1. Introduction
The pressure exerted on the agricultural production chains due to the continuous increase in world population will require increasing crop production by 70% by the year 2050 [
1]. Agriculture faces the challenge of producing more food in an available arable land area that is progressively decreasing and, at the same time, dealing with adverse conditions such as those generated by climate change [
2].
Salinized soils represent a problem with a significant impact on food security. Soil salinity reduces agricultural yields by causing metabolic changes that affect plant growth and development. It is estimated that more than 800 million hectares of land are currently affected by salinity globally, and it is expected that this figure will increase in the coming years [
3]. The use of different crop species or varieties of a given crop that perform better on land affected by salinity would be an economically viable option for increasing agricultural yields and food production [
4]. Eggplant (
Solanum melongena) is the fifth most economically important crop within the Solanaceae family and is considered by the Food and Agriculture Organization of the United Nations (FAO) as one of the 35 foods with the most considerable relevance for world food security [
5]. Eggplant is a species sensitive to salinity and, therefore, is not well adapted to saline soils [
6]. However, this trait could be enhanced by genetic improvement and also by the use as rootstocks of some related wild species that have more growth capacity in saline soils [
7,
8].
Solanum torvum has been used extensively as rootstock for eggplant and tomato [
8,
9,
10,
11] for its good agronomic performance and tolerance to some bacterial and fungal diseases. As
S. torvum and eggplant are genetically related, it is likely that their physiological and biochemical responses to salt stress are qualitatively similar, even though they may differ quantitatively. Correlation of the relative degree of salt tolerance of the two species with the stress-induced changes in the levels of different stress biomarkers associated to specific responses to salinity, may provide relevant information on the mechanisms of salt tolerance in these and related taxa.
The most general and rapid effect of salt stress (and other stress conditions) on plants, is the reduction of their growth, as they redirect metabolic and energetic resources from biomass accumulation to the activation of defense mechanisms [
12,
13]. Therefore, salt-induced inhibition of growth represents an appropriate criterion to compare the relative tolerance of different plant species. Salinity is often associated with the degradation of photosynthetic pigments. Under the same stress conditions, the decrease in pigments concentrations is generally more pronounced in less tolerant species; consequently, the concentration of chlorophylls and carotenoids can be used as biomarkers of stress [
14]. All plants share the same basic responses against salinity, but their behavior as more tolerant or susceptible relies mainly on the efficiency of these response mechanisms [
15]. Accumulation of toxic ions such as Na
+ and Cl
− and their sequestration in the vacuoles is a typical strategy of salt tolerant plants, which also insures an energetically “cheap” osmotic adjustment [
16]. To maintain the osmotic balance under stress, plants accumulate different types of organic osmolytes in their cytoplasm; these compatible solutes include, for example, proline, glycine betaine, polyalcohols, and soluble sugars [
16,
17]. These compounds play additional roles in the responses to stress, acting as low-molecular-weight chaperons in the direct protection of macromolecular structures, such as cell membranes and proteins, as scavengers of reactive oxygen species (ROS) or as signaling molecules [
12,
18].
Salinity is generally associated with the generation of oxidative stress as a secondary effect, by an excess production of ROS, which appear by the transfer to O
2 of one to three electrons, to form superoxide (O
2−), hydrogen peroxide (H
2O
2), or the hydroxyl radical (HO
−) [
19]. ROS are by-products of normal cell metabolism, generated during photorespiration, respiration and photosynthesis, and at low concentrations play a necessary physiological role as signaling molecules that regulate plant growth and responses to stress [
20]. When in excess, as it occurs in different stressful conditions, ROS alter the intracellular ionic homeostasis by reducing cytosolic K
+ levels, and subsequently activating proteases and endonucleases. Furthermore, ROS oxidize proteins, DNA, and unsaturated fatty acids in membrane lipids, which can lead to cell death [
21,
22]. Excessive ROS accumulation is prevented or counteracted by the activation of enzymatic and non-enzymatic antioxidant systems. Different types of chemical compounds are included in the latter category—for example, phenolic compounds, especially flavonoids, ascorbic acid, glutathione, or β-carotenes, among others [
19]. In addition, salinity stress causes changes in the major photosynthetic pigments accumulation. Superoxide dismutase (SOD), catalase (CAT), ascorbate peroxidase (APX) (and other peroxidases), or redox regulatory enzymes such as glutathione reductase (GR) are some of the commonest antioxidant plant enzymes [
23].
