vitamin C content in Japanese quince fruit. Folia Hortic. 2017, 29, 83–93. [Google Scholar] [CrossRef]
- Dziedzic, E.; Błaszczyk, J.; Bieniasz, M.; Dziadek, K.; Kopeć, A. Effect of modified (MAP) and controlled atmosphere (CA) storage on the quality and bioactive compounds of blue honeysuckle fruits (Lonicera caerulea L.). Sci. Hortic. 2020, 265, 109226. [Google Scholar] [CrossRef]
- Bieniek, A.; Lachowicz-Wiśniewska, S.; Bojarska, J. The Bioactive Profile, Nutritional Value, Health Benefits and Agronomic Requirements of Cherry Silverberry (Elaeagnus multiflora Thunb.): A Review. Molecules 2022, 27, 2719. [Google Scholar] [CrossRef]
- Bieniek, A.; Draganska, E.; Pranckietis, V. Assessment of climatic conditions for Actinidia argute cultivation in north-eastern Poland. Zemdirb. Agric. 2016, 103, 311–318. [Google Scholar] [CrossRef]
- Szot, I.; Łysiak, G.P. Effect of the Climatic Conditions in Central Europe on the Growth and Yield of Cornelian Cherry Cultivars. Agriculture 2022, 12, 1295. [Google Scholar] [CrossRef]
- Rashidinejad, A.; Jaiswal, A.K. (Eds.) Nutritional Composition and antioxidant Properties of Fruits and Vegetables; Academic Press: Cambridge, MA, USA, 2020; pp. 695–708. [Google Scholar] [CrossRef]
- Michalska, A.; Łysiak, G. Bioactive Compounds of Blueberries: Post-Harvest Factors Influencing the Nutritional Value of Products. Int. J. Mol. Sci. 2015, 16, 18642–18663. [Google Scholar] [CrossRef]
- Pertuzatti, P.B.; Barcia, M.T.; Gómez-Alonso, S.; Godoy, H.T.; Hermosin-Gutierrez, I. Phenolics profiling by HPLC-DAD-ESI-MSn aided by principal component analysis to classify Rabbiteye and Highbush blueberries. Food Chem. 2021, 340, 127958. [Google Scholar] [CrossRef]
- Varo, M.A.; Martin-Gomez, J.; Serratosa, M.P.; Merida, J. Effect of potassium metabisulphite and potassium bicarbonate on color, phenolic compounds, vitamin C and antioxidant activity of blueberry wine. LWT 2022, 163, 113585. [Google Scholar] [CrossRef]
- Behrends, A.; Weber, F. Influence of Different Fermentation Strategies on the Phenolic Profile of Bilberry Wine (Vaccinium myrtillus L.). J. Agric. Food Chem. 2017, 65, 7483–7490. [Google Scholar] [CrossRef]
- Mendes-Ferreira, A.; Coelho, E.; Barbosa, C.; Oliveira, J.M.; Mendes-Faia, A. Production of blueberry wine and volatile characterization of young and bottle-aging beverages. Food Sci. Nutr. 2019, 7, 617–627. [Google Scholar] [CrossRef]
