Phosphorus in Spring Barley and Italian Rye-Grass Biomass as an Effect of Inter-Species Interactions under Water Deficit
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
- water supply of the plants: optimal (OW), and reduced by 50% in relation to the optimal one (LW),
- sowing type: barley grown as a single species (BP), rye-grass grown as a single species (RP), barley in a mixture with rye-grass (BM), and rye-grass in a mixture with barley (RM).
2.2. Plant Sampling and Analysis
2.3. Calculations
2.4. Statistical Analysis
3. Results
3.1. Phosphorus Content and Accumulation
3.1.1. Spring Barley
3.1.2. Italian Rye-Grass
3.2. Competition for Phosphorus
4. Discussion
4.1. Phosphorus Content and Accumulation in Barley
4.2. Phosphorus Content and Accumulation in Rye-Grass
4.3. Relative Yields
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Suzuki, N.; Rivero, R.M.; Shulaev, V.; Blumwald, E.; Mittler, R. Abiotic and biotic stress combinations. New Phytol. 2014, 203, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Valliyodan, B.; Nguyen, H.T. Understanding regulatory networks and engineering for enhanced drought tolerance in plants. Curr. Opin. Plant Biol. 2006, 9, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Michalak, D. Adapting to climate change and effective water management in Polish agriculture—At the level of government institutions and farms. Ecohydrol. Hydrobiol. 2020, 20, 134–141. [Google Scholar] [CrossRef]
- Hänsel, S.; Ustrnul, Z.; Łupikasza, E.; Skalak, P. Assessing seasonal drought variations and trends over Central Europe. Adv. Water Resour. 2019, 127, 53–75. [Google Scholar] [CrossRef]
- Szwed, M. Variability of precipitation in Poland under climate change. Theor. Appl. Climatol. 2019, 135, 1003–1015. [Google Scholar] [CrossRef]
- da Silva, E.C.; Nogueira, R.; da Silva, M.A.; de Albuquerque, M.B. Drought stress and plant nutrition. Plant Stress 2011, 5, 32–41. [Google Scholar]
- Crush, J.R.; Easton, H.S.; Waller, J.E.; Hume, D.E.; Faville, M.J. Genotypic variation in patterns of root distribution, nitrate interception and response to moisture stress of a perennial ryegrass (Lolium perenne L.) mapping population. Grass Forage Sci. 2007, 62, 265–273. [Google Scholar] [CrossRef]
- Samarah, N.H.; Alqudah, A.M.; Amayreh, J.A.; McAndrews, G.M. The effect of late-terminal drought stress on yield components of four barley cultivars. J. Agron. Crop Sci. 2009, 195, 427–441. [Google Scholar] [CrossRef]
- Pecio, A.; Wach, D. Grain yield and yield components of spring barley genotypes as the indicators of their tolerance to temporal drought stress. Pol. J. Agron. 2015, 21, 19–27. [Google Scholar]
- Cyriac, D.; Hofmann, R.W.; Stewart, A.; Sathish, P.; Winefield, C.S.; Moot, D.J. Intraspecific differences in long-term drought tolerance in perennial ryegrass. PLoS ONE 2018, 13, e194977. [Google Scholar] [CrossRef]
- Aronsson, H.; Hansen, E.M.; Thomsen, I.K.; Liu, J.; Øgaard, A.F.; Känkänen, H.; Ulén, B. The ability of cover crops to reduce nitrogen and phosphorus losses from arable land in southern Scandinavia and Finland. J. Soil Water Conserv. 2016, 71, 41–55. [Google Scholar] [CrossRef]
- Żuk-Gołaszewska, K.; Wanic, M.; Orzech, K. The role of catch crops in field plant production—A review. J. Elem. 2019, 24, 575–587. [Google Scholar] [CrossRef]
