Repurposing Immunomodulatory Therapies against Coronavirus Disease 2019 (COVID-19) in the Era of Cardiac Vigilance: A Systematic Review
Abstract
1. Introduction
2. COVID-19 Clinical Observations
2.1. Viral Phase
2.2. Immune Response Phase
2.3. Thromboembolism
2.4. Cardiovascular Complications
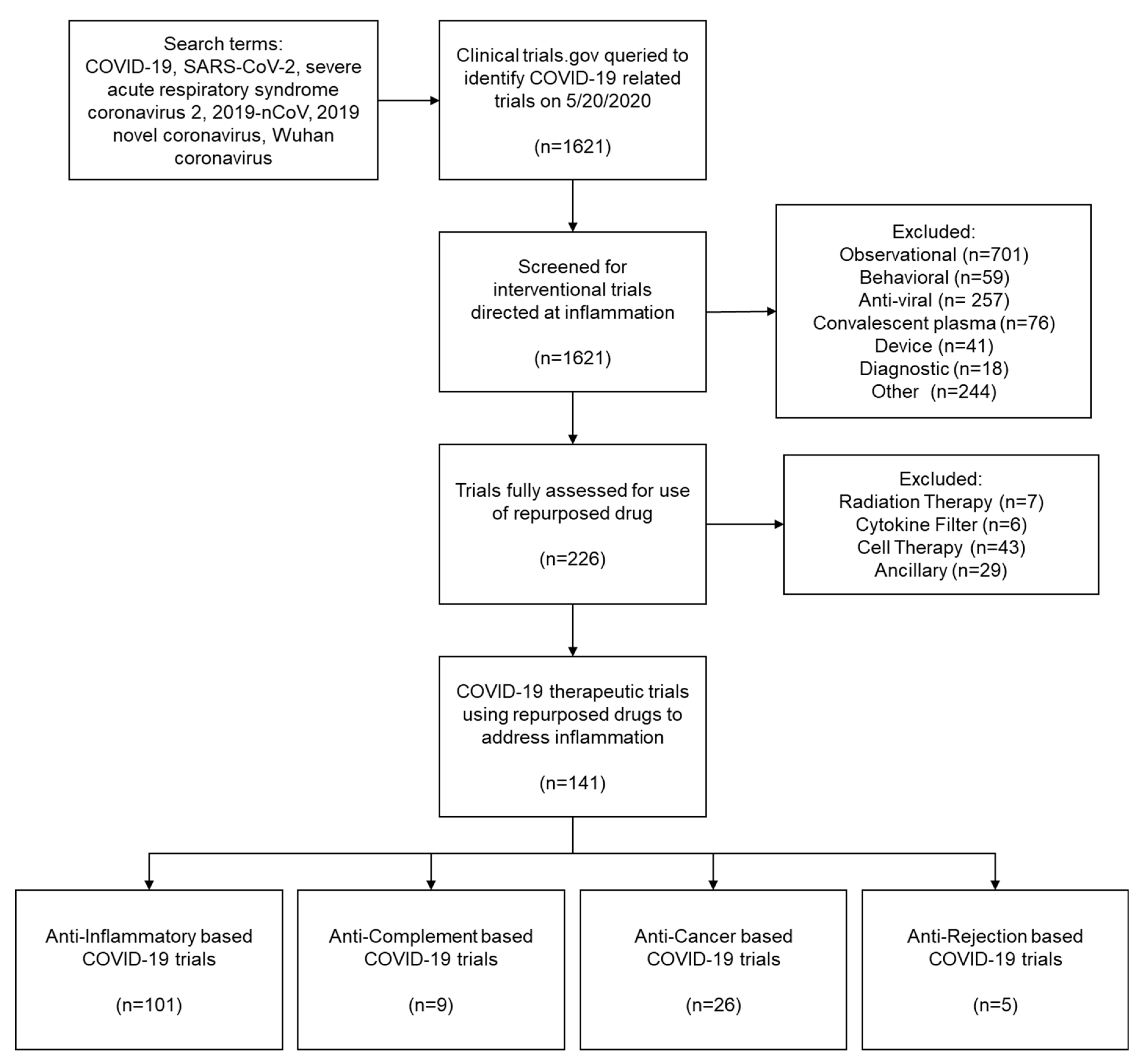
3. Systematic Review Methodology
3.1. Search Strategy
3.2. Inclusion/Exclusion Critieria
3.3. Screening Based on Inclusion/Exclusion Criteria
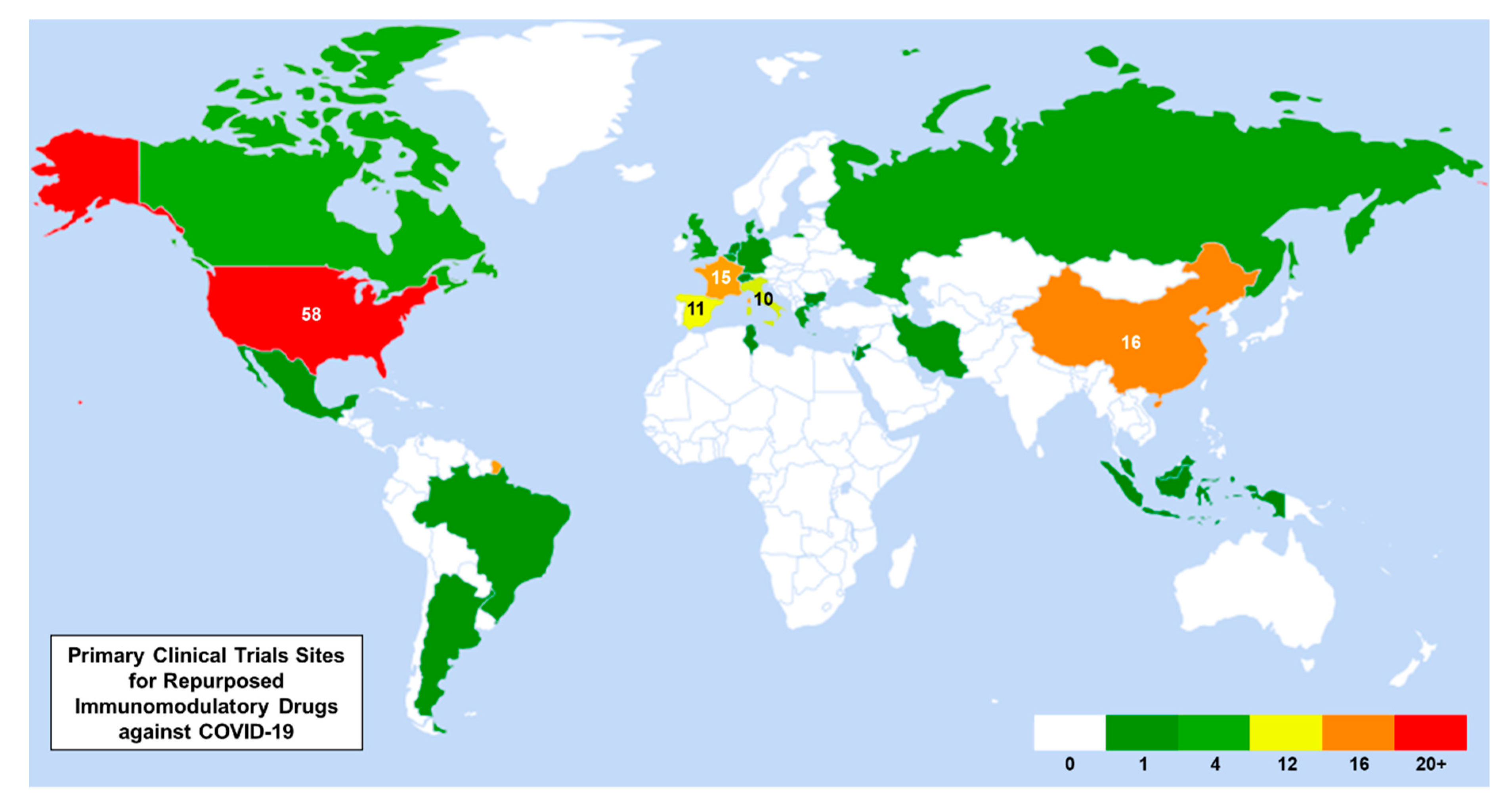
3.4. World Heat Map
4. Results of Repurposed Therapeutic Clinical Trials for COVID-19 Inflammation
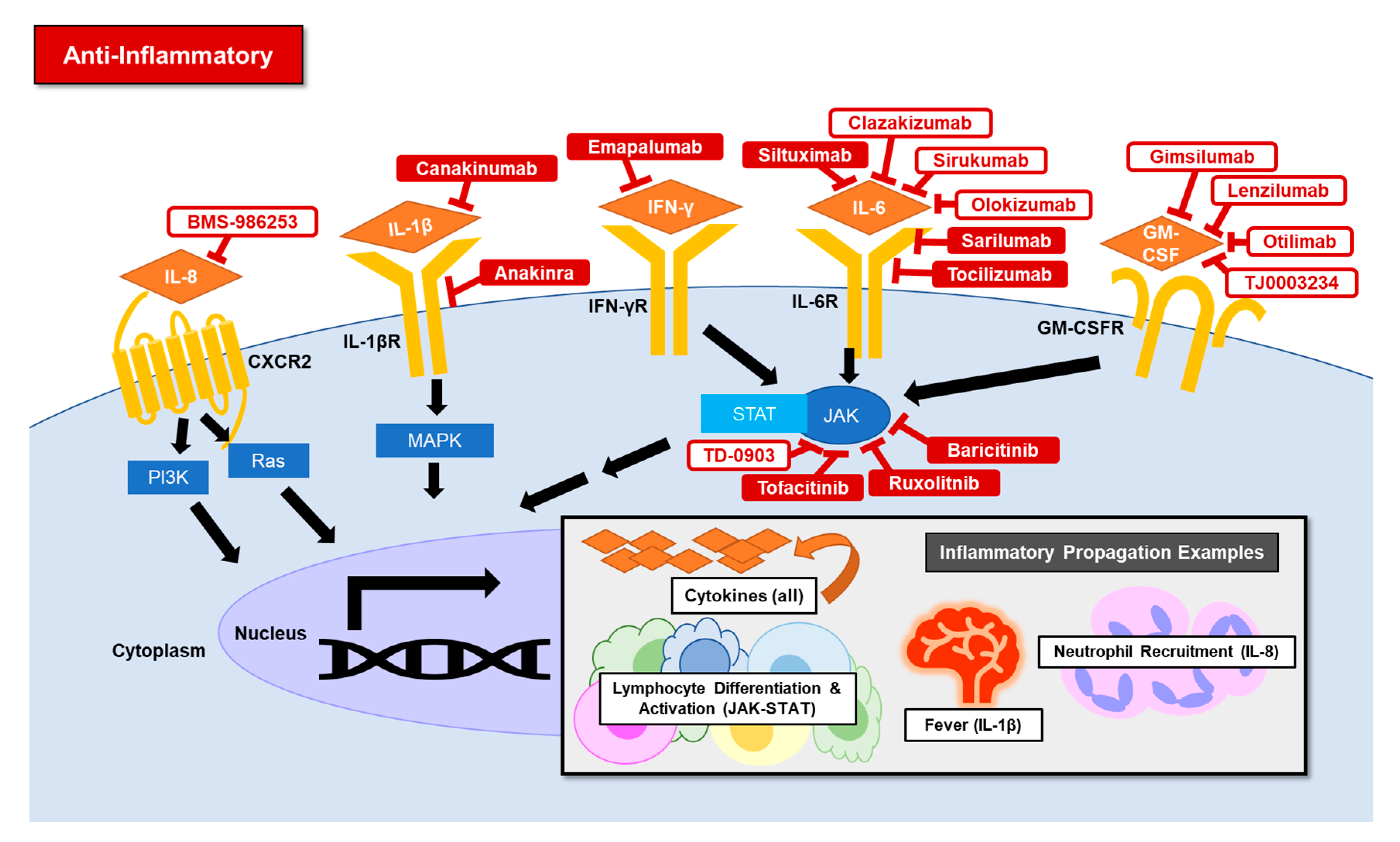
4.1. Anti-Inflammatory Agents
4.1.1. Overview
4.1.2. Clinical Uses
- JAK inhibitor: A pilot study of ruxolitinib demonstrated improvement in HLH [46]. In addition, an artificial intelligence algorithm predicted baricitinib to be a numb-associated kinase (NAK) inhibitor at the doses used for rheumatoid arthritis treatment [47]. In vitro NAK inhibition can reduce viral infection through clathrin-mediated endocytosis blockade [48]. These data suggest baricitinib may have both anti-inflammatory and anti-viral effects. Tofacitinib has not directly been trialed CRS but is a significant cytokine inhibitor [49]. TD-0903 is an investigational JAK inhibitor that is nebulized and lung-selective, per press releases [50].