This study aimed at identifying the most relevant mechanisms of salt tolerance in cultivated eggplant by comparing the responses to controlled salt stress treatments of S. melongena and its wild relative S. torvum. To address this objective, the relative salt tolerance of the two species, established by their degree of salt-induced growth inhibition, was correlated with the stress-induced changes in the contents of putative biomarkers associated to specific stress response mechanisms.
4. Discussion
The salt treatments applied to
S. melongena and
S. torvum plants in the present study were sufficient to cause significant inhibition of growth and changes in several biochemical markers, at 200 mM and higher NaCl concentrations. Glycophytes, including eggplant and all major crops, are sensitive to relatively low salt concentrations. They show optimal development when growing in soils with a salinity level below a particular limit, specific for each species (and for specific genotypes within a species); once this threshold is exceeded, growth is significantly reduced as plants invest their resources to activate metabolic pathways involved in the mechanisms of defense against salt stress [
12,
41]. Eggplant is a species sensitive to moderate salinity levels, and several studies have shown a reduction in biomass production and other growth parameters caused by the increase in salinity [
6,
42,
43]. The effects of salt stress on plant development and the general responses of plants to high salinity are well established [
12,
13], although the specific mechanisms of stress tolerance may differ widely in different species. The present work aimed to compare the responses to salt stress of the cultivated eggplant and its wild relative
S. torvum. Correlation of the relative tolerance of the two species with salt-induced changes in physiological and biochemical parameters may provide information on the mechanisms of salinity tolerance in these species, by telling apart those responses that are relevant for tolerance from those that are not. On the other hand, from a practical point of view, a higher salt tolerance of
S. torvum will support the use of this species as rootstock for eggplant in salinized farmland.
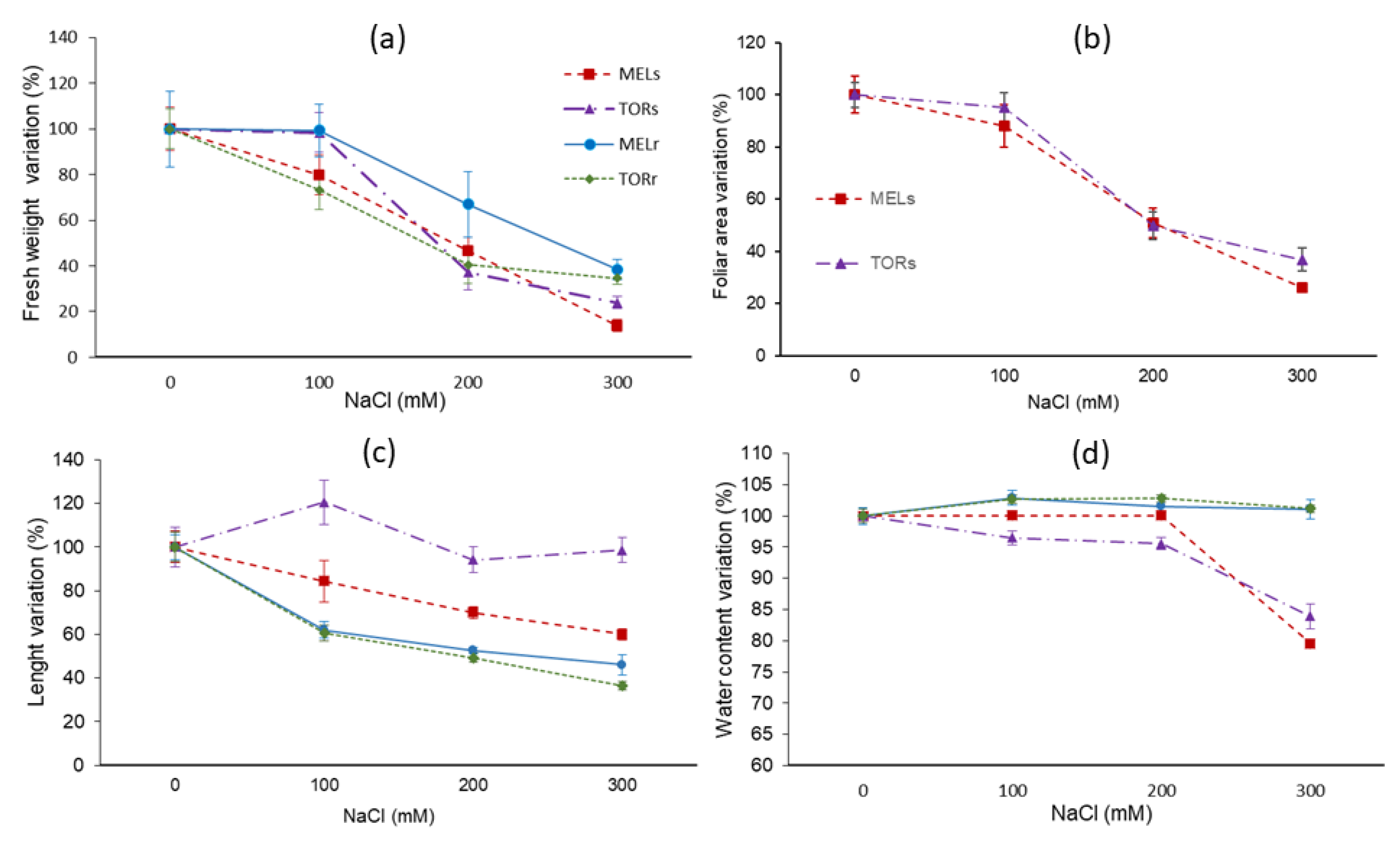