- Wanic, M.; Zuk-Golaszewska, K.; Orzech, K. Catch crops and the soil environment—A review of the literature. J. Elem. 2019, 24, 31–45. [Google Scholar] [CrossRef]
- Arlauskiene, A.; Maiksteniene, S.; Slepetiene, A. Application of environmental protection measures for clay loam cambisol used for agricultural purposes. J. Environ. Eng. Landsc. Manag. 2011, 19, 71–80. [Google Scholar] [CrossRef]
- Doltra, J.; Olesen, J.E. The role of catch crops in the ecological intensification of spring cereals in organic farming under Nordic climate. Eur. J. Agron. 2013, 44, 98–108. [Google Scholar] [CrossRef]
- Hansen, E.M.; Eriksen, J.; Vinther, F.P. Catch crop strategy and nitrate leaching following grazed grass-clover. Soil Use Manag. 2007, 23, 348–358. [Google Scholar] [CrossRef]
- Känkänen, H.; Eriksson, C. Effects of undersown crops on soil mineral N and grain yield of spring barley. Eur. J. Agron. 2007, 27, 25–34. [Google Scholar] [CrossRef]
- Malcolm, B.J.; Moir, J.L.; Cameron, K.C.; Di, H.J.; Edwards, G.R. Influence of plant growth and root architecture of Italian ryegrass (Lolium multiflorum) and tall fescue (Festuca arundinacea) on N recovery during winter. Grass Forage Sci. 2015, 70, 600–610. [Google Scholar] [CrossRef]
- Abdalla, M.; Hastings, A.; Cheng, K.; Yue, Q.; Chadwick, D.; Espenberg, M.; Truu, J.; Rees, R.M.; Smith, P. A critical review of the impacts of cover crops on nitrogen leaching, net greenhouse gas balance and crop productivity. Glob. Chang. Biol. 2019, 25, 2530–2543. [Google Scholar] [CrossRef]
- Guardia, G.; Aguilera, E.; Vallejo, A.; Sanz-Cobena, A.; Alonso-Ayuso, M.; Quemada, M. Effective climate change mitigation through cover cropping and integrated fertilization: A global warming potential assessment from a 10-year field experiment. J. Clean. Prod. 2019, 241. [Google Scholar] [CrossRef]
- Bert, S.; Bas, J.; Wiepie, H.; Wil, H.; Luis, A.J.; Jonas, K. Adoption of Cover Crops for Climate Change Mitigation in the EU; Joint Research Centre (Seville Site): Luxembourg, 2019. [Google Scholar]
- Sobkowicz, P. Uptake and use of nitrogen in the cultivation of spring barley with undersown Persian clover and serradella. Probl. Inż. Rol. 2009, 2, 99–106. [Google Scholar]
- Wanic, M.; Myśliwiec, M.; Orzech, K.; Michalska, M.; Denert, M. Competition for nitrogen, phosphorus, potassium and magnesium between spring wheat and persian clover depending on the density of plants. J. Elem. 2017, 22, 1081–1093. [Google Scholar] [CrossRef]
- Jastrzebska, M.; Kostrzewska, M.K.; Wanic, M.; Makowski, P.; Treder, K. Phosphorus content in spring Barley and red clover plants in pure and mixed sowing. Acta Sci. Pol. Agric. 2015, 14, 21–32. [Google Scholar]
- Kunelius, H.T.; McRae, K.B.; Dürr, G.H.; Fillmore, S.A.E. Management of Italian and perennial ryegrasses for seed and forage production in crop rotations. J. Agron. Crop Sci. 2004, 190, 130–137. [Google Scholar] [CrossRef]
- Salonen, J.; Ketoja, E. Undersown cover crops have limited weed suppression potential when reducing tillage intensity in organically grown cereals. Org. Agric. 2020, 10, 107–121. [Google Scholar] [CrossRef]
- Giraldo, P.; Benavente, E.; Manzano-Agugliaro, F.; Gimenez, E. Worldwide research trends on wheat and barley: A bibliometric comparative analysis. Agronomy 2019, 9, 352. [Google Scholar] [CrossRef]
- FAO (Food and Agriculture Organization of the United Nations). Crops Barley; 2018. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 7 February 2020).