- Szajdek, A.; Borowska, E.J. Bioactive Compounds and Health-Promoting Properties of Berry Fruits: A Review. Mater. Veg. 2008, 63, 147–156. [Google Scholar] [CrossRef]
- Kähkönen, M.P.; Hopia, A.I.; Vuorela, H.J.; Rauha, J.-P.; Pihlaja, K.; Kujala, T.S.; Heinonen, M. Antioxidant Activity of Plant Extracts Containing Phenolic Compounds. J. Agric. Food Chem. 1999, 47, 3954–3962. [Google Scholar] [CrossRef]
- Prior, R.L.; Cao, G.; Martin, A.; Sofic, E.; McEwen, J.; O’Brien, C.; Lischner, N.; Ehlenfeldt, M.; Kalt, W.; Krewer, A.G.; et al. Antioxidant Capacity As Influenced by Total Phenolic and Anthocyanin Content, Maturity, and Variety of Vaccinium Species. J. Agric. Food Chem. 1998, 46, 2686–2693. [Google Scholar] [CrossRef]
- Häkkinen, S.H.; Törrönen, A.R. Content of flavonols and selected phenolic acids in strawberries and Vaccinium species: Influence of cultivar, cultivation site and technique. Food Res. Int. 2000, 33, 517–524. [Google Scholar] [CrossRef]
- Rimando, A.M.; Kalt, W.; Magee, J.B.; Dewey, A.J.; Ballington, J.R. Resveratrol, Pterostilbene, and Piceatannol in Vaccinium Berries. J. Agric. Food Chem. 2004, 52, 4713–4719. [Google Scholar] [CrossRef]
- Youdim, K.A.; Shukitt-Hale, B.; Martin, A.; Wang, H.; Denisova, N.; Bickford, P.C.; Joseph, J.A. Short-Term Dietary Supplementation of Blueberry Polyphenolics: Beneficial Effects on Aging Brain Performance and Peripheral Tissue Function. Nutr. Neurosci. 2000, 3, 383–397. [Google Scholar] [CrossRef]
- DeBoer, J.D. Berries and their role in human health. In A Survey of Research into the Health Benefits of Berries; DeBoer Consulting: Victoria, BC, Canada, 2005; pp. 1–103. [Google Scholar]
- Piątkowska, E.; Kopeć, A.; Leszczyńska, T. Antocyjany—Charakterystyka, występowanie i oddziaływanie na organizm człowieka. ŻNTJ 2011, 4, 24–35. [Google Scholar]
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2017, 61, 1361779. [Google Scholar] [CrossRef]
- Gil Lee, S.; Vance, T.M.; Nam, T.-G.; Kim, D.-O.; Koo, S.I.; Chun, O.K. Evaluation of pH differential and HPLC methods expressed as cyanidin-3-glucoside equivalent for measuring the total anthocyanin contents of berries. J. Food Meas. Charact. 2016, 10, 562–568. [Google Scholar] [CrossRef]