An analysis of salt effects on growth parameters can be useful for the establishment of a salt stress tolerance scale when comparing related species or different cultivars of a given species [
44]. One of the criteria frequently used to assess the degree of tolerance to salinity (or other stresses) is the percentage of survival; in this case, however, all plants survived the stress period under the specific conditions used in the experiments. Therefore, the relative degree of salt-induced growth inhibition, most accurately indicated by the percentage of biomass reduction in relation to control plants, was used instead for ranking the selected genotypes. According to this criterion,
Solanum torvum was shown to be more tolerant to salinity than
S. melongena. Higher stress tolerance of eggplant wild relatives, compared to the cultivated species, is to be expected since they are often found in nature in arid or semiarid regions and in saline environments [
45,
46]. Nevertheless, it should be noted that the differences between the two species were relatively small and observed only at high external salinities (e.g., 300 mM NaCl) because the specific eggplant cultivar used in this work, MEL 1, seems to be also quite resistant to low and moderate salt concentrations [
25], as well as to water stress [
47].
Solanum melongena includes many varieties and commercial cultivars that differ considerably in their degree of salt tolerance [
43].
One of the multiple effects of salt stress on plant metabolism is the generation of reactive oxygen species (ROS), highly reactive oxygen-containing free radicals, and other molecules, which can damage many cellular macromolecular structures, such as DNA, proteins, and membranes, but also the photosynthetic machinery, leading to the degradation of photosynthetic pigments [
48,
49]. Indeed, a reduction in the levels of chlorophyll a, chlorophyll b and carotenoids, in response to salt, has been reported in many plant species, including eggplant [
50]. However, there are also many reports indicating that salt-tolerant plants can withstand relatively strong stress conditions without showing any degradation of chlorophylls or carotenoids [
51,
52]. In the present experiments, no significant decrease in pigment contents in response to salt was observed in