- Agegnehu, G.; Ghizaw, A.; Sinebo, W. Yield performance and land-use efficiency of barley and faba bean mixed cropping in Ethiopian highlands. Eur. J. Agron. 2006, 25, 202–207. [Google Scholar] [CrossRef]
- Sobkowicz, P. Shoot and root competition between spring triticale and field beans during early growth. Acta Sci. Pol. Agric. 2005, 4, 117–126. [Google Scholar]
- Wanic, M.; Michalska, M. The influence of competition between spring barley and field pea on content of macroelements in different parts of the plants. Fragm. Agron. 2009, 26, 162–174. [Google Scholar]
- Pappa, V.A.; Rees, R.M.; Walker, R.L.; Baddeley, J.A.; Watson, C.A. Legumes intercropped with spring barley contribute to increased biomass production and carry-over effects. J. Agric. Sci. 2012, 150, 584–594. [Google Scholar] [CrossRef]
- Andersen, A.; Olsen, C.C. Rye grass as a catch crop in spring barley. Acta Agric. Scand. Sect. B Soil Plant Sci. 1993, 43, 218–230. [Google Scholar] [CrossRef]
- Ohlander, L.; Bergkvist, G.; Stendahl, F.; Kvist, M. Yield of catch crops and spring barley as affected by time of undersowing. Acta Agric. Scand. Sect. B Soil Plant Sci. 1996, 46, 161–168. [Google Scholar] [CrossRef]
- Kuraszkiewicz, R.; Palys, E. The influence of cover crops on the yield of aboveground parts of undersown crops. Ann. Univ. Mariae Curie Sklodowska Sectio E Agric. (Poland) 2005, 57, 105–112. [Google Scholar]
- Gürel, F.; Öztürk, Z.N.; Uçarlı, C.; Rosellini, D. Barley genes as tools to confer abiotic stress tolerance in crops. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Akram, M. Growth and yield components of wheat under water stress of different growth stages. Bangladesh J. Agric. Res. 2011, 36, 455–468. [Google Scholar] [CrossRef]
- Ding, J.; Huang, Z.; Zhu, M.; Li, C.; Zhu, X.; Guo, W. Does cyclic water stress damage wheat yield more than a single stress? PLoS ONE 2018, 13, e0195535. [Google Scholar] [CrossRef]
- The Biology of Lolium multiflorum Lam (Italian ryegrass), Lolium perenne (perenial ryegrass) and Lolium arundinaceum (Schreb) Darbysh (tall fescue); Department of Primary Industries: Melbourne, VIC, Australia, 2008; p. 83.
- Varella, A.C.; Carassai, I.J.; Baldissera, T.C.; Nabinger, C.; Lustosa, S.B.C.; Moraes, A.; Teixeira, S.J.; Vargas, A.S.; Radin, B. Annual ryegrass dry matter yield and nitrogen responses to fertiliser N applications in southern Brazil. Agron. N. Z. 2010, 40, 33–42. [Google Scholar]
- Mandić, V.; Simić, A.; Vučković, S.; Stanisavljević, R.; Tomić, Z.; Bijelić, Z.; Krnjaja, V. Management practices effect on seed features of Italian ryegrass following storage period. Biotechnol. Anim. Husb. 2014, 30, 145–152. [Google Scholar] [CrossRef]