- Tsuda, T.; Horio, F.; Uchida, K.; Aoki, H.; Osawa, T. Dietary Cyanidin 3-O-β-D-Glucoside-Rich Purple Corn Color Prevents Obesity and Ameliorates Hyperglycemia in Mice. J. Nutr. 2003, 133, 2125–2130. [Google Scholar] [CrossRef] [PubMed]
- Łysiak, G.P. Ornamental Flowers Grown in Human Surroundings as a Source of Anthocyanins with High Anti-Inflammatory Properties. Foods 2022, 11, 948. [Google Scholar] [CrossRef]
- Faria, A.; Pestana, D.; Teixeira, D.; de Freitas, V.; Mateus, N.; Calhau, C. Blueberry anthocyanins and pyruvic acid adducts: Anticancer properties in breast cancer cell lines. Phytotherapy Res. 2010, 24, 1862–1869. [Google Scholar] [CrossRef] [PubMed]
- Bunea, A.; Rugină, D.; Sconţa, Z.; Pop, R.M.; Pintea, A.; Socaciu, C.; Tăbăran, F.; Grootaert, C.; Struijs, K.; VanCamp, J. Anthocyanin determination in blueberry extracts from various cultivars and their antiproliferative and apoptotic properties in B16-F10 metastatic murine melanoma cells. Phytochemistry 2013, 95, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Miyake, S.; Takahashi, N.; Sasaki, M.; Kobayashi, S.; Tsubota, K.; Ozawa, Y. Vision preservation during retinal inflammation by anthocyanin-rich bilberry extract: Cellular and molecular mechanism. Lab. Investig. 2011, 92, 102–109. [Google Scholar] [CrossRef]
- Lenart, A.; Wrona, D.; Klimek, K.; Kapłan, M.; Krupa, T. Assessment of the impact of innovative fertilization methods compared to traditional fertilization in the cultivation of highbush blueberry. PLoS ONE 2022, 17, e0271383. [Google Scholar] [CrossRef] [PubMed]
- Latocha, P.; Krupa, T.; Wołosiak, R.; Worobiej, E.; Wilczak, J. Antioxidant activity and chemical difference in fruit of different Actinidia sp. Int. J. Food Sci. Nutr. 2010, 61, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1999; Volume 299, pp. 152–178. [Google Scholar]
- Saint-Cricq de Gaulejac, N.; Provost, C.; Vivas, N. Comparative Study of Polyphenol Scavenging Activities Assessed by Different Methods. J. Agric. Food Chem. 1999, 47, 425–431. [Google Scholar] [CrossRef]
- Szpadzik, E.; Krupa, T.; Niemiec, W.; Jadczuk-Tobjasz, E. Yielding and fruit quality of selected sweet cherry (prunus avium) cultivars in the conditions of central poland. Acta Sci. Pol. Hortorum Cultus 2019, 18, 117–126. [Google Scholar] [CrossRef]