- IL-6 inhibitor: IL-6 levels correlate with severe disease in SARS [54]. Tocilizumab has been used to treat HLH, GvHD, and CRS induced by CAR T-cell therapy [55,56]. Preclinical studies are supportive of siltuximab for CRS induced by CAR T-cell therapy [57]. Sarilumab has not been directly tested in CRS.
- IL-8 inhibitor: IL-8 is elevated in CRS induced by CAR-T cell therapy [58]. No IL-8 inhibitors are currently FDA approved. IL-8 inhibitors are under investigation for the treatment of malignant solid tumors. High IL-8 levels are associated with tumor progression and epithelial-mesenchymal transition [59].
- IFN-beta-1: Interferon-beta-1 is a cytokine that has anti-viral, anti-proliferative, and immunomodulatory effects. It is FDA-approved to treat multiple sclerosis. IFN-beta increases the production of anti-inflammatory cytokines, such as IL-10, and limits leukocyte migration across the blood-brain-barrier [62,63]. IFN-beta was protective in septic shock and ARDS murine models [64,65]. In an open-label study, treatment with IFN-beta in ARDS was associated with decreased 28-day mortality [66]. However, IFN-beta did not improve outcomes in a recent randomized control trial for the treatment of moderate to severe ARDS [67].
- GM-CSF: No GM-CSF inhibitors under investigation for COVID-19 are currently FDA-approved for other indications. Lenzilumab and TJ003234 are under investigation to treat CRS induced by CAR T cell therapy. GM-CSF inhibitors are also under investigation to treat acute graft versus host disease, ankylosing spondylitis, and rheumatoid arthritis [68,69].
- TNF-alpha: Although TNF-alpha inhibitors are commonly used to treat rheumatoid arthritis, only one investigational TNF-alpha inhibitor, XPro1595, is being evaluated in a clinical trial for COVID-19. Murine models of severe influenza treated with TNF-alpha inhibitors had reduced cytokine production without changes in survival rates [70,71].
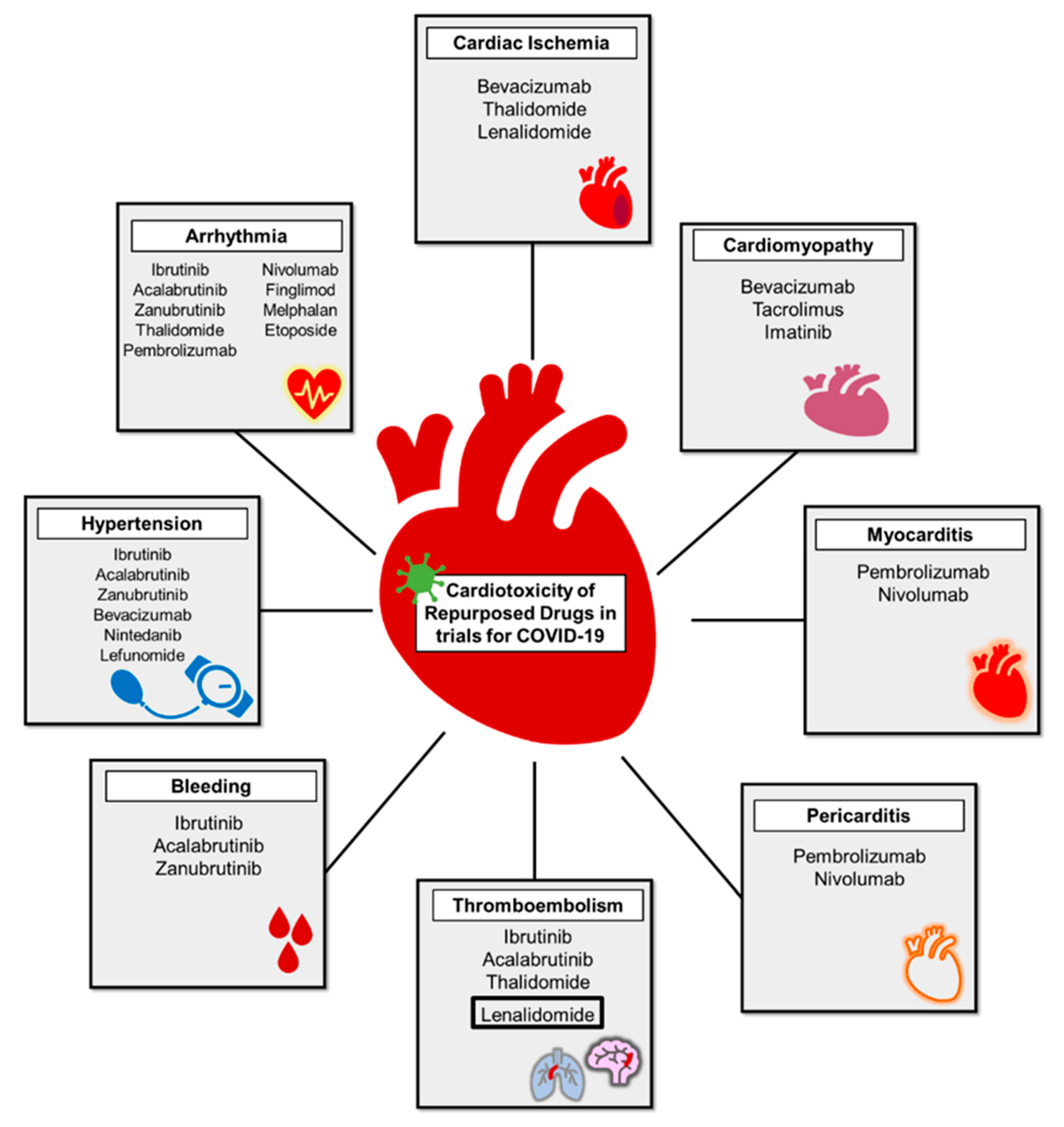
4.1.3. Cardiotoxicity
4.1.4. Early COVID-19 Experience
4.2. Anti-Rejection Agents
4.2.1. Overview
4.2.2. Clinical Uses
4.2.3. Cardiotoxicity
4.2.4. Early COVID-19 Experience
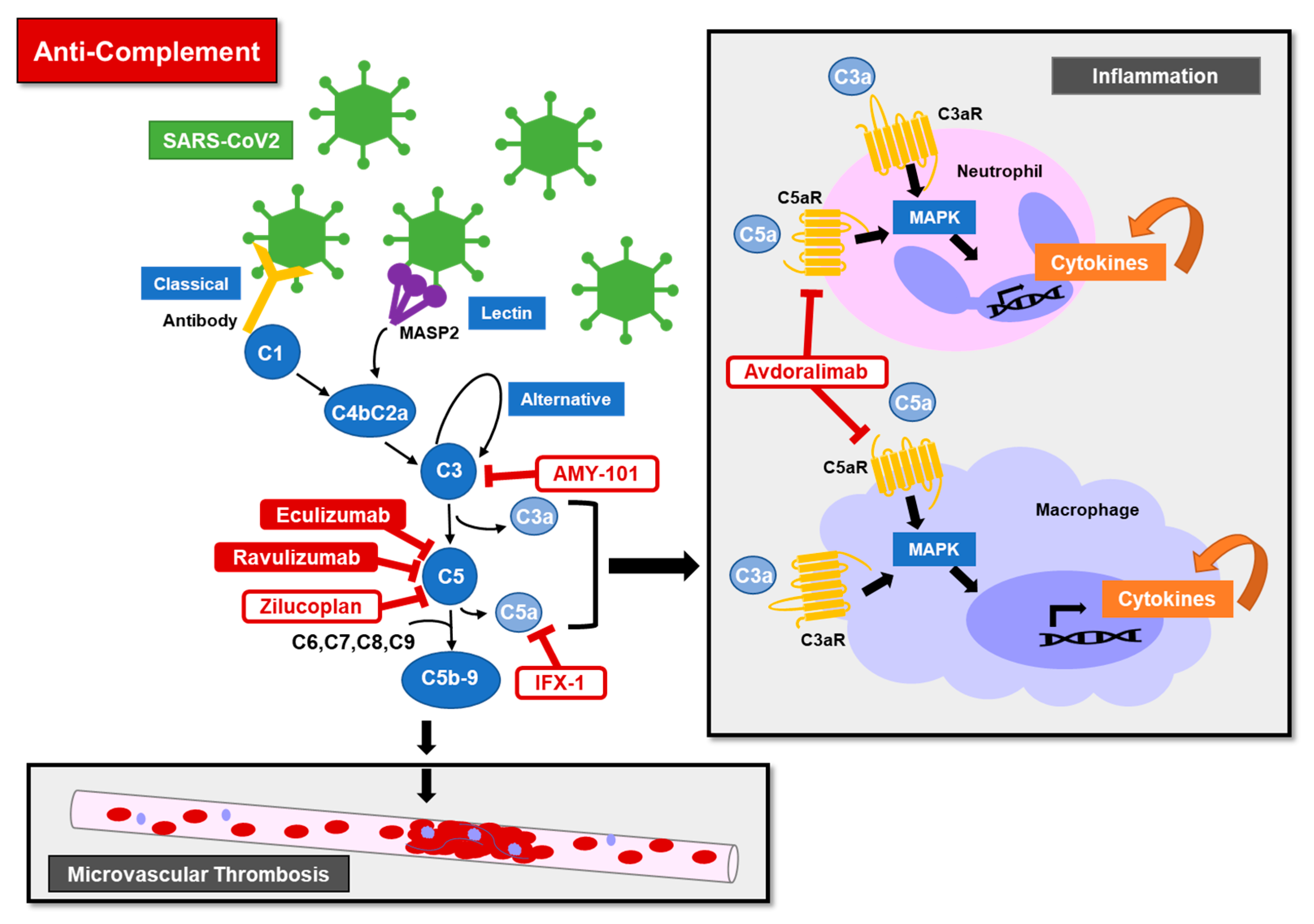
4.3. Anti-Complement Agents
4.3.1. Overview
4.3.2. Clinical Uses
4.3.3. Cardiotoxicity
4.3.4. Early COVID-19 Experience
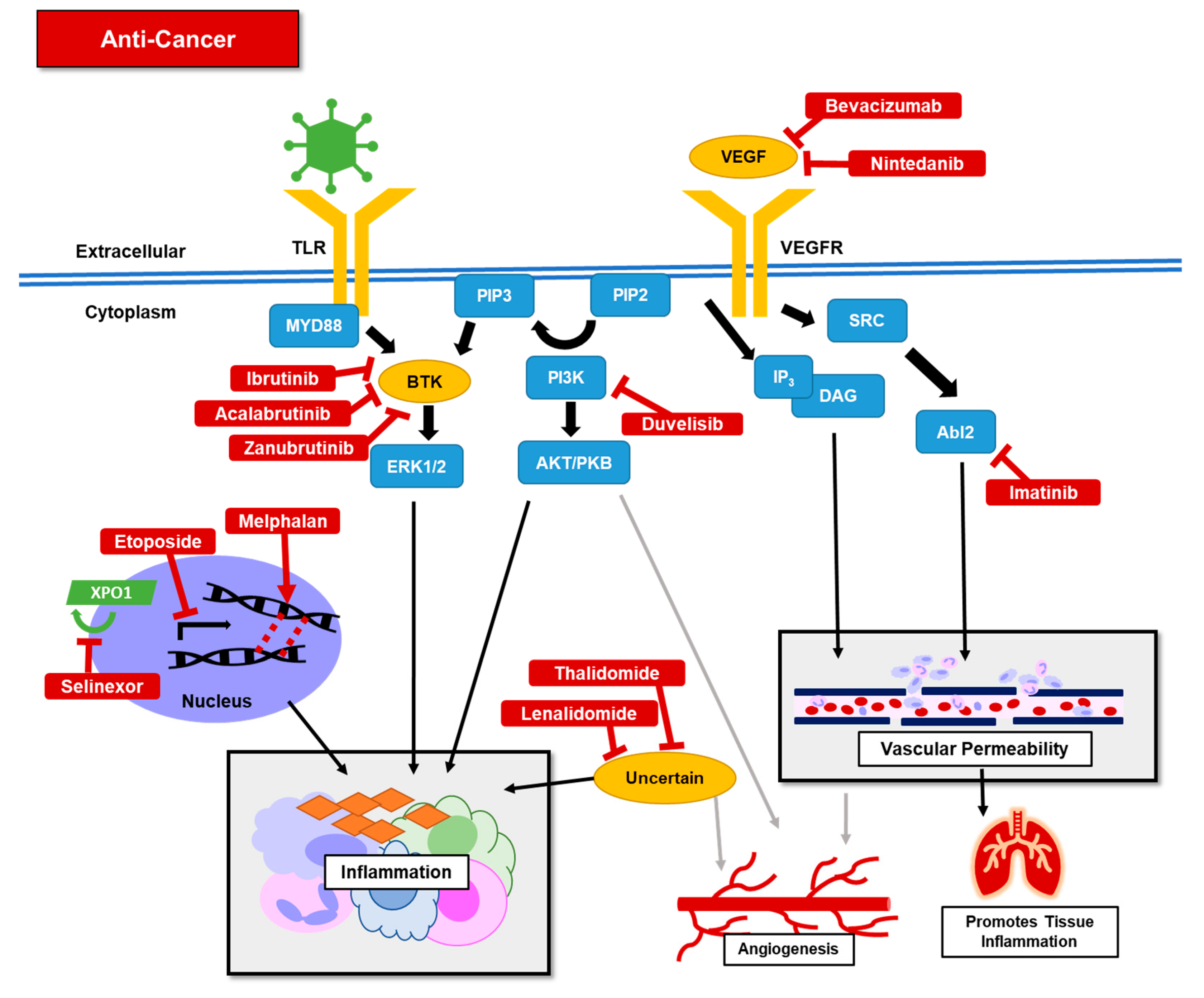
4.4. Anti-Cancer Agents
4.4.1. Overview
4.4.2. Clinical Uses
- Targeted therapies:
- Breakpoint cluster region (BCR)-Abelson’s (ABL) tyrosine kinase inhibitor: Imatinib may have both anti-viral and anti-inflammatory effects. In vitro studies demonstrate that the imatinib target, Abelson tyrosine-protein kinase 2, is required for efficient SARS-CoV1 and MERS-CoV replication [111]. In murine models, imatinib inhibited endothelial permeability, attenuating pulmonary edema in sepsis models [112,113].
- PI3 kinase inhibitors: Some isoforms of PI3 kinase are preferentially expressed in leukocytes, and inhibition resulted in blocking B and T cell proliferation and neutrophil migration in rodents [116]. Blockade of PI3 kinase also improved rodent models of arthritis, asthma, and systemic lupus erythematosus. Inhibition of PI3 decreased bronchoalveolar lavage eosinophils in a murine pulmonary inflammation model [117].
- VEGF inhibitors: Elevated VEGF levels have been observed in COVID-19 patients, and VEGF activation is associated with ARDS. Anti-VEGF therapies may suppress pulmonary edema, improving ARDS [118].
- Cytotoxic therapies: Etoposide, a topoisomerase II inhibitor, is used as part of the standard of care of HLH [119,120], likely effective through activated T cells ablation [121]. Melphalan, a DNA alkylator, used in non-cytotoxic doses, has been associated with anti-inflammatory effects through disruption of IL-2-beta and TNF-alpha receptor signaling [122]. Selinexor is an inhibitor of exportin-1, a nuclear export protein. Exportin-1 is thought to be important in both viral replication and mediating the inflammatory response through nuclear factor kappa-B signaling [123].
- Immunomodulatory: Thalidomide and lenalidomide have been shown to reduce the inflammatory response in patients with idiopathic pulmonary fibrosis and in rat models of paraquat lung toxicity [124,125]. In mouse studies of H1N1 influenza, thalidomide was shown to reduced inflammation and improved survival rate [126].
- Immune checkpoint inhibitors (ICIs): The use of ICIs in COVID-19 is controversial as the rare complication of inflammatory pneumonitis may overlap with COVID-19 interstitial pneumonia [127]. Clinical trials are aimed at ICI use in early viral clearance and the safety of continued ICI use in patients with cancer that become infected with COVID-19.
4.4.3. Cardiotoxicity
4.4.4. Early COVID-19 Experience
4.5. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| A3AR | A3 adenosine receptor |
| Abl2 | Abelson tyrosine kinase 2 |
| AKT/PKB | serine/threonine-specific protein kinase/protein kinase B; |
| BCR-ABL TK | breakpoint cluster region protein-Abelson tyrosine kinase; |
| BTK | Bruton’s tyrosine kinase |
| C5aR or C3aR | complement component receptor |
| CD | cluster of differentiation |
| CXCR2 | C-X-C motif chemokine receptor 2 |