- Kim, M.; Peng, J.L.; Sung, K. Causality between climatic and soil factors on Italian ryegrass yield in paddy field via climate and soil big data. J. Anim. Sci. Technol. 2019, 61, 324–332. [Google Scholar] [CrossRef]
- Olesen, J.E.; Hansen, E.M.; Askegaard, M.; Rasmussen, I.A. The value of catch crops and organic manures for spring barley in organic arable farming. Field Crop. Res. 2007, 100, 168–178. [Google Scholar] [CrossRef]
- Płaza, A.; Ceglarek, F.; Gąsiorowska, B.; Królikowska, M.A. The yielding and chemical composition of undersown crops. Fragm. Agron. 2009, 26, 93–99. [Google Scholar]
- Schachtman, D.P.; Reid, R.J.; Ayling, S.M. Phosphorus Uptake by Plants: From Soil to Cell. Plant Physiol. 1998, 116, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, F.; Ahmad, R.; Waraich, E.A.; Naeem, M.S.; Shabbir, R.N. Nutrient uptake, physiological responses, and yield attributes of wheat (triticum aestivum l.) exposed to early and late drought stress. J. Plant Nutr. 2012, 35, 961–974. [Google Scholar] [CrossRef]
- Suriyagoda, L.D.B.; Ryan, M.H.; Renton, M.; Lambers, H. Plant responses to limited moisture and phosphorus availability: A meta-analysis. In Advances in Agronomy; Academic Press Inc.: San Diego, CA, USA, 2014; Volume 124, pp. 143–200. [Google Scholar]
- Baligar, V.C.; Fageria, N.K.; He, Z.L. Nutrient use efficiency in plants. Commun. Soil Sci. Plant Anal. 2001, 32, 921–950. [Google Scholar] [CrossRef]
- Farahani, S.M.; Chaichi, M.R.; Mazaheri, D.; Afshari, R.T.; Savaghebi, G. Barley grain mineral analysis as affected by different fertilizing systems and by drought stress. J. Agric. Sci. Technol. 2011, 13, 315–326. [Google Scholar]
- Gonzalez-Dugo, V.; Durand, J.L.; Gastal, F.; Bariac, T.; Poincheval, J. Restricted root-to-shoot translocation and decreased sink size are responsible for limited nitrogen uptake in three grass species under water deficit. Environ. Exp. Bot. 2012, 75, 258–267. [Google Scholar] [CrossRef]
- Brodowska, M.S.; Filipek, T.; Kurzyna-Szklarek, M. Content of magnesium and calcium in cultivated plants depending on various soil supply with nitrogen, potassium, magnesium and sulfur. J. Elem. 2017, 22, 1167–1177. [Google Scholar] [CrossRef]
- Bista, D.R.; Heckathorn, S.A.; Jayawardena, D.M.; Mishra, S.; Boldt, J.K. Effects of drought on nutrient uptake and the levels of nutrient-uptake proteins in roots of drought-sensitive and -tolerant grasses. Plants 2018, 7, 28. [Google Scholar] [CrossRef]
- Dhima, K.V.; Lithourgidis, A.S.; Vasilakoglou, I.B.; Dordas, C.A. Competition indices of common vetch and cereal intercrops in two seeding ratio. Field Crop. Res. 2007, 100, 249–256. [Google Scholar] [CrossRef]