- Lenart, A.; Wrona, D. Biostymulacja a intensyfikacja wydajności biologicznej roślin. Czynniki wpływające na plonowanie i jakość owoców roślin sadowniczych, 29. Wydawnictwo SGGW. Warszawa 2019, 29, 59–66. [Google Scholar]
- Bulgari, R.; Cocetta, G.; Trivellini, A.; Vernieri, P.; Ferrante, A. Biostimulants and crop responses: A review. Biol. Agric. Hortic. 2014, 31, 1–17. [Google Scholar] [CrossRef]
- Benhamou, N.; Rey, P. Stimulators of natural plant defenses: A new phytosanitary strategy in the context of sustainable ecoproduction: II. Interest of the SND in crop protection. Phytoprotection 2012, 92, 24–35. [Google Scholar] [CrossRef]
- Durand, N.; Briand, X.; Meyer, C. The effect of marine bioactive substances (N PRO) and exogenous cytokinins on nitrate reductase activity in Arabidopsis thaliana. Physiol. Plant. 2003, 119, 489–493. [Google Scholar] [CrossRef]
- Du Jardin, P. Plant biostimulants: Definition, concept, main categories and regulation. Sci. Hortic. 2015, 196, 3–14. [Google Scholar] [CrossRef]
- Battacharyya, D.; Babgohari, M.Z.; Rathor, P.; Prithiviraj, B. Seaweed extracts as biostimulants in horticulture. Sci. Hortic. 2015, 196, 39–48. [Google Scholar] [CrossRef]
- Norrie, J.; Keathley, J.P. Benefits of ascophyllum nodosum marine-plant extract applications to ’thompson seedless’ grape production. Acta Hortic. 2006, 727, 243–248. [Google Scholar] [CrossRef]
- Crouch, I.J.; van Staden, J. Evidence for the presence of plant growth regulators in commercial seaweed products. Plant Growth Regul. 1993, 13, 21–29. [Google Scholar] [CrossRef]
- Graziani, G.; Cirillo, A.; Giannini, P.; Conti, S.; El-Nakhel, C.; Rouphael, Y.; Ritieni, A.; Di Vaio, C. Biostimulants Improve Plant Growth and Bioactive Compounds of Young Olive Trees under Abiotic Stress Conditions. Agriculture 2022, 12, 227. [Google Scholar] [CrossRef]
- Tamás, N.P.; Ádám, C.; Anita, S. Effects of algae products on nutrient uptake and fruit quality of apple. Nat. Resour. Sustain. Dev. 2019, 9, 80–91. [Google Scholar] [CrossRef]
- Ambroszczyk, A.M.; Liwińska, E.; Bieżanowska Kopeć, R. Rola procesów technologicznych w kształtowaniu jakości żywności: Zróżnicowanie wartości odżywczej oraz prozdrowotnej owoców pomidora w zależności od zastosowanych biostymulatorów wzrostu. Rola Procesów Technol. W Kształtowaniu Jakości Żywności 2016, 173, 173–182. [Google Scholar] [CrossRef]
