Assessing Viral Shedding and Infectivity of Asymptomatic or Mildly Symptomatic Patients with COVID-19 in a Later Phase
Abstract
:1. Introduction
2. Methods
2.1. Patients and Study Settings
2.2. Definitions
2.3. Nucleic Acid Extraction and RT-PCR for SARS-CoV-2
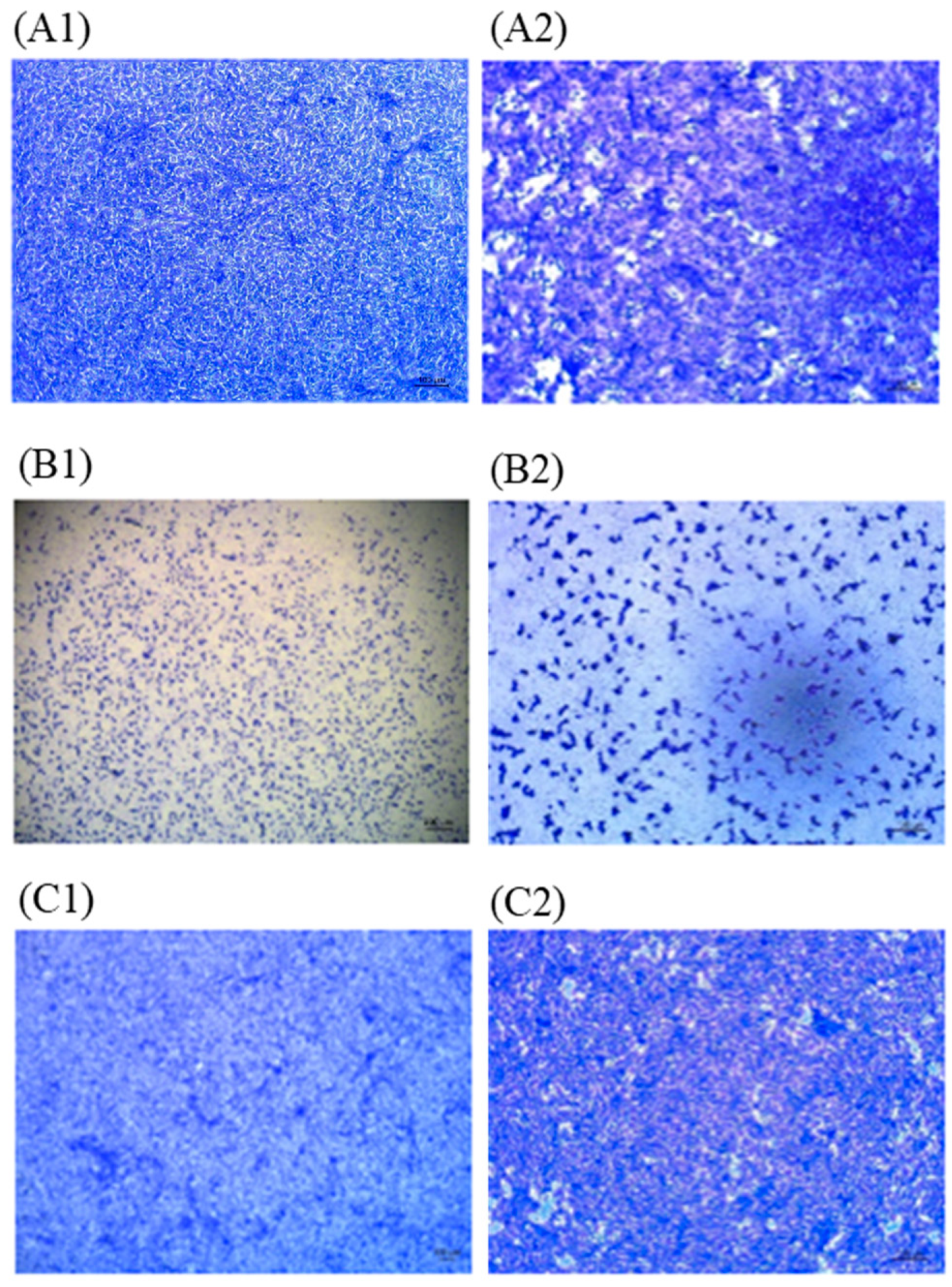
2.4. SARS-CoV-2 Cell Culture
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Coronavirus Disease (COVID-19) Situation Report—179. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200717-covid-19-sitrep-179.pdf?sfvrsn=2f1599fa_2 (accessed on 18 July 2020).