- Mariotti, M.; Masoni, A.; Ercoli, L.; Arduini, I. Above- and below-ground competition between barley, wheat, lupin and vetch in a cereal and legume intercropping system. Grass Forage Sci. 2009, 64, 401–412. [Google Scholar] [CrossRef]
- Eskandari, H.; Ghanbari, A. Effect of different planting pattern of wheat (Triticum aestivum) and bean (Vicia faba) on grain yield, dry matter production and weed biomass. Not. Sci. Biol. 2010, 2, 111–115. [Google Scholar] [CrossRef][Green Version]
- Alaru, M.; Talgre, L.; Luik, A.; Tein, B.; Eremeev, V.; Loit, E. Barley undersown with red clover in organic and conventional systems: Nitrogen aftereffect on legume growth. Zemdirbyste 2017, 104, 131–138. [Google Scholar] [CrossRef]
- Darch, T.; Giles, C.D.; Blackwell, M.S.A.; George, T.S.; Brown, L.K.; Menezes-Blackburn, D.; Shand, C.A.; Stutter, M.I.; Lumsdon, D.G.; Mezeli, M.M.; et al. Inter- and intra-species intercropping of barley cultivars and legume species, as affected by soil phosphorus availability. Plant Soil 2018, 427, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Weigelt, A.; Jolliffe, P. Indices of plant competition. J. Ecol. 2003, 91, 707–720. [Google Scholar] [CrossRef]
- Rahetlah, V.B.; Randrianaivoarivony, J.M.; Andrianarisoa, B.; Razafimpamoa, L.H.; Ramalanjaona, V.L. Yields and quality of Italian ryegrass (Lolium multiflorum) and Common Vetch (Vicia sativa) grown in monocultures and mixed cultures under irrigated conditions in the Highlands of Madagascar. Sustain. Agric. Res. 2013, 2. [Google Scholar] [CrossRef]
- Sobkowicz, P.; Podgórska-Lesiak, M. Assessment of barley interactions in mixture with triticale or field pea affected by nitrogen fertilizer rate. Fragm. Agron. 2009, 26, 115–126. [Google Scholar]
- Hinsinger, P.; Betencourt, E.; Bernard, L.; Brauman, A.; Plassard, C.; Shen, J.; Tang, X.; Zhang, F. P for two, sharing a scarce resource: Soil phosphorus acquisition in the rhizosphere of intercropped species. Plant Physiol. 2011, 156, 1078–1086. [Google Scholar] [CrossRef]
- Li, C.; Dong, Y.; Li, H.; Shen, J.; Zhang, F. Shift from complementarity to facilitation on P uptake by intercropped wheat neighboring with faba bean when available soil P is depleted. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Snaydon, R.W. Replacement or additive designs for competition studies? J. Appl. Ecol. 1991, 28, 930–946. [Google Scholar] [CrossRef]
- Semere, T.; Froud-Williams, R.J. The effect of pea cultivar and water stress on root and shoot competition between vegetative plants of maize and pea. J. Appl. Ecol. 2001, 38, 137–145. [Google Scholar] [CrossRef]