- Mikos-Bielak, M. Bioregulacja plonowania i chemicznej jakości plonu malin jako efekt zastosowania Asahi. Ann. Univ. Mariae Curie-Skłodowska. Sect. E. Agric. 2004, 59, 1471–1479. [Google Scholar]
- Kapłan, M.; Lenart, A.; Klimek, K.; Borowy, A.; Wrona, D.; Lipa, T. Assessment of the Possibilities of Using Cross-Linked Polyacrylamide (Agro Hydrogel) and Preparations with Biostimulation in Building the Quality Potential of Newly Planted Apple Trees. Agronomy 2021, 11, 125. [Google Scholar] [CrossRef]
- Campobenedetto, C.; Mannino, G.; Agliassa, C.; Acquadro, A.; Contartese, V.; Garabello, C.; Bertea, C.M. Transcriptome Analyses and Antioxidant Activity Profiling Reveal the Role of a Lignin-Derived Biostimulant Seed Treatment in Enhancing Heat Stress Tolerance in Soybean. Plants 2020, 9, 1308. [Google Scholar] [CrossRef] [PubMed]
- Parađiković, N.; Vinković, T.; Vinković Vrček, I.; Žuntar, I.; Bojić, M.; Medić-Šarić, M. Effect of natural biostimulants on yield and nutritional quality: An example of sweet yellow pepper (Capsicum annuum L.) plants. J. Sci. Food Agric. 2011, 91, 2146–2152. [Google Scholar] [CrossRef]
- Mukherjee, A.; Patel, J.S. Seaweed extract: Biostimulator of plant defense and plant productivity. Int. J. Environ. Sci. Technol. 2019, 17, 553–558. [Google Scholar] [CrossRef]
- Jeannin, I.; Lescure, J.-C.; Morot-Gaudry, J.-F. The Effects of Aqueous Seaweed Sprays on the Growth of Maize. Bot. Mar. 1991, 34, 469–474. [Google Scholar] [CrossRef]
- Mattner, S.W.; Wite, D.; Riches, D.A.; Porter, I.J.; Arioli, T. The effect of kelp extract on seedling establishment of broccoli on contrasting soil types in southern Victoria, Australia. Biol. Agric. Hortic. 2013, 29, 258–270. [Google Scholar] [CrossRef]
- Rayirath, P.; Benkel, B.; Hodges, D.M.; Allan-Wojtas, P.; MacKinnon, S.; Critchley, A.T.; Prithiviraj, B. Lipophilic components of the brown seaweed, Ascophyllum nodosum, enhance freezing tolerance in Arabidopsis thaliana. Planta 2009, 230, 135–147. [Google Scholar] [CrossRef]
- Alam, M.Z.; Braun, G.; Norrie, J.; Hodges, D.M. Effect of Ascophyllum extract application on plant growth, fruit yield and soil microbial communities of strawberry. Can. J. Plant Sci. 2013, 93, 23–36. [Google Scholar] [CrossRef]
- Mugnai, S.; Azzarello, E.; Pandolfi, C.; Salamagne, S.; Briand, X.; Mancuso, S. Enhancement of ammonium and potassium root influxes by the application of marine bioactive substances positively affects Vitis vinifera plant growth. J. Appl. Phycol. 2007, 20, 177–182. [Google Scholar] [CrossRef]