S. torvum or the eggplant cultivar MEL 1, supporting the relative salt resistance of both genotypes.
As the evolution of glycophytes occurred in non-saline environments, their ion capture and storage mechanisms are not adapted to face salinity, and their response mechanisms are mostly based on the restriction of intake of toxic ions by the roots and the limitation of their transport to the aboveground organs of the plants. In contrast, dicotyledonous halophytes possess active transport systems to accumulate the ions in their aerial parts, where they are mostly stored in the leaf vacuoles, to avoid their toxic effects in the cytoplasm [
12].
The cell has various ion channels and transporters, distributed throughout its cytoplasmic membrane and the tonoplast, which are responsible for controlling the passage of different ions, both inwardly and outwardly. High salt concentrations in the soil cause the water that enters the plant to carry an excess of ions, which have short and long-term deleterious effects for the plants. Na
+ competes with K
+ for protein binding since both elements have a similar atomic structure and can use K
+ transport systems of low selectivity [
53]. The increase in the intracellular concentration of Na
+ and Cl
− alone generates toxicity in different tissues since glycophytes have a limited capacity of compartmentalization in vacuoles, and also affects the absorption of mineral nutrients, thus contributing to growth inhibition [
54,
55]. Besides, the Na
+ ion, when in excess, can inhibit specific enzymatic activities and cellular processes by actively competing for the binding sites of enzymes that use K
+ or Mg
2+ as cofactors [
56,
57].
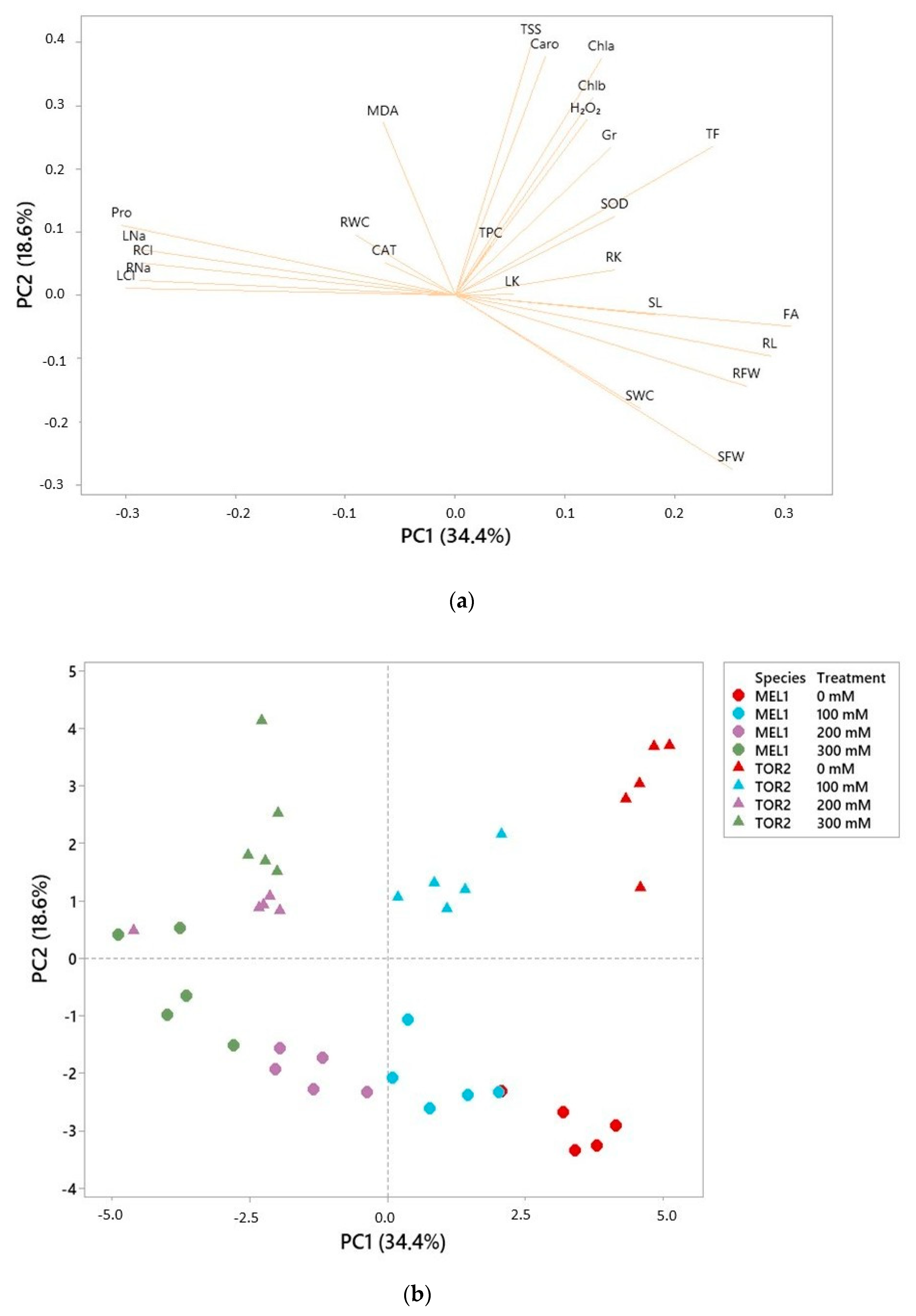
As expected, the levels of Na
+ and Cl
− increased in parallel to the increase of NaCl concentrations in the pots, in roots and leaves of
S. melongena and
S. torvum plants. This finding coincides with the variation in the levels of ions reported in other cultivated eggplant genotypes [
6,
58] and other solanaceous crops [
25,
59]. However, the patterns of accumulation of the ions in belowground and aboveground organs differed in the two species, providing some hints on the mechanisms of tolerance.