| DAG | diacylglycerol |
| DNA | deoxyribonucleic acid. |
| EF1A2 | elongation factor 1-alpha 2 |
| ERK1/2 | extracellular signal-related protein kinase 1/2 |
| FGFR | fibroblast growth factor receptor; |
| GM-CSF | granulocyte-macrophage colony-stimulating factor |
| GM-CSFR | granulocyte-macrophage colony-stimulating factor receptor |
| IFN | interferon |
| IFN-g | interferon gamma |
| IFN-gR | interferon gamma receptor |
| IL | interleukin |
| IL-1B | interleukin 1-beta |
| IL-1BR | interleukin-1-beta receptor |
| IL-6 | interleukin 6 |
| IL-6R | interleukin 6 receptor |
| IL-8 | interleukin 8 |
| IP3 | inositol trisphosphate |
| JAK | Janus tyrosine kinase |
| MAPK | mitogen-activated protein kinase |
| miR | micro-ribonucleic acid |
| mTOR | mammalian target of rapamycin |
| MYD88 | myeloid differentiation primary response 88 |
| PDGFR | platelet-derived growth factor receptor |
| PI3K | phosphoinositide 3-kinase |
| PIP2 | phosphatidylinositol (4,5)-bisphosphate |
| PIP3 | phosphatidylinositol (3,4,5)-triphosphate |
| Ras | rat sarcoma |
| SRC | cytoplasmic tyrosine kinase |
| ST2 | serum stimulation 2 |
| STAT | signal transducer and activator of transcription |
| TLR | Toll-like receptor |
| TNF | tumor necrosis factor |
| VEGF | vascular endothelial growth factor |
| VEGFR | vascular endothelial growth factor receptor |
| XPO1 | exportin-1 |
| ‘C’ | denotes complement component |
References
- Paules, C.I.; Marston, H.D.; Fauci, A.S. Coronavirus Infections-More Than Just the Common Cold. JAMA 2020, 323, 707. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and Clinical Characteristics of 99 Cases of 2019-Novel Coronavirus (2019-nCoV) Pneumonia in Wuhan, China. SSRN Electron. J. 2020, 395, 507–513. [Google Scholar] [CrossRef]
- Sanders, J.M.; Monogue, M.L.; Jodlowski, T.Z.; Cutrell, J.B. Pharmacologic Treatments for Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020, 323, 1824–1836. [Google Scholar] [CrossRef]
- Schoolman, H.M. The United States National Library of Medicine. Semin. Dermatol. 1989, 8. [Google Scholar]
- Roden, D.M.; Harrington, R.A.; Poppas, A.; Russo, A.M. Considerations for Drug Interactions on QTc Interval in Exploratory COVID-19 Treatment. J. Am. Coll. Cardiol. 2020, 75, 2623. [Google Scholar] [CrossRef]
- Siddiqi, H.K.; Mehra, M.R. COVID-19 illness in native and immunosuppressed states: A clinical–therapeutic staging proposal. J. Hear. Lung Transplant. 2020, 39, 405–407. [Google Scholar] [CrossRef]
- Mizumoto, K.; Kagaya, K.; Zarebski, A.; Chowell, G. Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the Diamond Princess cruise ship, Yokohama, Japan, 2020. Eurosurveillance 2020, 25, 2000180. [Google Scholar] [CrossRef]
- Pepperrell, T.; Pilkington, V.; Owen, A.; Wang, J.; Hill, A. Review of safety and minimum pricing of nitazoxanide for potential treatment of COVID-19. J. Virus Erad. 2020, 6, 52–60. [Google Scholar] [CrossRef]
- Guan, W.-J.; Ni, Z.-Y.; Hu, Y.; Liang, W.-H.; Ou, C.-Q.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-L.; Hui, D.S.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China. A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA 2020, 323, 1061. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yang, B.; Li, Q.; Wen, L.; Zhang, R. Clinical Features of 69 Cases with Coronavirus Disease 2019 in Wuhan, China. Clin. Infect. Dis. 2020, 71, 769–777. [Google Scholar] [CrossRef]
- Peiris, J.; Chu, C.; Cheng, V.; Chan, K.; Hung, I.F.-N.; Poon, L.L.M.; Law, K.; Tang, B.; Hon, T.; Chan, C.; et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia. A prospective study. Lancet 2003, 361, 1767–1772. [Google Scholar] [CrossRef]
- Cameron, M.J.; Ran, L.; Xu, L.; Danesh, A.; Bermejo-Martin, J.F.; Cameron, C.M.; Muller, M.P.; Gold, W.L.; Richardson, S.E.; Poutanen, S.; et al. Interferon-Mediated Immunopathological Events Are Associated with Atypical Innate and Adaptive Immune Responses in Patients with Severe Acute Respiratory Syndrome. J. Virol. 2007, 81, 8692–8706. [Google Scholar] [CrossRef]
- Stockman, L.J.; Bellamy, R.; Garner, P. SARS: Systematic Review of Treatment Effects. PLoS Med. 2006, 3, 343. [Google Scholar] [CrossRef]
- Lansbury, L.E.; Rodrigo, C.; Leonardi-Bee, J.; Nguyen-Van-Tam, J.; Lim, W.S. Corticosteroids as Adjunctive Therapy in the Treatment of Influenza. Crit. Care Med. 2020, 48, 98–106. [Google Scholar] [CrossRef]
- Arabi, Y.; Mandourah, Y.; Al-Hameed, F.; Sindi, A.A.; Almekhlafi, G.A.; Hussein, M.; Jose, J.; Pinto, R.; Al-Omari, A.; Kharaba, A.; et al. Corticosteroid Therapy for Critically Ill Patients with Middle East Respiratory Syndrome. Am. J. Respir. Crit. Care Med. 2018, 197, 757–767. [Google Scholar] [CrossRef]
- Russell, C.D.; Millar, J.E.; Baille, K.J. Clinical evidence does not support corticosteroid treatment for 2019-nCoV lung injury. Lancet 2020, 395, 473–475. [Google Scholar] [CrossRef]
- World Health Organization. WHO Welcomes Preliminary Results about Dexamethasone Use in Treating Critically Ill COVID-19 Patients. Available online: https://www.who.int/news-room/detail/16-06-2020-who-welcomes-preliminary-results-about-dexamethasone-use-in-treating-critically-ill-covid-19-patients (accessed on 16 June 2020).