- World Health Organization. Criteria for Releasing COVID-19 Patients from Isolation. Available online: https://www.who.int/news-room/commentaries/detail/criteria-for-releasing-covid-19-patients-from-isolation (accessed on 18 July 2020).
- Azzi, L.; Carcano, G.; Gianfagna, F.; Grossi, P.; Gasperina, D.D.; Genoni, A.; Fasano, M.; Sessa, F.; Tettamanti, L.; Carnici, F.; et al. Saliva is a reliable tool to detect SARS-CoV-2. J. Infect. 2020, 81, e45–e50. [Google Scholar] [CrossRef] [PubMed]
- Wolfel, R.; Corman, V.M.; Guggemos, W.; Seilmaier, M.; Zange, S.; Muller, M.A.; Niemeyer, D.; Jones, T.C.; Vollmar, P.; Rothe, C.; et al. Virological assessment of hospitalized patients with COVID-2019. Nature 2020, 81, 465–469. [Google Scholar] [CrossRef] [Green Version]
- Peng, L.; Liu, J.; Xu, W.; Luo, Q.; Chen, D.; Lei, Z.; Huang, Z.; Li, X.; Deng, K.; Lin, B.; et al. SARS-CoV-2 can be detected in urine, blood, anal swabs, and oropharyngeal swabs specimens. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- To, K.K.-W.; Tsang, O.T.-Y.; Leung, W.-S.; Tam, A.R.; Wu, T.-C.; Lung, D.C.; Yip, C.C.-Y.; Cai, J.-P.; Chan, J.M.-C.; Chik, T.S.-H.; et al. Temporal profiles of viral load in posterior oropharyngeal saliva samples and serum antibody responses during infection by SARS-CoV-2: An observational cohort study. Lancet Infect. Dis. 2020, 20, 565–574. [Google Scholar] [CrossRef] [Green Version]
- Widders, A.; Broom, A.; Broom, J. SARS-CoV-2: The viral shedding vs infectivity dilemma. Infect. Dis. Health 2020. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 95, 1054–1062. [Google Scholar] [CrossRef]
- Kim, S.W.; Lee, K.S.; Kim, K.; Lee, J.J.; Kim, J.-Y. Daegu Medical Association a brief telephone severity scoring system and therapeutic living centers solved acute hospital-bed shortage during the COVID-19 outbreak in Daegu, Korea. J. Korean Med. Sci. 2020, 35, e152. [Google Scholar] [CrossRef] [Green Version]
- Sung, H.; Roh, K.H.; Hong, K.H.; Seong, M.-W.; Ryoo, N.; Kim, H.S.; Lee, J.; Kim, S.Y.; Yoo, S.; Kim, M.-N.; et al. COVID-19 Molecular testing in Korea: Practical essentials and answers from experts based on experiences of emergency use authorization assays. Ann. Lab. Med. 2020, 40, 439–447. [Google Scholar] [CrossRef]
- Lai, C.-C.; Wang, C.-Y.; Ko, W.-C.; Hsueh, P.-R. In vitro diagnostics of coronavirus disease 2019: Technologies and application. J. Microbiol. Immunol. Infect. 2020. [Google Scholar] [CrossRef]
- Harcourt, J.; Tamin, A.; Lu, X.; Kamili, S.; Sakthivel, S.K.; Murray, J.; Queen, K.; Tao, Y.; Paden, C.R.; Zhang, J.; et al. Isolation and characterization of SARS-CoV-2 from the first US COVID-19 patient. bioRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- To, K.K.-W.; Tsang, O.T.-Y.; Yip, C.C.-Y.; Chan, K.-H.; Wu, T.-C.; Chan, J.M.-C.; Leung, W.-S.; Chik, T.S.-H.; Choi, C.Y.-C.; Kandamby, D.H.; et al. Consistent detection of 2019 novel coronavirus in saliva. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef] [Green Version]
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.; Bleicker, T.; Brünink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Eurosurveillance 2020, 25, 2000045. [Google Scholar] [CrossRef] [Green Version]