- Fan, D.; Hodges, D.M.; Critchley, A.T.; Prithiviraj, B. A Commercial Extract of Brown Macroalga (Ascophyllum nodosum) Affects Yield and the Nutritional Quality of Spinach In Vitro. Commun. Soil Sci. Plant Anal. 2013, 44, 1873–1884. [Google Scholar] [CrossRef]
- Bi, F.; Iqbal, S.; Arman, M.; Ali, A.; Hassan, M.-U. Carrageenan as an elicitor of induced secondary metabolites and its effects on various growth characters of chickpea and maize plants. J. Saudi Chem. Soc. 2011, 15, 269–273. [Google Scholar] [CrossRef]
- Vera, J.; Castro, J.; Contreras, R.A.; González, A.; Moenne, A. Oligo-carrageenans induce a long-term and broad-range protection against pathogens in tobacco plants (var. Xanthi). Physiol. Mol. Plant Pathol. 2012, 79, 31–39. [Google Scholar] [CrossRef]
| 2019 | 2020 | 2021 | ||||
|---|---|---|---|---|---|---|
| Month | Total Rainfall (mm·m−2) | Average Temp. (°C) | Total Rainfall (mm·m−2) | Average Temp. (°C) | Total Rainfall (mm·m−2) | Average Temp. (°C) |
| March | 25.1 | 6.8 | 13.2 | 5 | 18.3 | 4 |
| April | 2.2 | 10.7 | 7.4 | 9 | 55.3 | 6.5 |
| May | 77.8 | 14.1 | 65.2 | 11.9 | 62.3 | 12.4 |
| June | 16 | 22.7 | 140.9 | 19.6 | 69.2 | 19.7 |
| July | 34.8 | 19.5 | 45.9 | 19.1 | 118.8 | 21.7 |
| August | 34.4 | 21.1 | 83.1 | 20.7 | 140.1 | 17.2 |
| Salinity | pH | Content in mg/L | ||||||
|---|---|---|---|---|---|---|---|---|
| g NaCl/L | in H2O | N-NO3 | N-NH4 | P | K | Ca | Mg | Cl |
| 0.08 | 4.9 | <10.0 * | <10.0 * | <20.0 * | <20.0 * | 245 | 20 | <10.0 * |
| PB 02 ed.3 from 1 March 2018 ** | PB 01 ed.2 from 1 March 2018 ** | PB 06 ed.1 from 28 May 2004 ** | PB 69 ed.1 from 3 April 2017 ** | PB 03 ed.2 from 19 March 2007 ** | PB 04 ed.1 from 21 May 2004 ** | PB 04 ed.1 from 21 May 2004 ** | PB 05 ed.1 from 28 May 2004 ** | PB 07 ed.1 from 28 May 2004 ** |
| N (kg/ha−1) | P2O5 (kg/ha−1) | K2O (kg/ha−1) | SO3 (kg/ha−1) | CaCO3 (kg/ha−1) |
|---|---|---|---|---|
| 100 | 30 | 92.5 | 142 | 64 |
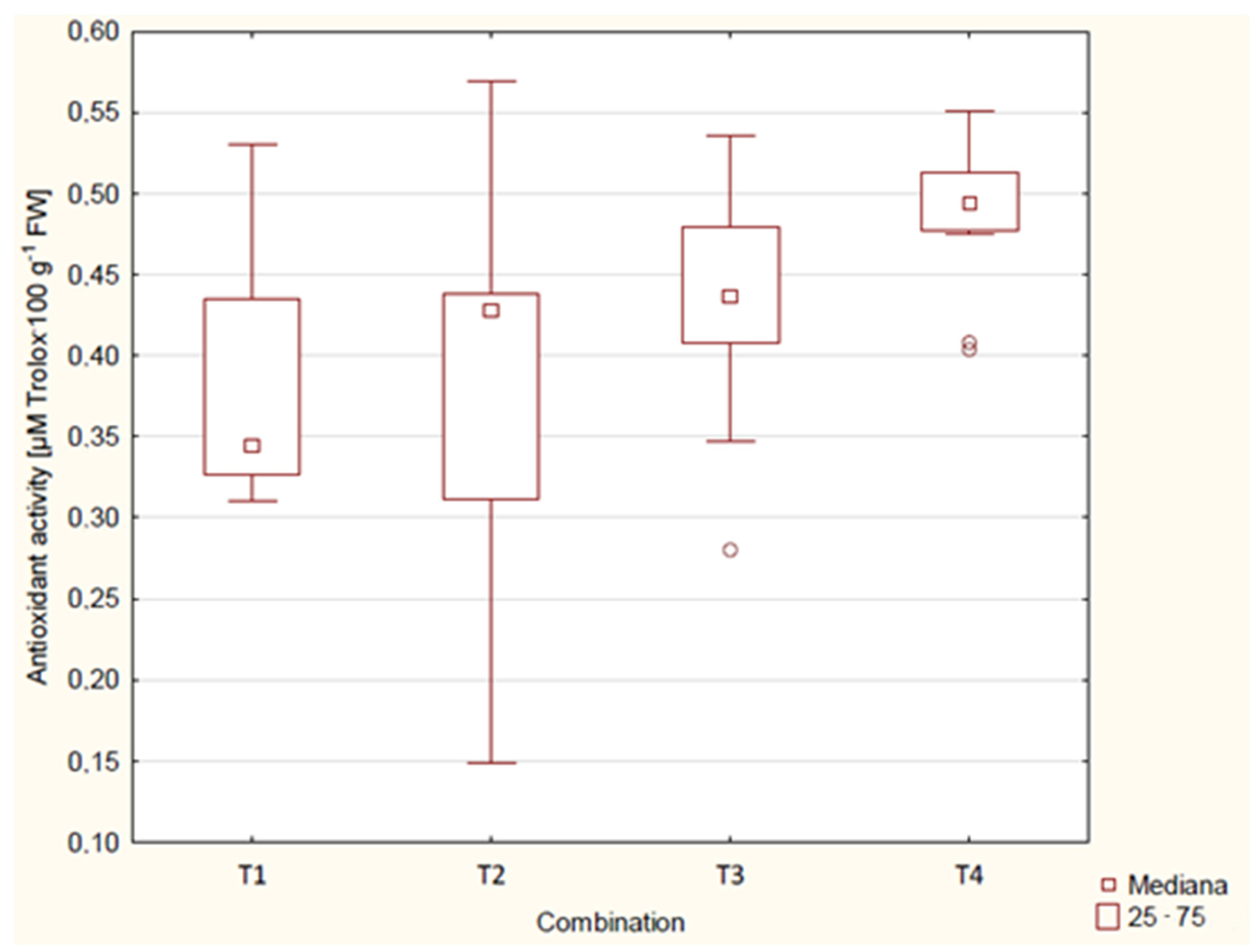
| Combination | Year | Antioxidant Activity [µM Trolox·100 g−1] | 3 Years Average Antioxidant Activity [µM Trolox·100 g−1] |
|---|---|---|---|
| T1 | 2019 | 0.356 ab * | 0.375 a |
| 2020 | 0.356 ab | ||
| 2021 | 0.414 bc | ||