- RECOVERY Central Coordinating Office, Randomized Evaluation of COVID-19 Therapy (RECOVERY) Protocol. Available online: https://www.recoverytrial.net/files/recovery-protocol-v6-0-2020-05-14.pdf (accessed on 18 June 2020).
- Bowles, L.; Platton, S.; Yartey, N.; Dave, M.; Lee, K.; Hart, D.P.; Macdonald, V.; Green, L.; Sivapalaratnam, S.; Pasi, K.J.; et al. Lupus Anticoagulant and Abnormal Coagulation Tests in Patients with Covid-19. N. Engl. J. Med. 2020, 383, 288–290. [Google Scholar] [CrossRef]
- Tang, N.; Li, D.; Wang, X.; Sun, Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 844–847. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Shi, L.; Wang, Y.; Zhang, J.; Huang, L.; Zhang, C.; Liu, S.; Zhao, P.; Liu, H.; Zhu, L.; et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020, 8, 420–422. [Google Scholar] [CrossRef]
- Magro, C.; Mulvey, J.J.; Berlin, D.; Nuovo, G.; Salvatore, S.; Harp, J.; Baxter-Stoltzfus, A.; Laurence, J. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: A report of five cases. Transl. Res. 2020, 220, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Menter, T.; Haslbauer, J.; Nienhold, R.; Savic, S.; Hopfer, H.; Deigendesch, N.; Frank, S.; Turek, D.; Willi, N.; Pargger, H.; et al. Post-mortem examination of COVID19 patients reveals diffuse alveolar damage with severe capillary congestion and variegated findings of lungs and other organs suggesting vascular dysfunction. Histopathology 2020, 77, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.; Bai, H.; Chen, X.; Gong, J.; Li, D.; Sun, Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J. Thromb. Haemost. 2020, 18, 1094–1099. [Google Scholar] [CrossRef]
- Giannis, D.; Ziogas, I.A.; Gianni, P. Coagulation disorders in coronavirus infected patients: COVID-19, SARS-CoV-1, MERS-CoV and lessons from the past. J. Clin. Virol. 2020, 127, 104362. [Google Scholar] [CrossRef]
- Wong, R.S.M.; Wu, A.; To, K.F.; Lee, N.; Lam, C.W.K.; Wong, C.K.; Chan, P.K.; Ng, M.H.L.; Yu, L.M.; Hui, D.S.C.; et al. Haematological manifestations in patients with severe acute respiratory syndrome. Retrospective analysis. BMJ 2003, 326, 1358–1362. [Google Scholar] [CrossRef]
- Xiang-Hua, Y.; Le-Min, W.; Ai-Bin, L.; Zhu, G.; Riquan, L.; Xu-You, Z.; Wei-Wei, R.; Ye-Nan, W. Severe Acute Respiratory Syndrome and Venous Thromboembolism in Multiple Organs. Am. J. Respir. Crit. Care Med. 2010, 182, 436–437. [Google Scholar] [CrossRef]
- Li, K.; Wohlford-Lenane, C.; Perlman, S.; Zhao, J.; Jewell, A.K.; Reznikov, L.R.; Gibson-Corley, K.N.; Meyerholz, D.K.; McCray, P.B. Middle East Respiratory Syndrome Coronavirus Causes Multiple Organ Damage and Lethal Disease in Mice Transgenic for Human Dipeptidyl Peptidase 4. J. Infect. Dis. 2015, 213, 712–722. [Google Scholar] [CrossRef]
- Nishiga, M.; Wang, D.W.; Han, Y.; Lewis, D.B.; Wu, J.C. COVID-19 and cardiovascular disease. From basic mechanisms to clinical perspectives. Nat. Rev. Cardiol. 2020, 17, 1–16. [Google Scholar] [CrossRef]
- Shi, S.; Qin, M.; Shen, B.; Cai, Y.; Liu, T.; Yang, F.; Gong, W.; Liu, X.; Liang, J.; Zhao, Q.; et al. Association of Cardiac Injury With Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA Cardiol. 2020, 5, 802–810. [Google Scholar] [CrossRef] [PubMed]
- Stefanini, G.G.; Montorfano, M.; Trabattoni, D.; Andreini, D.; Ferrante, G.; Ancona, M.B.; Metra, M.; Curello, S.; Maffeo, D.; Pero, G.; et al. ST-Elevation Myocardial Infarction in Patients With COVID-19. Circulation 2020, 141, 2113–2116. [Google Scholar] [CrossRef] [PubMed]
- Bangalore, S.; Sharma, A.; Slotwiner, A.; Yatskar, L.; Harari, R.; Shah, B.; Ibrahim, H.; Friedman, G.H.; Thompson, C.; Alviar, C.L.; et al. ST-Segment Elevation in Patients with Covid-19-A Case Series. N. Engl. J. Med. 2020, 382, 2478–2480. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Wu, D.; Chen, H.; Yan, W.; Yang, D.; Chen, G.; Ma, K.; Xu, D.; Yu, H.; Wang, H.; et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019. Retrospective study. BMJ 2020, 368, 1091. [Google Scholar] [CrossRef]
- Goyal, P.; Choi, J.J.; Pinheiro, L.C.; Schenck, E.J.; Chen, R.; Jabri, A.; Satlin, M.J.; Campion, T.R.; Nahid, M.; Ringel, J.B.; et al. Clinical Characteristics of Covid-19 in New York City. N. Engl. J. Med. 2020, 382, 2372–2374. [Google Scholar] [CrossRef]
- Grasselli, G.; Greco, M.; Zanella, A.; Albano, G.; Antonelli, M.; Bellani, G.; Bonanomi, E.; Cabrini, L.; Carlesso, E.; Castelli, G.; et al. Risk Factors Associated with Mortality Among Patients with COVID-19 in Intensive Care Units in Lombardy, Italy. JAMA Intern. Med. 2020. [Google Scholar] [CrossRef]
- Bonsu, J.M.; Guha, A.; Charles, L.; Yildiz, V.O.; Wei, L.; Baker, B.; Brammer, J.E.; Awan, F.; Lustberg, M.; Reinbolt, R.; et al. Reporting of Cardiovascular Events in Clinical Trials Supporting FDA Approval of Contemporary Cancer Therapies. J. Am. Coll. Cardiol. 2020, 75, 620–628. [Google Scholar] [CrossRef]
- Boulware, D.R.; Pullen, M.F.; Bangdiwala, A.S.; Pastick, K.A.; Lofgren, S.M.; Okafor, E.C.; Skipper, C.P.; Nascene, A.A.; Nicol, M.R.; Abassi, M.; et al. A Randomized Trial of Hydroxychloroquine as Postexposure Prophylaxis for Covid-19. N. Engl. J. Med. 2020, 383, 517–525. [Google Scholar] [CrossRef]
- Geleris, J.; Sun, Y.; Platt, J.; Zucker, J.; Baldwin, M.; Hripcsak, G.; Labella, A.; Manson, D.K.; Kubin, C.; Barr, R.G.; et al. Observational Study of Hydroxychloroquine in Hospitalized Patients with Covid-19. N. Engl. J. Med. 2020, 382, 2411–2418. [Google Scholar] [CrossRef]
- Borba, M.G.S.; Val, F.F.A.; Sampaio, V.S.; Alexandre, M.A.A.; Melo, G.C.; Brito, M.; Mourão, M.P.G.; Brito-Sousa, J.D.; Baía-Da-Silva, D.; Guerra, M.V.F.; et al. Effect of High vs Low Doses of Chloroquine Diphosphate as Adjunctive Therapy for Patients Hospitalized With Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection: A Randomized Clinical Trial. JAMA Netw. Open 2020, 3, 208857. [Google Scholar] [CrossRef]
- Villarino, A.V.; Kanno, Y.; O’Shea, J.J. Mechanisms and consequences of Jak–STAT signaling in the immune system. Nat. Immunol. 2017, 18, 374–384. [Google Scholar] [CrossRef]
- Shimabukuro-Vornhagen, A.; Gödel, P.; Subklewe, M.; Stemmler, H.J.; Schlößer, H.A.; Schlaak, M.; Kochanek, M.; Böll, B.; Von Bergwelt-Baildon, M.S. Cytokine release syndrome. J. Immunother. Cancer 2018, 6, 56. [Google Scholar] [CrossRef] [PubMed]
- Channappanavar, R.; Fehr, A.R.; Zheng, J.; Wohlford-Lenane, C.; Abrahante, J.E.; Mack, M.; Sompallae, R.; McCray, P.B.; Meyerholz, D.K.; Perlman, S. IFN-I response timing relative to virus replication determines MERS coronavirus infection outcomes. J. Clin. Investig. 2019, 130, 3625–3639. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.; Merrill, S.A.; Alsawah, F.; Bockenstedt, P.; Campagnaro, E.; Devata, S.; Gitlin, S.D.; Kaminski, M.; Cusick, A.; Phillips, T.; et al. Ruxolitinib in adult patients with secondary haemophagocytic lymphohistiocytosis. An open-label, single-centre, pilot trial. Lancet Haematol. 2019, 6, 630–637. [Google Scholar] [CrossRef]
- Stebbing, J.; Phelan, A.; Griffin, I.; Tucker, C.; Oechsle, O.; Smith, D.; Richardson, P. COVID-19. Combining antiviral and anti-inflammatory treatments. Lancet Infect. Dis. 2020, 20, 400–402. [Google Scholar] [CrossRef]
- Bekerman, E.; Neveu, G.; Shulla, A.; Brannan, J.M.; Pu, S.-Y.; Wang, S.; Xiao, F.; Barouch-Bentov, R.; Bakken, R.R.; Mateo, R.; et al. Anticancer kinase inhibitors impair intracellular viral trafficking and exert broad-spectrum antiviral effects. J. Clin. Investig. 2017, 127, 1338–1352. [Google Scholar] [CrossRef]
- McInnes, I.B.; Byers, N.L.; Higgs, R.E.; Lee, J.; Macias, W.L.; Na, S.; Ortmann, R.A.; Rocha, G.; Rooney, T.P.; Wehrman, T.; et al. Comparison of baricitinib, upadacitinib, and tofacitinib mediated regulation of cytokine signaling in human leukocyte subpopulations. Arthritis Res. 2019, 21, 1–10. [Google Scholar] [CrossRef]
- Biopharma, T. Theravance Biopharma Announces First Subject Dosed in Phase 1 Study of TD-0903, in Development for the Treatment of Hospitalized Patients with Acute Lung Injury Caused by COVID-19. Available online: https://investor.theravance.com/news-releases/news-release-details/theravance-biopharma-announces-first-subject-dosed-phase-1-study (accessed on 18 June 2020).