- Torres, I.; Sacoto, F. Localising an asset-based COVID-19 response in Ecuador. Lancet 2020, 395, 1339. [Google Scholar] [CrossRef]
- He, X.; Lau, E.H.Y.; Wu, P.; Deng, X.; Wang, J.; Hao, X.; Lau, Y.C.; Wong, J.Y.; Guan, Y.; Tan, X.; et al. Temporal dynamics in viral shedding and transmissibility of COVID-19. Nat. Med. 2020, 26, 672–675. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.-D.; Chang, S.-Y.; Wang, J.-T.; Tsai, M.-J.; Hung, C.-C.; Hsu, C.-L.; Chang, S.-C. Prolonged virus shedding even after seroconversion in a patient with COVID-19. J. Infect. 2020. [Google Scholar] [CrossRef]
- Zou, L.; Ruan, F.; Huang, M.; Liang, L.; Huang, H.; Hong, Z.; Yu, J.; Kang, M.; Song, Y.; Xia, J.; et al. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N. Engl. J. Med. 2020, 382, 1177–1179. [Google Scholar] [CrossRef]
- To, K.K.-W.; Lu, L.; Yip, C.C.; Poon, R.W.; Fung, A.M.; Cheng, A.; Lui, D.H.; Ho, D.T.; Hung, I.F.-N.; Chan, K.-H.; et al. Additional molecular testing of saliva specimens improves the detection of respiratory viruses. Emerg. Microbes Infect. 2017, 6, e49. [Google Scholar] [CrossRef] [Green Version]
- To, K.K.-W.; Yip, C.C.; Lai, C.Y.; Wong, C.K.; Ho, D.T.; Pang, P.K.P.; Ng, A.C.; Leung, K.-H.; Poon, R.W.; Chan, K.-H.; et al. Saliva as a diagnostic specimen for testing respiratory virus by a point-of-care molecular assay: A diagnostic validity study. Clin. Microbiol. Infect. 2019, 25, 372–378. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Ren, B.; Peng, X.; Hu, T.; Li, J.; Gong, T.; Tang, B.; Xu, X.; Zhou, X. Saliva is a non-negligible factor in the spread of COVID-19. Mol. Oral Microbiol. 2020, 5, 141–145. [Google Scholar] [CrossRef]
- Xu, H.; Zhong, L.; Deng, J.; Peng, J.; Dan, H.; Zeng, X.; Li, T.; Chen, Q. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int. J. Oral Sci. 2020, 12, 8. [Google Scholar] [CrossRef]
- Pan, X.; Chen, D.; Xia, Y.; Wu, X.; Li, T.; Ou, X.; Zhou, L.; Liu, J. Asymptomatic cases in a family cluster with SARS-CoV-2 infection. Lancet Infect. Dis. 2020, 20, 410–411. [Google Scholar] [CrossRef]
- La Scola, B.; Le Bideau, M.; Andreani, J.; Hoang, V.T.; Grimaldier, C.; Colson, P.; Gautret, P.; Raoult, D. Viral RNA load as determined by cell culture as a management tool for discharge of SARS-CoV-2 patients from infectious disease wards. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 9, 1059–1061. [Google Scholar] [CrossRef] [PubMed]

| Variables | Total (n = 48) | Symptomatic Group (n = 37) | Asymptomatic Group (n = 11) | p Value |
|---|---|---|---|---|
| Age, years | 32.62 ± 14.59 | 31.84 ± 13.74 | 35.27 ± 17.62 | 0.499 |
| Male, yes | 14 (29.2) | 12 (32.4) | 2 (18.2) | 0.361 |
| Smoker | 5 (10.4) | 5 (13.5) | 0 (0.0) | 0.198 |
| Comorbidity | ||||
| Hypertension | 4 (8.3) | 2 (5.4) | 2 (18.2) | 0.178 |
| Allergy | 4 (8.3) | 2 (5.4) | 2 (18.2) | 0.178 |
| Malignancy | 2 (4.2) | 2 (5.4) | 0 (0.0) | 0.431 |
| Diabetes | 1 (2.1) | 1 (2.7) | 0 (0.0) | 0.582 |
| Asthma | 1 (2.1) | 1 (2.7) | 0 (0.0) | 0.582 |
| Symptoms | ||||
| Myalgia | 12 (25.0) | 12 (32.4) | 0 (0.0) | - |