Solanum melongena behaves as a typical glycophyte, limiting ion transport to the leaves at high external salinities so that ion concentrations remain more elevated in the roots. In
S. torvum, on the contrary, under the same conditions transport of ions to the aerial part of the plant is activated, leading to higher concentrations in the leaves than in the roots, which is rather characteristic of dicotyledonous halophytes. Since higher Na
+ and Cl
− contents in leaves do not correlate with a stronger growth inhibition in
S. torvum, just the opposite, it should be assumed that the wild species possesses a better capacity than cultivated eggplant for the transport and storage of toxic ions in the vacuoles.
As mentioned above, Na
+ competes with K
+ for the same transport systems and, therefore, an increase in intracellular Na
+ concentration is generally accompanied by a decrease in K
+ contents [
56]. However, the concentration of K
+ was maintained at steady levels in roots and leaves of
S. melongena plants treated with increasing NaCl concentrations. This has been previously reported for the same eggplant cultivar, MEL 1 [
25], and probably contributes to the relative salt tolerance of this specific genotype. In
S. torvum, K
+ concentrations are maintained in the roots, in the control and at all tested salinities, whereas they decrease in leaves in the presence of 100 and 200 mM NaCl, thus following the general pattern mentioned above. Interestingly, however, at the highest salt concentration tested (300 mM NaCl), K
+ increases significantly again, suggesting the activation of uptake and transport mechanisms of this ″physiological cation″ to partly counteract the harmful effects of high Na
+ concentrations. Further studies will be required to identify and characterize the ion transporters of
S. torvum responsible for the efficient active transport of Na
+ and K
+ to the leaves at high external salinities, which appear to be relevant for the higher tolerance of the wild species. Transfer of the corresponding genes to
S. melongena, through classical breeding or by genetic transformation, could be a suitable strategy for the genetic improvement of salt tolerance of the cultivated eggplant.