- Shakoory, B.; Carcillo, J.A.; Chatham, W.W.; Amdur, R.L.; Zhao, H.; Dinarello, C.A.; Cron, R.Q.; Opal, S.M. Interleukin-1 Receptor Blockade Is Associated with Reduced Mortality in Sepsis Patients With Features of Macrophage Activation Syndrome. Crit. Care Med. 2016, 44, 275–281. [Google Scholar] [CrossRef]
- Miettunen, P.; Jayanthan, A.; Narendran, A. 7.3 Successful use of anakinra, a soluble IL-1 receptor antagonist, in pediatric rheumatic diseases associated macrophage activation syndrome/reactive hemophagocytic lymphohistiocytosis. Pediatr. Rheumatol. 2008, 6, 13. [Google Scholar] [CrossRef]
- Grom, A.A.; Ilowite, N.T.; Martini, A.; Leon, K.; Lheritier, K.; Abrams, K.; Pascual, V.; Brunner, H.I.; Lovell, D.; Ruperto, N.; et al. Rate and Clinical Presentation of Macrophage Activation Syndrome in Patients With Systemic Juvenile Idiopathic Arthritis Treated With Canakinumab. Arthritis Rheumatol. 2015, 68, 218–228. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, J.; Zhan, Y.; Wu, L.; Yu, X.; Zhang, W.; Ye, L.; Xu, S.; Sun, R.; Wang, Y.; et al. Analysis of Serum Cytokines in Patients with Severe Acute Respiratory Syndrome. Infect. Immun. 2004, 72, 4410–4415. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, G.A.; Varelias, A.; Vuckovic, S.; Le Texier, L.; Gartlan, K.H.; Zhang, P.; Thomas, G.P.; Anderson, L.; Boyle, G.M.; Cloonan, N.; et al. Addition of interleukin-6 inhibition with tocilizumab to standard graft-versus-host disease prophylaxis after allogeneic stem-cell transplantation. A phase 1/2 trial. Lancet Oncol. 2014, 15, 1451–1459. [Google Scholar] [CrossRef]
- Le, R.Q.; Li, L.; Yuan, W.; Shord, S.S.; Nie, L.; Habtemariam, B.A.; Przepiorka, D.; Farrell, A.T.; Pazdur, R. FDA Approval Summary: Tocilizumab for Treatment of Chimeric Antigen Receptor T Cell-Induced Severe or Life-Threatening Cytokine Release Syndrome. Oncology 2018, 23, 943–947. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Teachey, D.T.; Pequignot, E.; Frey, N.; Porter, D.; Maude, S.L.; Grupp, S.A.; June, C.H.; Melenhorst, J.J.; Lacey, S.F. Measuring IL-6 and sIL-6R in serum from patients treated with tocilizumab and/or siltuximab following CAR T cell therapy. J. Immunol. Methods 2016, 434, 1–8. [Google Scholar] [CrossRef]
- Teachey, D.T.; Lacey, S.F.; Shaw, P.A.; Melenhorst, J.J.; Maude, S.L.; Frey, N.; Pequignot, E.; Gonzalez, V.E.; Chen, F.; Finklestein, J.; et al. Identification of Predictive Biomarkers for Cytokine Release Syndrome after Chimeric Antigen Receptor T-cell Therapy for Acute Lymphoblastic Leukemia. Cancer Discov. 2016, 6, 664–679. [Google Scholar] [CrossRef]
- Bilusic, M.; Heery, C.R.; Collins, J.M.; Donahue, R.N.; Palena, C.; Madan, R.A.; Karzai, F.; Marté, J.L.; Strauss, J.; Gatti-Mays, M.E.; et al. Phase I trial of HuMax-IL8 (BMS-986253), an anti-IL-8 monoclonal antibody, in patients with metastatic or unresectable solid tumors. J. Immunother. Cancer 2019, 7, 240–248. [Google Scholar] [CrossRef]
- Lounder, D.T.; Bin, Q.; De Min, C.; Jordan, M.B. Treatment of refractory hemophagocytic lymphohistiocytosis with emapalumab despite severe concurrent infections. Blood Adv. 2019, 3, 47–50. [Google Scholar] [CrossRef]
- Vallurupalli, M.; Berliner, N. Emapalumab for the treatment of relapsed/refractory hemophagocytic lymphohistiocytosis. Blood 2019, 134, 1783–1786. [Google Scholar] [CrossRef]
- Kieseier, B.C. The Mechanism of Action of Interferon-β in Relapsing Multiple Sclerosis. CNS Drugs 2011, 25, 491–502. [Google Scholar] [CrossRef]
- Satyanarayanan, S.K.; El Kebir, D.; Soboh, S.; Butenko, S.; Sekheri, M.; Saadi, J.; Peled, N.; Assi, S.; Othman, A.; Schif-Zuck, S.; et al. IFN-β is a macrophage-derived effector cytokine facilitating the resolution of bacterial inflammation. Nat. Commun. 2019, 10, 1–16. [Google Scholar] [CrossRef]
- Hiruma, T.; Tsuyuzaki, H.; Uchida, K.; Trapnell, B.C.; Yamamura, Y.; Kusakabe, Y.; Totsu, T.; Suzuki, T.; Morita, S.; Doi, K.; et al. IFN-β Improves Sepsis-related Alveolar Macrophage Dysfunction and Postseptic Acute Respiratory Distress Syndrome-related Mortality. Am. J. Respir. Cell Mol. Boil. 2018, 59, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Yoo, C.-H.; Yeom, J.-H.; Heo, J.-J.; Song, E.-K.; Lee, S.-I.; Han, M.-K. Interferon β protects against lethal endotoxic and septic shock through SIRT1 upregulation. Sci. Rep. 2014, 4, 4220. [Google Scholar] [CrossRef]
- Bellingan, G.; Maksimow, M.; Howell, D.C.; Stotz, M.; Beale, R.; Beatty, M.; Walsh, T.; Binning, A.; Davidson, A.; Kuper, M.; et al. The effect of intravenous interferon-beta-1a (FP-1201) on lung CD73 expression and on acute respiratory distress syndrome mortality. An open-label study. Lancet Respir. Med. 2014, 2, 98–107. [Google Scholar] [CrossRef]
- Ranieri, V.M.; Pettilä, V.; Karvonen, M.K.; Jalkanen, J.; Nightingale, P.; Brealey, D.; Mancebo, J.; Ferrer, R.; Mercat, A.; Patroniti, N.; et al. Effect of Intravenous Interferon β-1a on Death and Days Free From Mechanical Ventilation Among Patients with Moderate to Severe Acute Respiratory Distress Syndrome. JAMA 2020, 323, 725. [Google Scholar] [CrossRef] [PubMed]
- Sterner, R.M.; Sakemura, R.; Cox, M.J.; Yang, N.; Khadka, R.H.; Forsman, C.L.; Hansen, M.J.; Jin, F.; Ayasoufi, K.; Hefazi, M.; et al. GM-CSF inhibition reduces cytokine release syndrome and neuroinflammation but enhances CAR-T cell function in xenografts. Blood 2018, 133, 697–709. [Google Scholar] [CrossRef]
- Gartlan, K.H.; Koyama, M.; Lineburg, K.E.; Chang, K.; Ensbey, K.S.; Kuns, R.D.; Henden, A.S.; Samson, L.D.; Clouston, A.D.; Lopez, A.F.; et al. Donor T-cell–derived GM-CSF drives alloantigen presentation by dendritic cells in the gastrointestinal tract. Blood Adv. 2019, 3, 2859–2865. [Google Scholar] [CrossRef]
- Salomon, R.; Hoffmann, E.; Webster, R. Inhibition of the cytokine response does not protect against lethal H5N1 influenza infection. Proc. Natl. Acad. Sci. USA 2007, 104, 12479–12481. [Google Scholar] [CrossRef]
- Szretter, K.J.; Gangappa, S.; Lu, X.; Smith, C.; Shieh, W.-J.; Zaki, S.R.; Sambhara, S.; Tumpey, T.M.; Katz, J.M. Role of Host Cytokine Responses in the Pathogenesis of Avian H5N1 Influenza Viruses in Mice. J. Virol. 2006, 81, 2736–2744. [Google Scholar] [CrossRef]
- Ikonomidis, I.; Pavlidis, G.; Katsimbri, P.; Andreadou, I.; Triantafyllidi, H.; Tsoumani, M.; Varoudi, M.; Vlastos, D.; Makavos, G.; Kostelli, G.; et al. Differential effects of inhibition of interleukin 1 and 6 on myocardial, coronary and vascular function. Clin. Res. Cardiol. 2019, 108, 1093–1101. [Google Scholar] [CrossRef]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef]
- Rozman, B.; Praprotnik, S.; Logar, D.; Tomsic, M.; Hojnik, M.; Kos-Golja, M.; Accetto, R.; Dolenc, P. Leflunomide and hypertension. Ann. Rheum. Dis. 2002, 61, 567–569. [Google Scholar] [CrossRef] [PubMed]
- Vargas, W.S.; Perumal, J.S. Fingolimod and cardiac risk. Latest findings and clinical implications. Ther. Adv. Drug Saf. 2013, 4, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Sciascia, S.; Aprà, F.; Baffa, A.; Baldovino, S.; Boaro, D.; Boero, R.; Bonora, S.; Calcagno, A.; Cecchi, I.; Cinnirella, G.; et al. Pilot prospective open, single-arm multicentre study on off-label use of tocilizumab in severe patients with COVID-19. Clin. Exp. Rheumatol 2020, 38, 529–532. [Google Scholar] [PubMed]
- Xu, X.; Han, M.; Li, T.; Sun, W.; Wang, D.; Fu, B.; Zhou, Y.; Zheng, X.; Yang, Y.; Li, X.; et al. Effective treatment of severe COVID-19 patients with tocilizumab. Proc. Natl. Acad. Sci. USA 2020, 117, 10970–10975. [Google Scholar] [CrossRef] [PubMed]
- Cantini, F.; Niccoli, L.; Matarrese, D.; Nicastri, E.; Stobbione, P.; Goletti, D. Baricitinib therapy in COVID-19: A pilot study on safety and clinical impact. J. Infect. 2020, 81, 318–356. [Google Scholar] [CrossRef]
- Cavalli, G.; De Luca, G.; Campochiaro, C.; Della-Torre, E.; Ripa, M.; Canetti, D.; Oltolini, C.; Castiglioni, B.; Din, C.T.; Boffini, N.; et al. Interleukin-1 blockade with high-dose anakinra in patients with COVID-19, acute respiratory distress syndrome, and hyperinflammation. A retrospective cohort study. Lancet Rheumatol. 2020, 2, 325–331. [Google Scholar] [CrossRef]