- Pasze—Oznaczanie Zawartości Fosforu—Metoda Spektrometryczna; Polski Komitet Normalizacyjny: Warszawa, Polska, 2000; p. 9.
- De Wit, C.T.; Van den Bergh, J.P. Competition between herbage plants. J. Agric. Sci. 1965, 13, 212–221. [Google Scholar]
- Wilson, J.B. Shoot competition and root competition. J. Appl. Ecol. 1988, 25, 279–296. [Google Scholar] [CrossRef]
- StatSoft, I. Statistica (Data Analysis Software System), Version 12; Statsoft Inc.: Tulsa, OK, USA, 2014. [Google Scholar]
- Kristoffersen, A.Ø.; Riley, H. Effects of soil compaction and moisture regime on the root and shoot growth and phosphorus uptake of barley plants growing on soils with varying phosphorus status. Nutr. Cycl. Agroecosyst. 2005, 72, 135–146. [Google Scholar] [CrossRef]
- Alghabari, F.; Ihsan, M.Z. Effects of drought stress on growth, grain filling duration, yield and quality attributes of barley (Hordeum vulgare L.). Bangladesh J. Bot. 2018, 47, 421–428. [Google Scholar] [CrossRef]
- Ali, A.H.; Mansorr, H.N. Effect of imposed water stress at certain growth stages on growth and yield of barley grown under different planting patterns. Plant Arch. 2018, 18, 1735–1744. [Google Scholar]
- Boudiar, R.; Casas, A.M.; Gioia, T.; Fiorani, F.; Nagel, K.A.; Igartua, E. Effects of low water availability on root placement and shoot development in landraces and modern barley cultivars. Agronomy 2020, 10, 134. [Google Scholar] [CrossRef]
- AbdElgawad, H.; Peshev, D.; Zinta, G.; Van Den Ende, W.; Janssens, I.A.; Asard, H. Climate extreme effects on the chemical composition of temperate grassland species under ambient and elevated CO2: A comparison of fructan and non-fructan accumulators. PLoS ONE 2014, 9, e92044. [Google Scholar] [CrossRef]
- Høgh-Jensen, H.; Schjoerring, J.K. Interactions between nitrogen, phosphorus and potassium determine growth and N2-fixation in white clover and ryegrass leys. Nutr. Cycl. Agroecosyst. 2010, 87, 327–338. [Google Scholar] [CrossRef]
- Brink, G.E.; Pederson, G.A.; Sistani, K.R.; Fairbrother, T.E. Forages: Uptake of selected nutrients by temperate grasses and legumes. Agron. J. 2001, 93, 887–890. [Google Scholar] [CrossRef]
- Burkitt, L.L.; Turner, L.R.; Donaghy, D.J.; Fulkerson, W.J.; Smethurst, P.J.; Roche, J.R. Characterisation of phosphorus uptake by perennial ryegrass (Lolium perenne L.) during regrowth. N. Z. J. Agric. Res. 2009, 52, 195–202. [Google Scholar] [CrossRef]
- Norris, I.B.; Thomas, H. Recovery of ryegrass species from drought. J. Agric. Sci. 1982, 98, 623–628. [Google Scholar] [CrossRef]
- Jerónimo, P.A.; Hrabě, F.; Knot, P.; Kvasnovský, M. Evaluation of suitability of grass species for dry conditions (water stress). Acta Univ. Agric. Silvic. Mendel. Brun. 2014, 62, 953–960. [Google Scholar] [CrossRef]