| Fever | 11 (22.9) | 11 (29.7) | 0 (0.0) | - |
| Chill | 9 (18.8) | 9 (24.3) | 0 (0.0) | - |
| Headache | 9 (18.3) | 9 (24.3) | 0 (0.0) | - |
| Anosmia | 8 (16.7) | 8 (21.6) | 0 (0.0) | - |
| Ageusia | 7 (14.6) | 7 (18.9) | 0 (0.0) | - |
| Rhinorrhea | 7 (14.6) | 7 (18.9) | 0 (0.0) | - |
| Nasal stuffiness | 7 (14.6) | 7 (18.9) | 0 (0.0) | - |
| Fatigue | 7 (14.6) | 7 (18.9) | 0 (0.0) | - |
| Cough | 6 (12.5) | 6 (16.2) | 0 (0.0) | - |
| Sputum | 6 (12.5) | 6 (16.2) | 0 (0.0) | - |
| Sore throat | 4 (8.3) | 4 (10.8) | 0 (0.0) | - |
| Dizziness | 3 (6.3) | 3 (8.1) | 0 (0.0) | - |
| Diarrhea | 3(6.3) | 3 (8.1) | 0 (0.0) | |
| Chest pain | 1 (2.1) | 1 (2.7) | 0 (0.0) | - |
| Vomiting | 1 (2.1) | 1 (2.7) | 0 (0.0) | - |
| Symptomatic Group (n = 37) | Asymptomatic Group (n = 11) | p Value | |
|---|---|---|---|
| Period from diagnosis to culture | |||
| Mean | 30.78 ± 5.85 | 29.09 ± 5.26 | 0.394 |
| Median | 31 (20–40) | 28 (21–41) | |
| Mean of Ct value on the day of culture | |||
| E gene | 30.55 ± 2.96 | 27.60 ± 0.47 | - |
| RdRP gene | 33.93 ± 3.43 | 31.56 ± 3.07 | - |
| N gene | 34.98 ± 2.89 | 33.94 ± 3.65 | - |
| Cases with rebound of Ct value | 11 (29.7) | 6 (54.5) | 0.131 |
| Items | Age | Sex | Comorbidity | Symptoms | Rebound | Ct Value | |
|---|---|---|---|---|---|---|---|
| NPS | Saliva | ||||||
| Patient1 | 51 | M | N | Y | N | 36.6 | 31.1 |
| Patient2 | 21 | M | Allergy | Y | N | 38.9 | 37.0 |
| Patient3 | 23 | F | N | Y | N | 35.1 | 31.4 |
| Patient4 | 25 | F | N | Y | N | 32.2 | 35.2 |
| Patient5 | 31 | F | N | Y | N | 33.2 | 36.9 |
| Patient6 | 54 | F | N | N | N | 34.5 | 35.1 |
| Patient7 | 27 | F | N | Y | N | 25.4 | 29.3 |
| Patient8 | 33 | M | N | Y | Y | 0.0 | 35.7 |
| Average of Ct value | 33.7 | 33.96 | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sohn, Y.; Jeong, S.J.; Chung, W.S.; Hyun, J.H.; Baek, Y.J.; Cho, Y.; Kim, J.H.; Ahn, J.Y.; Choi, J.Y.; Yeom, J.-S. Assessing Viral Shedding and Infectivity of Asymptomatic or Mildly Symptomatic Patients with COVID-19 in a Later Phase. J. Clin. Med. 2020, 9, 2924. https://doi.org/10.3390/jcm9092924
Sohn Y, Jeong SJ, Chung WS, Hyun JH, Baek YJ, Cho Y, Kim JH, Ahn JY, Choi JY, Yeom J-S. Assessing Viral Shedding and Infectivity of Asymptomatic or Mildly Symptomatic Patients with COVID-19 in a Later Phase. Journal of Clinical Medicine. 2020; 9(9):2924. https://doi.org/10.3390/jcm9092924
Chicago/Turabian StyleSohn, Yujin, Su Jin Jeong, Won Suk Chung, Jong Hoon Hyun, Yae Jee Baek, Yunsuk Cho, Jung Ho Kim, Jin Young Ahn, Jun Yong Choi, and Joon-Sup Yeom. 2020. "Assessing Viral Shedding and Infectivity of Asymptomatic or Mildly Symptomatic Patients with COVID-19 in a Later Phase" Journal of Clinical Medicine 9, no. 9: 2924. https://doi.org/10.3390/jcm9092924
APA StyleSohn, Y., Jeong, S. J., Chung, W. S., Hyun, J. H., Baek, Y. J., Cho, Y., Kim, J. H., Ahn, J. Y., Choi, J. Y., & Yeom, J.-S. (2020). Assessing Viral Shedding and Infectivity of Asymptomatic or Mildly Symptomatic Patients with COVID-19 in a Later Phase. Journal of Clinical Medicine, 9(9), 2924. https://doi.org/10.3390/jcm9092924