- Guaraldi, G.; Meschiari, M.; Cozzi-Lepri, A.; Milic, J.; Tonelli, R.; Menozzi, M.; Franceschini, E.; Cuomo, G.; Orlando, G.; Borghi, V.; et al. Tocilizumab in patients with severe COVID-19. A retrospective cohort study. Lancet Rheumatol. 2020, 2, 474–484. [Google Scholar] [CrossRef]
- La Rosée, P. Treatment of hemophagocytic lymphohistiocytosis in adults. Hematol. 2015, 2015, 190–196. [Google Scholar] [CrossRef]
- Elgebaly, S.A.; Elbayoumi, T.; Kreutzer, D.L. Cyclosporin h: A novel anti-inflammatory therapy for influenza flu patients. J. Egypt. Soc. Parasitol. 2017, 47, 25–33. [Google Scholar]
- Carbajo-Lozoya, J.; Mueller, M.A.; Kallies, S.; Thiel, V.; Drosten, C.; Von Brunn, A. Replication of human coronaviruses SARS-CoV, HCoV-NL63 and HCoV-229E is inhibited by the drug FK506. Virus Res. 2012, 165, 112–117. [Google Scholar] [CrossRef]
- De Wilde, A.H.; Zevenhoven-Dobbe, J.C.; Van Der Meer, Y.; Thiel, V.; Narayanan, K.; Makino, S.; Snijder, E.J.; Van Hemert, M.J. Cyclosporin A inhibits the replication of diverse coronaviruses. J. Gen. Virol. 2011, 92, 2542–2548. [Google Scholar] [CrossRef] [PubMed]
- Morrisett, J.D.; Abdel-Fattah, G.; Hoogeveen, R.; Mitchell, E.; Ballantyne, C.M.; Pownall, H.J.; Opekun, A.R.; Jaffe, J.S.; Oppermann, S.; Kahan, B.D. Effects of sirolimus on plasma lipids, lipoprotein levels, and fatty acid metabolism in renal transplant patients. J. Lipid Res. 2002, 43, 1170–1180. [Google Scholar] [CrossRef] [PubMed]
- McLeod, J.; Wu, S.; Grazette, L.; Sarcon, A. Tacrolimus-Associated Dilated Cardiomyopathy in Adult Patient After Orthotopic Liver Transplant. J. Investig. Med. High. Impact Case Rep. 2017, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Atkison, P.; Joubert, G.; Barron, A.; Grant, D.; Wall, W.; Rosenberg, H.; Howard, J.; Williams, S.; Stiller, C.; Paradis, K.; et al. Hypertrophic cardiomyopathy associated with tacrolimus in paediatric transplant patients. Lancet 1995, 345, 894–896. [Google Scholar] [CrossRef]
- Baruch, Y.; Weitzman, E.; Markiewicz, W.; Eisenman, A.; Eid, A.; Enat, R. Anasarca and hypertrophic cardiomyopathy in a liver transplant patient on FK506. Relieved after a switch to Neoral. Transplant. Proc. 1996, 28, 2250–2251. [Google Scholar]
- Roberts, C.A.; Stern, D.L.; Radio, S.J. Asymmetric cardiac hypertrophy at autopsy in patients who received FK506 (tacrolimus) or cyclosporine A after liver transplant1. Transplant. 2002, 74, 817–821. [Google Scholar] [CrossRef]
- Atkison, P.R.; Joubert, G.I.; Guiraudon, C.; Armstrong, R.; Wall, W.; Asfar, S.; Grant, D. arteritis and increased intracellular calcium as a possible mechanism for tacrolimus-related cardiac toxicity in a pediatric transplant recipient. Transplant. 1997, 64, 773–775. [Google Scholar] [CrossRef]
- Porter, G.A.; Bennett, W.M.; Sheps, S.G. Cyclosporine-Associated Hypertension. Arch. Intern. Med. 1990, 150, 280. [Google Scholar] [CrossRef]
- Luke, R.G. Mechanism of Cyclosporine-Induced Hypertension. Am. J. Hypertens. 1991, 4, 468–471. [Google Scholar] [CrossRef]
- Willicombe, M.; Thomas, D.; McAdoo, S. COVID-19 and Calcineurin Inhibitors: Should They Get Left Out in the Storm? J. Am. Soc. Nephrol. 2020, 31, 1145–1146. [Google Scholar] [CrossRef]
- Chaturvedi, S.; Braunstein, E.M.; Yuan, X.; Yu, J.; Alexander, A.; Chen, H.; Gavriilaki, E.; Alluri, R.K.; Streiff, M.B.; Petri, M.A.; et al. Complement activity and complement regulatory gene mutations are associated with thrombosis in APS and CAPS. Blood 2020, 135, 239–251. [Google Scholar] [CrossRef] [PubMed]
- Vasu, S.; Wu, H.; Satoskar, A.; Puto, M.; Roddy, J.; Blum, W.; Klisovic, R.; Andritsos, L.; Hofmeister, C.; Benson, D.M.; et al. Eculizumab therapy in adults with allogeneic hematopoietic cell transplant-associated thrombotic microangiopathy. Bone Marrow Transplant. 2016, 51, 1241–1244. [Google Scholar] [CrossRef] [PubMed]
- Wall, S.A.; Zhao, Q.; Yearsley, M.; Blower, L.; Agyeman, A.; Ranganathan, P.; Yang, S.; Wu, H.; Bostic, M.; Jaglowski, S.; et al. Complement-mediated thrombotic microangiopathy as a link between endothelial damage and steroid-refractory GVHD. Blood Adv. 2018, 2, 2619–2628. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.M.; Kahwash, R. Will Complement Inhibition Be the New Target in Treating COVID-19–Related Systemic Thrombosis? Circulation 2020, 141, 1739–1741. [Google Scholar] [CrossRef]
- Gralinski, L.E.; Baric, R.S. Molecular pathology of emerging coronavirus infections. J. Pathol. 2014, 235, 185–195. [Google Scholar] [CrossRef]
- Risitano, A.M.; Mastellos, D.C.; Huber-Lang, M.; Yancopoulou, D.; Garlanda, C.; Ciceri, F.; Lambris, J.D. Complement as a target in COVID-19? Nat. Rev. Immunol. 2020, 20, 343–344. [Google Scholar] [CrossRef]
- Gavriilaki, E.; Brodsky, R.A. Severe COVID-19 infection and thrombotic microangiopathy: Success doesn’t come easily. Br. J. Haematol. 2020, 189, 227–230. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhao, G.; Li, P.; Li, J.; Du, L.; Jiang, S.; Guo, R.; Sun, S.; Zhou, Y.; Song, N.; et al. Blockade of the C5a–C5aR axis alleviates lung damage in hDPP4-transgenic mice infected with MERS-CoV. Emerg. Microbes Infect. 2018, 7, 1–12. [Google Scholar] [CrossRef]
- Gralinski, L.E.; Sheahan, T.P.; Morrison, T.E.; Menachery, V.D.; Jensen, K.; Leist, S.R.; Whitmore, A.; Heise, M.T.; Baric, R.S.; Enjuanes, L.; et al. Complement Activation Contributes to Severe Acute Respiratory Syndrome Coronavirus Pathogenesis. mBio 2018, 9, 1753–1818. [Google Scholar] [CrossRef]
- Campbell, C.M.; Cassol, C.A.; Cataland, S.R.; Kahwash, R. Atypical haemolytic uraemic syndrome. A case report of a rare cause of reversible cardiomyopathy. Eur. Hear. J.-Case Rep. 2020, 4, 1–6. [Google Scholar] [CrossRef]
- Diurno, F.; Numis, F.G.; Porta, G.; Cirillo, F.; Maddaluno, S.; Ragozzino, A.; De Negri, P.; Di Gennaro, C.; Pagano, A.; Allegorico, E.; et al. Eculizumab treatment in patients with COVID-19. Preliminary results from real life ASL Napoli 2 Nord experience. Eur Rev. Med. Pharm. Sci 2020, 24, 4040–4047. [Google Scholar]
- Mastaglio, S.; Ruggeri, A.; Risitano, A.M.; Angelillo, P.; Yancopoulou, D.; Mastellos, D.C.; Huber-Lang, M.; Piemontese, S.; Assanelli, A.; Garlanda, C.; et al. The first case of COVID-19 treated with the complement C3 inhibitor AMY-101. Clin. Immunol. 2020, 215, 108450. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.; Lovly, C.M. Mechanisms of receptor tyrosine kinase activation in cancer. Mol. Cancer 2018, 17, 58. [Google Scholar] [CrossRef] [PubMed]
- Martini, M.; De Santis, M.C.; Braccini, L.; Gulluni, F.; Hirsch, E. PI3K/AKT signaling pathway and cancer. An updated review. Ann. Med. 2014, 46, 372–383. [Google Scholar] [CrossRef] [PubMed]
- Franks, M.E.; MacPherson, G.R.; Figg, W.D. Thalidomide. Lancet 2004, 363, 1802–1811. [Google Scholar] [CrossRef]
- Zhu, Y.X.; Kortuem, K.M.; Stewart, A.K. Molecular mechanism of action of immune-modulatory drugs thalidomide, lenalidomide and pomalidomide in multiple myeloma. Leuk. Lymphoma 2012, 54, 683–687. [Google Scholar] [CrossRef]
- Wei, S.; Duffy, C.R.; Allison, J. Fundamental Mechanisms of Immune Checkpoint Blockade Therapy. Cancer Discov. 2018, 8, 1069–1086. [Google Scholar] [CrossRef]
- Coleman, C.M.; Sisk, J.M.; Mingo, R.M.; Nelson, E.A.; White, J.M.; Frieman, M.B. Abelson Kinase Inhibitors Are Potent Inhibitors of Severe Acute Respiratory Syndrome Coronavirus and Middle East Respiratory Syndrome Coronavirus Fusion. J. Virol. 2016, 90, 8924–8933. [Google Scholar] [CrossRef]
- Aman, J.; Van Bezu, J.; Damanafshan, A.; Huveneers, S.; Eringa, E.C.; Vogel, S.M.; Groeneveld, A.J.; Noordegraaf, A.V.; Van Hinsbergh, V.W.; Amerongen, G.P.V.N. Effective Treatment of Edema and Endothelial Barrier Dysfunction With Imatinib. Circulation 2012, 126, 2728–2738. [Google Scholar] [CrossRef]
- Chislock, E.M.; Pendergast, A.M. Abl Family Kinases Regulate Endothelial Barrier Function In Vitro and in Mice. PLoS ONE 2013, 8, 85231. [Google Scholar] [CrossRef]
- Florence, J.M.; Krupa, A.; Booshehri, L.M.; Davis, S.A.; Matthay, M.A.; Kurdowska, A.K. Inhibiting Bruton’s tyrosine kinase rescues mice from lethal influenza-induced acute lung injury. Am. J. Physiol. Cell. Mol. Physiol. 2018, 315, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Treon, S.P.; Castillo, J.J.; Skarbnik, A.P.; Soumerai, J.D.; Ghobrial, I.M.; Guerrera, M.L.; Meid, K.E.; Yang, G. The BTK inhibitor ibrutinib may protect against pulmonary injury in COVID-19–infected patients. Blood 2020, 135, 1912–1915. [Google Scholar] [CrossRef] [PubMed]