- Staniak, M. The impact of drought stress on the yields and food value of selected forage grasses. Acta Agrobot. 2016, 69. [Google Scholar] [CrossRef]
- Paris, W.; Marchesan, R.; Cecato, U.; Martin, T.N.; Ziech, M.F.; Borges, G.D.S. Dynamics of yield and nutritional value for winter forage intercropping. Acta Sci. Anim. Sci. 2012, 34, 109–115. [Google Scholar] [CrossRef]
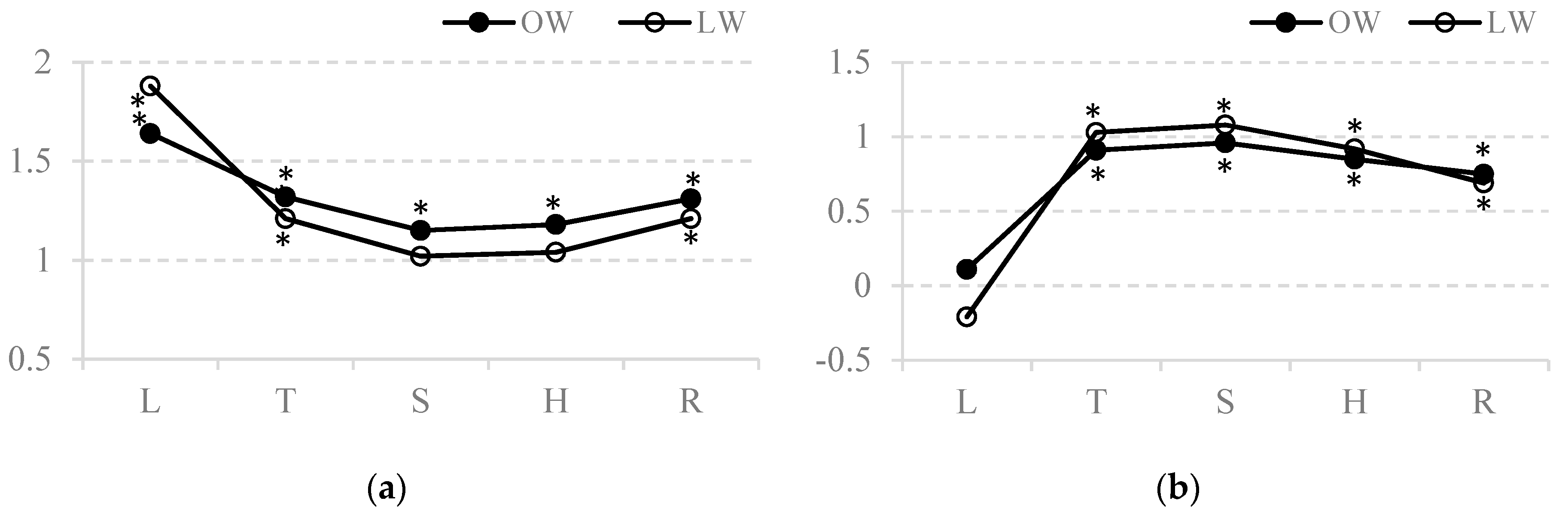
| Growth Stage of Barley | Plant Part | Water Supply | Sowing Type | Water Supply x Sowing Type | |||||
|---|---|---|---|---|---|---|---|---|---|
| OW | LW | BP | BM | BP-OW | BM-OW | BP-LW | BM-LW | ||
| Leaf development | shoots | 5.57 a | 4.10 b | 5.22 a | 4.44 b | 5.98 a | 5.15 b | 4.46 c | 3.74 d |
| Tillering | shoots | 22.47 a | 10.53 b | 17.13 a | 15.87 b | 23.13 a | 21.81 b | 11.13 c | 9.92 c |
| Stem elongation | shoots | 57.01 a | 22.77 b | 44.15 a | 35.64 b | 62.42 a | 51.60 b | 25.88 c | 19.67 d |
| leaves | 20.87 a | 10.02 b | 17.41 a | 13.48 b | 23.51 a | 18.23 b | 11.32 c | 8.72 d | |
| stems | 36.14 a | 12.76 b | 26.74 a | 22.16 b | 38.91 a | 33.37 b | 14.56 c | 10.95 d | |
| Heading | shoots | 68.28 a | 29.66 b | 54.37 a | 43.57 b | 74.75 a | 61.82 b | 33.99 c | 25.33 d |
| leaves | 18.37 a | 11.57 b | 17.47 a | 12.47 b | 22.52 a | 14.22 b | 12.43 c | 10.72 d | |
| stems | 35.74 a | 13.99 b | 26.61 a | 23.13 b | 36.57 a | 34.92 b | 16.65 c | 11.34 d | |
| spikes | 14.17a | 4.09 b | 10.29 a | 7.98 b | 15.66 a | 12.69 b | 4.91 c | 3.28 d | |
| Ripening | shoots | 48.88 a | 36.58 b | 46.21 a | 39.25 b | 51.85 a | 45.91 b | 40.58 c | 32.59 d |
| leaves | 10.46 a | 9.42 b | 10.04 a | 9.84 a | 10.40 ab | 10.51 a | 9.68 bc | 9.17 c | |
| stems | 20.15 a | 17.43 b | 19.86 a | 17.71 b | 19.44 a | 20.85 a | 20.28 a | 14.57 b | |
| spikes | 18.28 a | 9.74 b | 16.31 a | 11.70 b | 22.00 a | 14.55 b | 10.62 c | 8.86 d | |
| Growth Stage of Barley | Plant Part | Barley | Rye-Grass | ||
|---|---|---|---|---|---|
| P Content | Biomass | P Content | Biomass | ||
| Leaf development | shoots | 0.041 | 0.977 * | 0.291 * | 0.960 * |
| Tillering | shoots | 0.618 * | 0.944 * | 0.584 * | 0.993 * |
| Stem elongation | leaves | 0.481 * | 0.946 * | 0.293 * | 0.946 * |
| stems | 0.602 * | 0.968 * | 0.229 | 0.990 * | |
| Heading | leaves | 0.116 | 0.934 * | −0.024 | 0.963 * |
| stems | 0.557 * | 0.905 * | −0.229 | 0.980 * | |
| spikes | 0.274 | 0.942 * | – | – | |
| Ripening | leaves | 0.125 | 0.769 * | 0.110 | 0.850 * |
| stems | 0.493 * | 0.880 * | −0.591 * | 0.985 * | |
| spikes | −0.035 | 0.994 * | – | – | |
| Growth Stage of Barley | Plant Part | Water Supply | Sowing Type | Water Supply x Sowing Type | |||||