| T2 | 2019 | 0.241 a | 0.381 a |
| 2020 | 0.430 bc | ||
| 2021 | 0.472 c | ||
| T3 | 2019 | 0.481 c | 0.437 ab |
| 2020 | 0.413 bc | ||
| 2021 | 0.416 bc | ||
| T4 | 2019 | 0.517 c | 0.489 b |
| 2020 | 0.487 c | ||
| 2021 | 0.463 bc | ||
| p-value | <0.01 | <0.01 |
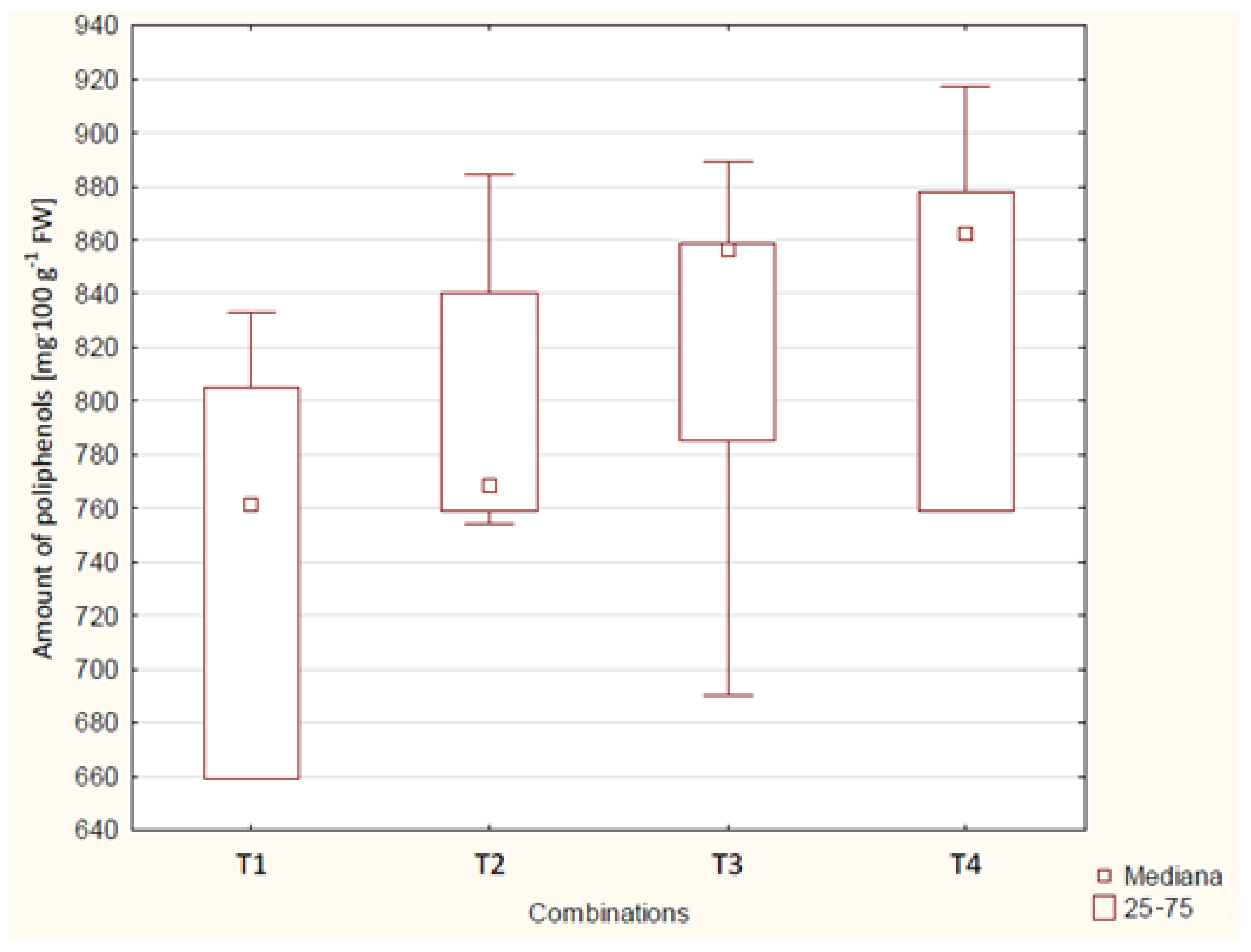
| Combination | Year | Amount of Polyphenols [mg 100 g−1 FW] | 3-Year Average [mg 100 g−1 FW] |
|---|---|---|---|
| T1 | 2019 | 659 a * | 742 a |
| 2020 | 766 ab | ||
| 2021 | 801 bc | ||
| T2 | 2019 | 759 ab | 802 ab |
| 2020 | 845 bc | ||
| 2021 | 802 bc | ||
| T3 | 2019 | 859 bc | 820 ab |
| 2020 | 752 ab | ||
| 2021 | 848 bc | ||
| T4 | 2019 | 759 ab | 837 b |
| 2020 | 878 c | ||
| 2021 | 875 c | ||
| p-value | <0.01 | <0.01 |
| Dp-3-glu | Dp-3-gal | Dp-3-ara | Pt-3-glu | Pt-3-gal | Pt-3-ara | Pn-3-glu | Mv-3-glu | Mv-3-gal | Mv-3-ara | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Combinations | T1 | 19.83 ab * | 10.58 a | 18.53 a | 10.76 a | 7.36 a | 6.42 a | 1.88 b | 26.5 a | 26.08 a | 19.00 a |
| T2 | 18.88 ab | 11.18 a | 20.22 a | 10.98 a | 7.56 a | 6.50 a | 2.0 ab | 36.73 b | 26.40 a | 19.09 a | |
| T3 | 17.19 a | 10.38 a | 21.10 a | 11.36 a | 7.90 a | 7.51 a | 2.36 b | 28.62 a | 27.52 a | 22.79 a | |
| T4 | 21.78 b | 12.96 b | 22.88 a | 12.74 a | 10.31 a | 8.04 a | 1.38 a | 26.16 a | 28.84 a | 23.43 a | |
| p-value | <0.0001 | <0.0001 | 0.0816 | 0.0816 | 0.0816 | 0.0816 | <0.0001 | <0.0001 | 0.0816 | 0.0816 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lenart, A.; Wrona, D.; Krupa, T. Health—Promoting Properties of Highbush Blueberries Depending on Type of Fertilization. Agriculture 2022, 12, 1741. https://doi.org/10.3390/agriculture12101741
Lenart A, Wrona D, Krupa T. Health—Promoting Properties of Highbush Blueberries Depending on Type of Fertilization. Agriculture. 2022; 12(10):1741. https://doi.org/10.3390/agriculture12101741
Chicago/Turabian StyleLenart, Agnieszka, Dariusz Wrona, and Tomasz Krupa. 2022. "Health—Promoting Properties of Highbush Blueberries Depending on Type of Fertilization" Agriculture 12, no. 10: 1741. https://doi.org/10.3390/agriculture12101741
APA StyleLenart, A., Wrona, D., & Krupa, T. (2022). Health—Promoting Properties of Highbush Blueberries Depending on Type of Fertilization. Agriculture, 12(10), 1741. https://doi.org/10.3390/agriculture12101741