- Winkler, D.G.; Faia, K.L.; DiNitto, J.P.; Ali, J.A.; White, K.F.; Brophy, E.E.; Pink, M.M.; Proctor, J.L.; Lussier, J.; Martin, C.M.; et al. PI3K-δ and PI3K-γ Inhibition by IPI-145 Abrogates Immune Responses and Suppresses Activity in Autoimmune and Inflammatory Disease Models. Chem. Boil. 2013, 20, 1364–1374. [Google Scholar] [CrossRef] [PubMed]
- McLeod, R.L.; Gil, M.A.; Chen, D.; Cabal, A.; Katz, J.; Methot, J.; Woodhouse, J.D.; Dorosh, L.; Geda, P.; Mehta, K.; et al. Characterizing Pharmacokinetic–Pharmacodynamic Relationships and Efficacy of PI3Kδ Inhibitors in Respiratory Models of TH2 and TH1 Inflammation. J. Pharmacol. Exp. Ther. 2019, 369, 223–233. [Google Scholar] [CrossRef]
- Ourradi, K.; Blythe, T.; Jarrett, C.; Barratt, S.L.; Welsh, G.I.; Millar, A.B. VEGF isoforms have differential effects on permeability of human pulmonary microvascular endothelial cells. Respir. Res. 2017, 18, 116. [Google Scholar] [CrossRef] [PubMed]
- Bergsten, E.; Horne, A.; Aricò, M.; Astigarraga, I.; Egeler, R.M.; Filipovich, A.; Ishii, E.; Janka, G.; Ladisch, S.; Lehmberg, K.; et al. Confirmed efficacy of etoposide and dexamethasone in HLH treatment. Long-term results of the cooperative HLH-2004 study. Blood 2017, 130, 2728–2738. [Google Scholar] [CrossRef]
- Ehl, S.; Astigarraga, I.; Greenwood, T.V.B.; Hines, M.; Horne, A.; Ishii, E.; Janka, G.; Jordan, M.B.; La Rosée, P.; Lehmberg, K.; et al. Recommendations for the Use of Etoposide-Based Therapy and Bone Marrow Transplantation for the Treatment of HLH: Consensus Statements by the HLH Steering Committee of the Histiocyte Society. J. Allergy Clin. Immunol. Pr. 2018, 6, 1508–1517. [Google Scholar] [CrossRef]
- Johnson, T.S.; Terrell, C.E.; Millen, S.H.; Katz, J.D.; Hildeman, D.A.; Jordan, M.B. Etoposide selectively ablates activated T cells to control the immunoregulatory disorder hemophagocytic lymphohistiocytosis. J. Immunol. 2013, 192, 84–91. [Google Scholar] [CrossRef]
- Pukhalsky, A.; Shmarina, G.; Alioshkin, V.; Sabelnikov, A. Alkylating drugs applied in non-cytotoxic doses as a novel compounds targeting inflammatory signal pathway. Biochem. Pharmacol. 2006, 72, 1432–1438. [Google Scholar] [CrossRef]
- Kashyap, T.; Argueta, C.; Aboukameel, A.; Unger, T.J.; Klebanov, B.; Mohammad, R.M.; Muqbil, I.; Azmi, A.S.; Drolen, C.; Senapedis, W.; et al. Selinexor, a Selective Inhibitor of Nuclear Export (SINE) compound, acts through NF-κB deactivation and combines with proteasome inhibitors to synergistically induce tumor cell death. Oncotarget 2016, 7, 78883–78895. [Google Scholar] [CrossRef]
- Horton, M.R.; Santopietro, V.; Mathew, L.; Horton, K.M.; Polito, A.J.; Liu, M.C.; Danoff, S.; Lechtzin, N. Thalidomide for the Treatment of Cough in Idiopathic Pulmonary Fibrosis. Ann. Intern. Med. 2012, 157, 398–406. [Google Scholar] [CrossRef]
- Li, D.; Zhang, X.-W.; Jiang, X.-Q.; Yin, Y.-J.; Fan, Z.; Sun, C.-B.; Chen, X.-H.; Li, Y.-H.; Li, D. Protective effects of thalidomide on pulmonary injuries in a rat model of paraquat intoxication. J. Inflamm. 2015, 12, 46. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Shi, X.; Ju, D.; Huang, H.; Wei, W.; Dong, X. Anti-Inflammatory Effect of Thalidomide on H1N1 Influenza Virus-Induced Pulmonary Injury in Mice. Inflammation 2014, 37, 2091–2098. [Google Scholar] [CrossRef] [PubMed]
- Bersanelli, M. Controversies about COVID-19 and anticancer treatment with immune checkpoint inhibitors. Immunotherapy 2020, 12, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Dickerson, T.; Wiczer, T.; Waller, A.; Philippon, J.; Porter, K.; Haddad, D.; Guha, A.; Rogers, K.A.; Bhatt, S.; Byrd, J.C.; et al. Hypertension and incident cardiovascular events following ibrutinib initiation. Blood 2019, 134, 1919–1928. [Google Scholar] [CrossRef] [PubMed]
- Leong, D.P.; Caron, F.; Hillis, C.; Duan, A.; Healey, J.S.; Fraser, G.; Siegal, D.M. The risk of atrial fibrillation with ibrutinib use. A systematic review and meta-analysis. Blood 2016, 128, 138–140. [Google Scholar] [CrossRef] [PubMed]
- Wiczer, T.E.; Levine, L.B.; Brumbaugh, J.; Coggins, J.; Zhao, Q.; Ruppert, A.S.; Rogers, K.; McCoy, A.; Mousa, L.; Guha, A.; et al. Cumulative incidence, risk factors, and management of atrial fibrillation in patients receiving ibrutinib. Blood Adv. 2017, 1, 1739–1748. [Google Scholar] [CrossRef] [PubMed]
- Guha, A.; Derbala, M.H.; Zhao, Q.; Wiczer, T.E.; Woyach, J.A.; Byrd, J.C.; Awan, F.T.; Addison, D. Ventricular Arrhythmias Following Ibrutinib Initiation for Lymphoid Malignancies. J. Am. Coll. Cardiol. 2018, 72, 697–698. [Google Scholar] [CrossRef]
- Wang, M.L.; Blum, K.A.; Martin, P.; Goy, A.; Auer, R.; Kahl, B.S.; Jurczak, W.; Advani, R.H.; Romaguera, J.E.; Williams, M.E.; et al. Long-term follow-up of MCL patients treated with single-agent ibrutinib. Updated safety and efficacy results. Blood 2015, 126, 739–745. [Google Scholar] [CrossRef]
- Levade, M.; David, E.; Garcia, C.; Laurent, P.A.; Cadot, S.; Michallet, A.-S.; Bordet, J.C.; Tam, C.; Sié, P.; Ysebaert, L.; et al. Ibrutinib treatment affects collagen and von Willebrand factor-dependent platelet functions. Blood 2014, 124, 3991–3995. [Google Scholar] [CrossRef]
- Mahmood, S.S.; Fradley, M.G.; Cohen, J.V.; Nohria, A.; Reynolds, K.L.; Heinzerling, L.M.; Sullivan, R.J.; Damrongwatanasuk, R.; Chen, C.L.; Gupta, D.; et al. Myocarditis in Patients Treated with Immune Checkpoint Inhibitors. J. Am. Coll. Cardiol. 2018, 71, 1755–1764. [Google Scholar] [CrossRef]
- Guha, A.; Al-Kindi, S.; Jain, P.; Tashtish, N.; ElAmm, C.; Oliveira, G.H. Association between myocarditis and other immune-related adverse events secondary to immune checkpoint inhibitor use. Int. J. Cancer 2020, 147, 1753–1754. [Google Scholar] [CrossRef] [PubMed]
- Waliany, S.; Sainani, K.L.; Park, L.S.; Zhang, C.A.; Srinivas, S.; Witteles, R.M. Increase in Blood Pressure Associated with Tyrosine Kinase Inhibitors Targeting Vascular Endothelial Growth Factor. JACC CardioOncology 2019, 1, 24–36. [Google Scholar] [CrossRef]
- El Accaoui, R.; Shamseddeen, W.; Taher, A.T. Thalidomide and thrombosis. A meta-analysis. Thromb. Haemost. 2007, 97, 1031–1036. [Google Scholar] [PubMed]
- Menon, S.P.; Rajkumar, S.V.; Lacy, M.; Falco, P.; Palumbo, A.; Rajkumar, S.V. Thromboembolic events with lenalidomide-based therapy for multiple myeloma. Cancer 2008, 112, 1522–1528. [Google Scholar] [CrossRef] [PubMed]
- Ben Kridis, W.; Khanfir, A.; Triki, F.; Frikha, M. An Exceptional Case of Atrial Fibrillation Arrhythmia Induced by Etoposide. Curr. Drug Saf. 2013, 8, 287–289. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.J.; Kobrossi, S.; Desai, A. Melphalan-Induced Atrial Fibrillation. Am. J. Ther. 2018, 25, 571–572. [Google Scholar] [CrossRef]
- Yanamandra, U.; Gupta, S.; Khadwal, A.; Malhotra, P. Melphalan-induced cardiotoxicity. Ventricular arrhythmias. BMJ Case Rep. 2016, 2016. [Google Scholar] [CrossRef]
- Bonomi, L.; Ghilardi, L.; Arnoldi, E.; Tondini, C.A.; Bettini, A.C. A rapid fatal evolution of Coronavirus Disease-19 (COVID-19) in an advanced lung cancer patient with a long time response to nivolumab. J. Thorac. Oncol. 2020, 15, 83–85. [Google Scholar] [CrossRef]
| Therapy | Mechanism of Action | FDA Approval Status | Primary Indications or Disease Targets | Cardiotoxicity (Reported) | Ongoing Clinical Trials |
|---|---|---|---|---|---|
| Anti-inflammatory agents | |||||
| Emapalumab | IFN-gamma inhibitor | Approved | Hemophagocytic lymphohistiocytosis | N/A | 1 trial |
| Interferon-beta-1a | Interferon-beta-1a | Approved | Multiple sclerosis | N/A | 9 trials |
| Ruxolitinib | JAK-1 and JAK-2 inhibitor | Approved | Myelofibrosis and polycythemia vera | N/A | 13 trials |
| Baricitinib | JAK-1 and JAK-2 inhibitor | Approved | Rheumatoid arthritis | N/A | 13 trials |
| Tofacitinib | JAK inhibitor | Approved | Rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, ulcerative colitis | N/A | 2 trials |
| TD-0903 | JAK inhibitor | Investigational | Lung transplant graft rejection | N/A | 1 trial |
| Tocilizumab | IL-6 receptor inhibitor | Approved | Rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, juvenile idiopathic arthritis | N/A | 31 trials |