The synthesis of compatible solutes, or organic osmolytes, is also a general response of plants to any stress condition including an osmotic component, such as drought, high salinity, cold or high temperatures, as they limit cellular dehydration. In the specific case of salt stress, accumulation of osmolytes in the cytoplasm contributes to cellular osmotic adjustment, compensating the accumulation of toxic ions in the vacuole [
12,
16]. Proline and glycine betaine are two of the main osmolytes synthesized by plants [
17,
60]. Accumulation of Pro to very high levels in response to the salt treatments has been observed in the two analyzed species, reaching higher values (more than 40-fold over those of the control plants) in
S. torvum, the most tolerant species. Therefore, there is a clear positive correlation of Pro accumulation with higher salt tolerance, as previously reported from other comparative studies with different eggplant cultivars [
43] or eggplant and related wild species [
25]. These data strongly support the direct participation of Pro in the mechanisms of salt tolerance in eggplant and related species. Although Pro accumulation in response to salt stress has been observed in many plants, its direct involvement in salt tolerance is not a general feature of Pro-accumulating species since other reports have shown higher Pro contents in the most susceptible genotypes of related species or cultivars of a given species [
61]. The accumulation to high concentrations of this osmolyte in wild species has been related to a greater expression or better functioning under stress conditions of the enzyme pyrroline-5-carboxylate synthetase, which has a fundamental role in the biosynthesis pathway of proline [
62].
Different soluble carbohydrates, sugars and polyalcohols, are often involved as osmolytes in the responses of plants to abiotic stresses [
63]. In eggplant, however, the measured levels of total soluble sugars are too low to have any relevant osmotic effect, and they increased only slightly with increasing salinity. In
S. torvum, although TSS contents in the controls were much higher than in
S. melongena, they actually decreased in response to the stress treatment. Soluble carbohydrates have multiple functions, as direct products of photosynthesis, energy source, metabolic precursors or signaling molecules, and it is not always simple to assess their specific roles in osmotic adjustment and osmoprotection [
16,
63]. Nevertheless, it does not seem that these compounds play any important role in the mechanisms of salt tolerance in eggplant or related species.
Salt stress usually generates oxidative stress on plants as a secondary effect, by increasing the level of ROS, and plants respond activating enzymatic and non-enzymatic antioxidant systems, [
19,
20,
64]. MDA, a product of membrane lipid peroxidation, is a reliable marker of oxidative stress [
65]. In the present study, however, MDA contents did not increase at all in response to the salt treatments in
S. torvum, and only in the presence of 300 mM NaCl in
S. melongena. An increase in ROS levels will also indicate the generation of oxidative stress, but, here again, leaf H
2O
2 contents showed no marked variation in response to the salt treatment.
These data indicate that, under the specific conditions used in the experiments, no oxidative stress was generated in salt-treated plants of either species. This is probably due to the relatively high salt tolerance of the selected genotypes. Response mechanisms based on the control of ion transport and the accumulation of high concentrations of Pro, with its ″ROS-scavenging″ activity, may be efficient enough to avoid a significant increase in ROS contents. Consequently, we did not observe the activation of the synthesis of antioxidant compounds in response to salt stress: TPC and TF contents did not increase, or even decreased, with increasing salinity. Similarly, we did not detect in the salt-treated plants any significant increase in the specific activity of some of the enzymes commonly involved in antioxidant defense mechanisms, namely SOD, CAT and GR. There are many reports on salt tolerant plants that do not activate antioxidant responses under high salinity conditions, as they possess efficient mechanisms to avoid oxidative stress [
36,
66].
Interestingly, mean values of MDA and H2O2 contents, the concentration of total flavonoids, and SOD, CAT and GR specific activities were generally higher in S. torvum than in S. melongena plants, in the controls and all applied treatments, although the differences between species were statistically significant only in some cases. It seems, therefore, that the basal redox equilibrium in S. torvum is based on higher levels of oxidant and antioxidant compounds than in S. melongena. This difference between the two species does not affect the conclusions of the present work, as it is independent of the salt treatments; as mentioned above, none of the determined biochemical parameters varied significantly with external salinity.