|---|---|---|---|---|---|---|---|---|---|
| OW | LW | RP | RM | RP-OW | RM-OW | RP- LW | RM-LW | ||
| Leaf development | shoots | 0.94 a | 0.65 b | 0.85 a | 0.74 b | 1.06 a | 0.82 b | 0.64 c | 0.66c |
| Tillering | shoots | 9.67 a | 3.34 b | 9.54 a | 3.47 b | 14.00 a | 5.33 b | 5.07 b | 1.62 c |
| Stem elongation | shoots | 28.34 a | 14.10 b | 32.71 a | 9.73 b | 43.03 a | 13.64 c | 22.39 b | 5.81 d |
| leaves | 14.02 a | 9.02 b | 16.50 a | 6.54 b | 18.80 a | 9.23 c | 14.20 b | 3.85 d | |
| stems | 14.32 a | 5.08 b | 16.21 a | 3.19 b | 24.23 a | 4.41 c | 8.19 b | 1.96 d | |
| Heading | shoots | 57.69 a | 29.82 b | 65.56 a | 21.95 b | 85.22 a | 30.16 c | 45.90 b | 13.74 d |
| leaves | 36.26 a | 18.98 b | 40.77 a | 14.47 b | 52.56 a | 19.96 c | 28.98 b | 8.99 d | |
| stems | 21.43 a | 10.83 b | 24.79 a | 7.47 b | 32.66 a | 10.20 c | 16.92 b | 4.75 d | |
| Ripening | shoots | 63.58 a | 35.71 b | 70.27 a | 29.02 b | 89.6 a | 37.55 c | 50.92 b | 20.49 d |
| leaves | 44.71 a | 25.71 b | 50.20 a | 20.22 b | 63.64 a | 25.78 c | 36.76 b | 14.66 d | |
| stems | 18.82 a | 10.00 b | 20.07 a | 8.75 b | 25.98 a | 11.66 c | 14.16 b | 5.83 d | |
| Growth Stage of Barley | Water Supply | |||
|---|---|---|---|---|
| OW | LW | |||
| RYB | RYR | RYB | RYR | |
| Leaf development | 0.86 bc* | 0.78 a* | 0.84 ab* | 1.04 a |
| Tillering | 0.94 a | 0.38 bc* | 0.89 a* | 0.32 bc* |
| Stem elongation | 0.83 c* | 0.32 c* | 0.76 c* | 0.26 c* |
| Heading | 0.83 c* | 0.35 bc* | 0.75 c* | 0.30 bc* |
| Ripening | 0.89 b* | 0.42 b* | 0.80 bc* | 0.40 b* |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kostrzewska, M.K.; Jastrzębska, M.; Treder, K.; Wanic, M. Phosphorus in Spring Barley and Italian Rye-Grass Biomass as an Effect of Inter-Species Interactions under Water Deficit. Agriculture 2020, 10, 329. https://doi.org/10.3390/agriculture10080329
Kostrzewska MK, Jastrzębska M, Treder K, Wanic M. Phosphorus in Spring Barley and Italian Rye-Grass Biomass as an Effect of Inter-Species Interactions under Water Deficit. Agriculture. 2020; 10(8):329. https://doi.org/10.3390/agriculture10080329
Chicago/Turabian StyleKostrzewska, Marta K., Magdalena Jastrzębska, Kinga Treder, and Maria Wanic. 2020. "Phosphorus in Spring Barley and Italian Rye-Grass Biomass as an Effect of Inter-Species Interactions under Water Deficit" Agriculture 10, no. 8: 329. https://doi.org/10.3390/agriculture10080329
APA StyleKostrzewska, M. K., Jastrzębska, M., Treder, K., & Wanic, M. (2020). Phosphorus in Spring Barley and Italian Rye-Grass Biomass as an Effect of Inter-Species Interactions under Water Deficit. Agriculture, 10(8), 329. https://doi.org/10.3390/agriculture10080329