| Sarilumab | IL-6 receptor inhibitor | Approved | Rheumatoid arthritis | N/A | 10 trials |
| Siltuximab | IL-6 inhibitor | Approved | Idiopathic multicentric Castleman’s disease | N/A | 3 trials |
| Sirukumab | IL-6 inhibitor | Investigational | Rheumatoid arthritis | N/A | 1 trial |
| Clazakizumab | IL-6 inhibitor | Investigational | Psoriatic arthritis | Unknown | 5 trials |
| Olokizumab | IL-6 inhibitor | Investigational | Rheumatoid arthritis | Unknown | 1 trial |
| Canakinumab | IL-1-beta inhibitor | Approved | Cryopyrin-associated periodic syndromes and systemic juvenile idiopathic arthritis | N/A; decreases cardiovascular events in trials | 2 trials |
| Anakinra | IL-1 receptor inhibitor | Approved | Rheumatoid arthritis, neonatal-onset multisystem inflammatory disease | N/A; decreases cardiovascular events in trials | 9 trials |
| BMS-986253 | IL-8 inhibitor | Investigational | Hematological malignancy, solid tumor | Unknown | 1 trial |
| Lenzilumab | GM-CSF inhibitor | Investigational | Cytokine release syndrome induced by CAR T cell therapy; graft versus host disease | Unknown | 1 trial |
| Gimsilumab | GM-CSF inhibitor | Investigational | Ankylosing Spondylitis | Unknown | 1 trial |
| Otilimab | GM-CSF inhibitor | Investigational | Rheumatoid arthritis | Unknown | 1 trial |
| TJ003234 | GM-CSF inhibitor | Investigational | CAR T cell cytokine storm | Unknown | 1 trial |
| XPro1595 | TNF-alpha soluble inhibitor | Investigational | Alzheimer’s disease | Unknown | 1 trial |
| ABX464 | miR-124 overexpression | Investigational | Ulcerative colitis, rheumatoid arthritis, Crohn’s disease, hepatocellular cancer, HIV | Unknown | 1 trial |
| Ulinastatin | Serine protesase inhibitor | Investigational | Acute pancreatitis, severe sepsis | Unknown | 1 trial |
| Piclidenoson | A3AR inhibitor | Investigational | Inflammatory bowel disease, rheumatoid arthritis | Unknown | 1 trial |
| Anti-ST2 | ST2 receptor inhibitor | Investigational | Asthma | Unknown | 1 trial |
| IC14 | CD14 inhibitor | Investigational | Amyotrophic lateral sclerosis | Unknown | 2 trials |
| IMU-838 | Dihydroorotate dehydrogenase inhibitor | Investigational | Ulcerative colitis | Unknown | 1 trial |
| Lefunomide | Dihydroorotate dehydrogenase inhibitor | Approved | Rheumatoid arthritis | Hypertension | 1 trial |
| Methotrexate | Multiple including IL-1-beta inhibitor and dihydrofolate reductase inhibitor | Approved | Rheumatoid arthritis, juvenile dermatomyositis, psoriasis, lupus, sarcoidosis, Crohn’s disease, eczema, vasculitis, multiple cancers | N/A | 1 trial |
| CM4620 | Calcium release-activated calcium channel inhibitor | Investigational | Pancreatitis | Unknown | 1 trial |
| CD24Fc | Danger-Associated Molecular Patterns (DAMPs) | Investigational | Graft-versus-host disease | Unknown | 1 trial |
| Finglimod | Sphingosine-1-phosphate receptor modulator | Approved | Multiple sclerosis | Bradycardia, atrioventricular block | 1 trial |
| Anti-rejection agents | |||||
| Sirolimus | mTOR pathway inhibitor | Approved | Organ transplant rejection | Hyperlipidemia | 3 trials |
| Tacrolimus | Calcineurin phosphatase inhibitor | Approved | Organ transplant rejection | Cardiomyopathy (rare) | 1 trial |
| Cyclosporine | Calcineurin inhibitor | Approved | Organ transplant rejection | Hypertension | 1 trial |
| Anti-complement agents | |||||
| AMY-101 | C3 inhibitor | Investigational | Complement 3 glomerulopathy, paroxysmal nocturnal hemoglobinuria, periodontitis | Unknown | 1 trial |
| Ravulizumab | C5 inhibitor | Approved | Paroxysmal nocturnal hemoglobinuria, atypical hemolytic uremic syndrome | N/A | 2 trials |
| Eculizumab | C5 inhibitor | Approved | Atypical hemolytic uremic syndrome | N/A | 3 trials |
| Zilucoplan | C5 inhibitor | Investigational | Myasthenia gravis | Unknown | 1 trial |
| IFX-1 | C5a inhibitor | Investigational | Hidradenitis suppurativa, ANCA-associated vasculitis, pyoderma gangrenosum | Unknown | 1 trial |
| Avdoralimab | C5a receptor inhibitor | Investigational | Hepatocellular carcinoma, non-small cell lung cancer | Unknown | 1 trial |
| Anti-Cancer agents | |||||
| Ibrutinib | BTK inhibitor | Approved | Mantel cell lymphoma, chronic lymphocytic leukemia, Waldenstrom’s macroglobulinemia | Atrial fibrillation, hypertension, bleeding, ventricular fibrillation | 1 trial |
| Acalabrutinib | BTK inhibitor | Approved | Mantle cell lymphoma, chronic lymphocytic leukemia | Atrial fibrillation, hypertension, bleeding | 2 trials |
| Zanubrutinib | BTK inhibitor | Approved | Mantle cell lymphoma | Atrial fibrillation, hypertension, bleeding | 1 trial |
| Imatinib | BCR-ABL TK inhibitor | Approved | Chronic myelogenous leukemia | Cardiomyopathy | 2 trials |
| Bevacizumab | VEGF inhibitor | Approved | Colorectal, lung, glioblastoma, kidney, cervical, and ovarian cancer | Hypertension, cardiac ischemia, congestive heart failure, venous thromboembolic events | 3 trials |
| Nintedanib | VEGF inhibitor, FGFR inhibitor, PDGFR inhibitor | Approved | Idiopathic pulmonary fibrosis, chronic fibrosing interstitial lung disease | Hypertension, thromboembolic events | 1 trial |
| Duvelisib | PI3 Kinase inhibitor | Approved | Chronic lymphocyte leukemia, small lymphocytic lymphoma | N/A | 1 trial |
| Thalidomide | Uncertain—angiogenesis inhibitor, anti-inflammatory, anti-proliferative | Approved | Multiple myeloma, graft-versus-host disease, leprosy | Cardiac ischemia, arrhythmias, venous thromboembolic events | 2 trials |
| Lenalidomide | Uncertain—angiogenesis inhibitor, anti-inflammatory, anti-proliferative | Approved | Myelodysplastic syndrome, multiple myeloma, mantle cell lymphoma | Venous thromboembolism (black box warning), cardiac ischemia | 1 trial |
| Plitidepsin | EF1A2 inhibitor (translation) | Investigational | Multiple myeloma | Unknown | 1 trial |
| Etoposide | Topoisomerase inhibitor | Approved | Testicular cancer, lung cancer, lymphoma, leukemia, neuroblastoma, ovarian cancer | Atrial fibrillation (rare) | 1 trial |
| Melphalan | DNA alkylation | Approved | Multiple myeloma, ovarian cancer, melanoma, amyloidosis | Case report of sustained ventricular tachycardia | 1 trial |
| Selinexor | Exportin 1 inhibitor | Approved | Multiple myeloma | N/A | 2 trials |
| Veru-111 | Tubulin inhibitor | Investigational | Prostate cancer | Unknown | 1 trial |
| Pembrolizumab | Immune checkpoint inhibitor | Approved | Melanoma, lung cancer, head and neck cancer, Hodgkin lymphoma, stomach cancer | Myocarditis, pericarditis, arrhythmia | 1 trial |
| Nivolumab | Immune checkpoint inhibitor | Approved | Melanoma, lung cancer, renal cell carcinoma, colon cancer, liver cancer, head and neck cancer, Hodgkin lymphoma | Myocarditis, pericarditis, arrhythmia | 3 trials |
| AVM0703 | Uncertain—lymphodepletion | Investigational | Non-Hodgkins lymphoma, acute lymphocytic leukemia, chronic lymphocytic leukemia | Unknown | 1 trial |
| TAK-981 | Small ubiquitin-like modifier | Investigational | Non-Hodgkin’s lymphoma | Unknown | 1 trial |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campbell, C.M.; Guha, A.; Haque, T.; Neilan, T.G.; Addison, D. Repurposing Immunomodulatory Therapies against Coronavirus Disease 2019 (COVID-19) in the Era of Cardiac Vigilance: A Systematic Review. J. Clin. Med. 2020, 9, 2935. https://doi.org/10.3390/jcm9092935
Campbell CM, Guha A, Haque T, Neilan TG, Addison D. Repurposing Immunomodulatory Therapies against Coronavirus Disease 2019 (COVID-19) in the Era of Cardiac Vigilance: A Systematic Review. Journal of Clinical Medicine. 2020; 9(9):2935. https://doi.org/10.3390/jcm9092935
Chicago/Turabian StyleCampbell, Courtney M., Avirup Guha, Tamanna Haque, Tomas G. Neilan, and Daniel Addison. 2020. "Repurposing Immunomodulatory Therapies against Coronavirus Disease 2019 (COVID-19) in the Era of Cardiac Vigilance: A Systematic Review" Journal of Clinical Medicine 9, no. 9: 2935. https://doi.org/10.3390/jcm9092935
APA StyleCampbell, C. M., Guha, A., Haque, T., Neilan, T. G., & Addison, D. (2020). Repurposing Immunomodulatory Therapies against Coronavirus Disease 2019 (COVID-19) in the Era of Cardiac Vigilance: A Systematic Review. Journal of Clinical Medicine, 9(9), 2935. https://doi.org/10.3390/jcm9092935





